Submitted:
03 February 2026
Posted:
04 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
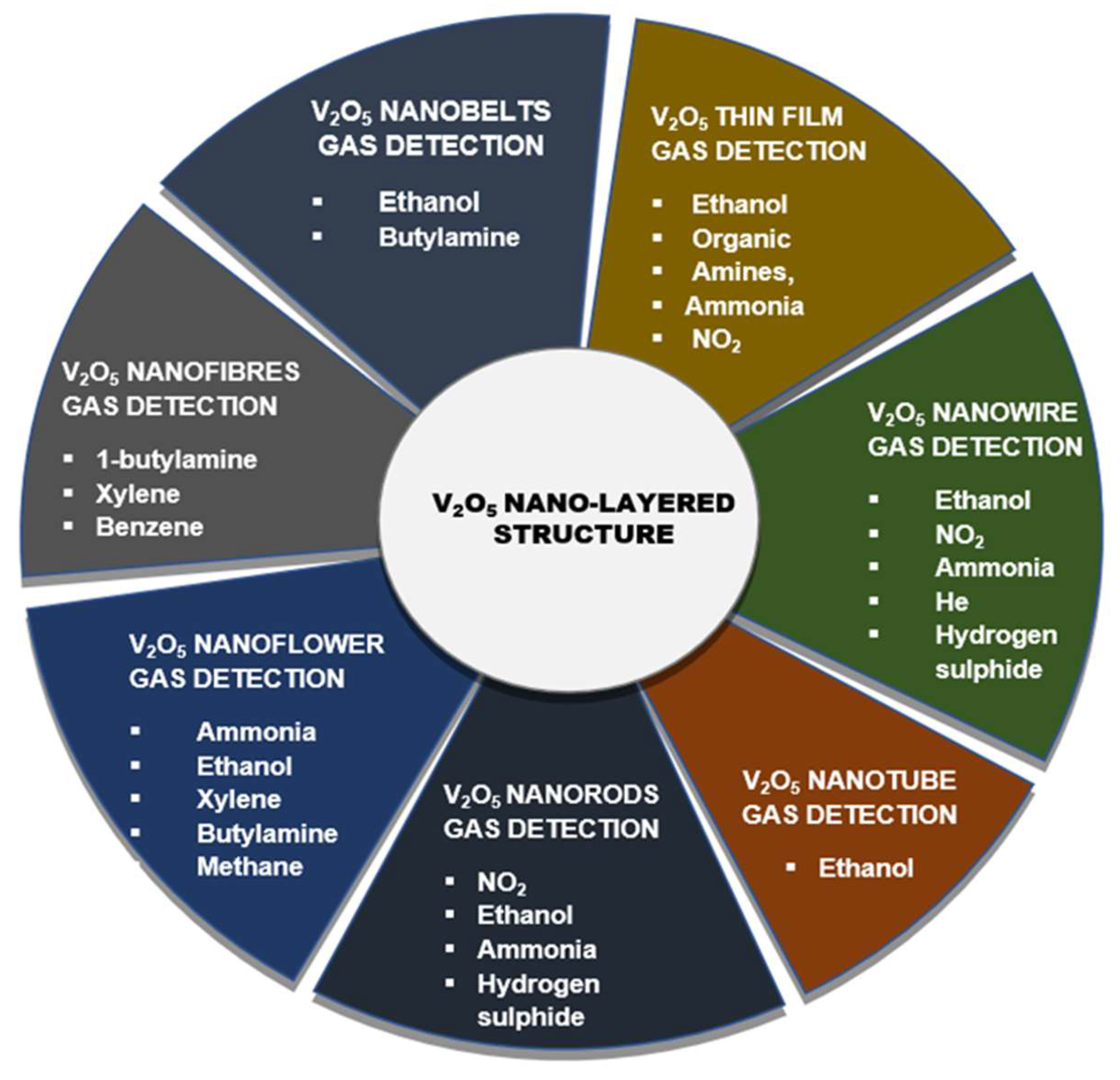
2. Hierarchical Nanostructures of V2O5
2.1. V2O5 Nanobelts
2.2. V2O5 Thin-Films
2.3. V2O5 Nanorods
2.4. V2O5 Nanofibers
2.5. V2O5 Nanoflowers
2.6. V2O5 nanowires
2.7. V2O5 Nanotubes
3. Density Functional Theory (DFT) Perspective
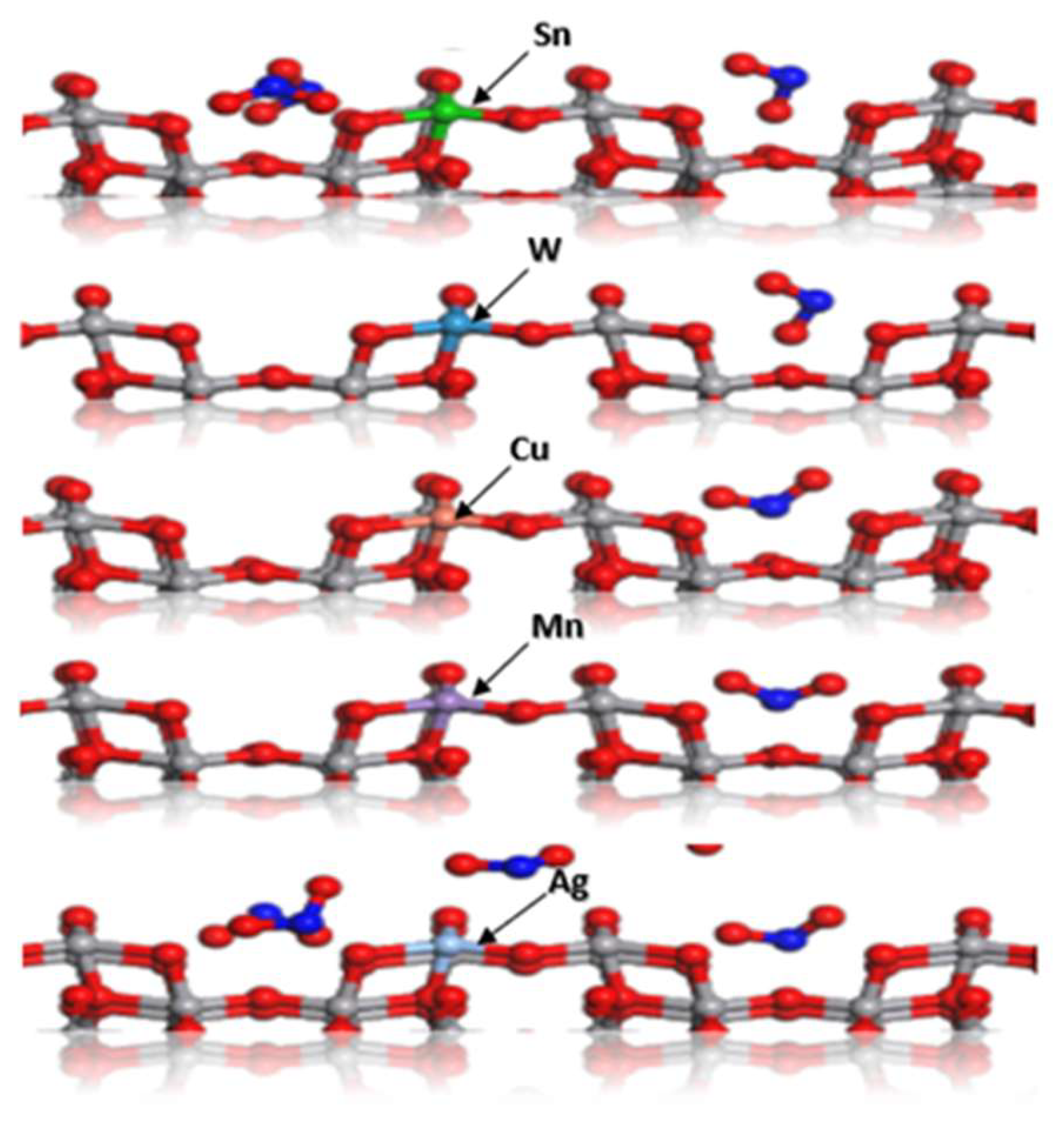
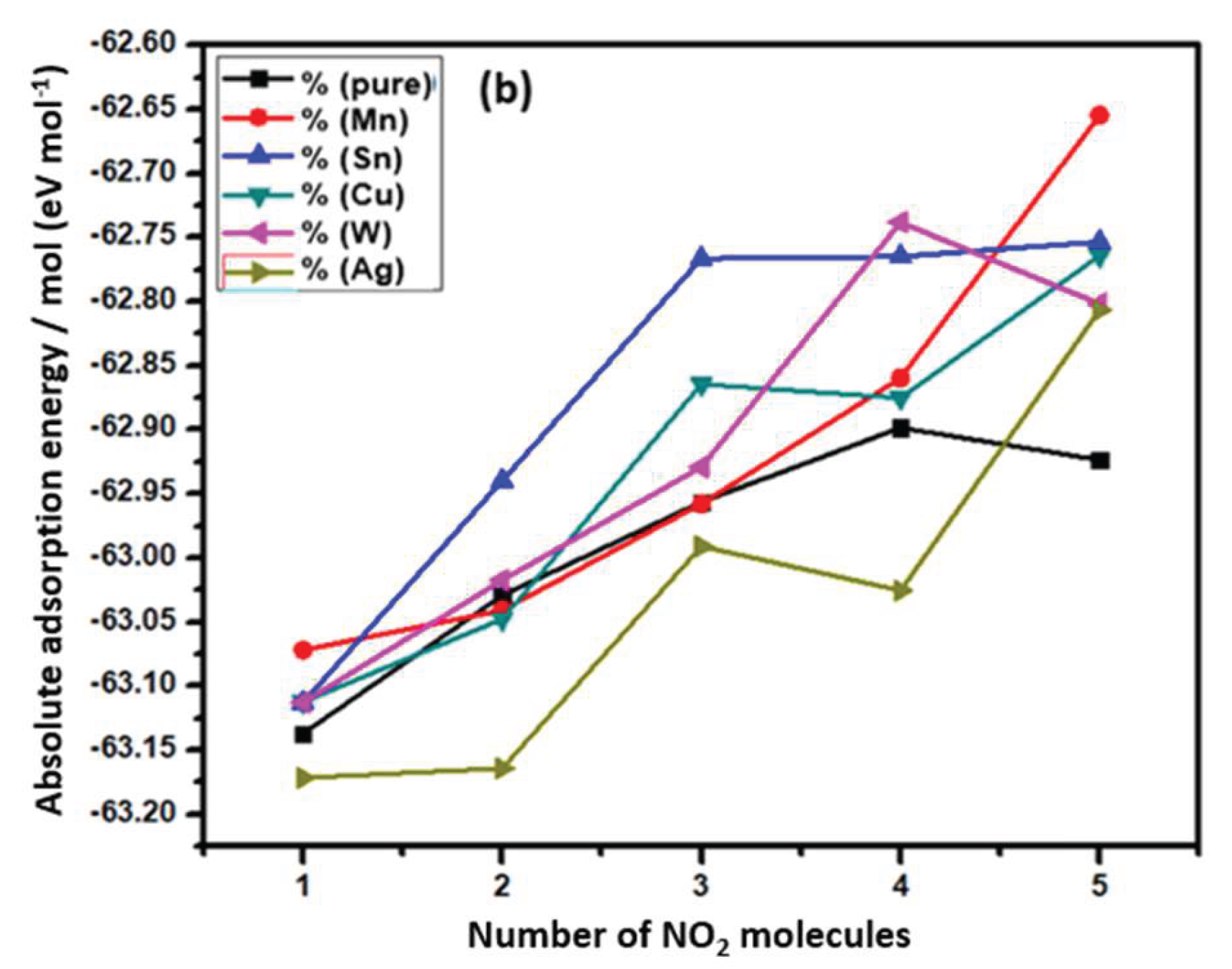
3.1. Density Functional Theory Study of Transition-Metal-Doped V₂O₅ for Enhanced Adsorption of NO₂
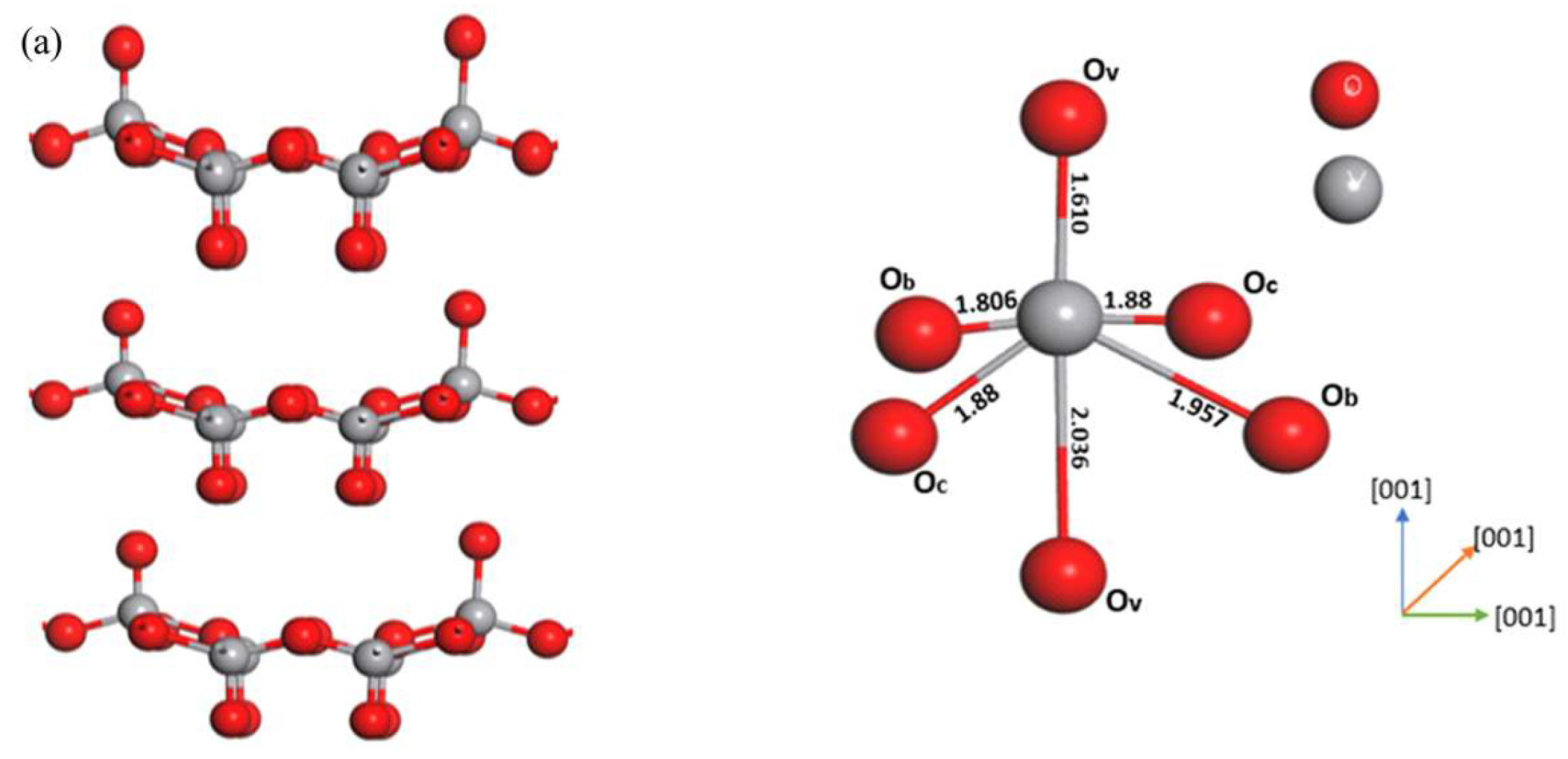
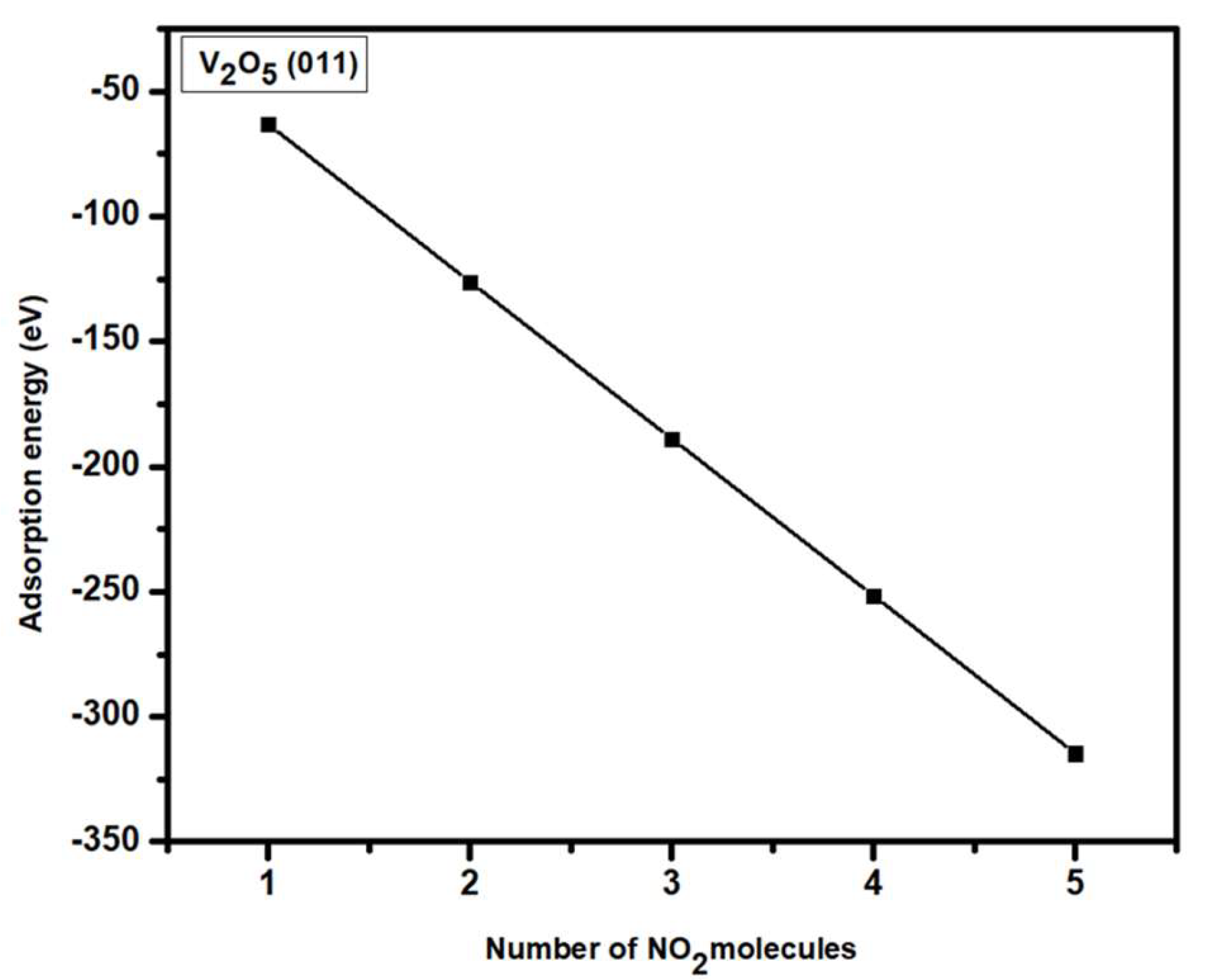
3.2. Adsorption of NO2 Molecule V2O5 (011) Surface
4. Conclusions
References
- Mane, A.A.; Suryawanshi, M.P.; Kim, J.H.; Moholkar, A.V. Highly selective and sensitive response of 30.5 % of sprayed molybdenum trioxide (MoO3) nanobelts for nitrogen dioxide (NO2) gas detection. Journal of Colloid and Interface Science 2016, vol. 483, 220–231. [Google Scholar] [CrossRef] [PubMed]
- Davies, M.; Hobbs, F.; Davis, R. Prevalence of left-ventricular systolic dysfunction and heart failure in the Echocardiographic Heart of England Screening study: a population based study. Lancet 2001, vol. 358, 439–44. [Google Scholar] [CrossRef]
- Redfi eld, M.M.; Jacobsen, S.J.; Burnett, J.C.; Mahoney, D.W.; Bailey, K.R.; Rodeheffer, R.J. Burden of systolic and diastolic ventricular dysfunction in the community: appreciating the scope of the heart failure epidemic. JAMA 2003, vol. 289, 194–202. [Google Scholar] [CrossRef]
- Nazar, A.S.; Majeed, G.; Murrawat, A.; Muhammad, A. Synthesis of Metal Oxide Semiconductor Nanostructures for Gas Sensors; intechopen, 2019. [Google Scholar]
- Naderi, H.; Hajati, S.; Ghaedi, M.; Dashtian, K.; Sabzehmeidani, M.M. Sensitive, selective and rapid ammonia-sensing by gold nanoparticle-sensitized V2O5/CuWO4 heterojunctions for exhaled breath analysis. Applied Surface Science 2020, vol. 501, 144270. [Google Scholar] [CrossRef]
- Roper, C.; Delgado, L.S.; Barrett, D.; Simonich, S.L.M.; Tanguay, R.L. PM2.5 filter extraction methods: implications for chemical and toxicological analyses. Environ. Sci. Technol. 2018, vol. 53, 434–442. [Google Scholar] [CrossRef] [PubMed]
- Roper, C.; Delgado, L.S.; Barrett, D.; Simonich, S.L.M.; Tanguay, R.L. PM2.5 filter extraction methods: implications for chemical and toxicological analyses. Environ. Sci. Technol 2018, vol. 53, 434–442. [Google Scholar] [CrossRef]
- Zhang, J.; Ouyang, Y.; Ye, Z.; Li, Q.; Lin, T.; Chen, Z.; Zhang, S. Mixed-valence cobalt (II/III) metal–organic framework for ammonia sensing with naked-eye color switching. ACS Appl. Mater. Interfaces 2018, vol. 10, 27465–27471. [Google Scholar] [CrossRef]
- Heiland, G. Zum Einfluß von Wasserstoff auf die elektrische Leitfähigkeit an der Oberfläche von Zinkoxydkristallen. Phys 1957, vol. 148, 15–27. [Google Scholar]
- Jeun, J.H.; Kim, D.H.; Hong, S.H. Synthesis of porous SnO2 foams on SiO2/Si substrate by electrochemical deposition and their gas sensing properties. Sens. Actuators B 2012, vol. 161, 784–790. [Google Scholar] [CrossRef]
- Liu, Y.; Hang, T.; Xie, Y.; Bao, Z.; Song, J.; Zhang, H.; Xie, E. Effect of Mg doping on the hydrogen-sensing characteristics of ZnO thin films. Sens. Actuators B 2011, vol. 160, 266–270. [Google Scholar] [CrossRef]
- Hassan, K.; Chung, G. Catalytically activated quantum size Pt/Pd bimetallic core-shell nanoparticles decorated on ZnO nanorod clusters for accelerated hydrogen gas detection. Sens. Actuators B 2017, vol. 239, 824–833. [Google Scholar] [CrossRef]
- Perfecto, T.M.; Zito, C.A.; Volanti, D.P. Design of nanostructured WO30.33H2O via combination of ultrasonic spray nozzle and microwave-assisted hydrothermal methods for enhancing isopropanol gas sensing at room temperature. Cryst. Eng. Comm. 2017, vol. 2733–2738, 19. [Google Scholar]
- Flak, D.; Braun, A.; Michalow, K.A.; Parlinska-Wojtan, J.W.M.; Graule, T.; Rekas, M. Differences in electrophysical and gas sensing properties of flame spray synthesized Fe2O3 (γ-Fe2O3 and α-Fe2O3). J. Nanosci. Nanotechnol. 2012, vol. 12, 6401–6411. [Google Scholar] [CrossRef] [PubMed]
- Zakrzewska, K.; Radecka, M.; Rekas, M. Effect of Nb, Cr, Sn additions on gas sensing properties of TiO2 thin. films. Thin Solid Films 1997, vol. 310, 161–166. [Google Scholar] [CrossRef]
- Lyson-Sypien, B.; Czapla, A.; Lubecka, M.; Gwizdz, P.; Schneider, K.; Zakrzewska, K.; Michalow, K.; Graule, T.; Reszka, A.; Rekas, M. Nanopowders of chromium doped TiO2 for gas sensors. Sens. Actuators B 2012, vol. 175, 163–172. [Google Scholar] [CrossRef]
- Qin, Y.; Guangtao, F.; Liu, K.; Ming, H. Vanadium pentoxide hierarchical structure networks for high performance ethanol gas sensor with dual working temperature characteristic. Sensors and Actuators B 2014, vol. 190, 141–148. [Google Scholar] [CrossRef]
- Wonbong, C.; Nitin, C.; Gang, H.; Han, J.; Park, D.; Young, H.L. Recent development of two-dimensional transition metal dichalcogenides and their applications. Materials Today 2017, vol. 20, 116. [Google Scholar] [CrossRef]
- Eunji, L.; Young, S.Y.; Dong, J. K. Two-Dimensional Transition Metal Dichalcogenides and Metal Oxide Hybrids for Gas Sensing. ACS Sens. 2018, vol. 3, 2045–2060. [Google Scholar]
- Liu, H.; Neal, A.T.; Zhu, Z.; Luo, Zhe.; Xu, X.; Toma´nek, D.; Ye, D.P. Phosphorene: An Unexplored 2D Semiconductor with a High Hole Mobility. ACS NANO 2014, vol. 8, 4033–4041. [Google Scholar] [CrossRef]
- Akande, A. A.; Linganiso, E.; CDhonge, B. P.; Rammutla, K. E.; Machatine, A.; Prinsloo, L.; Kunert, H.; Mwakikunga, B.W. Phase evolution of vanadium oxides obtained through temperature programmed calcinations of ammonium vanadate in hydrogen atmosphere and their humidity. Materials Chemistry and Physics 2015, vol. 151, 206–214. [Google Scholar] [CrossRef]
- Rao, M. C. Vanadium Pentoxide Cathode Material for Fabrication of all Solid State Lithium-Ion Batteries- a Case Study. Research Journal of Recent sciences 2013, vol. 2, 67–73. [Google Scholar]
- Reddy, I.N.; Sreedhar, A.; Reddy, C.V.; Cho, M.; Kim, D.; Shim, J. Facile synthesis and characterization of V2O5 nanobelt bundles containing plasmonic Ag for photoelectrochemical water splitting under visible light irradiation. Ceram. Int. 2019, vol. 45, 23333–23340. [Google Scholar] [CrossRef]
- Choi, Kyoung Jin; Jang, Ho Won. One-Dimensional Oxide Nanostructures as Gas-Sensing Materials: Review and Issues. sensors 2010, vol. 10, 4083–4099. [Google Scholar]
- Amiri, Vahid; Roshan, Hossein; Mirzaei, Ali; Sheikhi, Mohammad Hossein. A Review of Nanostructured Resistive-Based Vanadium Oxide Gas Sensors. Chemosensors 2020, vol. 8, 105. [Google Scholar] [CrossRef]
- Bouzidi, A.; Benramdane, N.; Bresson, S.; Mathieu, C.; Desfeux, R.; Marssi, M.E. X-ray and raman study of spray pyrolysed vanadium oxide thin films. Vib. Spectrosc. 2011, vol. 57, 182–186. [Google Scholar] [CrossRef]
- Su, Q.; Lan, W.; Wang, Y.Y.; Liu, X.Q. Structural characterization of β-v2o5 films prepared by dc reactive magnetron sputtering. Appl. Surf. Sci. 2009, vol. 255, 4177–4179. [Google Scholar] [CrossRef]
- Mwakikunga, B.W.; Sideras-Haddad, E.; Forbes, A.; Arendse, C.J. Raman spectroscopy of WO3 nano-wires and thermo-chromism study of VO2 belts produced by ultrasonic spray and laser pyrolysis techniques Phys. Status Solidi 2008, vol. 205, 150–4. [Google Scholar] [CrossRef]
- Schneider, k.; Maziarz, W. V2O5 Thin Films as Nitrogen Dioxide Sensors. sensors 2018, vol. 18, 4177. [Google Scholar] [CrossRef]
- Eunji, L.; Young, S.Y.; Dong, J.K. Two-Dimensional Transition Metal Dichalcogenides and Metal Oxide Hybrids for Gas Sensing. ACS Sens. 2018, vol. 3, 2045–2060. [Google Scholar]
- Mak, K.F.; Lee, C.; Hone, J.; Shan, J.; Heinz, T.F. Atomically Thin MoS2. Phys. Rev. Lett. 2010, vol. 105, 136805. [Google Scholar] [CrossRef]
- Liu, H.; Neal, A.T.; Zhu, Z.; Luo, Z.; Xu, X.; Tomanek, D.; Ye, P.D. Phosphorene: An unexplored 2D semiconductor with a high hole mobility. ACS Nano 2014, vol. 8, 4033–4041. [Google Scholar] [CrossRef]
- Liu, X.; Ma, T.; Pinna, N.; Zhang, J. Two-Dimensional Nanostructured Materials for Gas Sensing. Adv. Funct. Mater. 2017, vol. 27, 1702168. [Google Scholar] [CrossRef]
- Joshi, N.; Hayasaka, T.; Liu, Y.; Liu, H.; Oliveira, O.N., Jr.; Lin, L. A review on chemiresistive room temperature gas sensors based on metal oxidenanostructures, graphene and 2D transition metal dichalcogenides. Microchim. Acta 2018, vol. 185, 213. [Google Scholar] [CrossRef]
- Mhlongo, G.H.; Motaung, D.E.; Nkosi, S.S.; Swart, H.C.; Gerald, F.; Malgas, K.T.; Mwakikunga, B.W. Temperature-dependence on the structural, optical, and paramagnetic properties of ZnO nanostructures. Applied Surface Science 2014, vol. 293, 62–70. [Google Scholar] [CrossRef]
- Motuang, D. E.; Mhlongo, G. H.; Nkosi, S.S.; Malgas, G. F.; Mwakikunga, B. W.; Coetsee, E; Swart, H. C.; Aballah, H. M. I.; Moyo, T.; Ray, S. S. Shape-Selective Dependence of Room Temperature Ferromagnetism Induced by Hierarchical ZnO Nanostructures. Applied material & interfaces 2014, vol. 6, 8981–8995. [Google Scholar]
- Raseputuka, S.V.; Malwela, T.; Lemmer, Y.; Mervyn, B.; Mwakikunga, B.W. The hierarchical nanostructured Co-doped WO3/carbon and their improved acetone sensing performance. Materials Science in Semiconductor Processing 2020, vol. 117, 105157. [Google Scholar]
- Xu, L.; Zeng, W.; Li, Y. Synthesis of morphology and size-controllable SnO2 hierarchical structures and their gas-sensing performance. Applied Surface Science 2018, vol. 457, 1064–1071. [Google Scholar] [CrossRef]
- Xu, W.; Qiu, C.; Zhou, J.; Chen, Y. Regulation of specific surface area of 3D flower-like WO3 hierarchical structures for gas sensing application. Ceramics International 2020, vol. 46, 11372–11378. [Google Scholar] [CrossRef]
- Xu, L.; Zeng, W.; Li, Y. Synthesis of morphology and size-controllable SnO 2 hierarchical structures and their gas-sensing performance. Applied Surface Science 2018, vol. 457, 1064–1071. [Google Scholar] [CrossRef]
- Lee, J.H. Gas sensors using hierarchical and hollow oxide nanostructures: overview. Sens. Actuators B 2009, vol. 140, 319–336. [Google Scholar] [CrossRef]
- Bing, Y.; Zeng, Y.; Liu, C.; Qiao, L.; Sui, Y.; Zou, B.; Zheng, W.; Zou, G. Assembly of hierarchical ZnSnO3 hollow microspheres from ultra-thin nanorods and the enhanced ethanol-sensing performances. Sensors and Actuators B: Chemical 2014, vol. 190, 370–377. [Google Scholar] [CrossRef]
- Zhu, L.; Zeng, W.; Li, Y. New insight into gas sensing property of ZnO nanorods and nanosheets. Materials Letters 2018, vol. 228, 331–333. [Google Scholar] [CrossRef]
- Choi, K.; Kim, H.R.; Lee, J.H. Enhanced CO sensing characteristics of hierarchical and hollow In2O3 microspheres. Sensors and Actuators B: Chemical 2009, vol. 138, 497–503. [Google Scholar] [CrossRef]
- Yan, W.J.; Hu, M.; Zeng, P.; Ma, S.Y.; Li, M.D. Room temperature NO2-sensing properties of WO3 nanoparticles/porous silicon. Appl. Surf. Sci. 2014, vol. 292, 551–55. [Google Scholar] [CrossRef]
- Kanungo, J.; Saha, H.; Basu, S. Pd sensitized porous silicon hydrogen sensor –influence of ZnO thin film. Sens. Actuators B: Chem. 2010, vol. 147, 128–136. [Google Scholar] [CrossRef]
- Yan, D.; Hu, M.; Li, S.; Liang, J.; Wu, Y.; Ma, S. Electrochemical deposition of ZnO nanostructures onto porous silicon and their enhanced gas sensing to NO2 at room temperature. Electrochim. Acta 2014, vol. 115, 297–305. [Google Scholar] [CrossRef]
- Yan, W.; Hu, M.; Wang, D.; Li, C. Room temperature gas sensing properties of porous silicon/V2O5 nanorods composite. Applied Surface Science 2015, vol. 346, 216–222. [Google Scholar] [CrossRef]
- Pan, Z. W.; Dai, Z. R.; Wang, Z. L. Lead oxide nanobelts and phase transformation induced by electron bean irradiation. APPLIED PHYSICS LETTERS 2002, vol. 80, 2. [Google Scholar] [CrossRef]
- Rane, AV.; Kanny, K.; Abitha, VK.; Thomas, S. Methods for synthesis of nanoparticles and fabrication of nanocomposites; Elsevier Ltd., 2018. [Google Scholar]
- Lui, J.; Wang, X.; Peng, Q.; Li, Y. Vanadium Pentoxide Nanobelts: Highly Selective and Stable Ethanol Sensor Materials. Adv. mater. 2005, vol. 17, 6. [Google Scholar]
- Liu, J.; Wang, X.; Peng, Q.; Li, Y. Preparation and gas sensing properties of vanadium oxide nanobelts coated with semiconductor oxides. Sensors and Actuators B 2006, vol. 115, 481–487. [Google Scholar] [CrossRef]
- Liu, J.; Wang, X.; Peng, Q.; Li, Y. Preparation and gas sensing properties of vanadium oxide nanobelts coated with semiconductor oxides. Sensors and Actuators B 2006, vol. 115, 481–487. [Google Scholar] [CrossRef]
- Shi, S.; Cao, M.; He, X.; Xie, H. Surfactant-Assisted Hydrothermal Growth of Single-Crystalline Ultrahigh-Aspect-Ratio Vanadium Oxide Nanobelts. Crystal Growth & Design 2007, vol. 7, 9. [Google Scholar]
- Chen, H.; Di, J.; Wang, N.; Dong, H.; Wu, J.; Zhao, Y. Small. small 2011, vol. 7, 1779. [Google Scholar] [CrossRef]
- Zeng, W.; Chen, W.; Li, Z.; Zhang, He.; Li, T. Rapid and sensitive ethanol sensor based on hollow Au/V2O5 nanotubes via emulsion-electrospinning route. Materials Research Bulletin 2015, vol. 65, 157–162. [Google Scholar] [CrossRef]
- Liu, J.; Wang, X.; Peng, Q.; Li, Y. Preparation and gas sensing properties of vanadium oxide nanobelts coated with semiconductor oxides. Sensors and Actuators B 2006, vol. 115, 481–487. [Google Scholar] [CrossRef]
- Fu, H.; Xie, H. Hydrothermal synthesis of silver vanadium oxide (Ag0.35V2O5) nanobelts for sensing amines. Nanoscale Res Lett. 2015, vol. 10, 411. [Google Scholar] [CrossRef] [PubMed]
- Qiang, X.; Hu, M.; Zhou, L.; Liang, J. Pd nanoparticles incorporated porous silicon/V2O5nanopillars and their enhanced p-type NO2-sensing properties at room temperature. Mater Lett. 2018, vol. 231, 194–197. [Google Scholar] [CrossRef]
- Moshfegh, A. Z.; Ignatiev, A. Thin Solid Films 1991, vol. 198, 253. [CrossRef]
- Vernardou, D.; Paterakis, P.; Drosos, H.; Spanakis, E.; Povey, I.M.; Pemble, M.E.; Koudoumas, E.; Katsarakis, N. A study of the electrochemical performance of vanadium oxide thin films grown by atmospheric pressure chemical vapour deposition. Solar Energy Materials & Solar Cells 2011, vol. 95, 2842–2847. [Google Scholar]
- Vernardou, D.; Paterakis, P.; Drosos, H.; Spanakis, E.; Povey, I.M.; Pemble, M.E.; Koudoumas, E.; Katsarakis, N. A study of the electrochemical performance of vanadium oxide thin films grown by atmospheric pressure chemical vapour deposition. Solar Energy Materials and Solar Cells 2011, vol. 95, 2842–2847. [Google Scholar] [CrossRef]
- Gopalakrishnan, G.; Ramanathan, S. Compositional and metal–insulator transition characteristics of sputtered vanadium oxide thin films on yttria-stabilized zirconia. Journal of Materials Science 2011, vol. 46, 5768–5774. [Google Scholar] [CrossRef]
- Givernaud, J.; Crunteanu, A.; Orlianges, J.C.; Pothier, A.; Champeaux, C.; Catherinot, A.; Blondy, P. Microwave power limiting devices based on the semiconductor–metal transition in vanadium–dioxide thin films. IEEE Transactions on Microwave Theory and Techniques, 2010; pp. pp. 2352–2361. [Google Scholar]
- Ramana, C.V.; Smith, R.J.; Hussain, O.M.; Julien, C.M. On the growth mechanism of pulsed-laser deposited vanadium oxide thin films. Materials Science and Engineering B 2004, vol. 111, 218–225. [Google Scholar] [CrossRef]
- Huotari, J.; Lappalainen, J.; Puustinen, A.; Spetz, L. Gas sensing properties of pulsed laser deposited vanadium oxide thin films with various crystal structures. Sensors and Actuators B: Chemical 2013, vol. 187, 386–394. [Google Scholar] [CrossRef]
- Ramana, C.V.; Smith, R.J.; Hussain, O.M.; Julien, C.M. On the growth mechanism of pulsed-laser deposited vanadium oxide thin films. Materials Science and Engineering B 2004, vol. 111, 218–225. [Google Scholar] [CrossRef]
- Mane, AA.; Suryawanshi, M.P.; Kim, J.H.; Moholkar, A.V. Superior selectivity and enhanced response characteristics of palladium sensitized vanadium pentoxide nanorods for detection of nitrogen dioxide gas. J Colloid Interface Sci. 2017, vol. 495, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Vahid, A.; Hossein, R.; Ali, M.; Mohammad, H.S. A Review of Nanostructured Resistive-Based Vanadium Oxide Gas Sensors. Chemosensors 2020, vol. 8, 105. [Google Scholar] [CrossRef]
- Wang, X.Z.; Qiu, S.; He, C.Z.; Lu, G.X.; Liu, W.; Liu, J.R. RSC Adv 2013, vol. 3, 19002. [CrossRef]
- Schneider, K.; Lubecka, M.; Czapla, A. V2O5 thin films for gas sensor applications. Sens. Actuators B: Chem. 2016, vol. 236, 970–977. [Google Scholar] [CrossRef]
- Vijayakumar, y. V2O5 nanofibers: potential contestant for high performance xylene sensor. J. Alloy. Compd 2018, vol. 731, 85–812. [Google Scholar] [CrossRef]
- Alrammouz, R.; Lazerges, M.; Pironon, J.; Bin Taher, I.; Randi, A.; Halfaya, Y.; Gautier, S. V2O5 gas sensors: A review. Sensors and Actuators: A. Physical 2021, vol. 332, 13179. [Google Scholar] [CrossRef]
- Rizzo, G.; Arena, A.; Bonavita, A.; Donato, N.; neri, G.; Saitt, G. Gasochromic response of nanocrystalline vanadium pentoxide films deposited from ethanol dispersions. Thin Solid Films 2010, vol. 518, 7124–7127. [Google Scholar] [CrossRef]
- Schneider, K.; Lubecka, M.; Czapla, A. VOx thin films for gas sensor applications. Procedia Engineering 2015, vol. 120, 1153–1157. [Google Scholar] [CrossRef]
- Yu, D.; Zhang, S.; Liu, D.; Zhou, X.; Xie, S.; Zhang, Q.; Liu, Y.; Cao, G. Effect of manganese doping on Li-ion intercalation properties of V2O5 films, J. Mater. Chem. 2010, vol. 20, 10841–10846. [Google Scholar]
- Huotari, J.; Bjorklund, R.; Lappalainen, J; Lloyd Spetz, A. Pulsed Laser Deposited Nanostructured Vanadium Oxide Thin Films Characterized as Ammonia Sensors. Sensors and Actuators B: Chemical 2015, vol. 217, 22–29. [Google Scholar] [CrossRef]
- Dhayal Raj, A.; Pazhanivel, T.; Suresh Kumar, P.; Mangalaraj, D.; Nataraj, D.; Ponpandian, N. Self assembled V2O5 nanorods for gas sensors. Current Applied Physics 2010, vol. 10, 531–537. [Google Scholar] [CrossRef]
- Ostermann, R.; Li, D.; Yin, Y.; Jesse, T.; Xia, M. Y. V2O5 Nanorods on TiO2 Nanofibers: A New Class of Hierarchical Nanostructures Enabled by Electrospinning and Calcination. Nano Letters 2006, vol. 6, 1297–1302. [Google Scholar] [CrossRef]
- Huang, M.; Mao, S.; Feick, H.; Yan, H.; Wu, Y.; Kind, H.; Weber, E.; Russo, R.; Yang, P. D. Science 2001, vol. 292, 1897. [CrossRef]
- Mane, A.A.; Suryawanshi, M.P.; Kim, J.H.; Moholkar, A.V. Superior selectivity and enhanced response characteristics of palladium sensitized vanadium pentoxide nanorods for detection of nitrogen dioxide gas. Journal of Colloid and Interface Science 2017, vol. 495, 53–60. [Google Scholar] [CrossRef]
- Nirav, J.; Takeshi, H.; Yumeng, L.; Huiliang, L.; Osvaldo, N.; Oliveira, J.R.; Liwei, L. A review on chemiresistive room temperature gas sensors based on metal oxide nanostructures, graphene and 2D transition metal dichalcogenides. Microchim Acta 2018, vol. 185, 213. [Google Scholar]
- Abdul Hakim, S.; Liu, Y.; Lu, Y.; Chen, W. Room temperature highly selective ethanol sensing behavior of hydrothermally prepared Te-V2O5 nanorod nanocomposites. Mater Sci Semicond Process. 2015, vol. 31, 160–638. [Google Scholar] [CrossRef]
- Jimenez, V.M.; Espinos, J.P.; Gonzalez-Elipe, A.R. Effect of texture and annealing treatments in SnO and PdrSnO gas sensor materials. Sensors and Actuators B 1999, vol. 61, 23–32. [Google Scholar] [CrossRef]
- Ho, Y.K. Characterization of gasochromic vanadium oxides films by X-ray absorption spectroscopy, 6th Int. Conf. Technol.,". Adv. Thin Films Surf. Coat. 2013, vol. 544, 461–465. [Google Scholar] [CrossRef]
- Li, Y.; Huang, Z.; Rong, S. A vanadium oxide nanotube-based nitric oxide gas sensor. Sens. Mater. 2006, vol. 18, 241–249. [Google Scholar]
- Dong, Y.; Wei, H.; Liu, W.; Liu, Q.; Zhang, W.; Yang, Y. Template-free synthesis of V2O5 hierarchical nanosheet-assembled microspheres with excellent cycling stability. Journal of Power Sources 2015, vol. 285, 538–542. [Google Scholar] [CrossRef]
- Gibson, P.W.; Schreuder-Gibson, H.L.; Riven, D. AIChE J. 1999, vol. 45, 190. [CrossRef]
- Katti, D.S.; Robinson, K.W.; Attawia, M.A.; Ko, F.K.; Laurencin, C.T. Society for Biomaterials 28th Annual. Mtg Transactions, 2002; p. 143. [Google Scholar]
- Muster, J.; Kim, G. T.; Krstic, V.; Park, J. G.; Park, Y. W.; Roth, S.; Burghard, M. Adv. Mater. vol. 12, 420, 200.
- Raible, I.; Burghard, M.; Schlecht, U.; Yasuda, A.; Vossmeyer, T. V2O5 nanofibres: novel gas sensors with extremely high sensitivity and selectivity to amines. Sensors and Actuators B: Chemical 2005, vol. 106, 730–735. [Google Scholar]
- Li, D.; McCann, J.T.; Xia, Y.N.; Marquez, M. Electrospinning: a simple and versatile technique for producing ceramic nanofibers and nanotubes. J. Am. Ceram. Soc. 2006, vol. 89, 1861–1869. [Google Scholar] [CrossRef]
- Li, D.; Xia, Y.N. Electrospinning of nanofibers: reinventing the wheel. Adv. Mater. 2004, vol. 16, 1151–1170. [Google Scholar] [CrossRef]
- Patil, J.V.; Mali, S.S.; Kamble, A.S. a versatile technique for making of 1D growth of nanostructured nanofibers and its applications: an experimental approach. Appl Surf Sci. 2017, vol. 423, 641–674. [Google Scholar] [CrossRef]
- Muster, J.; Kim, G.T.; Krstic, V.; Park, J.G.; Park, Y.W.; Roth, S.; Burghard, M. Electrical transport through individual vanadium pentoxide nanowires. Adv. Mater. 2000, vol. 12, 420–424. [Google Scholar] [CrossRef]
- Modafferi, v. Highly sensitive ammonia resistive sensor based on electrospun v2o5 fibers. Sens. Actuators B: chem 2012, vol. 163, 61–68. [Google Scholar] [CrossRef]
- Feng, C; Li, X.; Wang, c. Facile synthesis benzene sensor based on V2O5-doped SnO2 nanofibers. RSC Adv. 2014, vol. 4, 47549–47555. [Google Scholar] [CrossRef]
- Viswanathamurthi, P.; Bhattarai, N.; Kim, H.Y.; Lee, D.R. Vanadium pentoxide nanofibers by electrospinning. Scripta Materialia 2003, vol. 49, 577–581. [Google Scholar] [CrossRef]
- Huang, M.H.; Wu, Y.Y.; Feick, H.; Tran, N.; Weber, E.; Yan, P.D. Catalytic growth of zinc oxide nanowires by vapor transport. Adv. Mater. 2001, vol. 13, 113–116. [Google Scholar] [CrossRef]
- Mounasamy, V.; Mani, G.K.; Ponnusamy, D.; Tsuchiya, K.; Prasad, A.K.; Madanagurusamy, S. Template-free synthesis of vanadium sesquioxide (V2O3) nanosheets and their room-temperature sensing performance. J. Mater. Chem. A. 2018, vol. 6, 6402–6413. [Google Scholar] [CrossRef]
- Qin, Y.; Fan, G.; Liu, K.; Hu, M. Vanadium pentoxide hierarchical structure networks for high performance ethanol gas sensor with dual working temperature characteristic. Sensors and Actuators B 2014, vol. 190, 141–148. [Google Scholar] [CrossRef]
- Pradhan, M.; Roy, A.; Sinha, A.K.; Sahoo, R.; Deb, D.; Pal, T. Solid-state transformation of single precursor vanadium complex nanostructures to V2O5 and VO2 : catalytic activity of V2O5 for oxidative coupling of 2-naphthol. Dalton Trans. 2015, vol. 44, 1889–1899. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Li, J.; Qiao, X.; Huang, J.; Ouyang, H. V2O5 nanoflowers assembled by nanorods as cathode material for lithium-ion batteries. Micro & Nano Lett. 2015, vol. 10, 686–688. [Google Scholar]
- Tang, Y.; Rui, X.; Zhang, Y.; Lim, T.M.; Dong, Z.; Hng, H.H.; Chen, X.; Yan, Q.; Chen, Z. Vanadium pentoxide cathode materials for high-performance lithium-ion batteries enabled by a hierarchical nanoflower structure via an electrochemical process. J. Mater. Chem. A. 2013, vol. 1, 82–88. [Google Scholar] [CrossRef]
- O‘Dwyer, C.; Navas, D.; Lavayen, V.; Benavente, E.; Santa Ana, M.A.; Gonzalez, G.; Newcomb, S.B.; Sotomayor Torres, C.M. Nano-urchin: the formation and structure of high-density spherical clusters of vanadium oxide nanotubes. Chem. Mater. 2006, vol. 18, 3016–3022. [Google Scholar] [CrossRef]
- Kaur, R.; Bhardwaj, S.K.; Kim, K.H.; Deep, A.; Vellingiri, K.; Kukkar, D. Nano-materials for sensing of formaldehyde in air: principles, applications, and performance evaluation. Nano Res. 2018, vol. 12, 225–246. [Google Scholar]
- Qin, Y.; Fan, G.; Liu, K.; Hu, M. Vanadium pentoxide hierarchical structure networks for high performance ethanol gas sensor with dual working temperature characteristic. Sensors Actuators B Chem 2014, vol. 190, 141–148. [Google Scholar] [CrossRef]
- Vijayakumar, Y.; Mani, G.K.; Reddy, M.V.R.; Rayappan, J.B.B. Nanostructured flower like V2O5 thin films and its room temperature sensing characteristics. Ceram Int 2015, vol. 41, 2221–2227. [Google Scholar] [CrossRef]
- Vijayakumar, Y.; Mani, G.K.; Reddy, M.V.R.; Rayappan, J.B.B. Nanostructured flower like V2O5 thin films and its room temperature sensing characteristics. Ceram Int 2015, vol. 41, 2221. [Google Scholar] [CrossRef]
- Yang, T.; Yu, H.; Xiao, B. Enhanced 1-butylamine gas sensing characteristics of flower-like V2O5 hierarchical architectures. J Alloys Compd 2017, vol. 699, 921–927. [Google Scholar] [CrossRef]
- Veena, M.; Ganesh, K.M.; Dhivya, P.; Kazuyoshi, T.; Reshma, P.R.; Arun, K.; Sridharan, M. Investigation on CH4 sensing characteristics of hierarchical V2O5 nanoflowers operated at relatively low temperature using chemiresistive approach. Analytica Chimica Acta 2020, vol. 1106, 148–160. [Google Scholar] [CrossRef]
- Liu, J.; Wang, X.; Peng, Q.; Li, Y. Preparation and gas sensing properties of vanadium oxide nanobelts coated with semiconductor oxides. Sens. Actuators B 2006, vol. 115, 481–487. [Google Scholar] [CrossRef]
- Yang, T.; Yu, H.; Xiao, B.; Li, Z.; Zhang, M. Enhanced 1-butylamine gas sensing characteristics of flower-like v2o5 hierarchical architectures. J. Alloys Compd. 2017, vol. 699, 921–927. [Google Scholar] [CrossRef]
- Liu, J.; Wang, X.; Peng, Q.; Li, Y. Vanadium Pentoxide Nanobelts:Highly Selective and Stable Ethanol Sensor Materials. Adv. Mater. 2005, vol. 17, 764–767. [Google Scholar] [CrossRef]
- Liu, J.; Wang, X.; Peng, Q.; Li, Y. Preparation and Gas Sensing Properties of Vanadium Oxide Nanobelts Coated with Semiconductor Oxides. Sens. Actuators, B 2006, vol. 115, 481–487. [Google Scholar] [CrossRef]
- Mai, L.; Xu, L.; Han, C.; Xu, X.; Luo, Y.; Zhao, S.; Zhao, Y. Electrospun Ultralong Hierarchical Vanadium Oxide Nanowires with High Performance for Lithium Ion Batteries. Nano Lett. 2010, vol. 10, 4750–4755. [Google Scholar] [CrossRef]
- Ramgir, N.; Datta, N.; Kaur, M.; Kailasaganapathi, S.; Debnath, A. K.; Aswal, D. K.; Gupta, S. K. Metal Oxide Nanowires for Chemiresistive Gas Sensors: Issues, Challenges and Prospects. Colloids Surf. A 2013, vol. 439, 101–106. [Google Scholar] [CrossRef]
- Tan, Ha Minh; Hung, Chu Manh; Minh Ngoc, Trinh; Nguyen, Hugo; Duc Hoa, Nguyen; Van Duy, Nguyen; Van Hieu, Nguyen. Novel Self-Heated Gas Sensors Using on-Chip Networked Nanowires with Ultralow Power Consumption. ACS Applied Materials & Interfaces 2017, vol. 9, 6153−6162. [Google Scholar]
- Jin, W.; Yan, S. Enhancement of ethanol gas sensing response based on ordered V2O5 nanowire microyarns. Sensors Actuators B Chem 2015, vol. 206, 284–290. [Google Scholar] [CrossRef]
- Mane, A.A.; Moholkar, A.V. Effect of film thickness on NO2 gas sensing properties of sprayed orthorhombic nanocrystalline V2O5 thin films. Appl. Surf. Sci. 2017, vol. 416, 511–520. [Google Scholar] [CrossRef]
- Yu, H.Y.; Kang, B.H.; Pi, U.H. V2O5 nanowire-based nanoelectronic devices for helium detection. Appl Phys Lett 2005, vol. 86, 1–3. [Google Scholar] [CrossRef]
- Yu, H.Y.; Kang, B.H.; Pi, U.H.; Park, C.W.; Choi, S.Y.; Kim, G.T. V2O5 nanowire-based nanoelectronic devices for helium detection. Appl. Phys. Lett. 2006, vol. 86, 25. [Google Scholar]
- Yeh, B.Y.; Jian, B.S.; Wang, G.J.; Tseng, W.J. CuO/V2O5 hybrid nanowires for highly sensitive and selective H2S gas sensor. RSC Adv 2017, vol. 7, 49605–49612. [Google Scholar] [CrossRef]
- Niranjan, R.; Niyanta, D.; Manmeet, K.S.; Kailasaganapathi, A.K.; Debnath, D.K.; Aswal, S.K.G. Metal oxide nanowires for chemiresistive gas sensors: Issues, challenges and prospects. Colloids and Surfaces A: Physicochem. Eng. Aspects 2013, vol. 439, 101–116. [Google Scholar]
- Wu, J.; Xing, X.; Zhu, Z.; Zheng, L.; Chen, J.; Wang, C.; Yang, D. Electrospun hollow CuO modified V2O5 nano-string of pearls with improved acetone sensitivity. Chemical Physics Letters 2019, vol. 727, 19–24. [Google Scholar] [CrossRef]
- Shahid, Muhammad; Shakir, Imran; Yang, Seok-Jo; Kang, Dae Joon. Facile synthesis of core–shell SnO2/V2O5 nanowires and their efficient photocatalytic property. Materials Chemistry and Physics 2010, vol. 124, 619–622. [Google Scholar] [CrossRef]
- Saliman, A.; Reza, H.; Aghabozorg, S.K. Synthesis and Characterization of Zr-Doped Vanadium Oxide Nanotubes. American Journal of Chemical Engineering 2018, vol. 6, 49–53. [Google Scholar] [CrossRef]
- Kanttcheva, M. Physical Chemistry Chemical Physics; 2000; p. pp. 3-43-3048. [Google Scholar]
- Spahr, M.E.; Bitterli, P.; Nesper, R.; Muller, M.; Krumeich, F.; Nissen, H.U. Chem. Int. Ed. 1998, vol. 37, 1263. [CrossRef]
- Lavayen, V.; O’Dwyer, C.; Santa Ana, M.A.; Newcomb, S.B.; Benavente, E.; González, G.; Sotomayor Torres, C.M. Comparative structural–vibrational study of nano-urchin and nanorods of vanadium oxide. Phys. Solid State (b) 2006, vol. 243, 3285–3289. [Google Scholar] [CrossRef]
- O’Dwyer, C.; Lavayen, V.; Newcomb, S.B.; Benavente, E.; Santa Ana, M.A.; González, G; Sotomayor Torres, C.M. Atomic layer structure of vanadium oxide nanotubes grown on nanourchin structures, Electrochem. Solid State Lett. 2007, vol. 10, A111–A114. [Google Scholar] [CrossRef]
- Mai, Liqiang; Chen, Wen; Xu, Qing; Zhu, Quanyao; Han, Chunhua; Peng, Junfeng. Cost-saving synthesis of vanadium oxide nanotubes. Solid State Communications 2003, vol. 126, 541–543. [Google Scholar] [CrossRef]
- Tenne, R.; Margulis, L; Genut, M.; modes, G. Nature 1992, vol. 360, 444. [CrossRef]
- Feldman, Y.; wasserman, E.; Srolouitz, D. j.; Tenne, R. Science 1995, vol. 267, 222. [CrossRef]
- Zeng, W.; Chen, W.; Li, Z.; Tianming, L. Rapid and sensitive ethanol sensor based on hollow Au/V2O5 nanotubes via emulsion-electrospinning route. Materials Research Bulletin 2015, vol. 65, 157–162. [Google Scholar] [CrossRef]
- Wang, L.W.; Wang, S.R.; Xun, M.J.; Hu, X.J. Chem. Phys 2013, vol. 15, 17179.
- Martin, E.; Saleta, J.C.; Troiani, H. E.; Guevara, S. R.; Sanchez, M.; Malta, R.T. vanadium oxide hexadecyl amine multiwall nanotube, science direct; 2007; pp. 333–336. [Google Scholar]
- Shimizu, Y.; Egashira, M. Basic aspects and challenges of semiconductor gas sensors. MRS Bull. 1999, vol. 24, 18–24. [Google Scholar] [CrossRef]
- Li, Z.Y.; Wang, X.G.; Lin, T. Mater. Chem. A 2014, vol. 2, 13655. [CrossRef]
- Wang, L.W.; Wang, S.R.; Xun, M.J.; Hu, X.J. Phys. Chem. Chem. Phys 2013, vol. 15, 17179. [CrossRef] [PubMed]
- Yu, M.; Liu, X.; Wang, Y.; Zheng, Y.; Zhang, J.; Li, M.; Lan, W.; Su, Q. Gas sensing properties of p-type semiconducting vanadium oxide nanotubes. Applied Surface Science 2012, vol. 258, 9554–9558. [Google Scholar] [CrossRef]
- Zhang, X.; Zhao1, M.; Yan, S.; He, T.; Li, W.; Lin, X.; Xi, Z.; Wang, Z.; Liu, X.; Xia, Y. First-principles study of ZnS nanostructures: nanotubes, nanowires and nanosheets. Nanotechnology 2008, vol. 19, 305708. [Google Scholar] [CrossRef]
- Benaissa, N.; Benaissa, T.; Garmim, Z.; El Jouad, A.; Louardi, B.; Hartiti, M.; Monkade, A.; El Kenz, A. Experimental and DFT TB-mBJ calculations studies of structural,morphological, electronic, optical and electrical properties of copper oxide thin films. Optical Materials 2023, vol. 136, 113433. [Google Scholar] [CrossRef]
- Shahzad, K.; Anurag, S.; Rajneesh, C.M.; Shahid, K.; Piyush, D. NH3and PH3adsorption through single walled ZnS nanotube:First principle insight. Chemical Physics Letters 2015, vol. 636, 103–109. [Google Scholar]
- Qin, Y.; Zhao, L.; Cui, M. Ultrathin vanadium pentoxide nanobelt for ethanol-sensing applications: Experimental and ab initio study. Journal of Alloys and Compounds 2018, vol. 735, 1480–1487. [Google Scholar] [CrossRef]
- Akande, A.A.; Mosuanga, T.; Ouma, C.N.M.; Benecha, E.M.; Tesfamicheal, T.; Rorof, K.; Machatine, A.G.J.; Mwakikunga, B.W. Ammonia gas sensing characteristics of V2O5 nanostructures: A combined experimental and ab initio density functional theory approach. Journal of Alloys and Compounds 2020, vol. 821, 153565. [Google Scholar] [CrossRef]
- Prayut, B.; Wandee, R.; Banchob, W.; Chanukorn, T. DFT investigation of NH3, PH3, and AsH3 adsorptions on Sc-, Ti-, V-,and Cr-doped single-walled carbon nanotubes. Applied Surface Science 2017, vol. 400, 506–514. [Google Scholar]
- Fu, H.; Xie, H.; Yang, X.; Xizhong, A.; Jiang, X.; Yu, A. Hydrothermal Synthesis of Silver Vanadium Oxide (Ag0.35V2O5) Nanobelts for Sensing Amines. Nanoscale Research Letters 2015, vol. 10, 411. [Google Scholar] [CrossRef]
| Nanobelt Material | Operation temperature (◦C) | Targeted gas | Concentration (ppm) | Response* | Response time/Recovery time | Reference |
|---|---|---|---|---|---|---|
| Pristine V2O5 | 250 | Ethanol | 100 | 1.7 | 32/30 | [35,36] |
| V2O5/TiO2 | 250 | Ethanol | 100 | 2. | 49/85 | [35,36] |
| V2O5/Fe2O3 | 250 | Ethanol | 100 | 2.3 | 36/64 | [35,36] |
| V2O5/SnO2 | 250 | Ethanol | 100 | 3.1 | 37/126 | [36,37] |
| Ag0.35V2O5 | 260 | Ethanol | 5-100 | 1 | 50 | [38,39] |
| Thin-Film Material | Operation temperature (◦C) | Targeted gas | Concentration (ppm) | Response* | Response time/Recovery time | Reference |
|---|---|---|---|---|---|---|
| Pristine V2O5 | 200 | NO2 | 100 | 41 | 20/150 | [48,49] |
| Pristine V2O5 | 300 | Ethanol | 300 | 1.25 | -5/8 | [50] |
| Pristine V2O5 | 350 | Hydrogen | 5 | 23% | 15/40 | [51] |
| Pristine V2O5 | 200 | Methane | 50 | 23% | 10 min | [51] |
| Pristine V2O5 | RT | Xylene | 5 | 27 | 80/50 | [52,53] |
| Pristine V2O5 | 200 | Propane | 50 | 0.05 | 15/30 | [36,51] |
| V2O5-TiO2 | 199.85-349.85 | O2 | 10 -21% | - | - | [27] |
| V2O5-V7O16 | 620 | NH3 | 0.16-0.32 | 1.4 | - | [54] |
| Material | Operation temperature (◦C) | Targeted gas | Concentration (ppm) | Response* | Response time/Recovery time | Reference |
|---|---|---|---|---|---|---|
| Pristine V2O5 | 200 | NO2 | 100 | 24.2 | 13/140 | [61] |
| p-type porous silicon (PS)/ V2O | 25 | NO2 | 0.25 | 7.4 | 2/6 | [28,62] |
| Pd-V2O5 | 200 | NO2 | 100 | 75 | 22/126 | [48,53] |
| Pristine V2O5 | RT | Ethanol | 100 | 3.7% | - | [58] |
| Pristine V2O5 | RT | Ammonia | 500 | 1.8% | 23/15 | [39,58] |
| Te-V2O5 | RT | Ethanol | 500 | 1.8% | 3.5 | [53,63] |
| Nanofibers Material | Operation temperature (◦C) | Targeted gas | Concentration (ppm) | Response* | Response time/Recovery time | Reference |
|---|---|---|---|---|---|---|
| Pristine V2O5 | RT | 1-butylamine | 0.03 | 42 | 250/700 | [53] |
| Pristine V2O5 | RT | Xylene | 5 | 1.7 | - | [52] |
| Pristine V2O5 | 150 | Ammonia | 0.1 | 11 | 50/350 | [39,75] |
| SnO2- V2O5 | 325 | Benzene | 25 | 6.35 | - | [76] |
| Nanofibers Material | Operation temperature (◦C) | Targeted gas | Concentration (ppm) | Response* | Response time/Recovery time | Reference |
|---|---|---|---|---|---|---|
| Pristine V2O5 | 250 | Ammonia | 100 | 4.5 | - | [86] |
| Pristine V2O5 | 250 | Ethanol | 5-1000 | 5.3 | - | [87] |
| Pristine V2O5 | 300 | Xylene | 100 | 2.2 | - | [88] |
| SnO2- V2O5 | 140 | Butylamine | 100 | 2.6 | 9/49 | [89] |
| Nanowire Material | Operation temperature (◦C) | Targeted gas | Concentration (ppm) | Response* | Response time/Recovery time | Reference |
|---|---|---|---|---|---|---|
| Pristine V2O5 | 330 | Ethanol | 9.09-1000 | 9 | - | [97] |
| Pristine V2O5 | 200 | NO2 | 20 | 41% | 20/150 | [98] |
| Pristine V2O5 | 330 | Ammonia | 1000 | 1.8 | - | [99] |
| Pristine V2O5 | -RT | He | 0-17.6 | 5% | - | [100] |
| CuO/V2O5 | 220 | Hydrogen Sulphide | 23 | 31.86 | - | [100] |
| SnO2-V2O5 | RT | Ethanol | 100 | 14 | - | [101] |
| Nanotube Material | Operation temperature (◦C) | Targeted gas | Concentration (ppm) | Response* | Response time/Recovery time (s) | Reference | |
|---|---|---|---|---|---|---|---|
| Pristine V2O5 | 330 | Ethanol | 100 | 2.6 | 5/5 | [112] | |
| V2O5/Au | 200 | Ethanol | 100 | 2.7 | 7/5 | [112] | |
| V2O5/Fe2O3 | 330 | Ethanol | 100 | 1.3 | 15/20 | [113] | |
| This work (GGA) | Experiment [124] | Literature (Calculated) [125] | |
|---|---|---|---|
| Lattice parameters (Å) | |||
| a | 3.62 | 3.56 | 3.56 |
| b | 4.79 | 4.37 | 4.37 |
| c | 11.55 | 11.51 | 11.51 |
| Bond length (Å) | |||
| vanadyl oxygen (Ov) | 1.61 | 1.54 | 1.61 |
| bridge oxygen (Ob) | 1.81 | 1.88 | 1.78 |
| chain oxygen (Oc) | 2.04 | 2.02 | 2.02 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




