Submitted:
06 February 2026
Posted:
11 February 2026
You are already at the latest version
Abstract
Keywords:
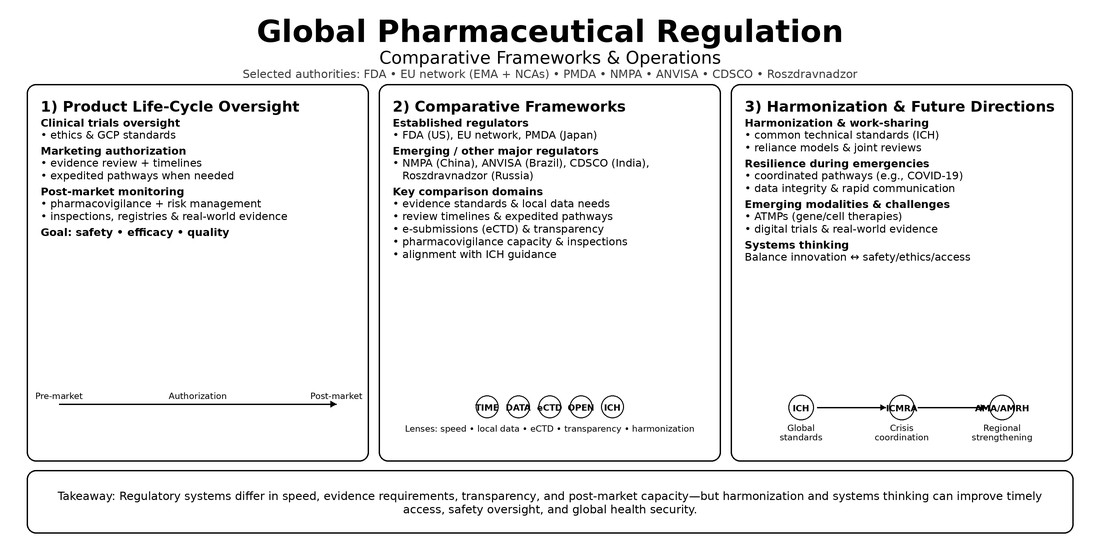
1. Introduction
2. Global Regulatory Landscape
2.1. Global Regulatory Authorities
2.1.1. Established Major Regulatory Authorities
- The US Food and Drug Administration (FDA)
- 2.
- Europe’s medicines regulatory network: the European Medicines Agency (EMA) and national competent authorities (NCAs).
- 3.
- Japan's Pharmaceuticals and Medical Devices Agency (PMDA)
2.1.2. Emerging Major Regulatory Authorities
- China's National Medical Products Administration (NMPA)
- 2.
- Brazil's National Health Surveillance Agency (ANVISA)
- 3.
- India's Central Drugs Standard Control Organization (CDSCO)
- 4.
- Russia's Federal Service for Surveillance in Healthcare (Roszdravnadzor)
2.1.3. Other Regulatory Authorities
- Argentina's National Administration of Drugs, Food and Medical Technology (ANMAT)
- 2.
- Australia's Therapeutic Goods Administration (TGA)
- 3.
- Selected African Regulatory Agencies (NAFDAC, SAHPRA, TMDA, EDA, Ghana’s FDA, MCAZ, ARP, Rwanda’s FDA)
- Nigeria (NAFDAC) – National Agency for Food and Drug Administration and Control
- South Africa (SAHPRA) - South African Health Products Regulatory Authority
- Tanzania (TMDA) - Tanzania Medicines and Medical Devices Authority
- Egypt (EDA) - Egyptian Drug Authority
- Ghana (FDA) – Food and Drugs Authority
- Zimbabwe (MCAZ) - Medicines Control Authority of Zimbabwe
- Senegal (ARP) - Agence Sénégalaise de Réglementation Pharmaceutique
- Rwanda (FDA) - Food and Drugs Authority
2.2. Harmonization Initiatives and Comparative Regulatory Frameworks
2.2.1. Global Harmonization Efforts
2.2.2. Regional Harmonization Networks
2.3. Comparative Analysis of Regulatory Authorities
3. Drug Development and Approval Process
3.1. Clinical Trial Oversight
3.1.1. Global Clinical Trial Standards
3.1.2. Innovation in Clinical Trial Regulation
3.2. Review and Approval Mechanisms
3.3. Challenges in Multinational Clinical Trial Regulations and Potential Solutions
4. Post-Market Safety Monitoring
4.1. Pharmacovigilance Systems
Internationally-recognized Pharmacovigilance (PV) Systems
4.2. Risk Management and Mitigation
4.2.1. Risk Management Plan (RMP)
4.2.2. Risk Evaluation and Mitigation Strategy (REMS)
5. Emerging Challenges and Future Directions
5.1. Advanced Therapy Medicinal Products (ATMPs)
5.2. Global Health Emergencies
5.2.1. Public Health Emergency of International Concern (PHEIC)
5.2.2. Examples of Major Scenarios of Global Health Emergencies
- Case Type 1: Quality or safety issues with medicines on the market that affect public health across multiple countries [31]. For example, investigating widespread problems with medication that could be causing harm.
- Case Type 2: Shortage of approved medicines during crises, where previously available products become scarce or unavailable due to supply problems [31]. A clear example is a viral outbreak where antiviral treatments or vaccines are in short supply.
- Case Type 3: Urgent need for new treatments or vaccines during emerging health threats such as the COVID-19 pandemic. These situations typically arise when the WHO declares a Public Health Emergency, prompting national health authorities and WHO to act using existing international health guidelines [31].
5.3. Case Studies in Regulatory Innovation
COVID-19 Vaccine Development
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADR | Adverse Drug Reaction |
| AMA | African Medicines Agency |
| BLA | Biologics License Application |
| CHMP | Committee for Medicinal Products for Human Use |
| CIRS | Center for Innovation in Regulatory Science |
| EUL | Emergency Use Listing |
| IND | Investigational New Drug |
| HCT/Ps | Cellular and Tissue-Based Products |
| CFR | Code of Federal Regulations |
| IND | Investigational New Drug |
| DCGI | Drugs Controller General of India |
| GMP | Good Manufacturing Practice |
| NDA | New Drug Application |
| PV | Pharmacovigilance |
| RA | Regulatory Affairs |
| REMS | Risk Evaluation and Mitigation Strategy |
| RMP | Risk Management Plan |
| RMAT | Regenerative Medicine Advanced Therapy |
| RWE | Real-World Evidence |
| WHO | World Health Organization |
References
- Rupnawar, M., and P. Wani. 2024. “Pharmaceutical Regulatory Affairs: An Overview of Global Regulatory Frameworks and Emerging Trends.” International Journal of Scientific Research and Technology 2 (12): 1–1. [CrossRef]
- Hussaarts, L., S. Mühlebach, V. P. Shah, et al. 2017. “Equivalence of Complex Drug Products: Advances in and Challenges for Current Regulatory Frameworks.” Annals of the New York Academy of Sciences 1407 (1): 39–49. [CrossRef]
- Raut, N., K. Bajaj, P. Matte, et al. 2023. “Building Bridges: Harmonization Efforts for Enhanced Collaboration between Developed and Developing Countries.” International Journal of Drug Regulatory Affairs 11 (3): 68–79. [CrossRef]
- Darrow, J. J., J. Avorn, and A. S. Kesselheim. 2020. “FDA Approval and Regulation of Pharmaceuticals, 1983–2018.” JAMA 323 (2): 164–176. [CrossRef]
- Tobin, J. J., and G. Walsh. 2023. Medical Product Regulatory Affairs: Pharmaceuticals, Diagnostics, Medical Devices. Hoboken, NJ: John Wiley & Sons.
- Gherghescu, I., and M. B. Delgado-Charro. 2021. “The Biosimilar Landscape: An Overview of Regulatory Approvals by the EMA and FDA.” Pharmaceutics 13 (1): 48. [CrossRef]
- Muselík, J., A. Komersová, K. Kubová, K. Matzick, and B. Skalická. 2021. “A Critical Overview of FDA and EMA Statistical Methods to Compare In Vitro Drug Dissolution Profiles of Pharmaceutical Products.” Pharmaceutics 13 (10): 1703. [CrossRef]
- Chen, R., R. Manochakian, L. James, et al. 2020. “Emerging Therapeutic Agents for Advanced Non-Small Cell Lung Cancer.” Journal of Hematology & Oncology 13 (1): 58. [CrossRef]
- Ali, F., A. Ilyas, and S. Ahmadeen. 2024. “Regulations in Japan.” In Global Regulations of Medicinal, Pharmaceutical, and Food Products. Boca Raton, FL: CRC Press.
- Clinical Stage. n.d. “Regulatory Differences across Regions.” Accessed March 20, 2025. https://www.clinicalstage.net/regulatory-differences-across-regions.
- US Food and Drug Administration (FDA). 2019. “Step 4: FDA Drug Review.” April 18, 2019. Accessed January 17, 2025. https://www.fda.gov/patients/drug-development-process/step-4-fda-drug-review.
- Van Norman, G. A. 2016. “Drugs and Devices: Comparison of European and U.S. Approval Processes.” JACC: Basic to Translational Science 1 (5): 399–412. [CrossRef]
- Hussain, F. 2013. “Kefauver-Harris Drug Amendments 1962.” Pharma Mirror Magazine, December 7. Accessed January 20, 2025. https://www.pharmamirror.com/knowledge-base/pharmaceutical-dictionary/kefauver-harris-drug-amendments-1962/.
- US Food and Drug Administration (FDA). 2018. Federal Food, Drug, and Cosmetic Act (FD&C Act). November 3, 2018. Accessed January 20, 2025. https://www.fda.gov/regulatory-information/laws-enforced-fda/federal-food-drug-and-cosmetic-act-fdc-act.
- US Food and Drug Administration (FDA). 2024a. “What We Do.” August 9, 2024. Accessed January 20, 2025. https://www.fda.gov/about-fda/what-we-do.
- Wang, W., and A. I. Wertheimer. 2022. “History, Status, and Politicization of the FDA.” Research in Social and Administrative Pharmacy 18 (5): 2811–2816. [CrossRef]
- Finnish Medicines Agency (Fimea). n.d.-a. “Pharmaceutical safety and information.” Accessed January 12, 2026. https://fimea.fi/en/pharmaceutical_safety_and_information.
- European Medicines Agency (EMA). 2015. “Adaptive Pathways.” November 20, 2015. Accessed January 21, 2025. https://www.ema.europa.eu/en/human-regulatory-overview/research-development/adaptive-pathways.
- European Medicines Agency (EMA). n.d.-b. “Marketing authorisation.” Accessed January 12, 2026. https://www.ema.europa.eu/en/human-regulatory-overview/marketing-authorisation.
- European Medicines Agency (EMA). n.d.-c. “National competent authorities (human).” Accessed January 12, 2026. https://www.ema.europa.eu/en/partners-networks/eu-partners/eu-member-states/national-competent-authorities-human.
- European Medicines Agency (EMA). 2019b. “Authorization of Medicines.” March 4, 2019. Accessed January 17, 2025. https://www.ema.europa.eu/en/about-us/what-we-do/authorisation-medicines.
- European Medicines Agency (EMA). n.d.-d. “Pharmacovigilance: Overview.” Accessed January 12, 2026. https://www.ema.europa.eu/en/human-regulatory-overview/pharmacovigilance-overview.
- Finnish Medicines Agency (Fimea). n.d.-b. “Pharmacovigilance.” Accessed January 12, 2026. https://fimea.fi/en/supervision/pharmacovigilance.
- Kanta Services. 2025a. “Prescriptions and medicines.” February 6, 2025. Accessed January 12, 2026. https://www.kanta.fi/en/prescriptions-and-medicines.
- Kanta Services. 2025b. “Prescription.” February 18, 2025. Accessed January 12, 2026. https://www.kanta.fi/en/professionals/prescription.
- Swedish Medical Products Agency (Läkemedelsverket). n.d. “About the Swedish Medical Products Agency.” Accessed January 12, 2026. https://www.lakemedelsverket.se/en/about-the-swedish-mpa.
- Socialstyrelsen (National Board of Health and Welfare). 2020. “National Prescribed Drug Register.” August 21, 2020. Accessed January 12, 2026. https://www.socialstyrelsen.se/en/statistics-and-data/registers/national-prescribed-drug-register/.
- Pharmaceuticals and Medical Devices Agency (PMDA). n.d.-a. “Outline of PMDA.” Accessed January 21, 2025. https://www.pmda.go.jp/english/about-pmda/outline/0005.html.
- Pharmaceuticals and Medical Devices Agency (PMDA). n.d.-b. “History.” Accessed January 21, 2025. https://www.pmda.go.jp/english/about-pmda/outline/0002.html.
- Wileman, H., and A. Mishra. 2010. “Drug Lag and Key Regulatory Barriers in the Emerging Markets.” Perspectives in Clinical Research 1 (2): 51–56. Accessed March 20, 2025. https://journals.lww.com/picp/fulltext/2010/01020/Drug_Lag_and_Key_Regulatory_Barriers_in_the.2.aspx.
- International Coalition of Medicines Regulatory Authorities (ICMRA). n.d. “Framework for the Involvement of Health Regulatory Authorities in the Management of Global Health Crises.” Accessed January 17, 2025. https://www.icmra.info/drupal/sites/default/files/2019-04/ICMRA%20SOP%20for%20Crisis%20Management.pdf.
- Ferreira, C. G. 2019. “ES10.05 Brazilian Health Regulatory Agency – Agência Nacional de Vigilância Sanitária.” Journal of Thoracic Oncology 14 (10): S39. [CrossRef]
- Huynh-Ba, K., and A. Beumer Sassi. 2018. “ANVISA: An Introduction to a New Regulatory Agency with Many Challenges.” AAPS Open 4 (1): 9. [CrossRef]
- Dhole, S. D., and R. B. Darade. 2024. “Indian Pharmaceutical Regulatory Authority: Review.” International Journal in Pharmaceutical Sciences 2 (5): 1109–1119. [CrossRef]
- Central Drugs Standard Control Organization (CDSCO). 2025. “Introduction.” Accessed January 21, 2025. https://cdsco.gov.in/opencms/opencms/hi/About-us/Introduction/.
- Federal Service for Surveillance in Healthcare. n.d. “Regulatory Framework for Federal Service for Surveillance in Healthcare.” Accessed January 21, 2025. https://roszdravnadzor.gov.ru/en.
- Vargas-Pelaez, C. M., M. T. Drago, A. Acosta, and M. R. Farias. 2017. “Pharmaceutical Policy in Argentina.” In Pharmaceutical Policy in Countries with Developing Healthcare Systems, 97–121. [CrossRef]
- Argentina.gob.ar. 2025. “What Is ANMAT?” Accessed March 20, 2025. https://www.argentina.gob.ar/anmat/anmat-en/what-anmat.
- Skerritt, J. 2022. “Australia’s Robust Strategy for Regional and Global Medical Product Engagement.” U.S. Food and Drug Administration. September 20, 2022. Accessed March 20, 2025. https://www.fda.gov/international-programs/australias-robust-strategy-regional-and-global-medical-product-engagement.
- FHI Clinical. n.d. “Increasing Regulatory Harmonization in Africa Is Streamlining Marketing Authorization of Your Products.” Accessed March 20, 2025. https://fhiclinical.com/increasing-regulatory-harmonization-in-africa/.
- World Health Organization (WHO). 2024. “Senegal and Rwanda Achieve WHO Maturity Level 3 in Medicines Regulation.” December 5, 2024. Accessed March 19, 2025. https://www.who.int/news/item/05-12-2024-senegal-and-rwanda-achieve-who-maturity-level-3-in-medicines-regulation.
- Ozili, P. K. 2021. “Covid-19 Pandemic and Economic Crisis: The Nigerian Experience and Structural Causes.” Journal of Economic and Administrative Sciences 37 (4): 401–418. [CrossRef]
- Olatunji, O. E. 2014. “Leadership Effectiveness and Regulatory Performance in the Public Sector: The Experience of the National Agency for Food and Drug Administration and Control (NAFDAC).” International Journal of Innovative Research & Development 3 (3): 217–225.
- Omijeh, B. O., and O. Raheem. 2021. “An Improved Drug Verification System for NAFDAC.” Accessed March 19, 2025. https://www.africanscholarpublications.com/wp-content/uploads/2021/06/AJASD_Vol20_No2_March_2021-2.pdf.
- Spink, J., D. C. Moyer, and M. R. Rip. 2016. “Addressing the Risk of Product Fraud: A Case Study of the Nigerian Combating Counterfeiting and Sub-standard Medicines Initiatives.” Journal of Forensic Science & Criminology 4 (2): 201.
- National Agency for Food and Drug Administration and Control (NAFDAC). n.d. “About NAFDAC.” Accessed March 20, 2025. https://nafdac.gov.ng/about-nafdac/.
- International Council for Harmonisation (ICH). n.d.-a “Welcome to the ICH Official Website.” Accessed January 21, 2025. https://www.ich.org/.
- Adleberg, J. 2024. “ICH Overview.” U.S. Food & Drug Administration. FDA/HC ICH Regional Public Meeting, February 22, 2024. Accessed January 21, 2025. https://www.fda.gov/media/177713/download.
- Vbnm,International Council for Harmonisation (ICH). n.d.-b. “Management Committee.” Accessed January 21, 2025. https://www.ich.org/page/management-committee.
- Reggi, V. 2017. “Medicines Regulatory Harmonization: International Collaboration as a Key to Improve Public Health.” Medicine Access @ Point of Care 1: maapoc.0000001. [CrossRef]
- Eurofins Scientific. n.d. “Three Key Regulations in the Pharmaceutical Industry: EMA, FDA and ANVISA.” Accessed March 21, 2025. https://www.eurofins.com/assurance/resources/articles/three-key-regulations-in-the-pharmaceutical-industry-ema-fda-and-anvisa/.
- Center for Innovation in Regulatory Science (CIRS). 2021. New Drug Approvals in Six Major Authorities 2011–2020: Focus on Facilitated Regulatory Pathways and Worksharing. https://www.cirsci.org/wp-content/uploads/dlm_uploads/2021/06/CIRS-RD-Briefing-81-6-agencies-v5.pdf.
- Vashisth, S., G. Singh, and A. Nanda. 2012. “A Comparative Study of Regulatory Trends of Pharmaceuticals in Brazil, Russia, India and China (BRIC) Countries.” Journal of Generic Medicines 9 (3): 128–143. [CrossRef]
- Clini India. 2024. “Understanding US FDA vs. EMA vs. CDSCO: How Do They Differ?” Accessed March 21, 2024. https://www.cliniindia.com/understanding-us-fda-vs-ema-vs-cdsco-how-do-they-differ/.
- National Medical Products Administration (NMPA). 2025. “Measures Facilitate Approval of 48 First-in-Class Innovative Drugs.” March 21, 2025. Accessed May 6, 2025. https://english.nmpa.gov.cn/2025-03/21/c_1080286.htm.
- Agência Nacional de Vigilância Sanitária (Anvisa). n.d. “Drugs.” Accessed May 6, 2025. https://www.gov.br/anvisa/pt-br/english/regulation-of-products/drugs.
- Deepika, M., and J. Karthi. 2025. “A Comparative Study of USFDA, EMA, and CDSCO Regulatory Guidelines.” International Journal of Research Publication and Reviews 6 (3): 1429–1431. https://ijrpr.com/uploads/V6ISSUE3/IJRPR39725.pdf.
- “Exploring CDSCO’s Approval Process for New Drugs.” n.d. Issuu. Accessed May 6, 2025. https://issuu.com/corpseedgroup/docs/exploring_cdsco_s_approval_process_for_new_drugs.
- US Food and Drug Administration (FDA). 2025. “FDA Acceptance of Foreign Clinical Studies Not Conducted under an IND: Frequently Asked Questions.” February 3, 2025. Accessed May 6, 2025. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/fda-acceptance-foreign-clinical-studies-not-conducted-under-ind-frequently-asked-questions.
- Naito, C. 1998. “Ethnic Factors in the Acceptability of Foreign Clinical Data.” Drug Information Journal 32 (1, suppl): 1283S–1292S. [CrossRef]
- National Medical Products Administration (NMPA). 2020. “Clinical Technical Requirements for Drugs Marketed Overseas but Not Marketed in China.” November 18, 2020. Accessed May 7, 2025. https://english.nmpa.gov.cn/2020-11/18/c_568155.htm.
- US Department of Health and Human Services. 2018. “Read the Belmont Report.” January 15, 2018. Accessed January 17, 2025. https://www.hhs.gov/ohrp/regulations-and-policy/belmont-report/read-the-belmont-report/index.html.
- Abou-El-Enein, M., A. Elsanhoury, and P. Reinke. 2016. “Overcoming Challenges Facing Advanced Therapies in the EU Market.” Cell Stem Cell 19 (3): 293–297. [CrossRef]
- International Council for Harmonisation (ICH). 2016. Guideline for Good Clinical Practice E6(R2). Accessed January 16, 2025. https://database.ich.org/sites/default/files/E6_R2_Addendum.pdf.
- Council for International Organizations of Medical Sciences, and World Health Organization, eds. 2002. International Ethical Guidelines for Biomedical Research Involving Human Subjects. Geneva: CIOMS.
- Rosa, C., L. A. Marsch, E. L. Winstanley, M. Brunner, and A. N. C. Campbell. 2021. “Using Digital Technologies in Clinical Trials: Current and Future Applications.” Contemporary Clinical Trials 100: 106219. [CrossRef]
- US Food and Drug Administration (FDA). 2024b. “FDA Promotes Clinical Trial Innovation.” FDA Voices. August 9, 2024. Accessed January 17, 2025. https://www.fda.gov/news-events/fda-voices/fda-promotes-clinical-trial-innovation.
- Scheppler, L., N. De Clercq, M. McGoldrick, and J. Dias. 2021. “Regulatory Harmonization and Streamlining of Clinical Trial Applications Globally Should Lead to Faster Clinical Development and Earlier Access to Life-Saving Vaccines.” Vaccine 39 (5): 790–796. [CrossRef]
- Hwenda, L., M. Sidibe, and M. Makanga. 2022. “The African Medicines Agency: The Key to Unlocking Clinical Research in Africa.” Lancet Global Health 10 (8): e1088–e1089. [CrossRef]
- Jackson, C. 2020. “Africa’s Missing Genomic Data and Its Impact on Health Care.” GEN – Genetic Engineering and Biotechnology News, September 8, 2020. Accessed January 17, 2025. https://www.genengnews.com/topics/omics/africas-missing-genomic-data-and-its-impact-on-health-care/.
- Khan, M. A. A., S. Hamid, and Z. U. D. Babar. 2023. “Pharmacovigilance in High-Income Countries: Current Developments and a Review of Literature.” Pharmacy 11 (1): 10. [CrossRef]
- Cui, X., L. X. Wang, G. Y. Liu, and Y. M. Xie. 2021. “Enlightenment of International Pharmacovigilance System on Establishment of Pharmacovigilance System of Chinese Medicine.” China Journal of Chinese Materia Medica 46: 5450–5455. [CrossRef]
- World Health Organization (WHO). 2020. 72nd and 73rd Report: WHO Technical Report Series No. 1030. Accessed May 7, 2025. https://www.who.int/publications/i/item/9789240024373.
- Dieck, G. S., and R. G. Sharrar. 2013. “Preparing for Safety Issues Following Drug Approval: Pre-Approval Risk Management Considerations.” Therapeutic Advances in Drug Safety 4 (5): 220–228. [CrossRef]
- US Food and Drug Administration (FDA). 2023b. “Risk Evaluation and Mitigation Strategies (REMS).” May 16, 2023. Accessed January 17, 2025. https://www.fda.gov/drugs/drug-safety-and-availability/risk-evaluation-and-mitigation-strategies-rems.
- Allison, S. J. 2022. “Novel Anti-Cancer Agents and Cellular Targets and Their Mechanism(s) of Action.” Biomedicines 10 (8): 1767. [CrossRef]
- Rothschild, J. 2020. “Ethical Considerations of Gene Editing and Genetic Selection.” Journal of General and Family Medicine 21 (3): 37–47. [CrossRef]
- “China Grants Conditional Approval for First COVID Vaccine.” 2020. National Health Commission of the People’s Republic of China. December 31, 2020. Accessed May 7, 2025. https://en.nhc.gov.cn/2020-12/31/c_82609.htm.
- Hanna, E., C. Rémuzat, P. Auquier, and M. Toumi. 2016. “Advanced Therapy Medicinal Products: Current and Future Perspectives.” Journal of Market Access & Health Policy 4: 31036. [CrossRef]
- Cuende, N., J. E. J. Rasko, M. B. C. Koh, M. Dominici, and L. Ikonomou. 2018. “Cell, Tissue and Gene Products with Marketing Authorization in 2018 Worldwide.” Cytotherapy 20 (11): 1401–1413. [CrossRef]
- International Council for Harmonisation (ICH). n.d.-c. “Mission.” Accessed May 7, 2025. https://www.ich.org/page/mission.
- US Food and Drug Administration (FDA). n.d.-a. “Regulation of Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps).” Accessed May 7, 2025. https://www.fda.gov/vaccines-blood-biologics/cellular-gene-therapy-products.
- US Food and Drug Administration (FDA). n.d.-b. “Regenerative Medicine Advanced Therapy (RMAT) Designation.” Accessed May 7, 2025. https://www.fda.gov/vaccines-blood-biologics/cellular-gene-therapy-products/regenerative-medicine-advanced-therapy-designation.
- European Medicines Agency (EMA). 2024. “Advanced Therapy Medicinal Products: Overview.” Updated March 2024. Accessed May 7, 2025. https://www.ema.europa.eu/en/human-regulatory/overview/advanced-therapy-medicinal-products-overview.
- Pharmaceuticals and Medical Devices Agency (PMDA). n.d.-d. “Review of Regenerative Medical Products.” Accessed May 7, 2025. https://www.pmda.go.jp/english/review-services/reviews/0003.html.
- National Health Surveillance Agency – Anvisa. 2020. “10154json-file-1.pdf.” Accessed May 7, 2025. https://www.gov.br/anvisa/pt-br/assuntos/reunioesdadicol/processos-deliberados/5de2020/arquivos/10154json-file-1/view.
- Brazilian Health Regulatory Agency (Anvisa). 2023. “Anvisa Approves Specific Good Manufacturing Practice Regulation for Advanced Therapy Products.” 2023. Accessed May 7, 2025. https://www.gov.br/anvisa/pt-br/assuntos/noticias-anvisa/2023/anvisa-aprova-norma-especifica-de-boas-praticas-de-fabricacao-para-produtos-de-terapias-avancadas.
- Roszdravnadzor. n.d. “Federal Law No. 180-FZ on Biomedical Cell Products.” Accessed May 7, 2025. https://www.consultant.ru/document/cons_doc_LAW_200349/.
- Jotwani, G. 2017. National Guidelines for Stem Cell Research. Indian Council of Medical Research and Department of Biotechnology. https://dbtindia.gov.in/sites/default/files/National_Guidelines_StemCellResearch-2017.pdf.
- Yin, C., J. Gao, G. Li, et al. 2022. “Gene and Cell Therapies in China: Booming Landscape under Dual-Track Regulation.” Journal of Hematology & Oncology 15 (1): 139. [CrossRef]
- Li, Y., F. Verter, B. Wang, and N. Gu. 2019. “Regulations on Cell Therapy Products in China: A Brief History and Current Status.” Regenerative Medicine 14 (8): 791–803. [CrossRef]
- International Coalition of Medicines Regulatory Authorities (ICMRA). 2016. “International Coalition of Medicines Regulatory Authorities (ICMRA).” European Medicines Agency. October 13, 2016. Accessed January 17, 2025. https://www.ema.europa.eu/en/partners-networks/international-activities/multilateral-coalitions-initiatives/international-coalition-medicines-regulatory-authorities-icmra.
- World Health Organization (WHO). 2021. “Joint Statement on Transparency and Data Integrity: International Coalition of Medicines Regulatory Authorities (ICMRA) and WHO.” May 7, 2021. Accessed January 17, 2025. https://www.who.int/news/item/07-05-2021-joint-statement-on-transparency-and-data-integrityinternational-coalition-of-medicines-regulatory-authorities-(icmra)-and-who.
- World Health Organization (WHO). 2019. “Emergencies: International Health Regulations and Emergency Committees.” December 19, 2019. Accessed January 22, 2025. https://www.who.int/news-room/questions-and-answers/item/emergencies-international-health-regulations-and-emergency-committees.
- Tanne, J. H. 2021. “Covid-19: FDA Approves Pfizer-BioNTech Vaccine in Record Time.” BMJ 374: n2096. [CrossRef]
- US Food and Drug Administration (FDA). 2020. “FDA Takes Key Action in Fight Against COVID-19 by Issuing Emergency Use Authorization for First COVID-19 Vaccine.” December 14, 2020. Accessed January 17, 2025. https://www.fda.gov/news-events/press-announcements/fda-takes-key-action-fight-against-covid-19-issuing-emergency-use-authorization-first-covid-19.
- European Medicines Agency (EMA). 2020. “EMA Recommends First COVID-19 Vaccine for Authorisation in the EU.” December 21, 2020. Accessed January 17, 2025. https://www.ema.europa.eu/en/news/ema-recommends-first-covid-19-vaccine-authorisation-eu.
- European Centre for Disease Prevention and Control (ECDC). 2020. “First COVID-19 Vaccine Authorised for Use in the European Union.” December 21, 2020. Accessed January 17, 2025. https://www.ecdc.europa.eu/en/news-events/first-covid-19-vaccine-authorised-use-european-union.
- Fauci, A. S. 2021. “The Story Behind COVID-19 Vaccines.” Science 372 (6538): 109. [CrossRef]
- Brear, M. R., and R. Gordon. 2021. “Translating the Principle of Beneficence into Ethical Participatory Development Research Practice.” Journal of International Development 33 (1): 109–126. [CrossRef]
- European Medicines Agency (EMA). 2016. “Conditional Marketing Authorisation.” February 15, 2016. Accessed May 7, 2025. https://www.ema.europa.eu/en/human-regulatory-overview/marketing-authorisation/conditional-marketing-authorisation.
- Pharmaceuticals and Medical Devices Agency (PMDA). 2021. “Special Approval for Emergency on First COVID-19 Vaccine in Japan.” February 16, 2021. Accessed May 7, 2025. https://www.pmda.go.jp/english/about-pmda/0003.pdf.
- Mattos Filho. 2020. “Anvisa Issues Emergency Use Authorization Rules to Accelerate Access to Covid-19 Vaccines.” December 8, 2020. Accessed May 7, 2025. https://www.mattosfilho.com.br/en/unico/anvisa-autorizacao-uso-emergencial-vacinas/.
- Mikule, E., T. Reissaar, J. Villers, A. S. Takoupo Penka, A. Temerev, and L. Rozanova. 2021. “The Fast Approval and Slow Rollout of Sputnik V: Why Is Russia’s Vaccine Rollout Slower than That of Other Nations?” Epidemiologia 2 (3): 360–376. [CrossRef]
- Ministry of Health and Family Welfare, Government of India. n.d. “General FAQ.” Accessed May 7, 2025. https://dshm.delhi.gov.in/(S(anjsnxdbajexzs2iqruzkarz))/COWIN/English/FAQHWFrontline.pdf.
|
Feature Category |
Feature | FDA (USA)a | EU regulatory network (EMA + NCAs)a | PMDA (Japan)b | NMPA (China)c | ANVISA (Brazil)d | CDSCO (India)e | Roszdravnadzor (Russia)c |
| General Framework | Public Health Mission | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Legal Basis | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |
| Organizational Model | Team | Rapporteur | Team | Provincial | Centralized | Centralized | Federal | |
| Pre-Market Requirements | Review Timeline (months) | 6–10f | 7–11g | 12h | ~10i | ~12j | 12–24k | 18–26 |
| Expedited Pathways | Multiple | PRIME | Sakigake | Yes | Yes | Limited | Limited | |
| Clinical Data: Local Required | Nol | Partialm | Yes | Yesn | Partial | Yes | Yes | |
| Electronic Submissions (eCTD) | Yes | Yes | Yes | In progress | In progress | Partial | Paper/electronic hybrid | |
| Post-Market Oversight | Safety Monitoring | REMS | RMP | Re-exam | Yes | Yes | Limited | Basic |
| Transparency | High | Moderate | Moderate | Low | Moderate | Low | Low | |
| Inspection Capacity | High | High | High | Medium | Medium | Limited | Periodic | |
| Global Alignment | ICH Participation | Full | Full | Full | Member | Observer | Limited | None |
| Accept RWE (Real Word Evidence) | Growing | Developing | Developing | Limited | Limited | Minimal | Minimal |
| Agency/Region | Terminology | Legal Framework | Approval Pathway | Special Features | Post-Marketing Oversight |
| FDA (USA)a | HCT/Ps, Cell/Gene Therapies | 21 CFR Part 1271 | IND + Biologics License Application (BLA) | RMAT, Breakthrough Therapy, Fast Track options | REMS and post-marketing surveillance |
| EU (EMA + Member States/NCAs)b | Advanced Therapy Medicinal Products (ATMPs) | Regulation (EC) No. 1394/2007 | Centralized authorization via EMA | Hospital exemption (implemented at Member State level via NCAs, e.g., Fimea); optional classification procedure | Risk Management Plans (RMPs) required |
| PMDA (Japan)c | Regenerative Medical Products | PMD Act (2014), Act on the Safety of Regenerative Medicine | Conditional/time-limited approval | Accelerated approval with post-market conditions | Mandatory follow-up studies |
|
ANVISA (Brazil)d |
Produtos de Terapias Avançadas | RDC 338/2020 | Clinical trials + GMP + regulatory submission | Special pathways for rare disease therapies | Traceability and pharmacovigilance |
| Roszdravnadzor (Russia)e | Biomedical Cell Products (BCPs) | Federal Law No. 180-FZ (2016), Law No. 61-FZ | State registration post-expert review | Centralized registry and annual reporting | Biosecurity, ethics board, and monitoring |
| CDSCO (India)f | Stem Cell and Cell-Based Products (SCCPs) | Guidelines for Stem Cell Research (2017) | Ethics + regulatory clearance from DCGI | Oversight by Institutional Ethics Committees | Ethical and informed consent focus |
| NMPA (China)g | Cell and Gene Therapy Products | Technical Guidelines (2021), ICH-aligned | IND-equivalent process + GMP | Fast-track review pathways | Enhanced post-marketing safety guidance |
| Agency/Region | Expedited Pathway | Clinical Data | Post Authorization Monitoring |
| FDA (USA)a | Issues Emergency Use Authorization | Phase III data from clinical trial | RWE and active safety monitoring required |
| EU (EMA + European Commission)b | Issues Conditional Marketing Authorization e.g. Comirnaty Approval | Phase III data except rare diseases. | Continued data submission required to maintain status |
| PMDA (Japan)c | Issues a Special Approval for Emergency e.g. Pfizer-BioNTech vaccine | Rely on Japan or foreign Clinical trial Phase III data | Post approval monitoring activities required. |
|
ANVISA (Brazil)d |
Issues Emergency Use Authorization | Require Phase III data | Follow-up data on vaccine quality, safety and efficacy required. |
| Roszdravnadzor (Russia)e | Issues Emergence Approval e.g. Sputnik V | Phase III Clinical data not required. | Post approval monitoring of data is required. |
| CDSCO (India)f | Issues Emergency Use Authorization e.g. Covishield |
Phase II Clinical data required. Bridging trials may be required. | Post Approval follow-up is required. |
| NMPA (China)g | Issues a conditional market approval e.g. Sinovac's CoronaVac |
Phase III data required. | Required to continue relevant research, fulfill conditional requirements and submit the results of subsequent research in a timely manner. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




