Submitted:
07 February 2026
Posted:
09 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
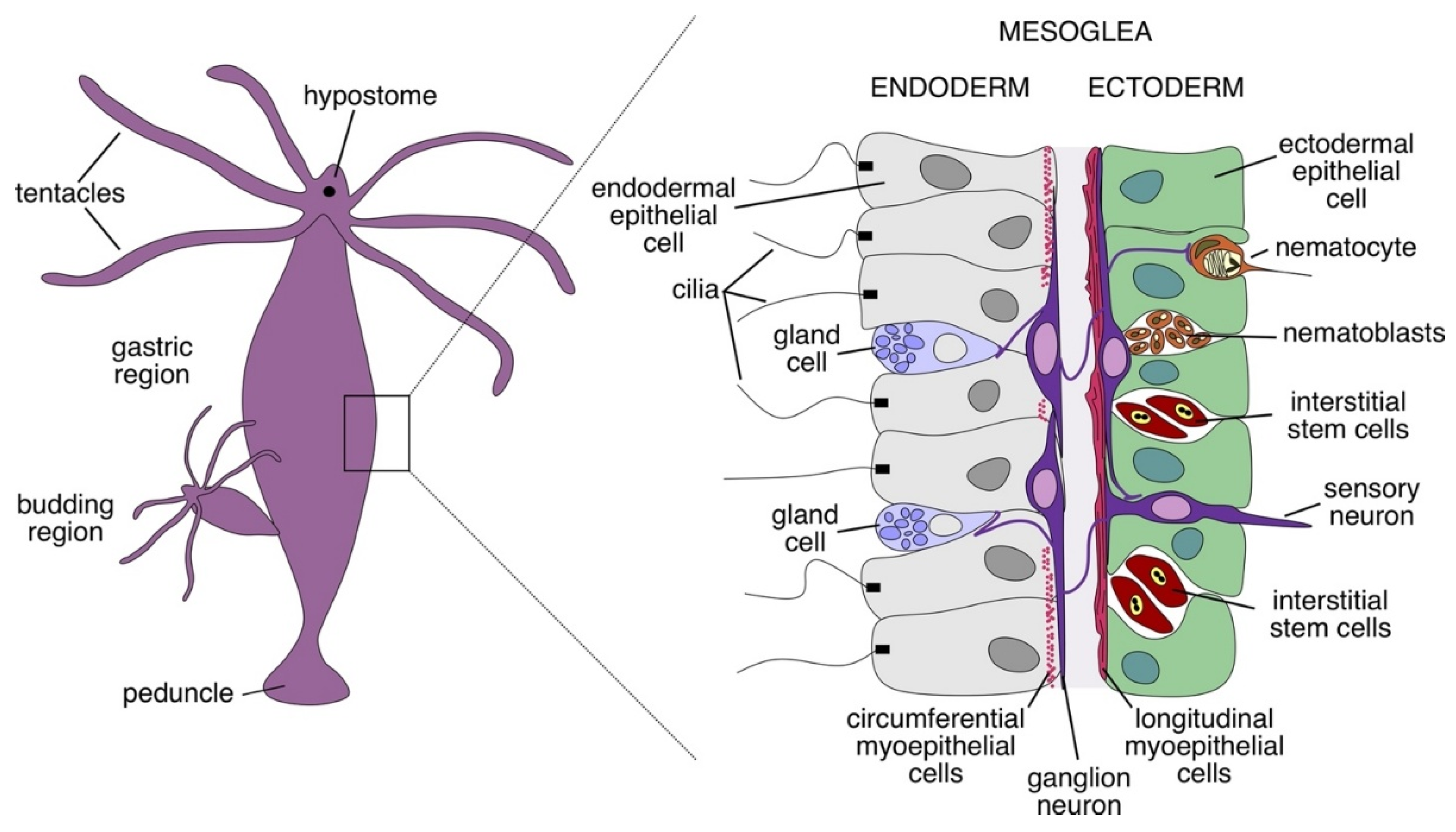
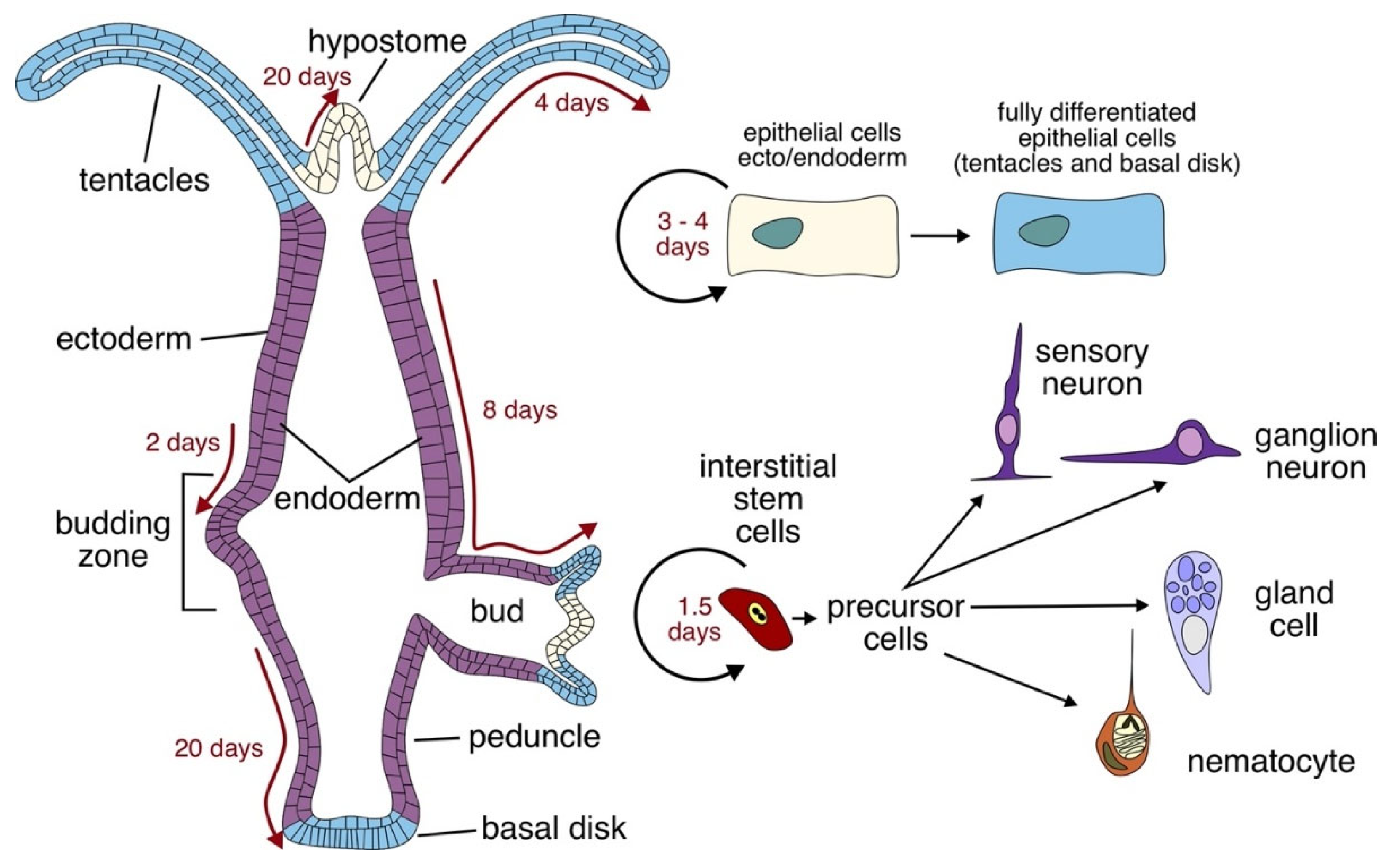
2. Peptidergic Signaling in Hydra
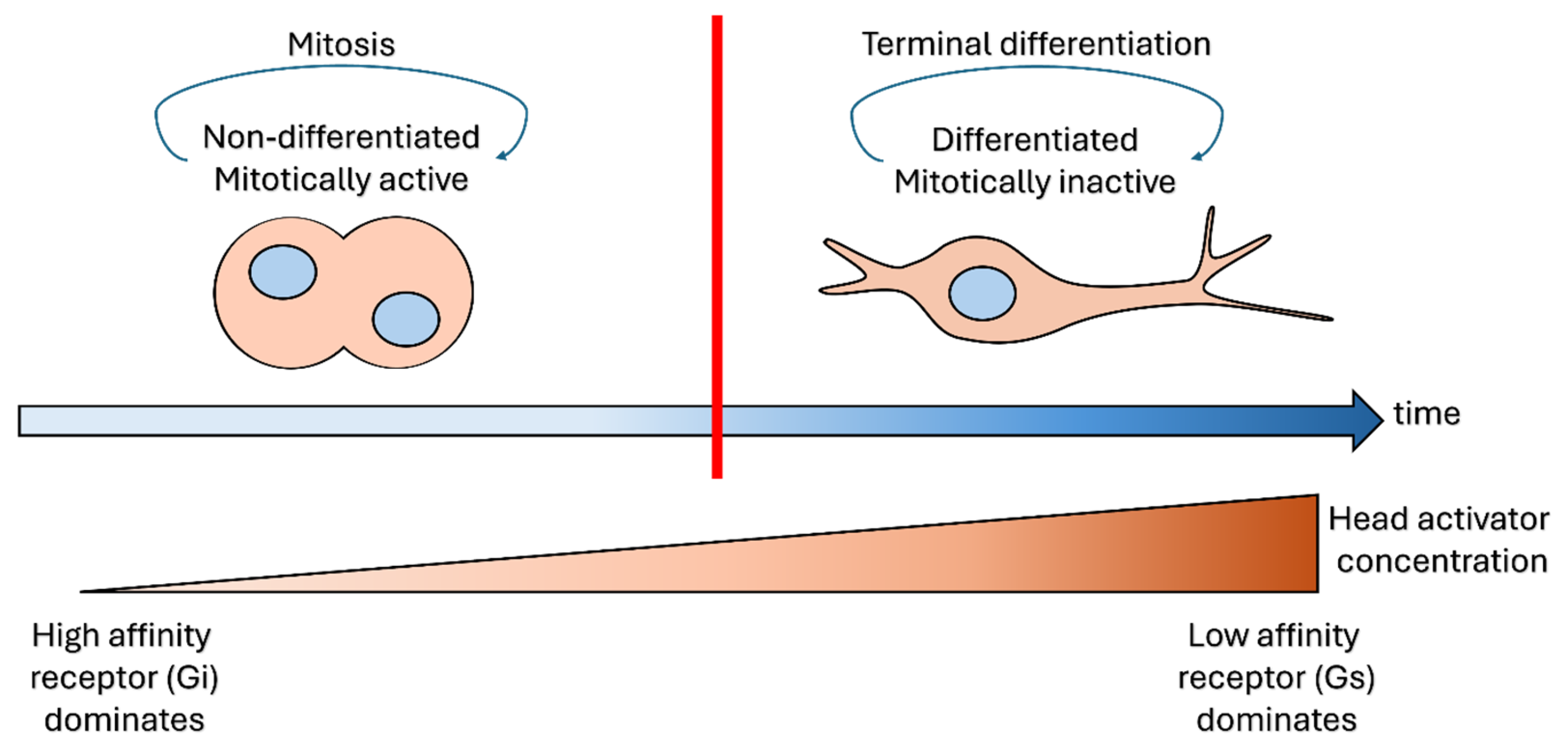
3. Context-Dependent Signaling of the Head Activator
4. Signaling Pathways of the Head Activator Peptide in Hydra
5. Effects of Exogenous Head Activator Applied to Mammals
6. Evolutionary Conservation of the Head Activator
7. Signaling Pathway of the Head Activator Peptide in Mammals
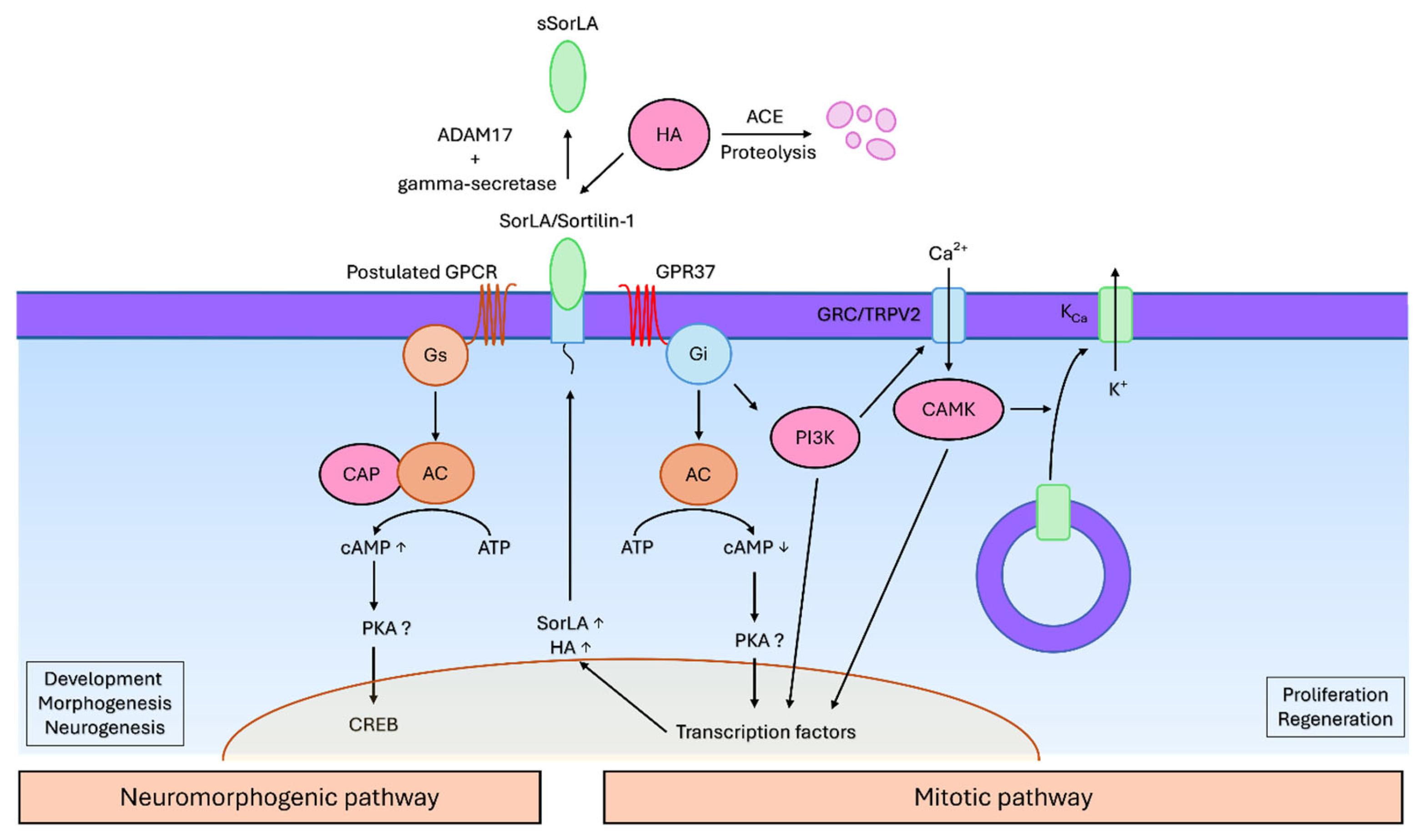
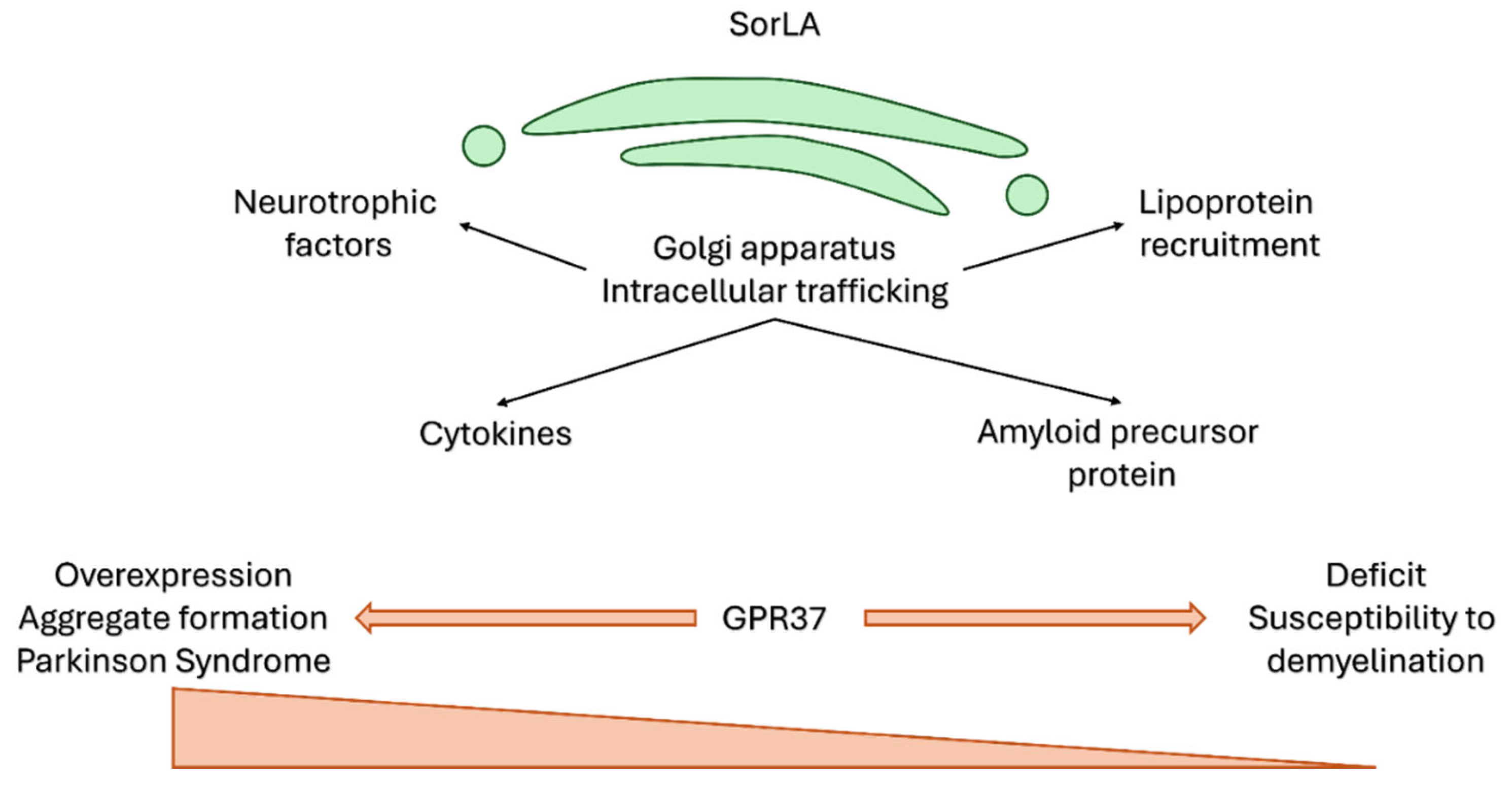
8. Potential Role of the Head Activator in Human Neuropathology
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Van Iten, H.; Leme, J.M.; Pacheco, Mírian L. A. F.; Simões, M.G.; Fairchild, T.R.; Rodrigues, F.; Galante, D.; Boggiani, P.C.; Marques, A.C. Origin and Early Diversification of Phylum Cnidaria: Key Macrofossils from the Ediacaran System of North and South America. Palaeontology 2014, 677–690. [CrossRef]
- Reddy, P.C.; Gungi, A.; Unni, M. Cellular and Molecular Mechanisms of Hydra Regeneration. Results and problems in cell differentiation 2019, 68, 259–290. [CrossRef]
- Yuste, R. Breaking the neural code of a cnidarian: Learning principles of neuroscience from the "vulgar" Hydra. Current Opinion in Neurobiology 2024, 86, 102869. [CrossRef]
- Tomczyk, S.; Fischer, K.; Austad, S.; Galliot, B. Hydra, a powerful model for aging studies. Invertebrate Reproduction & Development 2015, 59, 11–16. [CrossRef]
- Lucia Mattera. Functionalization, bio-conjugation and toxicity studies of quantum dots, 2016.
- Buzgariu, W.; Al Haddad, S.; Tomczyk, S.; Wenger, Y.; Galliot, B. Multi-functionality and plasticity characterize epithelial cells in Hydra. Tissue Barriers 2015, 3, e1068908. [CrossRef]
- Plachetzki, D.C.; Fong, C.R.; Oakley, T.H. Cnidocyte discharge is regulated by light and opsin-mediated phototransduction. BMC Biology 2012, 10, 17. [CrossRef]
- Bellantuono, A.J.; Bridge, D.; Martínez, D.E. Hydra as a tractable, long-lived model system for senescence. Invertebrate Reproduction & Development 2015, 59, 39–44. [CrossRef]
- Ojimi, M.C.; Isomura, N.; Hidaka, M. Telomerase activity is not related to life history stage in the jellyfish Cassiopea sp. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2009, 152, 240–244. [CrossRef]
- Pascual-Torner, M.; Carrero, D.; Pérez-Silva, J.G.; Álvarez-Puente, D.; Roiz-Valle, D.; Bretones, G.; Rodríguez, D.; Maeso, D.; Mateo-González, E.; Español, Y.; et al. Comparative genomics of mortal and immortal cnidarians unveils novel keys behind rejuvenation. Proc. Natl. Acad. Sci. U. S. A. 2022, 119, e2118763119. [CrossRef]
- Primack, A.S.; Cazet, J.F.; Little, H.M.; Mühlbauer, S.; Cox, B.D.; David, C.N.; Farrell, J.A.; Juliano, C.E. Differentiation trajectories of the Hydra nervous system reveal transcriptional regulators of neuronal fate. bioRxiv 2023, 2023.03.15.531610. [CrossRef]
- Noro, Y.; Shimizu, H.; Mineta, K.; Gojobori, T. A single neuron subset governs a single coactive neuron circuit in Hydra vulgaris, representing a possible ancestral feature of neural evolution. Sci Rep 2021, 11, 10828. [CrossRef]
- Dupre, C.; Yuste, R. Non-overlapping Neural Networks in Hydra vulgaris. Current biology : CB 2017, 27, 1085–1097. [CrossRef]
- Keramidioti, A.; Schneid, S.; Busse, C.; Laue, C.C. von; Bertulat, B.; Salvenmoser, W.; Heß, M.; Alexandrova, O.; Glauber, K.M.; Steele, R.E.; et al. A new look at the architecture and dynamics of the Hydra nerve net. eLife 2024, 12. [CrossRef]
- Badhiwala, K.N.; Primack, A.S.; Juliano, C.E.; Robinson, J.T. Multiple neuronal networks coordinate Hydra mechanosensory behavior. eLife Sciences Publications, Ltd [Online], July 30, 2021. Available online: https://elifesciences.org/articles/64108#bib64 (accessed on 25 June 2025).
- Rentzsch, F.; Layden, M.; Manuel, M. The cellular and molecular basis of cnidarian neurogenesis. Wiley Interdisciplinary Reviews. Developmental Biology 2017, 6, e257. [CrossRef]
- Hauser, F.; Koch, T.L.; Grimmelikhuijzen, C.J.P. Review: The evolution of peptidergic signaling in Cnidaria and Placozoa, including a comparison with Bilateria. Frontiers in endocrinology 2022, 13, 973862. [CrossRef]
- Liao, Q.; Feng, Y.; Yang, B.; Lee, S.M.-Y. Cnidarian peptide neurotoxins: a new source of various ion channel modulators or blockers against central nervous systems disease. Drug Discovery Today 2019, 24, 189–197. [CrossRef]
- Jayathilake, J.M.N.J.; Gunathilake, K.V.K. Cnidarian toxins: recent evidences for potential therapeutic uses. The European Zoological Journal 2020, 87, 708–713. [CrossRef]
- Schaller, H.C.; Bodenmüller, H. Structure and Function of the Head Activator in Hydra and in Mammals. In Advances in Metabolic Disorders : Gastrointestinal Hormones; Mutt, V., Ed.; Elsevier, 1988; pp 519–526, ISBN 0065-2903.
- Hicklin, J.; Hornbruch, A.; Wolpert, L.; Clarke, M. Positional information and pattern regulation in hydra: the formation of boundary regions following axial grafts. Development (Cambridge, England) 1973, 30, 701–725. [CrossRef]
- Chica Schaller, H. Neuropeptides in Hydra. Trends in Neurosciences 1979, 2, 120–122. [CrossRef]
- Takahashi, T.; Muneoka, Y.; Lohmann, J.; Lopez de Haro, M.S.; Solleder, G.; Bosch, T.C.; David, C.N.; Bode, H.R.; Koizumi, O.; Shimizu, H.; et al. Systematic isolation of peptide signal molecules regulating development in hydra: LWamide and PW families. Proc. Natl. Acad. Sci. U. S. A. 1997, 94, 1241–1246. [CrossRef]
- Fujisawa, T. Hydra peptide project 1993-2007. Development, growth & differentiation 2008, 50 Suppl 1, S257-68. [CrossRef]
- Takahashi, T.; Fujisawa, T. Important roles for epithelial cell peptides in hydra development. BioEssays : news and reviews in molecular, cellular and developmental biology 2009, 31, 610–619. [CrossRef]
- Takahashi, T.; Hayakawa, E.; Koizumi, O.; Fujisawa, T. Neuropeptides and their functions in Hydra. BIOLOGIA FUTURA 2008, 59 Suppl, 227–235. [CrossRef]
- Schaller, H.C.; Bodenmüller, H. Isolation and amino acid sequence of a morphogenetic peptide from hydra. Proc. Natl. Acad. Sci. U. S. A. 1981, 78, 7000–7004. [CrossRef]
- Hoffmeister, S.A. Isolation and characterization of two new morphogenetically active peptides from Hydra vulgaris. Development (Cambridge, England) 1996, 122, 1941–1948. [CrossRef]
- Schaller, H.C.; Hoffmeister, S.A.; Dübel, S. Role of the neuropeptide head activator for growth and development in hydra and mammals. Development (Cambridge, England) 1989, 107 Suppl, 99–107. [CrossRef]
- Lohmann, J.U.; Bosch, T.C. The novel peptide HEADY specifies apical fate in a simple radially symmetric metazoan. Genes & Development 2000, 14, 2771–2777. [CrossRef]
- Takahashi, T.; Koizumi, O.; Ariura, Y.; Romanovitch, A.; Bosch, T.C.; Kobayakawa, Y.; Mohri, S.; Bode, H.R.; Yum, S.; Hatta, M.; et al. A novel neuropeptide, Hym-355, positively regulates neuron differentiation in Hydra. Development (Cambridge, England) 2000, 127, 997–1005. [CrossRef]
- Bodenmüller, H.; Schaller, H.C. Conserved amino acid sequence of a neuropeptide, the head activator, from coelenterates to humans. Nature 1981, 293, 579–580. [CrossRef]
- Hoffmeister-Ullerich, S.A. The foot formation stimulating peptide pedibin is also involved in patterning of the head in hydra. Mechanisms of Development 2001, 106, 37–45. [CrossRef]
- Herrmann, D.; Hatta, M.; Hoffmeister-Ullerich, S.A.H. Thypedin, the multi copy precursor for the hydra peptide pedin, is a beta-thymosin repeat-like domain containing protein. Mechanisms of Development 2005, 122, 1183–1193. [CrossRef]
- Takahashi, T.; Takeda, N. Insight into the molecular and functional diversity of cnidarian neuropeptides. International journal of molecular sciences 2015, 16, 2610–2625. [CrossRef]
- Takahashi, T.; Koizumi, O.; Hayakawa, E.; Minobe, S.; Suetsugu, R.; Kobayakawa, Y.; Bosch, T.C.G.; David, C.N.; Fujisawa, T. Further characterization of the PW peptide family that inhibits neuron differentiation in Hydra. Archiv f mikr Anat u Entwicklungsmechanik 2009, 219, 119–129. [CrossRef]
- Morishita, F.; Nitagai, Y.; Furukawa, Y.; Matsushima, O.; Takahashi, T.; Hatta, M.; Fujisawa, T.; Tunamoto, S.; Koizumi, O. Identification of a vasopressin-like immunoreactive substance in hydra. Peptides 2003, 24, 17–26. [CrossRef]
- Schawaller, M.; Schenck, K.; Hoffmeister, S.A.; Schaller, H.; Schaller, H.C. Production and characterization of monoclonal antibodies recognizing head activator in precursor form and immunocytochemical localization of head activator precursor and head activator peptide in the neural cell line NH15-CA2 and in hydra. Differentiation; research in biological diversity 1988, 38, 149–160. [CrossRef]
- Schaller, H.C. Action of the head activator as a growth hormone in hydra. Cell Differentiation 1976, 5, 1–11. [CrossRef]
- Schaller, H.C. Action of the head activator on the determination of interstitial cells in hydra. Cell Differentiation 1976, 5, 13–25. [CrossRef]
- Schaller, H.C.; Hofmann, M.; Javois, L.C. Effect of head activator on proliferation, head-specific determination and differentiation of epithelial cells in hydra. Differentiation; research in biological diversity 1990, 43, 157–164. [CrossRef]
- Hobmayer, B.; Holstein, T.W.; David, C.N. Stimulation of tentacle and bud formation by the neuropeptide head activator in Hydra magnipapillata. Developmental biology 1997, 183, 1–8. [CrossRef]
- H. Schaller; I. Hermans-Borgmeyer; S. Hoffmeister. Neuronal control of development in hydra. International Journal of Developmental Biology 1996.
- Hampe, W.; Hermans-Borgmeyer, I.; Schaller, H.C. Function of the neuropeptide head activator for early neural and neuroendocrine development. Results and problems in cell differentiation 1999, 26, 323–337. [CrossRef]
- Galliot, B.; Welschof, M.; Schuckert, O.; Hoffmeister, S.; Schaller, H.C. The cAMP response element binding protein is involved in hydra regeneration. Development (Cambridge, England) 1995, 121, 1205–1216. [CrossRef]
- Neubauer, K.H.; Christians, S.; Hoffmeister, S.A.; Kreger, S.; Schaller, H.C. Characterisation of two types of head activator receptor on hydra cells. Mechanisms of Development 1990, 33, 39–47. [CrossRef]
- Christians, S.; Neubauer, K.H.; Ulrich, H. Purification and characterization of the head-activator receptor from a multi-headed mutant of Chlorohydra viridissima. FEBS Letters 1993, 316, 141–146. [CrossRef]
- Franke, I.; Buck, F.; Hampe, W. Purification of a head-activator receptor from hydra. Eur. J. Biochem. 1997, 244, 940–945. [CrossRef]
- Schaller, H.C.; Druffel-Augustin, S.; Dübel, S. Head activator acts as an autocrine growth factor for NH15-CA2 cells in the G2/mitosis transition. The EMBO journal 1989, 8, 3311–3318. [CrossRef]
- Fenger, U.; Hofmann, M.; Galliot, B.; Schaller, H.C. The role of the cAMP pathway in mediating the effect of head activator on nerve-cell determination and differentiation in hydra. Mechanisms of Development 1994, 47, 115–125. [CrossRef]
- v. d. Slepushkin; Prum, I.A.; Vinogradov, V.A.; Pavlov, V.A.; Titov, M.I.; Vasil'ev, N.V. Effect of hydra peptide morphogen on cyclic nucleotide levels in injured tissues. Bull Exp Biol Med 1989, 108, 1491–1493. [CrossRef]
- Bodenmüller, H.; Schilling, E.; Zachmann, B.; Schaller, H.C. The neuropeptide head activator loses its biological acitivity by dimerization. The EMBO journal 1986, 5, 1825–1829. [CrossRef]
- Schaller, H.C.; Roberge, M.; Zachmann, B.; Hoffmeister, S.; Schilling, E.; Bodenmüller, H. The head activator is released from regenerating Hydra bound to a carrier molecule. The EMBO journal 1986, 5, 1821–1824. [CrossRef]
- Lai, J.R.; Gellman, S.H. Reinvestigation of the proposed folding and self-association of the Neuropeptide Head Activator. Protein science : a publication of the Protein Society 2003, 12, 560–566. [CrossRef]
- Hampe, W.; Riedel, I.B.; Lintzel, J.; Bader, C.O.; Franke, I.; Schaller, H.C. Ectodomain shedding, translocation and synthesis of SorLA are stimulated by its ligand head activator. J Cell Sci 2000, 113 Pt 24, 4475–4485. [CrossRef]
- Hampe, W.; Urny, J.; Franke, I.; Hoffmeister-Ullerich, S.A.; Herrmann, D.; Petersen, C.M.; Lohmann, J.; Schaller, H.C. A head-activator binding protein is present in hydra in a soluble and a membrane-anchored form. Development (Cambridge, England) 1999, 126, 4077–4086. [CrossRef]
- Hermans-Borgmeyer, I.; Hampe, W.; Schinke, B.; Methner, A.; Nykjaer, A.; Süsens, U.; Fenger, U.; Herbarth, B.; Schaller, H.C. Unique expression pattern of a novel mosaic receptor in the developing cerebral cortex. Mechanisms of Development 1998, 70, 65–76. [CrossRef]
- Schmidt, V.; Subkhangulova, A.; Willnow, T.E. Sorting receptor SORLA: cellular mechanisms and implications for disease. Cell. Mol. Life Sci. 2017, 74, 1475–1483. [CrossRef]
- Jacobsen, L.; Madsen, P.; Jacobsen, C.; Nielsen, M.S.; Gliemann, J.; Petersen, C.M. Activation and functional characterization of the mosaic receptor SorLA/LR11. Journal of Biological Chemistry 2001, 276, 22788–22796. [CrossRef]
- Rezgaoui, M.; Süsens, U.; Ignatov, A.; Gelderblom, M.; Glassmeier, G.; Franke, I.; Urny, J.; Imai, Y.; Takahashi, R.; Schaller, H.C. The neuropeptide head activator is a high-affinity ligand for the orphan G-protein-coupled receptor GPR37. J Cell Sci 2006, 119, 542–549. [CrossRef]
- Rust, M.B.; Khudayberdiev, S.; Pelucchi, S.; Marcello, E. CAPt'n of Actin Dynamics: Recent Advances in the Molecular, Developmental and Physiological Functions of Cyclase-Associated Protein (CAP). Frontiers in cell and developmental biology 2020, 8, 586631. [CrossRef]
- Kaloulis, K.; Chera, S.; Hassel, M.; Gauchat, D.; Galliot, B. Reactivation of developmental programs: the cAMP-response element-binding protein pathway is involved in hydra head regeneration. Proc. Natl. Acad. Sci. U. S. A. 2004, 101, 2363–2368. [CrossRef]
- Niemann, S.; Schaller, H.C. Head-activator and the neuroectodermal differentiation of P19 mouse embryonal carcinoma cells. Neuroscience letters 1996, 207, 49–52. [CrossRef]
- Ulrich, H.; Tárnok, A.; Schaller, H.C. Head-activator induced mitosis of NH15-CA2 cells requires calcium influx and hyperpolarization. Journal of Physiology-Paris 1996, 90, 85–94. [CrossRef]
- Kayser, S.T.; Ulrich, H.; Schaller, H.C. Involvement of a Gardos-type potassium channel in head activator-induced mitosis of BON cells. European Journal of Cell Biology 1998, 76, 119–124. [CrossRef]
- Quach, T.T.; Duchemin, A.M.; Oliver, A.P.; Schrier, B.K.; Wyatt, R.J. Hydra head activator peptide has trophic activity for eukaryotic neurons. Developmental Brain Research 1992, 68, 97–102. [CrossRef]
- Kajiwara, S.; Sato, T. Growth promoting effect of head activator in cultured chick embryo brain cells. Acta Endocrinologica 1986, 113, 604–608. [CrossRef]
- Kalenikova, E.I.; Tishchenko, V.A.; Rubina, A.Y.; Sokolov, A.S.; Blidchenko, Y.A.; Kuznetsova, T.V.; Bespalova, Z.D. Distribution of a morphogenic peptide activating the head growth of hydra in rat organs following intravascular administration of3H-labeled peptide. Bull Exp Biol Med 1994, 118, 1162–1165. [CrossRef]
- Khomichuk, A.Y.; Timoshin, S.S. Participation of hydra peptide morphogen in regulation of proliferation of various albino rat tissues. Bull Exp Biol Med 1991, 112, 1332–1334. [CrossRef]
- Timoshin, S.S.; Yakovenko, I.G.; Berezina, G.P.; Obukhova, G.G.; Rubina, A.Y.; Murzina, N.B. Hydra peptide morphogen weakens poststress disturbances in albino rats. Bull Exp Biol Med 1997, 124, 965–967. [CrossRef]
- Lebed'ko, O.A.; Yatsenko, T.V.; Timoshin, S.S.; Rubina, A.Y. Effect of the hydra peptide morphogen on posthypoxic disorders in rats exposed to prenatal hypoxia. Bull Exp Biol Med 1997, 123, 232–234. [CrossRef]
- Lebed'ko, O.A.; Timoshin, S.S.; Rubina, A.Y. Effects of hydra peptide morphogen and its analogue and fragments on DNA synthesis in tracheal epithelium and smooth muscle cells in newborn albino rats. Bull Exp Biol Med 2000, 129, 550–552. [CrossRef]
- Fedoseev, V.A.; Kazimirskii, A.N.; Ionina, I.A.; Lomakina, E.B.; Rubina, A.Y.; Pylaev, A.S. Effect of hydra morphogen peptide on the dynamics of tissue components of the myocardial layers in compensatory hypertrophy of the heart. Bull Exp Biol Med 1993, 116, 1162–1164. [CrossRef]
- Fedoseev, V.A.; Kazimirskii, A.N.; Lomakina, E.B.; Ionina, I.A.; Rubina, A.Y.; Pylaev, A.S. Effect of hydra morphogen peptide on the structure of the tissue components of the myocardium layers in the early development of heart hypertrophy in rats. Bull Exp Biol Med 1993, 115, 339–341. [CrossRef]
- Sazonova, E.N.; Yakovenko, I.G.; Kryzhanovskaya, S.Y.; Budylev, A.A.; Timoshin, S.S. Effects of analogues of hydra peptide morphogen on DNA synthesis in the myocardium of newborn albino rats. Bull Exp Biol Med 2012, 152, 304–306. [CrossRef]
- Gan'cheva, E.A.; Kozlov, V.K.; Timoshin, S.S. Effect of the hydra peptide morphogen on cell proliferation in the thymus of newborn rats exposed to prenatal hypoxia. Bull Exp Biol Med 1997, 124, 929–931. [CrossRef]
- Murzina, N.B.; Khomichuk, A.Y.; Timoshin, S.S.; Obukhova, G.G.; Anosova, O.A.; Berezina, G.P. Effect of hydra peptide morphogen on levels of?-endorphin and certain hormones in albino rat blood and adrenals. Bull Exp Biol Med 1991, 112, 1510–1512. [CrossRef]
- Yarygin, K.N.; Kazimirskii, A.N.; Kositskii, G.I.; Rubina, A.Y.; Vinogradov, V.A.; Pylaev, A.S. Modulation of ornithine decarboxylase activity in the normal and regenerating rat liver by various doses of the peptide morphogen ofHydra. Bull Exp Biol Med 1986, 101, 755–757. [CrossRef]
- Kazimirskii, A.N.; Yarygin, K.N.; Kositskii, G.I.; Rubina, A.Y.; Vinogradova, V.A.; Pylaev, A.S. Effect of low doses of hydra peptide morphogen on protein synthesis in the intact and regenerating rat liver. Bull Exp Biol Med 1988, 105, 503–506. [CrossRef]
- Drozd, Y.V.; Yasnetsov, V.V.; Shashkov, V.S. Vestibuloprotective properties of some regulatory peptides. Bull Exp Biol Med 1988, 106, 971–973. [CrossRef]
- Sazonova, E.N.; Zhivotova, E.Y.; Sazonov, O.A.; Fleishman, M.Y.; Timoshin, S.S. Effects of regulatory peptides on DNA synthesis in duodenal smooth muscle tissues of albino rats during the early postnatal period. Bull Exp Biol Med 1999, 127, 593–595. [CrossRef]
- Feurle, G.E.; Bodenmüller, H.; Baća, I. The neuropeptide head activator stimulates amylase release from rat pancreas in vitro. Neuroscience letters 1983, 38, 287–289. [CrossRef]
- Khomichuk, A.Y.; Timoshin, S.S.; Orlov, S.N.; Pokudin, N.I.; Kubatiev, A.A. Hydra peptide morphogen activates Na/H exchange in human erythrocytes. Bull Exp Biol Med 1991, 111, 763–764. [CrossRef]
- Sakura, H.; Aoki, S.; Ozawa, T.; Hashimoto, T.; Sakura, N. The neuropeptide, head activator, in human placenta and serum from pregnant women. Acta Endocrinologica 1991, 125, 454–458. [CrossRef]
- Kume, H. Role of Airway Smooth Muscle in Inflammation Related to Asthma and COPD. Advances in experimental medicine and biology 2021, 1303, 139–172. [CrossRef]
- Miyata, S. New aspects in fenestrated capillary and tissue dynamics in the sensory circumventricular organs of adult brains. Frontiers in Neuroscience 2015, 9, 390. [CrossRef]
- Zhang, C.; Kaye, J.A.; Cai, Z.; Wang, Y.; Prescott, S.L.; Liberles, S.D. Area Postrema Cell Types that Mediate Nausea-Associated Behaviors. Neuron 2021, 109, 461-472.e5. [CrossRef]
- Wang, L.; Wang, N.; Zhang, W.; Cheng, X.; Yan, Z.; Shao, G.; Wang, X.; Wang, R.; Fu, C. Therapeutic peptides: current applications and future directions. Sig Transduct Target Ther 2022, 7, 48. [CrossRef]
- Nicosia, A.; Maggio, T.; Costa, S.; Salamone, M.; Tagliavia, M.; Mazzola, S.; Gianguzza, F.; Cuttitta, A. Maintenance of a Protein Structure in the Dynamic Evolution of TIMPs over 600 Million Years. Genome Biol Evol 2016, 8, 1056–1071. [CrossRef]
- Schaller, H.C. A neurohormone from hydra is also present in the rat brain. Journal of Neurochemistry 1975, 25, 187–188. [CrossRef]
- Bodenmüller, H.; Schaller, H.C.; Darai, G. Human hypothalamus and intestine contain a hydra-neuropeptide. Neuroscience letters 1980, 16, 71–74. [CrossRef]
- Roberge, M.; Escher, E.; Schaller, H.C.; Bodenmüller, H. The hydra head activator in human blood circulation. Degradation of the synthetic peptide by plasma angiotensin-converting enzyme. FEBS Letters 1984, 173, 307–313. [CrossRef]
- Schaller, H.C.; Schilling, E.; Theilmann, L.; Bodenmüller, H.; Sachsenheimer, W. Elevated levels of head activator in human brain tumors and in serum of patients with brain and other neurally derived tumors. J Neuro-Oncol 1988, 6, 251–258. [CrossRef]
- Winnikes, M.; Schaller, H.C.; Sachsenheimer, W. Head activator as a potential serum marker for brain tumour analysis. European journal of cancer (Oxford, England : 1990) 1992, 28, 421–424. [CrossRef]
- Lintzel, J.; Franke, I.; Riedel, I.B.; Schaller, H.C.; Hampe, W. Characterization of the VPS10 domain of SorLA/LR11 as binding site for the neuropeptide HA. Biological chemistry 2002, 383, 1727–1733. [CrossRef]
- Böhm, C.; Seibel, N.M.; Henkel, B.; Steiner, H.; Haass, C.; Hampe, W. SorLA signaling by regulated intramembrane proteolysis. Journal of Biological Chemistry 2006, 281, 14547–14553. [CrossRef]
- Gandía, J.; Fernández-Dueñas, V.; Morató, X.; Caltabiano, G.; González-Muñiz, R.; Pardo, L.; Stagljar, I.; Ciruela, F. The Parkinson's disease-associated GPR37 receptor-mediated cytotoxicity is controlled by its intracellular cysteine-rich domain. Journal of Neurochemistry 2013, 125, 362–372. [CrossRef]
- Meyer, R.C.; Giddens, M.M.; Schaefer, S.A.; Hall, R.A. GPR37 and GPR37L1 are receptors for the neuroprotective and glioprotective factors prosaptide and prosaposin. Proc. Natl. Acad. Sci. U. S. A. 2013, 110, 9529–9534. [CrossRef]
- Dunham, J.H.; Meyer, R.C.; Garcia, E.L.; Hall, R.A. GPR37 surface expression enhancement via N-terminal truncation or protein-protein interactions. Biochemistry 2009, 48, 10286–10297. [CrossRef]
- Southern, C.; Cook, J.M.; Neetoo-Isseljee, Z.; Taylor, D.L.; Kettleborough, C.A.; Merritt, A.; Bassoni, D.L.; Raab, W.J.; Quinn, E.; Wehrman, T.S.; et al. Screening β-arrestin recruitment for the identification of natural ligands for orphan G-protein-coupled receptors. Journal of biomolecular screening 2013, 18, 599–609. [CrossRef]
- Smith, N.J. Drug Discovery Opportunities at the Endothelin B Receptor-Related Orphan G Protein-Coupled Receptors, GPR37 and GPR37L1. Front. Pharmacol. 2015, 6, 275. [CrossRef]
- Boels, K.; Glassmeier, G.; Herrmann, D.; Riedel, I.B.; Hampe, W.; Kojima, I.; Schwarz, J.R.; Schaller, H.C. The neuropeptide head activator induces activation and translocation of the growth-factor-regulated Ca(2+)-permeable channel GRC. J Cell Sci 2001, 114, 3599–3606. [CrossRef]
- Hertz, E.; Terenius, L.; Vukojević, V.; Svenningsson, P. GPR37 and GPR37L1 differently interact with dopamine 2 receptors in live cells. Neuropharmacology 2019, 152, 51–57. [CrossRef]
- Kubota, K.; Niinuma, Y.; Kaneko, M.; Okuma, Y.; Sugai, M.; Omura, T.; Uesugi, M.; Uehara, T.; Hosoi, T.; Nomura, Y. Suppressive effects of 4-phenylbutyrate on the aggregation of Pael receptors and endoplasmic reticulum stress. Journal of Neurochemistry 2006, 97, 1259–1268. [CrossRef]
- Xiromerisiou, G.; Bourinaris, T.; Houlden, H.; Lewis, P.A.; Senkevich, K.; Hammer, M.; Federoff, M.; Khan, A.; Spanaki, C.; Hadjigeorgiou, G.M.; et al. SORL1 mutation in a Greek family with Parkinson's disease and dementia. Annals of Clinical and Translational Neurology 2021, 8, 1961–1969. [CrossRef]
- Pietilä, M.; Sahgal, P.; Peuhu, E.; Jäntti, N.Z.; Paatero, I.; Närvä, E.; Al-Akhrass, H.; Lilja, J.; Georgiadou, M.; Andersen, O.M.; et al. SORLA regulates endosomal trafficking and oncogenic fitness of HER2. Nat Commun 2019, 10, 2340. [CrossRef]
- Al-Akhrass, H.; Pietilä, M.; Lilja, J.; Vesilahti, E.-M.; Anttila, J.M.; Haikala, H.M.; Munne, P.M.; Klefström, J.; Peuhu, E.; Ivaska, J. Sortilin-related receptor is a druggable therapeutic target in breast cancer. Molecular oncology 2022, 16, 116–129. [CrossRef]
- Jiang, Z.; Bi, F.; Ge, Z.; Mansolf, M.; Hartwich, T.M.P.; Kolesnyk, V.; Yang, K.; Park, W.; Kim, D.; Grechukhina, O.; et al. SORL1-Mediated EGFR and FGFR4 Regulation Enhances Chemoresistance in Ovarian Cancer. Cancers 2025, 17, 244. [CrossRef]
- Talbot, H.; Saada, S.; Naves, T.; Gallet, P.-F.; Fauchais, A.-L.; Jauberteau, M.-O. Regulatory Roles of Sortilin and SorLA in Immune-Related Processes. Frontiers in pharmacology 2018, 9, 1507. [CrossRef]
- Glerup, S.; Lume, M.; Olsen, D.; Nyengaard, J.R.; Vaegter, C.B.; Gustafsen, C.; Christensen, E.I.; Kjolby, M.; Hay-Schmidt, A.; Bender, D.; et al. SorLA controls neurotrophic activity by sorting of GDNF and its receptors GFRα1 and RET. Cell reports 2013, 3, 186–199. [CrossRef]
- Rovelet-Lecrux, A.; Feuillette, S.; Miguel, L.; Schramm, C.; Pernet, S.; Quenez, O.; Ségalas-Milazzo, I.; Guilhaudis, L.; Rousseau, S.; Riou, G.; et al. Impaired SorLA maturation and trafficking as a new mechanism for SORL1 missense variants in Alzheimer disease. Acta Neuropathologica Communications 2021, 9, 196. [CrossRef]
- Caglayan, S.; Takagi-Niidome, S.; Liao, F.; Carlo, A.-S.; Schmidt, V.; Burgert, T.; Kitago, Y.; Füchtbauer, E.-M.; Füchtbauer, A.; Holtzman, D.M.; et al. Lysosomal sorting of amyloid-β by the SORLA receptor is impaired by a familial Alzheimer's disease mutation. Sci. Transl. Med. 2014, 6, 223ra20. [CrossRef]
- Mehmedbasic, A.; Christensen, S.K.; Nilsson, J.; Rüetschi, U.; Gustafsen, C.; Poulsen, A.S.A.; Rasmussen, R.W.; Fjorback, A.N.; Larson, G.; Andersen, O.M. SorLA complement-type repeat domains protect the amyloid precursor protein against processing. The Journal of biological chemistry 2015, 290, 3359–3376. [CrossRef]
- Shih, A.Z.L.; Chen, Y.-C.; Speckmann, T.; Søndergaard, E.; Schürmann, A.; Verchere, C.B.; Willnow, T.E. SORLA mediates endocytic uptake of proIAPP and protects against islet amyloid deposition. Molecular metabolism 2022, 65, 101585. [CrossRef]
- Bolinger, A.A.; Frazier, A.; La, J.-H.; Allen, J.A.; Zhou, J. Orphan G Protein-Coupled Receptor GPR37 as an Emerging Therapeutic Target. ACS chemical neuroscience 2023, 14, 3318–3334. [CrossRef]
- Wang, X.; Ju, J.; Xie, Y.; Hang, L. Emerging roles of the G-protein-coupled receptor 37 in neurological diseases and pain. Neuroscience 2024, 559, 199–208. [CrossRef]
- Argerich, J.; Garma, L.D.; López-Cano, M.; Álvarez-Montoya, P.; Gómez-Acero, L.; Fernández-Dueñas, V.; Muñoz-Manchado, A.B.; Aso, E.; Boxer, A.; Andres-Benito, P.; et al. GPR37 processing in neurodegeneration: a potential marker for Parkinson's Disease progression rate. npj Parkinsons Dis. 2024, 10, 172. [CrossRef]
- Bian, X.; Wang, Y.; Zhang, W.; Ye, C.; Li, J. GPR37 and its neuroprotective mechanisms: bridging osteocalcin signaling and brain function. Frontiers in cell and developmental biology 2024, 12, 1510666. [CrossRef]
- Yang, H.-J.; Vainshtein, A.; Maik-Rachline, G.; Peles, E. G protein-coupled receptor 37 is a negative regulator of oligodendrocyte differentiation and myelination. Nat Commun 2016, 7, 10884. [CrossRef]
- Smith, B.M.; Giddens, M.M.; Neil, J.; Owino, S.; Nguyen, T.T.; Duong, D.; Li, F.; Hall, R.A. Mice lacking Gpr37 exhibit decreased expression of the myelin-associated glycoprotein MAG and increased susceptibility to demyelination. Neuroscience 2017, 358, 49–57. [CrossRef]
- Ma, Q.; Tian, J.-L.; Lou, Y.; Guo, R.; Ma, X.-R.; Wu, J.-B.; Yang, J.; Tang, B.-J.; Li, S.; Qiu, M.; et al. Oligodendrocytes drive neuroinflammation and neurodegeneration in Parkinson's disease via the prosaposin-GPR37-IL-6 axis. Cell reports 2025, 44, 115266. [CrossRef]
- Ekman, R.; Salford, L.; Brun, A.; Larsson, I. Hydra head activator-like immunoreactivity in human brain astrocytomas grade III-IV and the surrounding brain tissue. Peptides 1990, 11, 271–275. [CrossRef]
| peptide | effect | reference |
|---|---|---|
| morphogenesis | ||
| pedibin (Hym-346) | foot formation and regeneration head patterning |
[28] |
| HEADY | head formation budding |
[30] |
| neurogenesis | ||
| Hym-355 | stem cell proliferation stimulation of neuronal differentiation |
[31] |
| Hym-33H | inhibition of neuronal differentiation | [23] |
| Hym-35 | ||
| Hym-37 | ||
| Hym-310 | ||
| morphogenesis and neurogenesis | ||
| head activator | head formation and regeneration stem cell proliferation stimulation of neuronal differentiation |
[32] |
| pedin (Hym-323) | axial body patterning foot formation and regeneration budding stem cell proliferation stimulation of neuronal differentiation |
[28] |
| condition | effect | |
|---|---|---|
| cell type | mitotic | mitosis and differentiation |
| post-mitotic | differentiation | |
| time point | early | mitosis |
| late | differentiation | |
| concentration (receptor affinity) | low (high) | mitosis |
| high (low) | differentiation | |
| pathway (AC-cAMP-PKA axis) | AC inhibition | mitosis |
| AC activation | differentiation, neurogenesis | |
| tissue/cell type/cell line | effect | reference |
|---|---|---|
| rat corneal epithelium | proliferation ↑ | [69,70] |
| rat lingual epithelium | proliferation ↑ | |
| rat tracheal epithelium | proliferation ↑ | [71,72] |
| rat tracheal smooth muscle cells | proliferation ↓ | |
| lipid peroxidation system in hypoxic rats | inhibition in lungs | |
| stress response in rats | reduction of lipid peroxidation, corticosterone, normalised thyrotropic hormone, T3, T4, and thymus weight | [70] |
| rat myocardium upon a left ventricular hypertrophy | myocardial remodelling | [73,74] |
| rat myocardium | proliferation ↑ | [75] |
| rats exposed to prenatal hypoxia | normalisation of thymocyte proliferation | [76] |
| rats exposed to prenatal hypoxia | normalisation of hepatocyte and tracheal epithelium proliferation, reduction of pulmonary lipid peroxidation |
[71] |
| rat β-endorphine | low dose: ↓ high dose: ↑ |
[77] |
| rat corticosterone | low dose: ↑ high dose: ↓ |
[77] [78,79] |
| rat noradrenaline | low dose: ↑ high dose: ↓ |
|
| rat thyroid hormones | low dose: = high dose: Τ3 ↑ |
|
| rat insulin | low dose: = high dose: ↑ |
|
| liver function in partially hepatectomised rats | ornithine decarboxylase activity in low/medium/high dose: =/↑/↓ protein content: ↑ |
|
| cat model of motion sickness | antiemesis | [80] |
| rat duodenal smooth myocytes | proliferation = | [81] |
| injured rat liver | 4 h after injury: cAMP ↑, cGMP = 24 h after injury: cAMP ↓, cGMP ↑ |
[51] |
| injured rat striated muscle | cAMP =, cGMP = | |
| rat cultured pancreatic cells | amylase secretion ↑ | [82] |
| human erythrocytes | activation of Na/H exchange | [83] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




