Submitted:
09 February 2026
Posted:
10 February 2026
You are already at the latest version
Abstract
Keywords:
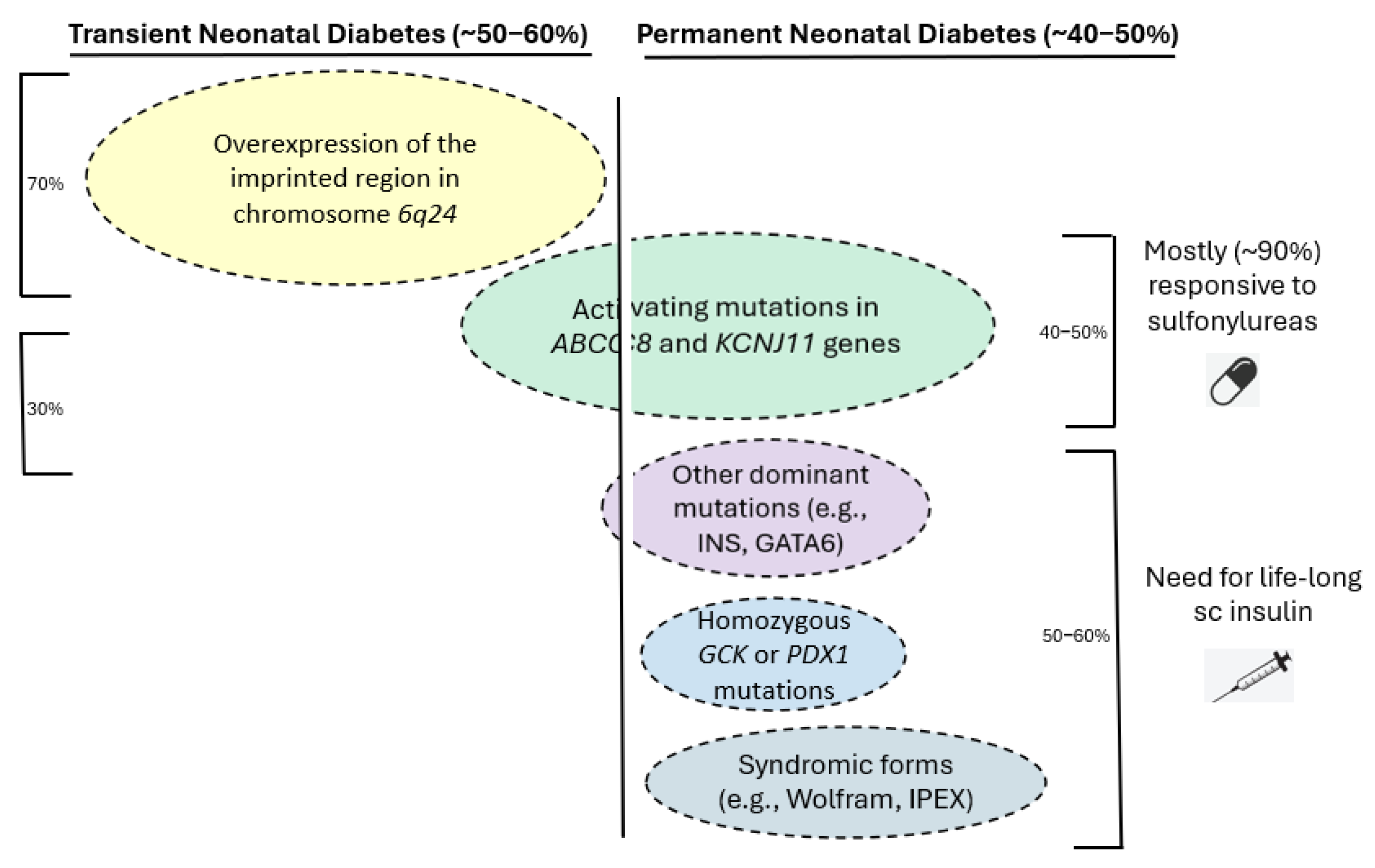
1. Introduction
2. Methods
3. Genetic Causes of NDM
3.1. Imprinting Defects of the 6q24 Locus
3.2. Defects in Beta-Cell Function and Insulin Secretion (Table 2)
3.2.1. ATP-Sensitive Potassium (KATP) Channels Mutations
3.2.2. Glucokinase (GCK) Gene Mutations
3.2.3. SLC2A2 Gene Mutations
3.3. Defects in Insulin Biosynthesis and Beta-Cell Destruction (Table 3)
3.3.1. Insulin (INS) Gene Mutations
3.3.2. Gene Mutations Associated with ER Stress Response
3.4. Defects in Pancreatic Morphogenesis
3.4.1. PDX1 Gene Mutations
3.4.2. PTF1A Gene Mutations
3.4.3. GATA6 Gene Mutations
3.4.4. HNF1B Gene Mutations
3.5. Defects in Beta-Cell Development (Table 4)
3.5.1. NEUROG3 Gene Mutations
3.5.2. NEUROD1 Gene Mutations
4. Transient Hyperglycemic States in Neonates
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hay, W. W.; Rozance, P. J. Neonatal Hyperglycemia—Causes, Treatments, and Cautions. J. Pediatr. 2018, 200, 6–8. [Google Scholar] [CrossRef]
- Beardsall, K. Hyperglycaemia in the Newborn Infant. Physiology Verses Pathology. Front. Pediatr. 2021, 9. [Google Scholar] [CrossRef]
- Angelis, D.; Jaleel, M. A.; Brion, L. P. Hyperglycemia and Prematurity: A Narrative Review. Pediatr. Res. 2023, 94, 892–903. [Google Scholar] [CrossRef]
- Beltrand, J.; Busiah, K.; Vaivre-Douret, L.; Fauret, A. L.; Berdugo, M.; Cavé, H.; Polak, M. Neonatal Diabetes Mellitus. Front. Pediatr. 2020, 8. [Google Scholar] [CrossRef]
- Hammoud, B.; Greeley, S. A. W. Growth and Development in Monogenic Forms of Neonatal Diabetes. Curr. Opin. Endocrinol. Diabetes Obes. 2022, 29, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Golshan-Tafti, M.; Dastgheib, S. A.; Bahrami, R.; Yeganegi, M.; Aghasipour, M.; Marzbanrad, Z.; Saeida-Ardekani, M.; Shahbazi, A.; Omidi, A.; Lookzadeh, M. H.; et al. Genetic Underpinnings of Neonatal Diabetes: A Review of Current Research. Egyptian Journal of Medical Human Genetics 2024, 25, 119. [Google Scholar] [CrossRef]
- Al-Khawaga, S.; Mohammed, I.; Saraswathi, S.; Haris, B.; Hasnah, R.; Saeed, A.; Almabrazi, H.; Syed, N.; Jithesh, P.; El Awwa, A.; et al. The Clinical and Genetic Characteristics of Permanent Neonatal Diabetes (PNDM) in the State of Qatar. Mol. Genet. Genomic Med. 2019, 7(10). [Google Scholar] [CrossRef]
- Habeb, A. M.; Al-Magamsi, M. S.; Eid, I. M.; Ali, M. I.; Hattersley, A. T.; Hussain, K.; Ellard, S. Incidence, Genetics, and Clinical Phenotype of Permanent Neonatal Diabetes Mellitus in Northwest Saudi Arabia. Pediatr. Diabetes 2012, 13, 499–505. [Google Scholar] [CrossRef] [PubMed]
- RubioCabezas, O.; Flanagan, S. E.; Damhuis, A.; Hattersley, A. T.; Ellard, S. KATP Channel Mutations in Infants with Permanent Diabetes Diagnosed after 6 Months of Life. Pediatr. Diabetes 2012, 13, 322–325. [Google Scholar] [CrossRef]
- Kabeer, A. S. VH. Genetics of Neonatal Diabetes: An Update. Genetic Clinics 2024, 17(4), 17–23. [Google Scholar]
- Lemelman, M. B.; Letourneau, L.; Greeley, S. A. W. Neonatal Diabetes Mellitus. Clin. Perinatol. 2018, 45, 41–59. [Google Scholar] [CrossRef]
- Shah, B.; Kataria, A.; Palliyil Gopi, R.; Mally, P. Neonatal Diabetes Mellitus: Current Perspective. Res. Rep. Neonatol. 2014, 55. [Google Scholar] [CrossRef]
- Dahl, A.; Kumar, S. Recent Advances in Neonatal Diabetes. Diabetes Metab. Syndr. Obes. 2020, Volume 13, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Letourneau, L. R.; Carmody, D.; Wroblewski, K.; Denson, A. M.; Sanyoura, M.; Naylor, R. N.; Philipson, L. H.; Greeley, S. A. W. Diabetes Presentation in Infancy: High Risk of Diabetic Ketoacidosis. Diabetes Care 2017, 40, e147–e148. [Google Scholar] [CrossRef] [PubMed]
- Novak, A.; Bowman, P.; Kraljevic, I.; Tripolski, M.; Houghton, J. A. L.; De Franco, E.; Shepherd, M. H.; Skrabic, V.; Patel, K. A. Transient Neonatal Diabetes: An Etiologic Clue for the Adult Diabetologist. Can. J. Diabetes 2020, 44, 128–130. [Google Scholar] [CrossRef]
- Greeley, S. A. W.; Polak, M.; Njølstad, P. R.; Barbetti, F.; Williams, R.; Castano, L.; Raile, K.; Chi, D. V.; Habeb, A.; Hattersley, A. T.; et al. ISPAD Clinical Practice Consensus Guidelines 2022: The Diagnosis and Management of Monogenic Diabetes in Children and Adolescents. Pediatr. Diabetes 2022, 23, 1188–1211. [Google Scholar] [CrossRef]
- Barbetti, F.; Deeb, A.; Suzuki, S. Neonatal Diabetes Mellitus around the World: Update 2024. J. Diabetes Investig. 2024, 15, 1711–1724. [Google Scholar] [CrossRef]
- Gobble, M. R. S.; Stone, S. I. Neonatal and Syndromic Forms of Diabetes. Curr. Diab. Rep. 2025, 25, 26. [Google Scholar] [CrossRef]
- Temple, I. K.; Shield, J. P. H. 6q24 Transient Neonatal Diabetes. Rev. Endocr. Metab. Disord. 2010, 11, 199–204. [Google Scholar] [CrossRef]
- Alkorta-Aranburu, G.; Sukhanova, M.; Carmody, D.; Hoffman, T.; Wysinger, L.; Keller-Ramey, J.; Li, Z.; Johnson, A. K.; Kobiernicki, F.; Botes, S.; et al. Improved Molecular Diagnosis of Patients with Neonatal Diabetes Using a Combined Next-Generation Sequencing and MS-MLPA Approach. Journal of Pediatric Endocrinology and Metabolism 2016, 29. [Google Scholar] [CrossRef]
- Mackay, D. J. G.; Temple, I. K. Transient Neonatal Diabetes Mellitus Type 1. Am. J. Med. Genet. C Semin. Med. Genet. 2010, 154C, 335–342. [Google Scholar] [CrossRef]
- Polak, M.; Cavé, H. Neonatal Diabetes Mellitus: A Disease Linked to Multiple Mechanisms. Orphanet J. Rare Dis. 2007, 2, 12. [Google Scholar] [CrossRef] [PubMed]
- Docherty, L. E.; Kabwama, S.; Lehmann, A.; Hawke, E.; Harrison, L.; Flanagan, S. E.; Ellard, S.; Hattersley, A. T.; Shield, J. P. H.; Ennis, S.; et al. Clinical Presentation of 6q24 Transient Neonatal Diabetes Mellitus (6q24 TNDM) and Genotype–Phenotype Correlation in an International Cohort of Patients. Diabetologia 2013, 56, 758–762. [Google Scholar] [CrossRef] [PubMed]
- Touati, A.; Errea-Dorronsoro, J.; Nouri, S.; Halleb, Y.; Pereda, A.; Mahdhaoui, N.; Ghith, A.; Saad, A.; Perez de Nanclares, G.; H’mida ben brahim, D. Transient Neonatal Diabetes Mellitus and Hypomethylation at Additional Imprinted Loci: Novel ZFP57 Mutation and Review on the Literature. Acta Diabetol. 2019, 56, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Vega-Benedetti, A. F.; Saucedo, C.; Zavattari, P.; Vanni, R.; Zugaza, J. L.; Parada, L. A. PLAGL1: An Important Player in Diverse Pathological Processes. J. Appl. Genet. 2017, 58, 71–78. [Google Scholar] [CrossRef]
- Liu, P.-Y.; Hsieh, T.-Y.; Liu, S.-T.; Chang, Y.-L.; Lin, W.-S.; Wang, W.-M.; Huang, S.-M. Zac1, an Sp1-like Protein, Regulates Human P21 Gene Expression in HeLa Cells. Exp. Cell Res. 2011, 317, 2925–2937. [Google Scholar] [CrossRef]
- Huang, S.-M.; Schönthal, A. H.; Stallcup, M. R. Enhancement of P53-Dependent Gene Activation by the Transcriptional Coactivator Zac1. Oncogene 2001, 20, 2134–2143. [Google Scholar] [CrossRef]
- Ma, D.; Shield, J. P. H.; Dean, W.; Leclerc, I.; Knauf, C.; Burcelin, R.; Rutter, G. A.; Kelsey, G. Impaired Glucose Homeostasis in Transgenic Mice Expressing the Human Transient Neonatal Diabetes Mellitus Locus, TNDM. Journal of Clinical Investigation 2004, 114, 339–348. [Google Scholar] [CrossRef]
- Hoffmann, A.; Spengler, D. Transient Neonatal Diabetes Mellitus Gene Zac1 Impairs Insulin Secretion in Mice through Rasgrf1. Mol. Cell. Biol. 2012, 32, 2549–2560. [Google Scholar] [CrossRef]
- Busiah, K.; Drunat, S.; Vaivre-Douret, L.; Bonnefond, A.; Simon, A.; Flechtner, I.; Gérard, B.; Pouvreau, N.; Elie, C.; Nimri, R.; et al. Neuropsychological Dysfunction and Developmental Defects Associated with Genetic Changes in Infants with Neonatal Diabetes Mellitus: A Prospective Cohort Study. Lancet Diabetes Endocrinol. 2013, 1, 199–207. [Google Scholar] [CrossRef]
- McCullough, M. E.; Letourneau-Freiberg, L. R.; Bowden, T. L.; Kandasamy, B.; Ray, A.; Wroblewski, K.; del Gaudio, D.; Mackay, D. J. G.; Philipson, L. H.; Greeley, S. A. W. Clinical Characteristics and Remission Monitoring of 6q24-Related Transient Neonatal Diabetes. Pediatr. Diabetes 2024. [Google Scholar] [CrossRef]
- Bonfanti, R.; Iafusco, D.; Rabbone, I.; Diedenhofen, G.; Bizzarri, C.; Patera, P. I.; Reinstadler, P.; Costantino, F.; Calcaterra, V.; Iughetti, L.; et al. Differences between Transient Neonatal Diabetes Mellitus Subtypes Can Guide Diagnosis and Therapy. Eur. J. Endocrinol. 2021, 184, 575–585. [Google Scholar] [CrossRef]
- Pietrusiński, M.; Grzybowska-Adamowicz, J.; Płoszaj, T.; Skoczylas, S.; Borowiec, M.; Piekarska, K.; Skowrońska, B.; Wajda-Cuszlag, M.; Mazur, A.; Zmysłowska, A. The Clinical and Diagnostic Characterization of 6q24-Related Transient Neonatal Diabetes Mellitus: A Polish Pediatric Cohort Study. Biomedicines 2025, 13, 2492. [Google Scholar] [CrossRef]
- Shoucri, B. M.; Thambundit, A.; Chia, D. J. 6q24-Related Transient Neonatal Diabetes Mellitus Presenting With Severe Diabetic Ketoacidosis and Multiorgan Failure. JCEM Case Reports 2025, 3. [Google Scholar] [CrossRef]
- Garcin, L.; Kariyawasam, D.; Busiah, K.; Fauret-Amsellem, A.-L.; Le Bourgeois, F.; Vaivre-Douret, L.; Cavé, H.; Polak, M.; Beltrand, J. Successful Off-Label Sulfonylurea Treatment of Neonatal Diabetes Mellitus Due to Chromosome 6 Abnormalities. Pediatr. Diabetes 2018, 19, 663–669. [Google Scholar] [CrossRef] [PubMed]
- Carmody, D.; Beca, F. A.; Bell, C. D.; Hwang, J. L.; Dickens, J. T.; Devine, N. A.; Mackay, D. J. G.; Temple, I. K.; Hays, L. R.; Naylor, R. N.; et al. Role of Noninsulin Therapies Alone or in Combination in Chromosome 6q24-Related Transient Neonatal Diabetes: Sulfonylurea Improves but Does Not Always Normalize Insulin Secretion. Diabetes Care 2015, 38, e86–e87. [Google Scholar] [CrossRef] [PubMed]
- Ashcroft, F. M. ATP-Sensitive Potassium Channelopathies: Focus on Insulin Secretion. Journal of Clinical Investigation 2005, 115, 2047–2058. [Google Scholar] [CrossRef] [PubMed]
- Pipatpolkai, T.; Usher, S.; Stansfeld, P. J.; Ashcroft, F. M. New Insights into KATP Channel Gene Mutations and Neonatal Diabetes Mellitus. Nat. Rev. Endocrinol. 2020, 16, 378–393. [Google Scholar] [CrossRef]
- Babenko, A. P.; Polak, M.; Cavé, H.; Busiah, K.; Czernichow, P.; Scharfmann, R.; Bryan, J.; Aguilar-Bryan, L.; Vaxillaire, M.; Froguel, P. Activating Mutations in the ABCC8 Gene in Neonatal Diabetes Mellitus. New England Journal of Medicine 2006, 355, 456–466. [Google Scholar] [CrossRef]
- Edghill, E. L.; Gloyn, A. L.; Goriely, A.; Harries, L. W.; Flanagan, S. E.; Rankin, J.; Hattersley, A. T.; Ellard, S. Origin of de Novo KCNJ11 Mutations and Risk of Neonatal Diabetes for Subsequent Siblings. J. Clin. Endocrinol. Metab. 2007, 92, 1773–1777. [Google Scholar] [CrossRef]
- Shimomura, K.; Maejima, Y. KATP Channel Mutations and Neonatal Diabetes. Internal Medicine 2017, 56, 2387–2393. [Google Scholar] [CrossRef] [PubMed]
- Matsutani, N.; Furuta, H.; Matsuno, S.; Oku, Y.; Morita, S.; Uraki, S.; Doi, A.; Furuta, M.; Iwakura, H.; Ariyasu, H.; et al. Identification of a Compound Heterozygous Inactivating ABCC 8 Gene Mutation Responsible for Young-onset Diabetes with Exome Sequencing. J. Diabetes Investig. 2020, 11, 333–336. [Google Scholar] [CrossRef] [PubMed]
- Ellard, S.; Flanagan, S. E.; Girard, C. A.; Patch, A.-M.; Harries, L. W.; Parrish, A.; Edghill, E. L.; Mackay, D. J. G.; Proks, P.; Shimomura, K.; et al. Permanent Neonatal Diabetes Caused by Dominant, Recessive, or Compound Heterozygous SUR1 Mutations with Opposite Functional Effects. The American Journal of Human Genetics 2007, 81, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Edghill, E. L.; Flanagan, S. E.; Ellard, S. Permanent Neonatal Diabetes Due to Activating Mutations in ABCC8 and KCNJ11. Rev. Endocr. Metab. Disord. 2010, 11, 193–198. [Google Scholar] [CrossRef]
- Besser, R. E. J.; Flanagan, S. E.; Mackay, D. G. J.; Temple, I. K.; Shepherd, M. H.; Shields, B. M.; Ellard, S.; Hattersley, A. T. Prematurity and Genetic Testing for Neonatal Diabetes. Pediatrics 2016, 138. [Google Scholar] [CrossRef]
- Ngoc, C. T. B.; Dien, T. M.; De Franco, E.; Ellard, S.; Houghton, J. A. L.; Lan, N. N.; Thao, B. P.; Khanh, N. N.; Flanagan, S. E.; Craig, M. E.; et al. Molecular Genetics, Clinical Characteristics, and Treatment Outcomes of KATP-Channel Neonatal Diabetes Mellitus in Vietnam National Children’s Hospital. Front. Endocrinol. (Lausanne) 2021, 12. [Google Scholar] [CrossRef]
- Mujahed, R. H.; Shawabka, A. M.; Daghlis, N. M.; Thweib, A. A.; Rabba, H. A.; Makhlouf, T. Z. De Novo KCNJ11 Mutation in an Infant With Neonatal Diabetes Mellitus Presenting as Diabetic Ketoacidosis: A Case Report and Literature Review. J. Investig. Med. High Impact Case Rep. 2025, 13. [Google Scholar] [CrossRef]
- Li, B.; Xi, X.; Roane, D. S.; Ryan, D. H.; Martin, R. J. Distribution of Glucokinase, Glucose Transporter GLUT2, Sulfonylurea Receptor-1, Glucagon-like Peptide-1 Receptor and Neuropeptide Y Messenger RNAs in Rat Brain by Quantitative Real Time RT-PCR. Molecular Brain Research 2003, 113, 139–142. [Google Scholar] [CrossRef]
- Ashcroft, F. M. ATP-Sensitive K + Channels and Disease: From Molecule to Malady. American Journal of Physiology-Endocrinology and Metabolism 2007, 293, E880–E889. [Google Scholar] [CrossRef]
- Esquiaveto-Aun, A. M.; De Mello, M. P.; Paulino, M. F. V. M.; Minicucci, W. J.; Guerra-Júnior, G.; De Lemos-Marini, S. H. V. A New Compound Heterozygosis for Inactivating Mutations in the Glucokinase Gene as Cause of Permanent Neonatal Diabetes Mellitus (PNDM) in Double-First Cousins. Diabetol. Metab. Syndr. 2015, 7, 101. [Google Scholar] [CrossRef]
- Osbak, K. K.; Colclough, K.; Saint-Martin, C.; Beer, N. L.; Bellanné-Chantelot, C.; Ellard, S.; Gloyn, A. L. Update on Mutations in Glucokinase ( GCK ), Which Cause Maturity-Onset Diabetes of the Young, Permanent Neonatal Diabetes, and Hyperinsulinemic Hypoglycemia. Hum. Mutat. 2009, 30, 1512–1526. [Google Scholar] [CrossRef]
- Abu Aqel, Y.; Alnesf, A.; Aigha, I. I.; Islam, Z.; Kolatkar, P. R.; Teo, A.; Abdelalim, E. M. Glucokinase (GCK) in Diabetes: From Molecular Mechanisms to Disease Pathogenesis. Cell. Mol. Biol. Lett. 2024, 29, 120. [Google Scholar] [CrossRef] [PubMed]
- Raimondo, A.; Chakera, A. J.; Thomsen, S. K.; Colclough, K.; Barrett, A.; De Franco, E.; Chatelas, A.; Demirbilek, H.; Akcay, T.; Alawneh, H.; et al. Phenotypic Severity of Homozygous GCK Mutations Causing Neonatal or Childhood-Onset Diabetes Is Primarily Mediated through Effects on Protein Stability. Hum. Mol. Genet. 2014, 23, 6432–6440. [Google Scholar] [CrossRef] [PubMed]
- Bolu, S. A Family With Novel Homozygous Deletion Mutation (c.1255delT; p.Phe419Serfs*12) in GCK Gene Which Is A Rare Cause of Permanent Neonatal Diabetes Mellitus. Turk Pediatri Ars. 2019. [Google Scholar] [CrossRef] [PubMed]
- Oza, C. M.; Karguppikar, M. B.; Khadilkar, V.; Khadilkar, A. Variable Presentations of GCK Gene Mutation in a Family. BMJ Case Rep. 2022, 15, e246699. [Google Scholar] [CrossRef]
- Al Senani, A.; Hamza, N.; Al Azkawi, H.; Al Kharusi, M.; Al Sukaiti, N.; Al Badi, M.; Al Yahyai, M.; Johnson, M.; De Franco, E.; Flanagan, S.; et al. Genetic Mutations Associated with Neonatal Diabetes Mellitus in Omani Patients. Journal of Pediatric Endocrinology and Metabolism 2018, 31, 195–204. [Google Scholar] [CrossRef]
- Sun, B.; Chen, H.; Xue, J.; Li, P.; Fu, X. The Role of GLUT2 in Glucose Metabolism in Multiple Organs and Tissues. Mol. Biol. Rep. 2023, 50, 6963–6974. [Google Scholar] [CrossRef]
- Guillam, M.-T.; Hümmler, E.; Schaerer, E.; Wu, J.-Y.; Birnbaum, M. J.; Beermann, F.; Schmidt, A.; Dériaz, N.; Thorens, B. Early Diabetes and Abnormal Postnatal Pancreatic Islet Development in Mice Lacking Glut-2. Nat. Genet. 1997, 17, 327–330. [Google Scholar] [CrossRef]
- Khandelwal, P.; Sinha, A.; Jain, V.; Houghton, J.; Hari, P.; Bagga, A. Fanconi Syndrome and Neonatal Diabetes: Phenotypic Heterogeneity in Patients with GLUT2 Defects. CEN Case Rep. 2018, 7, 1–4. [Google Scholar] [CrossRef]
- Sansbury, F. H.; Flanagan, S. E.; Houghton, J. A. L.; Shuixian Shen, F. L.; Al-Senani, A. M. S.; Habeb, A. M.; Abdullah, M.; Kariminejad, A.; Ellard, S.; Hattersley, A. T. SLC2A2 Mutations Can Cause Neonatal Diabetes, Suggesting GLUT2 May Have a Role in Human Insulin Secretion. Diabetologia 2012, 55, 2381–2385. [Google Scholar] [CrossRef]
- Yoo, H.-W.; Shin, Y.-L.; Seo, E.-J.; Kim, G.-H. Identification of a Novel Mutation in the GLUT2 Gene in a Patient with Fanconi-Bickel Syndrome Presenting with Neonatal Diabetes Mellitus and Galactosaemia. Eur. J. Pediatr. 2002, 161, 351–353. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Sun, J.; Cui, J.; Chen, W.; Guo, H.; Barbetti, F.; Arvan, P. INS-Gene Mutations: From Genetics and Beta Cell Biology to Clinical Disease. Mol. Aspects Med. 2015, 42, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Sui, L.; Du, Q.; Haataja, L.; Yin, Y.; Viola, R.; Xu, S.; Nielsson, C. U.; Leibel, R. L.; Barbetti, F.; et al. Permanent Neonatal Diabetes-Causing Insulin Mutations Have Dominant Negative Effects on Beta Cell Identity. Mol. Metab. 2024, 80, 101879. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Saint-Martin, C.; Xu, J.; Ding, L.; Wang, R.; Feng, W.; Liu, M.; Shu, H.; Fan, Z.; Haataja, L.; et al. Biological Behaviors of Mutant Proinsulin Contribute to the Phenotypic Spectrum of Diabetes Associated with Insulin Gene Mutations. Mol. Cell. Endocrinol. 2020, 518, 111025. [Google Scholar] [CrossRef]
- Haataja, L.; Manickam, N.; Soliman, A.; Tsai, B.; Liu, M.; Arvan, P. Disulfide Mispairing During Proinsulin Folding in the Endoplasmic Reticulum. Diabetes 2016, 65, 1050–1060. [Google Scholar] [CrossRef]
- Haataja, L.; Arunagiri, A.; Hassan, A.; Regan, K.; Tsai, B.; Dhayalan, B.; Weiss, M. A.; Liu, M.; Arvan, P. Distinct States of Proinsulin Misfolding in MIDY. Cellular and Molecular Life Sciences 2021, 78, 6017–6031. [Google Scholar] [CrossRef]
- Yang, Y.; Shu, H.; Hu, J.; Li, L.; Wang, J.; Chen, T.; Zhen, J.; Sun, J.; Feng, W.; Xiong, Y.; et al. A Novel Nonsense INS Mutation Causes Inefficient Preproinsulin Translocation Into the Endoplasmic Reticulum. Front. Endocrinol. (Lausanne). 2022, 12. [Google Scholar] [CrossRef]
- Balboa, D.; Saarimäki-Vire, J.; Borshagovski, D.; Survila, M.; Lindholm, P.; Galli, E.; Eurola, S.; Ustinov, J.; Grym, H.; Huopio, H.; et al. Insulin Mutations Impair Beta-Cell Development in a Patient-Derived IPSC Model of Neonatal Diabetes. Elife 2018, 7. [Google Scholar] [CrossRef]
- Edghill, E. L.; Flanagan, S. E.; Patch, A.-M.; Boustred, C.; Parrish, A.; Shields, B.; Shepherd, M. H.; Hussain, K.; Kapoor, R. R.; Malecki, M.; et al. Insulin Mutation Screening in 1,044 Patients With Diabetes. Diabetes 2008, 57, 1034–1042. [Google Scholar] [CrossRef]
- Garin, I.; Edghill, E. L.; Akerman, I.; Rubio-Cabezas, O.; Rica, I.; Locke, J. M.; Maestro, M. A.; Alshaikh, A.; Bundak, R.; del Castillo, G.; et al. Recessive Mutations in the INS Gene Result in Neonatal Diabetes through Reduced Insulin Biosynthesis. Proceedings of the National Academy of Sciences 2010, 107, 3105–3110. [Google Scholar] [CrossRef]
- Støy, J.; Steiner, D. F.; Park, S.-Y.; Ye, H.; Philipson, L. H.; Bell, G. I. Clinical and Molecular Genetics of Neonatal Diabetes Due to Mutations in the Insulin Gene. Rev. Endocr. Metab. Disord. 2010, 11, 205–215. [Google Scholar] [CrossRef]
- Rubio-Cabezas, O.; Patch, A.-M.; Minton, J. A. L.; Flanagan, S. E.; Edghill, E. L.; Hussain, K.; Balafrej, A.; Deeb, A.; Buchanan, C. R.; Jefferson, I. G.; et al. Wolcott-Rallison Syndrome Is the Most Common Genetic Cause of Permanent Neonatal Diabetes in Consanguineous Families. J. Clin. Endocrinol. Metab. 2009, 94, 4162–4170. [Google Scholar] [CrossRef]
- Habeb, A. M.; Flanagan, S. E.; Deeb, A.; Al-Alwan, I.; Alawneh, H.; Balafrej, A. A. L.; Mutair, A.; Hattersley, A. T.; Hussain, K.; Ellard, S. Permanent Neonatal Diabetes: Different Aetiology in Arabs Compared to Europeans. Arch. Dis. Child. 2012, 97, 721–723. [Google Scholar] [CrossRef] [PubMed]
- Asl, S. N.; Vakili, R.; Vakili, S.; Soheilipour, F.; Hashemipour, M.; Ghahramani, S.; De Franco, E.; Yaghootkar, H. Wolcott-Rallison Syndrome in Iran: A Common Cause of Neonatal Diabetes. Journal of Pediatric Endocrinology and Metabolism 2019, 32, 607–613. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Lv, Y.; Zhao, N.; Guan, G.; Wang, J. Protein Kinase R-like ER Kinase and Its Role in Endoplasmic Reticulum Stress-Decided Cell Fate. Cell Death Dis. 2015, 6, e1822–e1822. [Google Scholar] [CrossRef] [PubMed]
- Aldrian, D.; Bochdansky, C.; Kavallar, A. M.; Mayerhofer, C.; Deeb, A.; Habeb, A.; Romera Rabasa, A.; Khadilkar, A.; Uçar, A.; Knoppke, B.; et al. Natural History of Wolcott-Rallison Syndrome: A Systematic Review and Follow-up Study. Liver International 2024, 44, 811–822. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, F.; Habibi, M.; Enayati, S.; Bitarafan, F.; Razzaghy-Azar, M.; Sotodeh, A.; Omran, S. P.; Maroofian, R.; Amoli, M. M. A Genotype-First Approach for Clinical and Genetic Evaluation of Wolcott-Rallison Syndrome in a Large Cohort of Iranian Children With Neonatal Diabetes. Can. J. Diabetes 2018, 42, 272–275. [Google Scholar] [CrossRef] [PubMed]
- De Franco, E.; Caswell, R.; Johnson, M. B.; Wakeling, M. N.; Zung, A.; Dũng, V. C.; Bích Ngọc, C. T.; Goonetilleke, R.; Vivanco Jury, M.; El-Khateeb, M.; et al. De Novo Mutations in EIF2B1 Affecting EIF2 Signaling Cause Neonatal/Early-Onset Diabetes and Transient Hepatic Dysfunction. Diabetes 2020, 69, 477–483. [Google Scholar] [CrossRef]
- Yang, J.; Zhen, J.; Feng, W.; Fan, Z.; Ding, L.; Yang, X.; Huang, Y.; Shu, H.; Xie, J.; Li, X.; et al. IER3IP1 Is Critical for Maintaining Glucose Homeostasis through Regulating the Endoplasmic Reticulum Function and Survival of β Cells. Proceedings of the National Academy of Sciences 2022, 119. [Google Scholar] [CrossRef]
- Montaser, H.; Leppänen, S.; Vähäkangas, E.; Bäck, N.; Grace, A.; Eurola, S.; Ibrahim, H.; Lithovius, V.; Stephens, S. B.; Barsby, T.; et al. IER3IP1 Mutations Cause Neonatal Diabetes Due to Impaired Proinsulin Trafficking. Diabetes 2025, 74, 514–527. [Google Scholar] [CrossRef]
- Wu, L.; Zhang, J.; Li, D.; Zhang, Z.; Ni, Q.; Han, R.; Ye, L.; Zhang, Y.; Hong, J.; Wang, W.; et al. Novel WFS1 Variants Are Associated with Different Diabetes Phenotypes. Front. Genet. 2024, 15. [Google Scholar] [CrossRef]
- De Franco, E.; Flanagan, S. E.; Yagi, T.; Abreu, D.; Mahadevan, J.; Johnson, M. B.; Jones, G.; Acosta, F.; Mulaudzi, M.; Lek, N.; et al. Dominant ER Stress–Inducing WFS1 Mutations Underlie a Genetic Syndrome of Neonatal/Infancy-Onset Diabetes, Congenital Sensorineural Deafness, and Congenital Cataracts. Diabetes 2017, 66, 2044–2053. [Google Scholar] [CrossRef]
- Ebrahim, N.; Shakirova, K.; Dashinimaev, E. PDX1 Is the Cornerstone of Pancreatic β-Cell Functions and Identity. Front. Mol. Biosci. 2022, 9. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zanfardino, A.; Schiaffini, R.; Ishibashi, J.; Daniel, B.; Haemmerle, M. W.; Rapini, N.; Piscopo, A.; Miraglia del Giudice, E.; Digilio, M. C.; et al. Neonatal Diabetes–Associated Missense PDX1 Variant Disrupts Chromatin Association and Protein-Protein Interaction. JCI Insight 2025, 10. [Google Scholar] [CrossRef] [PubMed]
- Schwitzgebel, V. M.; Mamin, A.; Brun, T.; Ritz-Laser, B.; Zaiko, M.; Maret, A.; Jornayvaz, F. R.; Theintz, G. E.; Michielin, O.; Melloul, D.; et al. Agenesis of Human Pancreas Due to Decreased Half-Life of Insulin Promoter Factor 1. J. Clin. Endocrinol. Metab. 2003, 88, 4398–4406. [Google Scholar] [CrossRef] [PubMed]
- Stoffers, D. A.; Zinkin, N. T.; Stanojevic, V.; Clarke, W. L.; Habener, J. F. Pancreatic Agenesis Attributable to a Single Nucleotide Deletion in the Human IPF1 Gene Coding Sequence. Nat. Genet. 1997, 15, 106–110. [Google Scholar] [CrossRef]
- Jeffery, N.; Al Nimri, O.; Houghton, J. A. L.; Globa, E.; Wakeling, M. N.; Flanagan, S. E.; Hattersley, A. T.; Patel, K. A.; De Franco, E. Widening the Phenotypic Spectrum Caused by Pathogenic PDX1 Variants in Individuals with Neonatal Diabetes. BMJ Open Diabetes Res. Care 2024, 12, e004439. [Google Scholar] [CrossRef]
- De Franco, E.; Shaw-Smith, C.; Flanagan, S. E.; Edghill, E. L.; Wolf, J.; Otte, V.; Ebinger, F.; Varthakavi, P.; Vasanthi, T.; Edvardsson, S.; et al. Biallelic PDX1 (Insulin Promoter Factor 1) Mutations Causing Neonatal Diabetes without Exocrine Pancreatic Insufficiency. Diabetic Medicine 2013, 30. [Google Scholar] [CrossRef]
- Nicolino, M.; Claiborn, K. C.; Senée, V.; Boland, A.; Stoffers, D. A.; Julier, C. A Novel Hypomorphic PDX1 Mutation Responsible for Permanent Neonatal Diabetes With Subclinical Exocrine Deficiency. Diabetes 2010, 59, 733–740. [Google Scholar] [CrossRef]
- Sahebi, L.; Niknafs, N.; Dalili, H.; Amini, E.; Esmaeilnia, T.; Amoli, M.; Farrokhzad, N. Iranian Neonatal Diabetes Mellitus Due to Mutation in PDX1 Gene: A Case Report. J. Med. Case Rep. 2019, 13, 258. [Google Scholar] [CrossRef]
- Demirbilek, H.; Cayir, A.; Flanagan, S. E.; Yıldırım, R.; Kor, Y.; Gurbuz, F.; Haliloğlu, B.; Yıldız, M.; Baran, R. T.; Akbas, E. D.; et al. Clinical Characteristics and Long-Term Follow-up of Patients with Diabetes Due To PTF1A Enhancer Mutations. J. Clin. Endocrinol. Metab. 2020, 105, e4351–e4359. [Google Scholar] [CrossRef]
- Sellick, G. S.; Barker, K. T.; Stolte-Dijkstra, I.; Fleischmann, C.; J Coleman, R.; Garrett, C.; Gloyn, A. L.; Edghill, E. L.; Hattersley, A. T.; Wellauer, P. K.; et al. Mutations in PTF1A Cause Pancreatic and Cerebellar Agenesis. Nat. Genet. 2004, 36, 1301–1305. [Google Scholar] [CrossRef] [PubMed]
- Tutak, E.; Satar, M.; Yapicioğlu, H.; Altintaş, A.; Narli, N.; Hergüner, O.; Bayram, Y. A Turkish Newborn Infant with Cerebellar Agenesis/Neonatal Diabetes Mellitus and PTF1A Mutation. Genet. Couns. 2009, 20, 147–152. [Google Scholar] [PubMed]
- Al-Shammari, M.; Al-Husain, M.; Al-Kharfy, T.; Alkuraya, F. A Novel PTF1A Mutation in a Patient with Severe Pancreatic and Cerebellar Involvement. Clin. Genet. 2011, 80, 196–198. [Google Scholar] [CrossRef] [PubMed]
- Miguel-Escalada, I.; Maestro, M. Á.; Balboa, D.; Elek, A.; Bernal, A.; Bernardo, E.; Grau, V.; García-Hurtado, J.; Sebé-Pedrós, A.; Ferrer, J. Pancreas Agenesis Mutations Disrupt a Lead Enhancer Controlling a Developmental Enhancer Cluster. Dev. Cell 2022, 57, 1922–1936.e9. [Google Scholar] [CrossRef]
- Paksaz, M.; Saneifard, H.; Mirdehghan, A.; Mosallanejad, A.; Shakiba, M.; Saberi, M. A Rare PTF1A Enhancer Mutation Causing Neonatal Diabetes Mellitus with Pancreatic Agenesis: Case Report and Considerations for Genetic Evaluation. Int. J. Endocrinol. Metab. 2025, 23. [Google Scholar] [CrossRef]
- Gonc, E. N.; Ozon, A.; Alikasifoglu, A.; Haliloğlu, M.; Ellard, S.; Shaw-Smith, C.; Kandemir, N. Variable Phenotype of Diabetes Mellitus in Siblings with a Homozygous PTF1A Enhancer Mutation. Horm. Res. Paediatr. 2015, 84, 206–211. [Google Scholar] [CrossRef]
- Gabbay, M.; Ellard, S.; De Franco, E.; Moisés, R. S. Pancreatic Agenesis Due to Compound Heterozygosity for a Novel Enhancer and Truncating Mutation in the PTF1A Gene. J. Clin. Res. Pediatr. Endocrinol. 2017, 9, 274–277. [Google Scholar] [CrossRef]
- Evliyaoğlu, O.; Ercan, O.; Ataloğlu, E.; Zübarioğlu, Ü.; Özcabı, B.; Dağdeviren, A.; Erdoğan, H.; De Franco, E.; Ellard, S. Neonatal Diabetes: Two Cases with Isolated Pancreas Agenesis Due to Homozygous PTF1A Enhancer Mutations and One with Developmental Delay, Epilepsy, and Neonatal Diabetes Syndrome Due to KCNJ11 Mutation. J. Clin. Res. Pediatr. Endocrinol. 2018, 10, 168–174. [Google Scholar] [CrossRef]
- Yue, X.; Luo, Y.; Wang, J.; Huang, D. Monogenic Diabetes with GATA6 Mutations: Characterization of a Novel Family and a Comprehensive Analysis of the GATA6 Clinical and Genetics Traits. Mol. Biotechnol. 2024, 66, 467–474. [Google Scholar] [CrossRef]
- Allen, H. L.; Flanagan, S. E.; Shaw-Smith, C.; De Franco, E.; Akerman, I.; Caswell, R.; Ferrer, J.; Hattersley, A. T.; Ellard, S. GATA6 Haploinsufficiency Causes Pancreatic Agenesis in Humans. Nat. Genet. 2012, 44, 20–22. [Google Scholar] [CrossRef] [PubMed]
- Chao, C. S.; McKnight, K. D.; Cox, K. L.; Chang, A. L.; Kim, S. K.; Feldman, B. J. Novel GATA6 Mutations in Patients with Pancreatic Agenesis and Congenital Heart Malformations. PLoS One 2015, 10, e0118449. [Google Scholar] [CrossRef] [PubMed]
- Kishore, S.; De Franco, E.; Cardenas-Diaz, F. L.; Letourneau-Freiberg, L. R.; Sanyoura, M.; Osorio-Quintero, C.; French, D. L.; Greeley, S. A. W.; Hattersley, A. T.; Gadue, P. A Non-Coding Disease Modifier of Pancreatic Agenesis Identified by Genetic Correction in a Patient-Derived IPSC Line. Cell Stem Cell 2020, 27, 137-146.e6.
- De Franco, E. Neonatal Diabetes Caused by Disrupted Pancreatic and Β-cell Development. Diabetic Medicine 2021, 38. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Wasson, L. K.; AL Willcox, J.; Morton, S. U.; Gorham, J. M.; DeLaughter, D. M.; Neyazi, M.; Schmid, M.; Agarwal, R.; Jang, M. Y.; et al. GATA6 Mutations in HiPSCs Inform Mechanisms for Maldevelopment of the Heart, Pancreas, and Diaphragm. Elife 2020, 9. [Google Scholar] [CrossRef]
- Edghill, E. L.; Bingham, C.; Slingerland, A. S.; Minton, J. A. L.; Noordam, C.; Ellard, S.; Hattersley, A. T. Hepatocyte Nuclear Factor-1 Beta Mutations Cause Neonatal Diabetes and Intrauterine Growth Retardation: Support for a Critical Role of HNF-1β in Human Pancreatic Development. Diabetic Medicine 2006, 23, 1301–1306. [Google Scholar] [CrossRef]
- Yorifuji, T.; Kurokawa, K.; Mamada, M.; Imai, T.; Kawai, M.; Nishi, Y.; Shishido, S.; Hasegawa, Y.; Nakahata, T. Neonatal Diabetes Mellitus and Neonatal Polycystic, Dysplastic Kidneys: Phenotypically Discordant Recurrence of a Mutation in the Hepatocyte Nuclear Factor-1β Gene Due to Germline Mosaicism. J. Clin. Endocrinol. Metab. 2004, 89, 2905–2908. [Google Scholar] [CrossRef]
- Li, Y.; Han, X. The HNF1B Mutations and Deletion Associated with Diabetes and Their Resulting Diabetic Phenotypes: A Systematic Review. Int. J. Diabetes Dev. Ctries. 2025, 45, 24–32. [Google Scholar] [CrossRef]
- Poll, A. V; Pierreux, C. E.; Lokmane, L.; Haumaitre, C.; Achouri, Y.; Jacquemin, P.; Rousseau, G. G.; Cereghini, S.; Lemaigre, F. P. A VHNF1/TCF2-HNF6 Cascade Regulates the Transcription Factor Network That Controls Generation of Pancreatic Precursor Cells. Diabetes 2006, 55(1), 61–69. [Google Scholar] [CrossRef]
- Maestro, M. A.; Boj, S. F.; Luco, R. F.; Pierreux, C. E.; Cabedo, J.; Servitja, J. M.; German, M. S.; Rousseau, G. G.; Lemaigre, F. P.; Ferrer, J. Hnf6 and Tcf2 (MODY5) Are Linked in a Gene Network Operating in a Precursor Cell Domain of the Embryonic Pancreas. Hum. Mol. Genet. 2003, 12, 3307–3314. [Google Scholar] [CrossRef]
- Pezzino, G.; Ruta, R.; Rapini, N.; Chiodo, D. C.; Mucciolo, M.; Tomaselli, L.; Cianfarani, S.; Barbetti, F. A Rare Cause of Transient Neonatal Diabetes Mellitus: Spontaneous HNF1B Splice Variant. Diabetic Medicine 2024, 41. [Google Scholar] [CrossRef]
- Iafusco, F.; Meola, S.; Pecoraro, C.; Mazzaccara, C.; Iafusco, D.; Tinto, N. Prenatal Diagnosis of HNF1b Mutation Allows Recognition of Neonatal Dysglycemia. Acta Diabetol. 2021, 58(3), 393–395. [Google Scholar] [CrossRef]
- Gradwohl, G.; Dierich, A.; LeMeur, M.; Guillemot, F. Neurogenin3 Is Required for the Development of the Four Endocrine Cell Lineages of the Pancreas. Proceedings of the National Academy of Sciences 2000, 97(4), 1607–1611. [Google Scholar] [CrossRef] [PubMed]
- Wejaphikul, K.; Srilanchakon, K.; Kamolvisit, W.; Jantasuwan, S.; Santawong, K.; Tongkobpetch, S.; Theerapanon, T.; Damrongmanee, A.; Hongsawong, N.; Ukarapol, N.; et al. Novel Variants and Phenotypes in NEUROG3 -Associated Syndrome. J. Clin. Endocrinol. Metab. 2022, 108, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Pinney, S. E.; Oliver-Krasinski, J.; Ernst, L.; Hughes, N.; Patel, P.; Stoffers, D. A.; Russo, P.; De León, D. D. Neonatal Diabetes and Congenital Malabsorptive Diarrhea Attributable to a Novel Mutation in the Human Neurogenin-3 Gene Coding Sequence. J. Clin. Endocrinol. Metab. 2011, 96, 1960–1965. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Cabezas, O.; Jensen, J. N.; Hodgson, M. I.; Codner, E.; Ellard, S.; Serup, P.; Hattersley, A. T. Permanent Neonatal Diabetes and Enteric Anendocrinosis Associated With Biallelic Mutations in NEUROG3. Diabetes 2011, 60, 1349–1353. [Google Scholar] [CrossRef]
- Hancili, S.; Bonnefond, A.; Philippe, J.; Vaillant, E.; De Graeve, F.; Sand, O.; Busiah, K.; Robert, J.; Polak, M.; Froguel, P.; et al. A Novel NEUROG3 Mutation in Neonatal Diabetes Associated with a Neuro-intestinal Syndrome. Pediatr. Diabetes 2018, 19, 381–387. [Google Scholar] [CrossRef]
- Zhang, X.; McGrath, P. S.; Salomone, J.; Rahal, M.; McCauley, H. A.; Schweitzer, J.; Kovall, R.; Gebelein, B.; Wells, J. M. A Comprehensive Structure-Function Study of Neurogenin3 Disease-Causing Alleles during Human Pancreas and Intestinal Organoid Development. Dev. Cell 2019, 50, 367–380.e7. [Google Scholar] [CrossRef]
- Solorzano-Vargas, R. S.; Bjerknes, M.; Wang, J.; Wu, S. V.; Garcia-Careaga, M. G.; Pitukcheewanont, P.; Cheng, H.; German, M. S.; Georgia, S.; Martín, M. G. Null Mutations of NEUROG3 Are Associated with Delayed-Onset Diabetes Mellitus. JCI Insight 2020, 5. [Google Scholar] [CrossRef]
- Rubio-Cabezas, O.; Gómez, J. L.; Gleisner, A.; Hattersley, A. T.; Codner, E. Hypogonadotropic Hypogonadism and Short Stature in Patients with Diabetes Due to Neurogenin 3 Deficiency. J. Clin. Endocrinol. Metab. 2016, 101, 3555–3558. [Google Scholar] [CrossRef]
- Bohuslavova, R.; Smolik, O.; Malfatti, J.; Berkova, Z.; Novakova, Z.; Saudek, F.; Pavlinkova, G. NEUROD1 Is Required for the Early α and β Endocrine Differentiation in the Pancreas. Int. J. Mol. Sci. 2021, 22, 6713. [Google Scholar] [CrossRef]
- Bohuslavova, R.; Fabriciova, V.; Smolik, O.; Lebrón-Mora, L.; Abaffy, P.; Benesova, S.; Zucha, D.; Valihrach, L.; Berkova, Z.; Saudek, F.; et al. NEUROD1 Reinforces Endocrine Cell Fate Acquisition in Pancreatic Development. Nat. Commun. 2023, 14(1), 5554. [Google Scholar] [CrossRef]
- Demirbilek, H.; Hatipoglu, N.; Gul, U.; Tatli, Z. U.; Ellard, S.; Flanagan, S. E.; De Franco, E.; Kurtoglu, S. Permanent Neonatal Diabetes Mellitus and Neurological Abnormalities Due to a Novel Homozygous Missense Mutation in NEUROD1. Pediatr. Diabetes 2018, 19, 898–904. [Google Scholar] [CrossRef] [PubMed]
- Lucas, A.; Morley, R.; Cole, T. J. Adverse Neurodevelopmental Outcome of Moderate Neonatal Hypoglycaemia. BMJ 1988, 297, 1304–1308. [Google Scholar] [CrossRef] [PubMed]
- Jermendy, A.; Toschi, E.; Aye, T.; Koh, A.; Aguayo-Mazzucato, C.; Sharma, A.; Weir, G. C.; Sgroi, D.; Bonner-Weir, S. Rat Neonatal Beta Cells Lack the Specialised Metabolic Phenotype of Mature Beta Cells. Diabetologia 2011, 54, 594–604. [Google Scholar] [CrossRef] [PubMed]
- Richardson, C. C.; Hussain, K.; Jones, P. M.; Persaud, S.; Löbner, K.; Boehm, A.; Clark, A.; Christie, M. R. Low Levels of Glucose Transporters and Kþ ATP Channels in Human Pancreatic Beta Cells Early in Development. Diabetologia 2007, 50, 1000–1005. [Google Scholar] [CrossRef]
- Parappil, H.; Gaffari, M.; Paramban, R.; Rijims, M.; Skaria, S.; Ahmed, S. N. Management of Hyperglycemia in the Neonatal Unit. J. Clin. Neonatol. 2022, 11, 38–44. [Google Scholar] [CrossRef]
- Mitanchez, D. Glucose Regulation in Preterm Newborn Infants. Horm. Res. Paediatr. 2007, 68, 265–271. [Google Scholar] [CrossRef]
- Hey, E. Hyperglycaemia and the Very Preterm Baby. Semin. Fetal Neonatal Med. 2005, 10, 377–387. [Google Scholar] [CrossRef]
- Decaro, M. H.; Vain, N. E. Hyperglycaemia in Preterm Neonates: What to Know, What to Do. Early Hum. Dev. 2011, 87, S19–S22. [Google Scholar] [CrossRef]
- Mitanchez-Mokhtari, D.; Lahlou, N.; Kieffer, F.; Magny, J.-F.; Roger, M.; Voyer, M. Both Relative Insulin Resistance and Defective Islet β-Cell Processing of Proinsulin Are Responsible for Transient Hyperglycemia in Extremely Preterm Infants. Pediatrics 2004, 113, 537–541. [Google Scholar] [CrossRef]
- Chacko, S. K.; Ordonez, J.; Sauer, P. J. J.; Sunehag, A. L. Gluconeogenesis Is Not Regulated by Either Glucose or Insulin in Extremely Low Birth Weight Infants Receiving Total Parenteral Nutrition. J. Pediatr. 2011, 158, 891–896. [Google Scholar] [CrossRef]
- Cowett, R. M.; Andersen, G. E.; Maguire, C. A.; Oh, W. Ontogeny of Glucose Homeostasis in Low Birth Weight Infants. J. Pediatr. 1988, 112, 462–465. [Google Scholar] [CrossRef] [PubMed]
- Boehmer, B. H.; Limesand, S. W.; Rozance, P. J. The Impact of IUGR on Pancreatic Islet Development and β-Cell Function. Journal of Endocrinology 2017, 235, R63–R76. [Google Scholar] [CrossRef] [PubMed]
- Van Assche, F. A.; De Prins, F.; Aerts, L.; Verjans, M. The endocrine pancreas in small-for-dates infants. Bjog 1977, 84, 751–753. [Google Scholar] [CrossRef] [PubMed]
- Green, A. S.; Rozance, P. J.; Limesand, S. W. Consequences of a Compromised Intrauterine Environment on Islet Function. Journal of Endocrinology 2010, 205, 211–224. [Google Scholar] [CrossRef]
- Limesand, S. W.; Rozance, P. J.; Zerbe, G. O.; Hutton, J. C.; Hay, W. W. Attenuated Insulin Release and Storage in Fetal Sheep Pancreatic Islets with Intrauterine Growth Restriction. Endocrinology 2006, 147, 1488–1497. [Google Scholar] [CrossRef]
- Thorn, S.; Rozance, P.; Brown, L.; Hay, W. The Intrauterine Growth Restriction Phenotype: Fetal Adaptations and Potential Implications for Later Life Insulin Resistance and Diabetes. Semin. Reprod. Med. 2011, 29, 225–236. [Google Scholar] [CrossRef]
- Yates, D. T.; Macko, A. R.; Chen, X.; Green, A. S.; Kelly, A. C.; Anderson, M. J.; Fowden, A. L.; Limesand, S. W. Hypoxaemia-induced Catecholamine Secretion from Adrenal Chromaffin Cells Inhibits Glucose-stimulated Hyperinsulinaemia in Fetal Sheep. J. Physiol. 2012, 590, 5439–5447. [Google Scholar] [CrossRef]
- Park, J. H.; Stoffers, D. A.; Nicholls, R. D.; Simmons, R. A. Development of Type 2 Diabetes Following Intrauterine Growth Retardation in Rats Is Associated with Progressive Epigenetic Silencing of Pdx1. Journal of Clinical Investigation 2008. [Google Scholar] [CrossRef]
- Stoffers, D. A.; Desai, B. M.; DeLeon, D. D.; Simmons, R. A. Neonatal Exendin-4 Prevents the Development of Diabetes in the Intrauterine Growth Retarded Rat. Diabetes 2003, 52, 734–740. [Google Scholar] [CrossRef]
- Wang, C.; Jiang, H.; Wu, J.; Yu, Z.; Li, Q.; Jiang, C.-M. Association between Glycemia and Outcomes of Neonates with Hypoxic-Ischemic Encephalopathy: A Systematic Review and Meta-Analysis. BMC Pediatr. 2024, 24, 699. [Google Scholar] [CrossRef]
- Pedroza-García, K. A.; Calderón-Vallejo, D.; Quintanar, J. L. Neonatal Hypoxic–Ischemic Encephalopathy: Perspectives of Neuroprotective and Neuroregenerative Treatments. Neuropediatrics 2022, 53, 402–417. [Google Scholar] [CrossRef]
- Šumanović-Glamuzina, D.; Čulo, F.; Čulo, M. I.; Konjevoda, P.; Jerković-Raguž, M. A Comparison of Blood and Cerebrospinal Fluid Cytokines (IL-1β, IL-6, IL-18, TNF-α) in Neonates with Perinatal Hypoxia. Bosn. J. Basic Med. Sci. 2017. [Google Scholar] [CrossRef]
- Sheng, J. A.; Bales, N. J.; Myers, S. A.; Bautista, A. I.; Roueinfar, M.; Hale, T. M.; Handa, R. J. The Hypothalamic-Pituitary-Adrenal Axis: Development, Programming Actions of Hormones, and Maternal-Fetal Interactions. Front. Behav. Neurosci. 2021, 14. [Google Scholar] [CrossRef]
| Author | Population | Genetic etiology | Median age of diagnosis, days | Median age of remission, weeks | SGA | Congenital anomalies/disorders |
|---|---|---|---|---|---|---|
| Busiah, 2013[30] | 40 | 45% UPD6pat 33% 6q24 duplication 23% maternal hypomethylation |
5.0 (1.0–14.5) | 14.4 (7.8–28.3) | 92% | Macroglossia 32% Cardiac 8% Urinary tract 17% Neurological disorders 18% Umbilical hernia 8% |
| Docherty, 2013 [23] | 163 | 41% UPD6pat 33% paternal 6q24 duplication 26% maternal hypomethylation |
4.0 | 12.0 | ND | Macroglossia 44% Umbilical hernia 21% Dysmorphic facial features 18% Urinary tract 9% Cardiac 9% Hypothyroidism 4% |
| Bonfanti, 2021 [32] | 12 | 50% UPD6pat 17% paternal 6q24 duplication 33% maternal hypomethylation |
7.0 | 12.0 | ND | Macroglossia/Umbilical hernia 33% |
| McCullough, 2024 [31] | 33 | 52% UPD6pat 21% paternal 6q24 duplication 24% maternal hypomethylation |
2.0 (1.0–7.0) | 12.0 (8.0-24.0) | 71% | Macroglossia 56% Umbilical hernia 22% Speech pathologies 36% |
| Pietrusinski, 2025 [33] | 5 | 60% UPD6pat/maternal hypomethylation | 4.4 | 16.0 | 80% | Macroglossia 0% Umbilical hernia 40% Neurodevelopmental disorders 40% |
| Gene | Location | Mode of inheritance | Gene product | Type of NDM | Associated malformations |
|---|---|---|---|---|---|
| KCNJ11 | 11p15.1 | de novo/ AD | Kir6.2 | PNDM (more often)/TNDM | Neurodevelopmental impairment, DEND syndrome |
| ABCC8 | 11p15.1 | de novo/ AD | SUR1 | PNDM/TNDM (more often) | Neurodevelopmental impairment, DEND syndrome (very rare) |
| GCK | 7p15.3-p15.1 | AR | GCK | PNDM | Not present |
| SLC2A2 | 3q26.1-26.3 | AR | GLUT2 | PNDM/TNDM | Fanconi-Bickel syndrome (renal Fanconi syndrome, rickets, growth retardation, hepatomegaly, impaired galactose metabolism, diabetes) |
| SLC19A | 1q23.3 | AR | THTR1 | PNDM | Rogers syndrome (Thiamine-responsive megaloblastic anemia, sensorineural deafness, diabetes) |
| Gene | Location | Mode of inheritance | Gene product | Type of NDM | Associated malformations |
|---|---|---|---|---|---|
| INS | 11p15.5 | de novo, AD (more often)/AR | Insulin | PNDM (more often)/TNDM | Not present |
| EIF2AK3 | 2p11.2 | AR | PERK | PNDM | Wolcott-Rallison syndrome (neurodevelopmental disorders, skeletal dysplasias, renal disorders, liver impairment, diabetes) |
| EIF2B1 | 12q24.31 | de novo, AD | eEIF2Ba | PNDM | Liver impairment |
| IER3IP1 | 18q12 | AR | IER3IP1 | PNDM | MEDS syndrome (diabetes, microcephaly, epilepsy) |
| WFS1 | 4p16.1 | AR/ de novo, AD | wolframin | PNDM | Homozygous: Wolfram syndrome (diabetes mellitus, diabetes insipidus, optic atrophy, deafness) Heterozygous: diabetes, congenital sensorineural deafness, congenital cataract |
| Gene | Location | Mode of inheritance | Gene product | Type of NDM | Associated malformations |
|---|---|---|---|---|---|
| PDX1 | 13q12.1 | AD | PDX1 | PNDM | Nonsense mutations: pancreatic agenesis/hypoplasia Hypomorphic mutations: +/- exocrine pancreatic insufficiency |
| PTF1A | 10p12.2 | AR/AD | PTF1A | PNDM | Homozygous: pancreatic agenesis, cerebellar hypoplasia/agenesis Heterozygous: pancreatic agenesis Growth retardation, anemia, cholestasis |
| HNF1B | 17q12 | AD | HNF1B | PNDM/TNDM | Pancreatic hypoplasia, renal cysts |
| GATA6 | 18q11.2 | AD | GATA6 | PNDM | Pancreatic agenesis, cardiac, hepatobiliary, intestinal malformations |
| GATA4 | 8p23.1 | AD | GATA4 | PNDM | Pancreatic agenesis, congenital heart defects |
| CNOT1 | 16q21 | de novo, AD | CNOT1 | PNDM | Pancreatic agenesis, holoprosencephaly |
| RFX6 | 6q22.1 | AR | RFX6 | PNDM | Mitchell–Riley syndrome (diabetes, bowel atresia, gallbladder agenesis/hypoplasia) |
| NEUROG3 | 10q21.3 | AR | NEUROG3 | PNDM | Malabsorptive congenital diarrhea (enteric anendocrinosis), hypogonadotrophic hypogonadism, short stature |
| NEUROD1 | 2q31.3 | AR | NEUROD1 | PNDM | Cerebellar hypoplasia, sensorineural deafness, visual impairments |
| NKX2-2 | 20p11.22 | AR | NKX2-2 | PNDM | Neurodevelopmental impairment, corpus callosum hypoplasia |
| GLIS3 | 9p24.2 | AR | GLIS3 | PNDM | congenital hypothyroidism, polycystic kidneys, hepatomegaly, cholestasis, characteristic facial features |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





