Submitted:
03 February 2026
Posted:
04 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results & Discussion
3. Materials and Methods
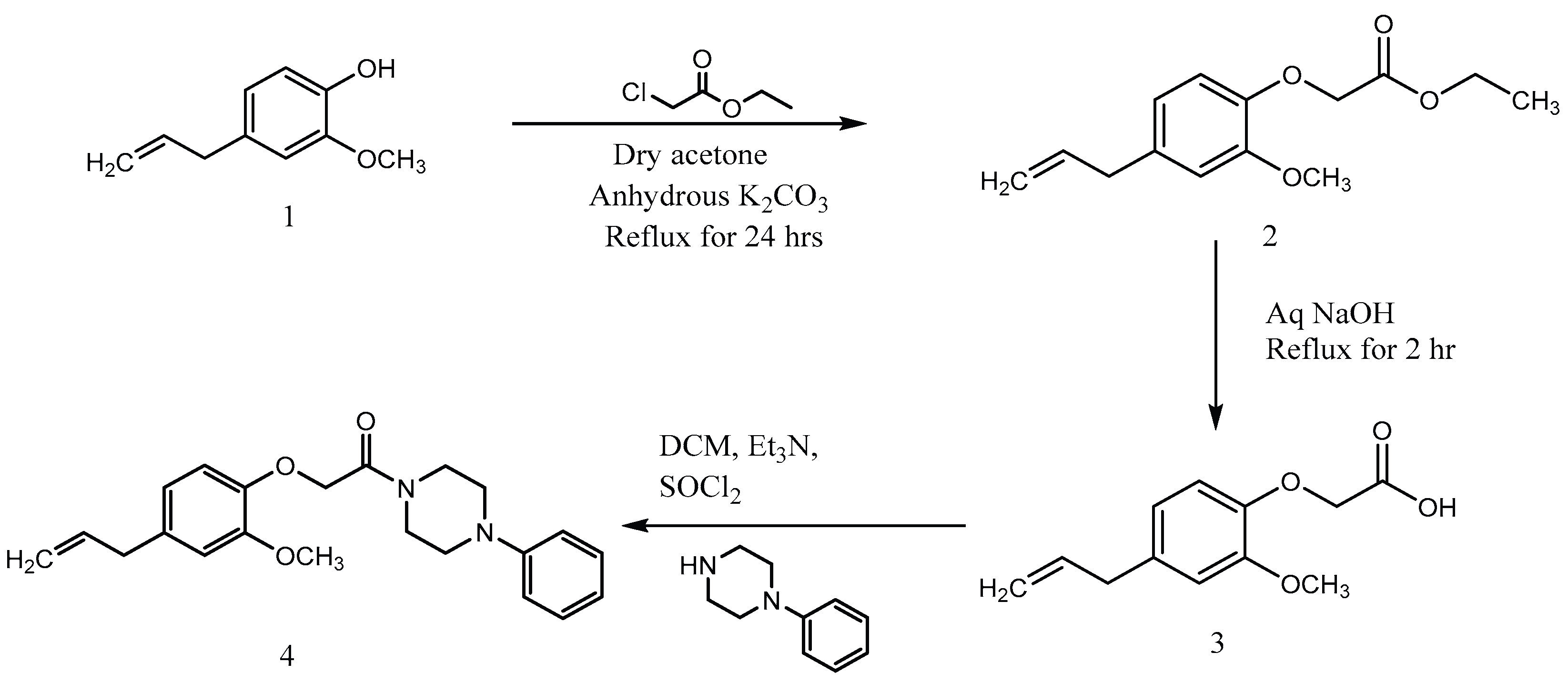
3.1. Synthesis of Ethyl [2-methoxy-4-(prop-2-en-1-yl)phenoxy]acetate (Compound 2)
3.2. Synthesis of 2-(4-allyl-2-methoxyphenoxy)acetic Acid (Compound 3)
3.3. Synthesis of 2-(4-allyl-2-methoxyphenoxy)-1-(4-phenylpiperazin-1-yl) ethan-1-one (Compound 4)
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Atanasov, A. G.; Zotchev, S. B.; Dirsch, V. M.; Supuran, C. T. Natural Products in Drug Discovery: Advances and Opportunities. Nat. Rev. Drug Discov. 2021, 20, 200–216. [Google Scholar] [CrossRef]
- Goel, B.; Jain, S. K. Semisynthesis: Bridging Natural Products and Novel Anticancer Therapies. Eur. J. Med. Chem. Rep. 2024, 12, 100218. [Google Scholar] [CrossRef]
- Naeem, A.; Hu, P.; Yang, M.; Zhang, J.; Liu, Y.; Zhu, W.; Zheng, Q. Natural Products as Anticancer Agents: Current Status and Future Perspectives. Molecules 2022, 27, 8367. [Google Scholar] [CrossRef] [PubMed]
- Newman, D. J.; Cragg, G. M. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R. H.; Guo, H. Y.; Deng, H.; Li, J.; Quan, Z. S. Piperazine Skeleton in the Structural Modification of Natural Products: A Review. J. Enzyme Inhib. Med. Chem. 2021, 36, 1165–1197. [Google Scholar] [CrossRef]
- Faizan, M.; Kumar, R.; Mazumder, A.; Salahuddin; Kukreti, N.; Kumar, A.; Chaitanya, M. V. N. L. The Medicinal Chemistry of Piperazines: A Review. Chem. Biol. Drug Des. 2024, 103, e14537. [Google Scholar] [CrossRef]
- Fadilah, F.; Yanuar, A.; Arsianti, A.; Andrajati, R. Phenylpropanoids, Eugenol Scaffold, and Its Derivatives as Anticancer Agents. Asian J. Pharm. Clin. Res. 2017, 10, 41–46. [Google Scholar] [CrossRef]
- Da Silva, F. F. M.; Monte, F. J. Q.; de Lemos, T. L. G.; et al. Eugenol Derivatives: Synthesis, Characterization, and Evaluation of Antibacterial and Antioxidant Activities. Chem. Cent. J. 2018, 12, 34. [Google Scholar] [CrossRef] [PubMed]
- FernandesMelo Reis, R. C.; Pontes Silva, A. V.; Veiga Torres, A. D.; de Cassia Alves Iemini, R.; Lapa, I. R.; Franco, L. L.; Pinto Braga, S. F.; Carvalho, D. T.; Dias, D. F.; de Souza, T. B. From Clove Oil to Bioactive Agents: Synthetic Routes, Antimicrobial and Antiparasitic Activities of Eugenol Derivatives. Future Med. Chem. 2024, 16(20), 2169–2188. [Google Scholar] [CrossRef] [PubMed]
- Nazreen, S.; Elbehairi, S. E. I.; Malebari, A. M.; et al. New Natural Eugenol Derivatives as Anti-proliferative Agents: Synthesis, Biological Evaluation, and Computational Studies. ACS Omega 2023, 8(21), 18811–18822. [Google Scholar] [CrossRef] [PubMed]
- Alam, M. M.; Elbehairi, S. E. I.; Shati, A. A.; Hussien, R. A.; Alfaifi, M. Y.; Malebari, A. M.; et al. Design, Synthesis, and Biological Evaluation of New Eugenol Derivatives Containing 1,3,4-Oxadiazole as Novel Inhibitors of Thymidylate Synthase. New J. Chem. 2023, 47(10), 5021–5032. [Google Scholar] [CrossRef]
- Anjum, N. F.; Shanmugarajan, D.; Shivaraju, V. K.; Faizan, S.; Naishima, N. L.; Prashantha Kumar, B. R.; Javid, S.; Purohit, M. N. Novel Derivatives of Eugenol as Potent Anti-Inflammatory Agents via PPARγAgonism: Rational Design, Synthesis, Biological Evaluation, and Computational Studies. RSC Adv. 2022, 12, 16966–16978. [Google Scholar] [CrossRef]
- Rohane, S. H.; Chauhan, A. J.; Fuloria, N. K.; Fuloria, S. Synthesis and in vitro antimycobacterial potential of novel hydrazones of eugenol. Arab. J. Chem. 2020, 13, 4496–4508. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



