Submitted:
04 February 2026
Posted:
06 February 2026
You are already at the latest version
Abstract
formation is an important mechanism in plant evolution that serves as the material basis for human survival. After double fertilization, the development of both the embryo and the endosperm requires a large amount of nutrients, and long-distance sucrose transportation is indispensable. We recently discovered a ring-shaped structure that can affect the unloading of nutrients into seeds. This structure is formed by the deposition of callose, which blocks the transport of nutrients by reducing the pore size of the plasmodesmata (PD). If fertilization is successful, the PD gate will form, but will remain open; if fertilization fails, the PD gate will be gradually closed—a strategy that efficiently prevents energy loss. A similar gating mechanism exists in rice, indicating that this strategy has substantial potential for agricultural production.
Keywords:
1. Introduction
2. Materials and Methods
2.1. The Aniline Blue Staining
2.2. Observation of Rice Pistils
3. Results and Discussion
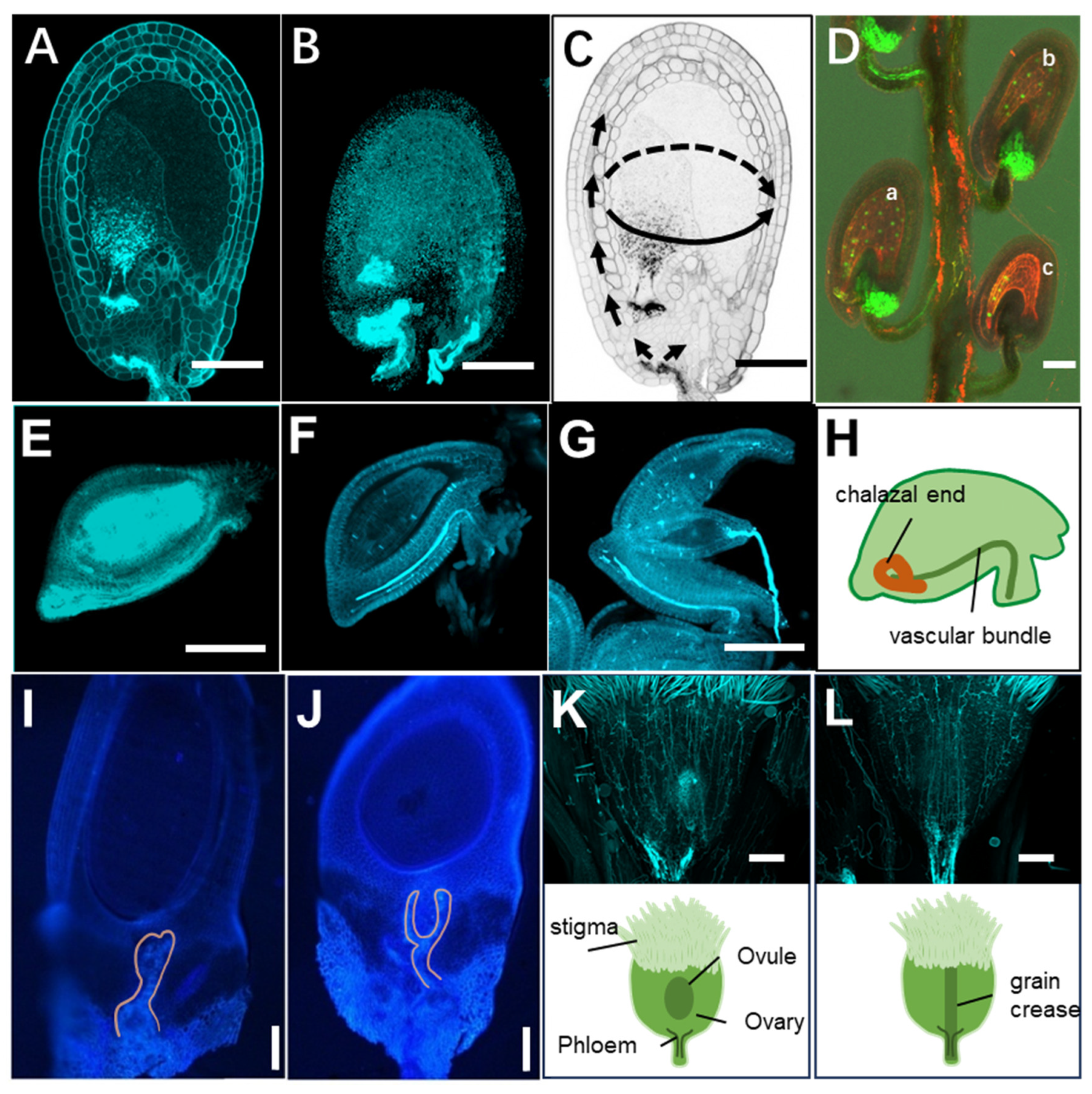
3.1. Dicotyledonous Plants Oxalis pes-caprae L. and Sterile Oxalis debilis Kunth
3.2. Oryza sativa L ssp. japonica
3.3. Pseudoroegneria stipifolia (Trautv.) Á.Löve
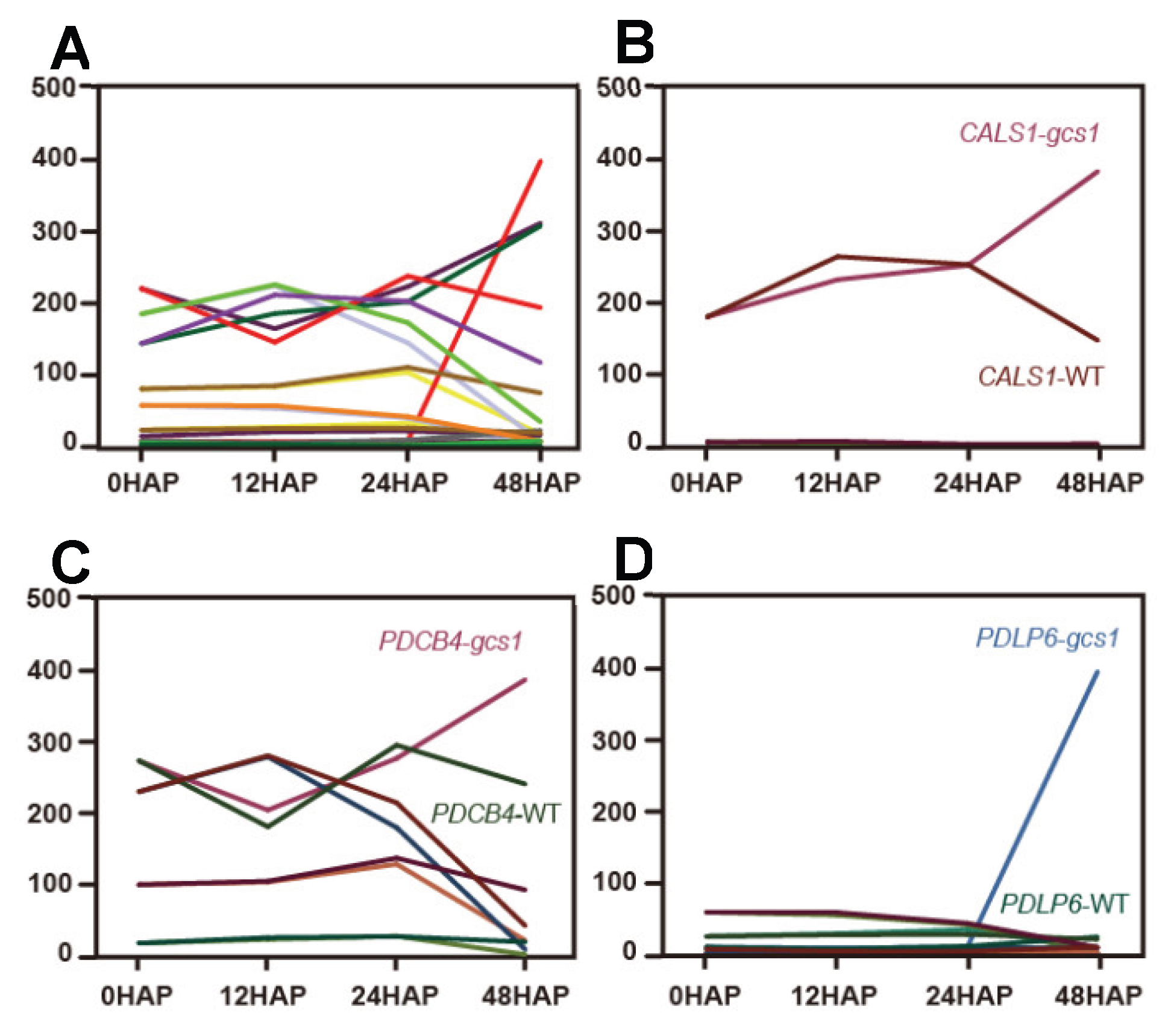
3.4. Fertilization of the Central Cell Unlocks the KGW
3.5. On/off Mechanism of the KGW
Author Contributions
Funding
Conflicts of Interest
References
- Münch, E. Material Flow in Plants; Milburn, J. A.; Kreeb, K. H., Translators; University of Bremen: Germany; Gustav Fischer Verlag: Jena Germany.
- Liu, X.; Nakajima, K.P.; Adhikari, P.B.; Wu, X.; Zhu, S.; Okada, K.; Kagenishi, T.; Kurotani, K.; Ishida, T.; Nakamura, M.; Sato, Y; Kawakatsu, Y.; Xie, L.; Huang, C.; He, J.; Yokawa, K.; Sawa, S.; Higashiyama, T.; Bradford, K.J.; Notaguchi, M.; Kasahara, R.D. Fertilization-dependent phloem end gate regulates seed size. Curr Biol. 2025, 35, 2049–63.e3. [Google Scholar] [CrossRef] [PubMed]
- Kasahara, R. D. Fertilization initiates seed nutrition via phloem end by a callose degradation enzyme. DNA Cell Biol. 2025, 44, 407–410. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, Kohdai P.; Kasahara, Ryushiro D. How Do Seeds Get Bigger? A Newly Identified Phloem-Terminal Tissue Controls Seed Enlargement through Callose Degradation. Academia Molecular Biology and Genomics 2025. [Google Scholar] [CrossRef]
- Mori, T.; Kuroiwa, H.; Higashiyama, T.; Kuroiwa, T. GENERATIVE CELL SPECIFIC 1 is essential for angiosperm fertilization. Nat. Cell Biol. 2006, 8, 64–71. [Google Scholar] [CrossRef] [PubMed]
- von Besser, K.; Frank, A. C.; Johnson, M. A.; Preuss, D. Arabidopsis HAP2 (GCS1) is a sperm-specific gene required for pollen tube guidance and fertilization. Development 2006, 133, 4761–4769. [Google Scholar] [CrossRef] [PubMed]
- Castro, S.; Castro, M.; Ferrero, V.; Costa, J.; Tavares, D.; Navarro, L.; Loureiro, J. Invasion fosters change: independent evolutionary shifts in reproductive traits after Oxalis pes-caprae L. introduction. Front. Plant Sci. 2016, 7, 874. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.; Zhang, D.; Renner, S. S. Oxalis debilis in China: distribution of flower morphs, sterile pollen and polyploidy. Ann Bot. 2006, 98, 459–64. [Google Scholar] [CrossRef] [PubMed]
- Honma, Y; Adhikari, P. B.; Kuwata, K.; Kagenishi, K.; Yo kawa, K.; Notaguchi, M.; Kurotani, K.; Toda, E.; Bessho-Uehara, K.; Liu, X.; Zhu, S.; Wu, X.; Kasahara, R. D. High-quality sugar production by osgcs1 rice. Commun. Biol. 2020, 3, 617. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y.; Khan, N.; Chaudhary, R.; Perumal, S.; Wang, Z.; Hucl, P.; Biligetu, B.; Sharpe, A.G.; Jin, L. Genomic insights into wheatgrass: unravelling genetic diversity, population structure, and evolutionary dynamics in Pseudoroegneria species. Mol. Phylogenet. Evol. 2025, 211, 108397. [Google Scholar] [CrossRef] [PubMed]
- Linkies, A.; Graeber, K.; Knight, C.; Leubner-Metzger, G. The evolution of seeds. New Phytol. 2010, 186, 817–831. [Google Scholar] [CrossRef] [PubMed]
- Ron, M.; Saez, M. A.; Williams, L. E.; Fletcher, J. C.; McCormick, S. Proper regulation of a sperm-specific cis-nat-siRNA is essential for double fertilization in Arabidopsis. Genes Dev. 2010, 24, 1010–1021. [Google Scholar] [CrossRef] [PubMed]
- Gooh, K.; Ueda, M.; Aruga, K.; Park, J.; Arata, H.; Higashiyama, T.; Kurihara, D. Live-cell imaging and optical manipulation of Arabidopsis early embryogenesis. Dev. Cell 2015, 34, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Kang, I. H.; Steffen, J. G.; Portereiko, M. F.; Lloyd, A.; Drews, G. N. The AGL62 MADS domain protein regulates cellularization during endosperm development in Arabidopsis. Plant Cell 2008, 20, 635–647. [Google Scholar] [CrossRef] [PubMed]
- Bayer, E. M.; Benitez-Alfonso, Y. Plasmodesmata: Channels under pressure. Annual Review of Plant Biology 2024, 75, 291–317. [Google Scholar] [CrossRef] [PubMed]
- Kasahara, R. D; Notaguchi, M.; Honma, Y. Discovery of pollen tube-dependent ovule enlargement morphology phenomenon, a new step in plant reproduction. Commun. Integr. Biol. 2017, 5, e1338989. [Google Scholar] [CrossRef]
- Chen, X.; Li, W.; Gao, J.; Wu, Z.; Du, J.; Zhang, X.; Zhu, Y. Arabidopsis PDLP7 modulated plasmodesmata function is related to BG10-dependent glucosidase activity required for callose degradation. Sci. Bull. 2024, 69, 3075–3088. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



