Submitted:
05 February 2026
Posted:
06 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methodology
2.1. Data Sources and Search Strategy
2.2. Eligibility Criteria
2.3. Study Selection Process
2.4. Data Extraction and Synthesis
2.5. Scope and Limitations
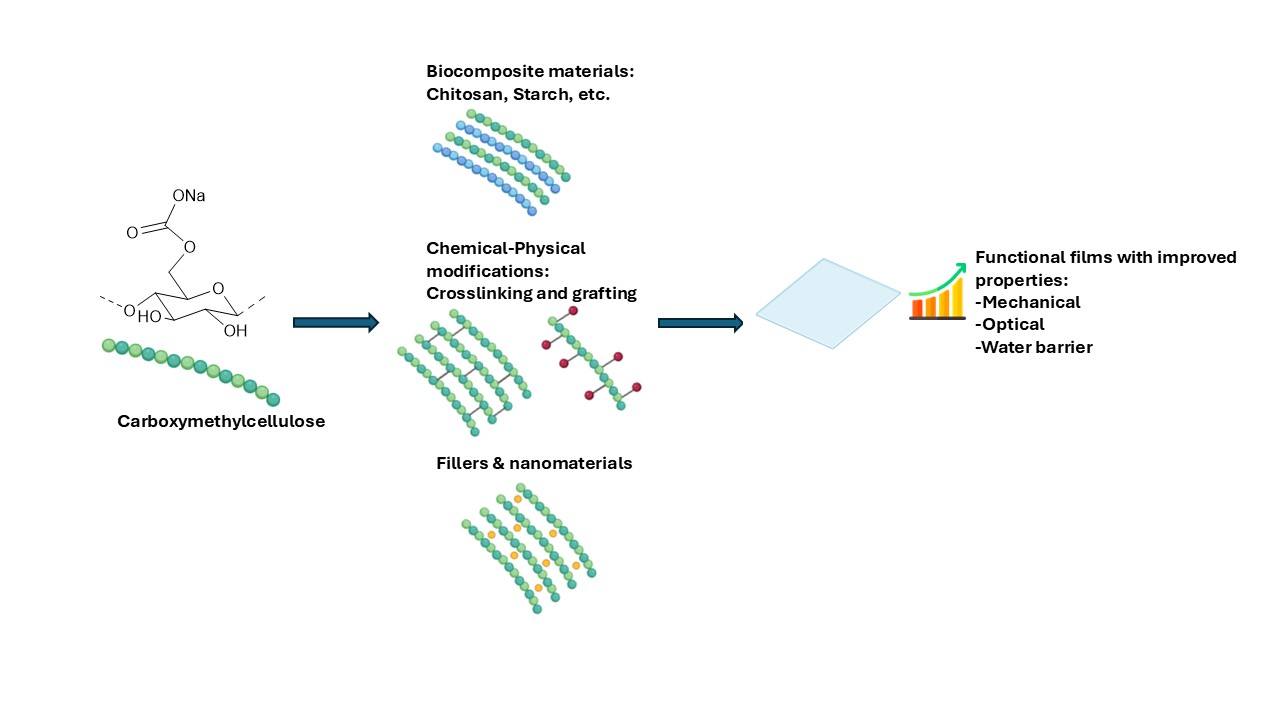
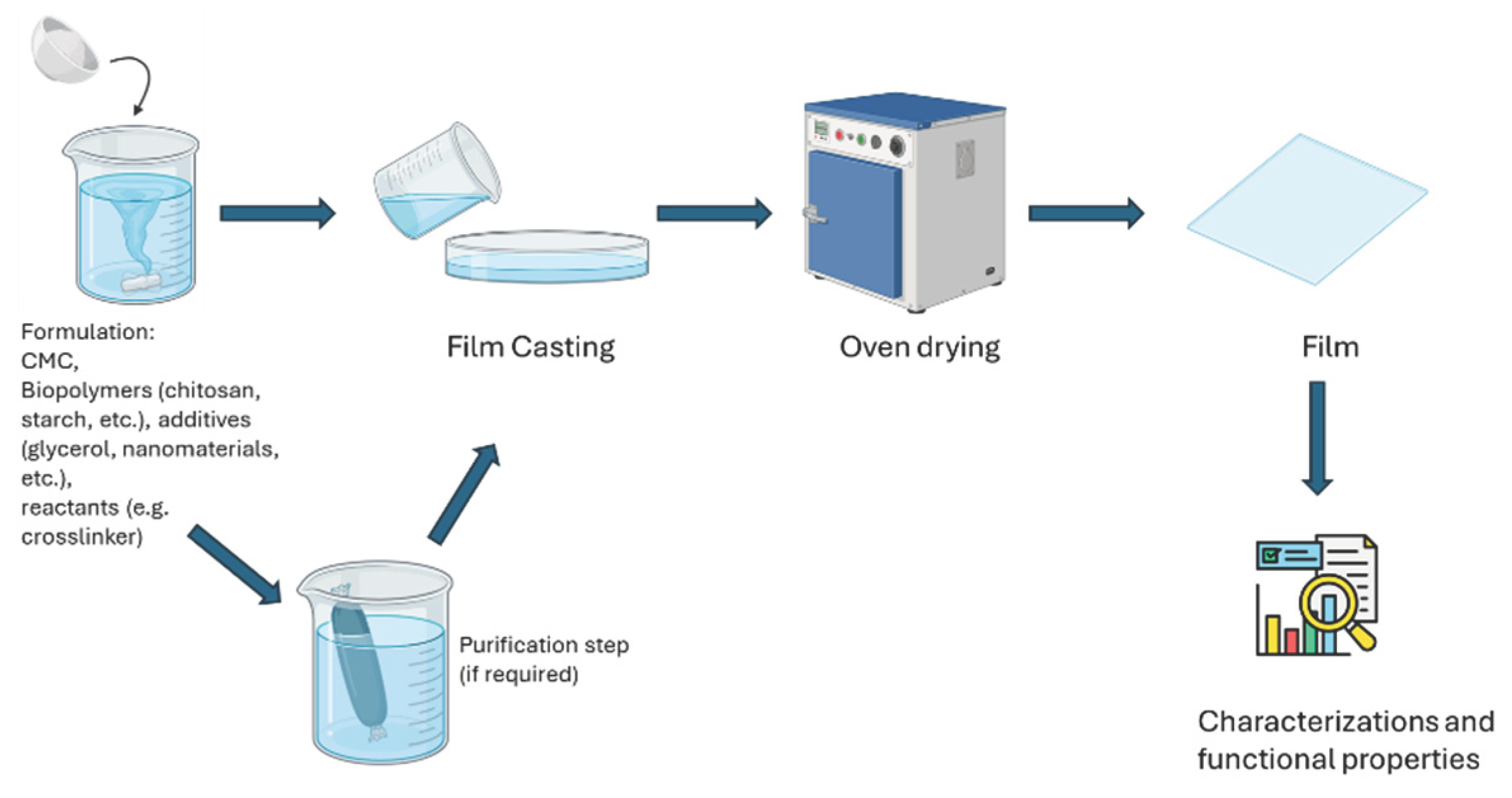
3. Preparation of CMC Films
4. Preparation of CMC Films
4.1. CMC Starch Polymer Blends
| Composition | MW(1) | DS(2) | TS(MPa)(3) | EB(%)(4) | E (MPa)(5) | Ɵ (°)(6) | WVP(7) | MC(%)(8) | Inh. Zone E. Coli(9) | Inh. Zone S. Aureus(9) | Ref |
|---|---|---|---|---|---|---|---|---|---|---|---|
| BOPP(10) | - | - | 120 | 150 | 102.1 | 25(11) | [97] | ||||
| LDPE(12) | - | - | 24 | 400 | 80 | 40(11) | |||||
| PLA(13) | - | - | 75 | 190 | 81 | 180(11) | |||||
| Mater Bi(14) | - | - | 30 | 280 | 90 | 220(11) | |||||
| Ecovio(15) | - | - | 25 | 500 | 75 | 520(11) | |||||
| CMC | - | - | 15.80±0.58 | 11.62±0.63 | 3.24×10-10 | 25.83±0.33 | - | - | [103] | ||
| CMC | - | - | 21.01±2.0 | 24.96 | 714.58±25.0 | 31.25±5.0 | 8.95×10-10 | 27.91±1.81 | - | - | [118] |
| CMC | - | - | 50.20±6.90 | 7.6±2.2 | 684.3±49.1 | 53.95±4.76(16) | - | - | - | - | [114] |
| CMC | - | - | 28.0±2.0 | 3.0±0.3 | 1700±50 | 380±5.0(11) | [119] | ||||
| CMC | - | - | 40.1±0.9 | 35.9±1.8 | 1040±40 | 39.2±1.8 | 1.40×10-9 | [120] | |||
| CMC | - | - | 21.85±3.12 | 23.42±2.0 | 385±63 | 9.2×10-11 | [69] | ||||
| CMC | - | - | 30.83±1.61 | 7.15±1.5 | [121] | ||||||
| CMC | - | - | 6.10±0.24 | 201.73±0.15 | 0.78×10-10 | [85] | |||||
| CS | - | - | 3.80±0.20 | 35.1±8.50 | 47.3±12.5 | 62.38±3.99(16) | 4.90×10-6 | [114] | |||
| CMC80/CS20 | - | - | 32.60±2.10 | 21.2±4.3 | 250.6±2.3 | 56.73±4.02(16) | 1.57×10-6 | - | - | - | |
| CMC60/CS40/ATH 0.9(17) | 26219 | 0.9 | 23.69±0.91 | 14.1±0.55 | - | - | - | 14.10±0.55 | - | - | [115] |
| CMC80/CS20/PSP 0.25(18) | 2.08±0.06 | 158.27±0.72 | 20.67±0.61 | [116] | |||||||
| CMC70/CSS30/Q/TBHQ(19) | 150 | - | [122] | ||||||||
| CMC50/CS50/LA 1.5 (20) | - | 0.9 | 4.80±0.56 | 73.17±2.90 | - | - | 5.56×10-11 | 23.67 | - | 4.17±0.21 | [123] |
| CMC50/CS50/Men2/Cur2(21) | 250 | 0.7 | 36.57±0.10 | 28.10±0.09 | 75.30±5.00 | 2.77×10-11 | 17.5 | 16.5 | [124] | ||
| CMC70/CHI30/OA50/CEO3(22) | 41 | - | 3.1±0.4 | 124.3±9.3 | 35.6±2.3 | 0.27×10-7 | [125] | ||||
| CMC70/CHI30/OA50/GEO3(22) | 41 | - | 2.7±0.4 | 61.4±8.8 | 29.3±1.9 | 0.99×10-7 | |||||
| CMC60/CHI40/EO12(23) | - | - | 5.83±0.30 | 51.20±6.97 | 0.12±0.02 | 119.0 | 19.21±1.10 | 7.19 | [126] | ||
| CMC/HTCC(24) | - | - | 21.82 | 2.27 | 28.61±0.37 | 117.0 | [127] | ||||
| CMC90/HTCC10/cry-CaCO3 4(24) | - | - | 35.02±1.68 | 14.43±0.65 | 18.72±0.85 | 68.0±2.6 | 5.9×10-11 | [128] | |||
| CMC/HTCMCh1(25) | - | - | 12.5±2.0 | 15.0±1.5 | 20±0.5 | 70.60±0.61 | 10.0±0.5 | 11.0±0.1 | [129] |
4.2. CMC Chitosan Polymer Blends
4.3. CMC Polymer Blends with Other Polysaccharides or Gelatin
| Composition | MW(1) | DS(2) | TS (MPa)(3) | EB(%)(4) | E (MPa)(5) | Ɵ (°)(6) | WVP(7) | MC(%)(8) | Inh. Zone E. Coli(9) | Inh. Zone S. Aureus(9) | Ref |
|---|---|---|---|---|---|---|---|---|---|---|---|
| CMC90/SCE10(10) | - | - | 22.33±2.35 | 6.56±0.86 | 111.48±3.38 | 3.64×10-10 | 20.97±0.82 | 11.07±0.78 | 14.25±0.37 | [102] | |
| CMC70/Gly30/CHPS 1(11) | - | - | 31.0±0.56 | 5.96±0.76 | 1.23×10-10 | 20.34±0.69 | 10.55±0.69 | 13.82±0.44 | [103] | ||
| CMC50/SA50(12) | - | - | 4.29±0.69 | 27.50±2.08 | [147] | ||||||
| CMC50/SA50/EGCG 040(13) | - | - | 10.78±0.15 | 11.20±1.57 | |||||||
| CMC50/Agar50 | - | - | 5.5 | 40 | 42.4±2.4 | 2.6×10-10 | [148] | ||||
| CMC50/Agar50/SSEO 1(14) | - | - | 7.5 | 55 | 58.3±2.4 | 3.0×10-10 | 33.45±2.76 | ||||
| CMC90/MLP10(15) | - | - | 34.86 | 14.23 | 970 | 65.45±5.0 | 5.21×10-10 | 14.12±1.70 | 11.75 | 9.41 | [118] |
| CMC90/P10/TO3(16) | - | - | 70.06 | 13.39 | 533.0 | 0.34×10-10 | [149] | ||||
| CMC70/Gelatin30/CaCl2 5 | - | - | 53.91±1.69 | 12.26±5.05 | 1.27×10-10 | [92] | |||||
| CMC85/SA/CS15 | - | - | 65.32±14.31 | 17.85±3.86 | 8.98±1.51 | 12.5 | 10.0 | [150] | |||
| SA/CMC/PS 1/1/1(17) | - | - | 31.95±0.66 | 13.78±0.44 | 62.79±1.00 | 2.26×10-9 | 16.21±0.34 | [64] |
4.4. CMC Ternary Polymer Blends
4.5. CMC Polymer Blends Containing Nano Polymer Compounds
| Composition | MW(1) | DS(2) | TS (MPa)(3) | EB(%)(4) | E (MPa)(5) | Ɵ (°)(6) | WVP(7) | MC(%)(8) | Inh. Zone S. Aureus(9) | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| CMC50/NC50(10) | - | - | 42.5±1.5 | 1.3±0.5 | 3750±100 | 4.32×10-11 | [119] | |||
| CMC50/NC50(10) | 80 | 0.8 | 34.86±2.90 | 2308±143 | 72.0±10.0 | 6.18×10-11 | [90] | |||
| CMC/CMC-NF4(11) | 226 | 0.8 | 52.5±2.0 | 3.5±0.3 | [121] | |||||
| CMC90/CNF10(12) | - | - | 49.2±2.1 | 26.6±2.2 | 1330±200 | 23.9±1.7 | 1.44×10-9 | [120] | ||
| CMC/CL-CNF(13) | - | - | 81.5±3.6 | 22.9±2.1 | 2637±59 | 31.5±4.5 | 1.75×10-9 | [157] | ||
| CMC70/Gly30/CHI-NC5/GSE(14) | 250 | 0.9 | 51.0±0.9 | 14.2±1.2 | 1900±4 | 1.36×10-9 | [158] | |||
| CMC/CHPS-NC5(15) | - | - | 32.95±4.06 | 20.32±0.32 | 1100±76.78 | 4.20×10-11 | [69] | |||
| CMC/NC1 | - | - | 12.3±0.3 | 89.53±0.18 | 0.28×10-10 | 22.0±1.3 | [85] | |||
| CMC/NC0.5/NCHI0.5(16) | - | - | 9.95±0.45 | 4.96±0.11 | 0.11×10-10 | 15.0±1.0 | 3.66±0.57 | |||
| CMC70/CS30/SB-NC2.5 | - | - | 99.06±4.95 | 20.88±0.83 | 1375.34±68.80 | 3.50×10-7 | [159] | |||
| CMC50/Agar50/OP-NC5/ShK10(17) | 250 | 0.9 | 61.7±5.2 | 8±2 | 2600±200 | 50±4 | 0.81×10-9 | [98] | ||
| CMC70/TS30/CHI-NP(18) | 700 | 0.9 | 81.08 | 11.3 | 3.57×10-7 | 12.17±0.89 | [87] | |||
| CMC50/Gelatin50/GMMT3/ATH1/PLE1.5(19) | 700 | 0.9 | 99.5±0.5 | 5.69×10-10 | 10.0±0.34 | [160] |
4.6. CMC Polymer Blends Containing Metal Nanoparticles
| Composition | MW(1) | DS(2) | TS (MPa)(3) | EB(%)(4) | E (MPa)(5) | Ɵ (°)(6) | WVP(7) | MC(%)(8) | Inh. Zone E. Coli(9) | Inh. Zone S. Aureus(9) | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|---|
| CMC70/CHI30/OA50 | 50-190 | 0.7-0.8 | 7.34±0.8 | 13.14±5.27 | 55.9±2.8 | 22.9±2.3 | [164] | ||||
| CMC70/CHI30/OA50/ZnO-NP1 | 50-190 | 0.7-0.8 | 4.38±0.59 | 42.37±3.82 | 30.27±3.84 | 47.40±1.25 | 8.27×10-7 | ||||
| CMC50/CHI50/ZnO-NP8 | 420000 | 0.7 | 12.6 | 95.6 | 9.0 | 11.0 | [165] | ||||
| CMC50/PVA50/Gly25 | 240.2 | - | 15.80 | 80.0 | [166] | ||||||
| CMC50/PVA50/Gly25/ZnO-NP 0.5 | 240.2 | - | 35.50 | 220.0 | 0.85±0.22 | 1.45±0.27 | |||||
| CMC50/PVA50 | - | 0.7 | 25 | 22 | 15 | 61.66±0.57 | [167] | ||||
| CMC50/PVA50/TiO2-NP 1/SiME 5(10) | - | 0.7 | 33 | 47 | 30 | 25.66±0.57 | |||||
| CMC80/CS20 | - | - | 20.83±2.00 | 25.87±3.09 | 45.50 | 1.49×10-10 | [104] | ||||
| CMC80/CS20/ZnO-NP3 | 14.91±2.50 | 31.29±2.90 | - | 87.85±1.43 | 1.18×10-10 | 14.76±0.55 | 27.92±0.13 | 25.27±0.26 | |||
| CMC80/CS20/ZnO-NP3/ATH 0.1g | 13.19±1.69 | 32.14±2.01 | - | 88.52±0.72 | 9.64×10-11 | 13.90±0.63 | 31.15±0.21 | 28.56±0.45 | |||
| CMC80/PS20(11) | 250 | 0.9 | 16.0±1.0 | 11.0±5.0 | 410(12) | [106] | |||||
| CMC80/PS20/Te-NP1/UDA1(11) | 250 | 0.9 | 14.85±0.32 | 12.45±3.85 | 374.2(12) |
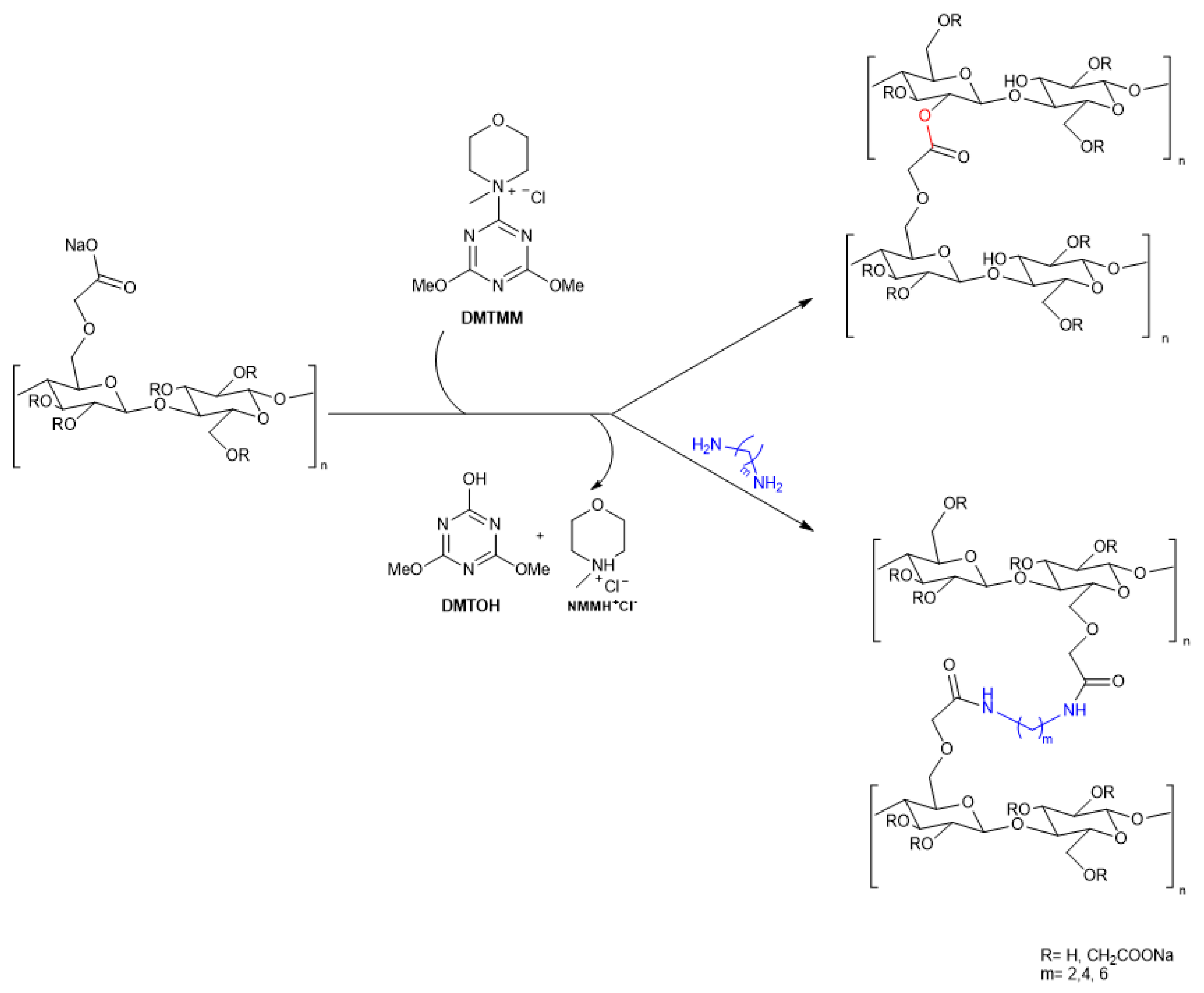
4.7. CMC Films Obtained by Crosslinking
4.7.1. CMC Films Obtained by Direct Cross-Linking
| Composition | MW(1) | DS(2) | TS (MPa)(3) | EB(%)(4) | E (MPa)(5) | Ɵ (°)(6) | WVP(7) | MC(%)(8) | Inh. Zone E. Coli(9) | Inh. Zone S. Aureus(9) | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|---|
| CMC/Gly50/DMTMM10(12) | 90 | 0.7 | 52.0±3.0 | 37.0±1.5 | 1.09×10-7 | 45.59±2.01 | [46] | ||||
| CMC/DMTMM1/EDA0.5 | 90 | 0.7 | 75±2.9 | 4.7±0.6 | 2.67±0.21x10-10 | 38.5±4.3 | [45] | ||||
| CMC/CA20 | 0.84 | 320(10) | 20±2 | [175] | |||||||
| CMC50/GG50/CA10(11) | - | - | 308±48(10) | [190] | |||||||
| CMC50/CS50/Gly15 | - | 0.25 | 6.19 | 55.48 | 13.37 | 1.05×10-10 | 16.00 | [191] | |||
| CMC50/CS50/Gly15/CA1.3 | - | 0.25 | 7.36 | 77.82 | 23.32 | 6.42×10-11 | 13.37 | ||||
| CMC/H2SO4 5 | - | - | 29.6±5.0 | 80.0 | [176] | ||||||
| CMC70/CHI30/CEO/OA/GA | 41 | - | 7.99±0.97 | 66.97±3.85 | 6.23×10-6 | [139] | |||||
| CMC60/PVA40 | - | - | 45.0 | 22.0 | 3.0×10-10 | [177] | |||||
| CMC60/PVA40/DCNC 3(13) | - | - | 90.0 | 7.0 | 2.0×10-10 | [177] | |||||
| CMC2/PVA1/GA1/PEI0.5 | - | - | 65.17 | 7.12 | 2800 | 85 | 0.3×10-10 | [178] | |||
| CMC8/CNF1/Gly/GA5/TP10(14) | - | - | 112.60 | 4.12 | 85.25±2.5 | [192] | |||||
| CMC80/NMMT20/CA20/TiO2 1 | 700 | 0.9 | 106.83±5.00 | 2.6±1.0 | 31.5±0.50 | 28.67±1.04 | [193] | ||||
| CMC50/CHI50/CA30/Gly38 | 150-250 | 0.55-0.65 | 7.12 | 35.95 | 0.22 | 45.19 | 2.10×10-12 | [194] | |||
| CMC50/CHI50/CA30/Gly38/ZnO-NP3 | 150-250 | 0.55-0.65 | 7.54 | 29.27 | 0.96 | 60.43 | 1.11×10-12 |
4.7.2. CMC Crosslinked Polymer Blends
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ATH | Anthocyanins |
| BBO | Bee bread oil |
| CA | Citric acid |
| CEO | Cinnamon essential oil |
| CESO | Clove essential oil |
| CHI | Chitosan |
| CHI-NF | Chitosan nanofibers |
| CHI-NP | Chitosan nanoparticles |
| CHPS | Chickpea hull polysaccharides |
| CHPS-NCs | Chickpea hull polysaccharide nanocrystals |
| CL-CNFs | Cotton linter cellulose nanofibrils |
| CMC | Carboxymethyl cellulose |
| CMC-NCs | Carboxymethyl cellulose nanocrystals |
| CMC-NFs | Carboxymethyl cellulose nanofibrils |
| CMP | Commiphora mukul polysaccharide |
| CNC/CNCs | Cellulose nanocrystal(s) |
| CNF/CNFs | Cellulose nanofiber(s) |
| CS | Corn starch |
| CIEO | Cinnamon essential oil (nano emulsion form) |
| DCNC | Dialdehyde cellulose nanofibers |
| DMTOH | 2,4-Dimethoxy-6-hydroxy-1,3,5-triazine |
| DMTMM | 4-(4,6-Dimethoxy-1,3,5-triazin-2-yl)-4-methylmorpholinium chloride |
| DS | Degree of substitution |
| EB% | Elongation at break |
| EGCG | Epigallocatechin gallate |
| EON | Essential oil nano emulsion |
| FT-IR | Fourier-transform infrared spectroscopy |
| GA | Glutaraldehyde |
| GEO | Ginger essential oil |
| GG | Guar gum |
| GMMT | Gelatin-modified montmorillonite |
| HTCC | N-(2-hydroxypropyl)-3-trimethylammonium chitosan chloride |
| HTCMCh | N-2-hydroxylpropyl-3-trimethylammonium-O-carboxymethyl chitosan |
| LA | Lactococcus lactis |
| MC% | Moisture content % |
| MLP | Mulberry leaf polysaccharides |
| MS-Q188 | Quaternized starch (Q188 grade) |
| MU% | Moisture uptake |
| NC | Nanocellulose |
| NCHI | Nanochitosan |
| NMMT | Nano-montmorillonite |
| OA | Oleic acid |
| OP-NC | Onion peel nanocellulose |
| OTR | Oxygen transmission rate |
| PEC | Polyelectrolyte complex |
| PE | Polyethylene |
| PEI | Polyethyleneimine |
| PLE | Pistacia leave extract |
| PRISMA | Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
| PS | Potato starch |
| PSP | Purple sweet potato pigment |
| PVA | Poly(vinyl alcohol) |
| RSM | Response surface methodology |
| SA | Sodium alginate |
| SB-NC | Sugarcane bagasse cellulose nanocrystals |
| SCG | Spent coffee grounds |
| SEM | Scanning electron microscopy |
| ShK | Shikonin |
| SSEO | Summer savory essential oil |
| TBHQ | tert-Butylhydroquinone |
| TEMPO | (2,2,6,6-Tetramethylpiperidin-1-yl)oxyl radical |
| TEMPO-CNF | TEMPO-oxidized cellulose nanofibers |
| TO | Thymol oil |
| TP | Tea polyphenols |
| TS | Tensile strength |
| UV | Ultraviolet |
| WVTR | Water vapor transmission rate |
| WVP | Water vapor permeability |
| WU% | Water uptake percentage |
| ZnO-NPs | Zinc oxide nanoparticles |
References
- Jambeck, J.R.; Geyer, R.; Wilcox, C.; Siegler, T.R.; Perryman, M.; Andrady, A.; Narayan, R.; Law, K.L. Plastic Waste Inputs from Land into the Ocean. Science 2015, 347, 768–771. [Google Scholar] [CrossRef]
- Basumatary, T.; Dey, R.; Nava, A.R.; Irnidayanti, Y.; Narayan, M.; Sarma, H. Micro, Nano, and Biodegradable Plastics: Hidden Threats to Plant Health and Soil Function. Rev. Environ. Contam. Toxicol. 2026, 264, 2. [Google Scholar] [CrossRef]
- Luo, D.; Chu, X.; Wu, Y.; Wang, Z.; Liao, Z.; Ji, X.; Ju, J.; Yang, B.; Chen, Z.; Dahlgren, R.; et al. Micro- and Nano-Plastics in the Atmosphere: A Review of Occurrence, Properties and Human Health Risks. J. Hazard. Mater. 2024, 465, 133412. [Google Scholar] [CrossRef]
- Rehman, M.U.; Nabi, S.U.; Yatoo, A.M.; Ali, S.; Raina, A.A.; Hussain, I.; Rashid, S.; Mushtaq, S.; Masoodi, M.H. Microplastic (MP) Pollution: Environmental Fate, Eco-Toxicity and Sustainable Remediation. Water. Air. Soil Pollut. 2024, 235, 384. [Google Scholar] [CrossRef]
- Menon, V.; Sharma, S.; Sharma, D.; Gupta, S. The Environmental Journey of Micro- and Nanoplastics: Fate, Transport, and Toxicity. Environ. Monit. Assess. 2026, 198, 130. [Google Scholar] [CrossRef] [PubMed]
- Wijesinghe, S.M.K.; Karunarathna, B.G.H.D.; Kumarawansha, W.K.M.G.; Liyanage, K.L.A.A.R.; Pahathkumbura, P.H.M.S.R.; Rajawardhana, A.G.M.I.; Samaranayake, W.M.N.K.; Sawmiya, M.I.F.; Wetthasinghe, K.S.; Ratnayake, A.S. Marine Pollution: The Global Challenge of Ocean Contaminants and Mitigation Efforts. Anthr. Coasts 2026, 9, 5. [Google Scholar] [CrossRef]
- Plastics the Fast Facts 2025. Available online: https://plasticseurope.org/wp-content/uploads/2025/09/PE_TheFacts_25_digital-1pager-scrollable.pdf.
- UNEP Plastic Pollution. Available online: https://www.unep.org/topics/chemicals-and-pollution-action/plastic-pollution.
- Visco, A.; Scolaro, C.; Facchin, M.; Brahimi, S.; Belhamdi, H.; Gatto, V.; Beghetto, V. Agri-Food Wastes for Bioplastics: European Prospective on Possible Applications in Their Second Life for a Circular Economy. Polymers 2022, 14, 2752. [Google Scholar] [CrossRef]
- Lambert, S.; Wagner, M. Environmental Performance of Bio-Based and Biodegradable Plastics: The Road Ahead. Chem. Soc. Rev. 2017, 46, 6855–6871. [Google Scholar] [CrossRef]
- Sokra, I.; Meta, H. Bioplastics in the Circular Bioeconomy: Production Pathways, Biodegradation Mechanisms, and Environmental Implications. J. Agric. Technol. 2026, 2, 23–35. [Google Scholar] [CrossRef]
- French, A.D. Glucose, Not Cellobiose, Is the Repeating Unit of Cellulose and Why That Is Important. Cellulose 2017, 24, 4605–4609. [Google Scholar] [CrossRef]
- Chi, K.; Catchmark, J.M. Improved Eco-Friendly Barrier Materials Based on Crystalline Nanocellulose/Chitosan/Carboxymethyl Cellulose Polyelectrolyte Complexes. Food Hydrocoll. 2018, 80, 195–205. [Google Scholar] [CrossRef]
- Putri, Rr.D.A.; Universitas Negeri Semarang, Indonesia; Anggraini, P.D.; Universitas Negeri Semarang, Indonesia Physical Properties of Edible Sorgum Starch Film Added with Carboxymethyl Cellulose. J. Phys. Sci. 2018, 29, 185–194. [CrossRef]
- Moustafa, H.; Nasr, H.E.; Youssef, A.M. Development of Antibacterial Carboxymethyl Cellulose/Quaternized Starch Bionanocomposites Based on Cinnamon Essential Oil Nanoemulsion for Wound Healing Applications. Biomass Convers. Biorefinery 2024, 14, 27477–27489. [Google Scholar] [CrossRef]
- Ahamed, A.; Loganathan, V.; Mullaivendhan, J.; Alodaini, H.A.; Akbar, I. Synthesis of Chitosan and Carboxymethyl Cellulose Connect Flavonoid (CH-Fla-CMC) Composite and Their Investigation of Antioxidant, Cytotoxicity Activities. Int. J. Biol. Macromol. 2025, 300, 140081. [Google Scholar] [CrossRef]
- Karmazsin, E. Thermal Analysis in the Cellulose, Paper and Textile Industry. Thermochim. Acta 1987, 110, 471–475. [Google Scholar] [CrossRef]
- Li, Y.; Liu, W.; Wang, Y.; Lu, S. Cellulose Based Nano-Scaffolds for Targeted Cancer Therapies: Current Status and Future Perspective. Int. J. Nanomedicine 2025, 20, 199–213. [Google Scholar] [CrossRef] [PubMed]
- More, V.R.; Antanitta, S.V.; Khonde, R.; Kandasubramanian, B. Cellulose and Derivatives Serving as Natural, Versatile and Biocompatible Polymers in Biomedical Applications. Int. J. Polym. Mater. Polym. Biomater. 2025, 74, 923–937. [Google Scholar] [CrossRef]
- Awere, C.O.; Anadebe, V.C.; Sabapathi, D.; Mounish, B.S.C.; Rakkammal, K.; Muthuramalingam, P.; Varadharajan, V.; Arinzechukwu, M.C.; Kumar, P.; Ebenso, E.E.; et al. From Waste to Wealth: Plant-Derived Cellulose Nanocrystal Hydrogel for Anticancer, Antibacterial, and Anticorrosion Studies. Carbohydr. Polym. Technol. Appl. 2025, 9, 100656. [Google Scholar] [CrossRef]
- Marinho, E. Cellulose: A Comprehensive Review of Its Properties and Applications. Sustain. Chem. Environ. 2025, 11, 100283. [Google Scholar] [CrossRef]
- Ates, B.; Koytepe, S.; Ulu, A.; Gurses, C.; Thakur, V.K. Chemistry, Structures, and Advanced Applications of Nanocomposites from Biorenewable Resources. Chem. Rev. 2020, 120, 9304–9362. [Google Scholar] [CrossRef]
- Trache, D.; Thakur, V.K.; Boukherroub, R. Cellulose Nanocrystals/Graphene Hybrids—A Promising New Class of Materials for Advanced Applications. Nanomaterials 2020, 10, 1523. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, V.; Thakur, A. Applications of Biodegradable Carboxymethyl Cellulose-Based Composites. Results Mater. 2023, 20, 100481. [Google Scholar] [CrossRef]
- Xu, Y.; Deng, Q.; Ruan, C.; Xu, D.; Zeng, K. Application of Carboxymethyl Cellulose and Its Edible Composite Coating in Fruit Preservation. Packag. Technol. Sci. 2024, 37, 781–792. [Google Scholar] [CrossRef]
- Cheng, J.; Gao, R.; Zhu, Y.; Lin, Q. Applications of Biodegradable Materials in Food Packaging: A Review. Alex. Eng. J. 2024, 91, 70–83. [Google Scholar] [CrossRef]
- Liu, Y.; Ahmed, S.; Sameen, D.E.; Wang, Y.; Lu, R.; Dai, J.; Li, S.; Qin, W. A Review of Cellulose and Its Derivatives in Biopolymer-Based for Food Packaging Application. Trends Food Sci. Technol. 2021, 112, 532–546. [Google Scholar] [CrossRef]
- Liu, X.; Qin, Z.; Ma, Y.; Liu, H.; Wang, X. Cellulose-Based Films for Food Packaging Applications: Review of Preparation, Properties, and Prospects. J. Renew. Mater. 2023, 11, 3203–3225. [Google Scholar] [CrossRef]
- Liu, X.; Xu, Y.; Liao, W.; Guo, C.; Gan, M.; Wang, Q. Preparation and Characterization of Chitosan/Bacterial Cellulose Composite Biodegradable Films Combined with Curcumin and Its Application on Preservation of Strawberries. Food Packag. Shelf Life 2023, 35, 101006. [Google Scholar] [CrossRef]
- Chen, L.; Yu, L.; Qi, L.; Eichhorn, S.J.; Isogai, A.; Lizundia, E.; Zhu, J.Y.; Chen, C. Cellulose Nanocomposites by Supramolecular Chemistry Engineering. Nat. Rev. Mater. 2025, 10, 728–749. [Google Scholar] [CrossRef]
- Rahman, A.F.A.; Arsad, A.; Abdurrahman, M.; Sidek, A. Sustainable Sensor Technologies: Intrinsically Conductive Polymer-Biowaste Cellulose Nanocomposites. Cellulose 2025, 32, 3529–3566. [Google Scholar] [CrossRef]
- Rhim, J.-W.; Ng, P.K.W. Natural Biopolymer-Based Nanocomposite Films for Packaging Applications. Crit. Rev. Food Sci. Nutr. 2007, 47, 411–433. [Google Scholar] [CrossRef]
- Antony Jose, S.; Cowan, N.; Davidson, M.; Godina, G.; Smith, I.; Xin, J.; Menezes, P.L. A Comprehensive Review on Cellulose Nanofibers, Nanomaterials, and Composites: Manufacturing, Properties, and Applications. Nanomaterials 2025, 15, 356. [Google Scholar] [CrossRef]
- Chandrika, K.S.V.P.; Singh, A.; Prasad, R.D.; Yadav, P.; Dhara, M.; Kavya, M.; Kumar, A.; Gopalan, B. Porous Crosslinked CMC-PVA Biopolymer Films: Synthesis, Standardization, and Application in Seed Coating for Improved Germination. Carbohydr. Polym. Technol. Appl. 2025, 11, 100900. [Google Scholar] [CrossRef]
- Yuan, H.; Wei, X.; Chen, C.; Yu, H.; Huang, J.; Tian, H. Recent Developments in Cellulose-Based Emulsion Systems for Food Applications: A Review. Food Hydrocoll. 2025, 163, 111089. [Google Scholar] [CrossRef]
- Sharma, C.; Raza, M.A.; Purohit, S.D.; Pathak, P.; Gautam, S.; Corridon, P.R.; Han, S.S. Cellulose-Based 3D Printing Bio-Inks for Biomedical Applications: A Review. Int. J. Biol. Macromol. 2025, 305, 141174. [Google Scholar] [CrossRef]
- Poudel, J.; Bhattarai, S.; Tara, A.; Rahman, Md.F.; Nath, N.; Tanti, B. Recent Progress in Microcrystalline Cellulose for Solar Cell Applications: A Comprehensive Review. J. Opt. 2025. [Google Scholar] [CrossRef]
- Abdullah; Cai, J.; Hafeez, M.A.; Wang, Q.; Farooq, S.; Huang, Q.; Tian, W.; Xiao, J. Biopolymer-Based Functional Films for Packaging Applications: A Review. Front. Nutr. 2022, 9, 1000116. [Google Scholar] [CrossRef] [PubMed]
- Pawase, P.A.; Rout, S.; Tripathy, S.; Pathare, A.M.; Srivastav, P.P.; Bashir, O.; Panghal, A. Recent Advances in Cellulose, Chitosan, and Protein-Based Edible Films for Sustainable Food Packaging: A Comprehensive Review. Int. J. Biol. Macromol. 2025, 321, 146172. [Google Scholar] [CrossRef]
- Elfawal, G.F.; Šišková, A.O.; Andicsová, A.E. Ecological Packaging and Creating Sustainable Solutions for Biodegradable Cellulose Derivatives: A Review. Int. J. Biol. Macromol. 2025, 309, 142348. [Google Scholar] [CrossRef]
- Santandrea, D.; Caldato, J.; Beghetto, V. Amidated Carboxymethylcellulose: Synthesis, Characterization and Evaluation of Their Performance in Paper Coating Application. Cellulose 2026, 33, 481–503. [Google Scholar] [CrossRef]
- Yang, F.; Chen, L.; Cui, S.; Yu, D.; Zheng, S.; Zhao, D.; Yin, X.; Lai, C.; Chen, J. Asymmetric Chitosan-Derivative/Carboxymethylcellulose Layer-by-Layer Film Combining Antimicrobial and Vascular Regeneration for the Repair of Infected Wounds. Int. J. Biol. Macromol. 2024, 269, 132031. [Google Scholar] [CrossRef]
- Seddiqi, H.; Oliaei, E.; Honarkar, H.; Jin, J.; Geonzon, L.C.; Bacabac, R.G.; Klein-Nulend, J. Cellulose and Its Derivatives: Towards Biomedical Applications. Cellulose 2021, 28, 1893–1931. [Google Scholar] [CrossRef]
- Li, W.; Liu, Q.; Zhang, Y.; Li, C.; He, Z.; Choy, W.C.H.; Low, P.J.; Sonar, P.; Kyaw, A.K.K. Biodegradable Materials and Green Processing for Green Electronics. Adv. Mater. 2020, 32, 2001591. [Google Scholar] [CrossRef]
- Santandrea, D.; Beghetto, V. 4-(4,6-Dimethoxy-1,3,5-Triazin-2-Yl)-4-Methyl-Morpholinium Chloride-Assisted Amide Crosslinking of Carboxymethyl Cellulose for High-Performing Films. RSC Adv. 2025, 15, 34846–34860. [Google Scholar] [CrossRef]
- Beghetto, V.; Gatto, V.; Conca, S.; Bardella, N.; Buranello, C.; Gasparetto, G.; Sole, R. Development of 4-(4,6-Dimethoxy-1,3,5-Triazin-2-Yl)-4-Methyl-Morpholinium Chloride Cross-Linked Carboxymethyl Cellulose Films. Carbohydr. Polym. 2020, 249, 116810. [Google Scholar] [CrossRef]
- Yang, W.; Li, W.; Lei, Y.; He, P.; Wei, G.; Guo, L. Functionalization of Cellulose-Based Sponges: Design, Modification, Environmental Applications, and Sustainability Analysis. Carbohydr. Polym. 2025, 348, 122772. [Google Scholar] [CrossRef] [PubMed]
- Kurhade, R.R.; Shaikh, M.S.; Nagulwar, V.; Kale, M.A. Advancements in Carboxymethyl Cellulose (CMC) Modifications and Their Diverse Biomedical Applications: A Comprehensive Review. Int. J. Polym. Mater. Polym. Biomater. 2025, 74, 1043–1067. [Google Scholar] [CrossRef]
- Madhushree, M.; Vairavel, P.; Mahesha, G.T.; Bhat, K.S. Oxidative Modifications of Cellulose: Methods, Mechanisms, and Emerging Applications. J. Nat. Fibers 2025, 22, 2497910. [Google Scholar] [CrossRef]
- Xing, W.; Wang, Z.; Zhang, K.; Xu, Y.; Pan, Y.; Zhang, G. Flame-Retardant and Antibacterial Multifunctional Cellulose Fibers with Carbamate Esterification and Phosphorylation Modification. Ind. Crops Prod. 2025, 233, 121482. [Google Scholar] [CrossRef]
- Liesiene, J.; Kazlauske, J. Functionalization of Cellulose: Synthesis of Water-Soluble Cationic Cellulose Derivative. Cellul. Chem. Technol. 2021, 47, 515–523. [Google Scholar]
- Heinze, T.; Pfeiffer, K. Studies on the Synthesis and Characterization of Carboxymethylcellulose. Angew. Makromol. Chem. 1999, 266, 37–45. [Google Scholar] [CrossRef]
- Hashem, A.; Farag, S.; Badawy, S.M. Carboxymethyl Cellulose: Past Innovations, Present Applications, and Future Horizons. Results Chem. 2025, 17, 102534. [Google Scholar] [CrossRef]
- Paganelli, S.; Massimi, N.; Di Michele, A.; Piccolo, O.; Rampazzo, R.; Facchin, M.; Beghetto, V. Use of Carboxymethyl Cellulose as Binder for the Production of Water-Soluble Catalysts. Int. J. Biol. Macromol. 2024, 270, 132541. [Google Scholar] [CrossRef] [PubMed]
- Suriyatem, R.; Auras, R.A.; Rachtanapun, P. Utilization of Carboxymethyl Cellulose from Durian Rind Agricultural Waste to Improve Physical Properties and Stability of Rice Starch-Based Film. J. Polym. Environ. 2019, 27, 286–298. [Google Scholar] [CrossRef]
- Uyanga, K.A.; Daoud, W.A. Carboxymethyl Cellulose-Chitosan Composite Hydrogel: Modelling and Experimental Study of the Effect of Composition on Microstructure and Swelling Response. Int. J. Biol. Macromol. 2021, 181, 1010–1022. [Google Scholar] [CrossRef] [PubMed]
- Rasheed, H.A.; Adeleke, A.; Nzerem, P.; Ajayi, O.; Ikubanni, P.; Yahya, A.M. A Review on the Use of Carboxymethyl Cellulose in Oil and Gas Field Operations. Cellulose 2023, 30, 9899–9924. [Google Scholar] [CrossRef]
- Zulqarnain, M.; Tabasum, S.; Zia, K.M.; Noreen, A. Synthesis and Characterization of Hydroxy Ethyl Cellulose (HEC)-Based Polyurethane Dispersions for Textile Applications. Arab. J. Sci. Eng. 2025. [Google Scholar] [CrossRef]
- Moura, H.O.M.A.; Souza, E.C.D.; Silva, B.R.D.; Pereira, E.S.; Bicudo, T.D.C.; Rodríguez-Castellón, E.; Carvalho, L.S.D. Optimization of Synthesis Method for Carboxymethylcellulose (CMC) from Agro-Food Wastes by Response Surface Methodology (RSM) Using D-Optimal Algorithm. Ind. Crops Prod. 2024, 220, 119413. [Google Scholar] [CrossRef]
- Badry, R.; Sabry, N.M.; Ibrahim, M.A. Enhancing the Structural and Optoelectronic Properties of Carboxymethyl Cellulose Sodium Filled with ZnO/GO and CuO/GO Nanocomposites for Antimicrobial Packaging Applications. Sci. Rep. 2024, 14, 30591. [Google Scholar] [CrossRef] [PubMed]
- Beghetto, V.; Gatto, V.; Samiolo, R.; Scolaro, C.; Brahimi, S.; Facchin, M.; Visco, A. Plastics Today: Key Challenges and EU Strategies towards Carbon Neutrality: A Review. Environ. Pollut. 2023, 334, 122102. [Google Scholar] [CrossRef]
- Yildirim-Yalcin, M.; Tornuk, F.; Toker, O.S. Recent Advances in the Improvement of Carboxymethyl Cellulose-Based Edible Films. Trends Food Sci. Technol. 2022, 129, 179–193. [Google Scholar] [CrossRef]
- Yao, Y.; Sun, Z.; Li, X.; Tang, Z.; Li, X.; Morrell, J.J.; Liu, Y.; Li, C.; Luo, Z. Effects of Raw Material Source on the Properties of CMC Composite Films. Polymers 2022, 14, 32. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishnan, R.; Kulandhaivelu, S.V.; Roy, S.; Viswanathan, V.P. Characterisation of Ternary Blend Film of Alginate/Carboxymethyl Cellulose/Starch for Packaging Applications. Ind. Crops Prod. 2023, 193, 116114. [Google Scholar] [CrossRef]
- Facchin, M.; Gatto, V.; Samiolo, R.; Conca, S.; Santandrea, D.; Beghetto, V. May 1,3,5-Triazine Derivatives Be the Future of Leather Tanning? A Critical Review. Environ. Pollut. 2024, 345, 123472. [Google Scholar] [CrossRef]
- Yaradoddi, J.S.; Banapurmath, N.R.; Ganachari, S.V.; Soudagar, M.E.M.; Mubarak, N.M.; Hallad, S.; Hugar, S.; Fayaz, H. Biodegradable Carboxymethyl Cellulose Based Material for Sustainable Packaging Application. Sci. Rep. 2020, 10, 21960. [Google Scholar] [CrossRef]
- Jiang, X.; Yang, Z.; Peng, Y.; Han, B.; Li, Z.; Li, X.; Liu, W. Preparation, Characterization and Feasibility Study of Dialdehyde Carboxymethyl Cellulose as a Novel Crosslinking Reagent. Carbohydr. Polym. 2016, 137, 632–641. [Google Scholar] [CrossRef]
- Yang, M.; Chen, Y.; Abdalkarim, S.Y.H.; Chen, X.; Yu, H.-Y. Efficient Cellulose Dissolution and Derivatization Enabled by Oxalic/Sulfuric Acid for High-Performance Cellulose Films as Food Packaging. Int. J. Biol. Macromol. 2024, 276, 133799. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Shi, H.; He, Y.; Fei, X.; Peng, L. Preparation and Characterization of Carboxymethyl Cellulose-Based Composite Films Reinforced by Cellulose Nanocrystals Derived from Pea Hull Waste for Food Packaging Applications. Int. J. Biol. Macromol. 2020, 164, 4104–4112. [Google Scholar] [CrossRef] [PubMed]
- Kong, P.; Rosnan, S.M.; Enomae, T. Carboxymethyl Cellulose–Chitosan Edible Films for Food Packaging: A Review of Recent Advances. Carbohydr. Polym. 2024, 346, 122612. [Google Scholar] [CrossRef]
- Kasle, P.; Bains, A.; Goksen, G.; Dhull, S.B.; Ali, N.; Nagarik, R.; Fareed, M.; Chawla, P. Exploring the Properties of Carboxymethyl Cellulose Blended With Other Polymeric Compounds for the Formulation of Biodegradable Packaging Films and Edible Coatings: A Review. Compr. Rev. Food Sci. Food Saf. 2025, 24, e70215. [Google Scholar] [CrossRef]
- Qin, S.; Sun, H.; Wan, X.; Wu, Y.; Lin, X.; Kan, H.; Hou, D.; Zheng, Z.; He, X.; Liu, C. Carboxymethylcellulose Reinforced Starch Films and Rapid Detection of Spoiled Beverages. Front. Bioeng. Biotechnol. 2023, 10, 1099118. [Google Scholar] [CrossRef]
- Arifin, H.R.; Djali, M.; Nurhadi, B.; Azlin-Hasim, S.; Masruchin, N.; Vania, P.A.; Hilmi, A. Corn Starch-Based Bionanocomposite Film Reinforced With ZnO Nanoparticles and Different Types of Plasticizers. Front. Sustain. Food Syst. 2022, 6, 886219. [Google Scholar] [CrossRef]
- Mirpoor, S.F.; Massironi, A.; Winning, D.; Lignou, S.; Ghawi, S.K.; Trotta, F.; Charalampopoulos, D. Agar/Carboxymethyl Cellulose Blended Films with Green-Synthesised Silver Nanoparticles as a Sustainable Alternative for Food Packaging Applications. Polymers 2025, 17, 3126. [Google Scholar] [CrossRef] [PubMed]
- Doveri, L.; Diaz Fernandez, Y.A.; Dacarro, G.; Grisoli, P.; Milanese, C.; Urena, M.; Sok, N.; Karbowiak, T.; Pallavicini, P. Active Pectin/Carboxymethylcellulose Composite Films for Bread Packaging. Molecules 2025, 30, 2257. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Li, W.; Wu, L.; Li, Y.; Li, H. Preparation and Characterization of Chitosan/Dialdehyde Carboxymethyl Cellulose Composite Film Loaded with Cinnamaldehyde@zein Nanoparticles for Active Food Packaging. Int. J. Biol. Macromol. 2024, 261, 129586. [Google Scholar] [CrossRef]
- Abdulkhani, A.; Najd Mazhar, A.; Hedjazi, S.; Hamzeh, Y. Preparation of Xylan Bio-Composite Films Reinforced with Oxidized Carboxymethyl Cellulose and Nanocellulose. Polym. Bull. 2020, 77, 6227–6239. [Google Scholar] [CrossRef]
- Franco, T.S.; Amezcua, R.M.J.; Rodrìguez, A.V.; Enriquez, S.G.; Urquíza, M.R.; Mijares, E.M.; De Muniz, G.B. Carboxymethyl and Nanofibrillated Cellulose as Additives on the Preparation of Chitosan Biocomposites: Their Influence Over Films Characteristics. J. Polym. Environ. 2020, 28, 676–688. [Google Scholar] [CrossRef]
- Zheng, T.; Yu, X.; Pilla, S. Mechanical and Moisture Sensitivity of Fully Bio-Based Dialdehyde Carboxymethyl Cellulose Cross-Linked Soy Protein Isolate Films. Carbohydr. Polym. 2017, 157, 1333–1340. [Google Scholar] [CrossRef]
- Kim, N.; Seo, E.; Kim, Y. Physical, Mechanical and Water Barrier Properties of Yuba Films Incorporated with Various Types of Additives. J. Sci. Food Agric. 2019, 99, 2808–2817. [Google Scholar] [CrossRef]
- Thungphotrakul, N.; Prapainainar, P. Development of Polyvinyl Alcohol/Carboxymethylcellulose-Based Bio-Packaging Film with Citric Acid Crosslinking and Clove Essential Oil Encapsulated Chitosan Nanoparticle Pickering Emulsion. Int. J. Biol. Macromol. 2024, 282, 137223. [Google Scholar] [CrossRef]
- Jia, Y.; Hsu, Y.-I.; Uyama, H. Dual-Crosslinked Starch/Carboxymethyl Cellulose Blend Film with Ion-Responsive Dissolution Properties. Polym. Degrad. Stab. 2023, 215, 110453. [Google Scholar] [CrossRef]
- Fernández-Santos, J.; Valls, C.; Cusola, O.; Roncero, M.B. Composites of Cellulose Nanocrystals in Combination with Either Cellulose Nanofibril or Carboxymethylcellulose as Functional Packaging Films. Int. J. Biol. Macromol. 2022, 211, 218–229. [Google Scholar] [CrossRef]
- Antosik, A.K.; Miądlicki, P.; Wilpiszewska, K.; Markowska-Szczupak, A.; Koren, Z.C.; Wróblewska, A. Polysaccharide Films Modified by Compounds of Natural Origin and Silver Having Potential Medical Applications. Cellulose 2021, 28, 7257–7271. [Google Scholar] [CrossRef]
- Jannatyha, N.; Shojaee-Aliabadi, S.; Moslehishad, M.; Moradi, E. Comparing Mechanical, Barrier and Antimicrobial Properties of Nanocellulose/CMC and Nanochitosan/CMC Composite Films. Int. J. Biol. Macromol. 2020, 164, 2323–2328. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Han, P.; Zhao, Y.; Lu, L.; Qi, W.; Zhao, K.; Shu, Y.; Zhang, Z. Preparation and Characteristics of Carboxymethyl Cellulose-Based Films Embedding Cinnamon Essential Oil and Their Application on Mutton Preservation. Front. Nutr. 2025, 12, 1559833. [Google Scholar] [CrossRef] [PubMed]
- Amaregouda, Y.; Kamanna, K. Carboxymethyl Cellulose/Starch-Based Films Incorporating Chitosan Nanoparticles for Multifunctional Food Packaging. Cellulose 2024, 31, 2413–2427. [Google Scholar] [CrossRef]
- Li, M.; Yang, G.; Zhou, P.; Li, Y.; Wang, B.; Liu, Z.; Wu, S.; Zhou, T.; Ruan, W.; Li, D. Preparation, Optimizations, and Applications of (Carboxymethyl Cellulose)-Chitosan Polyelectrolyte Composite Films in Food Packaging: A Review. Int. J. Biol. Macromol. 2025, 315, 144443. [Google Scholar] [CrossRef]
- Hashmi, M.; Ullah, S.; Ullah, A.; Saito, Y.; Haider, Md.K.; Bie, X.; Wada, K.; Kim, I.S. Carboxymethyl Cellulose (CMC) Based Electrospun Composite Nanofiber Mats for Food Packaging. Polymers 2021, 13, 302. [Google Scholar] [CrossRef]
- Nadeem, H.; Naseri, M.; Shanmugam, K.; Dehghani, M.; Browne, C.; Miri, S.; Garnier, G.; Batchelor, W. An Energy Efficient Production of High Moisture Barrier Nanocellulose/Carboxymethyl Cellulose Films via Spray-Deposition Technique. Carbohydr. Polym. 2020, 250, 116911. [Google Scholar] [CrossRef]
- Nadeem, H.; Dehghani, M.; Miri, S.; Pazirofteh, M.; Garnier, G.; Batchelor, W. Highly Hydrophobic and Moisture Barrier Nanocellulose Based Films Produced via Spray Deposition. Cellulose 2023, 30, 5157–5170. [Google Scholar] [CrossRef]
- He, B.; Wang, S.; Lan, P.; Wang, W.; Zhu, J. Topography and Physical Properties of Carboxymethyl Cellulose Films Assembled with Calcium and Gelatin at Different Temperature and Humidity. Food Chem. 2022, 382, 132391. [Google Scholar] [CrossRef]
- Chen, H.; Meng, X.; Zhang, F.; Chen, J.; Ding, X.; Jian, T.; Niu, G.; Tong, B.; Gai, Y.; Zhao, H.; et al. Development of Chitosan-Carboxymethyl Cellulose Edible Films Loaded with Blackberry Anthocyanins and Tea Polyphenols and Their Application in Beef Preservation. Food Hydrocoll. 2025, 164, 111198. [Google Scholar] [CrossRef]
- Peng, Y.; Zhang, X.; Liu, W.; Li, C. Development of Hydrophobic Sodium Carboxymethyl Cellulose/Polyvinyl Alcohol/Chitosan Paper-Based Antimicrobial Indicator Cards Based on a Non-Fluorinated Silane Coating for Monitoring Carp Freshness. Int. J. Biol. Macromol. 2025, 309, 142951. [Google Scholar] [CrossRef]
- Kumar, L.; Tyagi, P.; Lucia, L.; Pal, L. Innovations in Edible Packaging Films, Coatings, and Antimicrobial Agents for Applications in Food Industry. Compr. Rev. Food Sci. Food Saf. 2025, 24, e70217. [Google Scholar] [CrossRef]
- Yue, S.; Zhang, T.; Wang, S.; Han, D.; Huang, S.; Xiao, M.; Meng, Y. Recent Progress of Biodegradable Polymer Package Materials: Nanotechnology Improving Both Oxygen and Water Vapor Barrier Performance. Nanomaterials 2024, 14, 338. [Google Scholar] [CrossRef]
- Briassoulis, D.; Giannoulis, A. Evaluation of the Functionality of Bio-Based Food Packaging Films. Polym. Test. 2018, 69, 39–51. [Google Scholar] [CrossRef]
- Roy, S.; Kim, H.-J.; Rhim, J.-W. Synthesis of Carboxymethyl Cellulose and Agar-Based Multifunctional Films Reinforced with Cellulose Nanocrystals and Shikonin. ACS Appl. Polym. Mater. 2021, 3, 1060–1069. [Google Scholar] [CrossRef]
- Dąbrowska, G.B.; Antoszewski, M.; Szydłowska-Czerniak, A.; Raszkowska-Kaczor, A.; Jędrzejewski, T.; Wrotek, S.; Bartkowiak, M.; Swiontek Brzezinska, M.; Zborowska, M. New Biodegradable Carboxymethyl Cellulose-Based Films with Liquid Products of Wood Pine Pyrolysis with Antibacterial and Antioxidant Properties. Materials 2025, 18, 2228. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishnan, R.; Kim, J.T.; Roy, S.; Jayakumar, A. Recent Advances in Carboxymethyl Cellulose-Based Active and Intelligent Packaging Materials: A Comprehensive Review. Int. J. Biol. Macromol. 2024, 259, 129194. [Google Scholar] [CrossRef]
- Thomas, S.; Soloman, P.A.; Rejini, V.O. Preparation of Chitosan- CMC Blends and Studies on Thermal Properties. Procedia Technol. 2016, 24, 721–726. [Google Scholar] [CrossRef]
- Ballesteros, L.F.; Cerqueira, M.A.; Teixeira, J.A.; Mussatto, S.I. Production and Physicochemical Properties of Carboxymethyl Cellulose Films Enriched with Spent Coffee Grounds Polysaccharides. Int. J. Biol. Macromol. 2018, 106, 647–655. [Google Scholar] [CrossRef]
- Akhtar, H.M.S.; Riaz, A.; Hamed, Y.S.; Abdin, M.; Chen, G.; Wan, P.; Zeng, X. Production and Characterization of CMC-Based Antioxidant and Antimicrobial Films Enriched with Chickpea Hull Polysaccharides. Int. J. Biol. Macromol. 2018, 118, 469–477. [Google Scholar] [CrossRef]
- Li, N.; Jiang, D.; Zhou, Z.; Lu, Y.; Lei-Zhong; Law, W.C.; Tang, C.Y. Development of Carboxymethyl Cellulose/Starch Films Enriched with ZnO-NPs and Anthocyanins for Antimicrobial and pH-Indicating Food Packaging. Int. J. Biol. Macromol. 2024, 282, 136814. [Google Scholar] [CrossRef]
- Morandini, A.; Spadati, E.; Leonetti, B.; Sole, R.; Gatto, V.; Rizzolio, F.; Beghetto, V. Sustainable Triazine-Derived Quaternary Ammonium Salts as Antimicrobial Agents. RSC Adv. 2021, 11, 28092–28096. [Google Scholar] [CrossRef]
- Kyong, Y.Y.; Zhang, L.; Lu, Y.; Ryu, S.-J.; Han, K.; Wang, M.-H.; Kim, H.-Y.; Sathiyaseelan, A. Biocompatible Carboxymethyl Cellulose–Potato Starch Film with Biosynthesized Tellurium Nanoparticles and Undecanoic Acid for Enhanced Antimicrobial Protection in Post-Harvest Tomato Preservation. Int. J. Biol. Macromol. 2025, 322, 146858. [Google Scholar] [CrossRef]
- Beghetto, V. Waste Cooking Oils into High-Value Products: Where Is the Industry Going? Polymers 2025, 17, 887. [Google Scholar] [CrossRef]
- Roy, J.C.; Ferri, A.; Giraud, S.; Jinping, G.; Salaün, F. Chitosan–Carboxymethylcellulose-Based Polyelectrolyte Complexation and Microcapsule Shell Formulation. Int. J. Mol. Sci. 2018, 19, 2521. [Google Scholar] [CrossRef] [PubMed]
- Fekete, T.; Borsa, J.; Takács, E.; Wojnárovits, L. Synthesis of Carboxymethylcellulose/Starch Superabsorbent Hydrogels by Gamma-Irradiation. Chem. Cent. J. 2017, 11, 46. [Google Scholar] [CrossRef] [PubMed]
- Altam, A.A.; Zhu, L.; Huang, W.; Huang, H.; Yang, S. Polyelectrolyte Complex Beads of Carboxymethylcellulose and Chitosan: The Controlled Formation and Improved Properties. Carbohydr. Polym. Technol. Appl. 2021, 2, 100100. [Google Scholar] [CrossRef]
- Tavares, K.M.; Campos, A.D.; Mitsuyuki, M.C.; Luchesi, B.R.; Marconcini, J.M. Corn and Cassava Starch with Carboxymethyl Cellulose Films and Its Mechanical and Hydrophobic Properties. Carbohydr. Polym. 2019, 223, 115055. [Google Scholar] [CrossRef]
- Chen, J.; Li, H.; Fang, C.; Cheng, Y.; Tan, T.; Han, H. In Situ Synthesis and Properties of Ag NPs/Carboxymethyl Cellulose/Starch Composite Films for Antibacterial Application. Polym. Compos. 2020, 41, 838–847. [Google Scholar] [CrossRef]
- Arik Kibar, E.A.; Us, F. Starch–Cellulose Ether Films: Microstructure and Water Resistance. J. Food Process Eng. 2017, 40, e12382. [Google Scholar] [CrossRef]
- Tavares, K.M.; Campos, A.D.; Luchesi, B.R.; Resende, A.A.; Oliveira, J.E.D.; Marconcini, J.M. Effect of Carboxymethyl Cellulose Concentration on Mechanical and Water Vapor Barrier Properties of Corn Starch Films. Carbohydr. Polym. 2020, 246, 116521. [Google Scholar] [CrossRef]
- Jiang, G.; Hou, X.; Zeng, X.; Zhang, C.; Wu, H.; Shen, G.; Li, S.; Luo, Q.; Li, M.; Liu, X.; et al. Preparation and Characterization of Indicator Films from Carboxymethyl-Cellulose/Starch and Purple Sweet Potato (Ipomoea Batatas (L.) Lam) Anthocyanins for Monitoring Fish Freshness. Int. J. Biol. Macromol. 2020, 143, 359–372. [Google Scholar] [CrossRef] [PubMed]
- Yun, D.; Wu, Y.; Yong, H.; Tang, C.; Chen, D.; Kan, J.; Liu, J. Recent Advances in Purple Sweet Potato Anthocyanins: Extraction, Isolation, Functional Properties and Applications in Biopolymer-Based Smart Packaging. Foods 2024, 13, 3485. [Google Scholar] [CrossRef]
- Silva, M.T.D.C.; Cunha Júnior, P.C.D.; Miranda, C.G.; Sato, A.C.K.; Machado, M.T.D.C.; Ferreira, E.H.D.R. Development and Application of pH -indicator Film Based on Freeze-dried Purple-fleshed Sweet Potato, Starch, and CMC. Int. J. Food Sci. Technol. 2023, 58, 6689–6697. [Google Scholar] [CrossRef]
- Akhtar, H.M.S.; Ahmed, S.; Olewnik-Kruszkowska, E.; Gierszewska, M.; Brzezinska, M.S.; Dembińska, K.; Kalwasińska, A. Carboxymethyl Cellulose Based Films Enriched with Polysaccharides from Mulberry Leaves (Morus Alba L.) as New Biodegradable Packaging Material. Int. J. Biol. Macromol. 2023, 253, 127633. [Google Scholar] [CrossRef]
- Mandal, A.; Chakrabarty, D. Studies on Mechanical, Thermal, and Barrier Properties of Carboxymethyl Cellulose Film Highly Filled with Nanocellulose. J. Thermoplast. Compos. Mater. 2018, 32, 995–1014. [Google Scholar] [CrossRef]
- Oun, A.A.; Rhim, J.-W. Preparation and Characterization of Sodium Carboxymethyl Cellulose/Cotton Linter Cellulose Nanofibril Composite Films. Carbohydr. Polym. 2015, 127, 101–109. [Google Scholar] [CrossRef]
- Wei, J.; Jia, S.; Zhang, L.; Zhou, Y.; Lv, Y.; Zhang, X.; Shao, Z. Preparation of Treelike and Rodlike Carboxymethylated Nanocellulose and Their Effect on Carboxymethyl Cellulose Films. J. Appl. Polym. Sci. 2021, 138, 50092. [Google Scholar] [CrossRef]
- Tongdeesoontorn, W.; Mauer, L.J.; Wongruong, S.; Sriburi, P.; Rachtanapun, P. Physical and Antioxidant Properties of Cassava Starch–Carboxymethyl Cellulose Incorporated with Quercetin and TBHQ as Active Food Packaging. Polymers 2020, 12, 366. [Google Scholar] [CrossRef] [PubMed]
- Lan, W.; Zhang, R.; Ji, T.; Sameen, D.E.; Ahmed, S.; Qin, W.; Dai, J.; He, L.; Liu, Y. Improving Nisin Production by Encapsulated Lactococcus Lactis with Starch/Carboxymethyl Cellulose Edible Films. Carbohydr. Polym. 2021, 251, 117062. [Google Scholar] [CrossRef]
- Jiang, Q.; Liu, L.; Yuan, X.; Li, H.; Wang, Y.; Li, J. A Completely Bio-Based Corn Starch/Sodium Carboxymethyl Cellulose Film with Antibacterial and Antioxidant Performance Modified by a Natural Alcohol Ester of Amino Acid and Curcumin. Carbohydr. Polym. 2025, 362, 123686. [Google Scholar] [CrossRef]
- Noshirvani, N.; Ghanbarzadeh, B.; Gardrat, C.; Rezaei, M.R.; Hashemi, M.; Le Coz, C.; Coma, V. Cinnamon and Ginger Essential Oils to Improve Antifungal, Physical and Mechanical Properties of Chitosan-Carboxymethyl Cellulose Films. Food Hydrocoll. 2017, 70, 36–45. [Google Scholar] [CrossRef]
- How, Y.-H.; Lim, E.M.-Y.; Kong, I.; Kee, P.-E.; Pui, L.-P. Development of Carboxymethyl Cellulose–Chitosan Based Antibacterial Films Incorporating a Persicaria Minor Huds. Essential Oil Nanoemulsion. Sustain. Food Technol. 2024, 2, 400–414. [Google Scholar] [CrossRef]
- Wang, B.; Yang, X.; Qiao, C.; Li, Y.; Li, T.; Xu, C. Effects of Chitosan Quaternary Ammonium Salt on the Physicochemical Properties of Sodium Carboxymethyl Cellulose-Based Films. Carbohydr. Polym. 2018, 184, 37–46. [Google Scholar] [CrossRef]
- Li, Y.; Li, Y.; Chen, T.; Yang, X.; Qiao, C.; Hao, F.; Liu, M. N-(2-Hydroxyl)-Propyl-3-Trimethylammonium Chitosan Chloride/Carboxymethyl Cellulose Films Filled with in-Situ Crystallized Calcium Carbonate. Carbohydr. Polym. 2022, 278, 118975. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Yang, X.; Li, Y.; Qiao, C.; Wang, S.; Wang, X.; Xu, C.; Yang, H.; Li, T. Enhancement of a Zwitterionic Chitosan Derivative on Mechanical Properties and Antibacterial Activity of Carboxymethyl Cellulose-Based Films. Int. J. Biol. Macromol. 2020, 159, 1197–1205. [Google Scholar] [CrossRef]
- Liu, C.; Qin, S.; Xie, J.; Lin, X.; Zheng, Y.; Yang, J.; Kan, H.; Shi, Z. Using Carboxymethyl Cellulose as the Additive With Enzyme-Catalyzed Carboxylated Starch to Prepare the Film With Enhanced Mechanical and Hydrophobic Properties. Front. Bioeng. Biotechnol. 2021, 9, 638546. [Google Scholar] [CrossRef] [PubMed]
- Feng, S.; Liu, F.; Guo, Y.; Ye, M.; He, J.; Zhou, H.; Liu, L.; Cai, L.; Zhang, Y.; Li, R. Exploring the Role of Chitosan in Affecting the Adhesive, Rheological and Antimicrobial Properties of Carboxymethyl Cellulose Composite Hydrogels. Int. J. Biol. Macromol. 2021, 190, 554–563. [Google Scholar] [CrossRef]
- Mohammadi, M.; Azizi, M.H.; Zoghi, A. Antimicrobial Activity of Carboxymethyl Cellulose–Gelatin Film Containing Dianthus Barbatus Essential Oil against Aflatoxin-producing Molds. Food Sci. Nutr. 2020, 8, 1244–1253. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Gong, L.; Li, Q.; Fan, B.; Ma, C.; He, Y.-C. Preparation of a Biobased Polyelectrolyte Complex from Chitosan and Sodium Carboxymethyl Cellulose and Its Antibacterial Characteristics. Int. J. Biol. Macromol. 2023, 227, 524–534. [Google Scholar] [CrossRef]
- Uyanga, K.A.; Daoud, W.A. Green and Sustainable Carboxymethyl Cellulose-Chitosan Composite Hydrogels: Effect of Crosslinker on Microstructure. Cellulose 2021, 28, 5493–5512. [Google Scholar] [CrossRef]
- Sole, R.; Buranello, C.; Di Michele, A.; Beghetto, V. Boosting Physical-Mechanical Properties of Adipic Acid/Chitosan Films by DMTMM Cross-Linking. Int. J. Biol. Macromol. 2022, 209, 2009–2019. [Google Scholar] [CrossRef]
- Wu, J.-K.; Wang, X.-S.; Chen, K.-F.; Zhou, G.-J.; Li, X.; Xu, J.; An, Q.-F. The States of Sulfate Groups Affect the Mechanical and Separation Properties of Carboxymethyl Cellulose/Chitosan Complex Membranes. RSC Adv. 2016, 6, 26352–26360. [Google Scholar] [CrossRef]
- Riaz, A.; Lei, S.; Akhtar, H.M.S.; Wan, P.; Chen, D.; Jabbar, S.; Abid, M.; Hashim, M.M.; Zeng, X. Preparation and Characterization of Chitosan-Based Antimicrobial Active Food Packaging Film Incorporated with Apple Peel Polyphenols. Int. J. Biol. Macromol. 2018, 114, 547–555. [Google Scholar] [CrossRef]
- Riaz, A.; Lagnika, C.; Luo, H.; Nie, M.; Dai, Z.; Liu, C.; Abdin, M.; Hashim, M.M.; Li, D.; Song, J. Effect of Chinese Chives (Allium Tuberosum) Addition to Carboxymethyl Cellulose Based Food Packaging Films. Carbohydr. Polym. 2020, 235, 115944. [Google Scholar] [CrossRef] [PubMed]
- Valizadeh, S.; Naseri, M.; Babaei, S.; Hosseini, S.M.H.; Imani, A. Development of Bioactive Composite Films from Chitosan and Carboxymethyl Cellulose Using Glutaraldehyde, Cinnamon Essential Oil and Oleic Acid. Int. J. Biol. Macromol. 2019, 134, 604–612. [Google Scholar] [CrossRef]
- Thakkar, A.; Patel, B.; Sahu, S.K.; Yadav, V.K.; Patel, R.; Sahoo, D.K.; Joshi, M.; Patel, A. Potato Starch Bioplastic Films Reinforced with Organic and Inorganic Fillers: A Sustainable Packaging Alternative. Int. J. Biol. Macromol. 2025, 306, 141630. [Google Scholar] [CrossRef] [PubMed]
- Ragab, H.M.; Diab, N.S.; AlElaimi, M.; Alghamdi, A.M.; Farea, M.O.; Farea, A. Retraction of Fabrication and Characterization of Silver Nanoparticle-Doped Chitosan/Carboxymethyl Cellulose Nanocomposites for Optoelectronic and Biological Applications. ACS Omega 2026, 11, 3663–3663. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Rhim, J.-W. Carboxymethyl Cellulose-Based Antioxidant and Antimicrobial Active Packaging Film Incorporated with Curcumin and Zinc Oxide. Int. J. Biol. Macromol. 2020, 148, 666–676. [Google Scholar] [CrossRef]
- Jiang, S.; Qiao, C.; Liu, R.; Liu, Q.; Xu, J.; Yao, J. Structure and Properties of Citric Acid Cross-Linked Chitosan/Poly(Vinyl Alcohol) Composite Films for Food Packaging Applications. Carbohydr. Polym. 2023, 312, 120842. [Google Scholar] [CrossRef]
- Zhang, W.; Roy, S.; Ezati, P.; Yang, D.-P.; Rhim, J.-W. Tannic Acid: A Green Crosslinker for Biopolymer-Based Food Packaging Films. Trends Food Sci. Technol. 2023, 136, 11–23. [Google Scholar] [CrossRef]
- Hejna, A.; Barczewski, M.; Kosmela, P.; Aniśko, J.; Szulc, J.; Skórczewska, K.; Piasecki, A.; Kuang, T. More than Just a Beer – Brewers’ Spent Grain, Spent Hops, and Spent Yeast as Potential Functional Fillers for Polymer Composites. Waste Manag. 2024, 180, 23–35. [Google Scholar] [CrossRef]
- Visco, A.; Bardella, N.; Scolaro, C.; Belhamdi, H.; Brahimi, S.; Gatto, V.; Samiolo, R.; Beghetto, V. Reuse of Beer Spent Grain for the Industrial Production of Biodegradable Bio-Composites. Ind. Crops Prod. 2025, 235, 121684. [Google Scholar] [CrossRef]
- Ruan, C.; Zhang, Y.; Wang, J.; Sun, Y.; Gao, X.; Xiong, G.; Liang, J. Preparation and Antioxidant Activity of Sodium Alginate and Carboxymethyl Cellulose Edible Films with Epigallocatechin Gallate. Int. J. Biol. Macromol. 2019, 134, 1038–1044. [Google Scholar] [CrossRef]
- Abdollahi, M.; Damirchi, S.; Shafafi, M.; Rezaei, M.; Ariaii, P. Carboxymethyl Cellulose-Agar Biocomposite Film Activated with Summer Savory Essential Oil as an Antimicrobial Agent. Int. J. Biol. Macromol. 2019, 126, 561–568. [Google Scholar] [CrossRef]
- Spinei, M.; Oroian, M.; Ursachi, V.-F. Characterization of Biodegradable Films Based on Carboxymethyl Cellulose and Citrus Pectin Films Enriched with Bee Bread Oil and Thyme Oil. LWT 2024, 214, 117088. [Google Scholar] [CrossRef]
- Lan, W.; He, L.; Liu, Y. Preparation and Properties of Sodium Carboxymethyl Cellulose/Sodium Alginate/Chitosan Composite Film. Coatings 2018, 8, 291. [Google Scholar] [CrossRef]
- Reji, R.E.; Mathew, C.B.; V, J.; Sabu, C.S.; Roy, S. A Review on Gelatin Films and Coatings for Active Food Packaging: Functional Properties and Applications. Food Innov. Adv. 2025, 4, 423–436. [Google Scholar] [CrossRef]
- Hassan, A.M.; Altemimi, A.B.; Ghanbarzadeh, B.; Adun, P.; Arab, K.; Ibrahim, S.; Awlqadr, F.H.; Hesarinejad, M.A.; Abedelmaksoud, T.G. Extending the Shelf Life of Strawberries: Physicochemical and Antibacterial Effects of Carboxymethyl Cellulose and Gelatin Coatings With Lemon Essential Oil. Food Sci. Nutr. 2025, 13, e70222. [Google Scholar] [CrossRef]
- Wang, K.; Du, L.; Zhang, C.; Lu, Z.; Lu, F.; Zhao, H. Preparation of Chitosan/Curdlan/Carboxymethyl Cellulose Blended Film and Its Characterization. J. Food Sci. Technol. 2019, 56, 5396–5404. [Google Scholar] [CrossRef]
- Ke, F.; Liu, D.; Qin, J.; Yang, M. Functional pH-Sensitive Film Containing Purple Sweet Potato Anthocyanins for Pork Freshness Monitoring and Cherry Preservation. Foods 2024, 13, 736. [Google Scholar] [CrossRef]
- Hu, X.; Zhang, W.; Qi, X.; Cheng, L.; Shen, M.; Wen, H.; Xie, J. Development of a Highly Responsive and Stable pH-Responsive Smart Film Using Co-Pigmentation: Preparation, Characterization, and Application for Fish Freshness Monitoring. Food Packag. Shelf Life 2026, 53, 101680. [Google Scholar] [CrossRef]
- Plaeyao, K.; Talodthaisong, C.; Yingyuen, W.; Kaewbundit, R.; Tun, W.S.T.; Saenchoopa, A.; Kayunkid, N.; Wiwattananukul, R.; Sakulsombat, M.; Kulchat, S. Biodegradable Antibacterial Food Packaging Based on Carboxymethyl Cellulose from Sugarcane Bagasse/Cassava Starch/Chitosan/Gingerol Extract Stabilized Silver Nanoparticles (Gin-AgNPs) and Vanillin as Cross-Linking Agent. Food Chem. 2025, 466, 142102. [Google Scholar] [CrossRef] [PubMed]
- Oun, A.A.; Rhim, J.-W. Characterization of Carboxymethyl Cellulose-Based Nanocomposite Films Reinforced with Oxidized Nanocellulose Isolated Using Ammonium Persulfate Method. Carbohydr. Polym. 2017, 174, 484–492. [Google Scholar] [CrossRef] [PubMed]
- Oun, A.A.; Rhim, J.-W. Preparation of Multifunctional Carboxymethyl Cellulose-Based Films Incorporated with Chitin Nanocrystal and Grapefruit Seed Extract. Int. J. Biol. Macromol. 2020, 152, 1038–1046. [Google Scholar] [CrossRef]
- El Miri, N.; Abdelouahdi, K.; Barakat, A.; Zahouily, M.; Fihri, A.; Solhy, A.; El Achaby, M. Bio-Nanocomposite Films Reinforced with Cellulose Nanocrystals: Rheology of Film-Forming Solutions, Transparency, Water Vapor Barrier and Tensile Properties of Films. Carbohydr. Polym. 2015, 129, 156–167. [Google Scholar] [CrossRef]
- Sadi, A.; Ferfera-Harrar, H. Cross-Linked CMC/Gelatin Bio-Nanocomposite Films with Organoclay, Red Cabbage Anthocyanins and Pistacia Leaves Extract as Active Intelligent Food Packaging: Colorimetric pH Indication, Antimicrobial/Antioxidant Properties, and Shrimp Spoilage Tests. Int. J. Biol. Macromol. 2023, 242, 124964. [Google Scholar] [CrossRef]
- Mohammadi, H.; Rezaeigolestani, M.; Mohsenzadeh, M. Optimization of Antimicrobial Nanocomposite Films Based on Carboxymethyl Cellulose Incorporating Chitosan Nanofibers and Guggul Gum Polysaccharide. Sci. Rep. 2024, 14, 13693. [Google Scholar] [CrossRef]
- Wawrzyńczak, A.; Chudzińska, J.; Feliczak-Guzik, A. Metal and Metal Oxides Nanoparticles as Nanofillers for Biodegradable Polymers. ChemPhysChem 2024, 25, e202300823. [Google Scholar] [CrossRef]
- T.U., G; Jaiswal, S.; Jain, N.V.; Gowtham, G.K.; Somashekar, R. Biodegradable Polymer Composite Materials for Sustainable Food Packaging Applications: A Review. Polym. Bull. 2026, 83, 167. [Google Scholar] [CrossRef]
- Noshirvani, N.; Ghanbarzadeh, B.; Mokarram, R.R.; Hashemi, M.; Coma, V. Preparation and Characterization of Active Emulsified Films Based on Chitosan-Carboxymethyl Cellulose Containing Zinc Oxide Nano Particles. Int. J. Biol. Macromol. 2017, 99, 530–538. [Google Scholar] [CrossRef]
- Youssef, Ahmed.M.; EL-Sayed, S.M.; EL-Sayed, H.S.; Salama, H.H.; Dufresne, A. Enhancement of Egyptian Soft White Cheese Shelf Life Using a Novel Chitosan/Carboxymethyl Cellulose/Zinc Oxide Bionanocomposite Film. Carbohydr. Polym. 2016, 151, 9–19. [Google Scholar] [CrossRef]
- Helmiyati, H.; Hidayat, Z.S.Z.; Sitanggang, I.F.R.; Liftyawati, D. Antimicrobial Packaging of ZnO–Nps Infused into CMC–PVA Nanocomposite Films Effectively Enhances the Physicochemical Properties. Polym. Test. 2021, 104, 107412. [Google Scholar] [CrossRef]
- Hasanin, M.S.; Ibrahim, N.A.; Kamel, S. Fabrication and Characterization of Sustainable Multifunctional Films Based on Carboxymethyl Cellulose and Poly (Vinyl Alcohol) Doped with TiO2 Nanoparticles and Silicone Microemulsion. J. Inorg. Organomet. Polym. Mater. 2025. [Google Scholar] [CrossRef]
- Thakare, N.R.; Gogoi, P.; Bharali, P.; Hazarika, S. Influence of Copper Ion Cross-Linked CMC-PVA Film on Cell Viability and Cell Proliferation Study. Int. J. Biol. Macromol. 2024, 282, 136645. [Google Scholar] [CrossRef] [PubMed]
- Skulborisutsuk, C.; Saisriyoot, M.; Suramitr, S.; Yingchutrakul, Y.; Butkinaree, C.; Egashira, R.; Vayachuta, L.; Prompinit, P. Valorization of Pineapple Peel Waste through Immobilized Crude Bromelain for Enhanced Feed Protein Hydrolysis. ACS Omega 2026, acsomega.5c08847. [Google Scholar] [CrossRef]
- Vyas, A.; Ng, S.; Fu, T.; Anum, I. ZnO-Embedded Carboxymethyl Cellulose Bioplastic Film Synthesized from Sugarcane Bagasse for Packaging Applications. Polymers 2025, 17, 579. [Google Scholar] [CrossRef] [PubMed]
- Alavarse, A.C.; Frachini, E.C.G.; Da Silva, R.L.C.G.; Lima, V.H.; Shavandi, A.; Petri, D.F.S. Crosslinkers for Polysaccharides and Proteins: Synthesis Conditions, Mechanisms, and Crosslinking Efficiency, a Review. Int. J. Biol. Macromol. 2022, 202, 558–596. [Google Scholar] [CrossRef]
- Deng, H.; Zhang, W.; Ramezan, Y.; Riahi, Z.; Khan, A.; Huang, Z. Antibacterial and Antioxidant Plant-derived Aldehydes: A New Role as Cross-linking Agents in Biopolymer-based Food Packaging Films. Compr. Rev. Food Sci. Food Saf. 2025, 24, e70089. [Google Scholar] [CrossRef]
- Shahbazi, M.; Ahmadi, S.J.; Seif, A.; Rajabzadeh, G. Carboxymethyl Cellulose Film Modification through Surface Photo-Crosslinking and Chemical Crosslinking for Food Packaging Applications. Food Hydrocoll. 2016, 61, 378–389. [Google Scholar] [CrossRef]
- Hu, G.; Lan, X.; Peng, B.; Liao, J.; Xiong, Y. Water Resistant, Biodegradable and Flexible Corn Starch/Carboxymethyl Cellulose Composite Film for Slow-Release Fertilizer Coating Materials. Int. J. Biol. Macromol. 2024, 260, 129476. [Google Scholar] [CrossRef]
- Nongnual, T.; Butprom, N.; Boonsang, S.; Kaewpirom, S. Citric Acid Crosslinked Carboxymethyl Cellulose Edible Films: A Case Study on Preserving Freshness in Bananas. Int. J. Biol. Macromol. 2024, 267, 131135. [Google Scholar] [CrossRef]
- Wang, M.; Jia, X.; Liu, W.; Lin, X. Water Insoluble and Flexible Transparent Film Based on Carboxymethyl Cellulose. Carbohydr. Polym. 2021, 255, 117353. [Google Scholar] [CrossRef]
- Fu, X.; Xu, S.; Ma, M. Preparation of DCNC Chemically Cross-Linked CMC/PVA Composite Film for Sustainable and Strawberry Preservation Active Packaging. Int. J. Biol. Macromol. 2025, 306, 141671. [Google Scholar] [CrossRef] [PubMed]
- Shan, Z.; Huang, J.; Huang, Y.; Zhou, Y.; Li, Y. Glutaraldehyde Crosslinked Ternary Carboxymethylcellulose/Polyvinyl Alcohol/Polyethyleneimine Film with Enhanced Mechanical Properties, Water Resistance, Antibacterial Activity, and UV-Shielding Ability without Any UV Absorbents. Int. J. Biol. Macromol. 2024, 277, 134563. [Google Scholar] [CrossRef]
- Scrivanti, A.; Sole, R.; Bortoluzzi, M.; Beghetto, V.; Bardella, N.; Dolmella, A. Synthesis of New Triazolyl-Oxazoline Chiral Ligands and Study of Their Coordination to Pd(II) Metal Centers. Inorganica Chim. Acta 2019, 498, 119129. [Google Scholar] [CrossRef]
- Kunishima, M.; Kawachi, C.; Iwasaki, F.; Terao, K.; Tani, S. Synthesis and Characterization of 4-(4,6-Dimethoxy-1,3,5-Triazin-2-Yl)-4-Methylmorpholinium Chloride. Tetrahedron Lett. 1999, 40, 5327–5330. [Google Scholar] [CrossRef]
- Gatto, V.; Conca, S.; Bardella, N.; Beghetto, V. Efficient Triazine Derivatives for Collagenous Materials Stabilization. Materials 2021, 14, 3069. [Google Scholar] [CrossRef]
- Beghetto, V.; Agostinis, L.; Gatto, V.; Samiolo, R.; Scrivanti, A. Sustainable Use of 4-(4,6-Dimethoxy-1,3,5-Triazin-2-Yl)-4-Methylmorpholinium Chloride as Metal Free Tanning Agent. J. Clean. Prod. 2019, 220, 864–872. [Google Scholar] [CrossRef]
- Beghetto, V.; Gatto, V.; Conca, S.; Bardella, N.; Scrivanti, A. Polyamidoamide Dendrimers and Cross-Linking Agents for Stabilized Bioenzymatic Resistant Metal-Free Bovine Collagen. Molecules 2019, 24, 3611. [Google Scholar] [CrossRef]
- Sole, R.; Gatto, V.; Conca, S.; Bardella, N.; Morandini, A.; Beghetto, V. Sustainable Triazine-Based Dehydro-Condensation Agents for Amide Synthesis. Molecules 2021, 26, 191. [Google Scholar] [CrossRef]
- Petta, D.; Eglin, D.; Grijpma, D.W.; D’Este, M. Enhancing Hyaluronan Pseudoplasticity via 4-(4,6-Dimethoxy-1,3,5-Triazin-2-Yl)-4-Methylmorpholinium Chloride-Mediated Conjugation with Short Alkyl Moieties. Carbohydr. Polym. 2016, 151, 576–583. [Google Scholar] [CrossRef] [PubMed]
- Morandini, A.; Leonetti, B.; Riello, P.; Sole, R.; Gatto, V.; Caligiuri, I.; Rizzolio, F.; Beghetto, V. Synthesis and Antimicrobial Evaluation of Bis-morpholine Triazine Quaternary Ammonium Salts. ChemMedChem 2021, 16, 3172–3176. [Google Scholar] [CrossRef]
- D’Este, M.; Eglin, D.; Alini, M. A Systematic Analysis of DMTMM vs EDC/NHS for Ligation of Amines to Hyaluronan in Water. Carbohydr. Polym. 2014, 108, 239–246. [Google Scholar] [CrossRef]
- Amadio, E.; Scrivanti, A.; Bortoluzzi, M.; Bertoldini, M.; Beghetto, V.; Matteoli, U.; Chessa, G. A New Palladium(II)–Allyl Complex Containing a Thioether-Triazole Ligand as Active Catalyst in Suzuki–Miyaura Reaction. Use of Tetraalkylammonium Salts as Promoters: Influence of the Salt Anion and Cation on the Catalytic Activity. Inorganica Chim. Acta 2013, 405, 188–195. [Google Scholar] [CrossRef]
- Scrivanti, A.; Bortoluzzi, M.; Sole, R.; Beghetto, V. Synthesis and Characterization of Yttrium, Europium, Terbium and Dysprosium Complexes Containing a Novel Type of Triazolyl–Oxazoline Ligand. Chem. Pap. 2018, 72, 799–808. [Google Scholar] [CrossRef]
- Morais, M.A.P.; Silva, M.; Barros, M.; Halley, P.; Almeida, Y.; Vinhas, G. Impact of Citric Acid on Guar Gum Carboxymethylcellulose Crosslinked Blend Films. J. Appl. Polym. Sci. 2024, 141, e56162. [Google Scholar] [CrossRef]
- Santos, M.B.; Souza, J.A.D.S.; Cândido, V.S.; Del Nero, J.; Paschoal Junior, W.; Maia, A.A.B.; Viegas, B.M.; Alves Junior, S.; Alberto Brito Da Silva, C.; Vinícius Da Silva Paula, M. Corn Starch/Carboxymethyl Cellulose Films: Influence of Citric Acid on Water Susceptibility, Morphological and Tensile Properties. ACS Omega 2026, 11, 5479–5488. [Google Scholar] [CrossRef]
- Ge, J.; Lu, W.; Zhang, H.; Gong, Y.; Wang, J.; Xie, Y.; Chang, Q.; Deng, X. Exploring Sustainable Food Packaging: Nanocellulose Composite Films with Enhanced Mechanical Strength, Antibacterial Performance, and Biodegradability. Int. J. Biol. Macromol. 2024, 259, 129200. [Google Scholar] [CrossRef]
- Liang, Y.; Peng, Y.; Zhou, X.; Chen, S.; Li, W.; Li, J.; Niu, X.; Guo, W. A Robust and Biodegradable Carboxymethyl Cellulose-Based Bioplastic Inspired by Nacre. Int. J. Biol. Macromol. 2025, 310, 143513. [Google Scholar] [CrossRef]
- Zhao, C.; Li, L.; Xiao, H.; Huang, Y.; Zhao, Y.; Ma, L.; Tian, Y. Physicochemical Properties and Structural Characterization of Crosslinking Modifications of Carboxymethyl Cellulose with Varying Degrees of Substitution. Food Chem. 2025, 496, 146617. [Google Scholar] [CrossRef]
- Kadioglu, E.; Demirbas, O.; Nas, M.S.; Calimli, M.H.; Bekmezci, M.; Sen, F. Synthesis and Characterizations of a Novel Composite Films Composed of Polyethylene/Oxide Minerals. Mater. 2023, 1, 100037. [Google Scholar] [CrossRef]
- Sanchez-Salvador, J.L.; Duque, A.; Lopez-Monte, D.; Cañadas, R.; Tarrés, Q.; Delgado-Aguilar, M.; Blanco, A.; Negro, C. Process-Intensified Production of TEMPO-Oxidized Cellulose Nanofibrils: Application to Several Lignocellulosic Feedstocks. Carbohydr. Polym. Technol. Appl. 2026, 13, 101079. [Google Scholar] [CrossRef]
- Santandrea, D.; Sillard, C.; Beghetto, V.; Bras, J. Aqueous-Phase Surface Amidation of TEMPO-CNF Films for Improved Adsorption of Organic Pollutants in Water. ChemPlusChem 2025, 90, e202500398. [Google Scholar] [CrossRef] [PubMed]
- Thakur, V.; Guleria, A.; Kumar, S.; Sharma, S.; Singh, K. Recent Advances in Nanocellulose Processing, Functionalization and Applications: A Review. Mater. Adv. 2021, 2, 1872–1895. [Google Scholar] [CrossRef]
- Tang, Z.; Lin, X.; Yu, M.; Mondal, A.K.; Wu, H. Recent Advances in TEMPO-Oxidized Cellulose Nanofibers: Oxidation Mechanism, Characterization, Properties and Applications. Int. J. Biol. Macromol. 2024, 259, 129081. [Google Scholar] [CrossRef]
- Tahir, D.; Karim, M.R.A.; Hu, H.; Naseem, S.; Rehan, M.; Ahmad, M.; Zhang, M. Sources, Chemical Functionalization, and Commercial Applications of Nanocellulose and Nanocellulose-Based Composites: A Review. Polymers 2022, 14, 4468. [Google Scholar] [CrossRef]
- Ma, H.; Wang, W.; Yu, W.; Kong, F.; Wang, S.; Liu, K.; Zhou, Z.; Wei, G.; Wang, X.; Liu, Y. Preparation of Dispersible TEMPO-CNF Ultrafine Powder and Its Application in Achieving Superhydrophobicity. Cellulose 2025, 32, 2245–2260. [Google Scholar] [CrossRef]
- Sabzevari, S.; Farrokhzad, H.; Poorkhalil, A. Development of Citric Acid-Crosslinked Carboxymethyl Cellulose /Chitosan Hydrogel Films Reinforced with ZnO Nanoparticles for Active Broccoli Packaging. Food Packag. Shelf Life 2025, 52, 101623. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




