Submitted:
03 February 2026
Posted:
05 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cone and Seed Collection, and Parental Balance Curve
2.2. Data Analysis
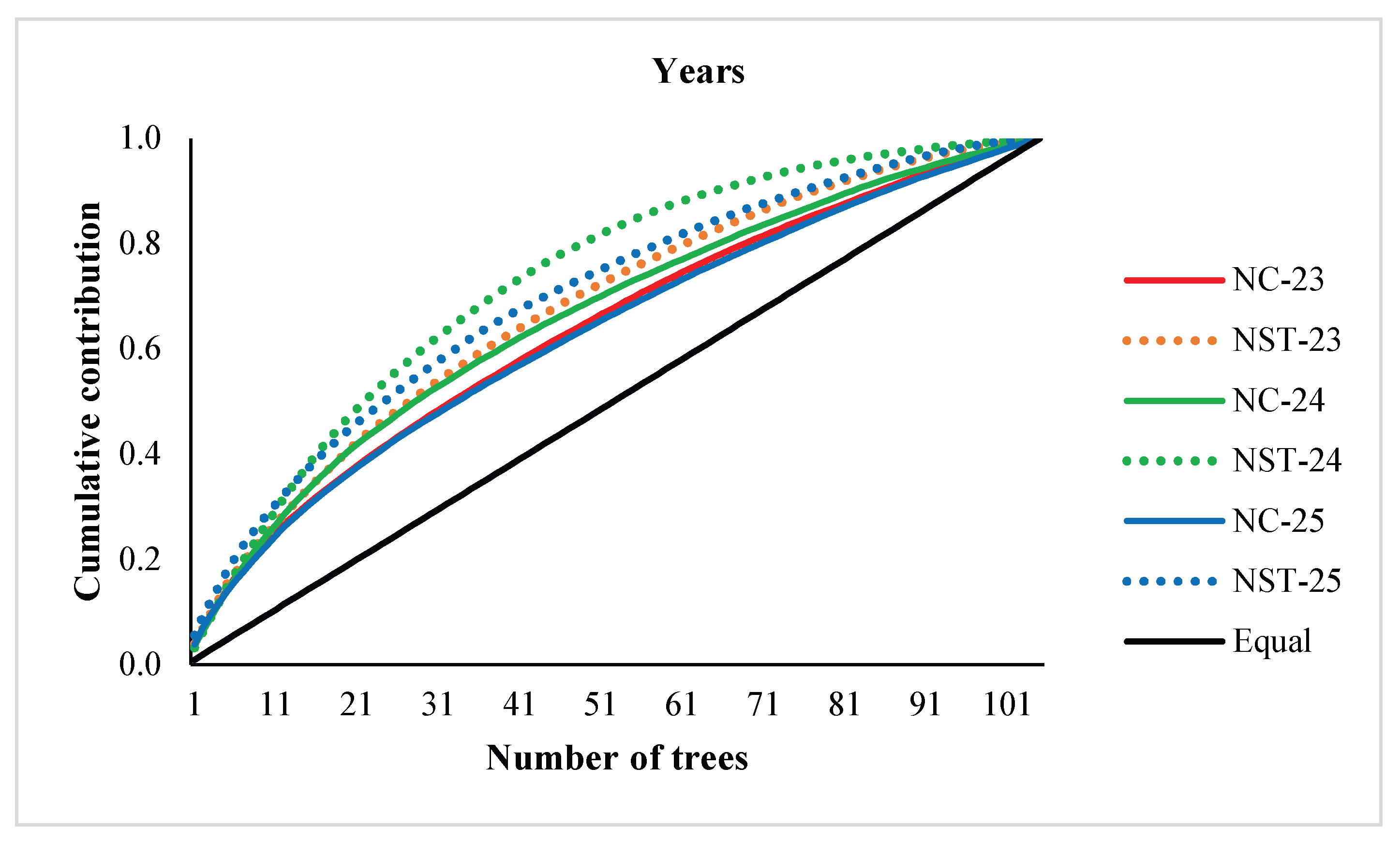
3. Results
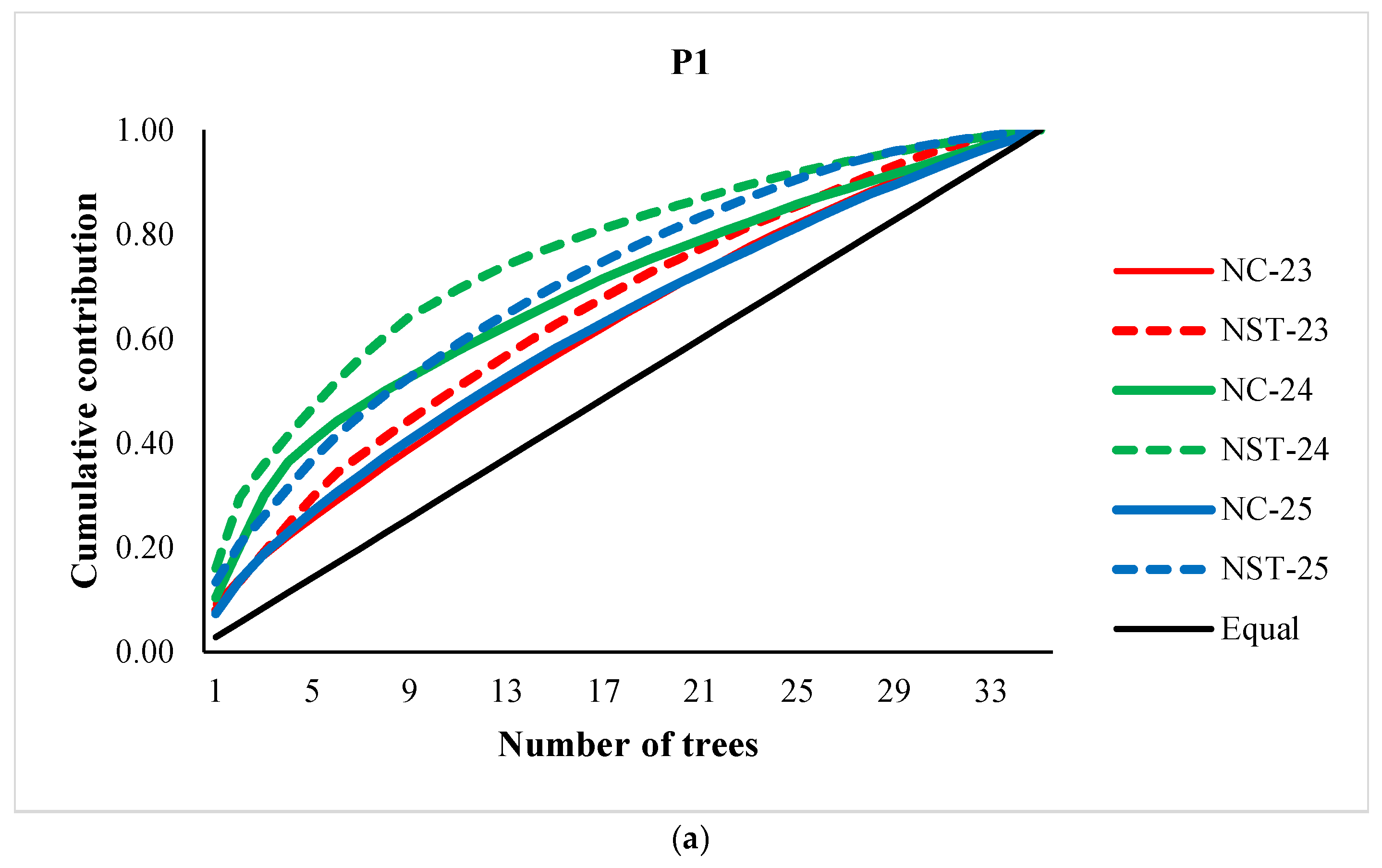
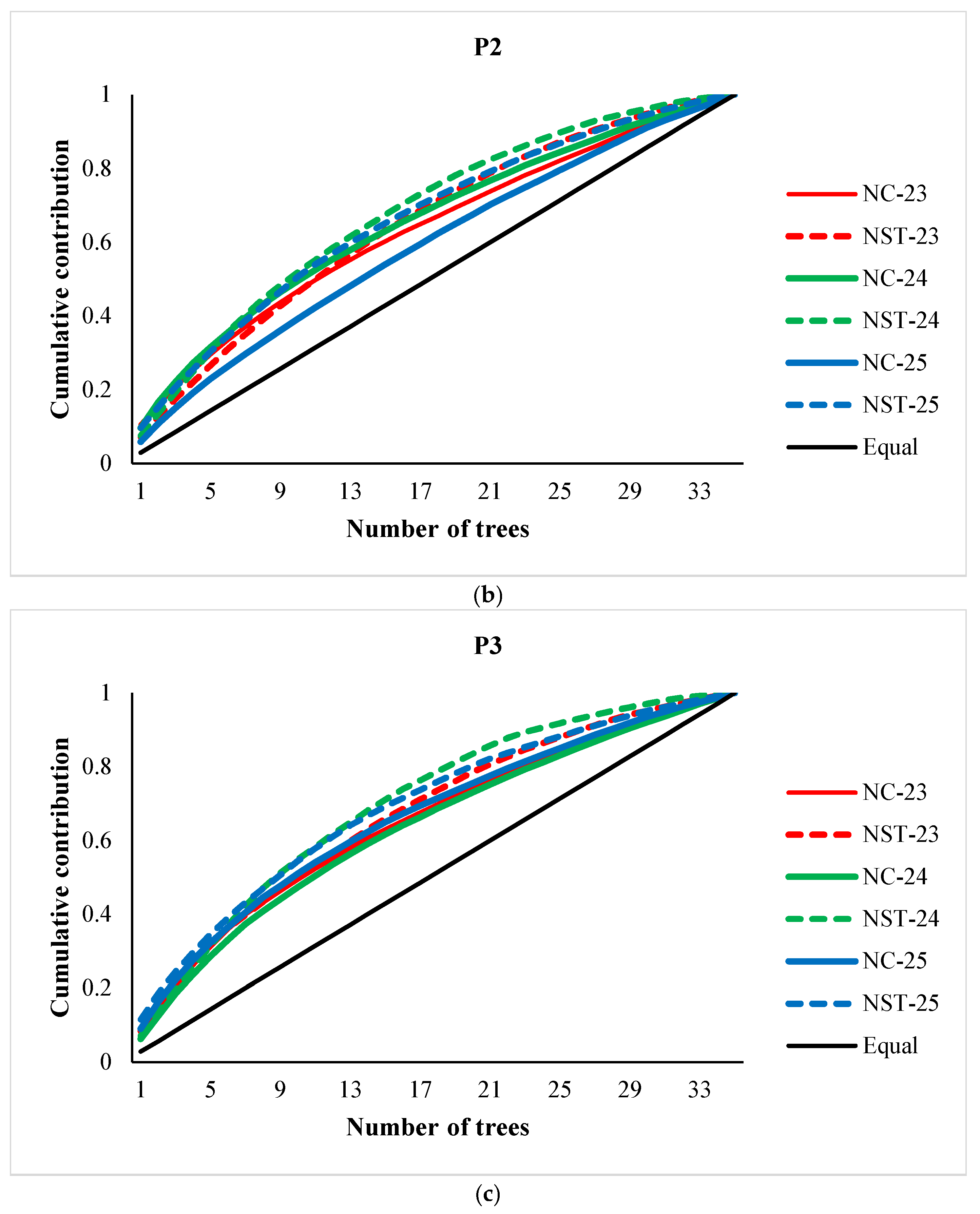
3.1. Cone and Seed Productions, and Parental Balance Curve
3.2. Fertility Variation, Effective Number of Parents and Gene Diversity
4. Discussion
5. Conclusion
Author Contributions
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tsanov, T.S.; Naidenov, Y.; Kalmoukov, K. Le Cedre (Cedrus) en Bulgarie Etat et Perspectives. International Cedar Symposium. Ankara, 1990, pp. 308-324.
- Ottone, R.J.; Carloni, C. Aspects of cedar cultivation in the Argentine Republic. International Cedar Symposium. Ankara, 1990, pp. 260-265.
- Fusaro, E. Characteristics and distribution of cedar in Italy. International Cedar Symposium. Antalya, 1990, pp. 273-285.
- Assadullahi, F.; Hedayati, M.A. Cedrus plantation experiments in Iran. International Cedar Symposium. Ankara, 1990, pp. 266-273.
- Toth, J. Le Cedre Dissemination, Extaction, Qualite, Germination, Conservation. La Foret Privee, 1980, 131.
- Bariteau, M.; Pommery, J.; Vauthier, D. Adaptation of Cedrus provenances tested in France in comparative plantations. Conference of Adaptation and Selection of Mediterranean Pinus and Cedrus for Sustainable Afforestations of Marginal Lands, Thessaloniki, 2000, pp. 15-19.
- Boydak, M. Regeneration of Lebanon cedar (Cedrus libani A. Rich.) on karstic lands in Turkey. Forest Ecology and Management 2003, 178, 231–243. [Google Scholar] [CrossRef]
- Boydak, M.; Çalıkoğlu, M. Biology and silviculture of Cedar of Lebanon (Cedrus libani A. Rich.); Ministry of Forestry Publication: Ankara, 2008. [Google Scholar]
- OGM. Forest inventory of Türkiye. General Directorated of Forestry, 2025, Ankara. https://www.ogm.gov.tr.
- ORTOHUM. Seed production areas. Research Directorate of Forest Tree Seeds and Tree Breeding, 2025, Ankara. https://ortohum.ogm.gov.tr.
- Zobel, B.J.; Talbert, J. Applied forest tree improvement; John Wiley and Scons: NY, 2003. [Google Scholar]
- Odabaşı, T. Research on cone and seed characters of Cedrus libani; General Directorate of Forestry Press: Ankara, 1990. [Google Scholar]
- Boydak, M. Ecology and silviculture of Cedar of Lebanon (Cedrus libani A. Rich.) and conservation of its natural forest; Ministry of Forestry Publication: Ankara, 1996. [Google Scholar]
- Saatçioğlu, F. Silviculture-I. Forestry Faculty of Istanbul University press: Istanbul, 1976. https://cerrahpasa.iuc.edu.tr/.
- Dirik, H. Effect of osmotic water stress on germination of Lebanon Cedar (Cedrus libani A. Rich.) seeds from the provenances of Turkey. Annals of Forest Science 2000, 57, 361–367. [Google Scholar] [CrossRef]
- Ayrancı, A.; Öner, M.N. Effect of some pretreatments of seeds on germination of different Taurus cedar (Cedrus libani A. Rich.) origins. Anatolian Journal of Forest Research 2019, 5, 61–70. [Google Scholar]
- Bila, AD. Fertility variation and its effects on gene diversity in forest tree populations. Ph.D. Thesis. Swedish University of Agricultural Science, Acta Universitatis Agriculturae Sueciae, Silvestria 166, Umeå, Sweden, 2000. https://www.upsc.se.
- Griffin, A.R. Clonal variation in Radiata pine seed orchards. I. Some flowering, cone and seed production traits. Aust. For. Res. 1982, 12, 295–302. http://jkv.50megs.com/afr.html.
- Roeder, K.; Devlin, B.; Lindsay, B.G. Application of maximum likelihood methods to population genetic data for the estimation of individual fertilities. Biometrics 1989, 45, 363–379. [Google Scholar] [CrossRef]
- El-Kassaby, Y.A.; Reynolds, S. Reproductive phenology, parental balance and supplemental mass pollination in a Sitka spruce seed orchard. For. Ecol. Manag. 1990, 31, 45–54. [Google Scholar] [CrossRef]
- Xie, C.Y.; Knowles, P. Male fertility variation in an open-pollinated plantation of Norway spruce (Picea abies). Canadian Journal of Forest Research 1992, 22, 1463–1468. [Google Scholar] [CrossRef]
- El-Kassaby, Y.A.; Cook, C. Female reproductive energy and reproductive success in a Douglas-fir seed orchard and its impact on genetic diversity. Silvae Genetica 1994, 43, 243–246. [Google Scholar]
- El-Kassaby, Y. Evaluation of the tree-improvement delivery system: factors affecting genetic potential. Tree Physiology 1995, 15, 545–550. [Google Scholar] [CrossRef]
- Shea, K. L. Effects of population structure and cone production on out crossing rates in Engelmann spruce and Subalpine fir. Evolution 1987, 41, 124–136. [Google Scholar] [CrossRef]
- Kang, KS.; Bila, A.D.; Harju, A.M.; Lindgren, D. Fertility variation in forest tree populations. Forestry 2003, 76, 329–344. [Google Scholar] [CrossRef]
- Kamalakannan, R.; Varghese, M.; Park, J.M.; Kwon, S.H.; Song, J.H.; Kang, K.S. Fertility variation and its impact on effective population size in seed stands of Tamarindus indica and Azadirachta indica. Silvae Genetica 2015, 64, 91–99. [Google Scholar] [CrossRef]
- Kang, K.S.; Bilir, N.; Jeon, K.; Kim, Y.-J. Fertility variation and gene diversity in forest populations. Forests 2023, 14, 2172. [Google Scholar] [CrossRef]
- Bilir, N.; Novikova, T.P.; Novikov, A.I. Variation in cone and seed productions of Taurus cedar (Cedrus libani A. Rich.) Populations. Geography, Environment, Sustainability 2025, 18, 63–69. [Google Scholar] [CrossRef]
- Ganatsas, P.; Tsakaldimi, M.; Thanos, C. Seed and cone diversity and seed germination of Pinus pinea in Strofylia Site of the Natura 2000 Network. Biodiversity and Conservation 2008, 17(10), 2427–2439. [Google Scholar] [CrossRef]
- ISTA. International Seed Testing Association; The International Seed Testing Association (ISTA): Bassersdorf, 2005, Switzerland.
- Chaisurisri, K.; El-Kassaby, YA. Estimation of clonal contribution to cone and seed crops in a Stika spruce seed orchard. Ann. Sci. For. 1993, 50, 461–467. [Google Scholar] [CrossRef]
- SAS Inst. Inc., 1988. SAS/STAT User’s Guide, Release 6.03, edition, Cary, NC. https://www.sas.com.
- Kang, K.S.; Lindgren, D. Fertility variation among clones of Korean pine (Pinus koraiensis S. et Z.) and its implications on seed orchard management. For. Genet. 1999, 6, 191–200. [Google Scholar]
- Lindgren, D.; Gea, L.; Jefferson, P. Loss of genetic diversity monitored by status number. Silvae Genetica 1996, 45, 52–59. https://www.degruyter.com.
- Lindgren, D.; Mullin, T.J. Relatedness and status number in seed orchard crops. Canadian Journal of Forest Research 1998, 28, 276–283. [Google Scholar] [CrossRef]
- Eriksson, G.; Jonsson, A.; Lindgren, D. Flowering in a clonal trial of Picea abies (Karst.). Studia Forestalia Sueccica 1973, 110, 4–45. [Google Scholar]
- Hedegart, T. Breeding systems, variation and genetic improvement of teak. Tropical trees: Variation, Breeding and Conservation Linnean Society Symposium Series:2, 1976, New York.
- Eser, Y. Variation in cone production of Anatolian black pine [Pinus nigra Arnold. subsp. pallasiana (Lamb.) Holmboe] over habitat classes. Biol Bull Russ Acad Sci. 2024, 51(1), 121–127. [Google Scholar] [CrossRef]
- Bilir, N.; Çetinkaya, D. Cone production by age groups in Cedrus libani. 2nd International Mediterranean Conference, Beirut, February 2-3, 2023; pp. 38–43. [Google Scholar]
- Takos, I.; Merou, T. Effect of storage conditions and treatment on germination of Cedrus deodara Loud. and Cedrus libani A. Rich. Silvae Genetica 2001, 50, 5–6. [Google Scholar]
- Atalay, I. General ecological properties of natural occurrence areas of cedar (Cedrus libani A. Rich.) forests and regioning of seed transfer of cedar in Turkey. General Directorate of Forestry press: Ankara, 1987. https://www.ogm.gov.tr.com.
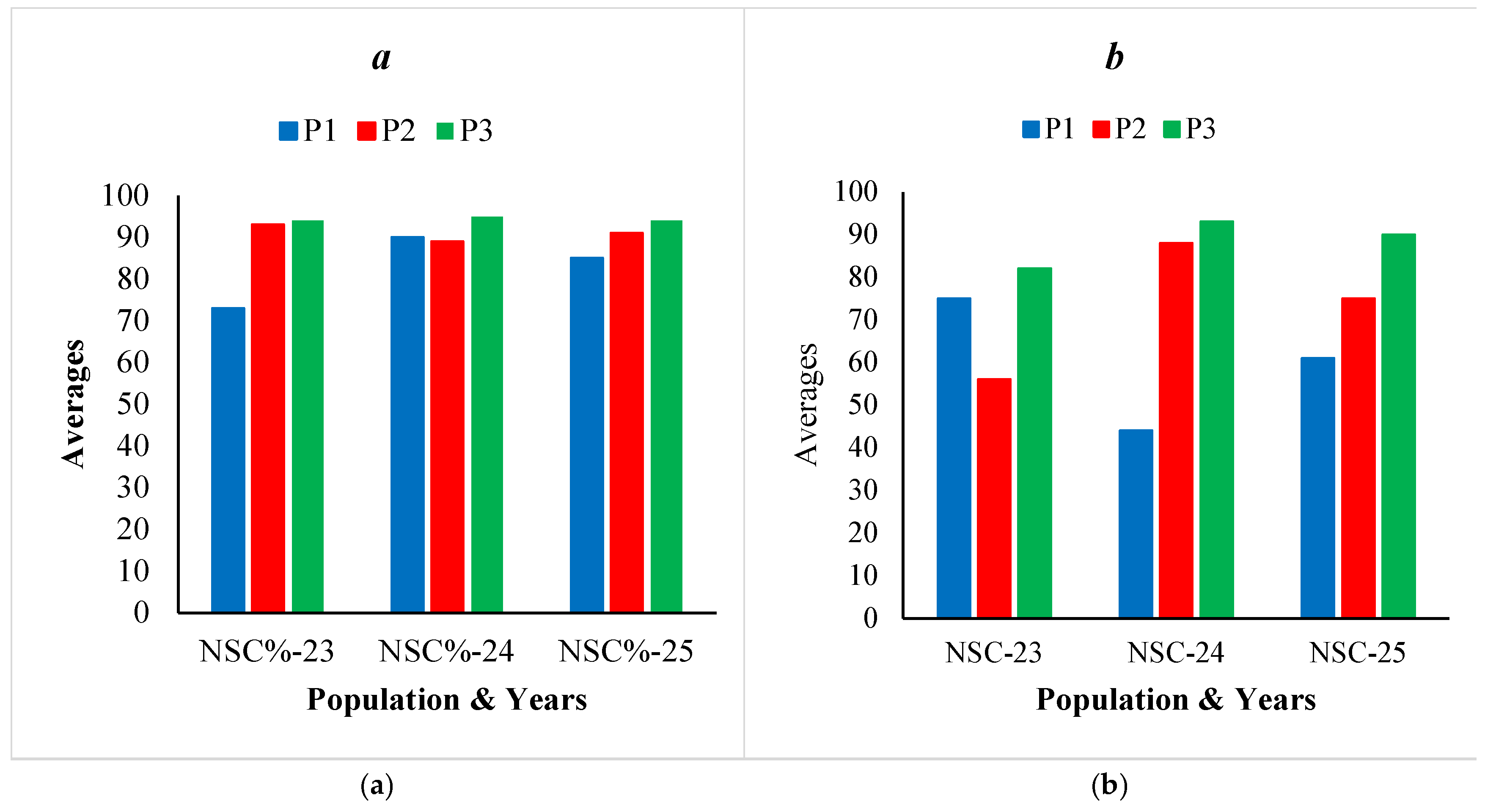
| Seed Stand code | Latitude (N) |
Longitude (E) | Altitude (m) |
Stand age (year) | Size (ha) |
Density (trees/ha) |
| P1 | 38°05′23′′ | 30°42′20′′ | 1,600 | 60 | 77.0 | 800 |
| P2 | 37°44′47′′ | 30°49′21′′ | 1,600 | 118 | 48.9 | 650 |
| P3 | 37°51′00′′ | 31°19′10′′ | 1,550 | 149 | 23.0 | 450 |
| P1 | P2 | P3 | Pooled | ||||||||
| Years | Mean | CV | Range | Mean | CV | Range | Mean | CV | Range | Mean | CV |
| 2023 | |||||||||||
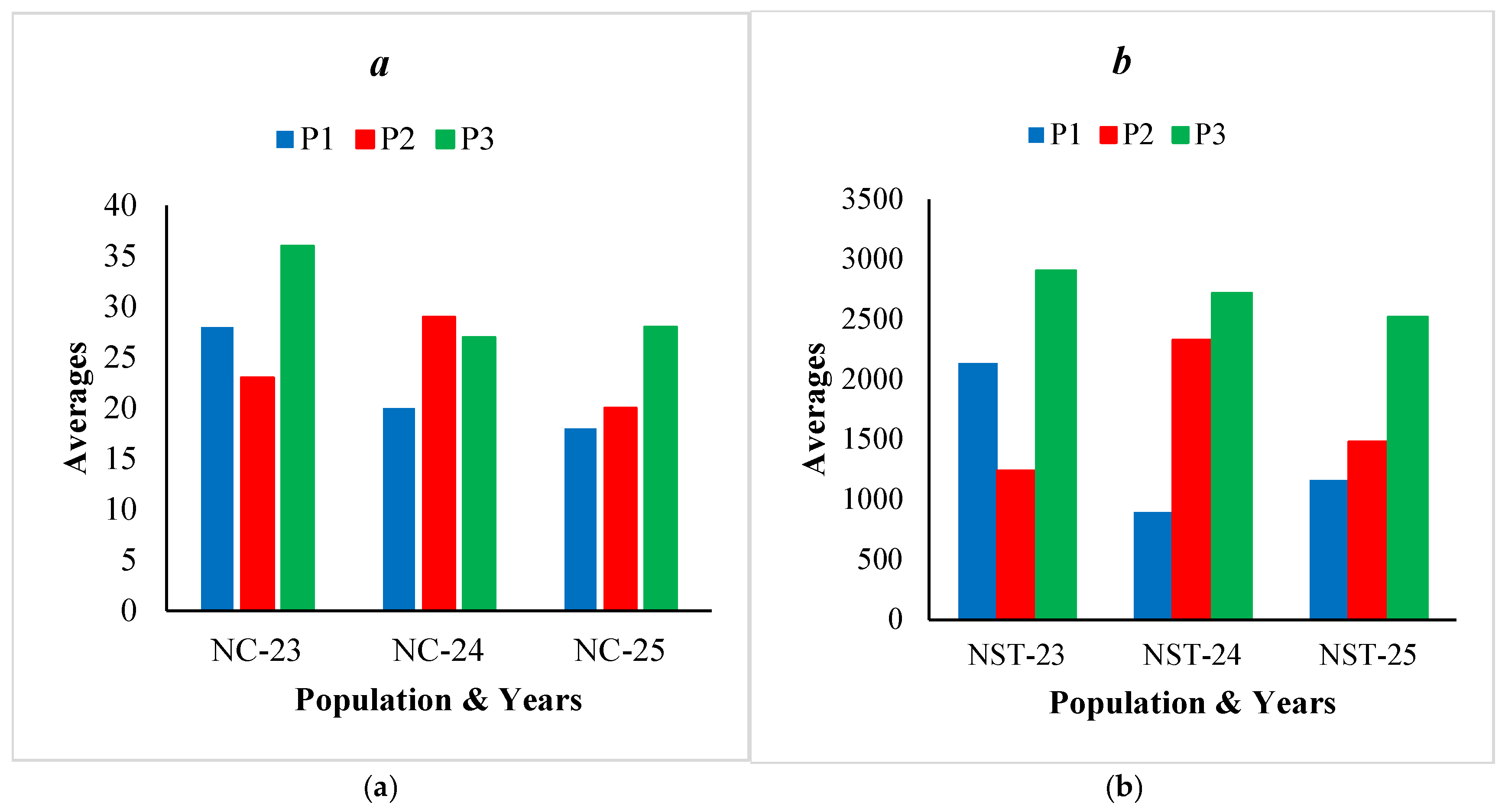
| NC | 28 | 0.48 | 12-90 | 23 | 0.58 | 12-86 | 36 | 0.58 | 14-110 | 29 | 0.59 |
| NSC% | 73 | 0.17 | 37-95 | 93 | 0.04 | 83-100 | 94 | 0.04 | 80-98 | 87 | 0.14 |
| NSC | 75 | 0.30 | 22-120 | 56 | 0.44 | 12-140 | 82 | 0.32 | 19-131 | 71 | 0.38 |
| NST | 2135 | 0.55 | 374-5940 | 1240 | 0.51 | 192-3080 | 2906 | 0.62 | 742-8580 | 2094 | 0.70 |
| 2024 | |||||||||||
| NC | 20 | 0.85 | 8-72 | 29 | 0.61 | 12-98 | 27 | 0.50 | 14-60 | 25 | 0.65 |
| NSC% | 90 | 0.11 | 66-100 | 89 | 0.11 | 65-100 | 95 | 0.04 | 84-100 | 91 | 0.09 |
| NSC | 44 | 0.59 | 13-110 | 88 | 0.53 | 18-177 | 93 | 0.43 | 16-173 | 75 | 0.59 |
| NST | 895 | 1.19 | 156-5040 | 2325 | 0.62 | 396-6109 | 2715 | 0.69 | 320-6688 | 1978 | 0.85 |
| 2025 | |||||||||||
| NC | 18 | 0.45 | 10-48 | 20 | 0.31 | 12-40 | 28 | 0.63 | 12-90 | 22 | 0.57 |
| NSC% | 85 | 0.14 | 51-100 | 91 | 0.06 | 72-97 | 94 | 0.04 | 85-100 | 90 | 0.01 |
| NSC | 61 | 0.53 | 16-120 | 75 | 0.43 | 18-130 | 90 | 0.37 | 19-164 | 75 | 0.45 |
| NST | 1165 | 0.85 | 160-5472 | 1482 | 0.62 | 288-5000 | 2518 | 0.76 | 760-10152 | 1722 | 0.85 |
| Pooled | |||||||||||
| NC | 22 | 0.63 | 8-90 | 24 | 0.58 | 12-98 | 31 | 0.59 | 12-110 | 26 | 0.61 |
| NSC% | 83 | 0.16 | 37-100 | 91 | 0.07 | 65-100 | 94 | 0.04 | 80-100 | 89 | 0.11 |
| NSC | 60 | 0.50 | 13-120 | 73 | 0.51 | 12-177 | 88 | 0.38 | 16-173 | 74 | 0.48 |
| NST | 1399 | 0.86 | 156-5940 | 1682 | 0.68 | 192-6109 | 2713 | 0.68 | 320-10152 | 1931 | 0.80 |
| Source | Variable | d.f. | Mean square | F | P |
|---|---|---|---|---|---|
| Stand (S) | NC | 2 | 2103.581 | 9.427 | ** |
| NSC% | 2 | 3774.421 | 60.731 | ** | |
| NSC | 2 | 20765.343 | 19.774 | ** | |
| NST | 2 | 50229040.060 | 26.239 | ** | |
| Year (Y) | NC | 2 | 1287.114 | 5.768 | * |
| NSC% | 2 | 661.029 | 10.636 | ** | |
| NSC | 2 | 557.867 | .531 | NS | |
| NST | 2 | 3811994.994 | 1.991 | NS | |
| S x Y interaction | NC | 4 | 625.881 | 2.805 | * |
| NSC% | 4 | 1204.025 | 19.373 | ** | |
| NSC | 4 | 8862.324 | 8.439 | ** | |
| NST | 4 | 11881578.603 | 6.207 | ** | |
| Error | NC | 306 | 223.147 | ||
| NSC% | 306 | 62.150 | |||
| NSC | 306 | 1050.127 | |||
| NST | 306 | 1914322.016 |
| P1 | P2 | P3 | Pooled | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Years | 23 | 24 | 25 | Total | 23 | 24 | 25 | Total | 23 | 24 | 25 | Total | 23 | 24 | 25 | Total |
| ΨC* | 1.22 | 1.71 | 1.20 | 1.40 | 1.33 | 1.36 | 1.10 | 1.33 | 1.33 | 1.24 | 1.38 | 1.34 | 1.34 | 1.42 | 1.32 | 1.36 |
| ΨS | 1.29 | 2.38 | 1.71 | 1.73 | 1.25 | 1.38 | 1.37 | 1.46 | 1.38 | 1.46 | 1.56 | 1.46 | 1.48 | 1.71 | 1.71 | 1.63 |
| NP(C) | 28.7 | 20.5 | 29.2 | 75.6 | 26.4 | 25.7 | 32.0 | 79.0 | 26.3 | 28.6 | 25.3 | 78.3 | 78.4 | 74.0 | 79.6 | 231.5 |
| Nr(C)** | 0.82 | 0.59 | 0.84 | 0.72 | 0.75 | 0.73 | 0.91 | 0.75 | 0.75 | 0.81 | 0.72 | 0.75 | 0.74 | 0.71 | 0.76 | 0.74 |
| NP(S) | 27.1 | 14.7 | 20.5 | 60.9 | 28.0 | 25.4 | 25.5 | 72.0 | 25.4 | 24.0 | 22.5 | 71.9 | 71.0 | 61.3 | 61.5 | 193.8 |
| Nr(S) | 0.77 | 0.42 | 0.59 | 0.58 | 0.80 | 0.73 | 0.73 | 0.69 | 0.73 | 0.69 | 0.64 | 0.69 | 0.68 | 0.58 | 0.59 | 0.62 |
| GDC | 0.983 | 0.976 | 0.983 | 0.993 | 0.981 | 0.981 | 0.984 | 0.994 | 0.981 | 0.982 | 0.980 | 0.994 | 0.994 | 0.993 | 0.994 | 0.998 |
| GDS | 0.982 | 0.966 | 0.976 | 0.992 | 0.982 | 0.980 | 0.980 | 0.993 | 0.980 | 0.979 | 0.978 | 0.993 | 0.993 | 0.992 | 0.992 | 0.997 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




