Submitted:
09 February 2026
Posted:
10 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
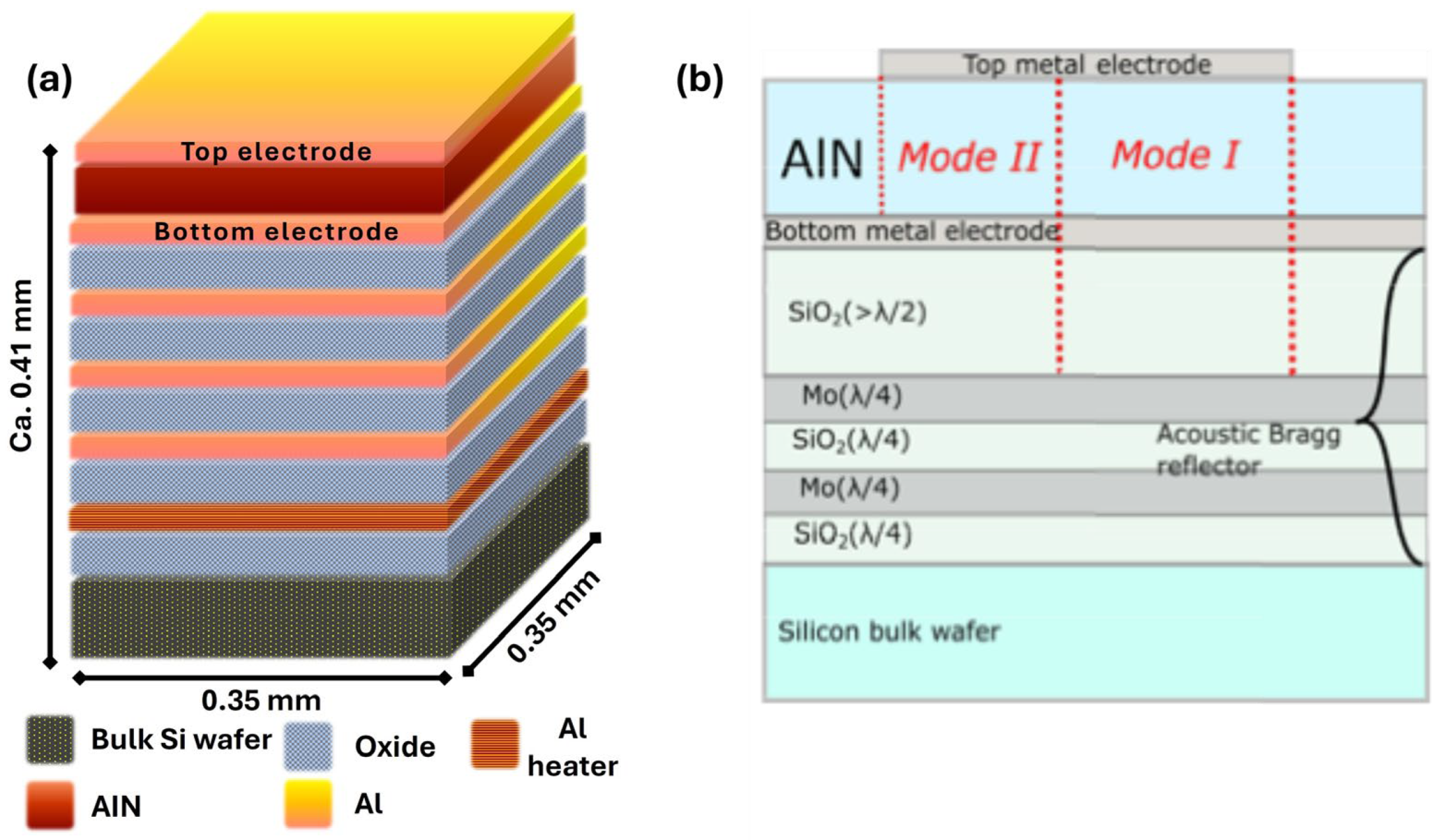
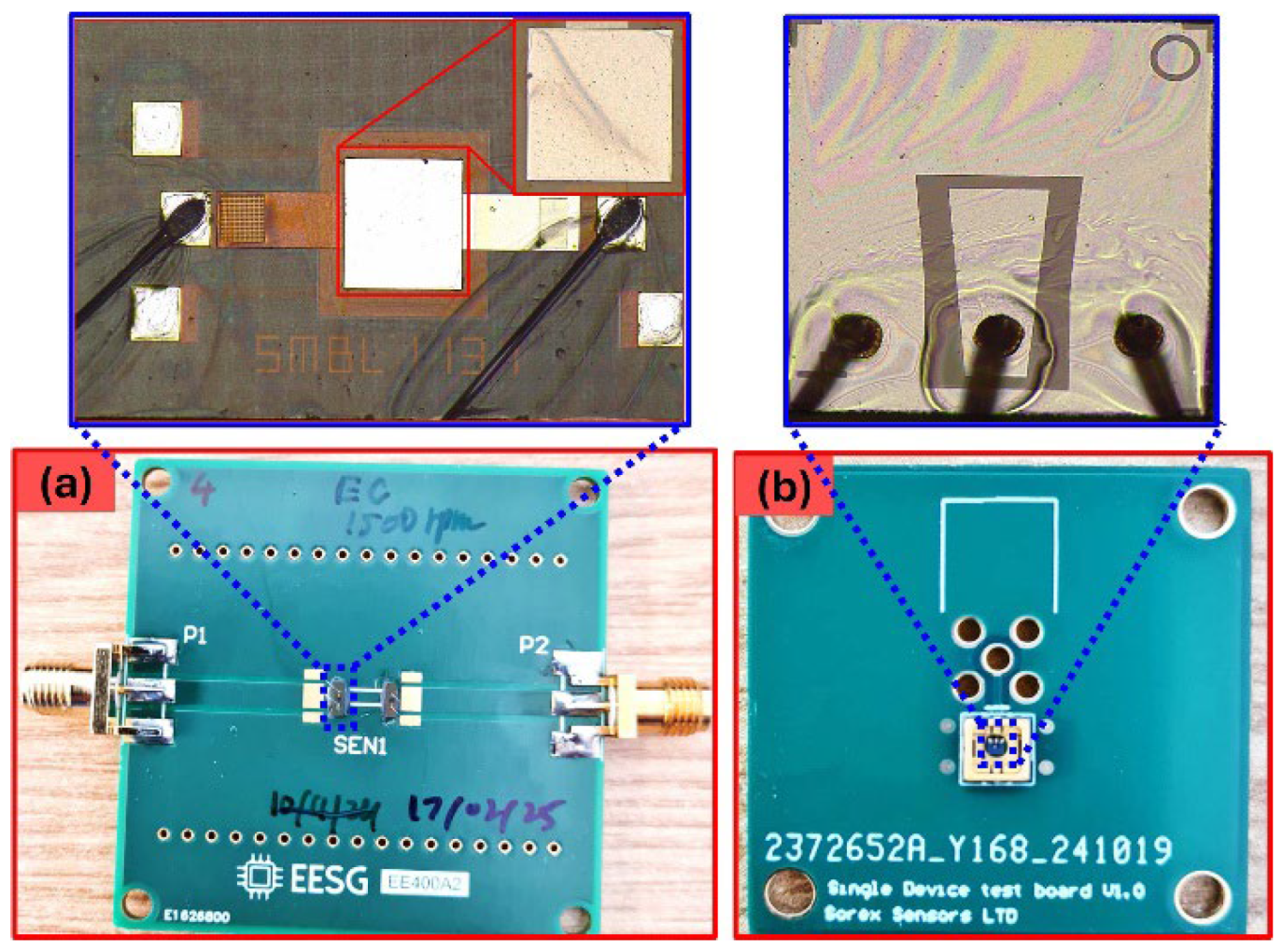
2.1. Sensor Fabrication and Functionalization
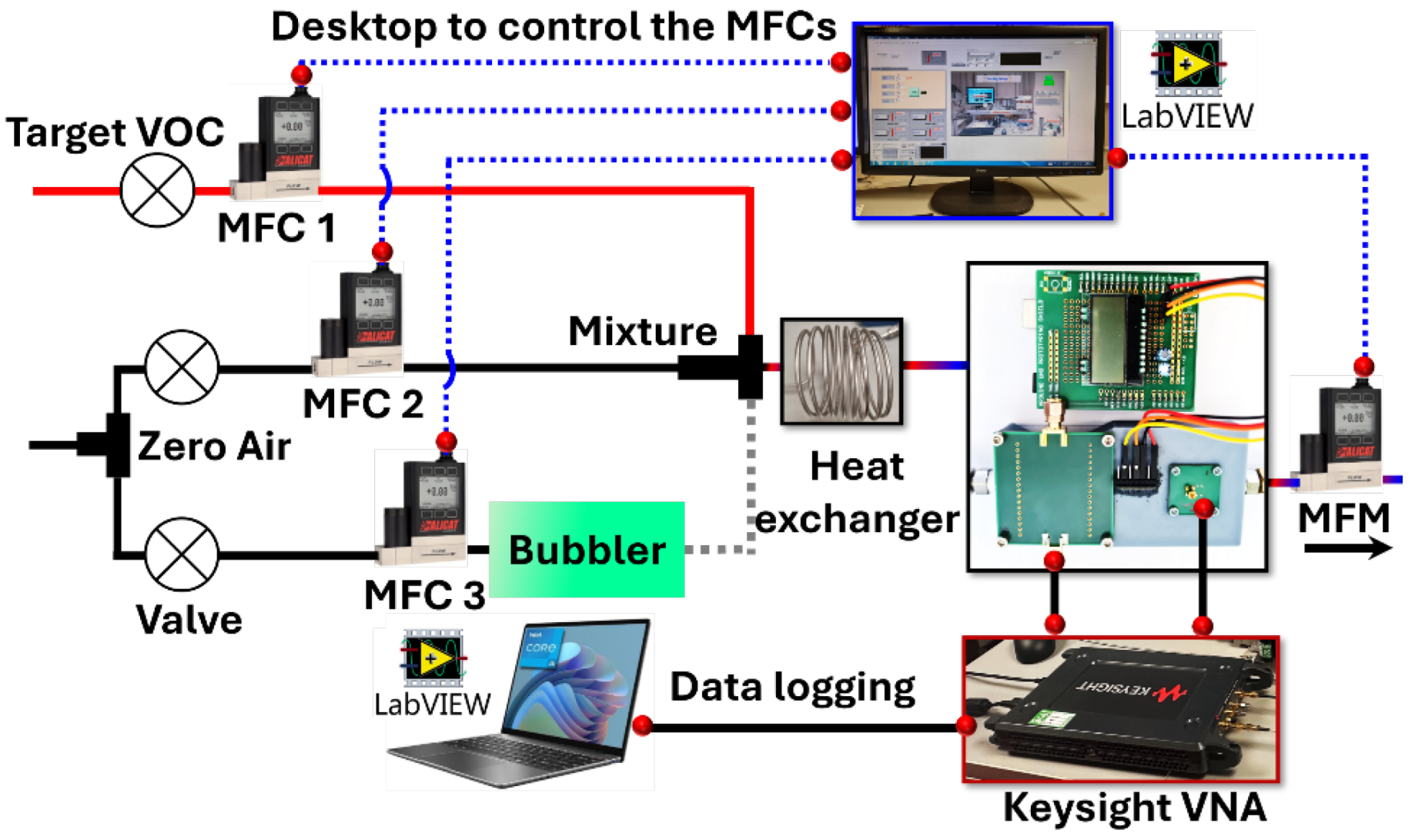
2.2. Sensor Testing Setup
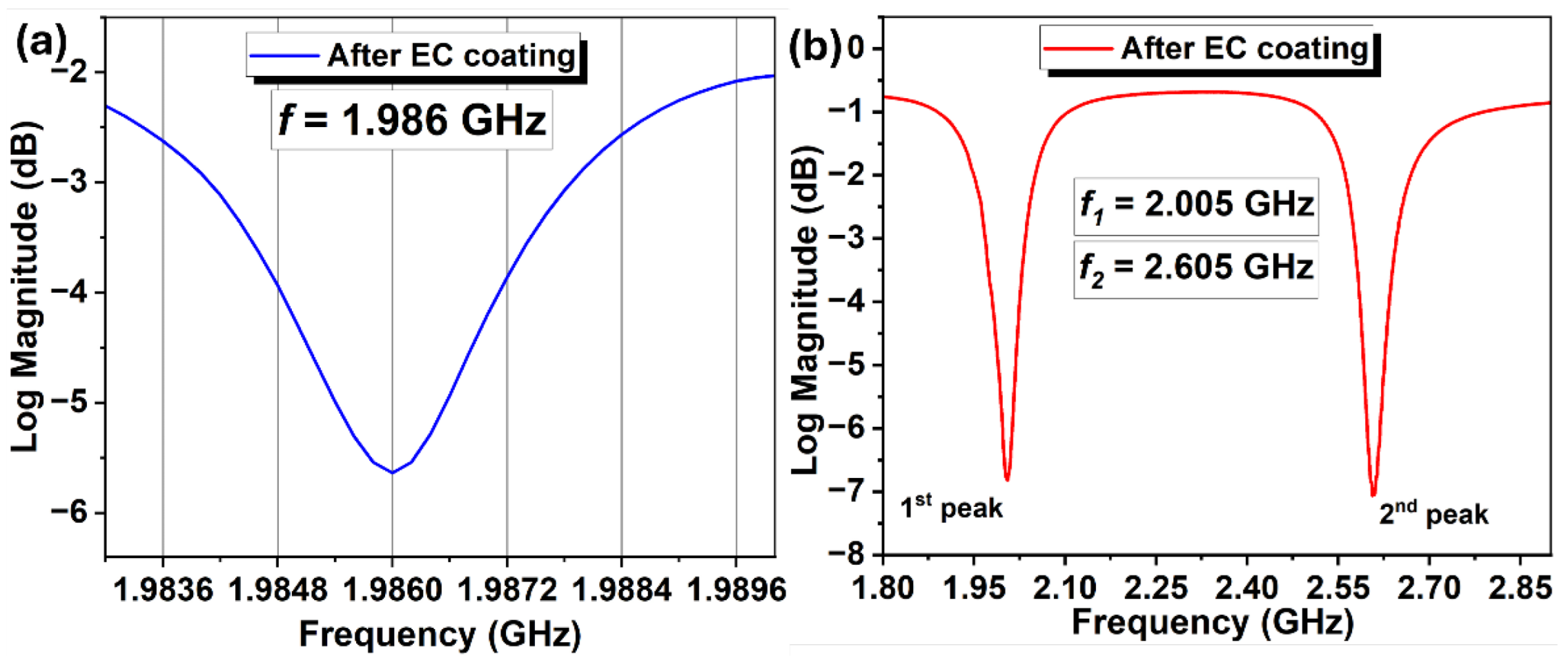
3. Results and Discussions
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Riccardo F.; Miroslav B.; Martin P.; Gunda T.; Carla C. M. A.; Maria L. I.; Ted C. J. T.; Jorge M. S. F.; Thomas J.; Damien B.; Alberto P.; Sergio A.; Luca C. The use of volatile organic compounds in preventing and managing invasive plant pests and pathogens. Front. Hort. 2024, 3.
- Pérez-H. M.; Gallego-G. C.; Forner-G. M. Á.; Ortells-F. R.; Urbaneja, A. Plant volatile-triggered defence in citrus against biotic stressors. Front. Plant Sci., 2024, 15.
- Li X.; Wang Q.; Li H.; Wang X.; Zhang R.; Yang X.; Jiang Q.; Shi Q. Revealing the mechanisms for linalool antifungal activity against Fusarium oxysporum and its efficient control of Fusarium wilt in tomato plants. Int. J. Mol. Sci., 2023, 24(1), 458. [CrossRef]
- Kessler A.; Baldwin I. T. Defensive function of herbivore-induced plant volatile emissions in nature, Science, 2001, 291, pp. 2141–2144.
- Dicheng M.; Guoxian W.; Jiamei Z.; Wei M.; Daolong D.; Feng L. Green-leaf volatile trans-2-hexenal inhibits the growth of Fusarium graminearum by inducing membrane damage, ROS accumulation, and cell dysfunction. J. Agric. Food Chem., 2022, 70, pp. 5646-5657.
- Ouyang Q.; Shi S.; Liu Y.; Yang Y.; Zhang Y.; Yuan X.; Tao N.; Li L. Inhibitory Mechanisms of trans-2-Hexenal on the Growth of Geotrichum citri-aurantii. J. Fungi. 2023, 9. [CrossRef]
- Yan W.; Liming G.; Zhanhong Z.; Kailong L.; Zhuo Z.; Deyong Z.; Jianbin C.; Jing P.; Yang G.; Jiao D.; Shuo Y.; Xiaobin S.; Yong L. D-Limonene affects the feeding behavior and the acquisition and transmission of Tomato Yellow Leaf Curl Virus by Bemisia tabaci. Viruses, 2024, 16(2), 300. [CrossRef]
- Pérez-H. M.; Gallego-G. C.; Forner-G. M. Á.; Ortells-F. R.; Urbaneja, A. Plant volatile-triggered defence in citrus against biotic stressors. Front. Plant Sci., 2024, 15.
- Alphus D. W.; Manuela B. Advances in electronic-nose technologies developed for biomedical applications. Sensors, 2011, 11(1), 1105–1176. [CrossRef]
- Alphus D. W. Applications of electronic-nose technologies for noninvasive early detection of plant, animal and human diseases. Chemosensors, 2018, 6(4), 45. [CrossRef]
- O’Sullivan C. K.; Guilbault, G. G. Commercial quartz crystal microbalances—Theory and applications. Biosens. Bioelectron., 1999, 14, pp. 663–670.
- Hajjaj A. Z.; Jaber N.; Alcheikh N.; Younis M. I. A resonant gas sensor based on multimode excitation of a buckled microbeam. IEEE Sensors Journal, 2020, 20, pp. 1778–1785. [CrossRef]
- Yaqoob U.; Jaber N.; Alcheikh N.; Younis M. I. Selective multiple analyte detection using multi-mode excitation of a MEMS resonator. Sci. Rep., 2022, 12, pp. 1–10. [CrossRef]
- Yaqoob U.; Lenz W. B.; Alcheikh N.; Jaber N.; and Younis M. I. Highly selective multiple gases detection using a thermal-conductivity-based MEMS resonator and machine learning. IEEE Sensors Journal, 2022, 22, pp. 19858-19866.
- Liu Y.; Wang L. Film bulk acoustic resonators (FBARs) as biosensors: A review. Biosens. Bioelectron., 2018, 100, pp. 256–270.
- Villa-López F. H.; Cole M.; Thomas S.; Gardner J. W. Indoor air quality monitor based on solidly mounted resonators for the detection of VOCs. in Proc. IEEE Sensors, 2017, Glasgow, U.K. pp. 1–3.
- Xuan W.; Cole M.; Gardner J. W.; Thomas S.; Villa-López F. H.; Wang X.; Dong S.; Luo J. A film bulk acoustic resonator oscillator based humidity sensor with graphene oxide as the sensitive layer. J. Micromech. Microeng., 2017, 27, p. 055017.
- Gao F.; Xuan W.; Bermak A.; Boussaid F.; Tsui C. Y.; Luo J. Dual transduction on a single sensor for gas identification. Sens. Actuators B Chem., 2019, 278, pp. 21–27.
- Johar A. K.; Sharma G. K.; Bhatt J. K.; Kumar A. VOCs sensors based on bulk acoustic resonators: A comprehensive review. Microsyst. Technol., 30, 7, 2024, pp. 821–843. [CrossRef]
- Specht J. P.; Esfahani S.; Tan T. H.; S-Ludwikowska F.; W-Teubenbacher R.; Kock A.; Cole M.; Gardner J. W. AlN FBAR particle sensor with a thermophoretic sampling mechanism. IEEE Sensors Journal, 2021, 21, pp. 19427–19435.
- Wang L.; Wang C.; Wang Y.; Quan A.; Keshavarz M.; Maderia B. P.; Zhang H.; Wang C.; Kraft M. A review on coupled bulk acoustic wave MEMS resonators. Sensors, 2022, 22, 10, p. 3857.
- Gao J.; Liu G.; Li J.; Li G. Recent developments of film bulk acoustic resonators (FBAR), Funct. Mater. Lett., 2016, 9, pp. 1630002.
- Thomas S.; Villa-López F. H.; Theunis J.; Peters J.; Cole M.; Gardner J. W. Particle sensor using solidly mounted resonators. IEEE Sensors Journal, 2016, 16 pp. 2282-2289.
- Specht J. P.; Esfahani S.; Xing Y.; Köck A.; Cole M.; Gardner J. W. Thermally modulated CMOS-compatible particle sensor for air quality monitoring. IEEE Trans. Instrum. Meas., 2022, 71, pp. 1–13.
- Carmona-Cejas J. M.; Mirea T.; Hervás-García R.; Olivares J.; Clement M. Dual-mode solidly mounted resonator-based sensor for temperature and humidity detection and discrimination. Sensors, 2024, 24, p. 2877. [CrossRef]
- Yaqoob U.; Urasinska-Wojcik B.; Esfahani S.; Cole M.; Gardner J. W. Rapid and Selective Detection of Linalool Using Solidly Mounted Resonators for Plant Health Monitoring. IEEE Sensors Journal, 2025. [CrossRef]
- Tamboli A.; Kale F.; Hrncirik M.; DeMiguel-Ramos, Flewitt A. J. Oven-Controlled Dual-Mode Solidly Mounted Resonator (SMR)-Based Biosensor. IEEE Sensors Journal, 2025, 25, pp. 39563-39573.
- Tamboli, A. Thermally modulated SMRs in biosensing, PhD thesis University of Cambridge, 2024.
- Midzi J.; Jeffery D. W.; Baumann U.; Rogiers S.; Tyerman S. D.; Pagay V. Stress-Induced Volatile Emissions and Signalling in Inter-Plant Communication. Plants, 2022,11, 2566. [CrossRef]
- Aratani Y.; Uemura T.; Hagihara T. Green leaf volatile sensory calcium transduction in Arabidopsis. Nat. Commun., 2023, 14, 6236.
- Niu D.; Xu L.; Lin K. Multitrophic and Multilevel Interactions Mediated by Volatile Organic Compounds. Insects, 2024, 15, 572. [CrossRef]
- Thuresson K.; Lindman B.; Nyström B. Effect of Hydrophobic Modification of a nonionic cellulose derivative on the interaction with surfactants. Rheology. J. Phys. Chem. B., 1997, 101, pp. 6450–6459.
- Ioelovich M. Adjustment of hydrophobic properties of cellulose materials. Polymers, 2021, 13, p. 1241. [CrossRef]
- Oh E.; Luner P. E. Surface free energy of ethylcellulose films and the influence of plasticizers. Int J Pharm. 1999, 25.
- Horvat G.; Žvab K.; Knez Ž.; Novak Z. Simple, One-Pot Method for Preparing Transparent Ethyl Cellulose Films with Good Mechanical Properties. Polymers, 2022, 14. [CrossRef]
- Zhang R.; Sun B.; Chen L.; Chen C.; Sun D. An ethyl cellulose-coated bacterial cellulose based hydrophobic and degradable straw-like materials towards drinking straws. Carbohydr Polym., 2025, 367. 124046. [CrossRef]
- Foo K. Y.; Hameed B. H. Insights into the modeling of adsorption isotherm systems. Chemical Engineering Journal, 2009, 156, pp. 2–10.

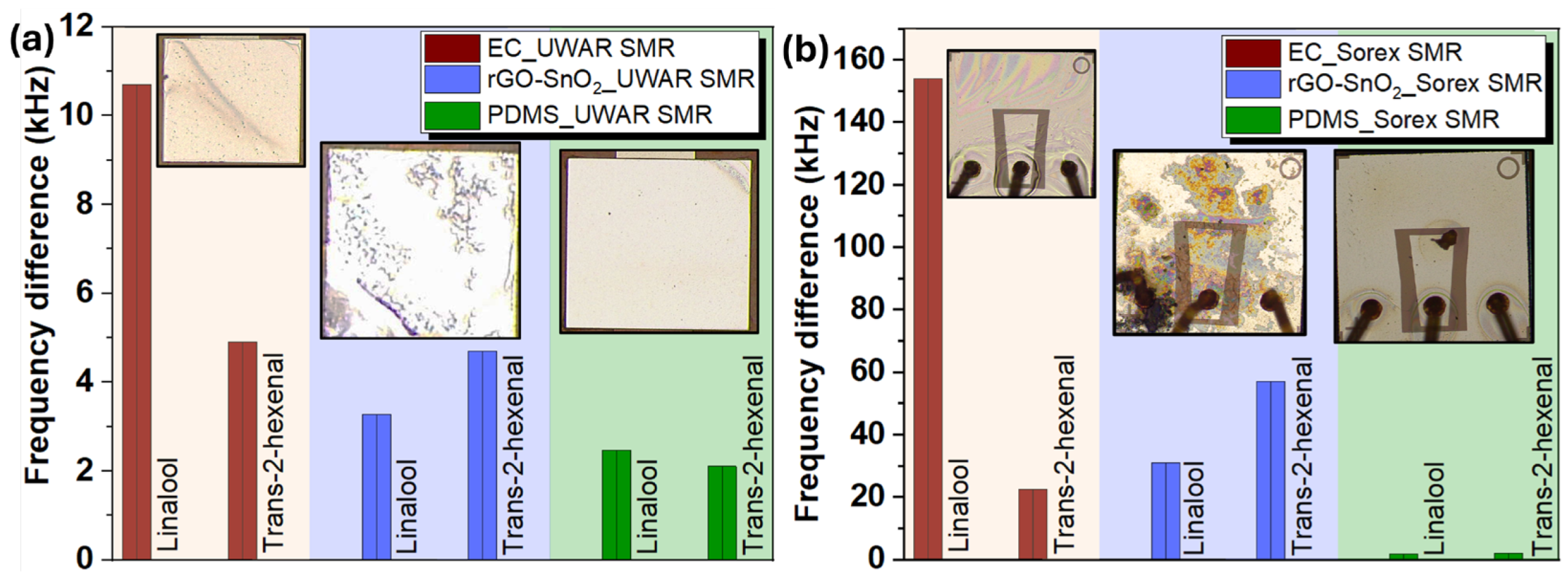
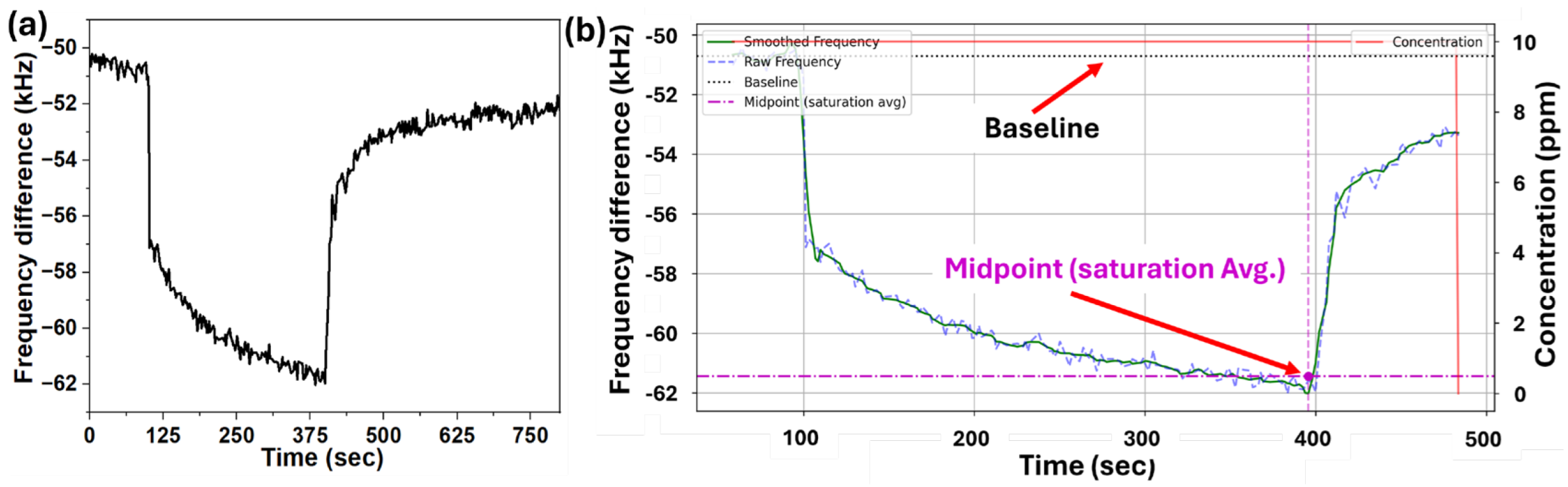
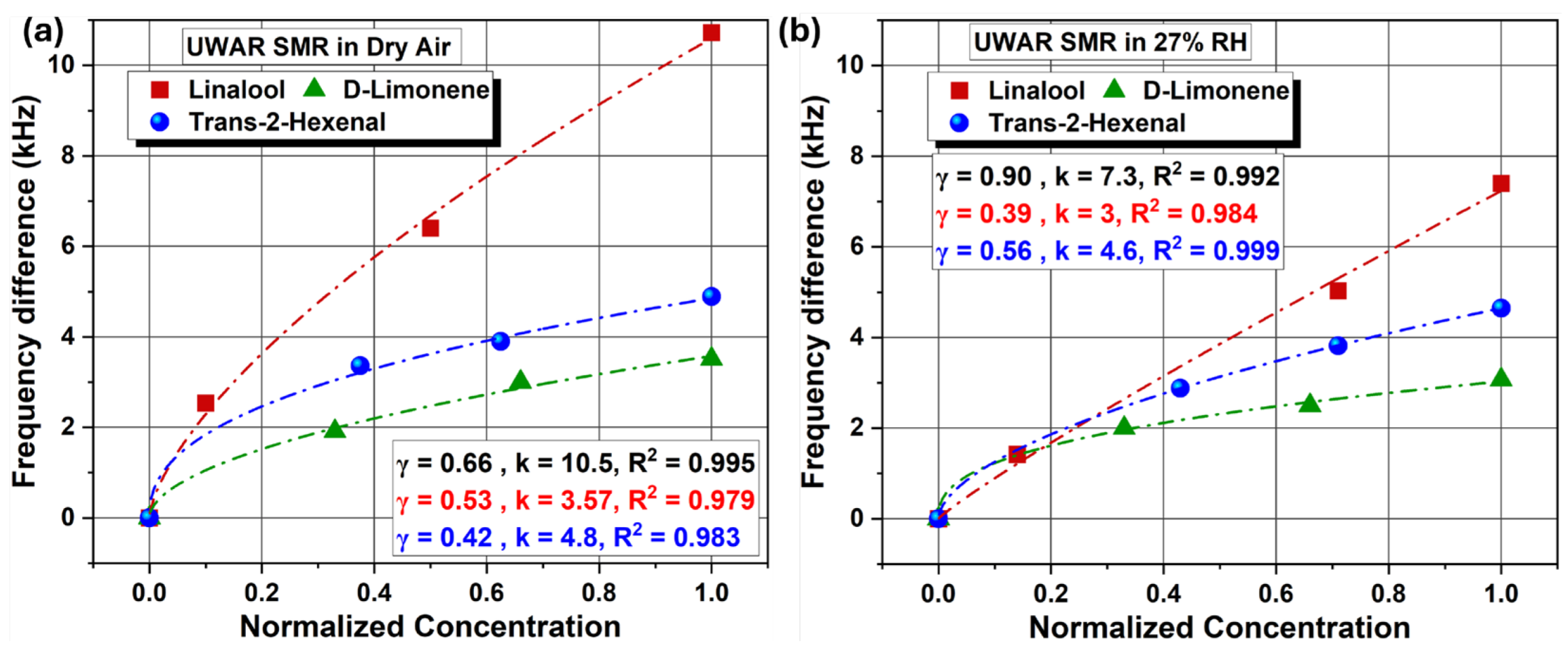
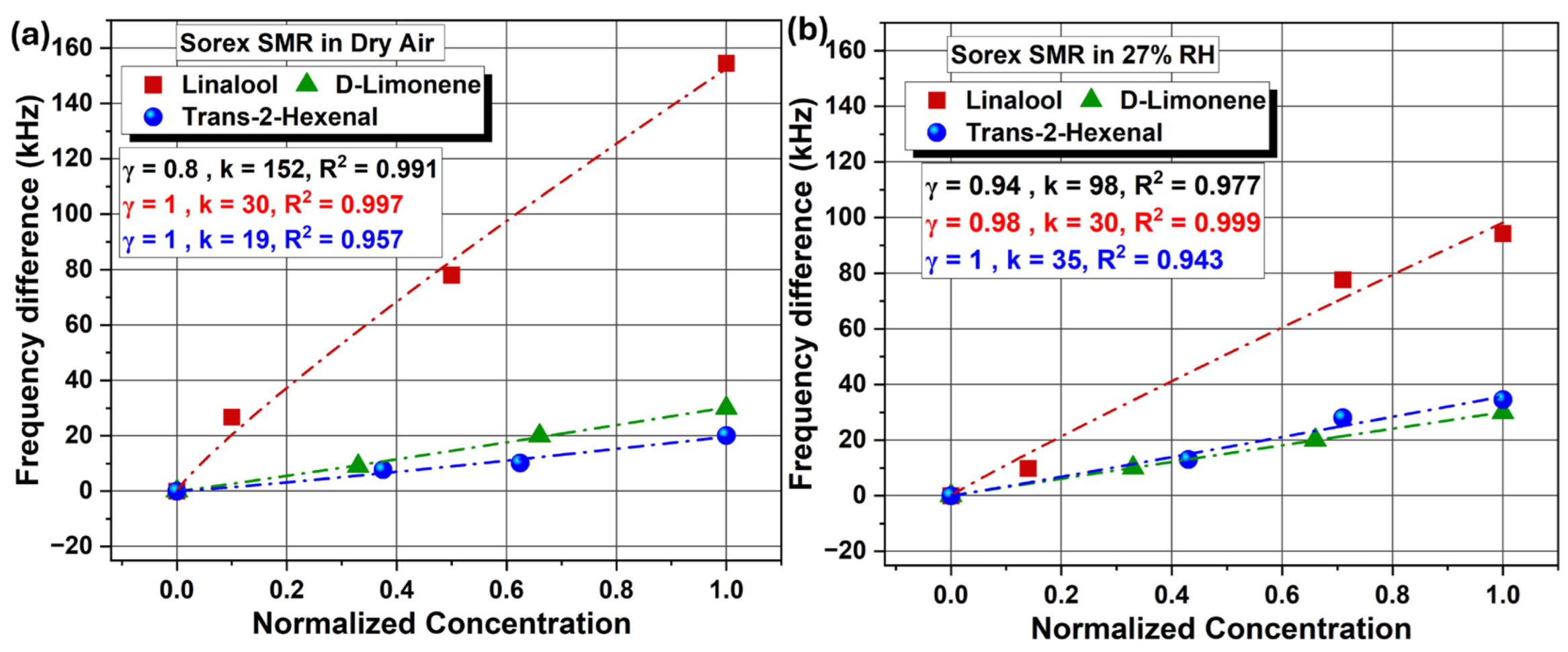
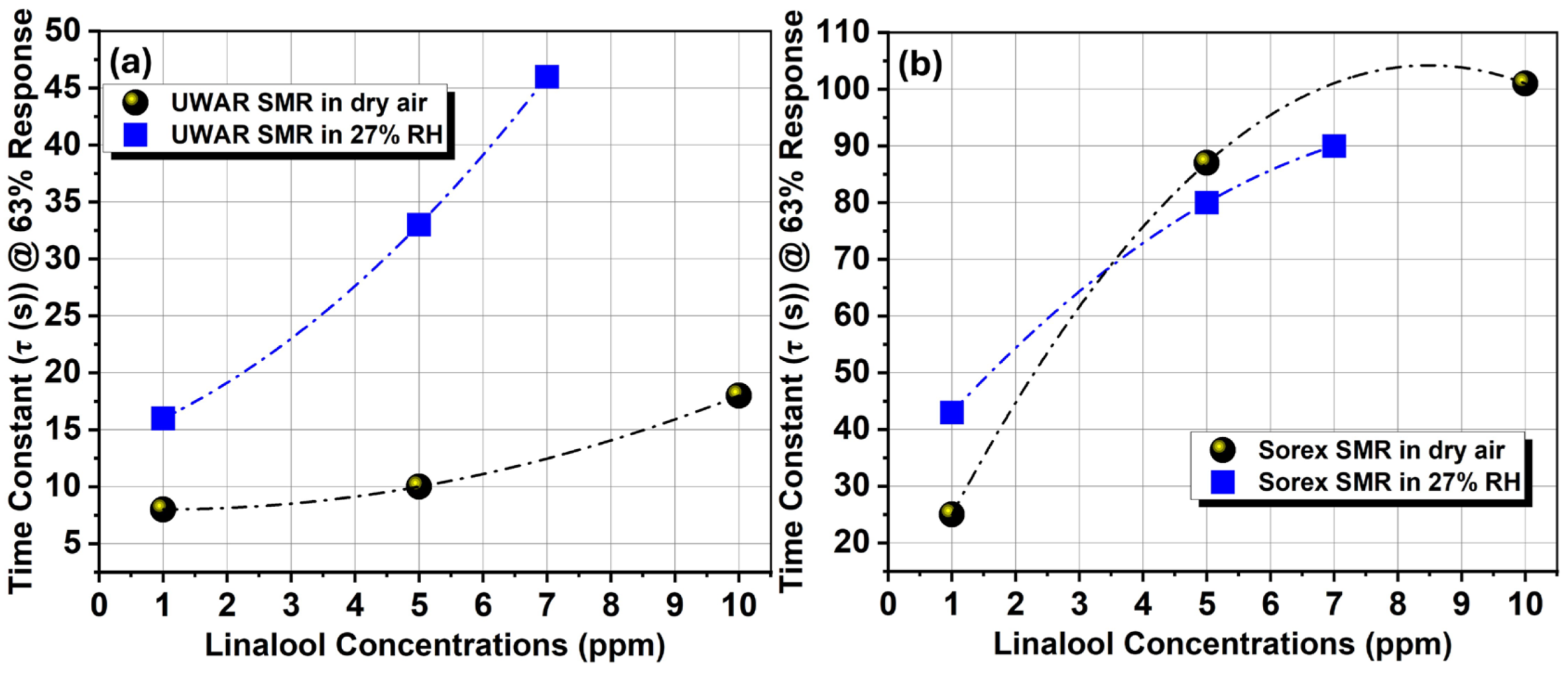
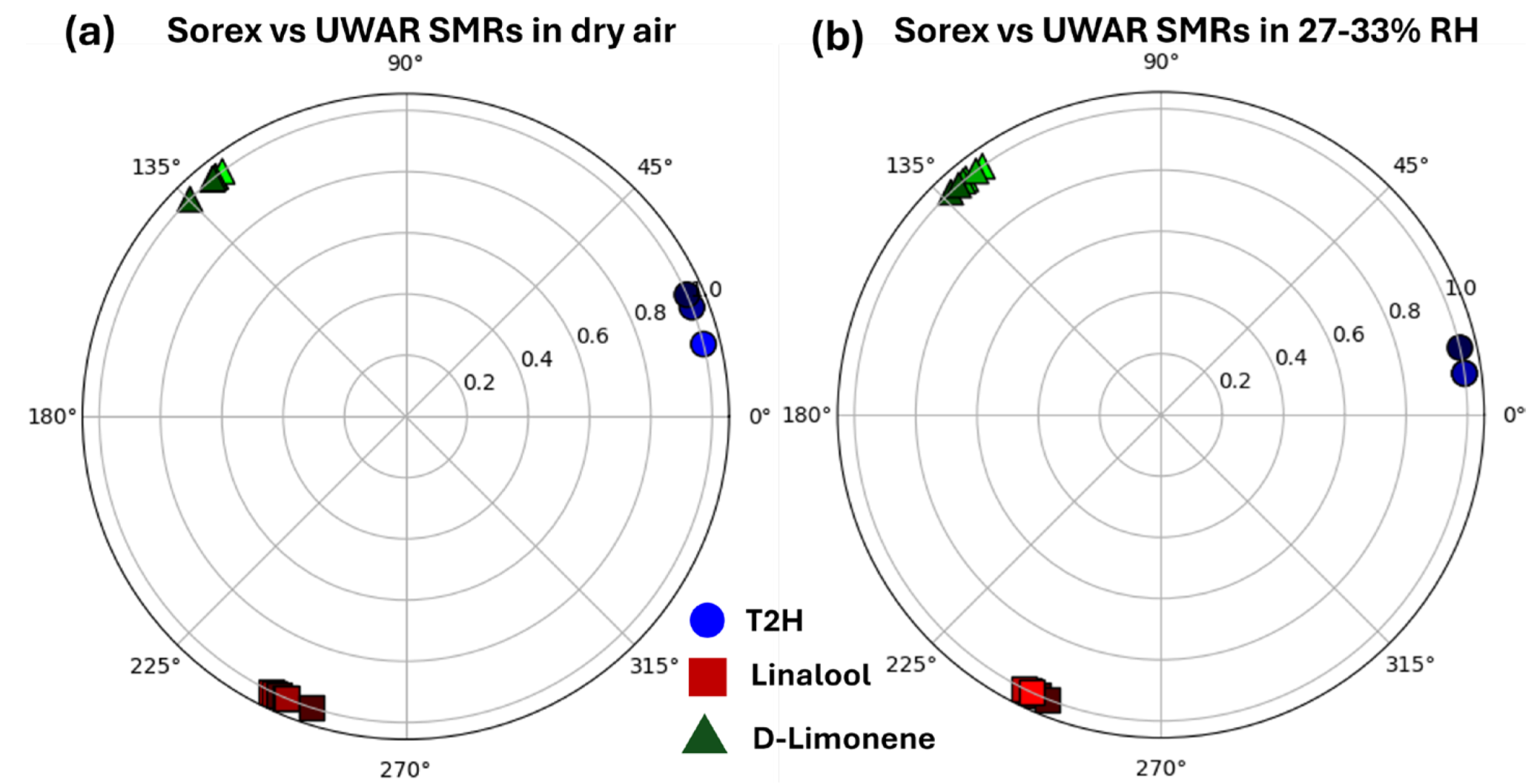
| Coating | Sensors | Linalool | D-limonene | T2H |
| Ethyl cellulose | UWAR | S = 1.03 kHz/ppm | S = 0.1 kHz/ppm | S = 0.059 kHz/ppm |
| Sorex | S = 15 kHZ/ppm | S = 0.89 kHZ/ppm | S = 0.22 kHZ/ppm |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




