Submitted:
09 February 2026
Posted:
10 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Conceptual Foundations of Functional ADE Platform Design
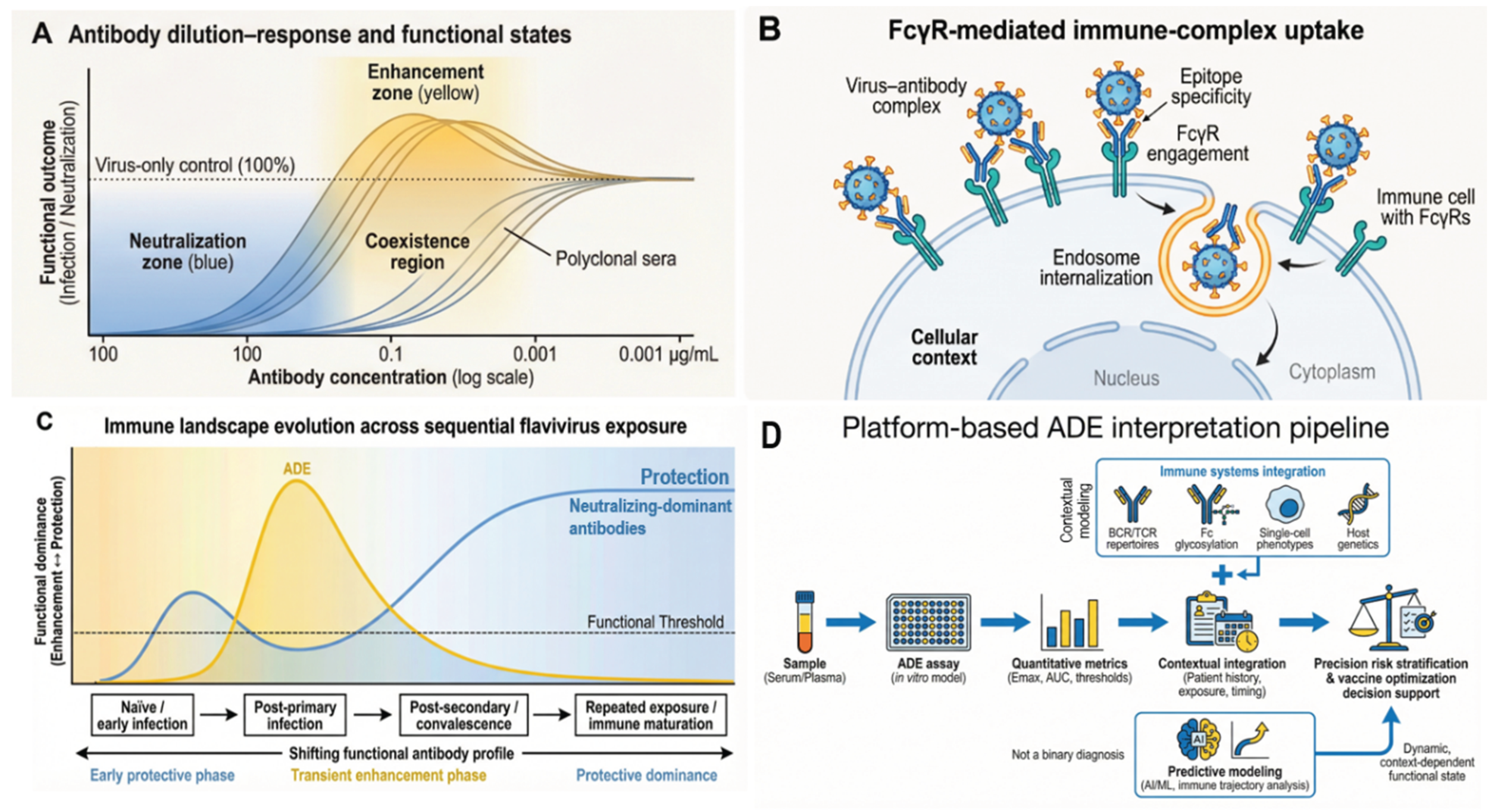
2.1. ADE as a Functional State, Not an Intrinsic Antibody Property
2.2. Immune-Complex Stoichiometry and Concentration Dependence
2.3. Fcγ Receptor Engagement Determines Entry Route and Intracellular Fate
- selection of FcγR-relevant target cells (cell lines or primary cells),
- systematic documentation and monitoring of FcγR expression stability across passages, and
- avoidance of overgeneralization from single-cell models.
2.4. Target-Cell and Viral Input Context as Hidden Confounders
2.5. Assay Architecture, Functional Endpoints, and Platform Logic
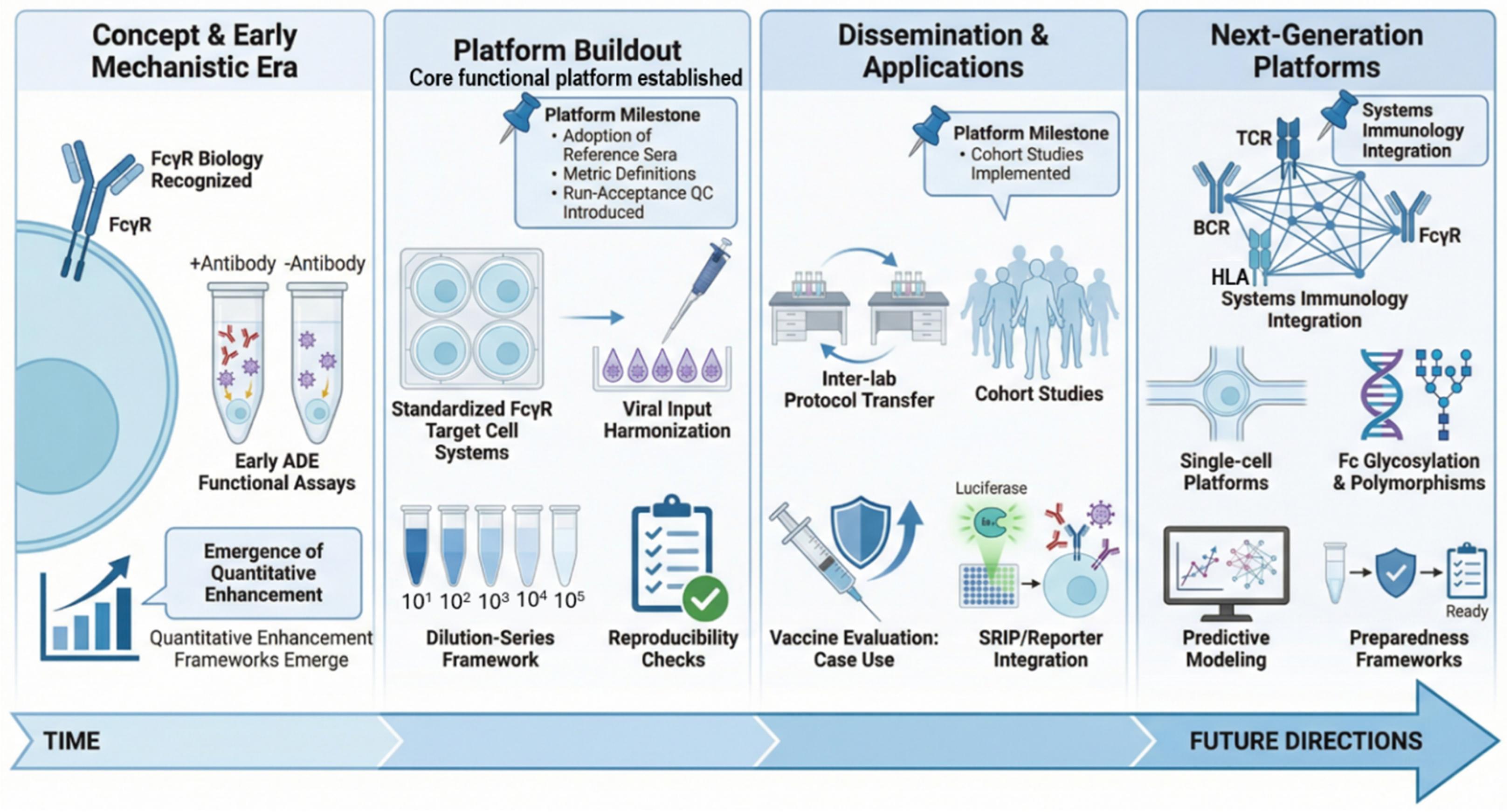
3. Application of Functional ADE Platforms Across Research and Translational Context
3.1. Clinical, Cohort, and Field Applications
3.2. Vaccine, Booster, and Therapeutic Evaluation
3.3. Cross-Platform and Systems-Level Integration
3.4. Operational Deployment and Translational Value
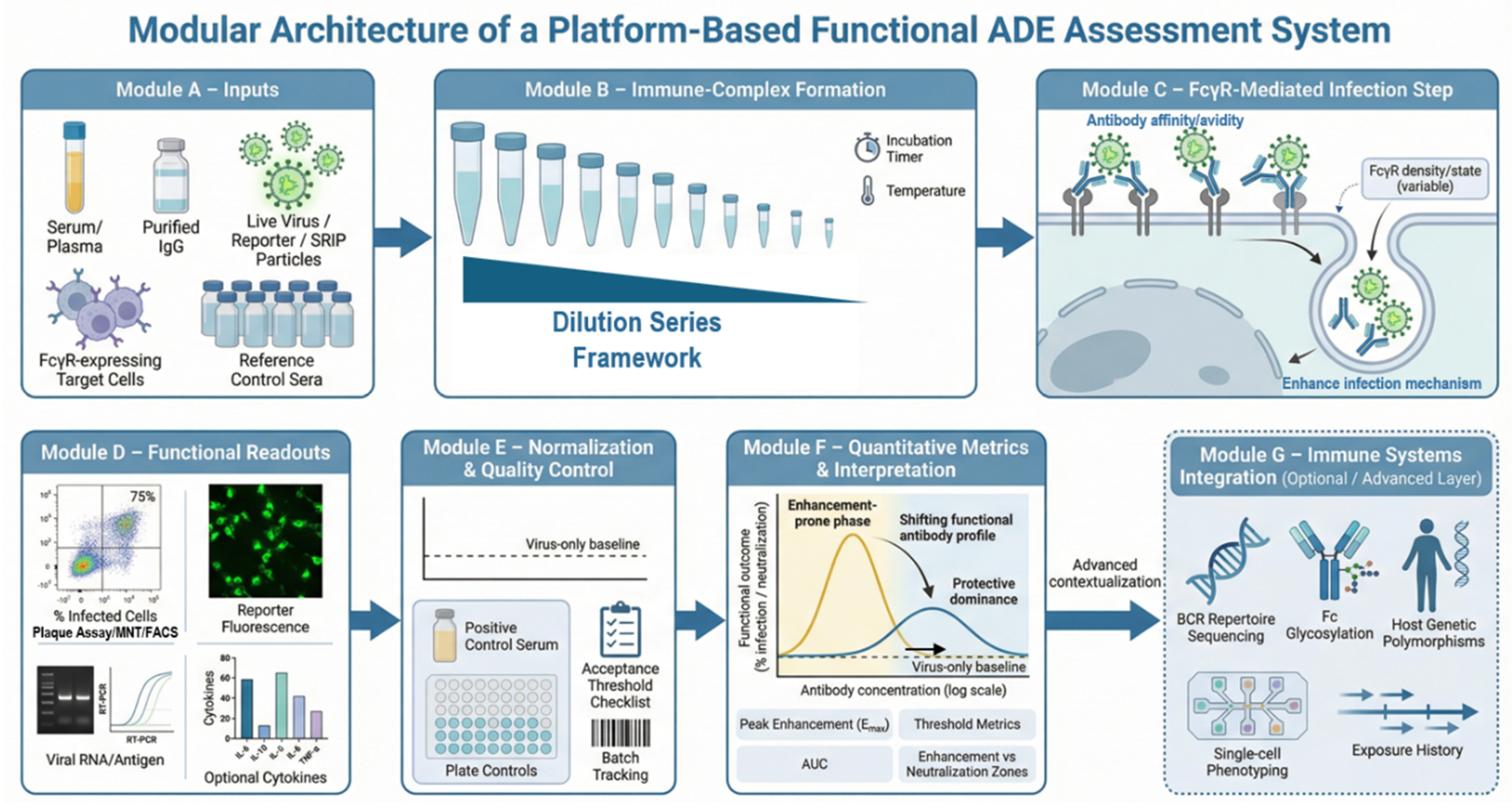
4. Methods and Reporting Standards for Platform-Based Functional ADE Assessment
4.1. Platform-Oriented Design and Experimental Architecture
4.2. Immune-Complex Formation, Viral Input Control, and Quality Assurance
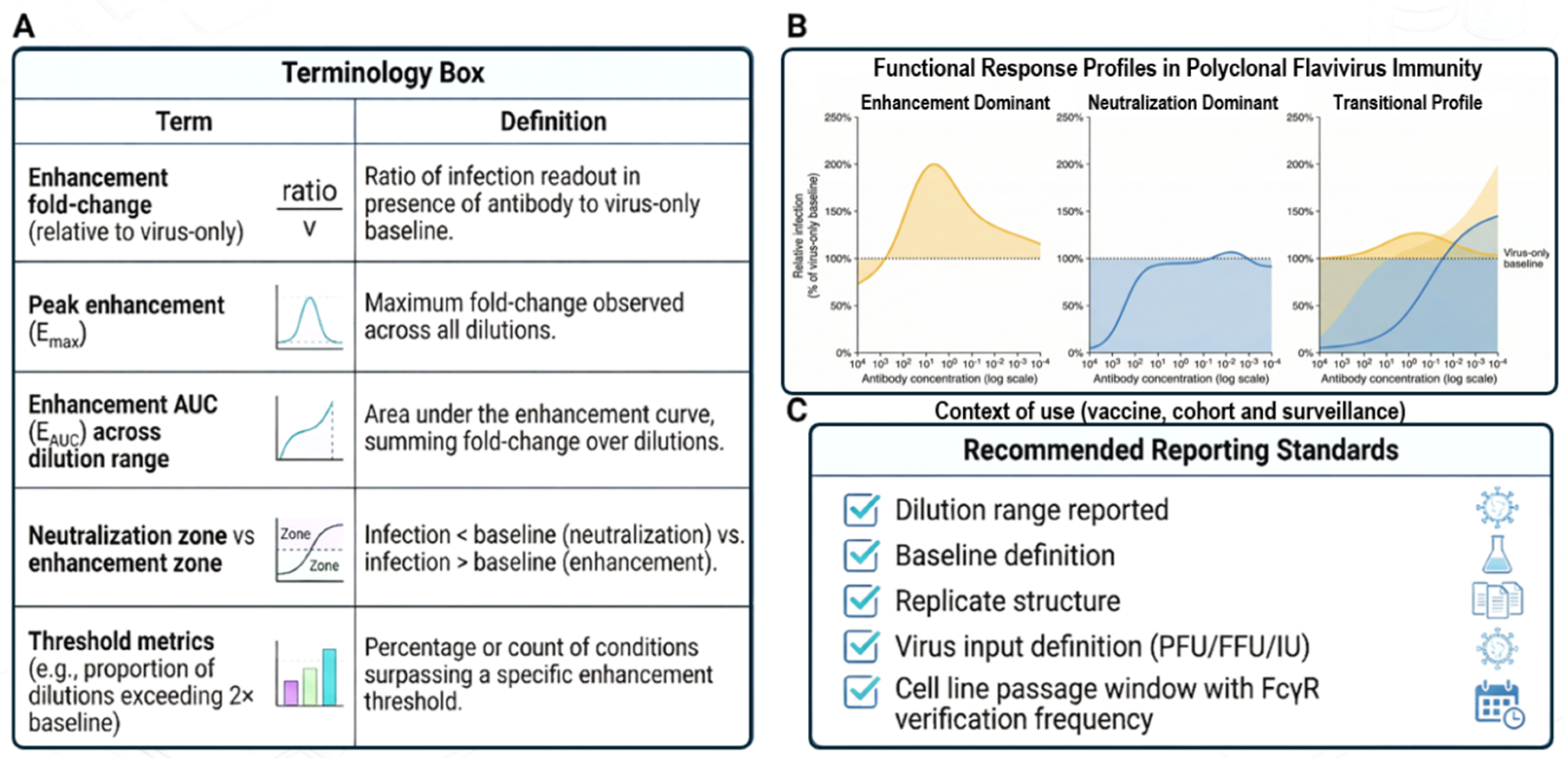
4.3. Readout Integration, Quantitative Metrics, and Interpretive Frameworks
4.4. Reporting Standards, Data Governance, and Translational Alignment
5. Discussion
6. Conclusions
Author Contributions
Funding
Acknowledgments
AI Usage Statement
Conflicts of Interest
Abbreviations
| ADE | Antibody-Dependent Enhancement |
| AUC | Area Under the Curve |
| BCR | B-Cell Receptor |
| DENV | Dengue Virus |
| ECBS | WHO Expert Committee on Biological Standardization |
| E_max | Maximum Enhancement |
| E_AUC | Enhancement Area Under the Curve |
| FcγR | Fc Gamma Receptor |
| FFU | Focus-Forming Unit |
| FRNT | Focus reduction neutralization test |
| HLA | Human Leukocyte Antigen |
| ICS | Intracellular Cytokine Staining |
| IFN-γ | Interferon Gamma |
| IgG | Immunoglobulin G |
| IU | Infectious Unit |
| JEV | Japanese Encephalitis Virus |
| MNT | Microneutralization test |
| PBMC | Peripheral Blood Mononuclear Cell |
| PFU | Plaque-Forming Unit |
| PRNT | Plaque reduction neutralization test |
| QC | Quality Control |
| RT-PCR | Reverse Transcription PCR |
| SRIP | Single-Round Infectious Particle |
| TCR | T-Cell Receptor |
References
- World Health Organization (WHO). Dengue: Guidelines for Diagnosis, Treatment, Prevention and Control; WHO Press: Geneva, Switzerland, 2009. [Google Scholar]
- World Health Organization (WHO). Guidelines on the Quality, Safety and Efficacy of Dengue Tetravalent Vaccines (Live, Attenuated): Annex 2; WHO Technical Report Series No. 979; WHO Press: Geneva, Switzerland, 2013. [Google Scholar]
- Moi, ML; Takasaki, T; Kurane, I. Human antibody response to dengue virus: implications for dengue vaccine design. Trop Med Health 2016, 44, 1. [Google Scholar] [CrossRef]
- Bournazos, S; Gupta, A; Ravetch, JV. The role of IgG Fc receptors in antibody-dependent enhancement. Nat Rev Immunol. 2020, 20(10), 633–643. [Google Scholar] [CrossRef] [PubMed]
- Sepúlveda-Delgado, J; Llorente, L; Hernández-Doño, S. A Comprehensive Review of Fc Gamma Receptors and Their Role in Systemic Lupus Erythematosus. Int J Mol Sci. 2025, 26(5), 1851. [Google Scholar] [CrossRef] [PubMed]
- Halstead, SB; Mahalingam, S; Marovich, MA; Ubol, S; Mosser, DM. Intrinsic antibody-dependent enhancement of microbial infection in macrophages: disease regulation by immune complexes. Lancet Infect Dis. 2010, 10(10), 712–22. [Google Scholar] [CrossRef]
- Wang, TT; Sewatanon, J; Memoli, MJ; Wrammert, J; Bournazos, S; Bhaumik, SK; Pinsky, BA; Chokephaibulkit, K; Onlamoon, N; Pattanapanyasat, K; Taubenberger, JK; Ahmed, R; Ravetch, JV. IgG antibodies to dengue enhanced for FcγRIIIA binding determine disease severity. Science 2017, 355(6323), 395–398. [Google Scholar] [CrossRef]
- Peiris, JS; Gordon, S; Unkeless, JC; Porterfield, JS. Monoclonal anti-Fc receptor IgG blocks antibody enhancement of viral replication in macrophages. Nature 1981, 289(5794), 189–91. [Google Scholar] [CrossRef]
- Moi, ML; Lim, CK; Takasaki, T; Kurane, I. Involvement of the Fc gamma receptor IIA cytoplasmic domain in antibody-dependent enhancement of dengue virus infection. J Gen Virol. 2010, 91 Pt 1, 103–11. [Google Scholar] [CrossRef]
- Moi, ML; Lim, CK; Kotaki, A; Takasaki, T; Kurane, I. Development of an antibody-dependent enhancement assay for dengue virus using stable BHK-21 cell lines expressing Fc gammaRIIA. J Virol Methods 2010, 163(2), 205–9. [Google Scholar] [CrossRef]
- Moi, ML; Lim, CK; Kotaki, A; Takasaki, T; Kurane, I. Discrepancy in dengue virus neutralizing antibody titers between plaque reduction neutralizing tests with Fcgamma receptor (FcgammaR)-negative and FcgammaR-expressing BHK-21 cells. Clin Vaccine Immunol. 2010, 17(3), 402–7. [Google Scholar] [CrossRef]
- Moi, ML; Lim, CK; Kotaki, A; Takasaki, T; Kurane, I. Detection of higher levels of dengue viremia using FcγR-expressing BHK-21 cells than FcγR-negative cells in secondary infection but not in primary infection. J Infect Dis. 2011, 203(10), 1405–14. [Google Scholar] [CrossRef]
- Moi, ML; Lim, CK; Chua, KB; Takasaki, T; Kurane, I. Dengue virus infection-enhancing activity in serum samples with neutralizing activity as determined by using FcγR-expressing cells. PLoS Negl Trop Dis. 2012, 6(2), e1536. [Google Scholar] [CrossRef]
- Moi, ML; Takasaki, T; Saijo, M; Kurane, I. Dengue virus infection-enhancing activity of undiluted sera obtained from patients with secondary dengue virus infection. Trans R Soc Trop Med Hyg. 2013, 107(1), 51–8. [Google Scholar] [CrossRef]
- Moi, ML; Takasaki, T; Saijo, M; Kurane, I. Determination of antibody concentration as the main parameter in a dengue virus antibody-dependent enhancement assay using FcγR-expressing BHK cells. Arch Virol. 2014, 159(1), 103–16. [Google Scholar] [CrossRef]
- Moi, ML; Takasaki, T; Kurane, I. Detection of Virus-Antibody Immune Complexes in Secondary Dengue Virus Infection. Methods Mol Biol. 2018, 1604, 331–337. [Google Scholar]
- Moi, ML; Ami, Y; Shirai, K; Lim, CK; Suzaki, Y; Saito, Y; Kitaura, K; Saijo, M; Suzuki, R; Kurane, I; Takasaki, T. Formation of infectious dengue virus-antibody immune complex in vivo in marmosets (Callithrix jacchus) after passive transfer of anti-dengue virus monoclonal antibodies and infection with dengue virus. Am J Trop Med Hyg. 2015, 92(2), 370–6. [Google Scholar] [CrossRef] [PubMed]
- Moi, ML; Takasaki, T; Omatsu, T; Nakamura, S; Katakai, Y; Ami, Y; Suzaki, Y; Saijo, M; Akari, H; Kurane, I. Demonstration of marmosets (Callithrix jacchus) as a non-human primate model for secondary dengue virus infection: high levels of viraemia and serotype cross-reactive antibody responses consistent with secondary infection of humans. J Gen Virol. 2014, 95 Pt 3, 591–600. [Google Scholar] [CrossRef]
- Moi, ML; Ami, Y; Muhammad Azami, NA; Shirai, K; Yoksan, S; Suzaki, Y; Kitaura, K; Lim, CK; Saijo, M; Suzuki, R; Takasaki, T; Kurane, I. Marmosets (Callithrix jacchus) as a non-human primate model for evaluation of candidate dengue vaccines: induction and maintenance of specific protective immunity against challenges with clinical isolates. J Gen Virol. 2017, 98(12), 2955–2967. [Google Scholar] [CrossRef]
- Ly, MHP; Moi, ML; Vu, TBH; Tun, MMN; Saunders, T; Nguyen, CN; Nguyen, AKT; Nguyen, HM; Dao, TH; Pham, DQ; Nguyen, TTT; Le, TQM; Hasebe, F; Morita, K. Dengue virus infection-enhancement activity in neutralizing antibodies of healthy adults before dengue season as determined by using FcγR-expressing cells. BMC Infect Dis. 2018, 18(1), 31. [Google Scholar] [CrossRef] [PubMed]
- Saito, Y; Moi, ML; Takeshita, N; Lim, CK; Shiba, H; Hosono, K; Saijo, M; Kurane, I; Takasaki, T. Japanese encephalitis vaccine-facilitated dengue virus infection-enhancement antibody in adults. BMC Infect Dis. 2016, 16(1), 578. [Google Scholar] [CrossRef] [PubMed]
- Mady, BJ; Kurane, I; Erbe, DV; Fanger, MW; Ennis, FA. Neuraminidase augments Fc gamma receptor II-mediated antibody-dependent enhancement of dengue virus infection. J Gen Virol. 1993, 74 Pt 5, 839–44. [Google Scholar] [CrossRef]
- Boonnak, K; Dambach, KM; Donofrio, GC; Tassaneetrithep, B; Marovich, MA. Cell type specificity and host genetic polymorphisms influence antibody-dependent enhancement of dengue virus infection. J Virol. 2011, 85(4), 1671–83. [Google Scholar] [CrossRef] [PubMed]
- Boonnak, K; Slike, BM; Donofrio, GC; Marovich, MA. Human FcγRII cytoplasmic domains differentially influence antibody-mediated dengue virus infection. J Immunol. 2013, 190(11), 5659–65. [Google Scholar] [CrossRef] [PubMed]
- Callaway, JB; Smith, SA; McKinnon, KP; de Silva, AM; Crowe, JE, Jr.; Ting, JP. Spleen Tyrosine Kinase (Syk) Mediates IL-1β Induction by Primary Human Monocytes during Antibody-enhanced Dengue Virus Infection. J Biol Chem. 2015, 290(28), 17306–20. [Google Scholar] [CrossRef]
- Chareonsirisuthigul, T; Kalayanarooj, S; Ubol, S. Dengue virus (DENV) antibody-dependent enhancement of infection upregulates the production of anti-inflammatory cytokines but suppresses anti-DENV free radical and pro-inflammatory cytokine production, in THP-1 cells. J Gen Virol. 2007, 88 Pt 2, 365–375. [Google Scholar] [CrossRef] [PubMed]
- Suhrbier, A; La Linn, M. Suppression of antiviral responses by antibody-dependent enhancement of macrophage infection. Trends Immunol. 2003, 24(4), 165–8. [Google Scholar] [CrossRef]
- Ong, EZ; Zhang, SL; Tan, HC; Gan, ES; Chan, KR; Ooi, EE. Dengue virus compartmentalization during antibody-enhanced infection. Sci Rep. 2017, 7, 40923. [Google Scholar] [CrossRef]
- Ayala-Nunez, NV; Hoornweg, TE; van de Pol, DP; Sjollema, KA; Flipse, J; van der Schaar, HM; Smit, JM. How antibodies alter the cell entry pathway of dengue virus particles in macrophages. Sci Rep. 2016, 6, 28768. [Google Scholar] [CrossRef]
- Chotiwan, N; Roehrig, JT; Schlesinger, JJ; Blair, CD; Huang, CY. Molecular determinants of dengue virus 2 envelope protein important for virus entry in FcγRIIA-mediated antibody-dependent enhancement of infection. Virology 2014, 456-457, 238–46. [Google Scholar] [CrossRef]
- Nakamura, Y; Moi, ML; Shiina, T; Shin-I, T; Suzuki, R. Idiotope-Driven T-Cell/B-Cell Collaboration-Based T-Cell Epitope Prediction Using B-Cell Receptor Repertoire Sequences in Infectious Diseases. Viruses 2023, 15(5), 1186. [Google Scholar] [CrossRef]
- Nguyen, TTN; Ngwe Tun, MM; Vu, TBH; Nguyen, TTT; Hoang, VMP; Nguyen, LKH; Phan, TL; Dang, DA; Raekiansyah, M; Suzuki, R; Balingit, JC; Takamatsu, Y; Buerano, CC; Urano, T; Le, TQM; Hasebe, F; Morita, K. Implications of immune responses to DENV, JEV, and ZIKV Infections for cross-reactivity and considerations for vaccine evaluation. Virus Res. 2026, 364, 199689. [Google Scholar] [CrossRef]
- Balingit, JC; Denis, D; Suzuki, R; Hayati, RF; Ngwe Tun, MM; Takamatsu, Y; Masyeni, S; Sasmono, RT; Morita, K. Impact of pre-existing cross-reactive antibodies on cyclic dengue outbreaks in the hyperendemic region of Bali, Indonesia. Virus Res. 2024, 348, 199445. [Google Scholar] [CrossRef]
- Urakami, A; Ngwe Tun, MM; Moi, ML; Sakurai, A; Ishikawa, M; Kuno, S; Ueno, R; Morita, K; Akahata, W. An Envelope-Modified Tetravalent Dengue Virus-Like-Particle Vaccine Has Implications for Flavivirus Vaccine Design. J Virol. 2017, 91(23), e01181-17. [Google Scholar] [CrossRef]
- Thoresen, D; Matsuda, K; Urakami, A; Ngwe Tun, MM; Nomura, T; Moi, ML; Watanabe, Y; Ishikawa, M; Hau, TTT; Yamamoto, H; Suzaki, Y; Ami, Y; Smith, JF; Matano, T; Morita, K; Akahata, W. A tetravalent dengue virus-like particle vaccine induces high levels of neutralizing antibodies and reduces dengue replication in non-human primates. J Virol. 2024, 98(5), e0023924. [Google Scholar] [CrossRef]
- Yamanaka, A; Moi, ML; Takasaki, T; Kurane, I; Matsuda, M; Suzuki, R; Konishi, E. Utility of Japanese encephalitis virus subgenomic replicon-based single-round infectious particles as antigens in neutralization tests for Zika virus and three other flaviviruses. J Virol Methods 2017, 243, 164–171. [Google Scholar] [CrossRef]
- Haga, K; Chen, ZN; Himeno, M; Majima, R; Moi, ML. Utility of an In-Vitro Micro-Neutralizing Test in Comparison to a Plaque Reduction Neutralization Test for Dengue Virus, Japanese Encephalitis Virus, and Zika Virus Serology and Drug Screening. Pathogens 2023, 13(1), 8. [Google Scholar] [CrossRef]
- Prajapati, S; Elong Ngono, A; Mc Cauley, M; Timis, J; Shrestha, S; Bastola, A; Mandal, SK; Ray Yadav, S; Napit, R; Moi, ML; Yamabhai, M; M Sessions, O; Shresta, S; Manandhar, KD. Genomic sequencing and neutralizing serological profiles during acute dengue infection: A 2017 cohort study in Nepal. PLOS Glob Public Health 2024, 4(11), e0002966. [Google Scholar] [CrossRef] [PubMed]
- Balingit, JC; Dimamay, MPS; Suzuki, R; Matsuda, M; Xayavong, D; Ngwe Tun, MM; Matias, RR; Natividad, FF; Moi, ML; Takamatsu, Y; Culleton, R; Buerano, CC; Morita, K. Role of pre-existing immunity in driving the dengue virus serotype 2 genotype shift in the Philippines: A retrospective analysis of serological data. Int J Infect Dis. 2024, 139, 59–68. [Google Scholar] [CrossRef]
- Balingit, JC; Abe, M; Suzuki, R; Xayavong, D; Ngwe Tun, MM; Takamatsu, Y; Sonoda, K; Morita, K. Cross-genotype immunogenicity and antibody-dependent enhancement of KD-382 dengue vaccine in flavivirus-naïve adults. NPJ Vaccines 2025, 10(1), 148. [Google Scholar] [CrossRef]
- Thomas, S; Smatti, MK; Ouhtit, A; Cyprian, FS; Almaslamani, MA; Thani, AA; Yassine, HM. Antibody-Dependent Enhancement (ADE) and the role of complement system in disease pathogenesis. Mol Immunol. 2022, 52, 172–182. [Google Scholar] [CrossRef] [PubMed]
- Okuya, K; Hattori, T; Saito, T; Takadate, Y; Sasaki, M; Furuyama, W; Marzi, A; Ohiro, Y; Konno, S; Hattori, T; Takada, A. Multiple Routes of Antibody-Dependent Enhancement of SARS-CoV-2 Infection. Microbiol Spectr 2022, 10(2), e0155321. [Google Scholar] [CrossRef] [PubMed]
- Mehlhop, E; Nelson, S; Jost, CA; Gorlatov, S; Johnson, S; Fremont, DH; Diamond, MS; Pierson, TC. Complement protein C1q reduces the stoichiometric threshold for antibody-mediated neutralization of West Nile virus. Cell Host Microbe 2009, 6(4), 381–91. [Google Scholar] [CrossRef]
- Wegman, AD; Fang, H; Rothman, AL; Thomas, SJ; Endy, TP; McCracken, MK; Currier, JR; Friberg, H; Gromowski, GD; Waickman, AT. Monomeric IgA Antagonizes IgG-Mediated Enhancement of DENV Infection. Front Immunol. 2021, 12, 777672. [Google Scholar] [CrossRef]
- Na, L; Zheng, Y; Yang, JB; Bao, HL; Tang, YD. The neonatal Fc receptor (FcRn): Guardian or Trojan Horse in viral infection? PLoS Pathog 2025, 21(7), e1013285. [Google Scholar] [CrossRef]
- Konishi, E; Tabuchi, Y; Yamanaka, A. A simple assay system for infection-enhancing and -neutralizing antibodies to dengue type 2 virus using layers of semi-adherent K562 cells. J Virol Methods 2010, 163(2), 360–7. [Google Scholar] [CrossRef]
- Yamanaka, A; Rattanaamnuaychai, P; Matsuda, M; Suzuki, R; Shimizu, J; Shioda, T; Miyazaki, K. Development of a rapid assay system for detecting antibody-dependent enhancement of dengue virus infection. J Virol Methods 2023, 311, 114641. [Google Scholar] [CrossRef]
- Chelluboina, S; Kshirsagar, D; Panzade, G; Mishra, AC; Arankalle, V; Shrivastava, S. Evaluation of methods for the measurement of antibody-dependent enhancement of dengue virus infection using different FcγRIIa expressing cell lines. PLoS One 2025, 20(8), e0331320. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, JK; Mastrodomenico, V; Hartnett, J; Heelan, WJ; Garvin, D; Cong, M; Grailer, JJ. A HiBiT-tagged pseudovirus-like particle platform for safe, rapid quantification of virus neutralization and antibody-dependent enhancement. J Virol. 2025, 99(11), e0099125. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y; Shang, J; Sun, S; Tai, W; Chen, J; Geng, Q; He, L; Chen, Y; Wu, J; Shi, Z; Zhou, Y; Du, L; Li, F. Molecular Mechanism for Antibody-Dependent Enhancement of Coronavirus Entry. J Virol. 2020, 94(5), e02015-19. [Google Scholar] [CrossRef] [PubMed]
- Furuyama, W; Marzi, A; Carmody, AB; Maruyama, J; Kuroda, M; Miyamoto, H; Nanbo, A; Manzoor, R; Yoshida, R; Igarashi, M; Feldmann, H; Takada, A. Fcγ-receptor IIa-mediated Src Signaling Pathway Is Essential for the Antibody-Dependent Enhancement of Ebola Virus Infection. PLoS Pathog 2016, 12(12), e1006139. [Google Scholar] [CrossRef]
- Jaume, M; Yip, MS; Cheung, CY; Leung, HL; Li, PH; Kien, F; Dutry, I; Callendret, B; Escriou, N; Altmeyer, R; Nal, B; Daëron, M; Bruzzone, R; Peiris, JS. Anti-severe acute respiratory syndrome coronavirus spike antibodies trigger infection of human immune cells via a pH- and cysteine protease-independent FcγR pathway. J Virol. 2011, 85(20), 10582–97. [Google Scholar] [CrossRef]
- WHO Expert Committee on Biological Standardization (ECBS). Clinical Evaluation of Dengue Vaccines: Amendments in Q&A and Guidelines; WHO/HIS/EMP/TSN, 15 October 2014. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



