1. Introduction
Chronic urticaria (CU) is a mast cell–driven inflammatory skin disorder characterized by the recurrent occurrence of wheals, angioedema, or both, persisting for more than six weeks and frequently leading to marked impairment in quality of life. Traditionally, CU has been regarded as a predominantly histamine-mediated condition, with disease management largely focused on the use of H1-antihistamines. However, this approach does not fully explain the clinical heterogeneity observed among patients, particularly with regard to disease severity and therapeutic response [
1,
2,
3].
Among the different forms of CU, chronic spontaneous urticaria (CSU) represents the most prevalent and clinically challenging subtype, characterized by the absence of identifiable external triggers [
2,
6]. Despite its apparent simplicity, CSU often follows an unpredictable trajectory, with marked interindividual variability in disease severity, duration, and response to treatment [
3]. These observations raise the question of whether CSU represents a single disease entity or a spectrum of immunologically distinct conditions. To further contextualize the complexity and thematic structure of current research in CSU, a bibliometric co-occurrence analysis of keywords was performed.
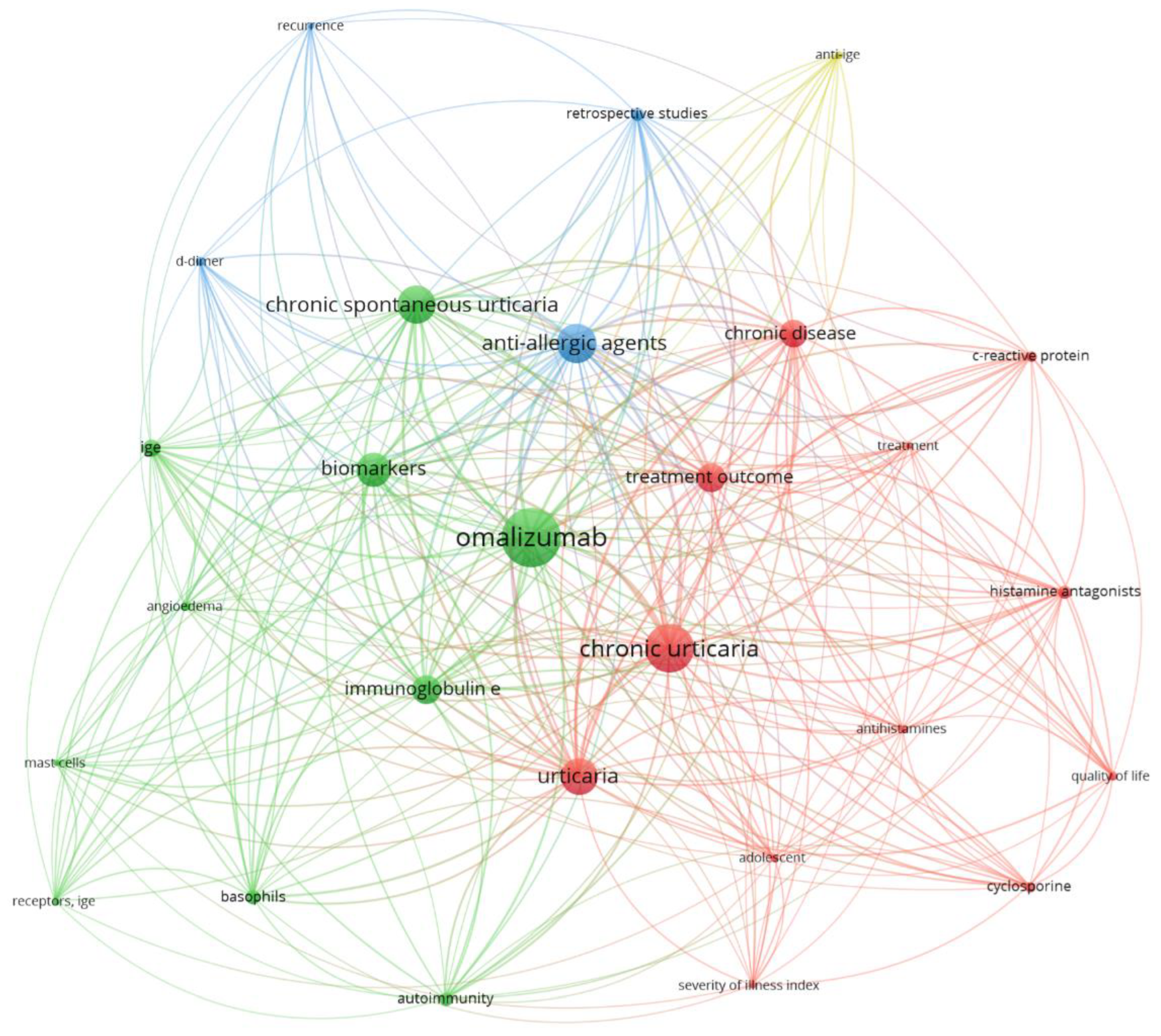
Figure 1.
Bibliometric co-occurrence network of keywords related to chronic urticaria and allergic disorders. The map reveals interconnected thematic clusters encompassing immunopathological mechanisms, disease endotypes, biomarkers of activity and severity, and therapeutic strategies, highlighting the central role of IgE-mediated pathways, mast cell activation, and targeted treatments in chronic urticaria research. The visualization was generated using VOSviewer (version 1.6.20).
Figure 1.
Bibliometric co-occurrence network of keywords related to chronic urticaria and allergic disorders. The map reveals interconnected thematic clusters encompassing immunopathological mechanisms, disease endotypes, biomarkers of activity and severity, and therapeutic strategies, highlighting the central role of IgE-mediated pathways, mast cell activation, and targeted treatments in chronic urticaria research. The visualization was generated using VOSviewer (version 1.6.20).
Advances in immunology over the past decade have further refined the understanding of CSU, supporting its classification as a heterogeneous immune-mediated disease. Rather than a single pathogenic mechanism, CSU encompasses distinct endotypes involving autoimmunity, autoallergy, and dysregulated inflammatory pathways that converge on mast cell activation [
2,
4]. Autoimmune mechanisms have emerged as central pathogenic pathways, including IgE-mediated autoallergy (type I autoimmunity) and IgG-mediated autoimmunity directed against FcεRI or IgE (type IIb autoimmunity) [
4,
9]. These mechanisms offer a biological explanation for disease chronicity, severity, and resistance to conventional antihistamine therapy.
This evolving pathophysiological framework carries important clinical implications. The recognition of distinct autoimmune and inflammatory endotypes challenges the traditional stepwise, symptom-based approach to CSU management and underscores the limitations of uniform treatment escalation [
4,
8]. Instead, it highlights the need for a mechanism-oriented strategy integrating immunological profiling, biomarker assessment, and targeted therapeutic selection [
10,
13].
In parallel, the therapeutic landscape of CSU has expanded considerably. The introduction of anti-IgE therapy marked a paradigm shift in disease control; however, heterogeneous response rates and disease relapse after treatment discontinuation reveal persistent unmet needs [
8]. Emerging therapies targeting intracellular signaling pathways, cytokine networks, and mast cell activation further reinforce the necessity of aligning therapeutic decisions with underlying disease mechanisms rather than clinical appearance alone [
14].
Beyond its cutaneous manifestations, chronic spontaneous urticaria shares fundamental immunopathological mechanisms with other allergic disorders, including asthma, allergic rhinitis, atopic dermatitis, and anaphylaxis. These conditions are unified by IgE-dependent and IgE-independent pathways, mast cell and basophil activation, type 2–skewed immune responses, and overlapping inflammatory and biomarker profiles [
1,
2,
4]. Although CSU presents a distinct clinical phenotype, its immunological landscape places it within a broader spectrum of allergic diseases characterized by immune dysregulation and heterogeneous therapeutic responses [
2,
8,
10]. Recognizing these shared mechanisms provides a conceptual framework for interpreting biomarker relevance and therapeutic advances across allergic disorders, while maintaining CSU as a focused model for mechanistic and translational investigation.
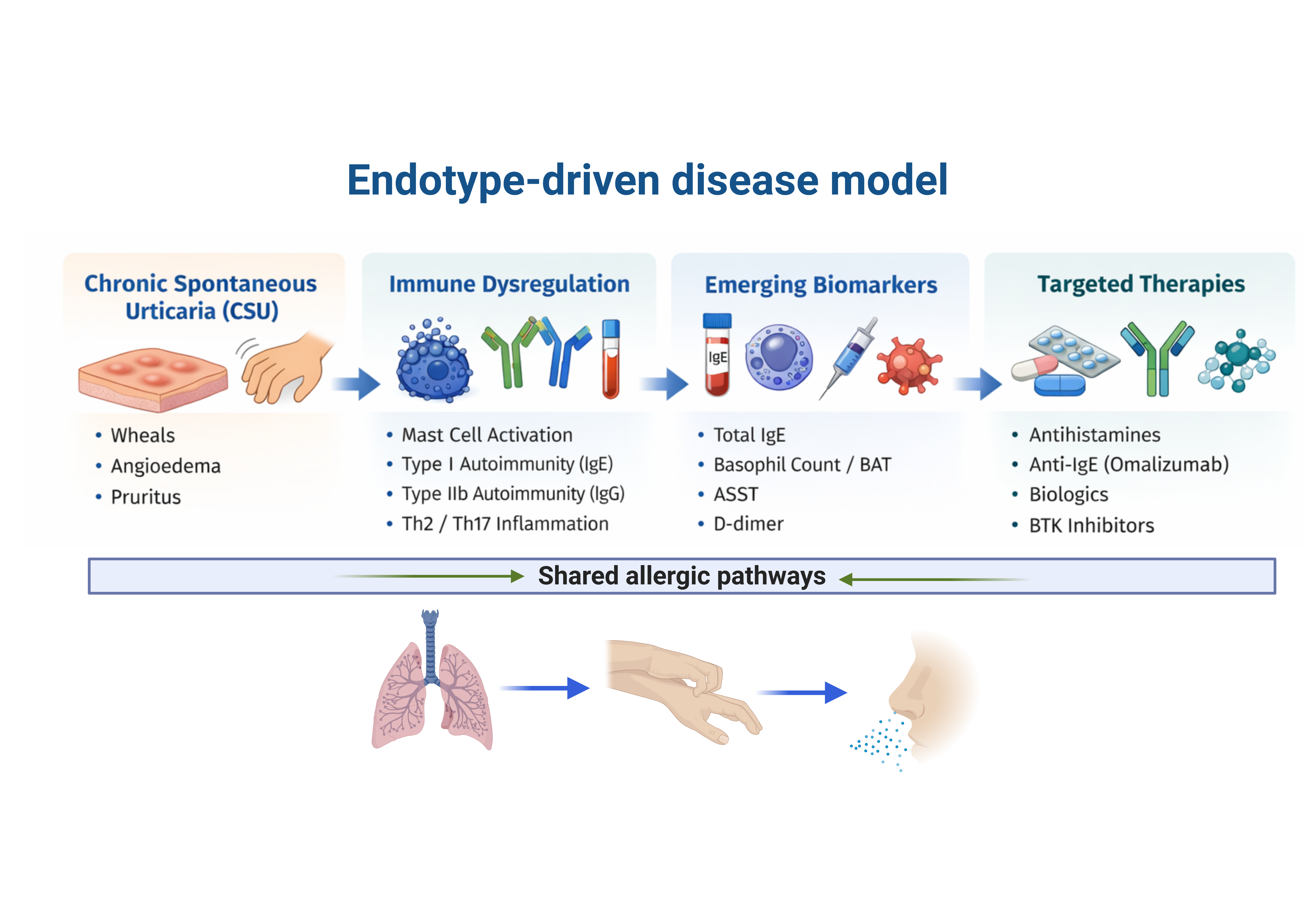
This review critically analyzes CSU as a complex immune-mediated disorder, emphasizing the interaction between mast cell biology, autoimmune mechanisms, and inflammatory amplification. It highlights emerging biomarkers with potential for clinical application in disease stratification and treatment decisions. The discussion includes both established and innovative therapies within an endotype-driven framework. By adopting a mechanistic and translational perspective, this review aims to advance personalized management of chronic urticaria.This approach aligns with current trends in allergy and immunology research, which seek to shift from symptom-based classifications to mechanism-oriented systems, fostering more targeted and effective patient care.
2. Classification and Epidemiological Aspects
According to international guidelines, chronic urticaria (CU) is classified into chronic spontaneous urticaria (CSU) and chronic inducible urticaria, the latter encompassing forms triggered by specific physical or environmental stimuli such as pressure, cold, heat, vibration, or physical exercise [
15,
16]. CSU accounts for approximately 70–80% of CU cases and predominantly affects women, with a peak incidence in middle age [
16,
17].
The clinical course of CSU is typically chronic and recurrent, with an average duration of several years, although marked interindividual variability exists regarding disease severity, frequency of exacerbations, and impact on quality of life [
17]. Epidemiological and real-world studies indicate that a substantial proportion of patients experience persistent or recurrent symptoms, frequently associated with functional limitations, sleep disturbances, and psychosocial impairment [
18].
From a therapeutic perspective, up to approximately half of patients with CSU fail to achieve adequate disease control with standard doses of second-generation H1 antihistamines, highlighting the limitations of conventional approaches and reinforcing the need for personalized therapeutic strategies based on specific pathogenic mechanisms [
18,
19].
3. Pathophysiological Mechanisms
3.1. Mast Cell and Basophil Activation
Mast cells constitute the primary effector cells in the pathophysiology of CSU and are responsible for the release of histamine, proteases, lipid mediators, and pro-inflammatory cytokines that culminate in wheal formation, angioedema, and the induction of pruritus [
20,
21]. Mast cell activation occurs predominantly in the superficial dermis, leading to increased vascular permeability and cellular recruitment.
Basophils also play a relevant role, particularly in subgroups of patients with autoimmune-associated CSU. Quantitative and functional alterations in these cells, including peripheral basopenia and aberrant responses to immunological stimulation, have been associated with disease severity and therapeutic response, especially to anti-IgE therapy [
15,
22].
Beyond classical IgE-mediated activation through the FcεRI receptor, growing evidence indicates that mast cells in CSU may be activated via alternative pathways, including complement receptors, protease-activated receptors, and the MRGPRX2 receptor [
21,
23]. These mechanisms contribute to the observed clinical heterogeneity and help explain inadequate responses to antihistamines and, in some cases, to anti-IgE therapy.
3.2. Autoimmune Endotypes
Autoimmunity is currently recognized as a central component of CSU pathophysiology in a significant proportion of patients. Two main autoimmune mechanisms have been described: type I autoimmune CSU, characterized by IgE autoantibodies directed against autoantigens, and type IIb autoimmune CSU, mediated by IgG autoantibodies against the FcεRI receptor or IgE itself [
21,
24].
Type I autoimmune CSU is frequently associated with elevated total IgE levels and a favorable response to omalizumab therapy. In contrast, type IIb CSU tends to present with more severe disease, reduced IgE levels, basopenia, and limited response to anti-IgE therapy, while in some cases demonstrating better responses to immunosuppressive or immunomodulatory agents [
24,
25].
Recognition of these endotypes has important clinical implications, as it provides a rational basis for patient stratification and targeted therapeutic selection, moving away from uniform approaches based solely on symptom intensity [
25]. Based on current immunopathological evidence, chronic spontaneous urticaria can be stratified into distinct immune endotypes characterized by different pathogenic mechanisms, clinical profiles, and therapeutic responses. These endotypes include IgE-mediated autoallergy (type I autoimmune CSU), IgG-mediated autoimmunity (type IIb autoimmune CSU), and inflammatory phenotypes driven by innate and adaptive immune pathways. The main immunological features, representative biomarkers, and potential therapeutic implications associated with these endotypes are summarized in
Table 1.
3.3. Inflammatory Networks
In addition to autoimmune mechanisms, CSU is sustained by complex inflammatory networks involving cytokines associated with Th2 and Th17 responses, chemokines, complement system activation, and interactions with the coagulation cascade [
21,
26]. These pathways act synergistically, amplifying mast cell activation and perpetuating cutaneous inflammation.
Activation of the coagulation system and the generation of fibrinolytic products, such as D-dimer, have been correlated with increased disease activity, therapeutic refractoriness, and poorer clinical outcomes, reinforcing the interplay between inflammation, hemostasis, and immune responses in CSU [
26,
27]. Taken together, current evidence supports the view that chronic spontaneous urticaria is a mast cell–driven disorder shaped by distinct autoimmune and inflammatory endotypes. IgE-mediated autoallergy, IgG-mediated autoimmunity, basophil dysfunction, and Th2/Th17-driven inflammatory amplification converge within the cutaneous microenvironment to sustain disease activity and clinical heterogeneity. An integrated overview of these immune pathways and their clinical implications is summarized in
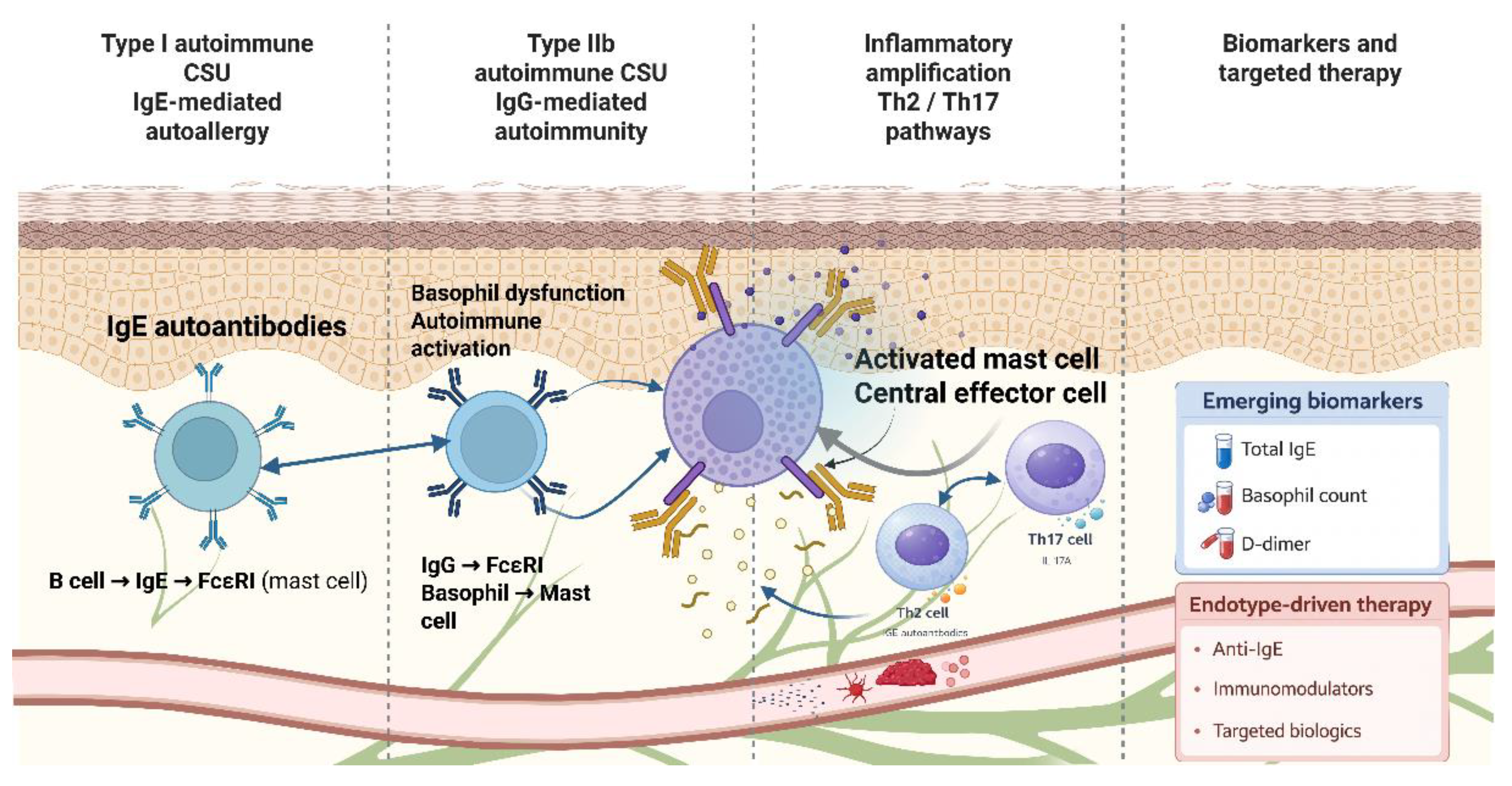
Figure 2.
Distinct autoimmune endotypes, including IgE-mediated autoallergy (type I autoimmune CSU) and IgG-mediated autoimmunity (type IIb autoimmune CSU), converge on mast cell activation within the dermis. Basophil dysfunction and Th2/Th17-driven inflammatory amplification loops contribute to disease persistence and clinical heterogeneity. Emerging biomarkers support disease stratification and an endotype-driven therapeutic approach.
4. Emerging Biomarkers
Despite significant therapeutic advances, the identification of reliable biomarkers for disease stratification and prediction of treatment response remains one of the most relevant unmet needs in chronic spontaneous urticaria (CSU) [
28]. The marked clinical and immunological heterogeneity of CSU limits the utility of single biomarkers and highlights the necessity of multidimensional approaches capable of capturing underlying disease endotypes. Functional assays aimed at detecting autoimmune mechanisms represent an important step toward mechanistic classification. Tests such as the autologous serum skin test and the basophil activation test provide indirect evidence of autoreactivity and have been associated with disease severity and therapeutic response, particularly in patients with suspected type IIb autoimmune CSU [
24,
28].
However, their limited availability, lack of standardization, and variable sensitivity restrict widespread clinical implementation. Total serum IgE has emerged as one of the most clinically accessible biomarkers in CSU. Elevated IgE levels are frequently observed in patients with type I autoimmune (autoallergic) CSU and have been associated with favorable responses to anti-IgE therapy. Conversely, low IgE levels, often accompanied by basopenia and positive functional autoantibody tests, are more characteristic of type IIb autoimmune CSU and correlate with reduced responsiveness to omalizumab [
22,
28].
Although total IgE alone is insufficient for definitive endotype assignment, it provides valuable contextual information when interpreted alongside clinical features and additional laboratory parameters. Markers of coagulation and fibrinolysis, particularly D-dimer, have gained attention as indicators of disease activity and inflammatory burden. Elevated D-dimer levels have been correlated with increased disease severity, treatment refractoriness, and impaired quality of life, reflecting the complex interplay between immune activation, endothelial dysfunction, and the coagulation cascade in CSU [
26]. While not specific, such markers contribute to a broader inflammatory profile that may inform prognosis and therapeutic monitoring.
Cellular biomarkers, including basophil counts and functional responsiveness, further refine disease stratification. Basopenia and altered basophil activation profiles have been associated with more severe disease and differential responses to therapy, reinforcing the role of basophils as both effectors and indicators of immune dysregulation in CSU [
22,
28]. Cytokine signatures and chemokine profiles reflecting Th2- and Th17-skewed responses are also under investigation, although their clinical applicability remains limited by methodological complexity and inter-study variability [
29]. Importantly, several of these biomarkers, including total IgE levels and type 2–associated inflammatory signatures, have been extensively explored across multiple allergic diseases, reinforcing their relevance as indicators of shared immunological endotypes rather than disease-specific markers [
2,
4,
28].
Collectively, current evidence suggests that no single biomarker is sufficient to capture the complexity of CSU. Instead, combined biomarker panels integrating immunoglobulin profiles, functional assays, cellular parameters, and inflammatory markers appear to offer greater potential for defining disease endotypes and guiding personalized therapeutic decisions [
28,
29]. The translation of these approaches into routine clinical practice will depend on further validation studies, standardization of assays, and the development of pragmatic algorithms that balance mechanistic insight with clinical feasibility.
5. Therapeutic Advances
The therapeutic management of chronic spontaneous urticaria (CSU) has evolved substantially over the past decade, transitioning from a predominantly symptom-oriented strategy toward a more mechanism-based approach. Second-generation H1 antihistamines remain the first-line treatment; however, real-world data consistently demonstrate that a significant proportion of patients fail to achieve adequate disease control even with dose escalation, underscoring the limitations of uniform therapeutic algorithms [
19]. The introduction of anti-IgE therapy represented a major paradigm shift in CSU management. Omalizumab has demonstrated robust efficacy in a large subset of patients refractory to antihistamines, primarily through modulation of IgE-mediated mast cell and basophil activation; however, studies demonstrate the need to evaluate its safety in situations such as risks related to anaphylaxis, pregnancy, malignancy, cardiovascular events, and infections [
22,
30] Clinical and immunological heterogeneity, however, strongly influences treatment outcomes. Patients with elevated total IgE levels and features consistent with type I autoimmune (autoallergic) CSU generally exhibit favorable and rapid responses, whereas those with low IgE levels, basopenia, and functional evidence of IgG-mediated autoimmunity often display delayed, partial, or absent responses [
24,
25]. These observations highlight the importance of integrating immunological biomarkers into therapeutic decision-making rather than relying solely on clinical severity.
Despite its clinical success, anti-IgE therapy does not fully address the spectrum of pathogenic mechanisms involved in CSU. Disease relapse following treatment discontinuation and incomplete responses in subsets of patients reveal persistent unmet needs and reinforce the necessity of alternative and adjunctive therapeutic strategies [
8]. In this context, immunomodulatory agents such as cyclosporine remain relevant, particularly for patients with features suggestive of type IIb autoimmune CSU, where T-cell–mediated inflammation and autoantibody-driven mechanisms predominate [
24,
25].
Emerging therapeutic approaches increasingly target intracellular signaling pathways and upstream immune regulators involved in mast cell activation and inflammatory amplification. Bruton’s tyrosine kinase inhibitors have gained attention due to their ability to interfere with Fc receptor–mediated signaling in mast cells and basophils, offering a rational option for patients with refractory disease and autoimmune-associated endotypes [
23]. Similarly, biologics targeting cytokine networks and alarmins implicated in Th2- and Th17-driven inflammation are under investigation, reflecting a growing recognition of non–IgE-dependent pathways in CSU pathogenesis [
27]. The success of targeted biologics in CSU parallels therapeutic advances observed in other allergic disorders, including asthma and atopic dermatitis, supporting the concept that shared pathogenic pathways may be effectively modulated across distinct clinical phenotypes [
8,
22,
24].
These developments support a shift toward endotype-driven therapeutic algorithms, in which treatment selection is informed by immunological profiling rather than linear escalation alone. Biomarkers such as total IgE levels, basophil-related parameters, and markers of systemic inflammation and coagulation may help identify patients more likely to benefit from specific interventions, optimize sequencing of therapies, and reduce unnecessary exposure to ineffective treatments [
22,
28]. Although such approaches remain largely investigative, they represent a critical step towards personalized management that has already been highlighted as a potential for modern medicine [
31].
Looking forward, the expanding therapeutic pipeline underscores the need for integrative frameworks that align pathogenic mechanisms with targeted interventions. Rather than viewing CSU treatment as a stepwise escalation of symptom control, future strategies are likely to involve dynamic, mechanism-guided decision-making aimed at achieving sustained disease remission and improving long-term patient outcomes. Continued clinical trials and real-world studies incorporating biomarker-driven stratification will be essential to translate these advances into routine clinical practice.
6. Conclusions
Chronic spontaneous urticaria (CSU) should no longer be regarded as a homogeneous, purely histamine-mediated skin disorder. Accumulating evidence supports the concept that CSU represents a spectrum of immune-mediated conditions. Distinct but overlapping pathogenic mechanisms converge on mast cell activation and drive disease heterogeneity.. IgE-mediated autoallergy, IgG-mediated autoimmunity, and sustained inflammatory amplification involving Th2 and Th17 pathways collectively contribute to disease heterogeneity, chronicity, and variable therapeutic responses. The recognition of autoimmune and inflammatory endotypes has important clinical implications. It challenges the traditional symptom-based, stepwise management approach and highlights the limitations of uniform treatment escalation. Instead, a mechanistic framework integrating immune profiling and biomarker assessment offers a more rational basis for disease stratification and personalized therapeutic decision-making. Although no single biomarker is currently sufficient for routine clinical use, combined approaches incorporating total IgE levels, basophil-related parameters, and markers of inflammatory and coagulation pathways hold promise for refining patient classification and predicting treatment response. Therapeutic advances over the past decade have substantially expanded the treatment landscape of CSU, particularly with the introduction of targeted biologics and immunomodulatory strategies.
Nevertheless, heterogeneous responses and disease relapse after treatment discontinuation underscore persistent unmet needs and reinforce the importance of aligning therapeutic choices with underlying pathogenic mechanisms rather than clinical phenotype alone. In this context, reframing CSU as a mast cell–driven disorder shaped by distinct immune endotypes provides a unifying conceptual model that bridges basic immunology and clinical practice. Future research should prioritize the validation of biomarker-driven stratification strategies and the development of targeted interventions aimed at achieving durable disease control and improving patient-centered outcomes. Such advances are essential to move CSU management toward a truly personalized and mechanism-based paradigm.
Author Contributions
Conceptualization, M.J.M.P. and L.C.A.; methodology, M.J.M.P.; validation, M.J.M.P., L.C.A. and W.B.D.A.; formal analysis, M.J.M.P. and G.S.S.; investigation, M.J.M.P., M.O.M., R.F.D.P., V.P.S.C., G.P.S.F. and I.A.M.D.; resources, W.B.D.A. and T.T.S.; data curation, M.J.M.P., G.S.S. and S.D.S.C.H.; writing—original draft preparation, M.J.M.P.; writing—review and editing, M.J.M.P., L.C.A., M.O.M., R.F.D.P., G.S.S., S.D.S.C.H., I.A.M.D., T.T.S., J.F.P and W.B.D.A.; visualization, M.J.M.P. and V.P.S.C.; supervision, W.B.D.A.; project administration, M.J.M.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Acknowledgments
The authors would like to thank all colleagues and institutions whose work contributed indirectly to the development of this review. During the preparation of this manuscript, the authors used artificial intelligence–assisted tools to support language refinement, structural organization, and figure conceptualization. The authors reviewed, edited, and validated all content and take full responsibility for the accuracy and integrity of the information presented.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| ASST |
Autologous Serum Skin Test |
| BAT |
Basophil Activation Test |
| BTK |
Bruton’s Tyrosine Kinase |
| CU |
Chronic Urticaria |
| CSU |
Chronic Spontaneous Urticaria |
| EAACI |
European Academy of Allergy and Clinical Immunology |
| FcεRI |
High-Affinity IgE Receptor |
| GA²LEN |
Global Allergy and Asthma European Network |
| H1 |
Histamine Type 1 Receptor |
| IgE |
Immunoglobulin E |
| IgG |
Immunoglobulin G |
| MRGPRX2 |
Mas-Related G Protein-Coupled Receptor X2 |
| RCT |
Randomized Controlled Trial |
| Th2 |
T Helper Type 2 Cells |
| Th17 |
T Helper Type 17 Cells |
| WAO |
World Allergy Organization |
References
- Zuberbier, T.; et al. Chronic urticaria: Unmet needs, emerging drugs, and new perspectives on personalised treatment. Lancet. 2024, 404, 1218–1231. [Google Scholar] [CrossRef]
- Kaplan, A.P.; Giménez-Arnau, A.M.; Saini, S.S. Chronic spontaneous urticaria: Focus on pathophysiology to unlock treatment advances. Allergy. 2022, 78, 389–401. [Google Scholar] [CrossRef]
- Bernstein, J.A.; Lang, D.M.; Khan, D.A. Chronic spontaneous urticaria. Immunol. Allergy Clin. North Am. 2024, 44, 381–398. [Google Scholar] [CrossRef]
- Kolkhir, P.; Church, M.K.; Weller, K.; Metz, M.; Schmetzer, O.; Maurer, M. Autoimmune chronic spontaneous urticaria. J. Allergy Clin. Immunol. 2022, 149, 1819–1831. [Google Scholar] [CrossRef]
- Greiner, A.N. Pathophysiology, diagnosis, and management of chronic spontaneous urticaria: A literature review. Clin. Rev. Allergy Immunol. 2022, 64, 194–211. [Google Scholar] [CrossRef]
- Kim, S.H.; et al. Advances in pathophysiology and therapeutic paradigm shifts in chronic spontaneous urticaria. Dermatol. Ther. 2025, 15, 1–18. [Google Scholar] [CrossRef]
- He, A.; et al. Chronic urticaria: Advances in understanding of the disease and clinical management. Clin. Rev. Allergy Immunol. 2021, 61, 424–448. [Google Scholar] [CrossRef]
- Yosipovitch, G.; et al. Current and emerging therapies for chronic spontaneous urticaria: A narrative review. Dermatol. Ther. 2023, 13, 1771–1794. [Google Scholar] [CrossRef] [PubMed]
- Asero, R.; et al. Biologics for chronic spontaneous urticaria: Toward a personalized treatment. Expert Rev. Clin. Immunol. 2022, 18, 1229–1240. [Google Scholar] [CrossRef] [PubMed]
- Giménez-Arnau, A.M.; et al. Recent updates in urticaria. Med. Clin. (Barc.). 2023, 161, 311–317. [Google Scholar] [CrossRef]
- Melchers, S.; et al. Chronic spontaneous urticaria—Status quo and future. Allergo J. Int. 2023, 32, 229–238. [Google Scholar] [CrossRef]
- Yolcu, S.; et al. Current view of chronic spontaneous urticaria. Astım Allerji İmmünoloji. 2023, 21, 67–76. [Google Scholar] [CrossRef]
- Puxeddu, I.; et al. Mast cell signaling and its role in urticaria. Ann. Allergy Asthma Immunol. 2024, 133, 256–265. [Google Scholar] [CrossRef]
- Grimalt, R. Chronic spontaneous urticaria: A changing paradigm. Allergies 2026, 6, 4. [Google Scholar] [CrossRef]
- Asero, R. Therapies for chronic spontaneous urticaria: Present and future. Pharmaceuticals. 2024, 17, 1499. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, J.A.; et al. Biomarkers for monitoring treatment response of omalizumab in patients with chronic urticaria. Int. J. Mol. Sci. 2023, 24, 11328. [Google Scholar] [CrossRef] [PubMed]
- Yanase, Y.; et al. Basophil characteristics as a marker of the pathogenesis of chronic spontaneous urticaria. Int. J. Mol. Sci. 2023, 24, 10320. [Google Scholar] [CrossRef]
- Sardina, D.S.; Valenti, G.; Papia, F.; Uasuf, C.G. Exploring machine learning techniques to predict the response to omalizumab in chronic spontaneous urticaria. Diagnostics 2021, 11, 2150. [Google Scholar] [CrossRef]
- Calzari, P.; et al. Omalizumab for chronic spontaneous urticaria in adults and adolescents: An eight-year real-life experience. J. Clin. Med. 2024, 13, 5610. [Google Scholar] [CrossRef]
- Manzoor, H.; et al. Meta-analysis of omalizumab dosing regimens in chronic spontaneous urticaria. Healthcare 2022, 10, 2579. [Google Scholar] [CrossRef]
- Bernstein, J.A.; et al. Why a complete response is the treatment aim in chronic spontaneous urticaria. J. Clin. Med. 2023, 12, 3561. [Google Scholar] [CrossRef] [PubMed]
- Manti, S.; et al. Monoclonal antibodies in the treatment of chronic spontaneous urticaria. J. Clin. Med. 2022, 11, 4453. [Google Scholar] [CrossRef]
- Robak, E.; et al. Bruton’s tyrosine kinase inhibitors for the treatment of immunological diseases. J. Clin. Med. 2022, 11, 2807. [Google Scholar] [CrossRef]
- Rizzi, A.; et al. Alarmins in Chronic Spontaneous Urticaria: Immunological Insights and Therapeutic Perspectives. Biomedicines. 2024, 12, 2765. [Google Scholar] [CrossRef]
- Bartosik, B.; et al. Chronic Spontaneous Urticaria—New Predictor on the Horizon? J. Clin. Med. 2024, 13, 6812. [Google Scholar] [CrossRef]
- Georgescu, M.; et al. Beyond the Skin: Exploring the Gut–Skin Axis in Chronic Spontaneous Urticaria and Other Inflammatory Skin Diseases. Biomedicines. 2025, 13, 2014. [Google Scholar] [CrossRef]
- Galletta, F.; et al. Efficacy and Safety of Omalizumab and Dupilumab in Pediatric Patients with Skin Diseases: An Observational Study J. Pers. Med. 2025, 15, 64. [Google Scholar] [CrossRef] [PubMed]
- Jutel, M.; et al. Nomenclature of allergic diseases and hypersensitivity reactions: Adapted to modern needs: An EAACI position paper. Allergy. 2023, 78, 2851–2874. [Google Scholar] [CrossRef]
- Chiang, V.; et al. Disproportionate rate of female referrals for suspected COVID-19 vaccine allergies. Allergy. 2022, 78, 308–310. [Google Scholar] [CrossRef]
- Pongdee, T. Omalizumab safety concerns. J. Allergy Clin. Immunol. Pract. 2024, 12, 01183-7. [Google Scholar] [CrossRef]
- Mulani, A.O.; et al. Transforming Drug Therapy with Deep Learning: The Future of Personalized Medicine. Drug Res. 2025, 75, 326–333. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).