1. Introduction
Aerobic granular sludge (AGS) has gained recognition as a promising alternative to conventional activated sludge (CAS) process in wastewater treatment [
1,
2]. AGS is a type of compact microbial aggregates formed under specific hydraulic and microbial selection pressures [
3]. AGS enables rapid solid-liquid separation and effective biomass retention, thereby improving treatment capacity and reducing the spatial requirement of wastewater treatment plants (WWTPs). Furthermore, the stratified structure of AGS promotes simultaneous nitrification and denitrification, which enhances nutrient removal efficiency and decrease the need for additional chemical additions. Overall, compared to CAS, AGS canreduce 25–50% of energy consumption and 25–75% of the footprint of the WWTPs [
3,
4].
With the advancement of aerobic granulation mechanisms and associated technologies, AGS has been successfully implemented in practical wastewater treatment applications, with approximately 100 AGS plants operated worldwide by the year of 2023 [
4,
5]. Besides, notable progress has been achieved in nutrient removal using AGS technology. Efficient and simultaneous removal of nitrogen and phosphorus has been demonstrated across a broad range of carbon-to-nitrogen (C/N) ratios, including at an excetionally low C/N ratio of 3.8 [
6,
7]. The widespread implementation of AGS has resulted in the generation of substantial quantities of waste AGS, thereby presenting new challenges and opportunities for treatment processes in both research and engineering domains. Currently, the research on AGS has progressed towards resource recovery and the promotion of sustainable development [
3,
5].
AGS has been demonstrated to be a promising raw material for resources reclamation [
8,
9,
10]. For instance, extracellular polymeric substances (EPS), which are integral to AGS formation, consist primarily of polysaccharides, proteins, nucleic acids, and other components [
11]. Polysaccharides provide the structural framework of AGS, whereas proteins enhance the hydrophobicity of EPS, thereby facilitating cellular adhesion [
3]. Notably, AGS secretes substantially greater quantities of EPS compared to CAS [
12]. Both polysaccharides and proteins represent valuable recyclable materials [
11]. These recoverable resources originate from the biologically mediated transformation and assimilation of organic pollutants and nutrients present in wastewater. They are contained within microbial cells, EPS, and inorganic constituents of AGS. To date, the recyclable substances identified in the literature predominantly include alginate-like exopolymers (ALE), polyhydroxyalkanoates (PHA), phosphate, tryptophan, xanthan, curdlan, and cellulose [
8,
9,
10].
The recovery of resources from waste AGS demonstrates considerable benefits [
10,
13,
14]. Firstly, if facilitates the efficient disposal of waste AGS. Secondly, it offers potential economic profits, particularly through the production of high value-added products [
13]. Thirdly, it contributes to the reduction of carbon emissions associated with wastewater treatment. Achieving resource recovery enables the reconstruction of carbon mass flow [
10]. Unlike the mineralization of organic carbon in wastewater, which results in carbon dioxide emissions, the microbial-mediated transformation of these organic compounds into high value-added polymers leads to lower carbon emissions. Furthermore, these bioproducts can serve as alternatives to the conventional industrial manufacturing processes, thereby contributing to the overall reduction in carbon emissions [
15,
16].
In this sense, the present paper aims to offer a comprehensive review of the recovery potential, methodologies and the current state of knowledge regarding each recyclable substance, including ALE, PHA, phosphate, tryptophan, xanthan, curdlan, and cellulose. Furthermore, recommendations for future research directions are proposed.
2. ALE Recovery from Waste AGS
2.1. Potential of ALE Recovery from Waste AGS
Alginate is a polysaccharide predominantly derived from brown algae and certain microorganisms, such as
Azotobacter sp. and
Pseudomonas sp. [
17,
18]. It serves multiple functional roles, including as a thickener, stabilizer, gelling agent, and medical materials. Consequently, alginate has been widely used in the fields of food, medicine, textiles, and environmental protection [
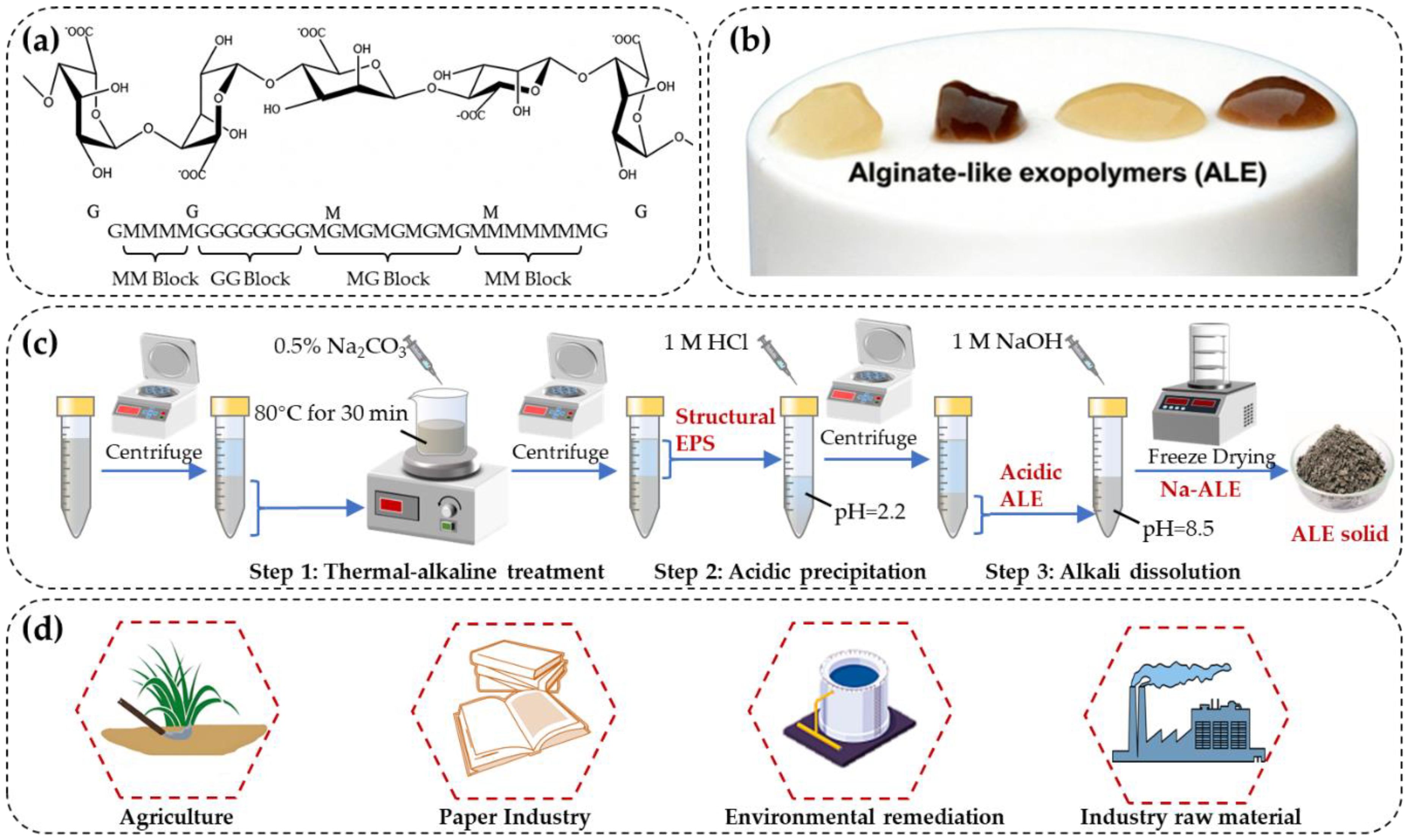
19]. Structurally, alginate is composed primarily of two uronic aced monomers: mannuronic acid (M) and glucuronic acid (G). These monomers are organized into homopolymeric blocks (MM and GG blocks) or heteropolymeric blocks (MG blocks), forming a linear, unbranched copolymer (
Figure 1a) [
16]. The GG blocks predominantly influence the gelation property of alginate, whereas the MG blocks affect the flexibility of polymer chain, and the MM blocks occupy the remaining structural regions [
5].
ALE extracted from sludge exhibits a more complex composition compared to natural alginate. In addition to the typical alginate structure, ALE contains proteins, various other polysaccharides, humic substances, lipids, and phosphate [
15]. ALE plays a critical role in the formation of AGS by interacting with calcium ions (
Figure 1b), which facilitates the formation of strong gels that provide a structural framework essential for AGS stability and integrity [
20,
21,
22,
23].
AGS demonstrates advantages over CAS in ALE recovery, particularly regarding ALE yield and gel-forming capacity. Comparative studies have indicated that the ALE content in the AGS (200–350 mg/g volatile suspended solids (VSS)) is substantially higher than that in CAS (55–190 mg/g VSS), highlighting AGS as a more promising source for ALE extraction [
24,
25,
26,
27,
28,
29]. In addition, ALE derived from AGS typically contains a greater proportion of GG blocks, which confers enhanced gel-forming properties [
5,
24,
30].
2.2. Factors Affecting ALE Generation in AGS
One aera of research focuses on improving ALE production through the optimization of operational parameters of the AGS system. Various factors influencing ALE production include sludge retention time (SRT), carbon sources, C/N ratio, organic loading rate, temperature, operational regime, salinity, and shear force, among others [
32].
Although AGS reactors are typically operated at high SRTs exceeding 30 d, evidence suggests that a moderately low SRT can enhance ALE yield. For instance, Rollemberg et al. (2021) reported that an SRT around 10 d promotes increased ALE production [
33]. Similarly, Amancio Frutuoso et al. (2025) identified an optimal SRT of 15 d for this purpose [
34]. Prolonged SRT may lead to biomass fermentation and consumption of ALE, whereas SRTs below 10 d could compromise the quality of the formed hydrogel [
34].
The type of carbon source exerts a significant effect on the production of ALE. Amanda et al. (2022) compared five different carbon sources and found that the highest ALE content was obtained when AGS was cultivated with acetate, yielding 418.7 mg ALE/g VSS. This was followed by propionate, glycerol, sucrose and glucose in descending order of ALE content [
35]. Similarly, Schambeck et al. (2020) reported that AGS fed with acetate and propionate produced substantially greater amounts of ALE (261 ± 33 mg ALE/g VSS) compared to sludge supplied with litter or no volatile fatty acids (VFAs) [
31]. These findings suggest that low molecular weight, readily biodegradable organic carbon sources promote ALE biosynthesis. Conversely, Li et al. (2022) reported divergent results, demonstrating that starch as the organic substrate yielded the highest ALE production (220.3 ± 8.0 mg/g VSS), surpassing that obtained with sodium acetate (156.4 ± 1.2 mg/g VSS) and glucose (125.7 ± 4.3 mg/g VSS) [
28].
In addition to the type of carbon source, both the organic load and C/N ratio affect ALE production. Li et al. (2022) reported the maximum ALE yield was obtained at a C/N ratio of 5 [
28]. A lower C/N ratio may lead to the utilization of EPS as an organic substrate for denitrification, whereas a higher C/N ratio tends to favor bacterial proliferation over ALE biosynthesis. A similar pattern was observed by Rollemberg et al. (2021), although they identified an optimal C/N ratio was determined at a considerably higher value of 20 [
33]. Concerning the organic load, a low organic load (0.20 kg BOD
5/kg VSS⋅d) led to a greater proportion of carbon towards ALE production, resulting in a specific ALE yield of 63.7 mg ALE/(g BOD
5), which is approximately double the yield observed under higher organic loads (0.36 and 0.50 kg BOD
5/kg VSS⋅d) [
28].
The particle size of AGS has been shown to affect ALE content, as reported by Rollemberg et al. (2021) [
33] and Zhang et al. (2025) [
36]. Despite variations in particle size ranges, granules of moderate particle size generally exhibited the highest ALE content.
The enhanced secretion of ALE has been recognized as an adaptive response to adverse environmental conditions [
32]. Consequently, the application of appropriate selective pressures, whether hydrodynamic or biological, can enhance ALE biosynthesis. For instance, the implementation of air pulse mixing during the anoxic period generating intermittent high sheer forces, resulting in significantly increased ALE production (172.4 ± 94.6 mg/g VSS) compared to recirculation mixing (63.7 ± 44.1 mg/g VSS) [
37]. Additionally, alternating feast and famine regimes and intermittent aeration, which preferentially select slow-growing heterotrophic microorganisms such as polyphosphate-accumulating organisms (PAOs) and glycogen-accumulating organisms (GAOs), promote ALE accumulation [
33,
38]. Prolongation of the anaerobic feast period further favors ALE synthesis; Frutuoso et al (2024) demonstrated ALE production peaked at the longest anaerobic duration (100 min) and diminished with the reduction of anaerobic duration in an AGS reactor fed with acetate [
39].
Other environment stressors, including reduced temperature, increased salinity, and the presence of microplastics, have also been shown to enhance ALE production. A decrease in temperature to 12 ◦C facilitated ALE production to 303.3 ± 21.5 mg/g VSS, approximately 2.5 times greater than that observed at 24 ◦C [
28]. Li et al (2017) reported that both ALE content and the proportion of GG blocks increased concomitantly with the rising proportions of seawater (0% to 100%) in the influent [
40]. Several investigations have further indicated that salt addition within the range of 2.5–10.0 g/L promotes ALE production in AGS systems relative to salt-free controls [
41,
42,
43]. Moreover, Jachimowicz et al. (2022) observed that microplastics stimulate ALE synthesis, with alginate content increasing from 238.7 ± 4.4 mg/g mixed liquor suspended solids (MLSS) in controls to 441.6 ± 13.8 mg/g MLSS at the highest polyethylene load (50 mg/L) in wastewater [
44]. However, elevated microplastic concentrations may adversely affect AGS settleability, causing biomass washout.
2.3. Optimization of ALE Recovery Methodology
The most widely employed protocol for ALE extraction was developed by Felz et al (2016) (
Figure 1c) [
45]. This protocol comprises three primary steps: (1) thermal-alkaline treatment of AGS to extract the structure EPS. AGS is treated with 0.5% sodium carbonate (Na
2CO
3) at 80◦C for 30 min, the insoluble ALE is transferred into sodium agminate, solubilized and separated from the solid phase. (2) acidic precipitation to isolate the acidic form of ALE. Following dialysis, the pH of supernatant obtained in step 1 is adjusted to 2.2 using 1 M HCl to precipitate the acidic form of ALE, which is collected as a gel-like pellet. (3) alkaline dissolution to obtain the sodium form of ALE. 0.5 M NaOH is gradually added to the gel to adjust the pH to 8.5, thereby solubilizing the acidic ALE and converting it into its sodium form. Finally, the solution undergoes dialysis and freeze-drying to yield solid ALE.
Efforts to optimize this extraction methodology have focused on improving both the recovery efficiency and purity of ALE. During the development of the protocol, Felz et al (2016) evaluated six commonly used EPS extraction techniques in step 1, including centrifugation, sonication, EDTA, formamide with NaOH, formaldehyde with NaOH, and Na
2CO
3 treatment [
45]. The results indicated that only the Na
2CO
3 method effectively solubilized the hydrogel matrix of AGS. Yang et al. (2025) investigated the influence of extraction temperature (50–95 ◦C) in step 1 on ALE yield [
46]. Results demonstrated that increasing the extraction temperature enhanced ALE recovery, with yield rising from 148.3 to 218.6 mg/g VSS when the temperature was elevated from 80◦C to 95◦C. Additionally, Liu et al. (2023) reported that substituting Na
2CO
3 with sodium percarbonate (2Na
2CO
3·3H
2O
2) during the thermal-alkaline treatment step improved ALE extraction efficiency by 30.11% [
47].
Pre-treatment strategies prior to the thermal-alkaline treatment step have also been explored. Li et al. (2022) employed various surfactants to enhance ALE extraction [
48]. The results indicated that surfactant application increased ALE yield from 124.1 mg/g VSS to 222.8–281.9 mg/g VSS, while simultaneously improving alginate purity to 54%–70%. This enhancement was attributed to surfactant-facilitated solubilization and desorption processes, the rule of “like dissolves like”, and adsorption via functional groups.
2.4. Potential Utilization of ALE
Natural alginate serves multiple functional roles, including thickeners, stabilizers, gelling agents, and biomedical materials, and finds extensive applications across the food, pharmaceutical, textile, and environmental protection sectors [
19]. However, the use of ALE in food and medical applications is generally discouraged due to concerns regarding their lower purity and the potential presence of harmful constituents. ALE may contain contaminants such as heavy metals, micropollutants, microplastics, and antibiotic resistance genes, which often originate from wastewater sources [
49,
50]. Although mass balance studies of sulfamethoxazole (SMX), a commonly used antibiotic, during ALE extraction have indicated minimal SMX presence in the ALE phase [
49], comprehensive investigations and evaluations of these hazardous components within ALE remain necessary.
To date, ALE has primarily been employed as a cost-effective alternative to natural alginate in the lower-value markets (
Figure 1d). In environmental applications, ALE has demonstrated utility as an adsorbent material. AGS-derived ALE has been employed for the adsorption of heavy metals such as Cd
2+ and Cu
2+ [
51,
52], exhibiting adsorption capacities of 132.31 ± 0.40 mg/g ALE for Cd
2+ and 71.85 ± 0.40 mg/g ALE for Cu
2+ [
51]. Notably, ALE recovered from a full-scale AGS plant achieved a high Cd
2+ removal efficiency of 94.4 ± 0.9% [
52]. Additionally, ALE has been applied for phosphate removal from wastewater [
53,
54,
55,
56]. For instance, Agnol et al. (2020) reported that AGS-derived ALE exhibited superior phosphorus removal capabilities compared to commercial seaweed-derived alginate [
53].Schambeck et al. (2021) utilized AGS-derived ALE to remove phosphate from municipal wastewater, attaining a removal efficiency of 90.8% under optimized conditions. Nevertheless, further modification of ALE hydrogels is warranted, as increases in chemical oxygen demand (COD), total nitrogen (TN), and total phosphorus (TP) in the bulk liquid were observed [
54]. Furthermore, Ladnorg et al. (2019) confirmed that AGS-derived ALE functions effectively as a biosorbent for the dye methylene blue, achieving a maximum removal efficiency of 69 % [
57].
Beyond environmental remediation, ALE has potential applications in agriculture as a fertilizer and in the paper packaging industry as a surface coating. Pagliaccia et al. (2024) assessed the feasibility of utilizing structured EPS (sEPS), predominantly comprising ALE, extracted from AGS in agricultural contexts. The results indicate that AGS-derived sEPS can be transformed into valuable biomaterials that enhance soil water retention and nutrient holding capacity [
58]. Moreover, studies have shown that AGS-derived ALE readily forms films on hydrophilic surfaces such as paper, functioning effectively as a water-resistant barrier. This property underscores its potential use as a surface coating material in paper packaging applications [
59,
60].
2.5. Reals Application of ALE Recovery from Waste AGS
Advancements in research on ALE production enhancement and extraction methods have facilitated the establishment of practical bio-ALE production installations in the Netherlands. The extraction of ALE from waste Nereda® AGS is also referred to as Kaumera Nereda® Gum. In 2020, two plants have been operated in Zutphen and Epe, producing 400 and 100 tons of Kaumera, respectively. The production yield of Kaumera was estimated at 18 kg of ALE per 80 kg of Nereda® AGS. By extracting Kaumera from the purified sludge, the amount of sludge requiring removal and processing is reduced by 20–35%, which favors the reduction in energy consumption and CO
2 emissions. Kaumera can be utilized in agriculture, the building sector, as well as the textile and paper industries [
13].
3. PHA Recovery Using Wasted AGS
3.1. Significance and Strategy of PHA Recovery in Wastewater Treatment
PHA represents a kind of natural polymers synthesized through microbial fermentation utilizing various organic carbon sources. In addition to exhibiting physical and chemical properties comparable to conventional petroleum-based plastics, PHA possesses advantageous characteristics such as biodegradability, biocompatibility, and effective gas-barrier properties [
61]. Consequently, PHA has been recognized as a promising alternative to conventional petroleum-based plastics, contributing to the mitigation of environmental pollution commonly referred to as “white pollution”. Their applications span diverse sectors, including industrial manufacturing, agricultural mulching, medical device fabrication, and the development of nanocomposite materials [
62].
The traditional production of PHA is cost-prohibitive, primarily due to the necessity of maintaining pure or genetically engineered bacterial cultures and the reliance on expensive organic substrates. Recently, attention has shifted towards the utilization of mixed microbial cultures, particularly those derived from waste biomass generated in wastewater treatment processes, as a more economical approach to PHA production [
63].
A broad spectrum of microorganisms is capable of intracellular PHA accumulation [
64]. Notably, PAOs and GAOs, commonly found in biological wastewater treatment systems, also demonstrate the capacity for PHA storage. This suggests that CAS can serve as aa viable for PHA production. A survey assessed the PHA accumulation potential (PAP) of sludge from fifteen WWTPs in the Netherlands, revealing activated sludge from four WWTPs exhibited PHP exceeding 0.38 g PHA/g VSS [
65]. Furthermore, Kumar et al (2020) reported that conditions characterized by an excess of organic carbon substrates, nutrient limitation, and low dissolved oxygen (DO) promote microbial PHA accumulation as an energy reserve during starvation periods [
63].
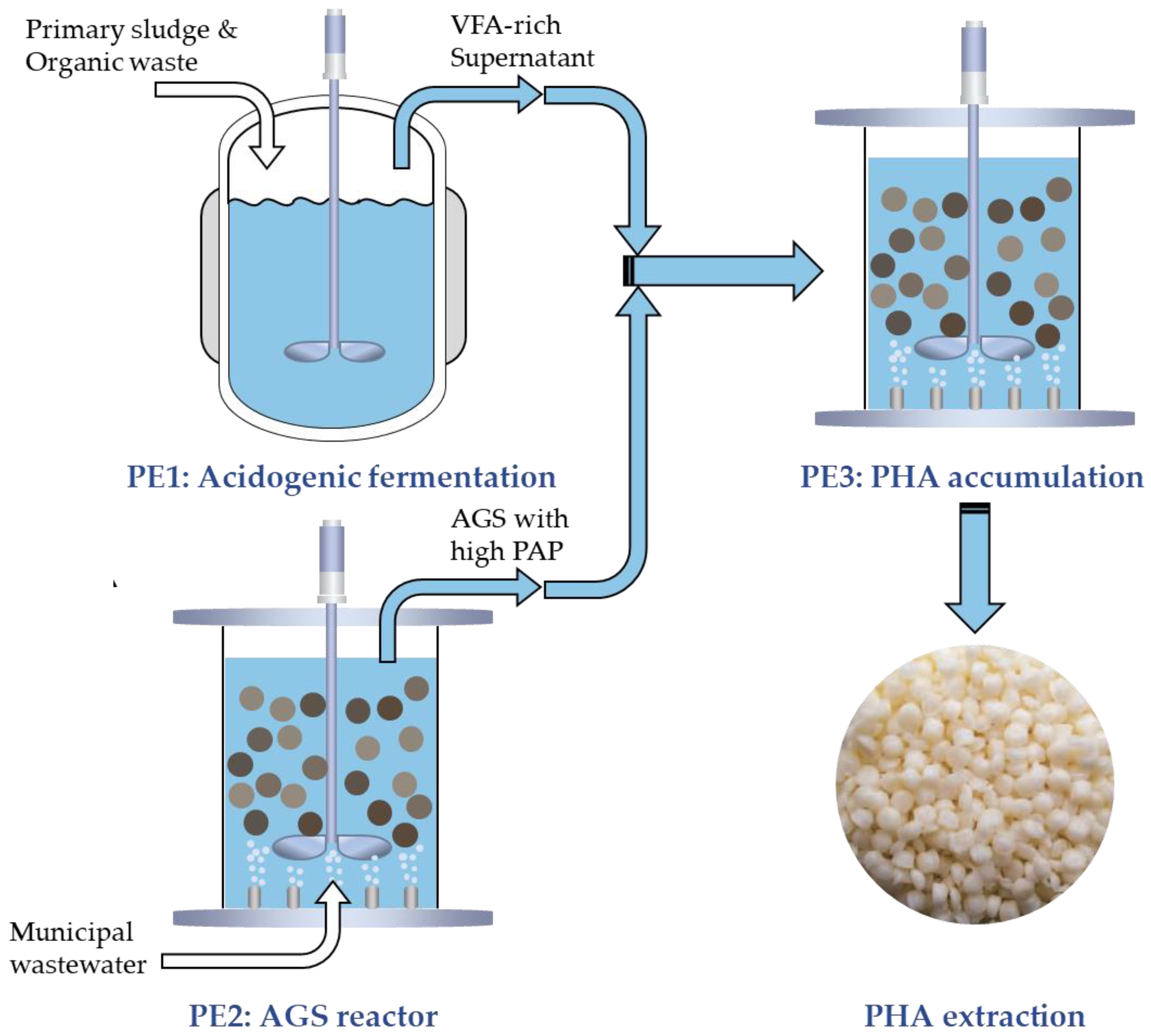
To facilitate consistent PHA production from activated sludge harvested from full-scale WWTPs, the PHARIO process has been developed [
65]. This process comprises three sequential biological process elements (PEs): PE1 involves acidogenic fermentation to generate VFAs from a mixture of primary sludge and organic waste; PE2 focuses on the enrichment and proliferation of biomass with PHA-storing capacities in the full-scale WWTP process; and PE3 entails PHA accumulation within biomass by utilizing surplus activated sludge from PE2 and fermentation filtrate from PE1 as feedstocks (
Figure 2). This integrated approach yielded PHA rich biomass with an average PHA content of 0.41 g PHA/g VSS.
3.2. PHA Recovery Using Waste AGS
In comparison to CAS process, the alternative anaerobic feast and aerobic famine regime is frequently employed in the cultivation of AGS [
66]. This approach promotes the enrichment of PHA-accumulating PAOs and GAOs, thereby potentially yielding a higher PAP relative to CAS. Numerous studies have reported elevated abundances of PAOs and GAOs within various AGS systems [
67,
68,
69].
However, direct extraction of PHA from waste AGS generated from WWTPs has demonstrated limited feasibility. The PHA content in waste AGS documented in the literature ranges from 0.1 to 0.215 g PHA/g VSS [
70,
71,
72], which falls below the commercial threshold criterion of 40% PHA content [
65]. Several factors contribute to the relatively low PHA yield observed in AGS. Firstly, a substantial portion of organic carbon present in the wastewater is diverted towards biological nutrient removal processes. Secondly, VFAs in wastewater may be preferentially utilized for EPS synthesis rather than PHA production, a mechanism that supports the structural integrity of the granules [
72,
73]. Thirdly, the extended SRT characteristic of AGS systems may lead to endogenous respiration of the stored PHA, thereby diminishing its overall production.
Given these limitations, the valorization of waste AGS as a feedstock for PHA production, such as its application as PE2 within the PHARIO process, may offer a more advantageous strategy, potentially yielding products with higher PHA content [
74].
4. Phosphorus Recovery Using AGS
4.1. Potential of Phosphorus Recovery Using AGS
Phosphate minerals constitute a critical raw material for fertilizer fabrication and various chemical industries. Nevertheless, phosphate minerals represent a finite resource, with global reserves projected to be depleted between 2070 and 2099, raising significant concerns worldwide [
75]. Phosphorus flow analyses indicate that 20–30% of mineral phosphorus is lost to wastewater through human excreta [
76]. Consequently, wastewater represents a substantial source of phosphorus and the recovery of phosphorus from these streams could mitigate the impending phosphorus scarcity.
Direct recovery of phosphorus from raw wastewater is challenging due to typically low phosphorus concentrations (<10 mg/L) [
77]. Within WWTPs, up to 90% of phosphorus is converted into the sludge phase via biological and chemical pathways. This phosphorus faction can be partially released during the anaerobic digestion of waste sludge, resulting in elevated phosphorus concentrations that are conducive to recovery efforts [
10].
AGS systems demonstrate considerable potential for phosphorus recovery. Compared to CAS systems, AGS offers enhanced biological phosphors removal (EBPR) capabilities, enabling the excessive uptake of phosphorus. The typical operational regime of AGS reactors, characterized by alternating anaerobic feast and aerobic famine phases, promotes the enrichment of slow-growing PAOs [
66]. Additionally, simultaneous nitrification/denitrification in AGS systems reduces organic carbon consumption, thereby preserving more organic substrates for PAOs. Excess AGS contains high levels of polyphosphate (Poly-P), which can be readily released during anaerobic digestion [
78]. Furthermore, the longer SRT typically employed in AGS systems contributes to the elevated phosphorus content relative to CAS systems [
68].
In addition to Poly-P, AGS systems exhibit accumulation of precipitated phosphate minerals, including hydroxyapatite (Ca
5(PO
4)
3OH), calcium phosphate (Ca
2(PO
4)
3), struvite (MgNH
4PO
4∙6H
2O) and iron phosphate (Fe
7(PO
4)
6), among others [
79,
80,
81,
82,
83,
84]. This accumulation results in phosphorus contents as high as 150.7 ± 28.5 mg P/g suspended solids (SS). The extent of this precipitation is influenced by the concentration of relevant metal ions in wastewaters. Extended SRTs (e.g., 30 d) and favorable micro-environmental conditions are critical for phosphorus precipitation. Moreover, PAO metabolism facilitates this process, particularly through the anaerobic release of phosphate, which elevates phosphate concentrations and promotes precipitation with Ca
2+ and Mg
2+ ions. Angela et al. (2011) reported that biologically induced phosphorus precipitation can account for 45% of total phosphorus removal under normal pH conditions (7.8–8.8) in AGS systems [
81].
The abundant EPS may also contribute to the high phosphorus content in AGS [
85,
86,
87,
88]. Reported contributions of EPS to total phosphorus content vary widely, ranging from 6.4% to 47% across different studies. This fraction of phosphorus can be concurrently recovered during EPS extraction procedures. For instance, Chen et al. (2022) reported that the EPS extracted from bacterial AGS contained approximately 25.10 ± 1.85 mg P/g SS [
87]. Furthermore, Bahgat et al. (2024) confirmed the predominance of organic phosphorus (OP) and orthophosphate (Ortho-P) within the EPS extracts [
88].
4.2. Species and Distribution of Phosphorus in AGS
Phosphorus in its existing forms is primarily categorized into OP and inorganic phosphorus (IP) [
78]. Specifically, OP predominantly comprises monoester-P and diester-P, which are present in adenosine triphosphate (ATP), cell membrane phospholipids, and nucleic acids. IP, classified based on the degree of phosphate polymerization, manly includes Ortho-P, pyrophosphate (Pyro-P), and Poly-P. These phosphorus species are distributed within microbial cells, EPS, and mineral precipitates in AGS.
The forms and spatial distribution of phosphorus vary among AGS derived from different sources, with the EBPR capacity of the AGS systems serving as a key influencing factor. Huang et al. (2015) examined the species and distribution of phosphorus in an AGS system capable of EBPR and reported that microbial cells, EPS, and mineral precipitates account for approximately 73.7%, 17.6% and 5.3–6.4% of the TP in AGS, respectively [
78]. Poly-P was identified as the predominant phosphorus species in both cells (64.1 ± 6.2%) and EPS (45.7 ± 6.8%), while OP was primarily localized in the cells. Furthermore, hydroxyapatite and calcium phosphate were the dominant phosphorus minerals accumulated in the granule cores. In contrast, Huang et al. (2015) observed that Ortho-P was the dominant phosphorus species in nitrifying AGS lacking PAOs, with hydroxyapatite and iron phosphate (Fe
7(PO
4)
6) constituting the major mineral phosphorus forms [
80].
4.3. Phostrip-like Sidestream Phosphorus Recovery Integrated with AGS Process
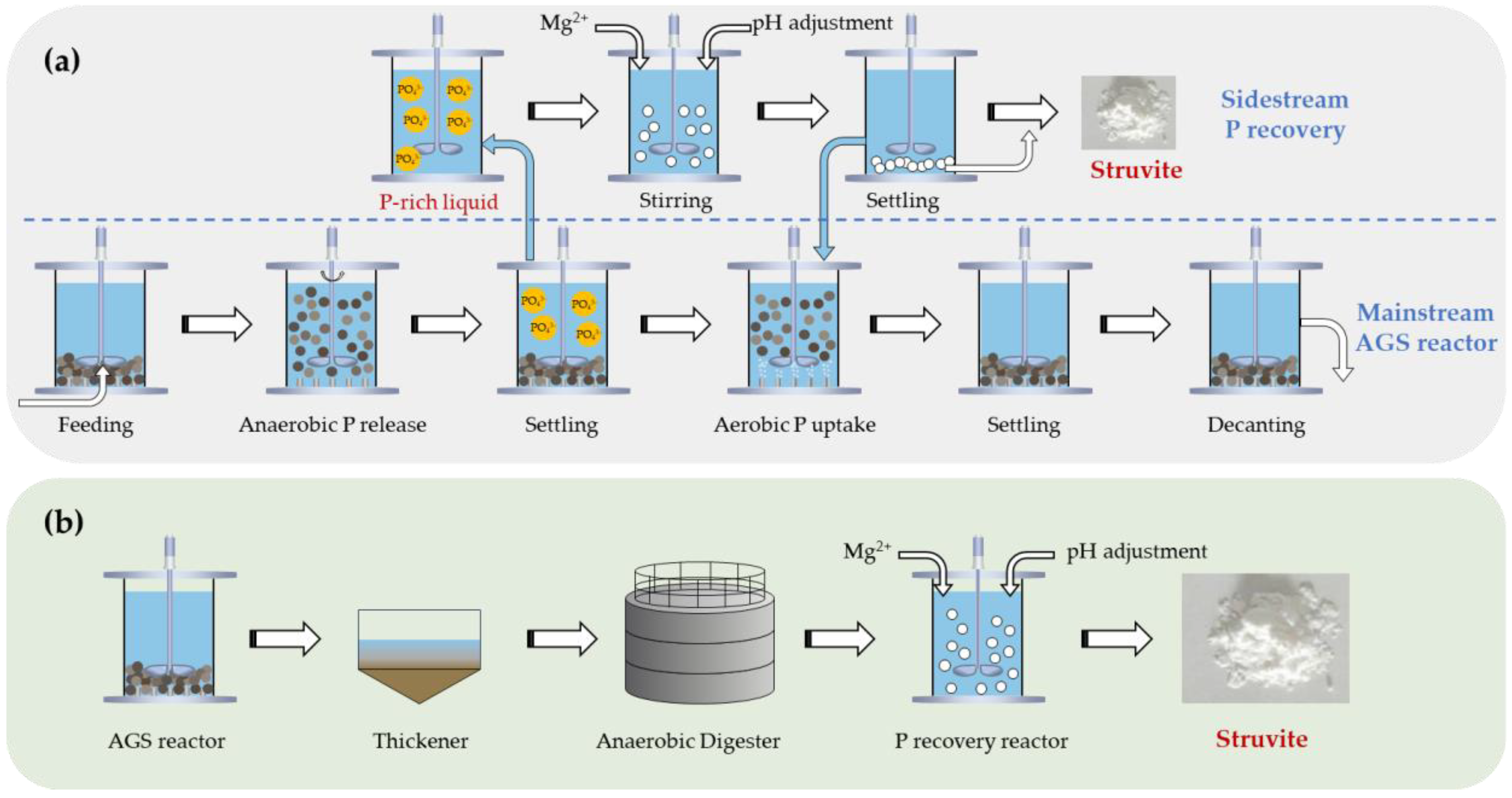
One approach for phosphorus recovery involves Phostrip-like sidestream phosphorus recovery utilizing the phosphate-rich supernatant obtained at the end of the anaerobic phase of AGS system, attributable to anaerobic phosphate release by PAOs [
89]. A portion of the supernatant, typically 20–30%, is extracted and introduced into a sidestream reactor for phosphorus recovery. The treated solution is subsequently returned to the AGS reactor for further treatment. This approach can decrease the phosphate load on WWTPs, reduce organic substrate demand, and enhance nutrient removal efficiency. Nevertheless, concerns exist regarding potential over-stripping of phosphate, which may destabilize EBPR performance, for instance, by enabling GAOs to outcompete PAOs.
He et al. (2023) extracted 30% of the phosphorus-rich supernatant from the AGS system for phosphorus recovery, estimating that 63.4% of the total influent phosphorus could be reclaimed without adversely affecting the nutrient removal performance [
89]. Wang et al. (2022) extracted 40 mL of phosphorus-rich liquid daily from the bottom of 250 mL AGS sequencing batch reactor (SBR), achieving a P recovery efficiency of 83.85 ± 0.57 % [
77].
An alternative phosphorus recovery approach was proposed by Lu et al. (2016) [
90]. Phosphorus-rich sludge was extracted at the end of aerobic phase at an amount of 1 L from an 8 L AGS-SBR once per day. This sludge was supplemented with 640 mg/L of COD in the form of acetate to enhance phosphorus release. Subsequently, the sludge was separated using the non-woven cloth and returned to the AGS-SBR, while phosphorus-rich supernatant was subjected to chemical precipitation phosphorus recovery. This method enables the transfer of 93.19% of influent phosphorus into the phosphorus-rich supernatant, which was further recovered as struvite. However, this strategy led to a decline in denitrification efficiency, indicating the need for further optimization.
Figure 3.
(a) Phostrip-like sidestream phosphorus recovery integrated with AGS process; (b) Phosphorus recovery from the anaerobic fermentation/digestion supernatant of waste AGS.
Figure 3.
(a) Phostrip-like sidestream phosphorus recovery integrated with AGS process; (b) Phosphorus recovery from the anaerobic fermentation/digestion supernatant of waste AGS.
4.4. Phosphorus Recovery from the Anaerobic Fermentation/Digestion Supernatant of Waste AGS
Another approach for phosphorus recovery is the utilization of the anaerobic fermentation/digestion supernatant of waste AGS, which contains high concentrations of phosphate. Struvite and hydroxyapatite are the predominant forms of recovered phosphorus [
10]. Struvite serves as a controlled-release fertilizer. A key advantage of struvite crystallization lies in its capacity to simultaneous remove phosphate and ammonium, thereby reducing the nitrogen load in WWTPs. Additionally, the supernatant from sludge digestion typically contains high concentrations of phosphorus, ammonium, and magnesium, which favor the crystallization of struvite. This process generally occurs under alkaline pH from 7 to 11 and a magnesium-to-ammonium-to-orthophosphate molar ratio of 1.5:1:1 [
10]. The pH level exerts a significant influence on phosphate precipitation. Cydzik-Kwiatkowska and Nosek (2020) reported that the precipitation efficiency was 30.13% ± 4.51% under uncontrolled pH conditions, increasing markedly to 81.73% ± 0.17% when the pH was maintained at 10 [
91].
It is important to note that the dense structure of AGS may impede phosphorus release during anaerobic fermentation/digestion. Various strategies have been proposed to enhance phosphorus release from AGS, including thermal pretreatment, optimization of fermentation pH, and increasing the food-to-microorganism (F/M) ratio. Zou and Li (2016) demonstrated that low-temperature thermal pretreatment (70°C for 60 min) improved the TP release efficiency from phosphorus-accumulating granular sludge to 93.4 ± 0.6 %, compared to 80.0 ± 7.1 % in untreated samples [
92]. Furthermore, Zou et al. (2018) examined the impact of fermentation pH on phosphorus release from nitrifying AGS [
93]. The results indicated that acidic conditions (pH = 4) significantly enhanced phosphorus release to 25.3 mg P/g SS, relative to alkaline and uncontrolled pH conditions, which yielded 4.4–5.4 mg P/g SS. However, acidic conditions also exhibited an inhibitory effect on the release of soluble COD (SCOD) and VFAs. Moreover, increasing the F/M ratios by the addition of external sodium acetate was found to augment the phosphorus release rate. The highest phosphorus release of 60.71 mg P/L from AGS was achieved at an F/M ratio of 0.04 [
91].
5. Methane Recovery from Waste AGS
5.1. Methane Recovery Potential from Waste AGS
Methane generation via the anaerobic digestion of waste AGS is a promising avenue for achieving energy neutrality in wastewater treatment processes. The implementation of AGS has been shown to reduce the energy consumption of WWTPs by 20–50% [
4]. Furthermore, the recovery of methane from waste AGS can enhance the energy self-sufficiency of WWTPs.
Typically, AGS exhibits a higher organic content, as indicated by elevated VSS/SS ratio, compared to CAS. However, numerous studies have reported that the potential for methane recovery from AGS is generally lower than that from CAS [
94,
95,
96,
97]. A primary factor limiting methane recovery is the dense EPS-rich structure of AGS, which may impede the release of soluble organic compounds, particularly in large granules. Wang et al. (2005) demonstrated that AGS possesses an outer EPS layer characterized by poor solubility and limited biodegradability, whereas the readily soluble and biodegradable EPS is predominantly located in the inner regions [
98]. Another critical factor influencing methane recovery is the long SRT commonly employed in AGS systems, often exceeding 30 d, which results in a higher proportion of refractory microbial products within the sludge. It has been documented that the biodegradability of proteins, polysaccharides, and lipids in sludge decreases with increasing sludge age [
99]. Additionally, Guo et al. (2020) reported that the polysaccharides and proteins constituting the structural EPS in AGS are more refractory compared to those found in CAS [
100].
5.2. Strategies to Enhance Methane Recovery from Waste AGS
Multiple approaches have been investigated to improve methane recovery from waste AGS, including regulating sludge discharge from AGS reactors, physical pretreatments such as homogenization and ultrasound, thermal-alkaline pretreatment, and optimization of the anaerobic digestion process.
Guo et al. (2020) evaluated the biochemical methane potential (BMP) of two types of waste AGS obtained a full-scale municipal WWTPs employing different sludge discharge strategies [
95]. The results indicated that selectively discharged AGS, which is discharged with the effluent in each cycle due to low particle size and settling velocity, exhibited a higher BMP of 296 ± 15 mL CH
4/g VS compared to mixed excess AGS removed for SRT control with 194 ± 10 mL CH
4/g VS. Dababat et al. (2023) also observed that the larger granules of 250 μm yield significantly less methane than smaller granules [
94]. Yang et al. (2023) developed a two-stage system to achieve autotrophic nitrogen removal from municipal wastewater. The AGS reactor functioned as the A-stage for organic matter removal, operating with EBPR regime under a high organic loading rate of 6.9 ± 1.5 kg COD/(m
3•d) and a short SRT of 8 days. Waste AGS from this system exhibited higher methane production (0.59 g COD
CH4/g SS) compared to CAS (0.35 g COD
CH4/g SS) [
68].
Physical pretreatment techniques have been applied to disrupt the granular structure of AGS. Cydzik-Kwiatkowska et al. (2022) demonstrated that ultrasound pretreatment releases approximately an order of magnitude more organic matter from AGS compared to homogenization, significantly enhancing methane-rich biogas production (455 L/kg VS, 66% CH
4) [
101]. Similarly, Kosar et al. (2025) reported that ultrasonication of 0.5 L of thickened AGS at 100 W for 1 min increased SCOD concentration from 3006 ± 61 mg/L to 5274 ± 59 mg/L relative to untreated AGS [
102]. These findings suggest that ultrasound pretreatment is an effective method to improve AGS disintegration and digestibility.
Thermal pretreatment has been employed to the anaerobic digestion of two different AGS, treating pig manure (G1) and simulated urban wastewater (G2), respectively [
97]. The results indicate that thermal pretreatment markedly enhanced the anerobic biodegradability of G1, with improvements of 20% at 60°C to 88% at 170 °C compared to untreated sludge. Conversely, only marginal increases in methane production were observed for G2 following thermal pretreatment. The authors advocate for the implementation of thermal pretreatment particularly when the initial anaerobic biodegradability is low. A similar pattern was reported by Liu et al. (2019), who applied steam explosion as a thermal pretreatment to AGS containing 10% (G1) and 39% mineral content (G2). Untreated G1 produced 30% more biogas than untreated G2; however, steam explosion was more effective in enhancing methane production in G2 [
103]. Cicekalan et al. (2025) reported that low-temperature thermal-alkaline pretreatment prior to anaerobic digestion increased sCOD release and methane production from AGS, achieving a maximum methane yield of 216 ± 22 mL CH
4/g VS at 100 ◦C and pH 10 [
104]. In contrast, pretreatment at higher pH levels (11 and 12) resulted in diminished improvements in methane production.
Beyond physicochemical pretreatment approaches, biological pretreatment methods such as dosing alginate-degrading consortia have been shown to enhance EPS hydrolysis and methane production (by 115%−185%) from waste sludge [
105].
Additional strategies to improve methane recovery from waste AGS involve optimizing anaerobic digestion operational parameters. For instance, Bernat et al. (2017) reported that co-digestion of AGS with primary sludge yielded higher biomass potential and methane content compared to digestion of AGS alone [
96]. Diniz et al. (2025) operated the anaerobic digestion process at elevated pH (~9.3) and alkalinity (~0.5 eq/L) using alkaline waste derived from EPS extraction from AGS [
106]. The biogas methane purity consistently exceeded 90%, indicating effective in-situ biogas purification.
5.3. Integration of Methane Recovery with Other Resource Reocovery from Waste AGS
The Waste AGS contains multiple recoverable organic compounds, including ALE, PHA, and tryptophan, which possess significantly higher economic value compared to methane [
107]. Consequently, the direct application of anaerobic digestion to waste AGS for methane production. Instead, anaerobic digestion can be employed as a terminal treatment step for the solid residues following the extraction of other valuable resource. This sequential approach not only maximizes the final economic benefits but also promotes sludge stabilization. Bahgat et al. (2023) investigated multiple resource recovery from AGS and estimated that approximately 30% of sludge organics could be recovered as EPS, while 25–30% could be converted to methane [
108]. Similarly, Chen et al. (2024) demonstrated that a conceptual AGS-based WWTP processing 10,000 m
3/d of wastewater, with ALE, phosphorus, and methane recovery, could generate an annual net profit of approximately 66,300 USD [
109]. It is important to note that prior resource recovery procedures may affect AGS biodigestibility. Liu et al. (2023) reported a potentially positive effect, whereby the extraction of polysaccharides induces cell lysis, thereby enhancing hydrolytic activity during anaerobic digestion and increasing methane production [
110]. Furthermore, anaerobic digestion can serve as a pretreatment step for phosphorus recovery. Chen et al. (2025) observed that anaerobic digestion of AGS produced a substantial methane yield of 193 mL/ g VS and phosphorus release of 393.22 mg/L to the digestate liquid [
111].
6. Other Recoverable Materials in AGS
6.1. Tryptophan
Tryptophan is an amino acid with diverse application across the chemical, agriculture, and particularly pharmaceutical industries. It is recognized as an essential dietary component for humans, as it cannot be endogenously synthesized [
10]. Within the EPS of AGS, Tryptophan is one of the predominant hydrophobic amino acids, alongside tyrosine [
112]. The elevated presence of tryptophan contributes to increase EPS hydrophobicity, thereby facilitating cell adhesion [
113,
114,
115]. Consequently, tryptophan plays a critical role in the formation and stability of AGS. Comparative analyses have demonstrated that the tryptophan content in AGS is nearly double that found in CAS [
116]. Furthermore, an assessment of resource recovery potential in a pilot-scale AGS reactor treating low-strength municipal wastewater indicated that the resultant sludge content a high concentration of tryptophan, reaching 0.048 g TRY/g VSS [
117].
Several factors influence tryptophan, including the type of carbon source, operational regime, SRT, granule size, C/N ratio, microbial community composition, and iron concentration. Amanda et al. (2022) investigated the effect of various carbon sources on tryptophan biosynthesis in AGS, reporting the highest biosynthesis level was achieved with acetate (4.1 mg TRY/g VSS), followed by propionate, glycerol, sucrose and glucose [
35]. Rollemberg et al. (2021) conducted a systematic study on factors affecting tryptophan content in AGS [
33]. Results indicate that a short anoxic phase within an anaerobic/aerobic/anoxic cycle and an SRT of 10 d enhanced tryptophan production. Granules with diameter of 0.5–1.0 mm exhibited the highest tryptophan content. Additionally, a high C/N ratio of 30 resulted a decline in tryptophan production. The presence of EPS-producing fermentative bacteria was suggested to be associated with tryptophan biosynthesis. Frutuoso et al (2024) observed increased tryptophan levels with shorter anaerobic phase periods in AGS systems fed with acetate and propitiate [
39]. Moreover, da Silva et al. (2025) reported that iron doping can enhanced tryptophan recovery, reaching up to 8.6 mg TRY/g VSS [
118].
6.2. Xanthan
Xanthan is a polysaccharides synthesized by various species of the genus
Xanthomonas and is widely employed as a thickening and suspending agent across food, chemical, and petroleum industries [
119]. Conventional production of xanthan involves aerobic fermentation of carbohydrate-rich substrates by strains such as
Xanthomonas campestris and
Xanthomonas pelargonii, which is energy-intensive, feedstock-depended and costly. Alternatively, xanthan production has been reported during wastewater treatment processes; for instance, yields ranging from 20.92 to 30.64 g/L have been documented in winery wastewater treatment systems [
120,
121]. AGS is a promising resource for xanthan recovery, with xanthan gum predominantly localized in loosely-bound EPS. Notably, the highest xanthan yield observed in AGS was 159 ± 53 mg/g VSS [
118].
The influence of operational parameters such as OLR, C/N ratio, and feeding strategy on xanthan production in AGS systems has been systematically investigated [
122]. A strong positive correlation was identified between OLR and xanthan yield, with the maximum yield occurring at an OLR of 2.1 kg COD/m³•d. Conversely, the C/N ratio exhibited a moderate but statistically non-significant negative correlation with xanthan production. Feeding strategy appeared to exert minimal impact on xanthan yield.
Rončević et al. (2019) developed a methodology for xanthan gum recovery [
121]. The first step involves the extraction of EPS from granular sludge, followed by xanthan precipitation using 96◦GL ethanol. Subsequent centrifugation and drying steps facilitate the recovery of xanthan solids. da Silva et al. (2026) optimized this methodology, achieving a maximum recovery efficiency of 97.5% through centrifugation at 4 ◦C followed by oven drying at 50–55 ◦C [
123]. Furthermore, results demonstrated a higher xanthan yield during the maturation phase of AGS of 139 ± 73 mg/g VSS compared to both the inoculum and granulation phases.
6.3. Curdlan
Curdlan is a distinct type of polysaccharide identified within EPS. It possesses the ability to form gels upon heating and has found applications across various sectors, including the food, pharmaceutical, and cement industries [
124,
125]. Microorganisms implicated in curdlan biosynthesis include
Agrobacterium,
Rhizobium, and
Alcaligene sp., and
Paenibacillus species [
125]. Reported curdlan contents in AGS range from 0.1 to 6.0 mg/g VSS [
118,
123]. Furthermore, enhanced curdlan production has been observed under saline and iron-doped conditions [
118].
The curdlan recovery process typically involves pH adjustment, centrifugation, washing of precipitates, and drying [
126]. da Silva et al. (2026) reported a maximum curdlan recovery efficiency of 98%, with no significant difference observed between centrifugation temperatures of 4 ◦C and room temperature. The average curdlan recovery was 5 ± 1 mg/g VSS, with loosely bound EPS demonstrating higher recovery efficiency [
123].
6.4. Cellulose
Cellulose present in municipal wastewater primarily originates from toilet paper. A significant proportion of cellulose, ranging from 13% to 15%, has been identified in the waste AGS produced by the Nereda® wastewater treatment technology. Espíndola et al. (2021) have developed a cellulose extraction method that enables the recovery of pulp with a purity exceeding 86 wt%. The cellulose fibers derived from wastewater represent a promising raw material for the production of recovered cellulose nanocrystals [
127].
7. Future Perspectives
7.1. Sequential recovery of multiple resources from AGS
Previous studies have predominantly concentrated on the extraction of individual resource from waste AGS. Nonetheless, AGS comprises a variety of recoverable materials, including ALE, PHA, phosphate, tryptophan, xanthan, curdlan, and cellulose [
8,
9,
10]. To optimize the overall recovery yield and economic benefits, it is imperative to explore the sequential recovery of multiple resources from waste AGS [
108,
128]. This approach necessitates the integration of the distinct recovery methodologies tailored to each resource, considering that the extraction process of one component may influence the subsequent recovery of others resources. Therefore, future research should focus on optimizing the sequential extraction protocols and evaluating the net benefits associated with such integrated recovery strategies.
7.2. Integration of AGS with Emerging Technologies to Enhance organic Recovery Efficacy
Most recyclable compounds within AGS, such as ALE, PHA, tryptophan, xanthan, and curdlan, originate from the biologically mediated transformation and assimilation of organic pollutants present in wastewater [
107]. However, organic carbon is also a critical factor in nutrient removal processes. The depletion of organic carbon can limit the availability of substrates necessary for the biosynthesis of recoverable materials, thereby diminishing their production.
Emerging technologies, including microalgal treatment and the partial nitritation/anammox (PN/A) process, have demonstrated potential in reducing the organic carbon demand for nutrient removal from wastewater and can be synergistically integrated with AGS technology. For instance, the development of algal-bacterial AGS not only introduces novel resource recovery opportunities, such as lipid extraction, but also influences the synthesis of other valuable compounds like ALE, PHA, and phosphate [
37,
67,
109]. Although preliminary investigations have been conducted, further research is warranted to optimize the resource recovery from algal-bacterial AGS. Additionally, AGS has been employed as a pretreatment stage in PN/A processes, functioning as the A-stage to facilitate EBPR and remove organic carbon, while PN/A achieves autotrophic nitrogen removal without the reliance on organic carbon sources [
68]. This integrated approach maximizes carbon flux towards resource recovery. Despite the demonstrated feasibly of utilizing AGS as the A-stage in PN/A systems [
68], comprehensive studies on the transformation of organic carbon and the associated resource recovery potential remain limited and should be addressed in future research.
8. Conclusions
This study presents a comprehensive review of resource recovery from AGS. Among the various recoverable resources, ALE is the most intensively studied component, demonstrating considerable technical maturity and practical application, exemplified by the Kaumera Nereda® Gum. AGS also shows significant potential for PHA accumulation, positioning it as a viable substrate for PHA production. Phosphorus can be recovered by chemical precipitation using Phostrip-like sidestream process coupled with AGS reactor or from the supernatant produced during anaerobic fermentation or digestion of waste AGS. Methane recovery from AGS is often constrained by the dense EPS matrix, necessitating enhancement via pretreatment techniques. Emerging recoverable compounds in AGS include tryptophan, xanthan, and curdlan. To enhance economic viability, strategies enabling the sequential recovery of multiple resources from AGS are essential. Furthermore, coupling AGS with microalgal treatment and partial nitritation/anammox process has potential to improve the efficiency of organic carbon reclamation from wastewater.
Author Contributions
Conceptualization, Y.Y. and S.Z.; methodology, Y.R., and Z.Q.; investigation, Z.Q., Y.R. and Y.Y.; writing—original draft preparation, Z.Q.; writing—review and editing, Y.Y. and S.Z.; Supervision, S.Z.; funding acquisition, Y.Y. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Natural Science Foundation of Shandong Province, China (Grant No. ZR2023ME202).
Data Availability Statement
Data are contained within the article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- van Loosdrecht, M.C.; Brdjanovic, D. Anticipating the next century of wastewater treatment. Science 2014, 344, 1452–1453. [Google Scholar] [CrossRef]
- Winkler, M.K.H.; van Loosdrecht, M.C.M. Intensifying existing urban wastewater. Science 2022, 375, 377–378. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Xiong, W.; Xiao, G.; Su, H. Aerobic granular sludge: Formation mechanism, accelerating granulation strategies, and emerging applications. J. Environ. Manage. 2025, 393, 126980. [Google Scholar] [CrossRef] [PubMed]
- Hamza, R.; Rabii, A.; Ezzahraoui, F.-z.; Morgan, G.; Iorhemen, O.T. A review of the state of development of aerobic granular sludge technology over the last 20 years: Full-scale applications and resource recovery. Case Studies in Chemical and Environmental Engineering 2022, 5, 100173. [Google Scholar] [CrossRef]
- Shi, C.; Zeng, R.-G.; Hao, L.-T.; Hao, X.-D.; Li, J. Extracting compositional blocks of alginate-like extracellular polymers (ALE) from conventional activated sludge (CAS). Sci. Total Environ. 2023, 867, 161371. [Google Scholar] [CrossRef]
- Campo, R.; Sguanci, S.; Caffaz, S.; Mazzoli, L.; Ramazzotti, M.; Lubello, C.; Lotti, T. Efficient carbon, nitrogen and phosphorus removal from low C/N real domestic wastewater with aerobic granular sludge. Bioresour. Technol. 2020, 305, 122961. [Google Scholar] [CrossRef]
- Wu, T.; Yang, S.-S.; Zhong, L.; Pang, J.-W.; Zhang, L.; Xia, X.-F.; Yang, F.; Xie, G.-J.; Liu, B.-F.; Ren, N.-Q.; et al. Simultaneous nitrification, denitrification and phosphorus removal: What have we done so far and how do we need to do in the future? Sci. Total Environ. 2023, 856, 158977. [Google Scholar] [CrossRef]
- Amorim de Carvalho, C.d.; Ferreira dos Santos, A.; Tavares Ferreira, T.J.; Sousa Aguiar Lira, V.N.; Mendes Barros, A.R.; Bezerra dos Santos, A. Resource recovery in aerobic granular sludge systems: is it feasible or still a long way to go? Chemosphere 2021, 274, 129881. [Google Scholar] [CrossRef]
- Wei, Z.; Qin, Y.; Li, X.; Gao, P. Resource recovery of high value-added products from wastewater: Current status and prospects. Bioresour. Technol. 2024, 398, 130521. [Google Scholar] [CrossRef]
- Tavares Ferreira, T.J.; Luiz de Sousa Rollemberg, S.; Nascimento de Barros, A.; Machado de Lima, J.P.; Bezerra Dos Santos, A. Integrated review of resource recovery on aerobic granular sludge systems: Possibilities and challenges for the application of the biorefinery concept. J. Environ. Manage. 2021, 291, 112718. [Google Scholar] [CrossRef]
- De Bruin, S.; Riisgaard-Jensen, M.; Hansen, S.H.; Van Loosdrecht, M.C.M.; Nielsen, P.H.; Lin, Y. Global insights into extracellular polymeric substances from activated sludge: Yield, composition, and microbial communities. Water Res. 2026, 289, 124726. [Google Scholar] [CrossRef]
- Liu, S.; Zhou, M.; Daigger, G.T.; Huang, J.; Song, G. Granule formation mechanism, key influencing factors, and resource recycling in aerobic granular sludge (AGS) wastewater treatment: A review. J. Environ. Manage. 2023, 338, 117771. [Google Scholar] [CrossRef]
- van Leeuwen, K.; de Vries, E.; Koop, S.; Roest, K. The Energy & Raw Materials Factory: Role and Potential Contribution to the Circular Economy of the Netherlands. Environmental Management 2018, 61, 786–795. [Google Scholar] [CrossRef]
- Ali, M.; Hong, P.-Y.; Mishra, H.; Vrouwenvelder, J.; Saikaly, P.E. Adopting the circular model: opportunities and challenges of transforming wastewater treatment plants into resource recovery factories in Saudi Arabia. Water Reuse 2022, 12, 346–365. [Google Scholar] [CrossRef]
- Cheng, M.; Shi, C.; Zhao, B.-H.; Zhang, N.; Shen, Q.-Y.; Hao, L.-T.; Wang, X.-Y. Recycling alginate-like extracellular polymers (ALE) from municipal sludge: Value-added products and external impact. Chem. Eng. J. 2024, 493, 152593. [Google Scholar] [CrossRef]
- Chen, X.; Lee, Y.-J.; Yuan, T.; Lei, Z.; Adachi, Y.; Zhang, Z.; Lin, Y.; van Loosdrecht, M.C.M. A review on recovery of extracellular biopolymers from flocculent and granular activated sludges: Cognition, key influencing factors, applications, and challenges. Bioresour. Technol. 2022, 363, 127854. [Google Scholar] [CrossRef] [PubMed]
- Hay, I.D.; Rehman, Z.U.; Ghafoor, A.; Rehm, B.H.A. Bacterial biosynthesis of alginates. J. Chem. Technol. Biotechnol. 2010, 85, 752–759. [Google Scholar] [CrossRef]
- Remminghorst, U.; Rehm, B.H.A. Bacterial alginates: from biosynthesis to applications. Biotechnol. Lett. 2006, 28, 1701–1712. [Google Scholar] [CrossRef]
- Xiao, R.; Zheng, Y. Overview of microalgal extracellular polymeric substances (EPS) and their applications. Biotechnol. Adv. 2016, 34, 1225–1244. [Google Scholar] [CrossRef] [PubMed]
- Sam, S.B.; Dulekgurgen, E. Characterization of exopolysaccharides from floccular and aerobic granular activated sludge as alginate-like-exoPS. Desalin. Water Treat. 2016, 57, 2534–2545. [Google Scholar] [CrossRef]
- Seviour, T.; Yuan, Z.; van Loosdrecht, M.C.M.; Lin, Y. Aerobic sludge granulation: A tale of two polysaccharides? Water Res. 2012, 46, 4803–4813. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.M.; Wang, L.; Chi, Z.M.; Liu, X.Y. Bacterial alginate role in aerobic granular bio-particles formation and settleability improvement. Sep. Sci. Technol. 2008, 43, 1642–1652. [Google Scholar] [CrossRef]
- Lin, Y.; de Kreuk, M.; van Loosdrecht, M.C.M.; Adin, A. Characterization of alginate-like exopolysaccharides isolated from aerobic granular sludge in pilot-plant. Water Res. 2010, 44, 3355–3364. [Google Scholar] [CrossRef]
- Sarvajith, M.; Nancharaiah, Y.V. Properties of alginate-like exopolymers recovered from flocculent and granular microbial sludges of different biological treatment systems treating real municipal wastewater. Sep. Purif. Technol. 2023, 313, 123460. [Google Scholar] [CrossRef]
- Rehman, Z.U.; Ghaani, M.; Mohamed, A.Y.A.; Gallagher, J.; Saikaly, P.E.; Ali, M. Alginate-like exopolysaccharides extracted from different waste sludges exhibit varying physicochemical and material properties. Front. Microbiol. 2024, 15. [Google Scholar] [CrossRef]
- Schambeck, C.M.; Magnus, B.S.; de Souza, L.C.R.; Leite, W.R.M.; Derlon, N.; Guimarães, L.B.; da Costa, R.H.R. Biopolymers recovery: dynamics and characterization of alginate-like exopolymers in an aerobic granular sludge system treating municipal wastewater without sludge inoculum. J. Environ. Manage. 2020, 263, 110394. [Google Scholar] [CrossRef]
- Li, J.; Hao, X.; Gan, W.; van Loosdrecht, M.C.M.; Wu, Y. Recovery of extracellular biopolymers from conventional activated sludge: Potential, characteristics and limitation. Water Res. 2021, 205, 117706. [Google Scholar] [CrossRef]
- Li, J.; Hao, X.; Gan, W.; C. M. van Loosdrecht, M.; Wu, Y. Controlling factors and involved mechanisms on forming alginate like extracellular polymers in flocculent sludge. Chem. Eng. J. 2022, 439, 135792. [Google Scholar] [CrossRef]
- Cydzik-Kwiatkowska, A.; Ciesielski, S.; Florczyk, M.; Pasieczna-Patkowska, S.; Komorowska-Kaufman, M.; Pomian, W.; Jóźwiak, K.; Oleskowicz-Popiel, P. Biopolymer Production in a Full-Scale Activated Sludge Wastewater Treatment Plant: Seasonal Changes and Promising Bacterial Producers. Energies 2024, 17, 6231. [Google Scholar] [CrossRef]
- Lin, Y.M.; Sharma, P.K.; van Loosdrecht, M.C.M. The chemical and mechanical differences between alginate-like exopolysaccharides isolated from aerobic flocculent sludge and aerobic granular sludge. Water Res. 2013, 47, 57–65. [Google Scholar] [CrossRef]
- Schambeck, C.M.; Girbal-Neuhauser, E.; Böni, L.; Fischer, P.; Bessière, Y.; Paul, E.; da Costa, R.H.R.; Derlon, N. Chemical and physical properties of alginate-like exopolymers of aerobic granules and flocs produced from different wastewaters. Bioresour. Technol. 2020, 312, 123632. [Google Scholar] [CrossRef]
- Zahra, S.A.; Abdullah, N.; Iwamoto, K.; Yuzir, A.; Mohamad, S.E. Alginate-like exopolysaccharides in aerobic granular sludge: A review. Mater. Today:. Proc. 2022, 65, 3046–3053. [Google Scholar] [CrossRef]
- Rollemberg, S.L.D.; dos Santos, A.F.; Ferreira, T.J.T.; Firmino, P.I.M.; dos Santos, A.B. Evaluation of the production of alginate-like exopolysaccharides (ALE) and tryptophan in aerobic granular sludge systems. Bioprocess. Biosyst. Eng. 2021, 44, 259–270. [Google Scholar] [CrossRef]
- Amancio Frutuoso, F.K.; P.S.G. da Silva, V.E.; C.V. Silva, T.F.; P. Vilar, V.J.; Bezerra dos Santos, A. Solids retention time (SRT) control in the co-treatment of leachate with domestic sewage in aerobic granular sludge systems: Impacts on system performance, operational stability, and bioresource production. Bioresour. Technol. 2025, 415, 131664. [Google Scholar] [CrossRef] [PubMed]
- Ferreira dos Santos, A.; Amancio Frutuoso, F.K.; de Amorim de Carvalho, C.; Sousa Aguiar Lira, V.N.; Mendes Barros, A.R.; Bezerra dos Santos, A. Carbon source affects the resource recovery in aerobic granular sludge systems treating wastewater. Bioresour. Technol. 2022, 357, 127355. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, R.; Du, Y.; Huang, S.; Zhao, F.; Kim, D.-H.; Ng, H.Y.; Shi, X.; Xu, B. From waste to wealth: Exploring the effect of particle size on biopolymer harvesting from aerobic granular sludge. Bioresour. Technol. 2025, 418, 131977. [Google Scholar] [CrossRef] [PubMed]
- Pereira, E.; Campos, H.; Sales, M.; Marinho, T.; Motteran, F.; Magnus, B.S.; Kato, M.T.; Florencio, L. Influence of mixing regimes on microbial community structure and recovery of alginate-like exopolymers and lipids from bacterial and algal-bacterial aerobic granular sludges. J. Water Process Eng. 2025, 79, 108926. [Google Scholar] [CrossRef]
- Svierzoski, N.D.S.; Castellano-Hinojosa, A.; Gallardo-Altamirano, M.J.; Mahler, C.F.; Bassin, J.P.; González-Martínez, A. Evaluating the effects of aeration intermittency on treatment performance and the granule microbiome in continuous-flow aerobic granular sludge systems. J. Environ. Chem. Eng. 2025, 13, 117660. [Google Scholar] [CrossRef]
- Frutuoso, F.K.A.; de Barros, A.N.; dos Santos, A.F.; Barros, A.R.M.; Rollemberg, S.L.D.; dos Santos, A.B. Impact of operating cycle type on alginate-like exopolysaccharide and tryptophan production in aerobic granular sludge systems. Revista Brasileira De Ciencias Ambientais 2024, 59. [Google Scholar] [CrossRef]
- Li, X.; Luo, J.; Guo, G.; Mackey, H.R.; Hao, T.; Chen, G. Seawater-based wastewater accelerates development of aerobic granular sludge: A laboratory proof-of-concept. Water Res. 2017, 115, 210–219. [Google Scholar] [CrossRef]
- Amancio Frutuoso, F.K.; Ferreira dos Santos, A.; da Silva França, L.L.; Mendes Barros, A.R.; Bezerra dos Santos, A. Influence of salt addition to stimulating biopolymers production in aerobic granular sludge systems. Chemosphere 2023, 311, 137006. [Google Scholar] [CrossRef]
- Amancio Frutuoso, F.K.; Ferreira dos Santos, A.; da Silva França, L.L.; Mendes Barros, A.R.; Bezerra dos Santos, A. Influence of operating regime on resource recovery in aerobic granulation systems under osmotic stress. Bioresour. Technol. 2023, 376, 128850. [Google Scholar] [CrossRef]
- Amancio Frutuoso, F.K.; Bandeira de Carvalho, C.; Porfiro Sales Gonçalves da Silva, V.E.; Bezerra dos Santos, A. Aerobic granulation and bioresource production under intermittent saline stress. J. Environ. Chem. Eng. 2024, 12, 112948. [Google Scholar] [CrossRef]
- Jachimowicz, P.; Jo, Y.-J.; Cydzik-Kwiatkowska, A. Polyethylene microplastics increase extracellular polymeric substances production in aerobic granular sludge. Sci. Total Environ. 2022, 851, 158208. [Google Scholar] [CrossRef]
- Felz, S.; Al-Zuhairy, S.; Aarstad, O.A.; van Loosdrecht, M.C.M.; Lin, Y.M. Extraction of Structural Extracellular Polymeric Substances from Aerobic Granular Sludge. JoVE 2016, e54534. [Google Scholar]
- Yang, Y.; Zhou, H.; Liu, W.; Wang, K.; Yu, T. Effect of extraction temperature on value-added biopolymer recovery in waste activated sludge. Water Cycle 2025, 6, 272–277. [Google Scholar] [CrossRef]
- Liu, X.; Ren, W.; Zhai, Y.; Xie, Y.; Liang, F.; Xu, Z. Enhanced Recovery of Alginate-like Extracellular Polymers (ALE) from Waste-Activated Sludge Using Sodium Percarbonate: Performance and Characteristics. Sustainability 2023, 15, 14573. [Google Scholar] [CrossRef]
- Li, J.; Hao, X.; Gan, W.; van Loosdrecht, M.C.M.; Wu, Y. Enhancing extraction of alginate like extracellular polymers (ALE) from flocculent sludge by surfactants. Sci. Total Environ. 2022, 837, 155673. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.; Qian, Y.; Zhao, B.; Zhang, N.; Sun, L.; Zhao, L.; Liu, R.; Hao, X.; Shi, C. Mass-balance analysis of sulfamethoxazole during extracellular-polymeric-substance extraction from excess sludge. J. Water Process Eng. 2025, 75, 107859. [Google Scholar] [CrossRef]
- Kedves, A.; Kedves, O.; Mikó, E.; Kónya, Z. Micro- and nanoplastics in granular sludge systems: mechanisms of disruption, retention, and microbial adaptation in wastewater treatment technologies. Water Res. X 2025, 28. [Google Scholar] [CrossRef]
- Zhang, S.; Ding, J.; Feng, Y.; Zhang, L.; Li, S.; Maddela, N.R.; Meng, F. Alginate-like extracellular polymers recovery from industrial activated sludge: characteristics and application potential. J. Water Process Eng. 2025, 78, 108759. [Google Scholar] [CrossRef]
- Cydzik-Kwiatkowska, A.; Gusiatin, M.Z.; Zielińska, M.; Wojnowska-Baryła, I.; Kulikowska, D.; Bernat, K. Alginate-like polymers from full-scale aerobic granular sludge: content, recovery, characterization, and application for cadmium adsorption. Sci. Rep. 2022, 12, 22260. [Google Scholar] [CrossRef]
- Dall’ Agnol, P.; Libardi, N.; Muller, J.M.; Xavier, J.A.; Domingos, D.G.; da Costa, R.H.R. A comparative study of phosphorus removal using biopolymer from aerobic granular sludge: A factorial experimental evaluation. J. Environ. Chem. Eng. 2020, 8, 103541. [Google Scholar] [CrossRef]
- Moraes Schambeck, C.; Ribeiro da Costa, R.H.; Derlon, N. Phosphate removal from municipal wastewater by alginate-like exopolymers hydrogels recovered from aerobic granular sludge. Bioresour. Technol. 2021, 333, 125167. [Google Scholar] [CrossRef] [PubMed]
- Dall'Agnol, P.; Libardi, N.; da Silva, E.C.; da Costa, R.H.R. Biosorption of Phosphorus Using Alginate-Like Exopolymers: Investigation of Removal Mechanism, Kinetic and Thermodynamic Properties. J. Polym. Environ. 2022, 30, 695–706. [Google Scholar] [CrossRef]
- Cabral, A.; Gommersbach, C.; Cavali, M.; Libardi, N.; da Costa, R.H.R. Biopolymers recovery from biological sludge and its use as phosphorus biosorbent. Engenharia Sanitaria E Ambiental 2024, 29, 1–8. [Google Scholar] [CrossRef]
- Ladnorg, S.; Junior, N.L.; Dall’ Agnol, P.; Domingos, D.G.; Magnus, B.S.; Wichern, M.; Gehring, T.; da Costa, R.H.R. Alginate-like exopolysaccharide extracted from aerobic granular sludge as biosorbent for methylene blue: Thermodynamic, kinetic and isotherm studies. J. Environ. Chem. Eng. 2019, 7, 103081. [Google Scholar] [CrossRef]
- Pagliaccia, B.; Campo, R.; Carretti, E.; Severi, M.; Lubello, C.; Lotti, T. Towards resource recovery-oriented solutions in agriculture exploiting structural extracellular polymeric substances (sEPS) extracted from aerobic granular sludge (AGS). Chem. Eng. J. 2024, 485, 149819. [Google Scholar] [CrossRef]
- Lin, Y.M.; Nierop, K.G.J.; Girbal-Neuhauser, E.; Adriaanse, M.; van Loosdrecht, M.C.M. Sustainable polysaccharide-based biomaterial recovered from waste aerobic granular sludge as a surface coating material. Sustainable Mater.Technol 2015, 4, 24–29. [Google Scholar] [CrossRef]
- Feng, C.; Bonetti, L.; Lu, H.; Zhou, Z.; Lotti, T.; Jia, M.; Rizzardi, G.; De Nardo, L.; Malpei, F. Extracellular polymeric substances as paper coating biomaterials derived from anaerobic granular sludge. Environmental Science and Ecotechnology 2024, 21, 100397. [Google Scholar] [CrossRef]
- Anderson, A.J.; Dawes, E.A. Occurrence, metabolism, metabolic role, and industrial uses of bacterial polyhydroxyalkanoates. Microbiological Reviews 1990, 54, 450–472. [Google Scholar] [CrossRef]
- Możejko-Ciesielska, J.; Kiewisz, R. Bacterial polyhydroxyalkanoates: Still fabulous? Microbiol. Res. 2016, 192, 271–282. [Google Scholar] [CrossRef]
- Kumar, M.; Rathour, R.; Singh, R.; Sun, Y.; Pandey, A.; Gnansounou, E.; Andrew Lin, K.-Y.; Tsang, D.C.W.; Thakur, I.S. Bacterial polyhydroxyalkanoates: Opportunities, challenges, and prospects. J. Cleaner Prod. 2020, 263, 121500. [Google Scholar] [CrossRef]
- Yadav, B.; Talan, A.; Tyagi, R.D.; Drogui, P. Concomitant production of value-added products with polyhydroxyalkanoate (PHA) synthesis: A review. Bioresour. Technol. 2021, 337, 125419. [Google Scholar] [CrossRef]
- Werker, A.; Bengtsson, S.; Korving, L.; Hjort, M.; Anterrieu, S.; Alexandersson, T.; Johansson, P.; Karlsson, A.; Karabegovic, L.; Magnusson, P.; et al. Consistent production of high quality PHA using activated sludge harvested from full scale municipal wastewater treatment – PHARIO. Water Sci. Technol. 2018, 78, 2256–2269. [Google Scholar] [CrossRef]
- de Kreuk, M.K.; van Loosdrecht, M.C.M. Selection of slow growing organisms as a means for improving aerobic granular sludge stability. Water Sci. Technol. 2004, 49, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wang, J.; Wang, Q.; Li, Z.; Yuan, T.; Lei, Z.; Zhang, Z.; Shimizu, K.; Lee, D.-J. A comparative study on simultaneous recovery of phosphorus and alginate-like exopolymers from bacterial and algal-bacterial aerobic granular sludges: Effects of organic loading rate. Bioresour. Technol. 2022, 357, 127343. [Google Scholar] [CrossRef]
- Yang, Y.; Peng, Y.; Cheng, J.; Zhang, S.; Liu, C.; Zhang, L. A novel two-stage aerobic granular sludge system for simultaneous nutrient removal from municipal wastewater with low C/N ratios. Chem. Eng. J. 2023, 462, 142318. [Google Scholar] [CrossRef]
- Ma, J.W.; Chen, L.; Ji, Y.N.; Sun, H.; Han, Y.; Zhu, L.; Bi, P.; He, Q.L. Integration of partial nitrification, endogenous denitrification and anaerobic ammonia oxidation in low dissolved oxygen anaerobic/oxic/ anoxic-aerobic granular sludge reactor treating low carbon to nitrogen ratios wastewater. Bioresour. Technol. 2025, 435, 132923. [Google Scholar] [CrossRef] [PubMed]
- Invernizzi, G.B.; Orozco, A.M.F.; Zaritzky, N.E.; Caravelli, A.H. Stable aerobic granulation for the selection of a mixed culture producing polyhydroxyalkanoates at high phenol loads. J. Chem. Technol. Biotechnol. 2025, 100, 2337–2356. [Google Scholar] [CrossRef]
- Karakas, I.; Sam, S.B.; Cetin, E.; Dulekgurgen, E.; Yilmaz, G. Resource recovery from an aerobic granular sludge process treating domestic wastewater. J. Water Process Eng. 2020, 34, 101148. [Google Scholar] [CrossRef]
- Rojas-Zamora, U.; Fajardo-Ortiz, M.d.C.; Cuetero-Martínez, Y.; Tavera-Mejía, W.; Salazar-Peláez, M.L. Aerobic granulation for polyhydroxyalkanoates accumulation using organic waste leachates. J. Water Process Eng. 2023, 51, 103464. [Google Scholar] [CrossRef]
- Traina, F.; Corsino, S.F.; Torregrossa, M.; Viviani, G. Biopolymer Recovery from Aerobic Granular Sludge and Conventional Flocculent Sludge in Treating Industrial Wastewater: Preliminary Analysis of Different Carbon Routes for Organic Carbon Utilization. Water 2023, 15, 47. [Google Scholar] [CrossRef]
- Wang, L.; Cui, Y.-W. Simultaneous treatment of epichlorohydrin wastewater and polyhydroxyalkanoate recovery by halophilic aerobic granular sludge highly enriched by Halomonas sp. Bioresour. Technol. 2024, 391, 129951. [Google Scholar] [CrossRef]
- Egle, L.; Rechberger, H.; Krampe, J.; Zessner, M. Phosphorus recovery from municipal wastewater: An integrated comparative technological, environmental and economic assessment of P recovery technologies. Sci. Total Environ. 2016, 571, 522–542. [Google Scholar] [CrossRef] [PubMed]
- Batstone, D.J.; Hülsen, T.; Mehta, C.M.; Keller, J. Platforms for energy and nutrient recovery from domestic wastewater: A review. CHEMOSPHERE 2015, 140, 2–11. [Google Scholar] [CrossRef]
- Wang, J.; Li, Z.; Wang, Q.; Lei, Z.; Yuan, T.; Shimizu, K.; Zhang, Z.; Adachi, Y.; Lee, D.-J.; Chen, R. Achieving stably enhanced biological phosphorus removal from aerobic granular sludge system via phosphorus rich liquid extraction during anaerobic period. Bioresour. Technol. 2022, 346, 126439. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Huang, W.; Li, H.; Lei, Z.; Zhang, Z.; Tay, J.H.; Lee, D.-J. Species and distribution of inorganic and organic phosphorus in enhanced phosphorus removal aerobic granular sludge. Bioresour. Technol. 2015, 193, 549–552. [Google Scholar] [CrossRef]
- Lan, Y.; Li, X.; Du, R.; Fan, X.; Cao, S.; Peng, Y. Hydroxyapatite (HAP) formation in acetate-driven partial denitrification process: Enhancing sludge granulation and phosphorus removal. Sci. Total Environ. 2023, 903, 166659. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Cai, W.; Huang, H.; Lei, Z.; Zhang, Z.; Tay, J.H.; Lee, D.-J. Identification of inorganic and organic species of phosphorus and its bio-availability in nitrifying aerobic granular sludge. Water Res. 2015, 68, 423–431. [Google Scholar] [CrossRef]
- Mañas, A.; Biscans, B.; Spérandio, M. Biologically induced phosphorus precipitation in aerobic granular sludge process. Water Res. 2011, 45, 3776–3786. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-Q.; Cinquepalmi, S. Exploration of mechanisms for calcium phosphate precipitation and accumulation in nitrifying granules by investigating the size effects of granules. Water Res. 2021, 206, 117753. [Google Scholar] [CrossRef]
- Li, Y.; Zou, J.; Zhang, L.; Sun, J. Aerobic granular sludge for simultaneous accumulation of mineral phosphorus and removal of nitrogen via nitrite in wastewater. Bioresour. Technol. 2014, 154, 178–184. [Google Scholar] [CrossRef]
- Lin, Y.M.; Bassin, J.P.; van Loosdrecht, M.C.M. The contribution of exopolysaccharides induced struvites accumulation to ammonium adsorption in aerobic granular sludge. Water Res. 2012, 46, 986–992. [Google Scholar] [CrossRef]
- Bi, P.; Yan, X.; Song, Y.; Miao, H.; Sun, H.; Han, Y.; Chen, L.; Yan, K.; Li, M.; Xiong, F.; et al. Microbially induced phosphate precipitation disentangles sludge retention time trade-off enabling efficient phosphorus removal with minimal sludge discharge in aerobic granular sludge. Chem. Eng. J. 2025, 522, 167305. [Google Scholar] [CrossRef]
- Li, W.-W.; Zhang, H.-L.; Sheng, G.-P.; Yu, H.-Q. Roles of extracellular polymeric substances in enhanced biological phosphorus removal process. Water Res. 2015, 86, 85–95. [Google Scholar] [CrossRef]
- Chen, X.; Wang, J.; Wang, Q.; Yuan, T.; Lei, Z.; Zhang, Z.; Shimizu, K.; Lee, D.-J. Simultaneous recovery of phosphorus and alginate-like exopolysaccharides from two types of aerobic granular sludge. Bioresour. Technol. 2022, 346, 126411. [Google Scholar] [CrossRef] [PubMed]
- Bahgat, N.T.; Wilfert, P.; Eustace, S.J.; Korving, L.; van Loosdrecht, M.C.M. Phosphorous speciation in EPS extracted from Aerobic Granular Sludge. Water Res. 2024, 262, 122077. [Google Scholar] [CrossRef]
- He, Q.; Yan, X.; Xie, Z.; Xu, P.; Fu, Z.; Li, J.; Liu, L.; Bi, P.; Xu, B.; Ma, J. Advanced low-strength wastewater treatment, side-stream phosphorus recovery, and in situ sludge reduction with aerobic granular sludge. Bioresour. Technol. 2023, 386, 129574. [Google Scholar] [CrossRef]
- Lu, Y.Z.; Wang, H.F.; Kotsopoulos, T.A.; Zeng, R.J. Advanced phosphorus recovery using a novel SBR system with granular sludge in simultaneous nitrification, denitrification and phosphorus removal process. Appl. Microbiol. Biotechnol. 2016, 100, 4367–4374. [Google Scholar] [CrossRef] [PubMed]
- Cydzik-Kwiatkowska, A.; Nosek, D. Biological release of phosphorus is more efficient from activated than from aerobic granular sludge. Sci. Rep. 2020, 10. [Google Scholar] [CrossRef]
- Zou, J.; Li, Y. Anaerobic fermentation combined with low-temperature thermal pretreatment for phosphorus-accumulating granular sludge: Release of carbon source and phosphorus as well as hydrogen production potential. Bioresour. Technol. 2016, 218, 18–26. [Google Scholar] [CrossRef]
- Zou, J.; Pan, J.; He, H.; Wu, S.; Xiao, N.; Ni, Y.; Li, J. Nitrifying aerobic granular sludge fermentation for releases of carbon source and phosphorus: The role of fermentation pH. Bioresour. Technol. 2018, 260, 30–37. [Google Scholar] [CrossRef]
- Dababat, S.; Berzio, S.; Wichern, M.; Lübken, M. Anaerobic digestibility of aerobic granular sludge from continuous flow reactors: the role of granule size distribution. Water Sci. Technol. 2023, 87, 3047–3058. [Google Scholar] [CrossRef]
- Guo, H.; van Lier, J.B.; de Kreuk, M. Digestibility of waste aerobic granular sludge from a full-scale municipal wastewater treatment system. Water Res. 2020, 173, 115617. [Google Scholar] [CrossRef]
- Bernat, K.; Cydzik-Kwiatkowska, A.; Wojnowska-Baryła, I.; Karczewska, M. Physicochemical properties and biogas productivity of aerobic granular sludge and activated sludge. Biochem. Eng. J. 2017, 117, 43–51. [Google Scholar] [CrossRef]
- Val del Río, A.; Morales, N.; Isanta, E.; Mosquera-Corral, A.; Campos, J.L.; Steyer, J.P.; Carrère, H. Thermal pre-treatment of aerobic granular sludge: Impact on anaerobic biodegradability. Water Res. 2011, 45, 6011–6020. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.W.; Liu, Y.; Tay, J.H. Distribution of EPS and cell surface hydrophobicity in aerobic granules. Appl. Microbiol. Biotechnol. 2005, 69, 469–473. [Google Scholar] [CrossRef]
- Chen, S.S.; Yang, D.H.; Dong, B.; Li, N.; Dai, X.H. Sludge age impacted the distribution, occurrence state and structure of organic compounds in activated sludge and affected the anaerobic degradability. Chem. Eng. J. 2020, 384. [Google Scholar] [CrossRef]
- Guo, H.; Felz, S.; Lin, Y.; van Lier, J.B.; de Kreuk, M. Structural extracellular polymeric substances determine the difference in digestibility between waste activated sludge and aerobic granules. Water Res. 2020, 181, 115924. [Google Scholar] [CrossRef] [PubMed]
- Cydzik-Kwiatkowska, A.; Bernat, K.; Zielińska, M.; Gusiatin, M.Z.; Wojnowska-Baryła, I.; Kulikowska, D. Valorization of full-scale waste aerobic granular sludge for biogas production and the characteristics of the digestate. Chemosphere 2022, 303, 135167. [Google Scholar] [CrossRef]
- Kosar, S.; Abdelrahman, A.M.; Teber, O.O.; Ozgun, H.; Ersahin, M.E. Impact of ultrasonication on sludge characteristics: Aerobic granular sludge versus waste activated sludge. Chem. Eng. Sci. 2025, 315, 121813. [Google Scholar] [CrossRef]
- Liu, Y.; Nilsen, P.J.; Maulidiany, N.D. Thermal pretreatment to enhance biogas production of waste aerobic granular sludge with and without calcium phosphate precipitates. Chemosphere 2019, 234, 725–732. [Google Scholar] [CrossRef]
- Cicekalan, B.; Atilgan, M.; Kosar, S.; Guven, H.; Koyuncu, I.; Ersahin, M.E.; Ozturk, I.; Ozgun, H. Enhancing biomethane production and phosphorus recovery from aerobic granular sludge through thermal alkali pretreatment. Biomass Bioenergy 2025, 203, 108305. [Google Scholar] [CrossRef]
- Zhang, F.; Qian, D.K.; Geng, Z.Q.; Dai, K.; Zhang, W.; van Loosdrecht, M.C.M.; Zeng, R.J. Enhanced Methane Recovery from Waste-Activated Sludge by Alginate-Degrading Consortia: The Overlooked Role of Alginate in Extracellular Polymeric Substances. Environ. Sci. Technol. Lett. 2021, 8, 86–91. [Google Scholar] [CrossRef]
- Diniz, B.C.; Wilfert, P.; Sorokin, D.Y.; van Loosdrecht, M.C.M. Anaerobic digestion at high-pH and alkalinity for biomethane production: Insights into methane yield, biomethane purity, and process performance. Bioresour. Technol. 2025, 429, 132505. [Google Scholar] [CrossRef]
- Zheng, M.; Hu, Z.T.; Liu, T.; Sperandio, M.; Volcke, E.I.P.; Wang, Z.Y.; Hao, X.D.; Duan, H.R.; Vlaeminck, S.E.; Xu, K.N.; et al. Pathways to advanced resource recovery from sewage. Nat. Sustainability 2024, 7, 1395–1404. [Google Scholar] [CrossRef]
- Bahgat, N.T.; Wilfert, P.; Korving, L.; van Loosdrecht, M. Integrated resource recovery from aerobic granular sludge plants. Water Res. 2023, 234, 119819. [Google Scholar] [CrossRef]
- Chen, X.; Wang, X.; Wang, Q.; Wang, J.; Lei, Z.; Yuan, T.; Zhang, Z.; Lee, D.-J. Energy and resource recovery from a future aerobic granular sludge wastewater treatment plant and benefit analysis. Chem. Eng. J. 2024, 487, 150558. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, Z.; Deng, Y.; Sato, Y.; Wu, D.; Chen, G. Coupling methane and bioactive polysaccharide recovery from wasted activated sludge: A sustainable strategy for sludge treatment. Water Res. 2023, 233, 119775. [Google Scholar] [CrossRef]
- Chen, X.; Wang, X.; Lei, Z.; Yuan, T.; Shimizu, K.; Zhang, Z.; Lee, D.-J. Preferable and feasible tests on phosphorus release/recovery from anaerobically digested bacterial and algal-bacterial aerobic granular sludges. Chem. Eng. J. 2025, 517, 164461. [Google Scholar] [CrossRef]
- Zhu, L.; Zhou, J.; Lv, M.; Yu, H.; Zhao, H.; Xu, X. Specific component comparison of extracellular polymeric substances (EPS) in flocs and granular sludge using EEM and SDS-PAGE. Chemosphere 2015, 121, 26–32. [Google Scholar] [CrossRef]
- Wang, L.L.; Wu, L.J.; Li, A.J.; Hou, B.L.; Jiang, X.M. Synergy of N-(3-oxohexanoyl)-L-homoserine lactone and tryptophan-like outer extracellular substances in granular sludge dominated by aerobic ammonia-oxidizing bacteria. Appl. Microbiol. Biotechnol. 2018, 102, 10779–10789. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, W.; Zhang, X.; Yu, L.; Giglio, G.L.; Huang, Z.; Peng, Z. Effect of the on/off ratio of anaerobic mixing duration on granulation in an anaerobic oxic treatment process. Sep. Purif. Technol. 2025, 364, 132572. [Google Scholar] [CrossRef]
- Sun, Y.; Sun, B.; Li, J.; Zhang, X. Fe3+ addition for enhancing the formation and stability of aerobic granular sludge to treat low-strength wastewater. J. Water Process Eng. 2025, 71, 107144. [Google Scholar] [CrossRef]
- Jahn, L.; Saracevic, E.; Svardal, K.; Krampe, J. Anaerobic biodegradation and dewaterability of aerobic granular sludge. J. Chem. Technol. Biotechnol. 2019, 94, 2908–2916. [Google Scholar] [CrossRef]
- Luiz de Sousa Rollemberg, S.; Queiroz de Oliveira, L.; Nascimento de Barros, A.; Igor Milen Firmino, P.; Bezerra Dos Santos, A. Pilot-scale aerobic granular sludge in the treatment of municipal wastewater: Optimizations in the start-up, methodology of sludge discharge, and evaluation of resource recovery. Bioresour. Technol. 2020, 311, 123467. [Google Scholar] [CrossRef] [PubMed]
- Porfiro Sales Gonçalves da Silva, V.E.; Amancio Frutuoso, F.K.; da Silva Xavier, R.; Santos Passos, E.; Iorhemen, O.; Bezerra dos Santos, A. New insights into the recovery of microbial gums and other bioresources in aerobic granular systems with and without photogranulation and metal ions. Chem. Eng. J. 2025, 523, 168582. [Google Scholar] [CrossRef]
- Kaur, M.; Guo, B.; Iorhemen, O.T. Potential for the recovery of xanthan from aerobic granular sludge wastewater systems–A review. Resources, Conservation and Recycling 2024, 207, 107688. [Google Scholar] [CrossRef]
- Roncevic, Z.Z.; Bajic, B.Z.; Vucurovic, D.G.; Dodic, S.N.; Grahovac, J.A.; Dodic, J.M. Xanthan production on wastewaters from wine industry. HEMIJSKA INDUSTRIJA 2017, 71, 145–153. [Google Scholar] [CrossRef]
- Rončević, Z.; Grahovac, J.; Dodić, S.; Vučurović, D.; Dodić, J. Utilisation of winery wastewater for xanthan production in stirred tank bioreactor: Bioprocess modelling and optimisation. Food Bioprod. Process. 2019, 117, 113–125. [Google Scholar] [CrossRef]
- Kaur, M.; Vesuwe, R.N.; dos Santos, A.B.; Morgan, K.D.; Iorhemen, O.T. Enhancing xanthan biosynthesis in aerobic granular sludge for resource recovery: The role of organic loading rate, carbon-to-nitrogen ratio, and feeding strategy. Cleaner Water 2025, 4, 100111. [Google Scholar] [CrossRef]
- Porfiro Sales Gonçalves da Silva, V.E.; Fiorani Ezequiel, T.; Iorhemen, O.; Bezerra dos Santos, A. Adaptation and application of methodologies for quantification of xanthan and curdlan in conventional AGS systems and algal-bacterial association. Biomass Bioenergy 2026, 205, 108558. [Google Scholar] [CrossRef]
- Mehta, K.; Shukla, A.; Saraf, M. Articulating the exuberant intricacies of bacterial exopolysaccharides to purge environmental pollutants. HELIYON 2021, 7. [Google Scholar] [CrossRef]
- Adekunle, A.; Ukaigwe, S.; Bezerra dos Santos, A.; Iorhemen, O.T. Potential for curdlan recovery from aerobic granular sludge wastewater treatment systems – A review. Chemosphere 2024, 362, 142504. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.; Shao, Z.Y.; Jiang, D.M.; Gao, H.L.; Yang, X.X. Curdlan production from cassava starch hydrolysates by Agrobacterium sp. DH-2. Bioprocess. Biosyst. Eng. 2022, 45, 969–979. [Google Scholar] [CrossRef]
- Espíndola, S.P.; Pronk, M.; Zlopasa, J.; Picken, S.J.; van Loosdrecht, M.C.M. Nanocellulose recovery from domestic wastewater. J. Cleaner Prod. 2021, 280, 124507. [Google Scholar] [CrossRef]
- Li, C.; Liu, J.; Lou, G.C.; Yu, C.N. The feasibility and applicability of sequential extraction of high value-added biogenic materials from sewage sludge. Water Sci. Technol. 2024, 89, 2812–2822. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).