Submitted:
03 February 2026
Posted:
04 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
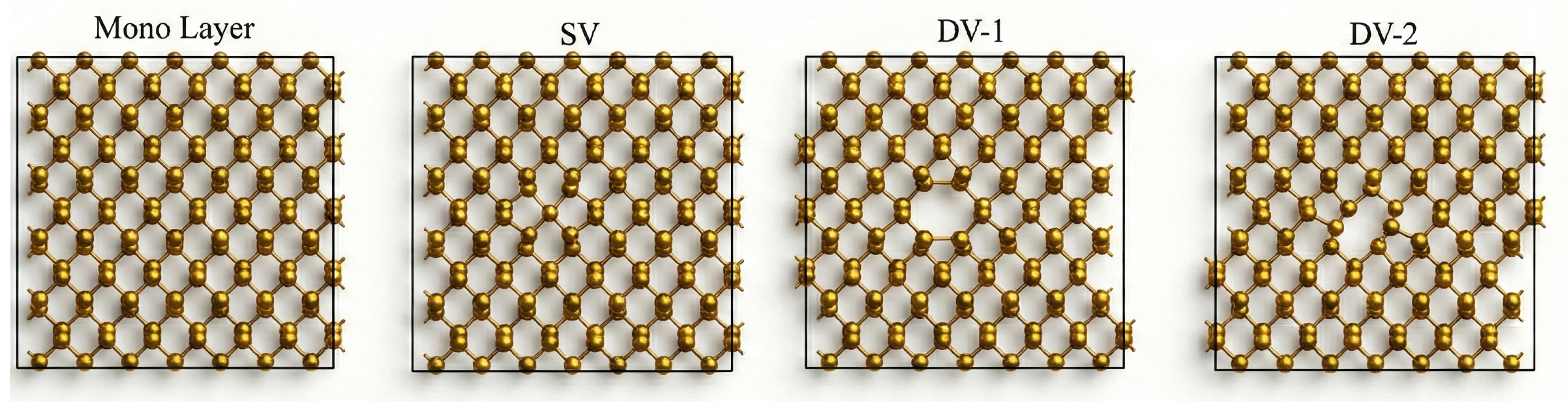
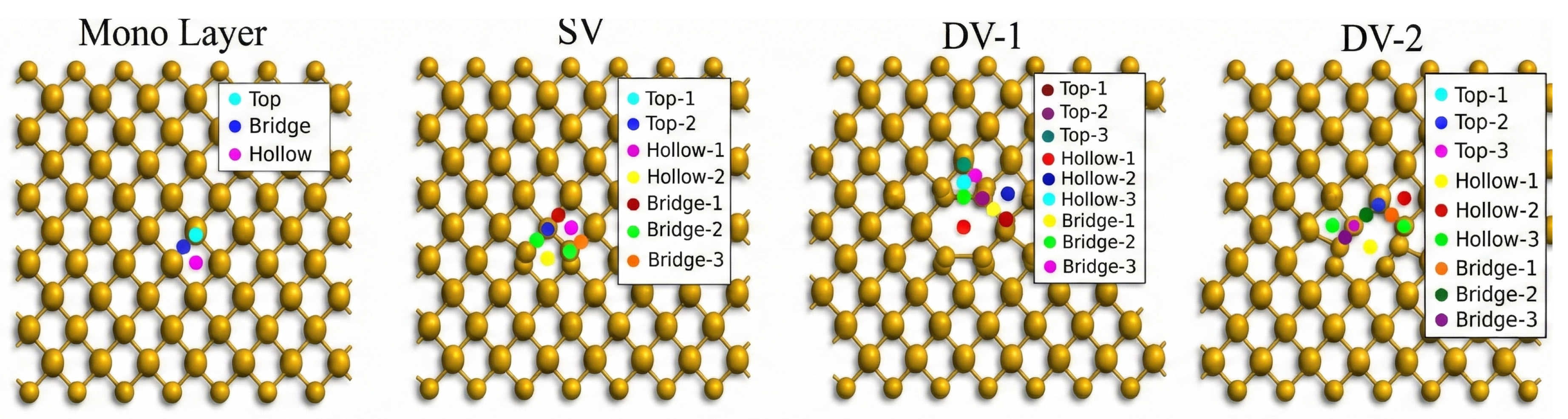
2. Methodology
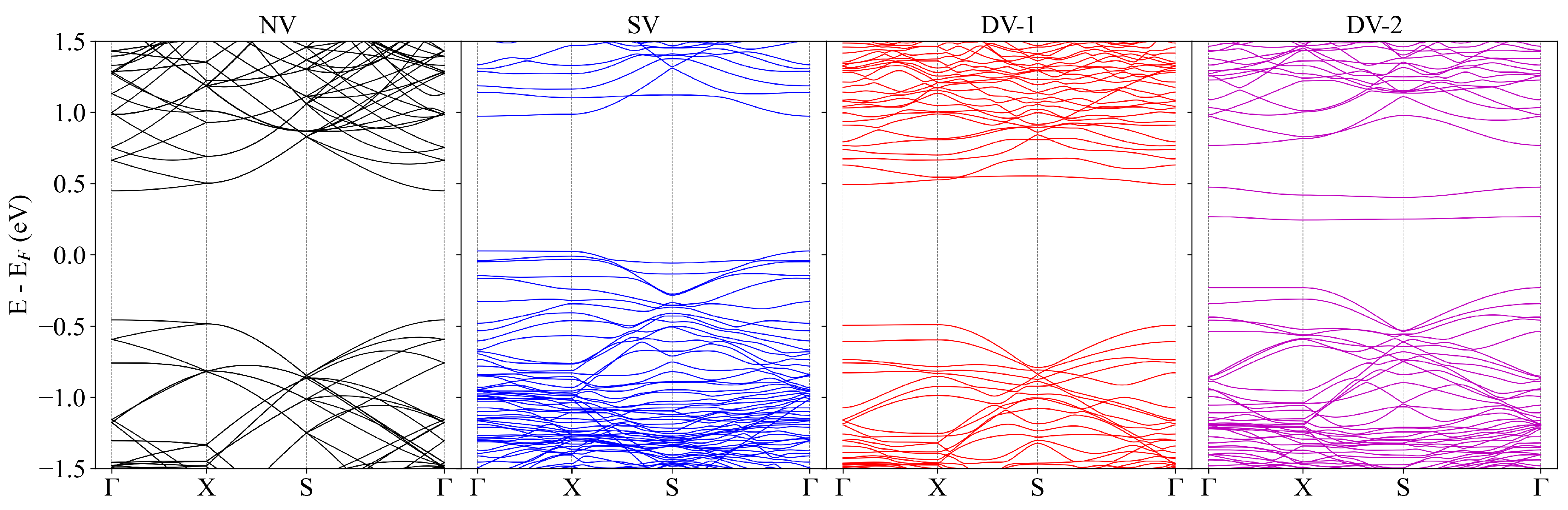
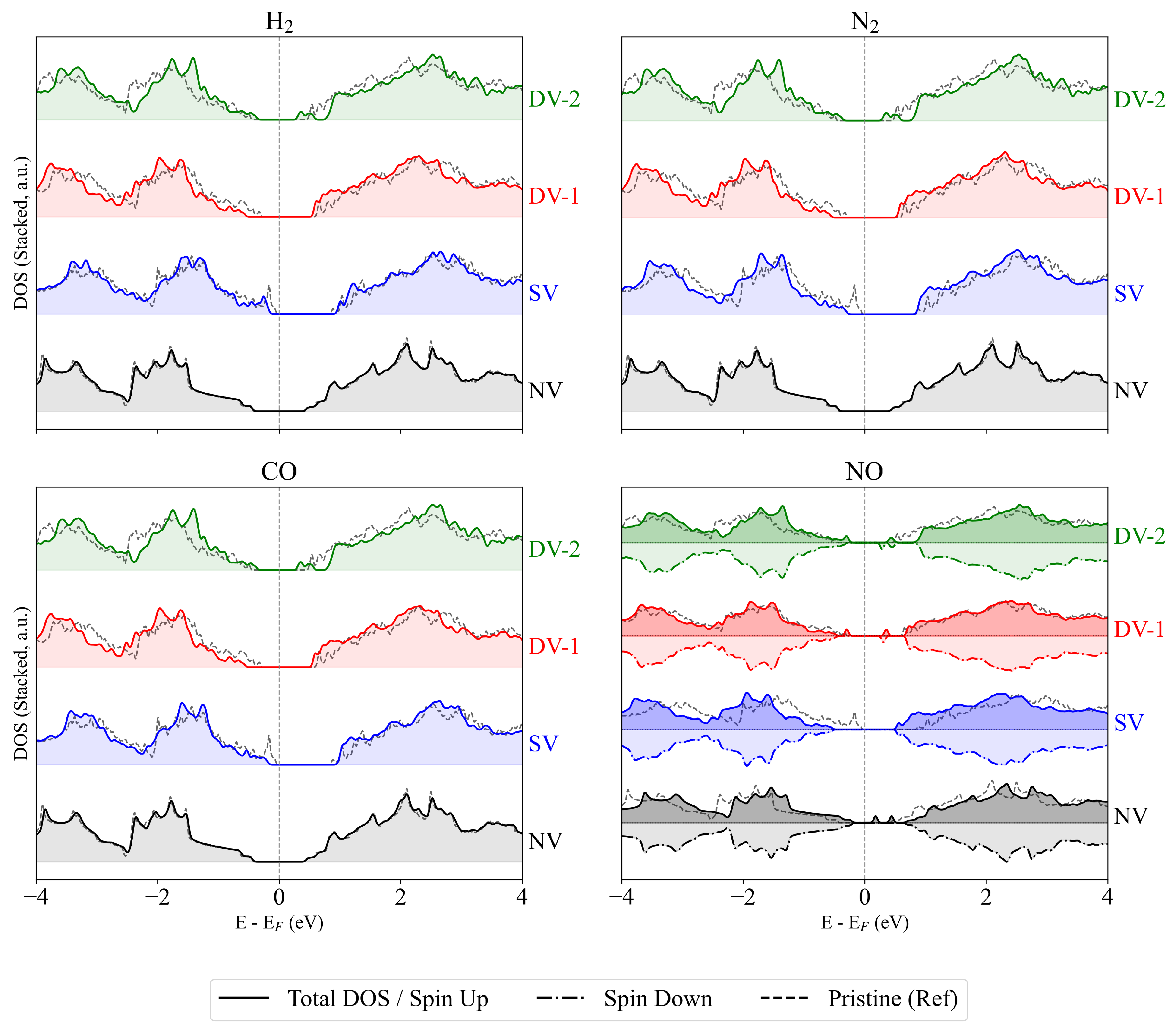
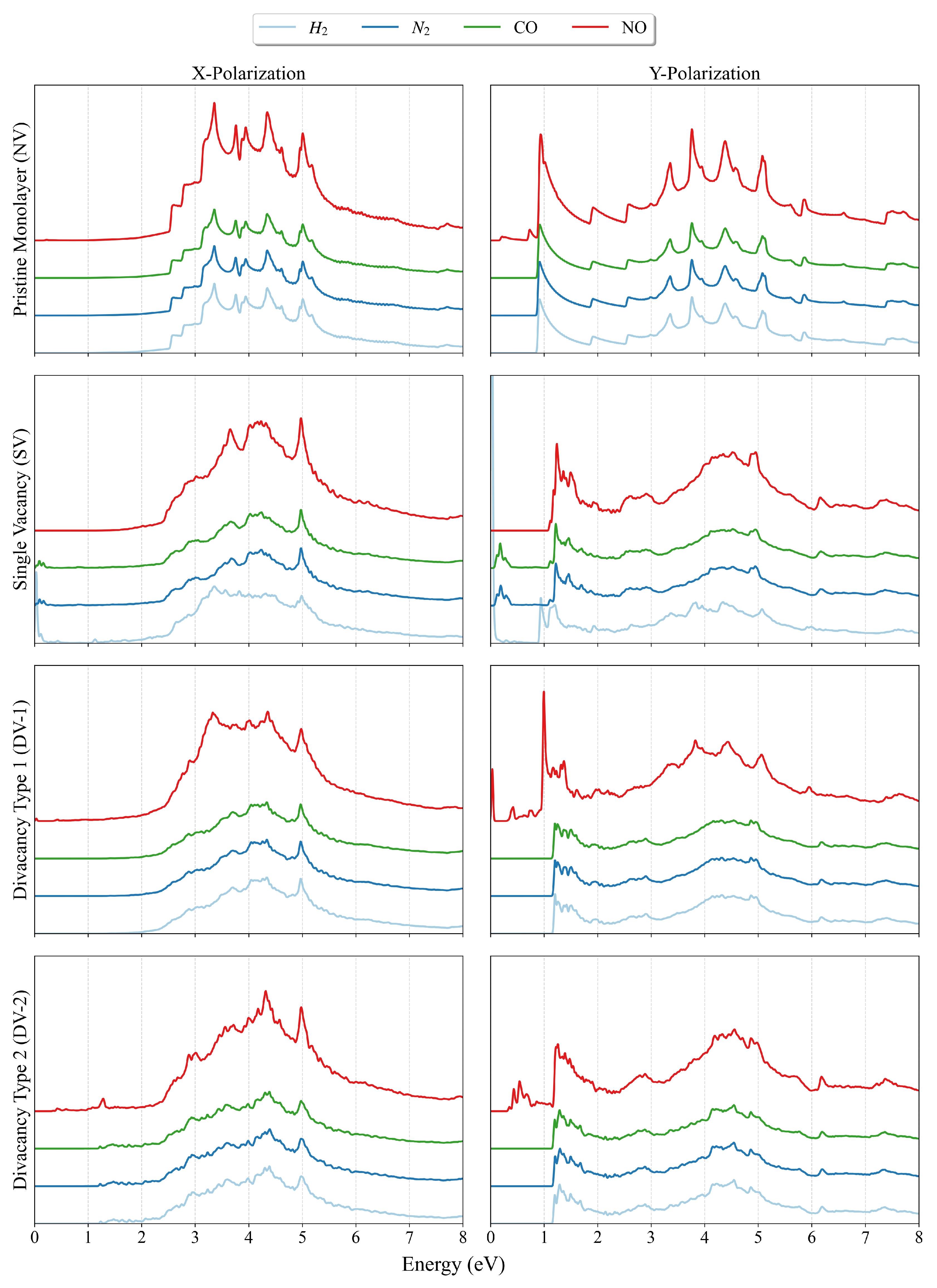
3. Result and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
References
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric field effect in atomically thin carbon films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef]
- Tang, S.; Cao, Z. Adsorption and dissociation of ammonia on graphene oxides: A first-principles study. J. Phys. Chem. C 2012, 116, 8778–8791. [Google Scholar] [CrossRef]
- Mattson, E.C.; Pande, K.; Unger, M.; Cui, S.; Lu, G.; Gajdardziska-Josifovska, M.; Weinert, M.; Chen, J.; Hirschmugl, C.J. Exploring adsorption and reactivity of NH3 on reduced graphene oxide. J. Phys. Chem. C 2013, 117, 10698–10707. [Google Scholar] [CrossRef]
- Zhu, S.; Sun, H.; Liu, X.; Zhuang, J.; Zhao, L. Room-temperature NH3 sensing of graphene oxide film and its enhanced response on the laser-textured silicon. Sci. Rep. 2017, 7, 14773. [Google Scholar] [CrossRef] [PubMed]
- Batmunkh, M.; Bat-Erdene, M.; Shapter, J.G. Phosphorene and phosphorene-based materials–prospects for future applications. Adv. Mater. 2016, 28, 8586–8617. [Google Scholar] [CrossRef]
- Li, L.; Yu, Y.; Ye, G.J.; Ge, Q.; Ou, X.; Wu, H.; Feng, D.; Chen, X.H.; Zhang, Y. Black phosphorus field-effect transistors. Nat. Nanotechnol. 2014, 9, 372–377. [Google Scholar] [CrossRef]
- Mohammadi, M.; Tavangar, Z. Adsorption of aromatic molecules on a black phosphorene surface: A first-principles study. New J. Chem. 2023, 47, 1842–1851. [Google Scholar] [CrossRef]
- Talukdar, D.; Mohanta, D.; Ahmed, G.A. Nitrogen doped compound defect in black phosphorene for enhanced gas sensing. Surf. Interfaces 2024, 51, 104699. [Google Scholar] [CrossRef]
- Tariq, M.; Khattak, J.I.; Iqbal, M.; Ullah, R.; Zeb, A.; Khan, M.; Khan, A.; Mahmood, T.; Ahmad, I. DFT study of the therapeutic potential of phosphorene as a drug delivery system for chlorambucil to treat cancer. RSC Adv. 2019, 9, 24325–24332. [Google Scholar] [CrossRef]
- Pica, M.; D’Amato, R. Chemistry of Phosphorene: Synthesis, Functionalization and Biomedical Applications in an Update Review. Inorganics 2020, 8, 29. [Google Scholar] [CrossRef]
- Kumar, A.; Kumar, M.; Kumar, M. Recent advances in phosphorene-based gas sensors. Sens. Actuators A Phys. 2019, 295, 523–537. [Google Scholar]
- de Sousa, F.E.B. Electronic, Transport and Optical Properties in Multilayer Phosphorene. Ph.D. Thesis, Federal University of Ceará, Brazil, 2023. [Google Scholar]
- Akilan, R.; Vinnarasi, S.; Mohanapriya, S.; Shankar, R. Adsorption of H2 molecules on B/N-doped defected graphene sheets—A DFT study. Struct. Chem. 2020, 31, 2413–2434. [Google Scholar] [CrossRef]
- Halder, P.; Mondal, I.; Kundu, M.; Ghosh, A.; Paul, B.K.; Biswas, S.; Sau, S.; Chattopadhyay, B.; Mondal, D.; Das, S. Optimizing phosphorene nanosheets for high performance all-solid asymmetric supercapacitors: A theoretical and experimental insight. J. Energy Storage 2024, 94, 112451. [Google Scholar] [CrossRef]
- Cui, X.; Tang, X.; Niu, Y.; Tong, L.; Zhao, H.; Yang, Y.; Jin, G.; Li, M.; Han, X. Functional phosphorene: Burgeoning generation, two-dimensional nanotherapeutic platform for oncotherapy. Coord. Chem. Rev. 2024, 507, 215744. [Google Scholar] [CrossRef]
- Zhu, Y.; Xie, Z.; Li, J.; Liu, Y.; Li, C.; Liang, W.; Huang, W.; Kang, J.; Cheng, F.; Kang, L.; Al-Hartomy, O.A.; Al-Ghamdi, A.; Wageh, S.; Xu, J.; Li, D.; Zhang, H. From phosphorus to phosphorene: Applications in disease theranostics. Coord. Chem. Rev. 2021, 446, 214110. [Google Scholar] [CrossRef]
- Zeng, X.; Liu, G.; Wang, W.; Wang, Y.; Liu, F.; Wu, M. Black phosphorus: Synthesis, stability, and applications. iScience 2021, 24, 103116. [Google Scholar] [CrossRef] [PubMed]
- Kaewmaraya, T.; Singh, D.; Kumar, A.; Park, S.; Lee, H. Drastic Improvement in Gas-Sensing Characteristics of Phosphorene Nanosheets under Vacancy Defects and Elemental Functionalization. J. Phys. Chem. C 2018, 122, 19864–19874. [Google Scholar] [CrossRef]
- Xiao, Y.; Zhou, M.; Zeng, M.; Fu, L. Atomic-scale structural modification of 2D materials. Adv. Sci. 2019, 6, 1801501. [Google Scholar] [CrossRef]
- Hu, W.; Yang, J. Defects in phosphorene. J. Phys. Chem. C 2015, 119, 20474–20480. [Google Scholar] [CrossRef]
- Pantha, N.; Chauhan, B.; Sharma, P.; Adhikari, N.P. Tuning structural and electronic properties of phosphorene with vacancies. J. Nepal Phys. Soc. 2020, 6, 7–15. [Google Scholar] [CrossRef]
- Cai, Y.; Chen, S.; Gao, J.; Zhang, G.; Zhang, Y.W. Evolution of intrinsic vacancies and prolonged lifetimes of vacancy clusters in black phosphorene. Nanoscale 2019, 11, 20987–20995. [Google Scholar] [CrossRef] [PubMed]
- Zhan, F.; Xu, W.; Zou, R.; Yang, J.; Fan, J.; Wu, X.; Wang, R. Interplay of charged states and oxygen dissociation induced by vacancies in phosphorene. J. Phys. Chem. C 2019, 123, 27080–27087. [Google Scholar] [CrossRef]
- Srivastava, P.; Hembram, K.P.S.S.; Mizuseki, H.; Lee, K.-R.; Han, S.S.; Kim, S. Tuning the electronic and magnetic properties of phosphorene by vacancies and adatoms. J. Phys. Chem. C 2015, 119, 6530–6538. [Google Scholar] [CrossRef]
- de Sousa, F.E.B.; Araújo, F.R.V.; Farias, G.A.; de Sousa, J.S.; da Costa, D.R. Effects on the electronic properties of multilayer phosphorene due to periodic arrays of vacancies: Band unfolding formalism. Physica E 2023, 152, 115750. [Google Scholar] [CrossRef]
- Li, J.-x.; Zhang, Q.; Ma, D.-w.; Liu, C.-k.; Li, F. Adsorption properties of CO2, NO2 and SO2 on pristine/defective/non-metallic element doping phosphorene by DFT method. SSRN Electron. J. 2024. [Google Scholar]
- Singsen, S.; Watwiangkham, A.; Ngamwongwan, L.; Fongkaew, I.; Jungthawan, S.; Suthirakun, S. Defect Engineering of Green Phosphorene Nanosheets for Detecting Volatile Organic Compounds: A Computational Approach. ACS Appl. Nano Mater. 2023, 6, 1496–1506. [Google Scholar] [CrossRef]
- Ghadiri, M.; Ghashghaee, M.; Ghambarian, M. Defective phosphorene for highly efficient formaldehyde detection: Periodic density functional calculations. Phys. Lett. A 2020, 384, 126792. [Google Scholar] [CrossRef]
- Kou, L.; Frauenheim, T.; Chen, C. Phosphorene as a superior gas sensor: Selective adsorption and distinct I-V response. J. Phys. Chem. Lett. 2014, 5, 2675–2681. [Google Scholar] [CrossRef] [PubMed]
- Donarelli, M.; Ottaviano, L. 2D materials for gas sensing applications: A review on graphene oxide, MoS2, WS2 and phosphorene. Sensors 2018, 18, 3638. [Google Scholar] [CrossRef]
- Khudair, S.A.M.; Mohaimeed, A.A. Gas Sensor Investigations through Adsorption of Toxic Gas Molecules on Single and Double Vacancy Graphene. NeuroQuantology 2020, 18, 87–95. [Google Scholar] [CrossRef]
- Qin, Y.; Cao, M.; Lei, M.; Feng, W. First-principles study on the selective gas adsorption of defective SnS. Vacuum 2021, 183, 109792. [Google Scholar] [CrossRef]
- Corona-García, C.A.; Lucas, H.R.; Ramírez-Pimentel, J.G.; Cruz-Martínez, H.; Cocoletzi, G.H. Adsorption of small pollutant molecules on monolayer aluminum-doped and single-vacancy blue phosphorene. J. Mol. Model. 2021, 27, 141. [Google Scholar] [CrossRef]
- Soler, J.M.; Artacho, E.; Gale, J.D.; García, A.; Junquera, J.; Ordejón, P.; Sánchez-Portal, D. The SIESTA method for ab initio order-N materials simulation. J. Phys. Condens. Matter 2002, 14, 2745. [Google Scholar] [CrossRef]
- Zuluaga-Hernandez, E.A.; Flórez, E.; Dorkis, L.; Mora-Ramos, M.E.; Correa, J.D. Small molecule gas adsorption onto blue phosphorene oxide layers. Appl. Surf. Sci. 2020, 530, 147039. [Google Scholar] [CrossRef]
- Klimeš, J.; Bowler, D.R.; Michaelides, A. Chemical accuracy for the van der Waals density functional. J. Phys. Condens. Matter 2009, 22, 022201. [Google Scholar] [CrossRef] [PubMed]
- Larsen, A.H.; Mortensen, J.J.; Blomqvist, J.; Castelli, I.E.; Christensen, R.; Dułak, M.; Friis, J.; Groves, M.N.; Hammer, B.; Hargus, C.; et al. The atomic simulation environment—a Python library for working with atoms. J. Phys. Condens. Matter 2017, 29, 273002. [Google Scholar] [CrossRef]
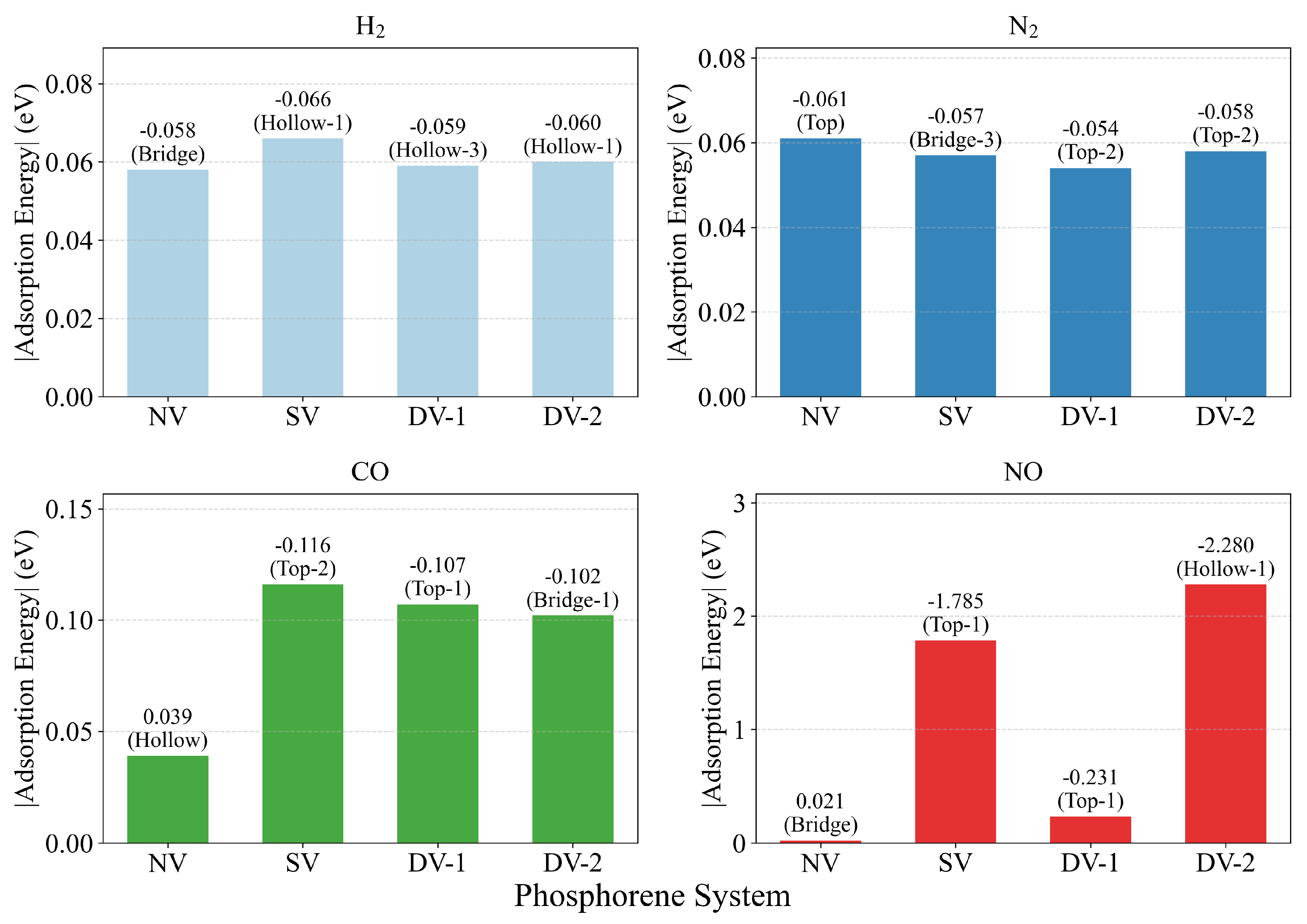
| Molecule | Hollow | Bridge | Top | |||
|---|---|---|---|---|---|---|
| d | d | d | ||||
| -0.057 | 0.778 | -0.058 | 0.778 | -0.048 | 0.778 | |
| -0.031 | 1.118 | -0.042 | 1.119 | -0.060 | 1.119 | |
| CO-v1 | -0.009 | 1.151 | 0.021 | 1.151 | 0.019 | 1.152 |
| NO-v1 | -0.391 | 1.178 | -0.383 | 1.179 | -0.341 | 1.179 |
| CO-v2 | 0.039 | 1.151 | 0.016 | 1.151 | 0.007 | 1.152 |
| NO-v2 | -0.396 | 1.182 | -0.374 | 1.183 | -0.396 | 1.185 |
| Defect Type | CO | NO | ||
|---|---|---|---|---|
| SV | 0.0 | 0.0 | 0.0 | 1.000 |
| DV-1 | 0.0 | 0.0 | 0.0 | 1.000 |
| DV-2 | 0.0 | 0.0 | 0.0 | 1.000 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




