Submitted:
10 February 2026
Posted:
10 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Study Identification and Search Strategy
2.2. Eligibility Criteria
2.3. Study Selection
2.4. Data Extraction
2.5. Summary of Measures and Synthesis of Results
2.6. Risk of Bias Assessment
2.7. Level of Evidence and Recommendation
3. Results
3.1. Study Selection
3.2. Study Characteristics
| Primary Author (Year) | Participants | Study Design | Methods | Primary Outcomes: | Primary Results: | |||
| N = |
Male: Female | Age (years) | Other Characteristics | |||||
| Lee [32] | 11 | 0:11 | 25.7 ± 4.2 | Professional female soccer athletes | RCT | RT + HC w/VC | ∙ PT Mechanical properties ∙ PT Material properties |
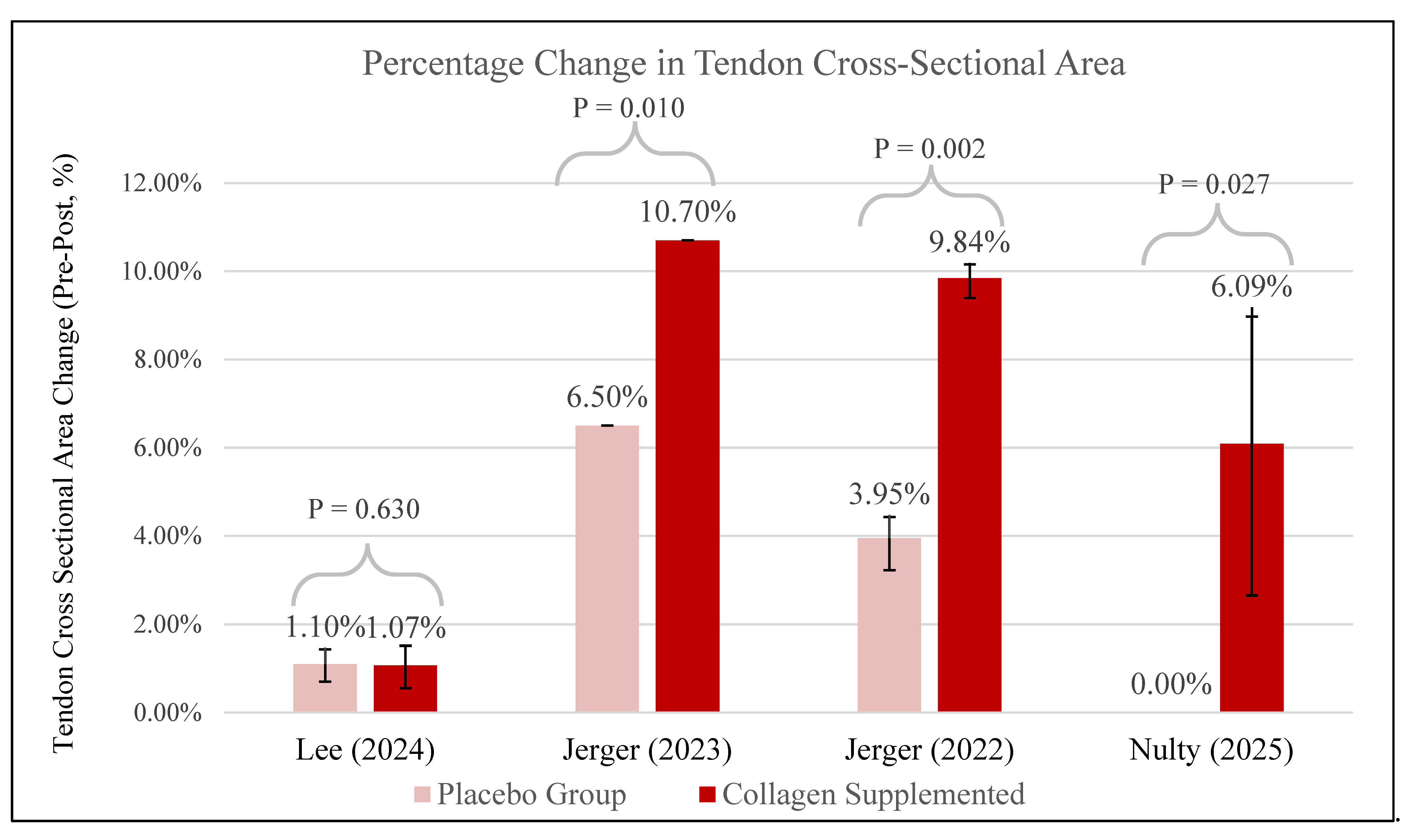
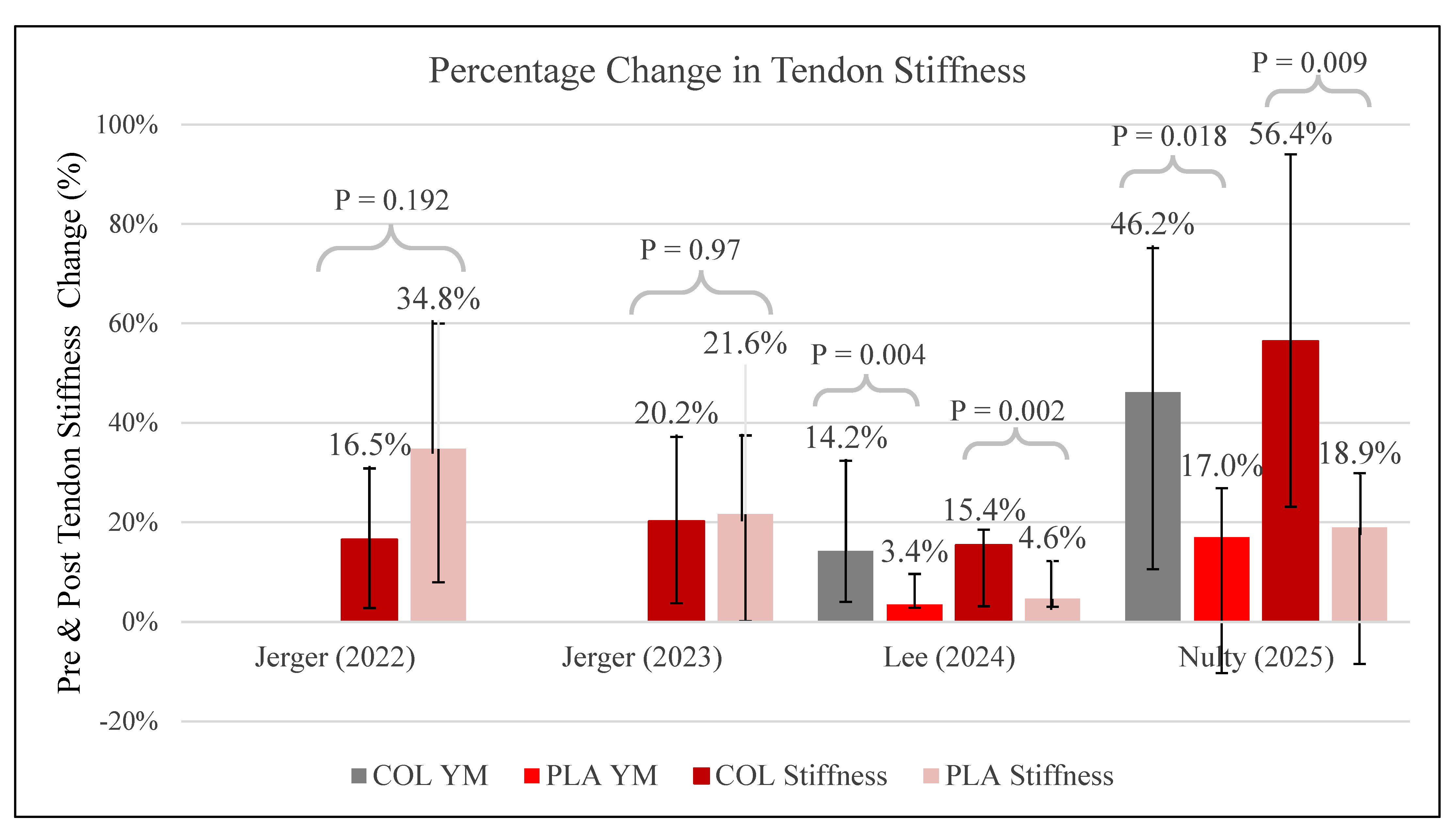
∙ ↑ Stiffness and YM ∙ X No effect on PT CSA |
| Nunez-Lisboa [43] | 9 | 9:0 | 32.5 ± 4.1 | Male Triathletes | RCT | PS + HC | ∙ Kvert ∙ Spatiotemporal Parameters |
∙ X No effect on Kvert or spatiotemporal parameters |
| Kirmse [26] | 57 | 57:0 | 24.0 ± 3 | Moderate-Trained Males | RCT | RT + CP | ∙ Body Composition, ∙ Strength ∙ Muscle fCSA |
∙ ∙ X No effect on Muscle strength or fCSA |
| Jerger [23] | 31 | 31:0 | 28.6 ± 5.1 | Healthy males with low/moderate physical activity (<120 min per week) | RCT | RT + CP | ∙ PT CSA & stiffness ∙ Maximal knee extension strength |
∙ ∙ ↑ PT CSA ∙ X No effect on PT Stiffness, knee extension strength |
| Balshaw [6] | 39 | 39:0 | CP: 27.0 ± 5.0 PLA: 24.4 ± 3.2 | Healthy males with low/moderate level of recreational physical activity | RCT | RT + CP | ∙ Functional, Structural, and contractile adaptations of skeletal muscle | ∙ ∙ ↑ Muscle volume, twitch peak torque, architectural remodeling ∙ X No effect on muscle strength |
| Lis [35] | 50 | 50:0 | 18–25 | Healthy male athletes participating in football, rugby or ROTC | RCT | PWT + HC w/VC | ∙ RFD | ∙ ∙ ↑ change in CM jumps eccentric deceleration impulse, eccentric deceleration RFD, recovery of RTD ∙ X No effect on maximal isometric squat force |
| Jerger [24] | 40 | 40:0 | 26.3 ± 4.0 | Healthy males with no recent history (>12 months) of RT or not involved in any systematic training (>60 min per week) | RCT | RT + CP | ∙ AT CSA ∙ AT stiffness ∙ Muscular strength ∙ Muscle thickness |
∙ ∙ ↑ Tendon CSA, Muscle thickness ∙ X No effect on tendon stiffness, Muscle Strength |
| Nulty [41] | 20 | 20:0 | 47 ± 5 | Healthy males with > 120 min moderate activity and/or sport training per week but naïve to LE resistance exercise |
RCT | RT + HC w/VC | ∙ PT CSA ∙ PT Stiffness ∙ Muscle Strength ∙ Muscle thickness ∙ pRTD |
∙ ∙ ↑ PT CSA, Youngs’ modulus, stiffness ∙ X No effect on strength, pRTD, and muscle thickness |
3.3. Risk of Bias Assessment
| PEDro Criteria | Lee [32] | Nunez-Lisboa [43] | Kirmse [26] | Jerger [23] | Balshaw [6] | Lis [35] | Jerger [24] | Nulty [41] |
| 1 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| 2 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| 3 | × | × | ✓ | ✓ | ✓ | × | × | ✓ |
| 4 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| 5 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| 6 | × | × | × | × | × | × | × | × |
| 7 | ✓ | × | × | × | × | × | × | ✓ |
| 8 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| 9 | × | × | × | × | × | × | × | × |
| 10 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| 11 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Total | 8 | 7 | 8 | 8 | 8 | 7 | 7 | 9 |
| Interpretation | Good | Good | Good | Good | Good | Good | Good | Excellent |
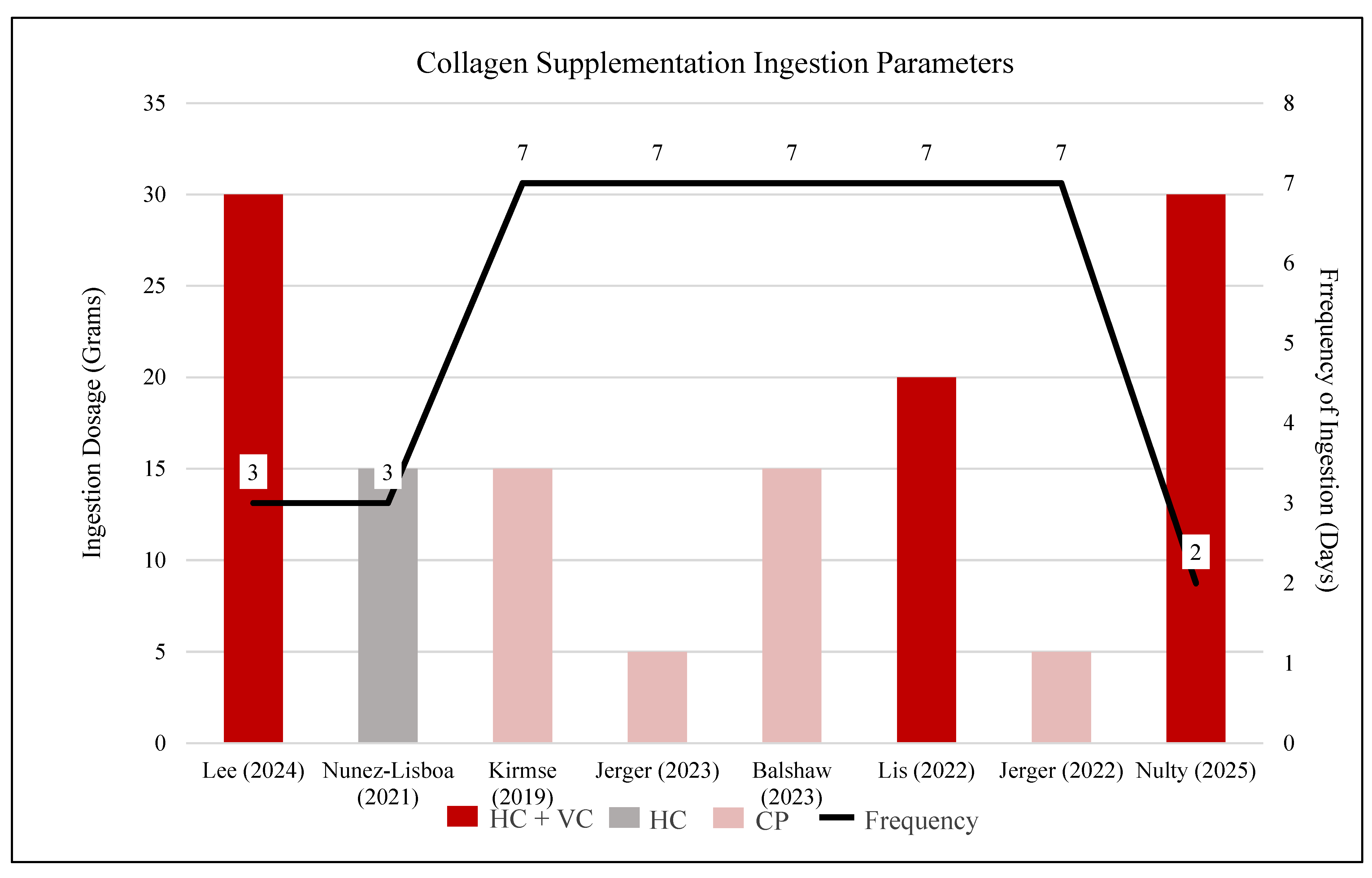
3.4. Collagen Ingestion
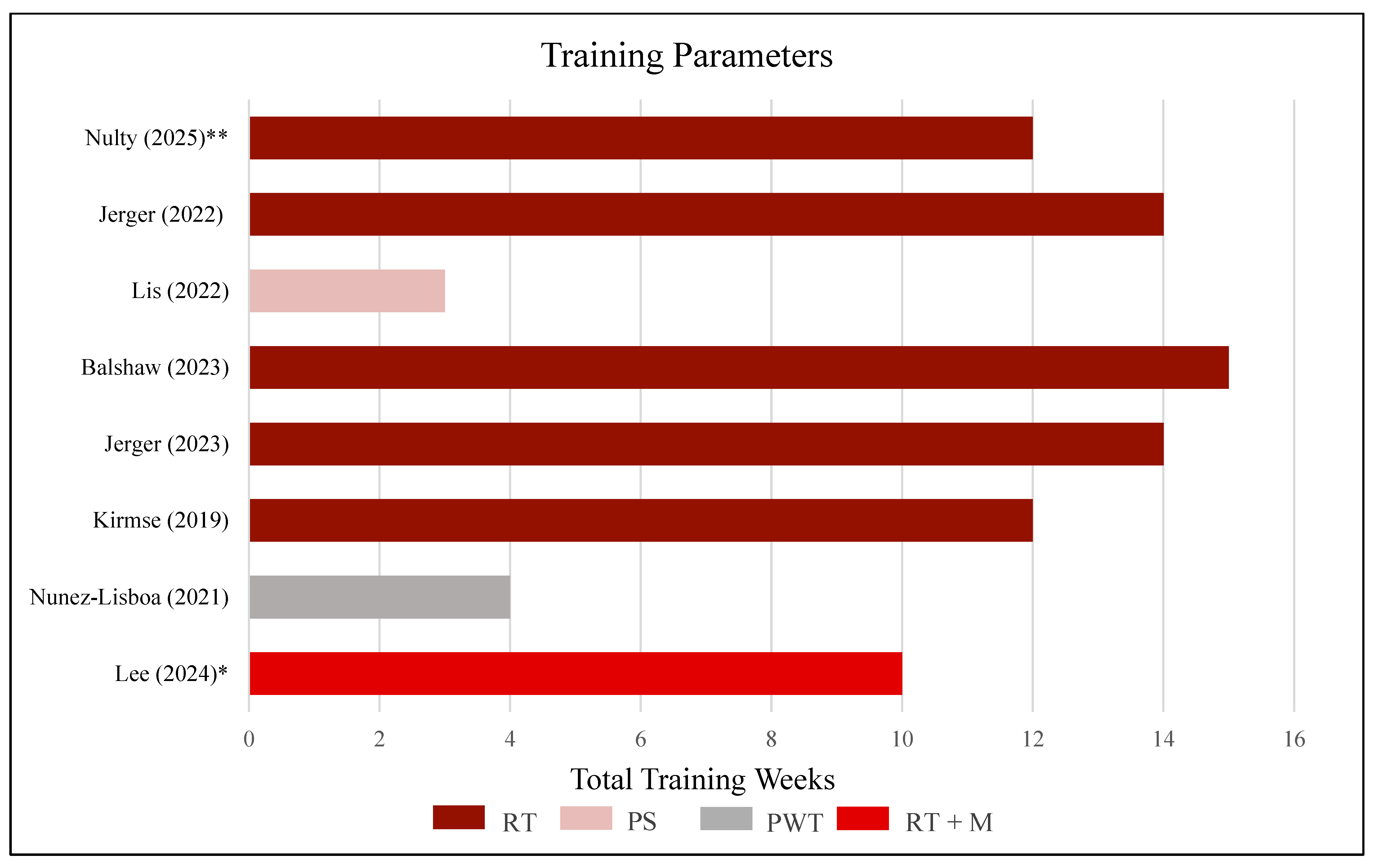
3.5. Training Protocol
3.6. Tendon Cross-Sectional Area (CSA)
3.7. Tendon Stiffness
3.8. Physical Performance:
3.9. Muscle Volume/Cross Sectional Area
3.10. Muscle Strength
| Author (Year) | Muscle Strength Tests | Group × Time Interaction | Between-Group Interpretation |
| Kirmse [26] | SL Ext. MViC, SQ 1RM, DL 1RM, BP 1RM, BOR 1RM | No sig. (p = 0.477–0.768), SQ trend p = 0.054 | No between-group difference |
| Jerger [24] | PF MVT | No sig. (p = 0.629) | No between-group difference |
| Balshaw [6] |
KE MViC, KE 1RM, KF MViC, Absolute Torque, Torque expressed relative to MVT (torque at 50ms intervals) | No sig. (p = 0.703–0.929 for %Δ; p = 0.054–0.862 for absolute) | No between-group difference |
| Lis [35] | Maximal Isometric SQ | No sig. (p = 0.32) | No between-group difference |
| Jerger [23] | LP 1RM, KE 1RM | No sig. (p = 0.396, 0.805) | No between-group difference |
| Nulty [41] | KE MViC, LP 10RM | No sig. (p > 0.05) | No between-group difference |
3.11. GRADE of Recommendations
4. Discussion
4.1. Limitations
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A



Appendix B
| Primary Author, Year | Intervention Prescription Variables | |||||
|
Duration: Weeks |
Training Frequency: sessions per/ week | Training Type: | Ingestion Type: | Ingestion Frequency: | Ingestion Timing: | |
| Lee [32] | 10 | 4 x RT + 1 x match |
∙ EL UE RT ∙ EL LE RT ∙ EL LE plyometric exercises ∙ Pitch-Based Sessions |
30g HC + 500mg VC | 3x per week: on days with training sessions |
Before each training session. |
| Nunez-Lisboa [43] | 4 | 3 x PS |
∙ Intervention Specific: ∙ PS: 4 × 100-meter sprints post-supplementation ∙ General Triathlon Training: ∙ Collagen Group:16.6 ± 2.1 h/week ∙ Control Group: 17.3 ± 1.4 h/week |
15g HC (Great lakes gelatin,12g of collagen hydrolysate, 36 mg of sodium) |
3x per week: on Plyometric Stimulus Days |
60 min before each training session |
| Kirmse [26] | 12 | 3 x RT |
∙ Whole-body RT: ∙ Squats, Bench press, Deadlift, Bent-over-Row, Knee extension ∙ Each exercise: 1 set of 10 at 50% 1RM followed by 3 sets of 10 repetitions with 70% (weeks 4-12) 1RM. Rest: 2 min |
15g CP (Bodybalance provided by Gelita AG) | Daily Ingestion |
Training Days: Immediately after training Non-Training Days: 24-hours after previous ingestion |
| Jerger [23] | 14 | 3 x RT |
∙ Whole-Body RT: ∙ Leg Press, Knee Extension, Calf Raise (Load progressed every four weeks from 70% to 85% 1RM). ∙ Latissimus Pull + Bench Press added to increase compliance, performed after LE protocol |
5g CP (Tendoforte, provided by Gelita AG) |
Daily Ingestion |
Training Days: Half ingested 30 min before training, half immediately after training Non-Training Days: 24-hours after previous ingestion |
| Balshaw [6] | 15 | 3 x RT |
∙ LE RT: ∙ Unilateral Knee Extension, Bilateral Knee Flexion, Leg Press. ∙ 2–4 sets per exercise (4 sets for each exercise by week 7) with undulating periodization between ~12RM and ~6RM |
15g CP (10g of BodyBalance and 5g of Tendoforte, Gelita AG) |
Daily Ingestion |
Training Days: Immediately after training Non-Training Days: Mid-Afternoon |
| Lis [35] | 3 | 3 x PWT |
∙ PWT: ∙ Progressive loading including vertical drop jumps, vertical box jumps, and body weight loaded ballistic squats. ∙ Rugby and ROTC participants: ∙ First 3 days: 2 x 10 reps, progressed to 3 x 10, reached 5 x 10 reps in final week ∙ Football players: ∙ Progressive PWT program: ballistic squats, plyometrics, and speed work, without the same progression |
20g HC + 50 mg VC |
Daily Ingestion |
Training Days: ∼60 min before training Non-Training Days: Ingestion with breakfast |
| Jerger [24] | 14 | 3 x LE RT |
∙ LE RT: ∙ Seated and standing calf ∙ Progressively increasing load from 70% to 85% 1RM over 14 weeks, 3 sets per exercise with reps decreasing from 12 to 6 |
5g CP (Tendoforte, provided by Gelita AG) |
Daily Ingestion | Training Days: within one hour after training Non-Training days: 24-hours after previous ingestion |
| Nulty [41] | 12 | 2 x LE RT |
∙ LE RT: ∙ Barbell back squats, dumbbell Romanian deadlifts, trap-bar deadlifts and dumbbell goblet squats. ∙ Starting: 4 sets of 8–10 reps at 90% 10-RM for 6 weeks, then progressed to 6–8 reps for the remaining 6 weeks, loads increased weekly based on performance |
30g HC (Collagen Protein, MyProtein) + 50 mg VC |
2x per week: on training days | Immediately post-training (5 minutes) |
References
- Alcock, R. D., Shaw, G. C., Tee, N., & Burke, L. M. (2019). Plasma Amino Acid Concentrations After the Ingestion of Dairy and Collagen Proteins, in Healthy Active Males. Frontiers in Nutrition, 6. [CrossRef]
- American Physical Therapy Association. (2014). Clinical practice guidelines (CPGs): Getting started. http://www.apta.org/EvidenceResearch/ImplementingEBP/CPGs/GettingStarted/.
- Arampatzis, A., Karamanidis, K., & Albracht, K. (2007). Adaptational responses of the human Achilles tendon by modulation of the applied cyclic strain magnitude. Journal of Experimental Biology, 210(15), 2743–2753. [CrossRef]
- Armijo-Olivo, S., Fuentes, J., Da Costa, B. R., Saltaji, H., Ha, C., & Cummings, G. G. (2017). Blinding in Physical Therapy Trials and Its Association with Treatment Effects: A Meta-epidemiological Study. American Journal of Physical Medicine & Rehabilitation, 96(1), 34–44. [CrossRef]
- Arya, S., & Kulig, K. (2010). Tendinopathy alters mechanical and material properties of the Achilles tendon. Journal of Applied Physiology, 108(3), 670–675. [CrossRef]
- Balshaw, T. G., Funnell, M. P., McDermott, E., Maden-Wilkinson, T. M., Abela, S., Quteishat, B., Edsey, M., James, L. J., & Folland, J. P. (2023). The effect of specific bioactive collagen peptides on function and muscle remodeling during human resistance training. Acta Physiologica 2023, 237(2), e13903. Correction in Acta Physiol (Oxf). 2023, 237(4), e13952. [CrossRef]
- Bobbert, M. F., & Casius, L. J. R. (2005). Is the Effect of a Countermovement on Jump Height due to Active State Development? Medicine & Science in Sports & Exercise, 37(3), 440–446. [CrossRef]
- Bohm, S., Mersmann, F., & Arampatzis, A. (2015). Human tendon adaptation in response to mechanical loading: A systematic review and meta-analysis of exercise intervention studies on healthy adults. Sports Medicine - Open, 1(1), 7. [CrossRef]
- Brueckheimer, P. J., Costa Silva, T., Rodrigues, L., Zague, V., & Isaia Filho, C. (2025). The Effects of Type I Collagen Hydrolysate Supplementation on Bones, Muscles, and Joints: A Systematic Review. Orthopedic Reviews, 17. [CrossRef]
- Cashin, A. G., & McAuley, J. H. (2020). Clinimetrics: Physiotherapy Evidence Database (PEDro) Scale. Journal of Physiotherapy, 66(1), 59. [CrossRef]
- Cook, J. L., & Purdam, C. R. (2009). Is tendon pathology a continuum? A pathology model to explain the clinical presentation of load-induced tendinopathy. British Journal of Sports Medicine, 43(6), 409–416. [CrossRef]
- Cormie, P., Mcguigan, M. R., & Newton, R. U. (2010). Adaptations in Athletic Performance after Ballistic Power versus Strength Training. Medicine & Science in Sports & Exercise, 42(8), 1582–1598. [CrossRef]
- Cormie, P., McGUIGAN, M. R., & Newton, R. U. (2010). Changes in the Eccentric Phase Contribute to Improved Stretch-Shorten Cycle Performance after Training. Medicine & Science in Sports & Exercise, 42(9), 1731–1744. [CrossRef]
- Folland, J. P., Buckthorpe, M. W., & Hannah, R. (2014). Human Capacity for Explosive Force Production: Neural and Contractile Determinants. Scandinavian Journal of Medicine & Science in Sports. [CrossRef]
- Gallagher Ford, L., & Melnyk, B. M. (2019). The Underappreciated and Misunderstood PICOT Question: A Critical Step in the EBP Process. Worldviews on Evidence-Based Nursing, 16(6), 422–423. [CrossRef]
- Galloway, M. T., Lalley, A. L., & Shearn, J. T. (2013). The Role of Mechanical Loading in Tendon Development, Maintenance, Injury, and Repair. Journal of Bone and Joint Surgery, 95(17), 1620–1628. [CrossRef]
- GRADE Working Group. (2005). Introduction: Grading the quality of evidence and the strength of recommendations. http://www.gradeworkinggroup.org/intro.htm.
- Heinemeier, K. M., & Kjaer, M. (2011). In vivo investigation of tendon responses to mechanical loading. Journal of Musculoskeletal & Neuronal Interactions, 11(2), 115–123.
- Heinemeier, K. M., Olesen, J. L., Haddad, F., Langberg, H., Kjaer, M., Baldwin, K. M., & Schjerling, P. (2007). Expression of collagen and related growth factors in rat tendon and skeletal muscle in response to specific contraction types. The Journal of Physiology, 582(3), 1303–1316. [CrossRef]
- Holwerda, A. M., & Van Loon, L. J. C. (2022). The impact of collagen protein ingestion on musculoskeletal connective tissue remodeling: A narrative review. Nutrition Reviews, 80(6), 1497–1514. [CrossRef]
- Jacinto, J. L., Nunes, J. P., Gorissen, S. H. M., Capel, D. M. G., Bernardes, A. G., Ribeiro, A. S., Cyrino, E. S., Phillips, S. M., & Aguiar, A. F. (2022). Whey Protein Supplementation Is Superior to Leucine-Matched Collagen Peptides to Increase Muscle Thickness During a 10-Week Resistance Training Program in Untrained Young Adults. International Journal of Sport Nutrition and Exercise Metabolism, 32(3), 133–143. [CrossRef]
- Jeanfavre, M., Husted, S., & Leff, G. (2018). Exercise therapy in the non-operative treatment of full-thickness rotator cuff tears: A systematic review. International Journal of Sports Physical Therapy, 13(3), 335–378. [CrossRef]
- Jerger, S., Centner, C., Lauber, B., Seynnes, O., Lolli, D., Gollhofer, A., & König, D. (2023). Specific collagen peptides increase adaptions of patellar tendon morphology following 14-weeks of high-load resistance training: A randomized- controlled trial.
- Jerger, S., Centner, C., Lauber, B., Seynnes, O., Sohnius, T., Jendricke, P., Oesser, S., Gollhofer, A., & König, D. (2022). Effects of specific collagen peptide supplementation combined with resistance training on Achilles tendon properties. Scandinavian Journal of Medicine & Science in Sports, 32(7), 1131–1141. [CrossRef]
- Khatri, M., Naughton, R. J., Clifford, T., Harper, L. D., & Corr, L. (2021). The effects of collagen peptide supplementation on body composition, collagen synthesis, and recovery from joint injury and exercise: A systematic review. Amino Acids, 53(10), 1493–1506. [CrossRef]
- Kirmse, M., Oertzen-Hagemann, V., De Marées, M., Bloch, W., & Platen, P. (2019). Prolonged Collagen Peptide Supplementation and Resistance Exercise Training Affects Body Composition in Recreationally Active Men. Nutrients, 11(5), 1154. [CrossRef]
- Kongsgaard, M., Reitelseder, S., Pedersen, T. G., Holm, L., Aagaard, P., Kjaer, M., & Magnusson, S. P. (2007). Region specific patellar tendon hypertrophy in humans following resistance training. Acta Physiologica, 191(2), 111–121. [CrossRef]
- Kubo, K., & Ikebukuro, T. (2019). Changes in joint, muscle, and tendon stiffness following repeated hopping exercise. Physiological Reports, 7(19). [CrossRef]
- Kubo, K., Kanehisa, H., Ito, M., & Fukunaga, T. (2001). Effects of isometric training on the elasticity of human tendon structures in vivo.
- Lavagnino, M., & Arnoczky, S. P. (2005). In vitro alterations in cytoskeletal tensional homeostasis control gene expression in tendon cells. Journal of Orthopaedic Research, 23(5), 1211–1218. [CrossRef]
- Lazarczuk, S. L., Maniar, N., Opar, D. A., Duhig, S. J., Shield, A., Barrett, R. S., & Bourne, M. N. (2022). Mechanical, Material and Morphological Adaptations of Healthy Lower Limb Tendons to Mechanical Loading: A Systematic Review and Meta-Analysis. Sports Medicine, 52(10), 2405–2429. [CrossRef]
- Lee, J., Robshaw, D. C., & Erskine, R. M. (2024). High-intensity resistance training and collagen supplementation improve patellar tendon adaptations in professional female soccer athletes. Experimental Physiology, EP092106. [CrossRef]
- Lee, J., Tang, J. C. Y., Dutton, J., Dunn, R., Fraser, W. D., Enright, K., Clark, D. R., Stewart, C. E., & Erskine, R. M. (2024). The Collagen Synthesis Response to an Acute Bout of Resistance Exercise Is Greater when Ingesting 30 g Hydrolyzed Collagen Compared with 15 g and 0 g in Resistance-Trained Young Men. The Journal of Nutrition, 154(7), 2076–2086. [CrossRef]
- Lis, D. M., & Baar, K. (2019). Effects of Different Vitamin C–Enriched Collagen Derivatives on Collagen Synthesis. International Journal of Sport Nutrition and Exercise Metabolism, 29(5), 526–531. [CrossRef]
- Lis, D. M., Jordan, M., Lipuma, T., Smith, T., Schaal, K., & Baar, K. (2022). Collagen and Vitamin C Supplementation Increases Lower Limb Rate of Force Development. International Journal of Sport Nutrition and Exercise Metabolism, 32(2), 65–73. [CrossRef]
- Maffiuletti, N. A., Aagaard, P., Blazevich, A. J., Folland, J., Tillin, N., & Duchateau, J. (2016). Rate of force development: Physiological and methodological considerations. European Journal of Applied Physiology, 116(6), 1091–1116. [CrossRef]
- Maganaris, C. N., Chatzistergos, P., Reeves, N. D., & Narici, M. V. (2017). Quantification of Internal Stress-Strain Fields in Human Tendon: Unraveling the Mechanisms that Underlie Regional Tendon Adaptations and Mal-Adaptations to Mechanical Loading and the Effectiveness of Therapeutic Eccentric Exercise. Frontiers in Physiology, 8. [CrossRef]
- Malliaras, P., Kamal, B., Nowell, A., Farley, T., Dhamu, H., Simpson, V., Morrissey, D., Langberg, H., Maffulli, N., & Reeves, N. D. (2013). Patellar tendon adaptation in relation to load-intensity and contraction type. Journal of Biomechanics, 46(11), 1893–1899. [CrossRef]
- Marshall, J., Bishop, C., Turner, A., & Haff, G. G. (2021). Optimal Training Sequences to Develop Lower Body Force, Velocity, Power, and Jump Height: A Systematic Review with Meta-Analysis. Sports Medicine, 51(6), 1245–1271. [CrossRef]
- Miller, B. F., Olesen, J. L., Hansen, M., Døssing, S., Crameri, R. M., Welling, R. J., Langberg, H., Flyvbjerg, A., Kjaer, M., Babraj, J. A., Smith, K., & Rennie, M. J. (2005). Coordinated collagen and muscle protein synthesis in human patella tendon and quadriceps muscle after exercise. The Journal of Physiology, 567(3), 1021–1033. [CrossRef]
- Nulty, C. D., Phelan, K., & Erskine, R. M. (2025). Hydrolysed Collagen Supplementation Enhances Patellar Tendon Adaptations to 12 Weeks’ Resistance Training in Middle-Aged Men. European Journal of Sport Science, 25(4), e12281. [CrossRef]
- Nulty, C. D., Tang, J. C. Y., Dutton, J., Dunn, R., Fraser, W. D., Enright, K., Stewart, C. E., & Erskine, R. M. (2024). Hydrolyzed collagen supplementation prior to resistance exercise augments collagen synthesis in a dose-response manner in resistance-trained, middle- aged men.
- Nunez-Lisboa, M., Dewolf, A. H., Cataldo, M., Castro-Sepulveda, M., Zbinden-Foncea, H., & Cancino-Lopez, J. (2021). Hydrolyzed Collagen Supplementation on Lower Body Stiffness in Recreational Triathletes. Asian Journal of Sports Medicine, 12(3). [CrossRef]
- OCEBM levels of evidence. (2011). https://www.cebm.ox.ac.uk/resources/levels-of-evidence/ocebm-levels-of-evidence.
- Oikawa, S. Y., Kamal, M. J., Webb, E. K., McGlory, C., Baker, S. K., & Phillips, S. M. (2020). Whey protein but not collagen peptides stimulate acute and longer-term muscle protein synthesis with and without resistance exercise in healthy older women: A randomized controlled trial. The American Journal of Clinical Nutrition, 111(3), 708–718. Correction in Am J Clin Nutr. 2020;111(3), 708-718. [CrossRef]
- Peñailillo, L., Blazevich, A., Numazawa, H., & Nosaka, K. (2015). Rate of force development as a measure of muscle damage. Scandinavian Journal of Medicine & Science in Sports, 25(3), 417–427. [CrossRef]
- Quinlan, J. I., Narici, M. V., Reeves, N. D., & Franchi, M. V. (2019). Tendon Adaptations to Eccentric Exercise and the Implications for Older Adults. Journal of Functional Morphology and Kinesiology, 4(3), 60. [CrossRef]
- Shaw, G., Lee-Barthel, A., Ross, M. L., Wang, B., & Baar, K. (2017). Vitamin C–enriched gelatin supplementation before intermittent activity augments collagen synthesis. The American Journal of Clinical Nutrition, 105(1), 136–143. [CrossRef]
- Swartz, M. K. (2011). The PRISMA Statement: A Guideline for Systematic Reviews and Meta-Analyses. Journal of Pediatric Health Care, 25(1), 1–2. [CrossRef]
- Thorlund, J. B., Michalsik, L. B., Madsen, K., & Aagaard, P. (2008). Acute fatigue-induced changes in muscle mechanical properties and neuromuscular activity in elite handball players following a handball match. Scandinavian Journal of Medicine & Science in Sports, 18(4), 462-472. [CrossRef]
- Waugh, C. M., Blazevich, A. J., Fath, F., & Korff, T. (2012). Age-related changes in mechanical properties of the Achilles tendon. Journal of Anatomy, 220(2), 144–155. [CrossRef]
- Zdzieblik, D., Oesser, S., Baumstark, M. W., Gollhofer, A., & König, D. (2015). Collagen peptide supplementation in combination with resistance training improves body composition and increases muscle strength in elderly sarcopenic men: A randomised controlled trial. British Journal of Nutrition, 114(8), 1237–1245. Correction in Br J Nutr. 2025, 134(5), 440. [CrossRef]


| Question Component | Inclusion Criteria | Exclusion Criteria |
| Population | ∙ Studies involving human participants of any age, sex, or ethnicity. ∙ Participants with healthy tendons or pathological tendons. |
∙ Studies involving animal models or non-human subjects. ∙ Studies focusing exclusively on populations with underlying comorbidities that may confound tendon health (e.g., severe systemic diseases). |
| Intervention | ∙ Studies that investigate collagen supplementation (including hydrolyzed collagen, gelatin, or collagen peptides). ∙ Studies that include proline or glycine as part of the supplementation regimen. |
∙ Studies that do not specifically investigate collagen, gelatin, proline, or glycine supplementation. |
| Comparison | ∙ Studies that include a control group receiving either a placebo or no treatment. ∙ Studies comparing different types of collagen supplementation. |
∙ Studies without a control group or appropriate comparison group. |
| Outcome | ∙ Studies reporting on tendon health outcomes, including but not limited to: o Tendon structure o Tendon mechanical properties o Muscle Performance o Physical Performance o Pain levels associated with tendon injuries |
∙ Studies that do not report tendon health outcomes |
| Study Design | ∙ RCTs, cohort studies, case-control studies. ∙ Hand selected articles that come from the review of references of the included studies. |
∙ Non-peer-reviewed studies, abstracts, conference proceedings, or unpublished data. ∙ Reviews, meta-analyses, or opinion pieces. ∙ Studies without accessible full text |
| Level of Evidence |
Study Characteristics |
| I | Evidence obtained from high-quality randomized controlled trials, prospective studies, or diagnostic studies. |
| II | Evidence obtained from lesser quality randomized control trials, prospective studies or diagnostic studies (e.g., improper randomization, no blinding, <80% follow-up) |
| III | Case controlled studies or retrospective studies. |
| IV | Case Series |
| V | Expert Opinion |
| Grade of Recommendation | Strength of Evidence | |
| A | Strong | A preponderance of level I and/or level II studies support the recommendation. Must include ≥ 1 level I study. |
| B | Moderate | A single high-quality randomized controlled trial or a preponderance of level II studies support the recommendation. |
| C | Weak | A single level II study or a preponderance of level III and level IV studies including statements of consensus by content experts support the recommendation. |
| D | Conflicting | Higher-quality studies conducted on this topic disagree with respect to their conclusions. The recommendation is based on these conflicting studies. |
| E | Theoretical/Foundational | A preponderance of evidence from animal or cadaver studies, from conceptual models/principles, or from basic sciences/bench research support this conclusion. |
| F | Expert Opinion | Best practice based on the clinical experience of the guidelines development team. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




