1. Introduction
Intra-abdominal infections are among the most common causes of acute surgical conditions in pediatric patients and frequently require operative management, particularly in cases of complicated appendicitis, bowel perforation, or intra-abdominal abscess formation. These infections encompass a heterogeneous group of clinical entities characterized by varying severity, microbial burden, and risk of postoperative complications. In children, age-related differences in immune response and disease progression further influence clinical presentation and outcomes, underscoring the need for pediatric-specific management strategies [
1].
Abdominal surgical procedures, especially those involving the gastrointestinal tract, are associated with a substantial risk of postoperative infectious complications. The high bacterial load of the intestinal flora and the frequent presence of contaminated or dirty surgical fields contribute to this risk, particularly in appendiceal and colorectal surgery. Among these complications, surgical site infections (SSIs) remain one of the most frequent and clinically relevant adverse outcomes, contributing to prolonged hospitalization, increased antimicrobial exposure, and higher healthcare costs [
2].
Bacterial pathogens represent the primary etiological agents of intra-abdominal infections and SSIs. Accordingly, perioperative antimicrobial prophylaxis and postoperative antibiotic therapy are integral components of pediatric surgical care [
3,
4,
5,
6,
7,
8,
9]. However, considerable variability persists in antibiotic selection and treatment duration, which typically ranges from 3 to 10 days [
9,
10,
11,
12,
15]. This variability reflects ongoing uncertainty regarding optimal antimicrobial strategies, particularly in pediatric populations.
Reported SSI incidence in abdominal surgery is approximately 10%, although this figure is likely underestimated due to infections developing after hospital discharge [
1,
2]. Pediatric patients appear to be at increased risk compared with adults, with reported SSI rates ranging from 6.3% in developed countries to 12.4–24.7% in developing regions [
14]. Despite this, current guidelines, including those from the American Society of Health-System Pharmacists (ASHP), do not distinguish between adult and pediatric patients when recommending antimicrobial prophylaxis for abdominal surgery [
16]. As a result, pediatric treatment protocols are often extrapolated from adult data.
Pediatric-specific information regarding the microbiological spectrum and antimicrobial susceptibility patterns of intra-abdominal infections and SSIs remains limited. Given the increasing prevalence of antimicrobial resistance and the importance of antimicrobial stewardship, detailed characterization of pathogen distribution and resistance patterns in children is essential to guide empiric therapy and support culture-guided de-escalation [
15]. Intra-abdominal infections in children are associated with significant morbidity and may rapidly progress to severe complications if diagnosis or treatment is delayed. Compared to adults, pediatric patients exhibit distinct immunological characteristics, inflammatory responses, and clinical evolution, necessitating tailored diagnostic and therapeutic strategies [
17].
There is a lack of pediatric-specific studies addressing the prognosis and management of SSIs. Currently, pediatric-specific data on microbiological profiles and antimicrobial susceptibility are limited, and management is frequently extrapolated from adult guidelines. Therefore, the present study aims to investigate the bacterial etiology of intra-abdominal SSIs in pediatric patients between 0 and 16 years. The SSI diagnosis was correlated with pediatric patients' age, surgical specimens, identified pathogens, and susceptibility patterns. Extensive statistical analysis supports the obtained results, with detailed correlations between all available data. Our findings confirm that continuous collaboration between pediatric surgeons and microbiologists is an essential factor for optimal surgical management and appropriate antimicrobial therapy.
2. Materials and Methods
2.1. Study Design and Population
This retrospective observational study was conducted at the “Saint Apostle Andrew” Clinical Hospital, Constanța, and included pediatric patients (0–16 years) who underwent surgical intervention for intra-abdominal pathology associated with confirmed intra-abdominal infection between January 2022 and November 2025.
Patients were eligible for inclusion if intra-abdominal infection was identified either intraoperatively or postoperatively and microbiological samples were obtained from the intra-abdominal site. Both patients undergoing surgery for infectious indications (e.g., complicated appendicitis, intra-abdominal abscess, generalized peritonitis) and those who developed postoperative intra-abdominal surgical site infections (SSIs) were included. The term surgical site infection was used in accordance with the Centers for Disease Control and Prevention (CDC) criteria and referred to infections involving the intra-abdominal space occurring within 30 days following surgery in the absence of implanted material.
Patients without microbiological confirmation, those managed non-operatively, or cases with incomplete clinical or laboratory data were excluded.
2.2. Data collection
Demographic data (age, sex), clinical diagnosis, type of surgical intervention, microbiological findings, and antimicrobial susceptibility results were extracted from electronic medical records. Patients were stratified into age groups (0–5, 6–10, and 11–16 years) to evaluate age-related differences in pathogen distribution and antimicrobial susceptibility patterns.
Surgical interventions were categorized according to the primary procedure performed, including appendectomy, abscess drainage, bowel resection, and other intra-abdominal procedures.
2.3. Microbiological Analysis
Biological samples (peritoneal fluid, pus, and other fluids) were collected under sterile conditions during surgical procedures. Samples were processed according to standard microbiological protocols. Bacterial identification was performed using conventional culture methods and, where available, automated identification systems. Polymicrobial infections were classified as dual Gram-negative (2GN), Gram-negative plus Gram-positive (1GN+1GP or 1GP+1GN), or triple associations (2GN+1GP or 1GN+2GP).
Intra-abdominal samples were collected intraoperatively or from postoperative drainage and processed according to standard microbiological protocols. Identification of bacterial isolates and antimicrobial susceptibility testing were performed using automated systems in the institutional microbiology laboratory.
Antimicrobial susceptibility was reported according to EUCAST criteria. Combination therapy susceptibility refers to predefined antibiotic combinations commonly used in clinical practice (e.g., β-lactam plus aminoglycoside or β-lactam plus metronidazole), as reported by the laboratory.
Anaerobic cultures were not routinely performed, and anaerobic pathogens were therefore not systematically identified.
2.4. Follow-Up and SSI Surveillance
Postoperative surveillance for intra-abdominal SSI was conducted during hospitalization and through outpatient follow-up visits or hospital readmissions documented in the electronic medical record. SSIs diagnosed after discharge were included when clinical documentation and microbiological confirmation were available within 30 days postoperatively.
2.5. Statistical Analysis
Descriptive statistics were used to summarize demographic, clinical, and microbiological data. Associations between categorical variables were assessed using chi-square tests, with adjusted Pearson residuals calculated to identify statistically significant deviations from expected frequencies. Associations between clinical variables, pathogen groups, and antibiotic susceptibility were assessed using the Chi-square test with Cramer's V coefficient (V), adjusted Pearson residuals (r*ij), and Fisher's exact test. A p-value < 0.05 was considered statistically significant.
The study was approved by the Ethics Committee of “Saint Apostle Andrew” Clinical Hospital, Constanța (approval number: 02/2026).
3. Results
3.1. Baseline Characteristics of the Pediatric Patients
A total of 177 pediatric patients were included in the study, of whom 64 (36.16%) were female, and 113 (63.84%) were male (
Table 1). Male predominance was observed across all age groups. The most significant proportion of patients was in the 11–16-year age group (44.07%), followed by those aged 7–10 years (22.03%). Age distribution differed significantly between sexes (p < 0.05).
Appendicitis was the most frequent diagnosis, accounting for 54.80% of cases, followed by intra-abdominal abscesses (22.03%) and peritonitis (3.95%). Several less common conditions, including intestinal perforation, occlusion, and hernias, were also identified. The distribution of SSI diagnoses did not show a statistically significant association with sex (p = 0.460; V = 0.273;
Table 1). The predominance of appendicitis and abscesses explains the high proportion of contaminated and dirty surgical fields, which are known to favor polymicrobial infections and increased antimicrobial resistance.
However, significant correlations were observed between SSI type and pediatric patient age (
Table 2), supported by the r*ij and
p-values. Appendicitis is substantially correlated with the 7-10 and 11-17 age groups, peritonitis with the 11-16 age group, and other SSIs with the 4-6 age group (r*ij > 2.8,
p < 0.05;
Table 2).
3.2. Biological Specimens, Surgical Interventions, and Pathogen Types
Peritoneal fluid was the most collected specimen (67.23%), followed by pus samples (29.38%) and other biological fluids (3.39%) (Figure 1A).
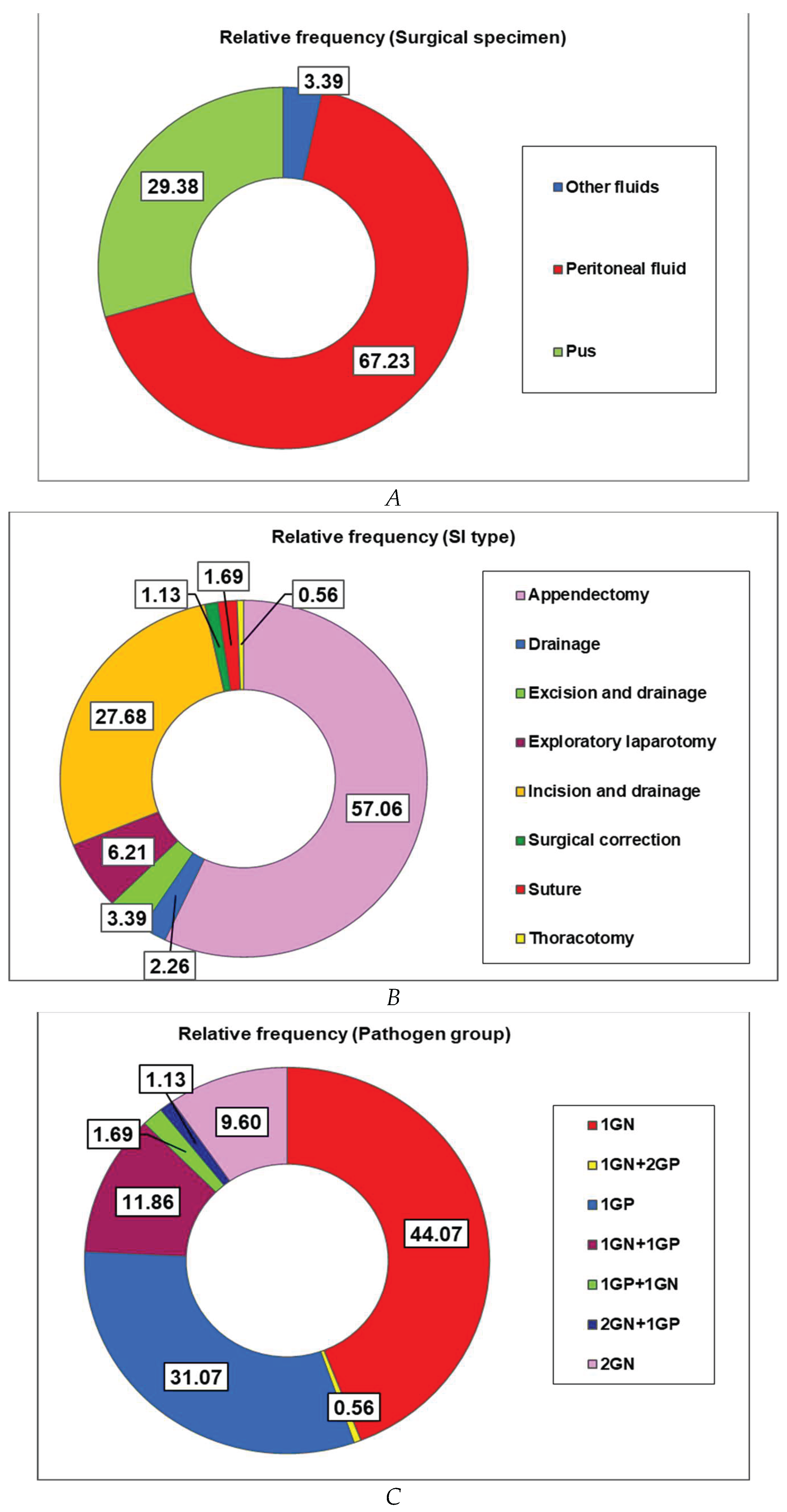
Appendectomy was the most frequently performed surgical procedure (57.06%), corresponding to the high incidence of appendicitis (54.80%) in the study cohort (Figure 1B and
Table 1). It is followed by incision and drainage (27.68%) and exploratory laparotomy (6.21%; Figure 1B).
Gram-negative bacteria (GN) were identified in most patients (44.07%), alone (42.94%), or in pairs of associated Gram-negative species (9.60%; Figure 1C). Gram-positive germs were identified alone in 27.68% of cases (Figure 1C) and combined with GN bacteria (1GP+1GN, 1.69%, Figure 1C). Gram-negative bacterial infections are frequently superinfected with Gram-positive germs: 1GN+1GP (11.86%), 2GN+1GP (1.13%), and 1 GN+2GP (0.56%; Figure 1C).
Appendectomy was the most frequently performed surgical procedure associated with a peritoneal fluid specimen (84.87%), while incision and drainage were predominantly associated with pus samples (90.38%;
Table 3). Exploratory laparotomy was mainly performed in patients with peritoneal fluid samples (9.24%;
Table 3). Biological specimens are substantially correlated with surgical intervention (SI) type and with SSI type (V = 1,
p < 0.0001,
Table 3).
However, no significant correlation was observed between surgical specimens and pathogen groups (V = 0.156, p = 0.613;
Table 3). Gram-negative bacteria isolated alone (1GN) predominated across all specimen types, particularly in other fluids (66.67%) and peritoneal fluid (46.22%). Gram-positive bacteria alone (1GP) were more frequently isolated in pus samples (38.47%) than in peritoneal fluid (28.57%) and other specimens (16.67%;
Table 3).
The correlations between biological specimens collected in intra- and post-surgical interventions and SI type and SSI type were explored using adjusted Pearson residuals and Fisher's exact test (
Table 4). Therefore, substantial positive associations were observed between peritoneal fluid, appendectomy, and appendicitis (r*ij > 10.000,
p < 0.0001;
Table 4). Pus is considerably correlated with incision and drainage and abscesses (r*ij > 9.500,
p < 0.0001), while other specimens are significantly associated with excision and drainage, thoracotomy, acute abdomen, and other SSIs ( r*ij > 2.800,
p < 0.05,
Table 4).
The same statistical tools helped us to understand the associations between SI type and SSIs (
Table 5).
Thus, appendectomy was performed mainly for appendicitis (r*ij > 12.000,
p < 0.0001), while incision and drainage were selected for abscess (r*ij > 10.000,
p < 0.0001), adenitis, and other SSIs (r*ij > 2.700,
p < 0.05;
Table 5). Surgical correction was preferred for inguinal and mesenteric hernias (r*ij > 9.000,
p < 0.02), exploratory laparotomy was selected for intestinal occlusion and perforation ( r*ij > 4.000,
p ≤ 0.01), suture was made for evisceration ( r*ij > 5.000,
p < 0.05), while excision and drainage were realized for ovarian tumors and other SSIs ( r*ij > 5.000,
p < 0.05,
Table 5).
Bacterial pathogens were also associated with SI type and SSIs; the corresponding significant correlations were assessed using adjusted Pearson residuals and Fisher's exact test. Thus, a dual association of 1GP+1GN was identified in pediatric patients undergoing exploratory laparotomy (r*ij = 4.374, p = 0.01) and in those diagnosed with evisceration (r*ij = 5.322, p = 0.034) and intestinal invagination (r*ij = 7.637, p = 0.017). Finally, triple association 2GN+1GP was identified in adenitis (r*ij = 5.322, p = 0.034).
3.3. Distribution of Isolated Bacterial Species
Among infections caused by Gram-negative bacteria alone (1GN),
E. coli was the most frequently isolated pathogen (83.33%), followed by
Klebsiella spp. and
P. aeruginosa, each identified in 6.41% of cases (
Table 6).
In the 1GP group,
S. aureus was the dominant microorganism (60%). In comparison, various
Enterococcus spp. and
Streptococcus spp. were also detected in 23.64% and 14.55% of pediatric patients, respectively, and accounted for the remaining isolates.
Gemella morbidum was identified in a single case (1.82%,
Table 6).
Polymicrobial infections involving two Gram-negative bacteria (2GN) were most frequently represented by combinations of
E. coli with
P. aeruginosa (52.94%),
Klebsiella spp. (17.65%), and
A. baumanii (11.76%,
Table 6).
Mixed Gram-negative and Gram-positive infections (1GN+1GP) predominantly involved
E. coli in association with
Streptococcus spp. (52.38%),
Enterococcus spp. (23.81%) and
S. aureus (9.52%,
Table 6). Three double associations were very rarely detected:
C. freundii + Enterococcus spp.,
H. alvei + Streptococcus spp., and
Klebsiella spp. +
Streptococcus spp. Double combinations (1GP+1GN) were rarely detected (3/177) and were based on
Enterococcus spp, associated with
Enterobacter spp.,
Klebsiella spp., and
P. aeruginosa (
Table 6). Triple bacterial associations (3/177) were rare and mainly included
E. coli,
P. aeruginosa, and
Streptococcus spp. (2GN+1GP, 2/177) and
E. coli + Enterococcus spp.
+ Streptococcus spp. (1GN+2GP, 1/177,
Table 6).
Notably, pus samples were more commonly obtained from younger age groups (< 7 years; r*ij > 5.500, p < 0.0001), whereas peritoneal fluid samples were mainly obtained from patients aged 7-16 years (r*ij > 4.900, p < 0.0001). Moreover, 2GN pathogens are significantly associated with the 7-10 years group (r*ij = 3.234, p = 0.003). E. coli + P aeruginosa is substantially associated with the 7-10 years group (r*ij = 4.967, p < 0.0001), while Klebsiella spp. was highly correlated with the 4-6 years group (r*ij = 4.085, p = 0.002) and P. aeruginosa with 11-16 years (r*ij = 2.556, p = 0.015).
Detailed correlations between Gram-negative and Gram-positive bacteria identified in specimens collected in intra- and post-surgical interventions and SSI type were explored using adjusted Pearson residuals and Fisher's exact test (
Table 7 and
Table 8).
Data from
Table 7 show that
E. coli is significantly associated with appendicitis (r > 3.800,
p < 0.001), while
P. aeruginosa is involved in complicated SSIs, such as peritonitis and pyloric stenosis (r*ij > 5.800,
p < 0.05).
Klebsiella spp. are detected in ovarian tumor (r*ij > 5.800,
p < 0.05). Moreover, both
Klebsiella and
Enterobacter spp. were identified in biological specimens of pediatric patients with other SSIs (r*ij > 4.200,
p < 0.05;
Table 7).
Gram-positive bacteria were associated with severe SSIs (
Table 8).
Enterococcus spp. is significantly correlated with intestinal occlusion and perforation (r*ij > 3.900,
p < 0.05), while
Streptococcus spp. is strongly associated with mesenteric hernia (r*ij > 4.600,
p < 0.05). Both Gram-negative bacteria were highly correlated with other SSIs (r*ij > 2.500,
p < 0.05,
Table 8).
Moreover, Gram-negative bacteria in dual combination (2GN) were considerably associated with peritonitis (Klebsiella spp.+ P. aeruginosa, r*ij = 4.943, p = 0.04), appendicitis (E. coli + P. aeruginosa, r*ij = 2.796, p = 0.004), and abscess (E. coli +A. Baumanii, r*ij = 2.675, p = 0.048).
Triple bacterial associations tip 2GN+1GP (E. coli + P. aeruginosa + Streptococcus spp.) were highly associated with adenitis (r*ij = 5.322, p = 0.034).
Dual associations tip 1GP+1GN) are substantially correlated with complicated SSIs, as intestinal invagination (Enterococcus spp. + P. aeruginosa, r*ij = 13.304, p = 0.006) and evisceration (Enterococcus spp. + Klebsiella spp., r*ij = 9.381, p = 0.011).
3.4. Antimicrobial Susceptibility Patterns
Previous findings emphasize the importance of empiric antimicrobial coverage against Gram-negative organisms in pediatric intra-abdominal infections, particularly in appendiceal pathology. The high rate of polymicrobial infections supports the need for broad-spectrum empiric therapy and careful de-escalation based on culture results.
Overall susceptibility was highest for cephalosporins (53.67%), combination regimens (51.98%), monobactams (45.19%), and aminoglycosides (40.68%,
Table 9).
Aminoglycosides demonstrated good activity, particularly against polymicrobial Gram-negative infections (61.54% in the 2GN group).
Cephalosporins are significantly correlated with Enterococcus spp. (r*ij = 2.344, p = 0.022), monobactams with E. coli + Enterococcus spp. (r*ij = 2.498, p = 0.018), quinolones with E. coli (r*ij = 2.171, p = 0.034) and E. coli + Klebsiella spp. (r*ij = 2.948, p = 0.017), carbapenems with P. aeruginosa (r*ij = 2.349, p = 0.049), aminoglycosides with E. coli + Enterococcus spp. (r*ij = 2.739, p = 0.010) and E. coli + P. aeruginosa (r*ij = 2.326, p = 0.034), and combination with E. coli + Streptococcus spp (r*ij = 2.669, p = 0.010).
Clinically relevant resistance trends included:
- ○
Low susceptibility to penicillins across all pathogen groups (4.52%), limiting their utility as monotherapy.
- ○
Moderate carbapenem susceptibility (19.20%), indicating the emergence of resistant Gram-negative strains and supporting restricted use in severe or refractory cases.
- ○
Reduced quinolone susceptibility in mixed infections (GN+GP), suggesting limited effectiveness in polymicrobial disease.
- ○
Higher activity of glycopeptides, lincosamides, and oxazolidinones against Gram-positive organisms, particularly S. aureus and Enterococcus spp.
These resistance patterns underscore the importance of avoiding unnecessary prolonged prophylaxis and highlight the need for culture-guided antimicrobial stewardship in pediatric surgery.
4. Discussions
This study provides a comprehensive analysis of pediatric intra-abdominal infections associated with surgical intervention, highlighting age-related differences in microbiological profiles and antimicrobial susceptibility patterns. Pediatric-specific data in this field remain limited, and treatment strategies are often extrapolated from adult populations; therefore, these findings contribute valuable real-world evidence to support empiric antimicrobial decision-making in pediatric surgery.
4.1. Principal Findings:
Consistent with existing pediatric literature, appendicitis-particularly in its complicated form—represented the most frequent diagnosis associated with intra-abdominal infection. Gram-negative bacteria predominated across all age groups, with Escherichia coli identified as the most common pathogen, particularly in monomicrobial infections. Gram-positive organisms and polymicrobial infections were more frequently observed in younger children and in cases involving abscess formation or diffuse peritonitis, reflecting the complexity of these clinical presentations.
Age-related differences in pathogen distribution were observed, with adolescents more frequently exhibiting Gram-negative monomicrobial infections, while younger age groups demonstrated a higher proportion of polymicrobial infections. These findings suggest an association between age, disease severity, and microbial complexity, although causal relationships cannot be inferred due to the retrospective nature of the study.
Antimicrobial susceptibility analysis demonstrated generally favorable sensitivity of Gram-negative isolates to third-generation cephalosporins and combination regimens, supporting their continued use as empiric therapy in uncomplicated cases. However, reduced susceptibility observed in polymicrobial infections and abscess-related cases underscores the importance of broader-spectrum empiric coverage in clinically severe presentations, followed by culture-guided de-escalation. These results align with current pediatric surgical and infectious disease recommendations emphasizing antimicrobial stewardship.
4.2. Antibacterial Susceptibility Patterns
In line with World Society of Emergency Surgery (WSES) and Infectious Diseases Society of America (IDSA) guidelines, Gram-negative bacteria—particularly Escherichia coli—were the predominant pathogens identified in this pediatric cohort, especially in cases of appendicitis and peritonitis [
18,
19,
20]. The predominance of Enterobacterales supports guideline recommendations advocating empiric antimicrobial regimens with reliable Gram-negative coverage for community-acquired intra-abdominal infections in children.
Polymicrobial infections were identified in nearly one-quarter of cases, particularly among patients with intra-abdominal abscesses and complicated appendicitis. This observation is consistent with Surgical Infection Society (SIS) and ESPID guidance, which highlights the polymicrobial nature of complicated intra-abdominal infections and the necessity for broad-spectrum empiric therapy combined with adequate source control [
20,
21,
22].
The frequent isolation of
Streptococcus spp in mixed infections is clinically significant, as these pathogens are strongly associated with abscess formation and invasive disease. Current guidelines emphasize the importance of considering these organisms when managing localized intra-abdominal infections requiring drainage [
19,
23].
Cephalosporins and combination antibiotic regimens demonstrated the highest overall susceptibility rates in this study, supporting their role as first-line empiric therapy, as recommended by ASHP, IDSA, and WSES guidelines [
18,
20,
24]. However, susceptibility rates slightly exceeding 50% suggest that cephalosporin monotherapy may be insufficient in settings with a high prevalence of polymicrobial infections or resistant organisms.
The low susceptibility to penicillins observed across all pathogen groups limits their utility as empiric monotherapy. It aligns with guideline recommendations discouraging the use of narrow-spectrum agents in complicated intra-abdominal infections [
20,
24]. Moderate carbapenem susceptibility rates indicate emerging resistance among Gram-negative organisms, reinforcing antimicrobial stewardship principles that advocate carbapenem-sparing strategies and reserve their use for severe infections, septic shock, or treatment failure [
18,
25].
Reduced quinolone susceptibility, particularly in mixed infections, further supports current pediatric guidelines that recommend restricting fluoroquinolone use due to safety concerns and
the increasing risk of resistance [
21,
26]. In contrast, Gram-positive organisms retained susceptibility to glycopeptides, lincosamides, and oxazolidinones, consistent with guideline-endorsed targeted therapy for Staphylococcus aureus and Enterococcus spp. infections [
20,
24].
Age-related differences in pathogen distribution were observed: adolescents were predominantly affected by Gram-negative monomicrobial infections, whereas younger children exhibited more heterogeneous, polymicrobial patterns. These findings support guideline statements emphasizing that age, immune status, and disease severity should guide empiric antimicrobial therapy in pediatric patients [
21,
22].
Appendicitis was primarily associated with Gram-negative organisms, whereas abscesses showed increased polymicrobial involvement. This distinction reinforces current recommendations for narrower empiric coverage in uncomplicated appendicitis and broader-spectrum regimens in complicated or perforated disease [
18,
19].
Consistent with WSES and SIS recommendations, effective surgical source control remains the cornerstone of management for pediatric intra-abdominal infections [
18,
20]. The high proportion of appendectomies and drainage procedures in this cohort underscores the essential role of timely surgical intervention in limiting infection severity and duration of antimicrobial exposure.
Despite advances in antimicrobial therapy, spontaneous bacterial peritonitis remains associated with high in-hospital mortality (27.8%) [
27], with fever and systemic inflammatory response syndrome (SIRS) identified as independent predictors of death. These findings emphasize the need for early recognition and prompt initiation of appropriate empirical antibiotic therapy, particularly in the context of increasing antimicrobial resistance [
27].
Our findings support guideline-endorsed strategies advocating short-course, culture-guided postoperative antibiotic therapy following adequate source control, particularly in uncomplicated cases. Prolonged empiric antibiotic use may contribute to antimicrobial resistance without improving clinical outcomes [
18,
24,
28].
4.3. Strengths and Limitations of the Present Study
This study provides comprehensive pediatric-specific data on intra-abdominal infections, integrating microbiological findings with surgical interventions and patient age. The inclusion of both monomicrobial and polymicrobial infections allows a nuanced understanding of pathogen distribution and resistance patterns, which are often underreported in pediatric cohorts. Detailed antimicrobial susceptibility profiling offers actionable guidance for empiric therapy and supports antimicrobial stewardship initiatives. Furthermore, the study provides a foundation for age- and diagnosis-stratified empiric treatment strategies, addressing the critical gap in pediatric-specific recommendations often extrapolated from adult data. By combining microbiological, surgical, and demographic information, this study contributes valuable evidence to guide clinical decision-making in pediatric intra-abdominal infections
A major limitation of this study is the absence of systematic anaerobic pathogen identification. Anaerobic bacteria play a significant role in intra-abdominal infections, particularly in perforated appendicitis and abscess formation, and their exclusion likely resulted in an underestimation of polymicrobial infection rates. Consequently, the antimicrobial susceptibility patterns reported here primarily reflect aerobic and facultative anaerobic organisms and should be interpreted with caution when extrapolating to infections with a strong anaerobic component.
Additional limitations include the single-center design and retrospective methodology, which may limit generalizability and preclude assessment of causality. Nevertheless, the relatively large pediatric cohort and integration of clinical, surgical, and microbiological data represent important strengths of this study.
5. Conclusions
Pediatric intra-abdominal infections associated with surgical intervention demonstrate distinct microbiological patterns influenced by age, clinical diagnosis, and surgical complexity. Gram-negative organisms predominate, particularly in older children and monomicrobial infections, while polymicrobial infections are more frequently associated with younger age groups and complicated disease presentations.
The observed antimicrobial susceptibility patterns support the use of targeted empiric therapy based on clinical severity, with broader-spectrum combination regimens reserved for complicated infections and subsequent de-escalation guided by culture results. These findings reinforce the importance of age- and diagnosis-specific considerations in pediatric antimicrobial stewardship.
Further prospective, multicenter studies incorporating systematic anaerobic pathogen identification are needed to refine empiric treatment strategies and improve outcomes in pediatric intra-abdominal surgical site infections.
Author Contributions
Conceptualization, F.D.E., T.C., V.V.L., V.P., and C.M.M.; methodology T.C. and V.P.; software, T.C. and V.P.; validation A.L.B., A.L., E.A., G.F.P., I.L.S., and I.M.; formal analysis, V.P.; investigation, F.D.E., T.C., A.L.B., G.F.P., I.L.S., R.M.S.,I.M., C.M.M.; resources, T.C., F.D.E., A.L., I.L.S.,; data curation, C.M.M., T.C., A.L.B.; writing— original draft, F.D.E., T.C., G.F.P., I.L.S., V.P., R.M.S., A.L.B., I.M.; writing—review and editing, F.D.E., A.L., T.C., A.L.B., E.A., V.V.L., V.P., R.M.S and C.M.M.; visualization: F.D.E., A.L., T.C., A.L.B., V.V.L., A.L., V.P., R.M.S and C.M.M; supervision, C.M.M.; A.L., T.C., E.A. and R.M.S. contributed equally with F.D.E. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding
Institutional Review Board Statement
This study was conducted following the Declaration of Helsinki and approved by the Faculty of Medicine Ethics Committee, Ovidius University of Constanta. Protocol no 02 was approved on 14.01.2026
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data presented in this study are available on request from the first author and the first corresponding author due to the ethical reasons.
Conflicts of Interest
The authors declare no conflicts of interest
References
- European Centre for Disease Prevention and Control (ECDC). Healthcare-Associated Infections: Surgical Site Infections—Annual Epidemiological Report for 2017; ECDC: Stockholm, Sweden, 2019; Available online: https://www.ecdc.europa.eu/en/publications-data/healthcare-associated-infections-surgical-site-infections-annual-1 (accessed on 20 December 2025).
- Centers for Disease Control and Prevention (CDC). Surgical Site Infections. Available online: https://www.cdc.gov/infection-control/hcp/surgical-site-infection/index.html (accessed on 20 December 2025).
- Donskey, C.J.; Chowdhry, T.K.; Hecker, M.T.; Hoyen, C.K.; Hanrahan, J.A.; Hujer, A.M.; Hutton-Thomas, R.A.; Whalen, C.C.; Bonomo, R.A.; Rice, L.B. Effect of antibiotic therapy on the density of vancomycin-resistant enterococci in the stool of colonized patients. N. Engl. J. Med. 2000, 343, 1925–1932. [Google Scholar] [CrossRef]
- Louie, T. J.; Chubb, H.; Bow, E. J.; Conly, J. M.; Harding, G. K.; Rayner, E.; James, M. Preservation of colonization resistance parameters during empiric therapy with aztreonam in the febrile neutropenic patient. Reviews of infectious diseases 1985, 7 Suppl 4, S747–S761. [Google Scholar] [CrossRef]
- Samonis, G.; Gikas, A.; Anaissie, E. J.; Vrenzos, G.; Maraki, S.; Tselentis, Y.; Bodey, G. P. Prospective evaluation of effects of broad-spectrum antibiotics on gastrointestinal yeast colonization of humans. Antimicrobial agents and chemotherapy 1993, 37(1), 51–53. [Google Scholar] [CrossRef]
- Cosgrove, S.E. The relationship between antimicrobial resistance and patient outcomes: Mortality, length of hospital stay, and health care costs. Clin. Infect. Dis. 2006, 42, S82–S89. [Google Scholar] [CrossRef]
- Maragakis, L.L.; Perencevich, E.N.; Cosgrove, S.E. Clinical and economic burden of antimicrobial resistance. Expert Rev. Anti Infect. Ther. 2008, 6, 751–763. [Google Scholar] [CrossRef] [PubMed]
- Kociolek, L.; Patel, S.; Shulman, S.; Zheng, X.; Todd, K.; Gerding, D.N. Clinical and microbiologic assessment of pediatric community-associated Clostridioides difficile infection reveals opportunities for improved testing decisions. Pediatr. Infect. Dis. J. 2016, 35, 157–161. [Google Scholar] [CrossRef]
- Allegranzi, B.; Bischoff, P.; de Jonge, S.; Kubilay, N.Z.; Zayed, B.; Gomes, S.M.; Abbas, M.; Atema, J.J.; Gans, S.; van Rijen, M.; et al. New WHO recommendations on preoperative measures for surgical site infection prevention: An evidence-based global perspective. Lancet Infect. Dis. 2016, 16, e276–e287. [Google Scholar] [CrossRef]
- Allegranzi, B.; Zayed, B.; Bischoff, P.; Kubilay, N.Z.; de Jonge, S.; de Vries, F.; Gomes, S.M.; Gans, S.; Wallert, E.D.; Wu, X.; et al. New WHO recommendations on intraoperative and postoperative measures for surgical site infection prevention: An evidence-based global perspective. Lancet Infect. Dis. 2016, 16, e288–e303. [Google Scholar] [CrossRef] [PubMed]
- Ban, K.A.; Minei, J.P.; Laronga, C.; Harbrecht, B.G.; Jensen, E.; Fry, D.E.; Itani, K.M.; Dellinger, E.P.; Ko, C.Y.; Duane, T.M. American College of Surgeons and Surgical Infection Society: Surgical site infection guidelines, 2016 update. J. Am. Coll. Surg. 2017, 224, 59–74. [Google Scholar] [CrossRef] [PubMed]
- Berríos-Torres, S.I.; Umscheid, C.A.; Bratzler, D.W.; Leas, B.; Stone, E.C.; Kelz, R.R.; Reinke, C.E.; Morgan, S.; Solomkin, J.S.; Mazuski, J.E.; et al. Centers for Disease Control and Prevention guideline for the prevention of surgical site infection, 2017. JAMA Surg. 2017, 152, 784–791. [Google Scholar] [CrossRef]
- European Centre for Disease Prevention and Control (ECDC). Surveillance of Surgical Site Infections in Europe 2010–2011. Available online: https://www.ecdc.europa.eu/sites/default/files/media/en/publications/Publications/SSI-in-europe-2010-2011.pdf (accessed on 10 December 2025).
- GlobalSurg Collaborative. Surgical site infection after gastrointestinal surgery in children: An international, multicentre, prospective cohort study. BMJ Glob. Health 2020, 5, e003429. [Google Scholar] [CrossRef] [PubMed]
- Gorter, R.R.; Eker, H.H.; Gorter-Stam, M.A.W.; Abis, G.S.A.; Acharya, A.; Ankersmit, M.; et al. Diagnosis and management of acute appendicitis: EAES consensus development conference 2015. Surg. Endosc. 2016, 30, 4668–4690. [Google Scholar] [CrossRef]
- Bratzler, D.W.; Dellinger, E.P.; Olsen, K.M.; Perl, T.M.; Auwaerter, P.G.; Bolon, M.K.; et al. Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am. J. Health Syst. Pharm. 2013, 70, 195–283. [Google Scholar] [CrossRef] [PubMed]
- Goovaerts, C.; Miserez, M.; van Hoeve, K.; Hoffman, I. Antibiotics post-appendectomy in pediatric patients: A literature review leading to proposition of a new treatment protocol. Belg. J. Paediatr. 2024, 26, 194–204. Available online: https://www.belgjpaediatrics.com/index.php/bjp/article/view/319/432.
- Sartelli, M.; Chichom-Mefire, A.; Labricciosa, F. M.; Hardcastle, T.; Abu-Zidan, F. M.; Adesunkanmi, A. K.; Ansaloni, L.; Bala, M.; Balogh, Z. J.; Beltrán, M. A.; Ben-Ishay, O.; Biffl, W. L.; Birindelli, A.; Cainzos, M. A.; Catalini, G.; Ceresoli, M.; Che Jusoh, A.; Chiara, O.; Coccolini, F.; Coimbra, R.; Catena, F. The management of intra-abdominal infections from a global perspective: 2017 WSES guidelines for management of intra-abdominal infections. World journal of emergency surgery: WJES 2017, 12, 29. [Google Scholar] [CrossRef]
- Di Saverio, S.; Podda, M.; De Simone, B.; Ceresoli, M.; Augustin, G.; Gori, A.; Boermeester, M.; Sartelli, M.; Coccolini, F.; Tarasconi, A.; De' Angelis, N.; Weber, D. G.; Tolonen, M.; Birindelli, A.; Biffl, W.; Moore, E. E.; Kelly, M.; Soreide, K.; Kashuk, J.; Ten Broek, R.; Catena, F. Diagnosis and treatment of acute appendicitis: 2020 update of the WSES Jerusalem guidelines. World journal of emergency surgery: WJES 2020, 15(1), 27. [Google Scholar] [CrossRef]
- Solomkin, Joseph S.; Mazuski, John E.; Bradley, John S.; Rodvold, Keith A; Goldstein, Ellie J.C.; Baron, Ellen J.; O'Neill, Patrick J.; Chow, Anthony W.; Dellinger, E. Patchen; Eachempati, Soumitra R.; Gorbach, Sherwood; Hilfiker, Mary; May, Addison K.; Nathens, Avery B.; Sawyer, Robert G.; Bartlett, John G. Diagnosis and Management of Complicated Intra-abdominal Infection in Adults and Children: Guidelines by the Surgical Infection Society and the Infectious Diseases Society of America. Clinical Infectious Diseases, Volume 50, Issue 2, 15 January 2010, Pages 133–164. [CrossRef]
- Bradley, J. S.; Byington, C. L.; Shah, S. S.; Alverson, B.; Carter, E. R.; Harrison, C.; Kaplan, S. L.; Mace, S. E.; McCracken, G. H., Jr.; Moore, M. R.; St Peter, S. D.; Stockwell, J. A.; Swanson, J. T.; Pediatric Infectious Diseases Society and the Infectious Diseases Society of America. The management of community-acquired pneumonia in infants and children older than 3 months of age: clinical practice guidelines by the Pediatric Infectious Diseases Society and the Infectious Diseases Society of America. Clinical infectious diseases: an official publication of the Infectious Diseases Society of America 2011, 53(7), e25–e76. [Google Scholar] [CrossRef]
- Bielicki, J.; Lundin, R.; Patel, S.; Paulus, S. Antimicrobial stewardship for neonates and children: a global approach. The Pediatric infectious disease journal 2015, 34(3), 311–313. [Google Scholar] [CrossRef] [PubMed]
- Whiley, R. A.; Beighton, D. Current classification of the oral streptococci. Oral microbiology and immunology 1998, 13(4), 195–216. [Google Scholar] [CrossRef]
- Bratzler, D. W.; Dellinger, E. P.; Olsen, K. M.; Perl, T. M.; Auwaerter, P. G.; Bolon, M. K.; Fish, D. N.; Napolitano, L. M.; Sawyer, R. G.; Slain, D.; Steinberg, J. P.; Weinstein, R. A.; American Society of Health-System Pharmacists; Infectious Disease Society of America; Surgical Infection Society; Society for Healthcare Epidemiology of America. Clinical practice guidelines for antimicrobial prophylaxis in surgery. American journal of health-system pharmacy: AJHP: official journal of the American Society of Health-System Pharmacists 2013, 70(3), 195–283. [Google Scholar] [CrossRef]
- Branstetter, J. W.; Barker, L.; Yarbrough, A.; Ross, S.; Stultz, J. S. Challenges of Antibiotic Stewardship in the Pediatric and Neonatal Intensive Care Units. The journal of pediatric pharmacology and therapeutics: JPPT: the official journal of PPAG 2021, 26(7), 659–668. [Google Scholar] [CrossRef]
- Choi, S. H.; Kim, E. Y.; Kim, Y. J. Systemic use of fluoroquinolone in children. Korean journal of pediatrics 2013, 56(5), 196–201. [Google Scholar] [CrossRef] [PubMed]
- Popoiag, R; Panaitescu, E; Suceveanu, A; Suceveanu, A; Micu, SI; Mazilu, L; Parepa, I; Voinea, F; Costea, DO; Enache, F; Enache, F. Spontaneous bacterial peritonitis mortality trends of cirrhotic patients in the last decade in Constanta County. Experimental and Therapeutic Medicine 2021, 22, 732. [Google Scholar] [CrossRef] [PubMed]
- Sawyer, R. G.; Claridge, J. A.; Nathens, A. B.; Rotstein, O. D.; Duane, T. M.; Evans, H. L.; Cook, C. H.; O'Neill, P. J.; Mazuski, J. E.; Askari, R.; Wilson, M. A.; Napolitano, L. M.; Namias, N.; Miller, P. R.; Dellinger, E. P.; Watson, C. M.; Coimbra, R.; Dent, D. L.; Lowry, S. F.; Cocanour, C. S.; STOP-IT Trial Investigators. Trial of short-course antimicrobial therapy for intraabdominal infection. The New England journal of medicine 2015, 372(21), 1996–2005. [Google Scholar] [CrossRef] [PubMed]
Table 1.
Baseline data of pediatric patients.
Table 1.
Baseline data of pediatric patients.
| Parameter |
Total |
F |
M |
Chi-Square test |
| N |
% |
n |
% |
n |
% |
p-value |
V |
| 177 |
100 |
64 |
36.16 |
113 |
63.84 |
< 0.0001 |
1 |
| Age group |
| < 1 year |
12 |
6.78 |
4 |
33.33 |
8 |
66.67 |
0.860 |
0.088 |
| 1-3 years |
21 |
11.86 |
8 |
38.10 |
13 |
61.90 |
| 4-6 years |
27 |
15.25 |
9 |
33.33 |
18 |
66.67 |
| 7-10 years |
39 |
22.03 |
17 |
43.59 |
22 |
56.41 |
| 11-16 years |
78 |
44.07 |
26 |
33.33 |
52 |
66.67 |
| SSI |
| Abscess |
39 |
22.03 |
15 |
38.46 |
24 |
61.54 |
0.460 |
0.273 |
| Acute abdomen |
1 |
0.56 |
0 |
0.00 |
1 |
100.00 |
| Adenitis |
3 |
1.69 |
1 |
33.33 |
2 |
66.67 |
| Appendicitis |
97 |
54.80 |
37 |
38.14 |
60 |
61.86 |
| Evisceration |
2 |
1.13 |
2 |
100.00 |
0 |
0.00 |
| Inguinal hernia |
1 |
0.56 |
0 |
0.00 |
1 |
100.00 |
| Intestinal invagination |
1 |
0.56 |
1 |
100.00 |
0 |
0.00 |
| Intestinal occlusion |
2 |
1.13 |
0 |
0.00 |
2 |
100.00 |
| Intestinal perforation |
3 |
1.69 |
0 |
0.00 |
3 |
100.00 |
| Mesenteric hernia |
1 |
0.56 |
0 |
0.00 |
1 |
100.00 |
| Ovarian tumor |
1 |
0.56 |
1 |
100.00 |
0 |
0.00 |
| Peritonitis |
7 |
3.95 |
2 |
28.57 |
5 |
71.43 |
| Pyloric stenosis |
1 |
0.56 |
0 |
0.00 |
1 |
100.00 |
| Others |
18 |
10.17 |
5 |
27.78 |
13 |
72.22 |
Table 2.
Associations between pediatric patients' age and SSI type.
Table 2.
Associations between pediatric patients' age and SSI type.
|
Age group |
< 1 year |
1-3 years |
4-6 years |
7-10 years |
11-16 years |
| SSI type |
|
p-value
|
r*ij |
p-value
|
r*ij |
p-value
|
r*ij |
p-value
|
r*ij |
p-value
|
r*ij |
| Abscess |
<0.0001 |
4.585 |
<0.0001 |
8.060 |
0.135 |
1.539 |
<0.0001 |
–3.760 |
<0.0001 |
–5.547 |
| Acute abdomen |
1.000 |
–0.270 |
1.000 |
–0.368 |
1.000 |
–0.425 |
1.000 |
–0.533 |
0.441 |
1.130 |
| Adenitis |
1.000 |
–0.471 |
1.000 |
–0.641 |
0.061 |
2.498 |
1.000 |
–0.929 |
1.000 |
–0.378 |
| Appendicitis |
<0.0001 |
–3.951 |
<0.0001 |
–5.375 |
0.000 |
–3.695 |
<0.0001 |
6.423 |
0.006 |
2.815 |
| Evisceration |
1.000 |
–0.384 |
1.000 |
–0.522 |
1.000 |
–0.603 |
1.000 |
–0.756 |
0.193 |
1.602 |
| Inguinal hernia |
1.000 |
–0.270 |
1.000 |
–0.368 |
1.000 |
–0.425 |
1.000 |
–0.533 |
0.441 |
1.130 |
| Intestinal invagination |
1.000 |
–0.270 |
1.000 |
–0.368 |
1.000 |
–0.425 |
1.000 |
–0.533 |
0.441 |
1.130 |
| Intestinal occlusion |
1.000 |
–0.384 |
1.000 |
–0.522 |
1.000 |
–0.603 |
1.000 |
–0.756 |
0.193 |
1.602 |
| Intestinal perforation |
1.000 |
–0.471 |
1.000 |
–0.641 |
0.393 |
0.878 |
1.000 |
–0.929 |
0.584 |
0.795 |
| Mesenteric hernia |
1.000 |
–0.270 |
1.000 |
–0.368 |
1.000 |
–0.425 |
1.000 |
–0.533 |
0.441 |
1.130 |
| Others |
0.108 |
1.760 |
1.000 |
–0.104 |
0.000 |
4.326 |
0.014 |
–2.380 |
0.077 |
–1.970 |
| Ovarian tumor |
1.000 |
–0.270 |
1.000 |
–0.368 |
1.000 |
–0.425 |
1.000 |
–0.533 |
0.441 |
1.130 |
| Peritonitis |
1.000 |
–0.728 |
1.000 |
–0.991 |
0.597 |
–1.145 |
0.350 |
–1.435 |
0.003 |
3.041 |
| Pyloric stenosis |
1.000 |
–0.270 |
1.000 |
–0.368 |
1.000 |
–0.425 |
1.000 |
–0.533 |
0.441 |
1.130 |
Table 3.
Specimens collected during surgical intervention, associated with SSI type, and bacterial pathogen groups.
Table 3.
Specimens collected during surgical intervention, associated with SSI type, and bacterial pathogen groups.
| Specimen |
Peritoneal fluid |
Pus |
Other |
Chi-square test |
| |
n |
% |
n |
% |
n |
% |
p-value
|
V |
| SI type |
| Appendectomy |
101 |
84.87 |
0 |
0.00 |
0 |
0.00 |
<0.0001 |
1 |
| Drainage |
1 |
0.84 |
2 |
3.85 |
1 |
16.67 |
| Excision and drainage |
1 |
0.84 |
3 |
5.77 |
2 |
33.33 |
| Exploratory laparotomy |
11 |
9.24 |
0 |
0.00 |
0 |
0.00 |
| Incision and drainage |
0 |
0.00 |
47 |
90.38 |
2 |
33.33 |
| Surgical correction |
2 |
1.68 |
0 |
0.00 |
0 |
0.00 |
| Suture |
3 |
2.52 |
0 |
0.00 |
0 |
0.00 |
| Thoracotomy |
0 |
0.00 |
0 |
0.00 |
1 |
16.67 |
| SSI |
| Abscess |
2 |
1.68 |
36 |
69.23 |
1 |
16.67 |
<0.0001 |
1 |
| Acute abdomen |
1 |
0.84 |
0 |
0.00 |
0 |
0.00 |
| Adenitis |
0 |
0.00 |
2 |
3.85 |
1 |
16.67 |
| Appendicitis |
97 |
81.51 |
0 |
0.00 |
0 |
0.00 |
| Evisceration |
2 |
1.68 |
0 |
0.00 |
0 |
0.00 |
| Inguinal hernia |
1 |
0.84 |
0 |
0.00 |
0 |
0.00 |
| Intestinal invagination |
1 |
0.84 |
0 |
0.00 |
0 |
0.00 |
| Intestinal occlusion |
2 |
1.68 |
0 |
0.00 |
0 |
0.00 |
| Intestinal perforation |
3 |
2.52 |
0 |
0.00 |
0 |
0.00 |
| Mesenteric hernia |
1 |
0.84 |
0 |
0.00 |
0 |
0.00 |
| Others |
0 |
0.00 |
14 |
26.92 |
4 |
66.67 |
| Ovarian tumor |
1 |
0.84 |
0 |
0.00 |
0 |
0.00 |
| Peritonitis |
7 |
5.88 |
0 |
0.00 |
0 |
0.00 |
| Pyloric stenosis |
1 |
0.84 |
0 |
0.00 |
0 |
0.00 |
| Pathogen type |
| 1GN |
55 |
46,22 |
19 |
36,54 |
4 |
66,67 |
0.613 |
0.156 |
| 2GN |
13 |
10,92 |
4 |
7,69 |
0 |
0,00 |
| 1GN+1GP |
13 |
10,92 |
7 |
13,46 |
1 |
16,67 |
| 2GN+1GP |
1 |
0,84 |
1 |
1,92 |
0 |
0,00 |
| 1GP |
34 |
28,57 |
20 |
38,46 |
1 |
16,67 |
| 1GP+1GN |
3 |
2,52 |
0 |
0,00 |
0 |
0,00 |
| 1GN+2GP |
0 |
0,00 |
1 |
1,92 |
0 |
0,00 |
Table 4.
Associations between biological specimens collected from pediatric patients and SI and SSI types.
Table 4.
Associations between biological specimens collected from pediatric patients and SI and SSI types.
| Aspect |
Peritoneal fluid |
Pus |
Other fluids |
|
p-value
|
r*ij |
p-value
|
r*ij |
p-value
|
r*ij |
| SI type |
| Appendectomy |
<0.0001 |
10.707 |
<0.0001 |
–9.892 |
0.006 |
–2.873 |
| Drainage |
0.104 |
–1.820 |
0.582 |
0.916 |
0.130 |
2.416 |
| Excision and drainage |
0.015 |
–2.685 |
0.361 |
1.128 |
0.014 |
4.124 |
| Exploratory laparotomy |
0.017 |
2.391 |
0.035 |
–2.209 |
1.000 |
–0.642 |
| Incision and drainage |
<0.0001 |
–11.791 |
<0.0001 |
12.025 |
0.669 |
0.315 |
| Surgical correction |
1.000 |
0.993 |
1.000 |
–0.917 |
1.000 |
–0.266 |
| Suture |
0.552 |
1.220 |
0.556 |
–1.127 |
1.000 |
–0.327 |
| Thoracotomy |
0.328 |
–1.436 |
1.000 |
–0.647 |
0.034 |
5.354 |
| SSI type |
| Abscess |
<0.0001 |
–9.358 |
<0.0001 |
9.771 |
1.000 |
–0.323 |
| Acute abdomen |
1.000 |
0.700 |
1.000 |
–0.647 |
1.000 |
–0.188 |
| Adenitis |
0.034 |
–2.502 |
0.207 |
1.430 |
0.099 |
2.891 |
| Appendicitis |
<0.0001 |
10.227 |
<0.0001 |
–9.449 |
0.008 |
–2.744 |
| Evisceration |
1.000 |
0.993 |
1.000 |
–0.917 |
1.000 |
–0.266 |
| Inguinal hernia |
1.000 |
0.700 |
1.000 |
–0.647 |
1.000 |
–0.188 |
| Intestinal invagination |
1.000 |
0.700 |
1.000 |
–0.647 |
1.000 |
–0.188 |
| Intestinal occlusion |
1.000 |
0.993 |
1.000 |
–0.917 |
1.000 |
–0.266 |
| Intestinal perforation |
0.552 |
1.220 |
0.556 |
–1.127 |
1.000 |
–0.327 |
| Mesenteric hernia |
1.000 |
0.700 |
1.000 |
–0.647 |
1.000 |
–0.188 |
| Others |
<0.0001 |
–6.412 |
<0.0001 |
4.756 |
0.001 |
4.658 |
| Ovarian tumor |
1.000 |
0.700 |
1.000 |
–0.647 |
1.000 |
–0.188 |
| Peritonitis |
0.097 |
1.885 |
0.107 |
–1.741 |
1.000 |
–0.506 |
| Pyloric stenosis |
1.000 |
0.700 |
1.000 |
–0.647 |
1.000 |
–0.188 |
Table 5.
Associations between SI types and SSI types in pediatric patients.
Table 5.
Associations between SI types and SSI types in pediatric patients.
|
SI |
Appendectomy |
Drainage |
Excision and drainage |
Exploratory laparotomy |
Incision and drainage |
Surgical correction |
Suture |
Thoracotomy |
| SSI |
|
|
p-value
|
| Abscess |
<0.0001 |
0.211 |
0.341 |
0.460 |
<0.0001 |
1.000 |
1.000 |
1.000 |
| Acute abdomen |
0.429 |
1.000 |
1.000 |
0.062 |
1.000 |
1.000 |
1.000 |
1.000 |
| Adenitis |
0.077 |
1.000 |
1.000 |
1.000 |
0.020 |
1.000 |
1.000 |
1.000 |
| Appendicitis |
<0.0001 |
0.040 |
0.008 |
0.000 |
<0.0001 |
0.203 |
0.090 |
0.452 |
| Evisceration |
0.183 |
1.000 |
1.000 |
0.121 |
1.000 |
1.000 |
0.034 |
1.000 |
| Inguinal hernia |
0.429 |
1.000 |
1.000 |
1.000 |
1.000 |
0.011 |
1.000 |
1.000 |
| Intestinal invagination |
0.429 |
1.000 |
1.000 |
0.062 |
1.000 |
1.000 |
1.000 |
1.000 |
| Intestinal occlusion |
0.183 |
1.000 |
1.000 |
0.004 |
1.000 |
1.000 |
1.000 |
1.000 |
| Intestinal perforation |
0.077 |
1.000 |
1.000 |
0.010 |
0.562 |
1.000 |
0.050 |
1.000 |
| Mesenteric hernia |
0.429 |
1.000 |
1.000 |
1.000 |
1.000 |
0.011 |
1.000 |
1.000 |
| Others |
<0.0001 |
0.052 |
<0.0001 |
0.606 |
0.010 |
1.000 |
1.000 |
0.102 |
| Ovarian tumor |
0.429 |
1.000 |
0.034 |
1.000 |
1.000 |
1.000 |
1.000 |
1.000 |
| Peritonitis |
1.000 |
1.000 |
1.000 |
0.062 |
0.192 |
1.000 |
0.115 |
1.000 |
| Pyloric stenosis |
0.429 |
1.000 |
1.000 |
0.062 |
1.000 |
1.000 |
1.000 |
1.000 |
| r*ij |
| Abscess |
–8.153 |
1.365 |
–1.325 |
–1.069 |
10.215 |
–0.756 |
–0.929 |
–0.533 |
| Acute abdomen |
–1.156 |
–0.152 |
–0.188 |
3.896 |
–0.620 |
–0.107 |
–0.132 |
–0.076 |
| Adenitis |
–2.014 |
–0.266 |
–0.327 |
–0.450 |
2.823 |
–0.187 |
–0.229 |
–0.132 |
| Appendicitis |
12.708 |
–2.228 |
–2.744 |
–3.771 |
–9.064 |
–1.566 |
–1.924 |
–1.104 |
| Evisceration |
–1.640 |
–0.216 |
–0.266 |
2.579 |
–0.880 |
–0.152 |
5.322 |
–0.107 |
| Inguinal hernia |
–1.156 |
–0.152 |
–0.188 |
–0.258 |
–0.620 |
9.381 |
–0.132 |
–0.076 |
| Intestinal invagination |
–1.156 |
–0.152 |
–0.188 |
3.896 |
–0.620 |
–0.107 |
–0.132 |
–0.076 |
| Intestinal occlusion |
–1.640 |
–0.216 |
–0.266 |
5.525 |
–0.880 |
–0.152 |
–0.187 |
–0.107 |
| Intestinal perforation |
–2.014 |
–0.266 |
–0.327 |
4.374 |
–1.081 |
–0.187 |
4.282 |
–0.132 |
| Mesenteric hernia |
–1.156 |
–0.152 |
–0.188 |
–0.258 |
–0.620 |
9.381 |
–0.132 |
–0.076 |
| Others |
–5.160 |
2.666 |
6.033 |
–1.152 |
2.788 |
–0.479 |
–0.588 |
2.981 |
| Ovarian tumor |
–1.156 |
–0.152 |
5.354 |
–0.258 |
–0.620 |
–0.107 |
–0.132 |
–0.076 |
| Peritonitis |
0.004 |
–0.411 |
–0.506 |
2.500 |
–1.670 |
–0.289 |
2.633 |
–0.203 |
| Pyloric stenosis |
–1.156 |
–0.152 |
–0.188 |
3.896 |
–0.620 |
–0.107 |
–0.132 |
–0.076 |
Table 6.
Bacterial pathogens identified in pediatric patients' specimens.
Table 6.
Bacterial pathogens identified in pediatric patients' specimens.
| Bacteria |
n |
% |
| 1GN |
| E. coli |
65 |
83.33 |
| Klebsiella spp |
5 |
6.41 |
| P. aeruginosa |
5 |
6.41 |
| Enterobacter spp |
2 |
2.56 |
| 2GN |
| E. coli + P. aeruginosa |
9 |
52.94 |
| E. coli + Klebsiella spp |
3 |
17.65 |
| E. coli + A. baumannii |
2 |
11.76 |
| C. freundii + Enterobacter spp |
1 |
5.88 |
| Enterobacter spp + Klebsiella spp |
1 |
5.88 |
| 1GN+1GP |
| E. coli + Streptococcus spp |
11 |
52.38 |
| E. coli + Enterococcus spp |
5 |
23.81 |
| E. Coli + S. aureus |
2 |
9.52 |
| C. freundii + Enterococcus spp |
1 |
4.76 |
| H. alvei + Streptococcus spp |
1 |
4.76 |
| Klebsiella spp + Streptococcus spp |
1 |
4.76 |
| 2GN+1GP |
| E. coli + P. aeruginosa + Streptococcus spp |
2 |
100.00 |
| 1GN+2GP |
| E. coli + Enterococcus spp + Streptococcus spp |
1 |
100.00 |
| 1GP+1GN |
| Enterococcus spp + Enterobacter spp |
1 |
33.33 |
| Enterococcus spp + Klebsiella spp |
1 |
33.33 |
| Enterococcus spp + P. aeruginosa |
1 |
33.33 |
| 1GP |
| S. aureus |
33 |
60.00 |
| Enterococcus spp |
13 |
23.64 |
| Streptococcus spp |
8 |
14.55 |
| Gemella morbillum |
1 |
1.82 |
Table 7.
Associations between Gram-negative bacterial pathogens and SSI types in pediatric patients.
Table 7.
Associations between Gram-negative bacterial pathogens and SSI types in pediatric patients.
| 1GN |
E. coli |
Enterobacterspp
|
Klebsiellaspp
|
P. aeruginosa |
| SSI type |
p-value
|
r*ij |
p-value
|
r*ij |
p-value
|
r*ij |
p-value
|
r*ij |
| Abscess |
0.575 |
0.631 |
1.000 |
-0.756 |
0.588 |
-1.206 |
0.588 |
-1.206 |
| Acute abdomen |
1.000 |
-0.764 |
1.000 |
-0.107 |
1.000 |
-0.171 |
1.000 |
-0.171 |
| Adenitis |
1.000 |
-0.123 |
1.000 |
-0.187 |
1.000 |
-0.298 |
1.000 |
-0.298 |
| Appendicitis |
0.000 |
3.878 |
0.203 |
-1.566 |
0.018 |
-2.498 |
0.018 |
-2.498 |
| Evisceration |
0.533 |
-1.084 |
1.000 |
-0.152 |
1.000 |
-0.242 |
1.000 |
-0.242 |
| Inguinal hernia |
1.000 |
-0.764 |
1.000 |
-0.107 |
1.000 |
-0.171 |
1.000 |
-0.171 |
| Intestinal invagination |
1.000 |
-0.764 |
1.000 |
-0.107 |
1.000 |
-0.171 |
1.000 |
-0.171 |
| Intestinal occlusion |
0.533 |
-1.084 |
1.000 |
-0.152 |
1.000 |
-0.242 |
1.000 |
-0.242 |
| Intestinal perforation |
0.299 |
-1.331 |
1.000 |
-0.187 |
1.000 |
-0.298 |
1.000 |
-0.298 |
| Mesenteric hernia |
1.000 |
-0.764 |
1.000 |
-0.107 |
1.000 |
-0.171 |
1.000 |
-0.171 |
| Others |
0.000 |
-3.410 |
0.010 |
4.227 |
0.000 |
5.241 |
1.000 |
-0.763 |
| Ovarian tumor |
1.000 |
-0.764 |
1.000 |
-0.107 |
0.028 |
5.882 |
1.000 |
-0.171 |
| Peritonitis |
0.048 |
-2.057 |
1.000 |
-0.289 |
1.000 |
-0.460 |
<0.0001 |
8.851 |
| Pyloric stenosis |
1.000 |
-0.764 |
1.000 |
-0.107 |
1.000 |
-0.171 |
0.028 |
5.882 |
Table 8.
Associations between Gram-positive bacterial pathogens and SSI types in pediatric patients.
Table 8.
Associations between Gram-positive bacterial pathogens and SSI types in pediatric patients.
| 1GP |
Enterococcusspp
|
S. aureus |
Streptococcusspp
|
| SSI |
p-value
|
r*ij |
p-value
|
r*ij |
p-value
|
r*ij |
| Abscess |
0.303 |
-1.296 |
0.103 |
1.736 |
0.203 |
-1.539 |
| Acute abdomen |
1.000 |
-0.282 |
0.186 |
2.095 |
1.000 |
-0.218 |
| Adenitis |
1.000 |
-0.492 |
1.000 |
-0.836 |
1.000 |
-0.380 |
| Appendicitis |
0.021 |
-2.388 |
0.333 |
1.130 |
0.001 |
-3.187 |
| Evisceration |
1.000 |
-0.400 |
1.000 |
-0.681 |
0.089 |
3.114 |
| Inguinal hernia |
0.073 |
3.562 |
1.000 |
-0.480 |
1.000 |
-0.218 |
| Intestinal invagination |
1.000 |
-0.282 |
1.000 |
-0.480 |
1.000 |
-0.218 |
| Intestinal occlusion |
0.005 |
5.052 |
1.000 |
-0.681 |
1.000 |
-0.309 |
| Intestinal perforation |
0.014 |
3.973 |
1.000 |
-0.836 |
1.000 |
-0.380 |
| Mesenteric hernia |
1.000 |
-0.282 |
1.000 |
-0.480 |
0.045 |
4.609 |
| Others |
0.030 |
2.553 |
0.027 |
-2.143 |
0.000 |
5.012 |
| Ovarian tumor |
1.000 |
-0.282 |
1.000 |
-0.480 |
1.000 |
-0.218 |
| Peritonitis |
1.000 |
-0.760 |
0.351 |
-1.292 |
0.281 |
1.269 |
| Pyloric stenosis |
1.000 |
-0.282 |
1.000 |
-0.480 |
1.000 |
-0.218 |
Table 9.
Bacterial susceptibility (laboratory results).
Table 9.
Bacterial susceptibility (laboratory results).
| |
Total |
1GN |
2GN |
1GP |
| n |
% |
n |
% |
n |
% |
n |
% |
| Penicillines |
8 |
4.52 |
4 |
5.13 |
1 |
5.88 |
2 |
3.64 |
| Cephalosporines |
95 |
53.67 |
40 |
51.28 |
10 |
58.82 |
31 |
56.36 |
| Monobactams |
80 |
45.20 |
31 |
39.74 |
7 |
41.18 |
23 |
41.82 |
| Quinolones |
46 |
25.99 |
26 |
33.33 |
6 |
35.29 |
13 |
23.64 |
| Carbapenems |
34 |
19.21 |
17 |
21.79 |
7 |
41.18 |
8 |
14.55 |
| Aminoglycosides |
72 |
40.68 |
33 |
42.31 |
9 |
52.94 |
19 |
34.55 |
| Combination |
92 |
51.98 |
38 |
48.72 |
8 |
47.06 |
25 |
45.45 |
| Macrolides |
10 |
5.65 |
6 |
7.69 |
0 |
0.00 |
2 |
3.64 |
| Tetracyclines |
14 |
7.91 |
9 |
11.54 |
1 |
5.88 |
4 |
7.27 |
| Peptides (glico. poli) |
21 |
11.86 |
12 |
15.38 |
2 |
11.76 |
6 |
10.91 |
| Lyncosamydes |
18 |
10.17 |
10 |
12.82 |
0 |
0.00 |
8 |
14.55 |
| Steroids |
30 |
16.95 |
14 |
17.95 |
0 |
0.00 |
15 |
27.27 |
| Oxazolidinones |
23 |
12.99 |
12 |
15.38 |
3 |
17.65 |
7 |
12.73 |
| |
1GN+1GP |
1GP+1GN |
2GN+1GP |
1GN+2GP |
| n |
% |
n |
% |
n |
% |
n |
% |
| Penicillines |
1 |
4.76 |
0 |
0.00 |
0 |
0.00 |
0 |
0.00 |
| Cephalosporines |
12 |
57.14 |
1 |
33.33 |
0 |
0.00 |
1 |
100.00 |
| Monobactams |
14 |
66.67 |
2 |
66.67 |
2 |
100.00 |
1 |
100.00 |
| Quinolones |
1 |
4.76 |
0 |
0.00 |
0 |
0.00 |
0 |
0.00 |
| Carbapenems |
2 |
9.52 |
0 |
0.00 |
0 |
0.00 |
0 |
0.00 |
| Aminoglycosides |
7 |
33.33 |
2 |
66.67 |
2 |
100.00 |
0 |
0.00 |
| Combination |
16 |
76.19 |
2 |
66.67 |
2 |
100.00 |
1 |
100.00 |
| Macrolides |
2 |
9.52 |
0 |
0.00 |
0 |
0.00 |
0 |
0.00 |
| Tetracyclines |
0 |
0.00 |
0 |
0.00 |
0 |
0.00 |
0 |
0.00 |
| Peptides (glico. poli) |
1 |
4.76 |
0 |
0.00 |
0 |
0.00 |
0 |
0.00 |
| Lyncosamydes |
0 |
0.00 |
0 |
0.00 |
0 |
0.00 |
0 |
0.00 |
| Steroids |
0 |
0.00 |
1 |
33.33 |
0 |
0.00 |
0 |
0.00 |
| Oxazolidinones |
0 |
0.00 |
1 |
33.33 |
0 |
0.00 |
0 |
0.00 |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).






