Submitted:
06 February 2026
Posted:
06 February 2026
You are already at the latest version
Abstract
Keywords:
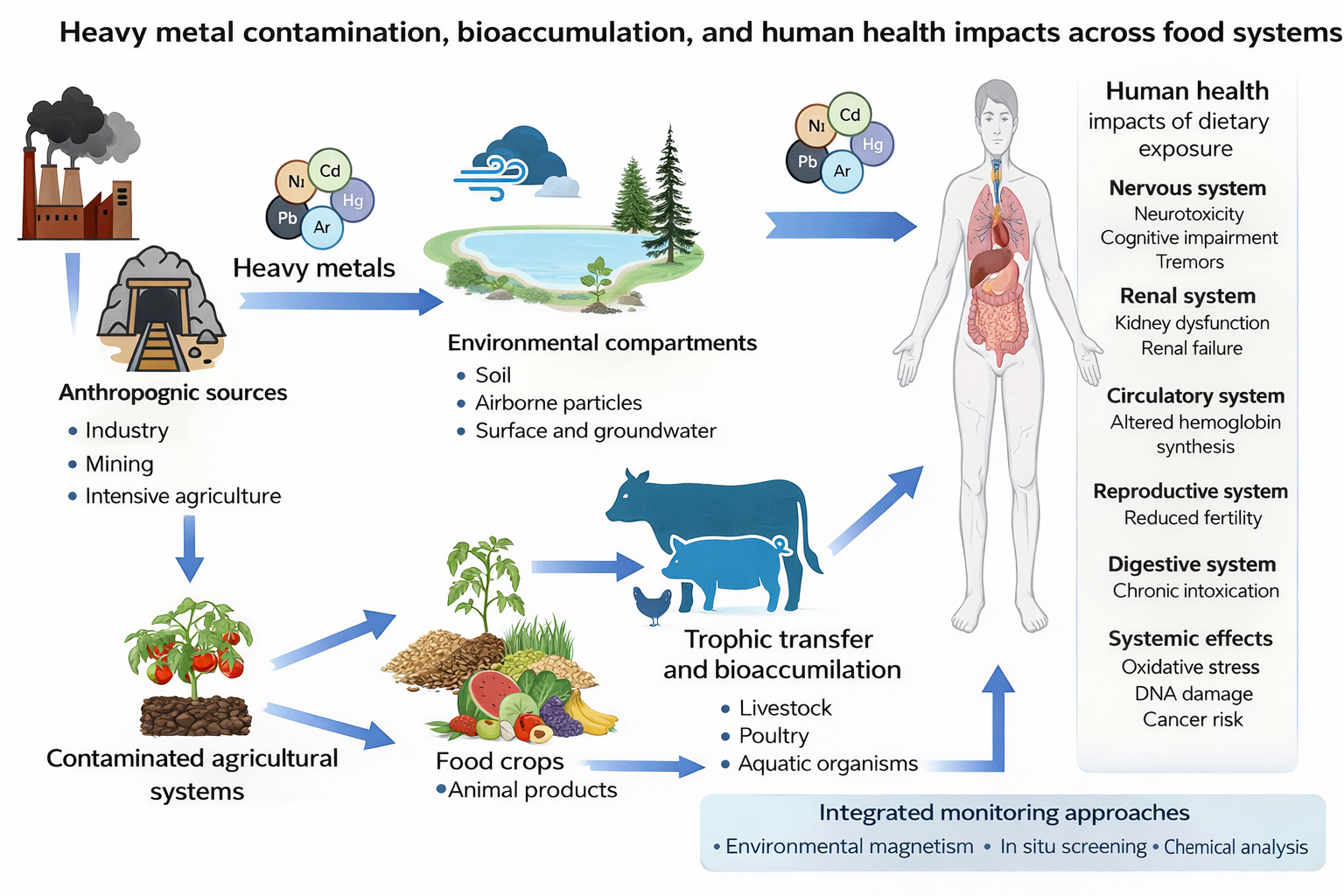
1. Introduction
2. Materials and Methods
2. Methodology
2.1. Literature Sources and Search Strategy
2.2. Study Selection Criteria
2.3. Study Screening and Selection Process
2.4. Data Extraction and Organisation
2.5. Evidence Synthesis
2.6. Considerations on Methodological Limitations
3. Results and Discussion
3.1. Heavy Metals
3.1.1. Sources of Heavy Metal Contamination in Foods
3.1.2. Dietary Exposure to Heavy Metals Through Food Consumption
3.2. Methods and Analytical Techniques for Quantifying Heavy Metals in Foods
3.2.1. Atomic Absorption Spectrometry (AAS)
3.2.2. Inductively Coupled Plasma Mass Spectrometry (ICP–MS)
3.2.3. Inductively Coupled Plasma Optical Emission Spectrometry (ICP–OES)
3.2.4. Liquid Chromatography–ICP–MS (LC–ICP–MS)
3.3. Emerging Techniques for Measuring Heavy Metals in Foods
3.3.1. X-Ray Fluorescence Spectrometry (XRF)
3.3.2. Raman Spectroscopy and SERS
3.3.3. Electronic Tongues
3.3.4. Electrochemical Nanomaterial-Based Biosensors
3.3.5. In Situ Magnetism
3.4. Heavy Metals and Human Health Impacts
4. Conclusions
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Afonne, O.J.; Ifediba, E.C. Heavy metals risks in plant foods – need to step up precautionary measures. Curr. Opin. Toxicol. 2020, 22, 1–6. [Google Scholar] [CrossRef]
- Sarker, A.; Kim, J.-E.; Islam, A.R.M.T.; Bilal, M.; Rakib, R.J.; Nandi, R.; Rahman, M.M.; Islam, T. Heavy metals contamination and associated health risks in food webs—a review focuses on food safety and environmental sustainability in Bangladesh. Environ. Sci. Pollut. Res. 2021, 29, 3230–3245. [Google Scholar] [CrossRef]
- Scutarașu, E.C.; Trincă, L.C. Heavy Metals in Foods and Beverages: Global Situation, Health Risks and Reduction Methods. Foods 2023, 12, 3340. [Google Scholar] [CrossRef]
- Hou, D.; Jia, X.; Wang, L.; McGrath, S.P.; Zhu, Y.-G.; Hu, Q.; Zhao, F.-J.; Bank, M.S.; O’cOnnor, D.; Nriagu, J. Global soil pollution by toxic metals threatens agriculture and human health. Science 2025, 388, 316–321. [Google Scholar] [CrossRef]
- Abdel-Rahman, G.N.-E. Heavy metals, definition, sources of food contamination, incidence, impacts and remediation: A literature review with recent updates. Egypt. J. Chem. 2021, 65, 419–437. [Google Scholar] [CrossRef]
- Abdelmonem, B.H.; Kamal, L.T.; Elbaz, R.M.; Khalifa, M.R.; Abdelnaser, A. From contamination to detection: The growing threat of heavy metals. Heliyon 2025, 11, e41713. [Google Scholar] [CrossRef]
- Castañeda-Miranda, A.G.; Böhnel, H.N.; Molina-Garza, R.S.; Chaparro, M.A. Magnetic evaluation of TSP-filters for air quality monitoring. Atmospheric Environ. 2014, 96, 163–174. [Google Scholar] [CrossRef]
- Miranda, A.G.C.; Chaparro, M.A.; Böhnel, H.N. Magnetic properties of Tillandsia recurvata L. and its use for biomonitoring a Mexican metropolitan area. Ecol. Indic. 2016, 60, 125–136. [Google Scholar] [CrossRef]
- Zaynab, M.; Al-Yahyai, R.; Ameen, A.; Sharif, Y.; Ali, L.; Fatima, M.; Khan, K.A.; Li, S. Health and environmental effects of heavy metals. J. King Saud Univ. - Sci. 2022, 34. [Google Scholar] [CrossRef]
- Mandal, R.R.; Bashir, Z.; Raj, D. Microbe-assisted phytoremediation for sustainable management of heavy metal in wastewater - A green approach to escalate the remediation of heavy metals. J. Environ. Manag. 2025, 375, 124199. [Google Scholar] [CrossRef]
- Buledi, J.A.; Amin, S.; Haider, S.I.; Bhanger, M.I.; Solangi, A.R. A review on detection of heavy metals from aqueous media using nanomaterial-based sensors. Environ. Sci. Pollut. Res. 2020, 28, 58994–59002. [Google Scholar] [CrossRef] [PubMed]
- Munir, N.; Jahangeer, M.; Bouyahya, A.; El Omari, N.; Ghchime, R.; Balahbib, A.; Aboulaghras, S.; Mahmood, Z.; Akram, M.; Shah, S.M.A.; et al. Heavy Metal Contamination of Natural Foods Is a Serious Health Issue: A Review. Sustainability 2021, 14, 161. [Google Scholar] [CrossRef]
- Castañeda-Miranda, A.G.; Chaparro, M.A.E.; Pacheco-Castro, A.; Böhnel, H.N. Magnetic biomonitoring of atmospheric dust using tree leaves of Ficus benjamina in Querétaro (México). Environ. Monit. Assess. 2020, 192, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Dávila-Cisneros, S.; Castañeda-Miranda, A.G.; Bautista-Capetillo, C.F.; Mattos-Villarroel, E.D.; Rodríguez-Abdalá, V.I.; Rovelo, C.O.R.; Pinedo-Torres, L.A.; Rodríguez-Trejo, A.; Ibarra-Delgado, S. Monitoring Landform Changes in a Mining Area in Mexico Using Geomatic Techniques. Geomatics 2025, 5, 63. [Google Scholar] [CrossRef]
- Lu, Y.; Chen, X.; Lan, H.; Nag, A.; Chen, Y.; Gao, J.; Deng, S. A review of multi-walled carbon nanotubes-based electrochemical sensors to detect heavy metals for food packaging applications. J. Alloy. Compd. 2025, 1017. [Google Scholar] [CrossRef]
- Mititelu, M.; Neacșu, S.M.; Busnatu, Ș.S.; Scafa-Udriște, A.; Andronic, O.; Lăcraru, A.-E.; Ioniță-Mîndrican, C.-B.; Lupuliasa, D.; Negrei, C.; Olteanu, G. Assessing Heavy Metal Contamination in Food: Implications for Human Health and Environmental Safety. Toxics 2025, 13, 333. [Google Scholar] [CrossRef]
- Madjar, R.M.; Scăețeanu, G.V. An Overview of Heavy Metal Contamination in Water from Agriculture: Origins, Monitoring, Risks, and Control Measures. Sustainability 2025, 17, 7368. [Google Scholar] [CrossRef]
- Shetty, B.R.; Jagadeesha, P.B.; Salmataj, S.A. Heavy metal contamination and its impact on the food chain: exposure, bioaccumulation, and risk assessment. CyTA - J. Food 2025, 23. [Google Scholar] [CrossRef]
- Angon, P.B.; Islam, S.; Kc, S.; Das, A.; Anjum, N.; Poudel, A.; Suchi, S.A. RETRACTED: Sources, effects and present perspectives of heavy metals contamination: Soil, plants and human food chain. Heliyon 2024, 10. [Google Scholar] [CrossRef]
- Koch, W.; Czop, M.; Iłowiecka, K.; Nawrocka, A.; Wiącek, D. Dietary Intake of Toxic Heavy Metals with Major Groups of Food Products—Results of Analytical Determinations. Nutrients 2022, 14, 1626. [Google Scholar] [CrossRef]
- Haidar, Z.; Fatema, K.; Shoily, S.S.; Sajib, A.A. Disease-associated metabolic pathways affected by heavy metals and metalloid. Toxicol. Rep. 2023, 10, 554–570. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; He, X.; LE, Y.; Al-Tohamy, R.; Ali, S.S. Potential applications of extremophilic bacteria in the bioremediation of extreme environments contaminated with heavy metals. J. Environ. Manag. 2024, 352, 120081. [Google Scholar] [CrossRef]
- Alawadhi, N.; Abass, K.; Khaled, R.; Osaili, T.M.; Semerjian, L. Heavy metals in spices and herbs from worldwide markets: A systematic review and health risk assessment. Environ. Pollut. 2024, 362, 124999. [Google Scholar] [CrossRef] [PubMed]
- Broek, S.v.D.; Nybom, I.; Hartmann, M.; Doetterl, S.; Garland, G. Opportunities and challenges of using human excreta-derived fertilizers in agriculture: A review of suitability, environmental impact and societal acceptance. Sci. Total. Environ. 2024, 957, 177306. [Google Scholar] [CrossRef] [PubMed]
- Ungureanu, E.L.; Mocanu, A.L.; Stroe, C.A.; Duță, D.E.; Mustățea, G. Assessing Health Risks Associated with Heavy Metals in Food: A Bibliometric Analysis. Foods 2023, 12, 3974. [Google Scholar] [CrossRef]
- Rostamzadeh, M.; Sadrabad, E.K.; Mohajeri, F.A.; Dehghani-Tafti, A.; Askari, E. Heavy Metals in Foods Consumed by Copper Miners: A Health Risk Assessment. Food Sci. Nutr. 2025, 13, e70267. [Google Scholar] [CrossRef]
- Rai, P.K.; Lee, S.S.; Zhang, M.; Tsang, Y.F.; Kim, K.-H. Heavy metals in food crops: Health risks, fate, mechanisms, and management. Environ. Int. 2019, 125, 365–385. [Google Scholar] [CrossRef]
- Rashid, A.; Schutte, B.J.; Ulery, A.; Deyholos, M.K.; Sanogo, S.; Lehnhoff, E.A.; Beck, L. Heavy Metal Contamination in Agricultural Soil: Environmental Pollutants Affecting Crop Health. Agronomy 2023, 13, 1521. [Google Scholar] [CrossRef]
- Xu, W.; Jin, Y.; Zeng, G. Introduction of heavy metals contamination in the water and soil: a review on source, toxicity and remediation methods. Green Chem. Lett. Rev. 2024, 17. [Google Scholar] [CrossRef]
- Fei, Y.; Hu, Y.H. Recent progress in removal of heavy metals from wastewater: A comprehensive review. Chemosphere 2023, 335, 139077. [Google Scholar] [CrossRef]
- Witkowska, D.; Słowik, J.; Chilicka, K. Heavy Metals and Human Health: Possible Exposure Pathways and the Competition for Protein Binding Sites. Molecules 2021, 26, 6060. [Google Scholar] [CrossRef]
- Shrestha, R.; Ban, S.; Devkota, S.; Sharma, S.; Joshi, R.; Tiwari, A.P.; Kim, H.Y.; Joshi, M.K. Technological trends in heavy metals removal from industrial wastewater: A review. J. Environ. Chem. Eng. 2021, 9. [Google Scholar] [CrossRef]
- Ye, M.; Huang, H. Transition metal oxide electrode materials in electrochemical sensors for heavy metal ions: cutting-edge developments and challenges. Talanta 2025, 298, 128994. [Google Scholar] [CrossRef] [PubMed]
- Akinyele, I.; Shokunbi, O. Comparative analysis of dry ashing and wet digestion methods for the determination of trace and heavy metals in food samples. Food Chem. 2015, 173, 682–684. [Google Scholar] [CrossRef]
- Wang, L.; Peng, X.; Fu, H.; Huang, C.; Li, Y.; Liu, Z. Recent advances in the development of electrochemical aptasensors for detection of heavy metals in food. Biosens. Bioelectron. 2020, 147, 111777. [Google Scholar] [CrossRef] [PubMed]
- Das, A.K.; Chakraborty, R.; Cervera, M.L.; de la Guardia, M. Metal speciation in biological fluids — a review. Microchim. Acta 1996, 122, 209–246. [Google Scholar] [CrossRef]
- Bunu, S.J.; Ebeshi, B.U.; Kpun, H.F.; Kashimawo, A.J.; Vaikosen, E.N.; Itodo, C.B. Atomic Absorption Spectroscopic (AAS) Analysis of Heavy Metals and Health Risks Assessment of some Common Energy Drinks. Pharmacol. Toxicol. Nat. Med. 2023, 1, 1–10. [Google Scholar] [CrossRef]
- Dong, Y.; Hao, J.; Wang, Y.; Cao, X.; Dong, Z.; Zhang, Z.; Liu, Y. Recent advances in polyoxometalate-based functional materials for sustainable remediation of radionuclide and heavy metal pollution. Chin. Chem. Lett. 2025. [Google Scholar] [CrossRef]
- Meng, R.; Zhu, Q.; Long, T.; He, X.; Luo, Z.; Gu, R.; Wang, W.; Xiang, P. The innovative and accurate detection of heavy metals in foods: A critical review on electrochemical sensors. Food Control 2023, 150. [Google Scholar] [CrossRef]
- Gori, A.; Armani, A.; Pedonese, F.; Benini, O.; Mancini, S.; Nuvoloni, R. Heavy metals (Pb, Cd, Ni) in insect-based products for human consumption sold by e-commerce in the EU market: Occurrence and potential health risk associated with dietary exposure. Food Control 2024, 167. [Google Scholar] [CrossRef]
- Cantoral, A.; Betanzos-Robledo, L.; Collado-López, S.; García-Martínez, B.A.; Lamadrid-Figueroa, H.; Mariscal-Moreno, R.M.; Díaz-Ruiz, A.; Ríos, C.; Téllez-Rojo, M.M. Lead Levels in the Most Consumed Mexican Foods: First Monitoring Effort. Toxics 2024, 12, 318. [Google Scholar] [CrossRef]
- Ávila, F.G.; Cabrera-Sumba, J.; Valdez-Pilataxi, S.; Villalta-Chungata, J.; Valdiviezo-Gonzales, L.; Alegria-Arnedo, C. Removal of heavy metals in industrial wastewater using adsorption technology: Efficiency and influencing factors. Clean. Eng. Technol. 2025, 24. [Google Scholar] [CrossRef]
- Barlow, N.L.; Bradberry, S.M. Investigation and monitoring of heavy metal poisoning. J. Clin. Pathol. 2022, 76, 82–97. [Google Scholar] [CrossRef] [PubMed]
- Guagliardi, I.; Ricca, N.; Cicchella, D. Comparative evaluation of ICP-MS and XRF techniques for screening potentially toxic elements in soil. Toxics 2025, 13. [Google Scholar] [CrossRef]
- Mao, Y.; Wang, M.; Wei, H.; Gong, N.; Wang, F.; Zhu, C. Heavy Metal Pollution and Risk Assessment of Vegetables and Soil in Jinhua City of China. Sustainability 2023, 15, 4241. [Google Scholar] [CrossRef]
- Azevedo, R.; Oliveira, A.R.; Almeida, A.; Gomes, L.R. Determination by ICP-MS of Essential and Toxic Trace Elements in Gums and Carrageenans Used as Food Additives Commercially Available in the Portuguese Market. Foods 2023, 12, 1408. [Google Scholar] [CrossRef] [PubMed]
- Alabi, O.O.; Akande, T.O.; Gbadeyan, O.J.; Deenadayalu, N. Advanced technologies for plastic waste recycling: examine recent developments in plastic waste recycling technologies. RSC Adv. 2025, 15, 40541–40557. [Google Scholar] [CrossRef] [PubMed]
- Kojo, K.; Oguri, T.; Tanaka, T.; Ikeda, A.; Shimizu, T.; Fujimoto, S.; et al. ICP-MS performance for measurement of key serum minerals. Journal of Clinical Laboratory Analysis 2025, 39. [Google Scholar]
- Czarnek, K.; Tatarczak-Michalewska, M.; Wójcik, G.; Szopa, A.; Majerek, D.; Fila, K.; Hamitoglu, M.; Gogacz, M.; Blicharska, E. Nutritional Risks of Heavy Metals in the Human Diet—Multi-Elemental Analysis of Energy Drinks. Nutrients 2024, 16, 4306. [Google Scholar] [CrossRef] [PubMed]
- Kaya, T.N.; Zurnacı, M.; Şener, İ. Determination of heavy metal accumulation in milk and packaging materials. Journal of Engineering Technology and Applied Sciences 2024, 9, 113–130. [Google Scholar] [CrossRef]
- Zahir, A.; Ge, Z.; Khan, I.A. Public Health Risks Associated with Food Process Contaminants – A Review. J. Food Prot. 2024, 88, 100426. [Google Scholar] [CrossRef]
- Sable, H.; Singh, V.; Kumar, V.; Roy, A.; Pandit, S.; Kaur, K.; Rustagi, S.; Malik, S. Toxicological and bioremediation profiling of nonessential heavy metals (mercury, chromium, cadmium, aluminium) and their impact on human health: A review. Toxicol. Anal. et Clin. 2024, 36, 205–234. [Google Scholar] [CrossRef]
- Hu, Y.; Wang, J.; Yang, Y.; Li, S.; Wu, Q.; Nepovimova, E.; Zhang, X.; Kuca, K. Revolutionizing soil heavy metal remediation: Cutting-edge innovations in plant disposal technology. Sci. Total. Environ. 2024, 918, 170577. [Google Scholar] [CrossRef] [PubMed]
- Bonthula, S.; Bonthula, S.R.; Pothu, R.; Srivastava, R.K.; Boddula, R.; Radwan, A.B.; Al-Qahtani, N. Recent Advances in Copper-Based Materials for Sustainable Environmental Applications. Sustain. Chem. 2023, 4, 246–271. [Google Scholar] [CrossRef]
- Gogoi, B.; Acharjee, S.A.; Bharali, P.; Sorhie, V.; Walling, B. A critical review on the ecotoxicity of heavy metal on multispecies in global context: A bibliometric analysis. Environ. Res. 2024, 248, 118280. [Google Scholar] [CrossRef]
- Hadian, Z.; Shariatifar, N.; Arabameri, M.; Moazzen, M.; Mousavi Khaneghah, A. Risk assessment of metal exposure from nuts consumption in Iran using ICP-OES. Journal of Agriculture and Food Research 2025, 21, 101865. [Google Scholar] [CrossRef]
- Hammani, O.; El-Otmani, N.; Ben Lenda, O.; El Azhari, H.; Rfaki, A.; Lahlouhi, N.; Mourad, Y.; Abdallaoui, A. Exploratory analysis of potential toxic elements in Moroccan couscous and health risk evaluation utilizing ICP-OES. J. Trace Elements Miner. 2024, 11. [Google Scholar] [CrossRef]
- López-Alonso, M.; Rivas, I.; Miranda, M. Trace Mineral Imbalances in Global Health: Challenges, Biomarkers, and the Role of Serum Analysis. Nutrients 2025, 17, 2241. [Google Scholar] [CrossRef]
- Ivanišević, I. The Role of Silver Nanoparticles in Electrochemical Sensors for Aquatic Environmental Analysis. Sensors 2023, 23, 3692. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, A.G.; Renu, K.; Gopalakrishnan, A.V.; Veeraraghavan, V.P.; Vinayagam, S.; Paz-Montelongo, S.; Dey, A.; Vellingiri, B.; George, A.; Madhyastha, H.; et al. Heavy Metal and Metalloid Contamination in Food and Emerging Technologies for Its Detection. Sustainability 2023, 15, 1195. [Google Scholar] [CrossRef]
- Tejada-Purizaca, T.R.; Garcia-Chevesich, P.A.; Ticona-Quea, J.; Martínez, G.; Martínez, K.; Morales-Paredes, L.; Romero-Mariscal, G.; Arenazas-Rodríguez, A.; Vanzin, G.; Sharp, J.O.; et al. Heavy Metal Bioaccumulation in Peruvian Food and Medicinal Products. Foods 2024, 13, 762. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, F.M.; Braunger, M.L.; Riul, A. Heavy Metal/Toxins Detection Using Electronic Tongues. Chemosensors 2019, 7, 36. [Google Scholar] [CrossRef]
- Xu, H.; Newton, R.; Love, D.C.; Zhao, Y.; Toppe, J.; Zhang, W. Heavy metal risks in aquatic foods. Environ. Int. 2025, 205, 109831. [Google Scholar] [CrossRef]
- Gu, M.; Huang, H.; Jiao, Q.; Ma, D.; Xu, Y.; Liu, C.; Tan, X. In situ multi-element soil analysis using LIBS. Journal of Analytical Atomic Spectrometry 2026. [Google Scholar] [CrossRef]
- Singh, R.; Gupta, R.; Bansal, D.; Bhateria, R.; Sharma, M. A Review on Recent Trends and Future Developments in Electrochemical Sensing. ACS Omega 2024, 9, 7336–7356. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, X.; Yang, M.; Li, N. Magnetic monitoring and source traceability of heavy metal pollution in urban topsoil. Sustainability 2025, 17. [Google Scholar] [CrossRef]
- Jordanova, N.; Petrovský, E.; Kapicka, A.; Jordanova, D.; Petrov, P. Magnetic methods for assessment of soil restoration near a metallurgical plant. Environmental Monitoring and Assessment 2017, 189. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Wu, J.; Shi, T.; Chen, C.; Li, J.; Wang, P.; Song, Y.; Yu, Q.; Zhu, Z. New magnetic proxies to reveal source and bioavailability of heavy metals in contaminated soils. J. Hazard. Mater. 2024, 479, 135665. [Google Scholar] [CrossRef]
- Jordanova, N.; Jordanova, D.; Tcherkezova, E.; Georgieva, B.; Ishlyamski, D. Advanced mineral magnetic and geochemical investigations of road dusts for assessment of pollution in urban areas near the largest copper smelter in SE Europe. Sci. Total. Environ. 2021, 792, 148402. [Google Scholar] [CrossRef]
- Ibrahim, A.Y.; Markandan, K.; Peter, A.P.; Tiong, Y.W.; Sankaran, R. Magneto-priming for Rice: Mechanisms, Agronomic Benefits, and Sustainability Potential. J. Crop. Heal. 2026, 78, 19. [Google Scholar] [CrossRef]
- Menshov, O.; Kruglov, O.; Vyzhva, S.; Horoshkova, L.; Pereira, P.; Pastushenko, T.; Dindaroglu, T. Landscape Position Effects on Magnetic Properties of Soils in the Agricultural Land Pechenigy, Ukraine. Earth Syst. Environ. 2021, 5, 739–750. [Google Scholar] [CrossRef]
- Ayoubi, S.; Abazari, P.; Zeraatpisheh, M. Soil great groups discrimination using magnetic susceptibility technique in a semi-arid region, central Iran. Arab. J. Geosci. 2018, 11, 616. [Google Scholar] [CrossRef]
- Hasani, S.; Asghari, O.; Ardejani, F.D.; Yousefi, S. Spatial modelling of hazardous elements at waste dumps using geostatistical approach: a case study Sarcheshmeh copper mine, Iran. Environ. Earth Sci. 2017, 76. [Google Scholar] [CrossRef]
- Zou, X.; Zhang, Z.; Wu, M.; Wan, Y. Slope-scale spatial variability of fractal dimension of soil particle size distribution at multiple depths. Soil Sci. Soc. Am. J. 2020, 85, 117–131. [Google Scholar] [CrossRef]
- Sharafi, A.; Beigi, H.; Rezaei, B.; Sargheini, J. The spatial modeling of hazardous elements in one of the Angouran mine waste dumps using a geostatistical approach. Journal of Mining Engineering 2020, 15, 41–49. [Google Scholar]
- Liu, X.; Wang, C.; Wang, W.; Qiu, Y.; Tang, Y.; Wang, C.; Li, H.; Li, G.; An, T. Combined pollution of heavy metals and polycyclic aromatic hydrocarbons in non-ferrous metal smelting wastewater treatment plant: Distribution profiles, removal efficiency, and ecological risks to receiving river. J. Hazard. Mater. 2025, 486, 137118. [Google Scholar] [CrossRef]
- Hou, R.; Liu, L.; Dong, Z.; Ma, F.; Liu, X.; Wang, Y.; Lai, J. Construction and verification of soil heavy metal establishment identification method based on dual-threshold of magnetic susceptibility. J. Hazard. Mater. 2025, 497, 139741. [Google Scholar] [CrossRef] [PubMed]
- Vodyanitskii, Y.N.; Shoba, S.A. Magnetic susceptibility as an indicator of heavy metal contamination of urban soils. Moscow University Soil Science Bulletin 2015, 70, 10–16. [Google Scholar] [CrossRef]
- Sudarningsih, S.; Pratama, A.; Bijaksana, S.; Fahruddin, F.; Zanuddin, A.; Salim, A.; et al. Magnetic susceptibility and heavy metal contents in river sediments, Indonesia. Heliyon 2023, 9. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Zhang, J.; Ma, R.; Luo, H.; Wan, T.; Yu, D.; Hong, Y. Worldwide Examination of Magnetic Responses to Heavy Metal Pollution in Agricultural Soils. Agriculture 2024, 14, 702. [Google Scholar] [CrossRef]
- Kapper, K.L.; Bautista, F.; Goguitchaishvili, A.; Bógalo, M.F.; Cejudo-Ruíz, R.; Solano, M.C. The use and misuse of magnetic methods to monitor urban pollution. Boletín de la Sociedad Geológica Mexicana 2020, 72, 1–44. [Google Scholar] [CrossRef]
- Frydrych, A.; Jurowski, K. Portable X-ray fluorescence as a tool for in situ food analysis. TrAC Trends in Analytical Chemistry 2023, 166. [Google Scholar] [CrossRef]
- Roberts, A.A.; Guimarães, D.; Tehrani, M.W.; Lin, S.; Parsons, P.J. Field-based evaluation of portable XRF for screening toxic metals in seafood. X-Ray Spectrometry 2024, 53, 506–519. [Google Scholar] [CrossRef] [PubMed]
- Sloan-Dennison, S.; Wallace, G.Q.; Hassanain, W.A.; Laing, S.; Faulds, K.; Graham, D. Advancing SERS as a quantitative technique: challenges, considerations, and correlative approaches to aid validation. Nano Converg. 2024, 11, 1–30. [Google Scholar] [CrossRef]
- Zhou, H.; Li, X.; Wang, L.; Liang, Y.; Jialading, A.; Wang, Z.; et al. Application of quantitative SERS methods in food safety detection. Reviews in Analytical Chemistry 2021, 40, 173–186. [Google Scholar] [CrossRef]
- Mousavizadegan, M.; Firoozbakhtian, A.; Hosseini, M.; Ju, H. Machine learning in analytical chemistry. TrAC Trends in Analytical Chemistry 2023, 167, 117216. [Google Scholar] [CrossRef]
- Wu, X.; Yuan, Z.; Gao, S.; Zhang, X.; El-Mesery, H.S.; Lu, W.; Dai, X.; Xu, R. Electrochemical Biosensors Driving Model Transformation for Food Testing. Foods 2025, 14, 2669. [Google Scholar] [CrossRef]
- Oladimeji, T.E.; Oyedemi, M.; Emetere, M.E.; Agboola, O.; Adeoye, J.B.; Odunlami, O.A. Impact of heavy metals from industrial wastewater and removal technologies. Heliyon 2024, 10. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Xuan, T.D. Heavy metals polluting drinking water and their individual health hazards. International Journal of Molecular Sciences 2025, 26. [Google Scholar] [CrossRef]
- Jomova, K.; Alomar, S.Y.; Nepovimova, E.; Kuca, K.; Valko, M. Heavy metals: toxicity and human health effects. Arch. Toxicol. 2024, 99, 153–209. [Google Scholar] [CrossRef] [PubMed]
- Onakpa, M.M.; Njan, A.A.; Kalu, O.C. A Review of Heavy Metal Contamination of Food Crops in Nigeria. Ann. Glob. Heal. 2018, 84, 488–494. [Google Scholar] [CrossRef] [PubMed]
- Castañeda-Miranda, A.G.; Chaparro, M.A.; Böhnel, H.N.; Castañeda-Miranda, R.; Pacheco-Castro, A.; Martinez-Fierro, M.L.; Solis-Sánchez, L.O.; Ornelas-Vargas, G. Bursera fagaroides bark as a bioindicator for air particle pollution using magnetic properties. J. South Am. Earth Sci. 2021, 108. [Google Scholar] [CrossRef]
- Castañeda-Miranda, A.G.; Böhnel, H.N.; Chaparro, M.A.E.; Pinedo-Torres, L.A.; Rodríguez-Trejo, A.; Castañeda-Miranda, R.; Sandoval-Aréchiga, R.; Rodríguez-Abdalá, V.I.; Gomez-Rodriguez, J.R.; Dávila-Cisneros, S.; et al. Magnetic Biomonitoring of PM in a Semi-Arid Urban Park of North-Central Mexico Using Tillandsia recurvata as a Particulate Matter Biocollector. Atmosphere 2025, 17, 55. [Google Scholar] [CrossRef]
- Hama Aziz, K.H.; Mustafa, F.S.; Omer, K.M.; Hama, S.; Hamarawf, R.F.; Rahman, K.O. Heavy metal pollution in aquatic environments and removal approaches. RSC Advances 2023, 13, 17595–17610. [Google Scholar] [CrossRef]
- vila Vázquez, V.; Aguilera Flores, M.M.; Veyna Robles, A.N.; Solís Lerma, L.E.; Sánchez Mata, O.; Durón Torres, S.M. Lead in fruit grown near mine tailings in Zacatecas, Mexico. Toxics 2025, 13. [Google Scholar] [CrossRef] [PubMed]
- Eddie-Amadi, B.F.; Vangone, R.; Guerretti, V.; Ozoani, H.A.; Okolo, K.O.; Awolayeofori, D.; Odinga-Israel, T.-B.; Nkpaa, K.W.; Sivieri, E.M.; Orisakwe, O.E.; et al. Ovary Metal Toxicity Remediation by Agro-Food Waste: Evidence for a Regulatory Mechanism of Oxidative Stress by Banana (Musa cavendish) Peel Extract. Antioxidants 2025, 14, 1129. [Google Scholar] [CrossRef]
- Bunu, S.J.; George, D.; Alfred-Ugbenbo, D.; Ebeshi, B.U. Heavy metals quantification and correlative carcinogenic-risks evaluation in selected energy drinks sold in Bayelsa state using atomic absorption spectroscopic technique. Int. J. Chem. Res. 2023, 1–4. [Google Scholar] [CrossRef]
- Salgado-Souto, S.A.; Escorcia-Reynoso, M.U.; Del Rio-Salas, R.; Talavera-Mendoza, O.; Bottini-Luzardo, M.B.; Núñez-Martínez, G.; Aguirre-Noyola, J.L.; Hernández-Flores, G. Assessing bioaccumulation of Pb, Cd, and As in goats: Impacts of exposure to mine tailings in a polymetallic mining region. J. South Am. Earth Sci. 2025, 153. [Google Scholar] [CrossRef]
- Fu, Z.; Xi, S. The effects of heavy metals on human metabolism. Toxicol. Mech. Methods 2019, 30, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Chen, P.; Yosri, N.; Chen, Q.; Elseedi, H.R.; Zou, X.; et al. Detection of heavy metals in food using SERS. Food Reviews International 2023, 39, 1440–1461. [Google Scholar] [CrossRef]
- Luo, Y.; Wang, Z.; He, X.; Hafeez, A.; Murtaza, G.; Deng, G.; Zaman, Q.U.; Xia, X.; Zhang, X.; Fahad, S.; et al. Soil application of vermicompost modulates physiological and biochemical responses enhancing lead tolerance in hemp cultivated in lead-contaminated soils. J. Hazard. Mater. Adv. 2025, 20. [Google Scholar] [CrossRef]
- Ramírez-Cota, M.; Escobar-Sánchez, O.; Betancourt-Lozano, M.; Frías-Espericueta, M.G.; Zamora-Arellano, N.Y.; Osuna-Martínez, C.C. Heavy metals in drinking water in northern Mexico. Journal of Water and Health 2025, 23, 684–700. [Google Scholar] [CrossRef]
- Pinedo-Torres, L.A.; Bonilla-Petriciolet, A.; García-Arreola, M.E.; Villagrana-Pacheco, Y.; Castañeda-Miranda, A.G.; Berber-Mendoza, M.S. Adsorption of arsenic, lead, cadmium and chromium using protonated chabazite. Adsorption Science and Technology 2023. [Google Scholar] [CrossRef]
- Yang, Y.; Hassan, M.F.; Ali, W.; Zou, H.; Liu, Z.; Ma, Y. Effects of Cadmium Pollution on Human Health: A Narrative Review. Atmosphere 2025, 16, 225. [Google Scholar] [CrossRef]
- Charkiewicz, A.E.; Omeljaniuk, W.J.; Nowak, K.; Garley, M.; Nikliński, J. Cadmium Toxicity and Health Effects—A Brief Summary. Molecules 2023, 28, 6620. [Google Scholar] [CrossRef]
- Sable, H.; Kumar, V.; Singh, V.; Rustagi, S.; Chahal, S.; Chaudhary, V. Advanced nanomaterials for heavy metal remediation from wastewater. Coordination Chemistry Reviews 2024, 518, 216079. [Google Scholar] [CrossRef]
- Zeng, S.; Liu, X.; Kafuti, Y.S.; Kim, H.; Wang, J.; Peng, X.; Li, H.; Yoon, J. Fluorescent dyes based on rhodamine derivatives for bioimaging and therapeutics: recent progress, challenges, and prospects. Chem. Soc. Rev. 2023, 52, 5607–5651. [Google Scholar] [CrossRef] [PubMed]
| Metal | Food or matrix | Maximum limit | Source | Remarks |
|---|---|---|---|---|
| Arsenic (As) | Edible fats and oils | 0.10 mg/kg | Codex 193-1995 Rev. 2024 | General limit for fats and oils |
| Salt and food-grade salt | 0.50 mg/kg | Codex 193-1995 Rev. 2024 | ||
| Husked rice | 0.35 mg/kg | EU 1881/2006 | ||
| Polished rice | 0.20 mg/kg | EU 1881/2006 | ||
| Natural mineral water | 0.01 mg/L | Codex 193-1995 Rev. 2024 | Most restrictive value | |
| Grains (wheat, maize, legumes) | 0.20 mg/kg | Codex / EU 2023/915 | ||
| Cadmio (Cd) | Natural mineral water | 0.003 mg/L | Codex 193-1995 | |
| Rice | 0.40 mg/kg | EU 1881/2006 | ||
| Leafy vegetables | 0.20 mg/kg | EU 1881/2006 | Higher bioaccumulation | |
| Legumes and tubers | 0.10 mg/kg | EU 1881/2006 | ||
| Chocolate (>50% cocoa) | 0.80 mg/kg | EU 1881/2006 | ||
| Chocolate (>70% cocoa) | 0.90 mg/kg | EU 1881/2006 | ||
| Seafood (mollusks, bivalves, cephalopods) | 2.00 mg/kg | Codex 193-1995 Rev. 2024 | ||
| Food-grade salt | 0.50 mg/kg | Codex 193-1995 Rev. 2024 | ||
| Wine | 0.01 mg/L | OIV 2021 | ||
| Plomo (Pb) | Infant foods | 0.01 mg/kg | Codex / EU 1881/2006 | Foods intended for infants and children up to 36 months |
| Milk and dairy products | 0.02 mg/kg | Codex / EU 1881/2006 | ||
| Fruits and vegetables | 0.10 mg/kg | Codex 193-1995 Rev. 2024 | ||
| Cereals and grains | 0.20 mg/kg | Codex 193-1995 Rev. 2024 | ||
| Meat (beef, pork, poultry) | 0.10 mg/kg | Codex 193-1995 Rev. 2024 | ||
| Fish | 0.30 mg/kg | Codex 193-1995 Rev. 2024 | ||
| Wine (year ≥ 2019) | 0.10 mg/L | OIV 2021 | Updated limit | |
| Wine-based spirits | 0.15 mg/L | OIV 2021 | ||
| Food-grade salt | 1.00 mg/kg | Codex 193-1995 Rev. 2024 | ||
| Mercurio (Hg) | Natural mineral water | 0.001 mg/L | Codex 193-1995 Rev. 2024 | |
| Food-grade salt | 0.10 mg/kg | Codex 193-1995 Rev. 2024 | ||
| Fish (general) | 0.50 mg/kg | Codex 193-1995 Rev. 2024 | Total Hg | |
| Methylmercury in large predatory fish (shark, marlin, tuna, etc.) | 1.0–1.7 mg/kg | Codex 193-1995 Rev. 2024 |
| Technique | Principle | Metals | LOD | Strengths | Limitations |
|---|---|---|---|---|---|
| AAS (FAAS / GF-AAS) | Atomic absorption by free atoms | Pb, Cd, Hg, As, Ni | µg/L–ng/L | Low cost; good precision; reference method | Single-element analysis; acid digestion required |
| ICP–MS | Plasma ionization with mass spectrometric detection | Pb, Cd, As, Hg, Ni | ng/L–pg/L | Ultra-high sensitivity; multi-element capability | High capital and operating costs; skilled personnel |
| ICP–OES | Optical emission of excited atoms in plasma | Pb, Cd, Ni, Cu, Zn | µg/L | Robust; multi-element; high throughput | Lower sensitivity than ICP–MS |
| LC–ICP–MS | Chromatographic separation coupled to ICP–MS | As species, Hg, Se | ng/L | Elemental speciation; high selectivity | High analytical complexity and cost |
| XRF | X-ray–induced fluorescence emission | Pb, Cd, Hg, As | mg/kg | Non-destructive; rapid; portable | Higher detection limits; limited trace sensitivity |
| Raman / SERS | Inelastic light scattering (surface-enhanced for SERS) | Pb, Cd, As (indirect) | ng/L (SERS) | Rapid; non-destructive; high chemical specificity | Limited quantitative reproducibility |
| Electronic tongues | Multisensor electrochemical pattern recognition | Pb2+, Cd2+, Cu2+ | nmol/L | Low cost; simultaneous multi-ion detection | Limited standardisation and regulatory validation |
| Electrochemical biosensors | Nanomaterial-enhanced electrochemical detection | Pb2+, Cd2+, Hg2+, As3+ | pg/L–ng/L | Portable; real-time; high sensitivity | Matrix effects; limited regulatory acceptance |
| In situ magnetic methods | Magnetic susceptibility as indirect proxy | Fe-linked metals; Pb, Cd (indirect) | Qualitative–semiquantitative | Rapid; non-destructive; low cost | Indirect signal; requires chemical correlation |
| Metal | Main food sources | Target organs | Health effects | Toxic mechanism |
|---|---|---|---|---|
| Pb | Rice, cereals, spices, vegetables | Nervous system, blood, kidney | Neurotoxicity, anemia, cognitive deficits | Oxidative stress, Ca2+ interference |
| Cd | Rice, legumes, vegetables, seafood | Kidney, liver, bone | Nephrotoxicity, bone loss, cancer | Bioaccumulation, mitochondrial damage |
| Hg | Fish, seafood, rice | CNS, kidney | Tremors, memory impairment | ROS generation, antioxidant inhibition |
| As | Rice, drinking water, seafood | Skin, lung, liver | Cancer, skin lesions, systemic toxicity | DNA damage, oxidative stress |
| Ni | Vegetables, cereals, water | Lung, skin | Allergy, carcinogenic potential | Protein binding, inflammation |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





