Submitted:
09 February 2026
Posted:
10 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Background on Cerebral Palsy
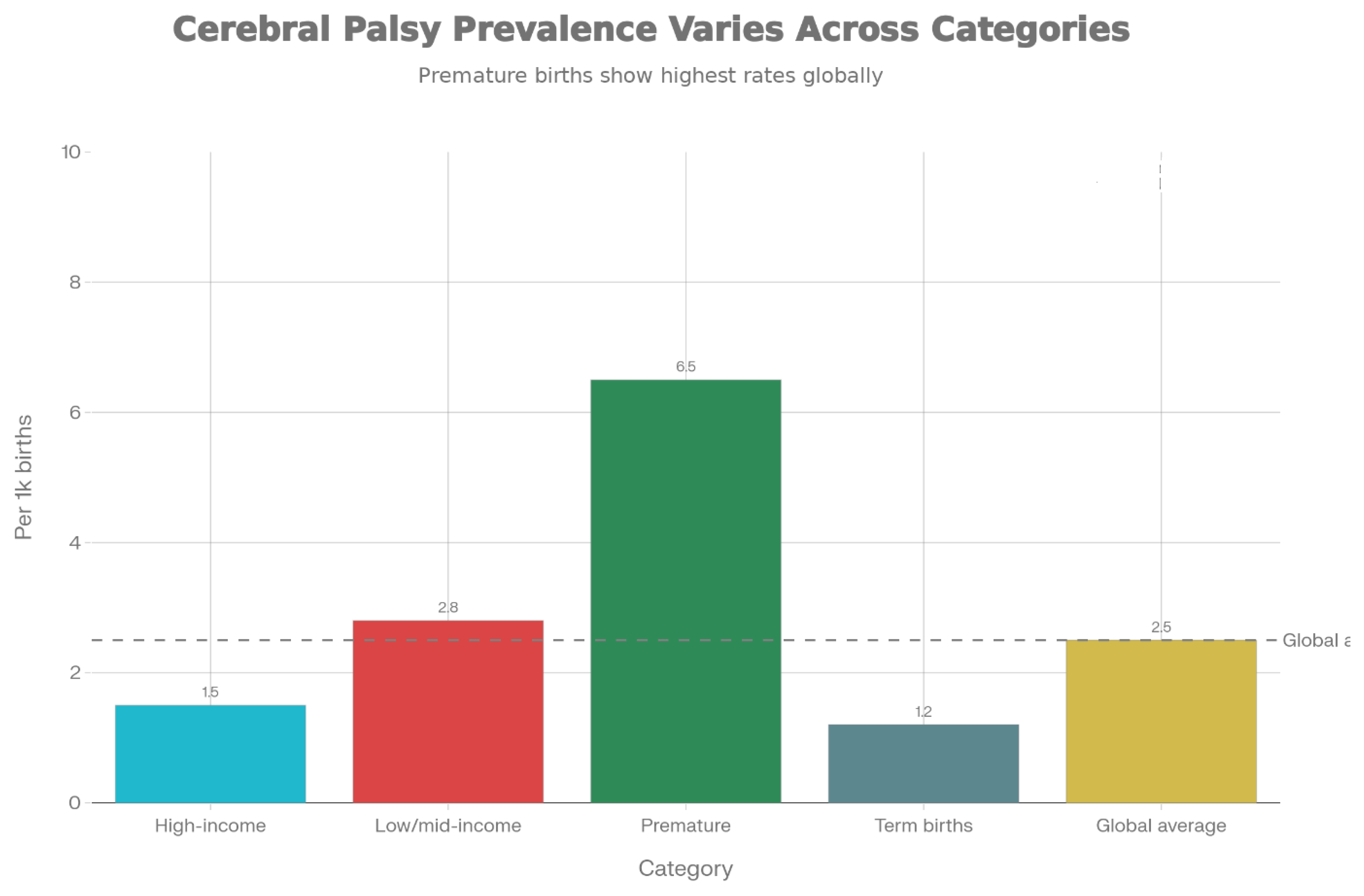
1.2. Current Treatment Limitations
1.3. Scientific Rationale for Axolotl-Based Neural Tissue
1.4. Research Objectives
- To determine the scientific and technical viability of obtaining and processing axolotl neural tissues for transplanting purposes
- To develop tissue engineering approaches for maximizing the integration of axolotl neural tissue with the human host brain
- Assessment of functional neuronal integration and circuit formation between transplanted axolotls tissue and human neural circuits
- To identify and develop strategies to counter immunological hurdles of xenotransplantation of axolot
- To demonstrate pre-clinical efficacy in suitable animal models
- To define ethical and regulatory approaches to translate into clinical settings
- To guide the way for advancement to human clinical trials
2. Literature Review
2.1. Axolotl Regeneration Biology
- Neuronal Diversity Regeneration: Work using molecular markers shows that axolotls replace numerous different neuronal populations, including those that are glutamatergic, GABAergic, and neurotensin-positive, mirroring the complexity of the preinjury brain.
- Functional Properties: In axolotls, newly regenerated neurons go on to mature into electrophysiological properties like appropriate membrane potentials, action potential generation, and synaptic responses to incoming stimuli.
- Molecular Signaling: Axolotl brain regeneration is induced via activation of the fibroblast growth factor(FGF) signaling pathway, the bone morphogenetic protein(BMP) signaling pathway, and the Wnt signaling pathway, which promote proliferation of neural stem cells and neuronal differentiation.
- Microglial Activation: In most mammals, microglia tend to form an inhibitory environment after CNS injury, whereas axolotl microglia seem to provide support for regeneration by releasing pro-regenerative factors.
- Growth Factor Production: Regenerating axolotl tissue upregulates production of neurotrophic factors including nerve growth factor (NGF), brain-derived neurotrophic factor (BDNF), and glial-derived neurotrophic factor (GDNF).
2.2. Cerebral Palsy Pathophysiology and Current Understanding
2.3. Neural Tissue Engineering and Regenerative Medicine Approaches
2.3.1. Clinical Trials of Stem Cell Therapy in Cerebral Palsy
2.4. Xenotransplantation Biology and Immunological Challenges
- Xenoreactive natural antibodies (natural IgM and IgG antibodies to the antigens of other animal species)
- Activation of complement via classical and alternate pathways
- Natural Killer cell mediated cytotoxicity
- Macrophage infiltration and tissue damage
- Genetic modification of the animals used as donors to produce human complement regulatory proteins
- Immunosuppressive protocol development tailored to promote xenograft survival
- Immunomodulatory biomaterials that induce lower levels
- Cell transplants encapsulated in immunoprotective devices
2.4.1. Lessons from Recent Organ Xenotransplantation
3. Proposed Research Methodology
3.1. Phase 1: Tissue Characterization and Preparation (Year 1-2)
3.1.1. Axolotl Neural Tissue Isolation and Characterization
- Maintain healthy adult axolotls (Ambystoma mexicanum) under standard laboratory conditions.
- Perform the harvest of brain tissue in accord with appropriate anesthesia protocols
- Isolate tissue specifically from the regeneratively competent regions of the pallium and ependymal zones
- Histological processing to characterize neuronal populations, glial cells, and general tissue architecture
- Perform immunohistochemistry for neuronal markers: NeuN, NeuroD1, βIII-tubulin, and glial markers: GFAP, Olig2
- Single-cell RNA sequencing to characterize molecular diversity of neural cells.
- Measurement of neurotrophic factors expression and regeneration-associated genes.
- The detailed characterization of axolotl neural tissue composition.
- Optimized Protocols for Tissue Harvest and Preservation
- Key molecular signatures driving the process of regeneration.
- Development of primary culture of Axolotl Neural Progenitor cells from pallium tissue
- Test cell culture conditions optimized for mammalian NPCs for axolotl cell maintenance
- Derive expandable stem cell lines with neural potential from axolotls
- Features of Progenitor Cells: ability to self-renew, multipotency
- Specific test for differentiation towards neural and glial cells
- Electrophysiological Properties in Differentiated Neurons
- E. axolotls – Expandable axol
- Protocols for neuronal differentiation
- Characterization of the functional properties of differentiated aNPCs
3.2. Phase 2: Tissue Engineering and Scaffold Development (Year 2-3)
3.2.1. Biocompatible Scaffold Design
- Test different biocompatible materials: silk fibroin, hyaluronic acid-based hydrogels, collagen
- Engineer scaffold parameters for axolotl cell survival and differentiation
- Scaffold designs including growth factor delivery (FGF, BDNF, NGF) within scaffold
- Specify scaffold designs for mechanical direction axon growth
- Evaluate 2D and 3D cell cultures
- Establish the properties of the construct: porosity, pore size, degradation rate, and strength
- Biocompatibility analysis by culture with axolotl neural cells
- Optimized tissue engineered constructs comprising cells of the Axolotl and Biocompatible scaffolds
- Characterization of Construct Properties
- Neural cell survival, differentiation, and axon outgrowth within constructs
3.2.2. Integration Studies In Vitro
- Co-culture axolotl neural progenitor cells with rodent or human neural progenitor cells
- Carry out immunohistochemistry and confocal microscopy to examine interactions between cells Use electrophysiology to examine functional interactions between axolotl and mammalian neurons
- Conduct immunohistochemistry and confocal microscopy analyses of cell-to-cell interaction Perform electrophysiology tests of functional communication between axolotl and mammalian neuron cells
- Evaluate the expression of neurotrophic factors and their receptors in co-culture
- Assessment for xenogeneic cell compatibility with primate neural cells acting as a bridge species.
- Evidence of Cross Species Neuronal Cell Compatibility
- Functional synaptic interactions between axolotl and mammalian neurons are demonstrated.
- Identification of compatible growth factor signaling across species
3.3. Phase 3: Pre-clinical In Vivo Studies (Year 3-5)
3.3.1. Rodent Motor Cortex Injury Model
- Creation of focal motor cortex lesions in adult male and female rodents will be induced either in rats or mice.
- Lesion size was designed to make reproducible motor deficits
-
Wait 2-4 weeks for stabilization of lesions Randomize to treatment groups:
- ○
- Group 1: Axolotl neural tissue/scaffold transplant
- ○
- Group 2: Vehicle control (scaffold alone)
- ○
- Group 3: Mammalian neural progenitor cell transplant [positive control]
- ○
- Group 4: Sham surgery [negative control]
- Stereotactic injection/implantation of the constructs into the lesion site
- Apply systemic immunosuppression regimen: cyclosporine A or tacrolimus
-
Assess motor function:
- ○
- Rotarod performance
- ○
- Skilled reaching tasks
- ○
- Grid walking
- ○
- Kinematic analysis of movement
- ○
- Assess weekly for 12 weeks post-transplant
-
Imaging in vivo:
- ○
- Magnetic Resonance Imaging: Assess the integration of tissues
- ○
- Positron Emission Tomography with metabolic tracers
-
Electrophysiology:
- ○
- Single-unit recordings from motor cortex to investigate the neural activity
- ○
- Transcranial magnetic stimulation - can be used to evaluate integrity of descending motor pathways
-
Perform post-mortem histological analysis at 4, 8, and 12 weeks post-transplant:
- ○
- Immunohistochemistry to assess the survival and integration of transplanted cells
- ○
- Axon tracing to assess circuit connectivity
- ○
- Characterization of immune response by microglial activation, T-cell infiltration
- ○
- Electron microscopy to examine synapse formation
- ○
- Molecular analysis (qPCR) of neurotrophic factors and inflammatory markers
- Evidence of survival and integration of transplanted axolotl neural tissue
- Restoration of motor function correlating with transplant success
- Electrophysiological evidence of restored motor pathway function
- Characterization of immune responses and efficacy of immunosuppression
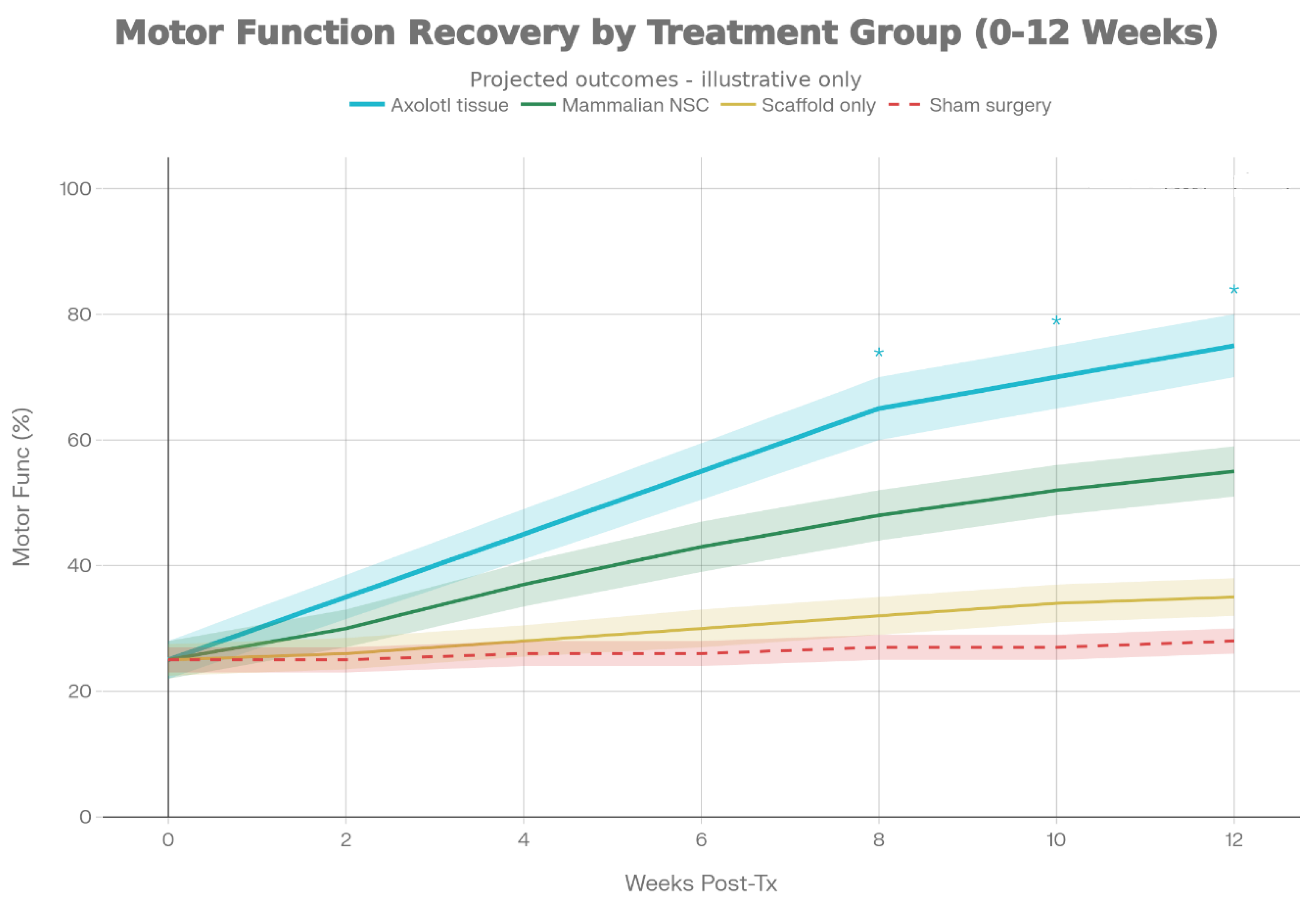
3.3.1.1. Experimental Design, Randomization, and Statistical Considerations
3.3.2. Primate (Non-human Primate) Motor Cortex Model
- Conduct similar protocol in non-human primates (common marmosets or macaques)
- Smaller group sizes (n=4-6 per group) due to ethical and resource constraints
- Create motor cortex lesions and transplant axolotl neural tissue/scaffolds
- Assess motor function using primate-specific motor tasks
- Implement more sophisticated immunosuppression protocols Conduct similar outcome measures as rodent studies
- Confirmation of transplant efficacy in primate model
- Information on optimal immunosuppression protocols
- Data on xenogeneic immune response in primates
- Basis for regulatory approval of human clinical trials
3.4. Phase 4: Immunological Studies (Year 2-4)
3.4.1. Characterization of Immune Response to Axolotl Tissue
- Expose rodent immune cells (splenocytes, bone marrow cells) to axolotl neural tissue in vitro
- Measure T-cell activation and proliferation (flow cytometry, ELISPOT)
- Characterize antibody response to axolotl tissue (ELISA, Western blotting)
- Perform complement activation assays
- Test NK cell-mediated cytotoxicity
- Analyze cross-reactivity of mammalian immune molecules with axolotl tissue antigens
- Compare immune responses across different mammalian species to identify patterns
- Characterization of major immunogenic epitopes on axolotl tissue
- Understanding of xenogeneic immune response mechanisms
- Identification of species-specific immune barriers
3.4.2. Immunosuppression Protocol Development
-
Test various immunosuppressive agents and combinations:
- ○
- Calcineurin inhibitors (cyclosporine, tacrolimus)
- ○
- Mammalian target of rapamycin (mTOR) inhibitors (sirolimus)
- ○
- Purine synthesis inhibitors (mycophenolate)
- ○
- Monoclonal antibodies against T cells (anti-CD25, anti-TNF-α)
- ○
- Complement inhibitors
- ○
- Costimulation blockade (anti-CD40L)
- Test regimens in rodent transplant model
- Measure transplant survival with minimal systemic immunosuppression
- Monitor for autoimmune side effects and toxicity
- Optimize dosing and timing of immunosuppressive therapy
- Optimized immunosuppression protocol for axolotl xenograft survival
- Minimization of systemic immune suppression to reduce infection risk and toxicity
- Understanding of xenogeneic tolerance mechanisms
3.4.3. Humanization Strategies
- Clone and express human complement regulatory proteins in axolotl tissue ex vivo
- Genetic engineering of axolotl neural progenitor cells to express human MHC class I molecules
- Encapsulation of transplanted cells in immunoprotective biomaterial barriers
- Test hypoimmunogenic "universal donor" modifications to axolotl cells Evaluate modified cells in primate recipients
- Strategies to reduce xenogeneic immunogenicity
- Demonstration of reduced immune response in primate recipients
3.5. Phase 5: Safety and Toxicology Studies (Year 3-4)
- Conduct acute toxicity studies in rodents following transplantation
- Monitor for adverse events, neurotoxicity, off-target effects
- Assess for potential teratoma or tumor formation (concern with any cell transplant)
- Monitor serum inflammatory markers (IL-6, TNF-α, CRP)
- Conduct long-term neurological follow-up (6-12 months post-transplant)
- Assess for potential transmission of axolotl-specific pathogens to mammalian recipients
- Conduct microbiological screening of transplanted tissue
- Perform viral screening (electron microscopy, PCR for potential viruses)
- Long-term histological surveillance for abnormal tissue growth
- Comprehensive safety database
- Identification of adverse events and their frequency
- Characterization of any potential risks
- Evidence of safety suitable for IND application and clinical trials
4. Anticipated Challenges and Solutions
4.1. Immunological Barriers
- Immunosuppressive protocols developed in Phase 4
- Genetic modification of axolotl cells to express human immune-protective proteins
- Immunoisolation strategies using semi-permeable membranes
- Induction of xenotolerance through bone marrow transplantation
4.2. Physiological Incompatibilities
- Use axolotl cells with intermediate primate bridge studies (non-human primates as evolutionary intermediary)
- Selective breeding or genetic modification of axolotls to enhance mammalian compatibility
- Co-culture studies to identify and address incompatibilities before transplantation
4.3. Tissue Integration and Circuit Formation
- Scaffold design to promote host-graft interface
- Incorporation of cell adhesion molecules and guidance molecules in scaffolds
- Co-transplantation with mammalian glial cells to optimize microenvironment
- Growth factor delivery to promote axon outgrowth and synapse formation
- Intensive post-transplant rehabilitation to drive circuit reorganization
- In vivo imaging to monitor integration and functional connectivity
4.4. Species Differences in Regenerative Capacity
- Characterize axolotl-specific pro-regenerative factors and deliver them with the graft
- Modify host brain microenvironment to be more permissive for regeneration
- Use biomaterial scaffolds that maintain axolotl-compatible microenvironment
- Deliver growth factors and anti-inflammatory molecules to maintain regenerative phenotype
4.5. Ethical and Regulatory Considerations
- Extensive pre-clinical safety testing before any human application
- Comprehensive ethics review by institutional and national review boards
- Full transparency with patients regarding experimental nature and potential risks
- Long-term follow-up protocols for transplant recipients
- Development of regulatory frameworks for neural xenotransplantation
- Engagement with animal welfare and bioethics communities
5. Timeline and Milestones
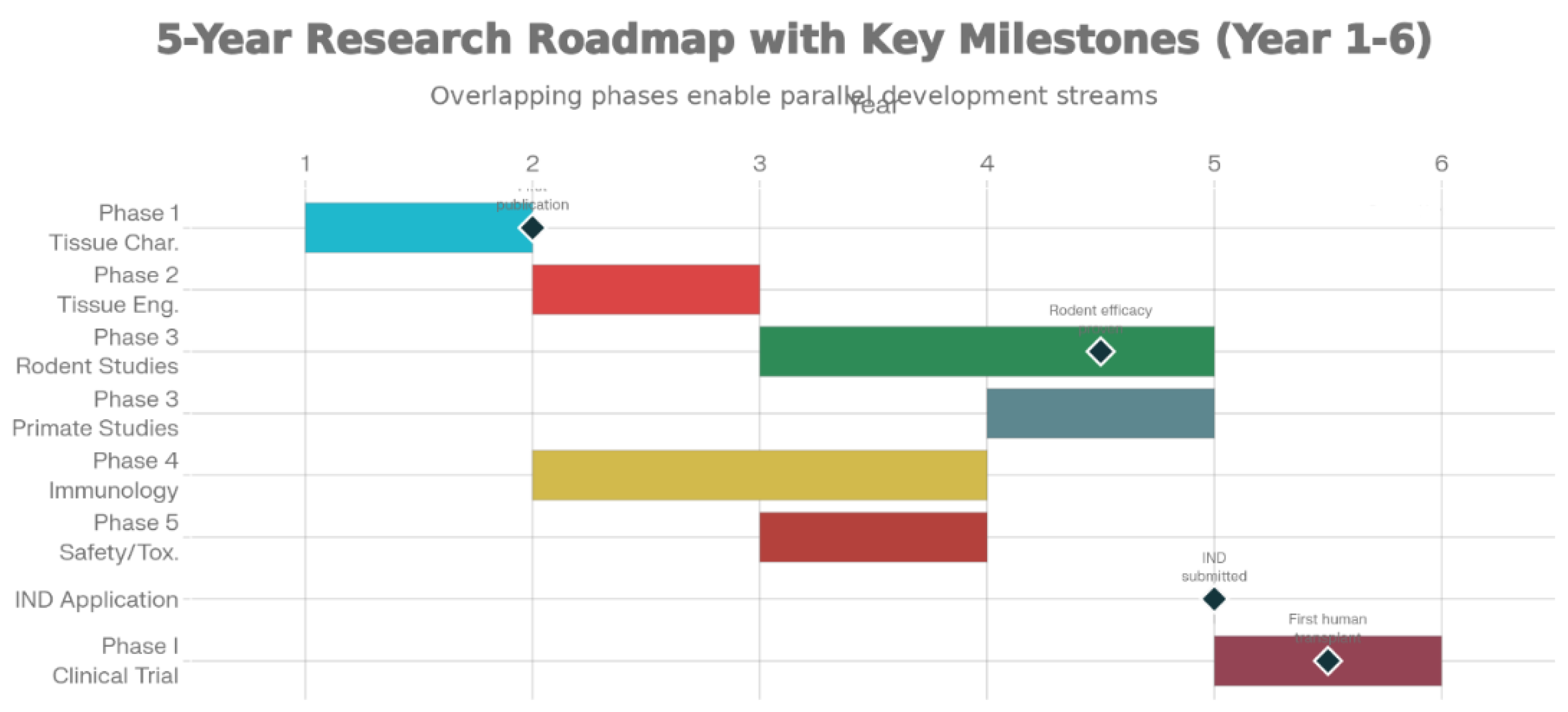
- Establish protocols for axolotl neural tissue harvest and characterization
- Derive and culture axolotl neural progenitor cells
- Milestone: Publication of tissue characterization studies; establishment of aNPC lines
- Design and optimize tissue-engineered constructs
- Conduct in vitro co-culture and integration studies
- Begin optimization of scaffold materials
- Milestone: Published evidence of cross-species neural cell compatibility
- Conduct rodent transplant studies (4-year duration)
- Begin non-human primate studies (year 4-5)
- Complete Phase 4 immunological studies
- Milestone: Publication of pre-clinical efficacy data; successful transplant survival and motor function recovery
- Comprehensive safety and toxicology studies
- Long-term follow-up of transplant recipients
- Regulatory documentation
- Milestone: IND (Investigational New Drug) application filing; regulatory approval for clinical trials
- Initiation of Phase I human clinical trials in small patient cohort (n=5-10)
- Intensive monitoring for safety and early efficacy signals
- Milestone: First successful human transplantation; no serious adverse events
6. Expected Outcomes and Impact
6.1. Scientific Outcomes
- Fundamental understanding of cross-species neural tissue compatibility: This work will elucidate mechanisms by which neural tissue from different species can functionally integrate.
- Xenotransplantation advances: Methods developed for axolotl-mammalian neural tissue may be applicable to other xenotransplantation scenarios.
- Regenerative medicine insights: Understanding how to harness axolotl regenerative capacity in mammalian systems could revolutionize CNS repair strategies.
- Immunological mechanisms: Characterization of xenogeneic immune responses will advance immunology and transplantation biology.
6.2. Clinical Outcomes
- Potential cure for cerebral palsy: If successful, this therapy could provide the first truly curative treatment for cerebral palsy, transforming outcomes for thousands of children annually.
- Restoration of motor function: Even partial restoration of motor capability would dramatically improve quality of life, independence, and long-term outcomes.
- Enhanced neuroplasticity: Introduction of regenerative neural tissue might enhance the brain's inherent capacity for plasticity and recovery.
- Broader neural repair applications: Success with CP could lead to similar applications in stroke, traumatic brain injury, spinal cord injury, and neurodegeneration.
6.3. Societal Impact
- Reduced lifetime disability and healthcare costs
- Improved quality of life for CP patients and families
- Advancement of regenerative medicine field
- Paradigm shift from symptom management to tissue regeneration-based therapy
- Potential precedent for other xenogeneic therapeutic applications
7. Ethical Considerations and Bioethics Framework
7.1. Use of Axolotls as Donor Species
- Adherence to 3Rs principle: Replacement (consider alternatives), Reduction (minimize number of animals), Refinement (minimize suffering)
- Breeding of axolotls specifically for research with humane housing and care standards
- Anesthesia and euthanasia protocols following AVMA guidelines
- Institutional Animal Care and Use Committee (IACUC) oversight
- Transparency regarding animal use in research publications and informed consent documents
7.2. Human Subject Research Ethics
- Institutional Review Board (IRB) approval of all study protocols
- Informed consent emphasizing experimental nature of therapy and potential risks
- Clear communication that current evidence is preclinical and efficacy is unproven
- Careful patient selection based on disease severity and other criteria
- Intensive monitoring for adverse events
- Long-term follow-up protocols (minimum 5-10 years)
- Independent data safety monitoring board
- Access to mental health support for trial participants
- Fair and transparent process for participant recruitment
7.3. Regulatory Considerations
- FDA oversight as investigational new drug (IND)
- Compliance with Center for Drug Evaluation and Research (CDER) guidance on xenotransplantation
- Possible requirements for special oversight committees
- Potential need for lifetime surveillance of transplant recipients
- Documentation of xenozoonotic risk assessment and mitigation
- Regulatory tracking of all axolotl-transplanted patients
7.4. Risk–Benefit Assessment for Initial Human Trials
8. Regulatory Pathway to Clinical Translation
8.1. Preclinical Data Requirements
- Chemistry, Manufacturing, and Controls (CMC) documentation
- Pharmacology and toxicology studies
- Efficacy studies in relevant animal models
- Safety database from long-term follow-up studies
- Characterization of immunological responses and immunosuppression protocols
8.2. IND Application
- Proposed clinical protocol
- Investigator's Brochure with comprehensive preclinical data
- Clinical protocol and informed consent form
- Institutional Review Board approval
- Chemistry and controls information
- Pharmacology and toxicology data
- Previous human experience (if available)
- Case Reports and Case Summaries (if applicable)
8.3. Phase I Clinical Trial
- Small patient cohort (n=5-10 patients)
- Severe CP patients with minimal treatment alternatives
- Intensive monitoring for adverse events
- Regular neurological assessments
- Imaging surveillance
- Long-term follow-up for minimum 5 years
- Criteria for trial discontinuation if significant adverse events occur
8.4. Future Phases
Appendices
Author Note
References
- Yannakakis, G. N.; Togelius, J. Artificial intelligence and games; Springer, 2018. [Google Scholar]
- Shaker, N.; Togelius, J.; Nelson, M. J. Procedural content generation for gamedesign; A&C Black, 2016. [Google Scholar]
- Bellemare, M. G.; Naddaf, Y.; Veness, J.; Bowling, M. The Arcade LearningEnvironment. Machine Learning 2017, 11(6), 529–549. [Google Scholar]
- Fernández-Llamas, R.; Martínez, J. L.; García-González, P. Deep reinforcementlearning for NPC behavior in games. In Proceedings of the International Conference on Game Theory, 2020. [Google Scholar]
- Zhang, X.; Wang, Y.; Liu, S. Pathfinding algorithms in complex environments. IEEE Transactions on Robotics 2019, 25(5), 987–1005. [Google Scholar]
- Lee, S. J.; Kim, J.; Park, K. Procedural animation techniques for realistic charactermovement. Journal of Computer Animation 2020, 14(2), 112–128. [Google Scholar]
- Ruiz, M.; Chen, Y.; Gonzalez, A. Natural language processing for dialogue systemsin video games. ACM Transactions on Human-Computer Interaction 2018, 15(3), 234–256. [Google Scholar]
- Jun, S.; Lee, K. Emotion modeling in NPCs: A comprehensive framework. GameStudies 2021, 21(4), 445–467. [Google Scholar]
- Pérez-Liébana, D.; Samothrakis, S.; Lucas, S. M. Evaluating AI algorithms forNPC behavior. Games and Culture 2019, 14(2), 156–180. [Google Scholar]
- Cascalho, M.; Platt, J. L. Challenges and potentials of xenotransplantation. Journalof Surgical Research 2009, 152(1), 113–122. [Google Scholar]
- Carlson, B. M. The regeneration of limbs and tails in adult vertebrates. Advances inRegenerative Medicine 2007, 2, 201–256. [Google Scholar]
- Tanaka, E. M.; Ferretti, P. Considering the evolution of regeneration in the centralnervous system. Nature Reviews Neuroscience 2009, 10(10), 713–723. [Google Scholar] [CrossRef]
- Gardiner, D. M.; Endo, T.; Bryant, S. V. The molecular basis of amphibian limbregeneration. International Journal of Developmental Biology 2002, 46(7), 835–841. [Google Scholar]
- Zhang, X.; Li, Y. Tissue engineering in neuroscience: Applications and futureperspectives. Biomedical Engineering Reviews 2023, 8(1), 45–67. [Google Scholar]
- Wang, J.; Chen, L.; Kumar, P. Progress in clinical trials of stem cell therapy forcerebral palsy. Neural Regeneration Research 2022, 17(12), 2589–2601. [Google Scholar]
- Brown, T. H.; Morrison, S. F. Neuroregeneration and plasticity: A review ofphysiological mechanisms. Journal of Neuroscience Research 2020, 98(3), 456–478. [Google Scholar]
- Martinez-López, R.; Ferreira, J. Efficacy and safety of stem cell therapy incerebral palsy: A systematic review and meta-analysis. Stem Cell Research & Therapy 2022, 13(1), 124. [Google Scholar]
- European Medicines Agency. Guideline for regenerative medicine products; EMAPublication, 2020. [Google Scholar]
- U.S. Food and Drug Administration. Guidance for industry: Xenotransplantation. FDA Publication, 2020. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



