Submitted:
02 February 2026
Posted:
04 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Phthalates: An Overview
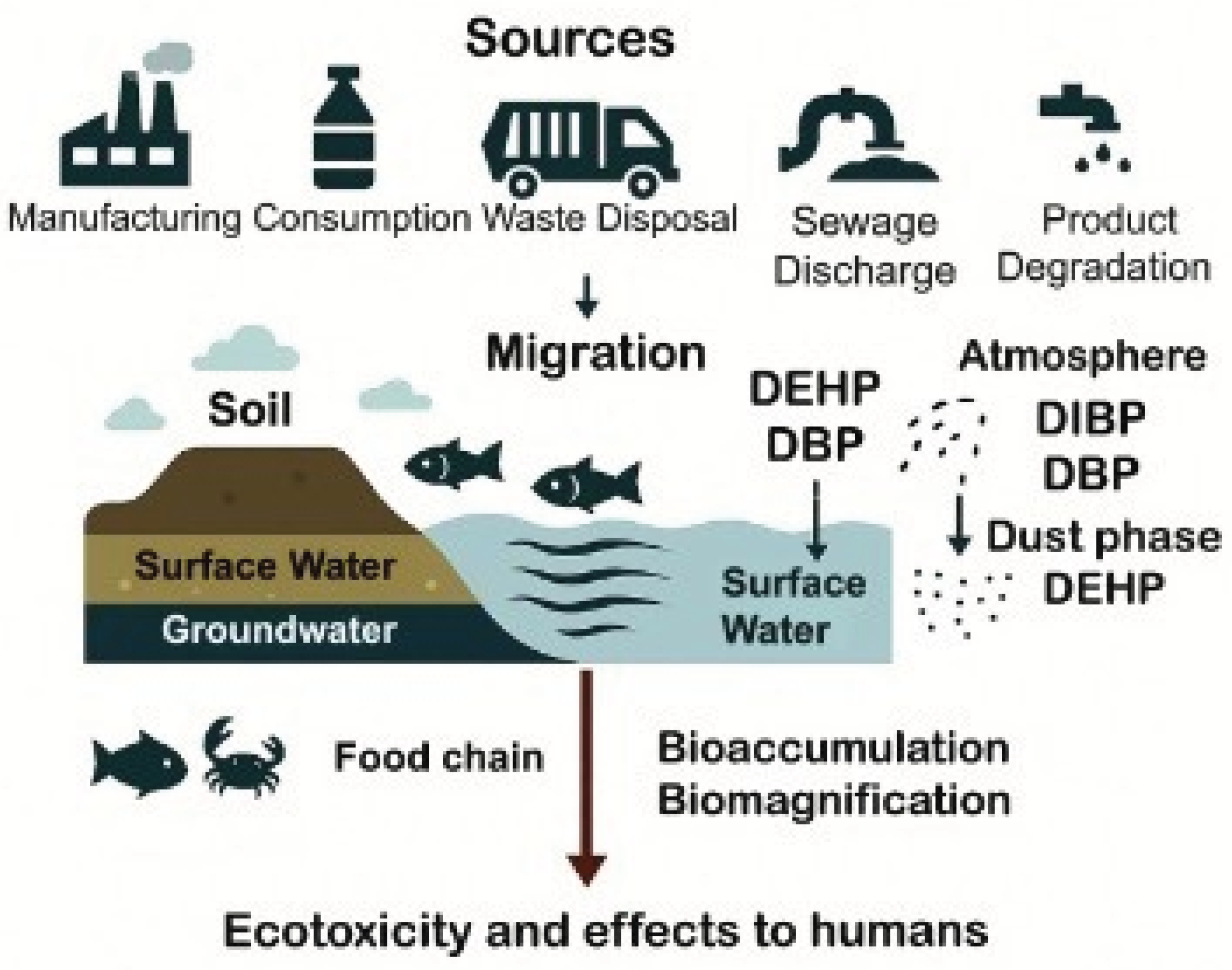
2.1. Phthalate Regulations
2.2. Health Effects and Toxicity of Phthalates
3. Phthalates Exposure and Ischemic Heart Disease: Epidemiological Evidence
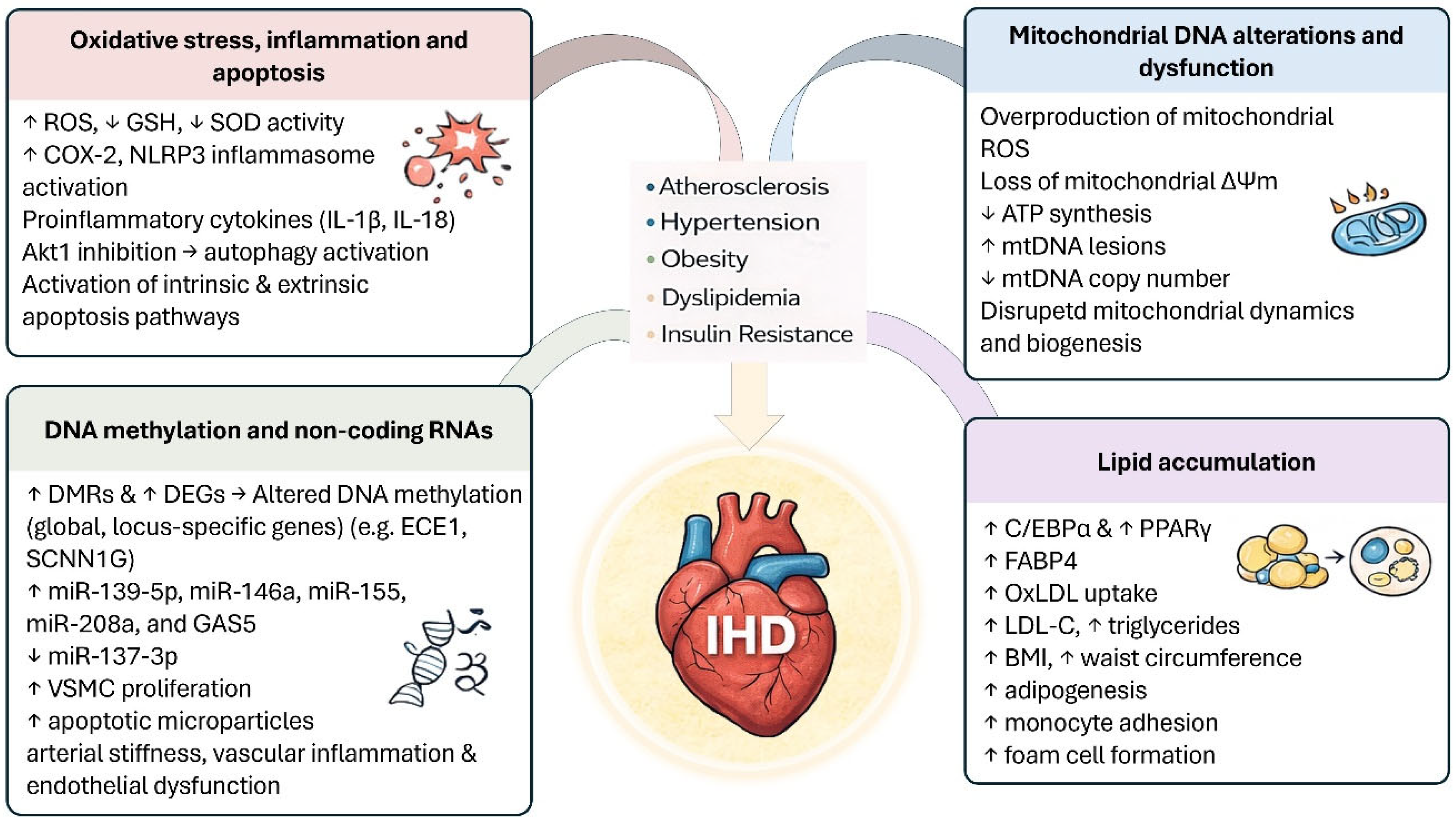
4. Cellular and Molecular Mechanisms Underlying Phthalates-Induced Ischemic Heart Disease
4.1. Oxidative Stress, Inflammation, and Apoptosis
4.2. Mitochondrial DNA Alterations and Dysfunction
4.3. DNA Methylation and Non-Coding RNAs
4.4. Lipid Accumulation
5. The Present and Future of Research: Intelligent Approaches in Studying Phthalate Exposure and Cardiovascular Risk
5.1. AI for Detecting and Modeling Phthalate Exposure and Health Outcomes
5.2. AI in Cardiovascular Risk Prediction and IHD
5.3. Phthalates and Cardiometabolic Pathways: AI Role in Their Integration
5.4. Tips for Future Development
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| 5mdC | 5-methyl-2-deoxycytidine |
| AI | Artificial Intelligence |
| AMI | Acute myocardial infarction |
| ASCVD | Atherosclerotic cardiovascular diseases |
| AUC | Area Under the Curve |
| AUROC | Area Under the Receiving Operating Characteristics curve |
| BBP | Butyl benzyl phthalate |
| BBzP | Butylbenzyl phthalate |
| BMI | Body mass index |
| bw | Body weight |
| CAC | Coronary Artery Calcium |
| CCTA | Coronary Computed Tomography Angiography |
| cGAN | Conventional Generative Adversarial Network |
| CHD | Coronary heart disease |
| CNN | Convolutional Neural Network |
| CT | Computed Tomography |
| CVD | Cardiovascular disease |
| dG | 2-deoxyguanine |
| DBP | Di-n-butyl phthalate |
| DEHP | 2-ethylhexyl phthalate |
| DEP | Diethyl phthalate |
| DIBP | Diisobutyl phthalate |
| DIDP | Di-iso-decyl phthalate |
| DINP | Diisononyl phthalate |
| DIPP | Diisobutyl phthalate |
| DL | Deep Learning |
| DMP | Dimethyl phthalate |
| DnBP | Di-n-butyl phthalate |
| DnOP | di-n-octyl phthalate |
| DOP | Dinoctyl phthalate |
| eGFR | Estimated glomerular filtration rate |
| EBP | Elevated blood pressure |
| EHR | Electronic Health Record |
| EMP | Endothelial microparticle |
| FPG | Fasting plasma glucose |
| GBM | Gradient Boosting Machine |
| GSH | Glutathione |
| GSM | Gray-scale median |
| HMW | High-molecular weight |
| hs-CRP | High-sensitivity C-reactive protein |
| hscTn | High-sensitivity cardiac troponin I |
| IHD | Ischemic heart disease |
| IMT | Intima-media thickness |
| IL | Interleukin |
| lncRNA | Long non-coding RNA |
| LLE-GC-MS | Liquid–liquid extraction/gas chromatography/mass spectrometry |
| LMW | Low-molecular weight |
| MBP | Mono-butyl phthalate |
| MBzP | Monobenzyl phthalate |
| MCPP | Mono-3-carboxy propyl phthalate |
| MDA | Malondialdehyde |
| MECPP | mono-2-ethyl-5-carboxypentyl phthalate |
| MEHHP | Mono(ethyl-5-hydroxyhexyl) phthalate |
| MEHOP | Mono(2-ethly-5-oxoheyl) phthalate |
| MEHP | Mono (2 ethylhexyl) phthalate |
| MEP | Mono-ethyl phthalate |
| MiBP | Mono-isobutyl phthalate |
| miRNA | Micro RNA |
| ML | Machine Learning |
| MMP | Mono-methyl phthalate |
| MnBP | Mono-n-butyl phthalate |
| mtDNA | Mitochondrial DNA |
| PLBoost | Phthalate Boosting |
| PMP | Platelet microparticle |
| ROS | Reactive oxygen species |
| SDI | Sociodemographic index |
| SMC | Smooth muscle cell |
| SOD | Superoxide dismutase |
| T2D | Type 2 diabetes |
| TDI | Tolerable daily intake |
| VSMC | Vascular smooth muscle cell |
References
- Hahad, O.; A Gilan, D.; Chalabi, J.; Al-Kindi, S.; Schuster, A.K.; Wicke, F.; Büttner, M.; Tüscher, O.; Lackner, K.J.; Galle, P.R.; et al. Cumulative social disadvantage and cardiovascular disease burden and mortality. Eur. J. Prev. Cardiol. 2023, 31, 40–48. [CrossRef]
- Nedkoff, L.; Briffa, T.; Zemedikun, D.; Herrington, S.; Wright, F.L. Global Trends in Atherosclerotic Cardiovascular Disease. Clin. Ther. 2023, 45, 1087–1091. [CrossRef]
- WHO, World Health Organization, 2025. Cardiovascular Diseases (CVDs). Available online: https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds) (accessed on 27 November 2023).
- GBD 2021 Diseases and Injuries Collaborators. Global incidence, prevalence, years lived with disability (YLDs), disability-adjusted life-years (DALYs), and healthy life expectancy (HALE) for 371 diseases and injuries in 204 countries and territories and 811 subnational locations, 1990-2021: a systematic analysis for the Global Burden of Disease Study 2021. Lancet. 2024, 403, 2133-2161.
- Li, Y.; Cao, G.-Y.; Jing, W.-Z.; Liu, J.; Liu, M. Global trends and regional differences in incidence and mortality of cardiovascular disease, 1990−2019: findings from 2019 global burden of disease study. Eur. J. Prev. Cardiol. 2022, 30, 276–286. [CrossRef]
- GBD 2021 Causes of Death Collaborators. Global burden of 288 causes of death and life expectancy decomposition in 204 countries and territories and 811 subnational locations, 1990-2021: a systematic analysis for the Global Burden of Disease Study 2021. Lancet. 2024, 403, 1988.
- Tang, J.; Hu, S.; Liu, X.; Li, H.; Kuang, L.; Zhang, L.; Cao, W.; Zhang, T.; Guan, X.; Li, L.; et al. Global, regional, and national time trends in ischaemic heart disease incidence over three decades (1990–2019): an age-period-cohort analysis of the global burden of disease study 2019. Front. Cardiovasc. Med. 2024, 11, 1396380. [CrossRef]
- Buja, L.M.; Heide, R.S.V. Pathobiology of Ischemic Heart Disease: Past, Present and Future. Cardiovasc. Pathol. 2016, 25, 214–220. [CrossRef]
- Jovin, D.G.; Sumpio, B.E.; Greif, D.M. Manifestations of human atherosclerosis across vascular beds. Jvs-Vascular Insights 2024, 2. [CrossRef]
- Russo, M.; Gurgoglione, F.L.; Russo, A.; Rinaldi, R.; Triglia, L.T.; Foschi, M.; Vigna, C.; Vergallo, R.; Montone, R.A.; Benedetto, U.; et al. Coronary Artery Disease and Atherosclerosis in Other Vascular Districts: Epidemiology, Risk Factors and Atherosclerotic Plaque Features. Life 2025, 15, 1226. [CrossRef]
- Khera, A.V.; Emdin, C.A.; Drake, I.; Natarajan, P.; Bick, A.G.; Cook, N.R.; Chasman, D.I.; Baber, U.; Mehran, R.; Rader, D.J.; et al. Genetic Risk, Adherence to a Healthy Lifestyle, and Coronary Disease. N. Engl. J. Med. 2016, 375, 2349–2358. [CrossRef]
- pinar, J. Hypertension and ischemic heart disease. Cor et vasa. 2012, 54, e433-e438.
- Pirillo, A.; Norata, G.D. The burden of hypercholesterolemia and ischemic heart disease in an ageing world. Pharmacol. Res. 2023, 193, 106814. [CrossRef]
- Shen, N.; Liu, J.; Wang, Y.; Qiu, Y.; Li, D.; Wang, Q.; Chai, L.; Chen, Y.; Hu, H.; Li, M. The global burden of ischemic heart disease attributed to high fasting plasma glucose: Data from 1990 to 2019. Heliyon 2024, 10, e27065. [CrossRef]
- Sohail, M.U.; Aisha, E.; Waqas, S.A.; Saad, M.; Arshad, M.S.; Ahmed, A.; Sohail, M.O.; Naveed, Z.; Amin, E.; Arora, S.; et al. Trends in obesity-related ischemic heart disease mortality among adults in the United States from 1999 to 2020. Futur. Cardiol. 2025, 21, 479–487. [CrossRef]
- Tang, S.; Meng, J.; Zhao, X.; Sun, W. Trends of ischemic heart disease mortality attributable to smoking in the five countries with the highest number of smokers during 1990–2019: an age-period-cohort analysis. Arch. Med Sci. 2024, 20, 43–53. [CrossRef]
- Wang, Y.; Yan, D.; Xu, W.; Min, B.; Fan, Z.; Su, H.; Zhao, X.; Wang, D.; Zhu, Y. Global burden of ischemic heart disease attributable to dietary factors: insights from the global burden of disease study 2021. Front. Nutr. 2025, 12, 1634566. [CrossRef]
- Scimeca, M.; Palumbo, V.; Giacobbi, E.; Servadei, F.; Casciardi, S.; Cornella, E.; Cerbara, F.; Rotondaro, G.; Seghetti, C.; Scioli, M.P.; et al. Impact of the environmental pollution on cardiovascular diseases: From epidemiological to molecular evidence. Heliyon 2024, 10, e38047. [CrossRef]
- Lamas, G.A.; Bhatnagar, A.; Jones, M.R.; Mann, K.K.; Nasir, K.; Tellez-Plaza, M.; Ujueta, F.; Navas-Acien, A.; the American Heart Association Council on Epidemiology and Prevention; Council on Cardiovascular and Stroke Nursing; Council on Lifestyle and Cardiometabolic Health; Council on Peripheral Vascular Disease; and Council on the Kidney in Cardiovascular Disea Contaminant Metals as Cardiovascular Risk Factors: A Scientific Statement From the American Heart Association. J. Am. Hear. Assoc. 2023, 12, e029852. [CrossRef]
- Yang, L.; Zheng, B.; Gong, Y. Global, regional and national burden of ischemic heart disease and its attributable risk factors from 1990 to 2021: a systematic analysis of the Global Burden of Disease study 2021. BMC Cardiovasc. Disord. 2025, 25, 1–15. [CrossRef]
- WHO, World Health Organization. The public health impact of chemicals: knowns and unknowns. Available online: https://www.who.int/publications/i/item/WHO-FWC-PHE-EPE-16.01-eng (accessed 17 November 2025).
- Bu, S.; Wang, Y.; Wang, H.; Wang, F.; Tan, Y. Analysis of global commonly-used phthalates and non-dietary exposure assessment in indoor environment. Build. Environ. 2020, 177. [CrossRef]
- Gorini, F.; Tonacci, A.; Sanmartin, C.; Venturi, F. Phthalates and Non-Phthalate Plasticizers and Thyroid Dysfunction: Current Evidence and Novel Strategies to Reduce Their Spread in Food Industry and Environment. Toxics 2025, 13, 222. [CrossRef]
- Gao, D.; Li, Z.; Wang, H.; Liang, H. An overview of phthalate acid ester pollution in China over the last decade: Environmental occurrence and human exposure. Sci. Total. Environ. 2018, 645, 1400–1409. [CrossRef]
- Statista. Global Plasticizers Market Volume 2015-2030. Available online: https://www.statista.com/statistics/1245193/plasticizer-market-volume-worldwide/#:~:text=In%202022,%20market%20volume%20of,it%20softer%20and%20more%20flexible (accessed on 20 November 2025).
- Naveen, K.V.; Saravanakumar, K.; Zhang, X.; Sathiyaseelan, A.; Wang, M.-H. Impact of environmental phthalate on human health and their bioremediation strategies using fungal cell factory- A review. Environ. Res. 2022, 214, 113781. [CrossRef]
- TEDX, The Endocrine Disruption Exchange. List of Potential Endocrine Disruptors. Available online: https://endocrinedisruption.org/interactive-tools/tedx-list-of-potential-endocrine-disruptors/search-the-tedx-list (accessed on 20 November 2025).
- Kelly, M.; Connolly, L.; Dean, M. Public Awareness and Risk Perceptions of Endocrine Disrupting Chemicals: A Qualitative Study. Int. J. Environ. Res. Public Heal. 2020, 17, 7778. [CrossRef]
- Eales, J.; Bethel, A.; Galloway, T.; Hopkinson, P.; Morrissey, K.; Short, R.; Garside, R. Human health impacts of exposure to phthalate plasticizers: An overview of reviews. Environ. Int. 2022, 158, 106903. [CrossRef]
- Mariana, M.; Castelo-Branco, M.; Soares, A.M.; Cairrao, E. Phthalates’ exposure leads to an increasing concern on cardiovascular health. J. Hazard. Mater. 2023, 457, 131680. [CrossRef]
- Hyman, S.; Acevedo, J.; Giannarelli, C.; Trasande, L. Phthalate exposure from plastics and cardiovascular disease: global estimates of attributable mortality and years life lost. EBioMedicine 2025, 117, 105730. [CrossRef]
- Zhang, J.; Wang, Z.; Li, X.; Zhang, Y.; Yuan, J.; Wang, Z.; Xu, F.; Chen, Y.; Li, C. Association between phthalates exposure and myocardial damage in the general population: A cross-sectional study. Environ. Res. 2024, 261, 119632. [CrossRef]
- Gong, W.; Zhu, H.; Sun, X.; Zhang, J.; Lin, M.; Sun, P. The association between exposure to phthalates and cardiovascular disease: A comprehensive study utilizing NHANES data from 2005 to 2018 and network toxicology. Chem. Interactions 2025, 420, 111651. [CrossRef]
- Kabekkodu, S.P.; Gladwell, L.R.; Choudhury, M. The mitochondrial link: Phthalate exposure and cardiovascular disease. Biochim. et Biophys. Acta (BBA) - Mol. Cell Res. 2024, 1871, 119708. [CrossRef]
- Moawad, A.M.; Awady, S.; Ali, A.A.E.R.; Abdelgwad, M.; Belal, S.; Taha, S.H.N.; Mohamed, M.I.; Hassan, F.M. Phthalate Exposure and Coronary Heart Disease: Possible Implications of Oxidative Stress and Altered miRNA Expression. Chem. Res. Toxicol. 2024, 37, 723–730. [CrossRef]
- Hasan, M.; Tama, R.T.; Dona, H.A.; Hoque, N.S.; Rahaman, A.; Alam, A. Comprehensive review of phthalate exposure: Health implications, biomarker detection and regulatory standards. J. Steroid Biochem. Mol. Biol. 2024, 247, 106671. [CrossRef]
- Martínez-Ibarra, A.; Martínez-Razo, L.; MacDonald-Ramos, K.; Morales-Pacheco, M.; Vázquez-Martínez, E.; López-López, M.; Dorantes, M.R.; Cerbón, M. Multisystemic alterations in humans induced by bisphenol A and phthalates: Experimental, epidemiological and clinical studies reveal the need to change health policies. Environ. Pollut. 2021, 271, 116380. [CrossRef]
- Mansuri, A.; Trivedi, C.; Chokshi, S.; Jantrania, K.; Kumar, A. Phthalate Exposure: Prevalence, Health Effects, Regulatory Frameworks, and Remediation. Chem. Res. Toxicol. 2025, 38, 1291–1308. [CrossRef]
- Šimunović, A.; Tomić, S.; Kranjčec, K. Medical devices as a source of phthalate exposure: a review of current knowledge and alternative solutions. Arch. Ind. Hyg. Toxicol. 2022, 73, 179–190. [CrossRef]
- Giuliani, A.; Zuccarini, M.; Cichelli, A.; Khan, H.; Reale, M. Critical Review on the Presence of Phthalates in Food and Evidence of Their Biological Impact. Int. J. Environ. Res. Public Heal. 2020, 17, 5655. [CrossRef]
- Das, M.T.; Ghosh, P.; Thakur, I.S. Intake estimates of phthalate esters for South Delhi population based on exposure media assessment. Environ. Pollut. 2014, 189, 118–125. [CrossRef]
- Wang, Y.; Zhu, H.; Kannan, K. A Review of Biomonitoring of Phthalate Exposures. Toxics 2019, 7, 21. [CrossRef]
- Fromme, H.; Gruber, L.; Schlummer, M.; Wolz, G.; Böhmer, S.; Angerer, J.; Mayer, R.; Liebl, B.; Bolte, G. Intake of phthalates and di(2-ethylhexyl)adipate: Results of the Integrated Exposure Assessment Survey based on duplicate diet samples and biomonitoring data. Environ. Int. 2007, 33, 1012–1020. [CrossRef]
- Fromme, H.; Lahrz, T.; Piloty, M.; Gebhart, H.; Oddoy, A.; Ruden, H. Occurrence of phthalates and musk fragrances in indoor air and dust from apartments and kindergartens in Berlin (Germany). Indoor Air 2004, 14, 188–195. [CrossRef]
- Wormuth, M.; Scheringer, M.; Vollenweider, M.; Hungerbühler, K. What Are the Sources of Exposure to Eight Frequently Used Phthalic Acid Esters in Europeans? Risk Anal. 2006, 26, 803–824. [CrossRef]
- Stroustrup, A.; Bragg, J.B.; Busgang, S.A.; Andra, S.S.; Curtin, P.; Spear, E.A.; Just, A.C.; Arora, M.; Gennings, C. Sources of clinically significant neonatal intensive care unit phthalate exposure. J. Expo. Sci. Environ. Epidemiology 2018, 30, 137–148. [CrossRef]
- Tran, H.T.; Lin, C.; Bui, X.-T.; Nguyen, M.K.; Cao, N.D.T.; Mukhtar, H.; Hoang, H.G.; Varjani, S.; Ngo, H.H.; Nghiem, L.D. Phthalates in the environment: characteristics, fate and transport, and advanced wastewater treatment technologies. Bioresour. Technol. 2022, 344, 126249. [CrossRef]
- EC: Commission Decision of 7 December 1999 adopting measures prohibiting the placing on the market of toys and childcare articles intended to be placed in the mouth by children under three years of age made of soft PVC containing one or more of the substances di-iso-nonyl phthalate (DINP), di(2-ethylhexyl) phthalate (DEHP), dibutyl phthalate (DBP), di-iso-decyl phthalate (DIDP), di-n-octyl phthalate (DNOP), and butylbenzyl phthalate (BBP) (notified under document number C(1999) 4436). Available online: https://op.europa.eu/en/publication-detail/-/publication/da264be1-6ef2-4063-b725-f36cc1f1a20f (accessed on 13 December 2025).
- Palandri, L.; Monti, M.; Scasserra, M.R.; Lugli, C.; Fasano, M.; Lucaccioni, L.; Righi, E. Regulatory framework of phthalates and two common alternatives: A review of the European Union legislation. Int. J. Hyg. Environ. Heal. 2025, 271, 114704. [CrossRef]
- European Commission. REACH Regulation. Available online: https://environment.ec.europa.eu/topics/chemicals/reach-regulation_en (accessed on 13 December 2025).
- European Union. Directive 2005/84/EC of the European Parliament and of the Council of 14 December 2005 amending for the 22nd time Council Directive 76/769/EEC on the approximation of the laws, regulations and administrative provisions of the Member States relating to restrictions on the marketing and use of certain dangerous substances and preparations (phthalates in toys and childcare articles). Available online: https://eur-lex.europa.eu/eli/dir/2005/84/oj/eng (accessed on 13 December 2025).
- European Union. Commission Regulation (EU) 2018/2005 of 17 December 2018 Amending Annex XVII to Regulation (EC) No 1907/2006 of the European Parliament and of the Council Concerning the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) as Regards Bis(2-Ethylhexyl) Phthalate (DEHP), Dibutyl Phthalate (DBP), Benzyl Butyl Phthalate (BBP) and Diisobutyl Phthalate (DIBP). Available online: https://eur-lex.europa.eu/eli/reg/2018/2005/oj/eng (accessed on 13 December 2025).
- EFSA Panel on Food Contact Materials, Enzymes and Processing Aids (CEP); Silano, V.; Barat Baviera, J.M.; Bolognesi, C.; Chesson, A.; Cocconcelli, P.S.; Crebelli, R.; Gott, D.M.; Grob, K.; Lampi, E.; et al. Update of the risk assessment of di-butylphthalate (DBP), butyl-benzyl-phthalate (BBP), bis(2-ethylhexyl)phthalate (DEHP), di-isononylphthalate (DINP) and di-isodecylphthalate (DIDP) for use in food contact materials. EFSA J. 2019, 17, e05838.
- European Commission. Commission Regulation (EU) 2021/2045 of 23 November 2021 amending Annex XIV to Regulation (EC) No 1907/2006 of the European Parliament and of the Council concerning the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) (Text with EEA relevance). Available online: https://eur-lex.europa.eu/eli/reg/2021/2045/oj/eng (accessed on 14 December 2025).
- European Commission. Commission Regulation (EU) 2023/2482 of 13 November 2023 amending Regulation (EC) No 1907/2006 of the European Parliament and of the Council as regards the substance bis(2-ethylhexyl) phthalate (DEHP) in medical devices. Available online: https://eur-lex.europa.eu/eli/reg/2023/2482/oj/eng (accessed on 14 December 2025).
- United States Environmental Protection Agency: Assessing and Managing Chemicals under TSCA. URL: https://www.epa.gov/assessing-and-managing-chemicals-under-tsca/phthalates (accessed on).
- United States Environmental Protection Agency, 2025. Risk Evaluation for Di-isodecyl phthalate (1,2-Benzene- dicarboxylic acid, 1,2- diisodecyl ester) (DIDP). Available online: https://www.epa.gov/assessing-and-managing-chemicals-under-tsca/risk-evaluation-di-isodecyl-phthalate-12-benzene (accessed on 15 December 2025).
- United States Environmental Protection Agency, 2025. Risk Evaluation for Diisononyl phthalate (1,2-Benzene- dicarboxylic acid, 1,2- diisononyl ester) (DINP). Available online: https://www.epa.gov/assessing-and-managing-chemicals-under-tsca/risk-evaluation-diisononyl-phthalate-12-benzene (accessed on 15 December 2025).
- GreenSoft Technology, Inc., 2024. China RoHS Regulation Updated to Restrict Four Phthalates in Electronics. Available online: https://www.greensofttech.com/blog-2024-china-rohs-regulation-updated-to-restrict-four-phthalates-in-electronics/ (accessed on 15 December 2025).
- Hlisníková, H.; Petrovičová, I.; Kolena, B.; Šidlovská, M.; Sirotkin, A. Effects and Mechanisms of Phthalates’ Action on Reproductive Processes and Reproductive Health: A Literature Review. Int. J. Environ. Res. Public Heal. 2020, 17, 6811. [CrossRef]
- Caporossi, L.; Alteri, A.; Campo, G.; Paci, E.; Tranfo, G.; Capanna, S.; Papaleo, E.; Pigini, D.; Viganò, P.; Papaleo, B. Cross Sectional Study on Exposure to BPA and Phthalates and Semen Parameters in Men Attending a Fertility Center. Int. J. Environ. Res. Public Heal. 2020, 17, 489. [CrossRef]
- Hond, E.D.; Tournaye, H.; De Sutter, P.; Ombelet, W.; Baeyens, W.; Covaci, A.; Cox, B.; Nawrot, T.S.; Van Larebeke, N.; D'HOoghe, T. Human exposure to endocrine disrupting chemicals and fertility: A case–control study in male subfertility patients. Environ. Int. 2015, 84, 154–160. [CrossRef]
- Jensen, M.S.; Anand-Ivell, R.; Nørgaard-Pedersen, B.; Jönsson, B.A.G.; Bonde, J.P.; Hougaard, D.M.; Cohen, A.; Lindh, C.H.; Ivell, R.; Toft, G. Amniotic Fluid Phthalate Levels and Male Fetal Gonad Function. Epidemiology 2015, 26, 91–99. [CrossRef]
- Das, D.; Dutta, H.K.; Borbora, D.; Brahma, R.C.; Das, J.M. Assessing the relationship between hypospadias risk and parental occupational exposure to potential endocrine-disrupting chemicals. Occup. Environ. Med. 2022, 80, 93–96. [CrossRef]
- Dorman, D.C.; Chiu, W.; Hales, B.F.; Hauser, R.; Johnson, K.J.; Mantus, E.; Martel, S.; Robinson, K.A.; Rooney, A.A.; Rudel, R.; et al. Systematic reviews and meta-analyses of human and animal evidence of prenatal diethylhexyl phthalate exposure and changes in male anogenital distance. J. Toxicol. Environ. Heal. Part B 2018, 21, 207–226. [CrossRef]
- Ghazarian, A.A.; Trabert, B.; Robien, K.; Graubard, B.I.; McGlynn, K.A. Maternal use of personal care products during pregnancy and risk of testicular germ cell tumors in sons. Environ. Res. 2018, 164, 109–113. [CrossRef]
- Li, J.; Deng, T.; Rao, W.; Liao, H.; Wang, Y.; Guo, N.; Du, Y.; Guo, Q.; Zeng, Q.; Liu, C.; et al. Phthalate metabolites in urine and follicular fluid in relation to menstrual cycle characteristics in women seeking fertility assistance. Environ. Int. 2023, 183, 108362. [CrossRef]
- Golestanzadeh, M.; Riahi, R.; Kelishadi, R. Association of phthalate exposure with precocious and delayed pubertal timing in girls and boys: a systematic review and meta-analysis. Environ. Sci. Process. Impacts 2020, 22, 873–894. [CrossRef]
- Hu, W.; Jin, Z.; Wang, H.; Wang, F.; Qu, F. Relationship between phthalates exposure, risk of decreased ovarian reserve, and oxidative stress levels. Toxicol. Ind. Heal. 2024, 40, 156–166. [CrossRef]
- Al-Saleh, I. The relationship between urinary phthalate metabolites and polycystic ovary syndrome in women undergoing in vitro fertilization: Nested case-control study. Chemosphere 2022, 286, 131495. [CrossRef]
- Cai, W.; Yang, J.; Liu, Y.; Bi, Y.; Wang, H. Association between Phthalate Metabolites and Risk of Endometriosis: A Meta-Analysis. Int. J. Environ. Res. Public Health 2019, 16, 3678. [CrossRef]
- Tiburcio, D.; Parsell, M.; Shapiro, H.; Adolphe, S.; Naranjo, O.; George, S.; Toborek, M. Endocrine disruption to metastasis: How phthalates promote breast carcinogenesis. Ecotoxicol. Environ. Saf. 2025, 303, 118874–118874. [CrossRef]
- Derakhshan, A.; Shu, H.; Broeren, M.A.; Lindh, C.H.; Peeters, R.P.; Kortenkamp, A.; Demeneix, B.; Bornehag, C.-G.; Korevaar, T.I. Association of phthalate exposure with thyroid function during pregnancy. Environ. Int. 2021, 157, 106795. [CrossRef]
- Al-Saleh, I.; Elkhatib, R.; Alghamdi, R.; Alrushud, N.; Alnuwaysir, H.; Alnemer, M.; Aldhalaan, H.; Shoukri, M. Phthalate exposure during pregnancy and its association with thyroid hormones: A prospective cohort study. Int. J. Hyg. Environ. Heal. 2024, 261, 114421. [CrossRef]
- Li, N.; Papandonatos, G.D.; Calafat, A.M.; Yolton, K.; Lanphear, B.P.; Chen, A.; Braun, J.M. Identifying periods of susceptibility to the impact of phthalates on children's cognitive abilities. Environ. Res. 2019, 172, 604–614. [CrossRef]
- Polanska, K.; Ligocka, D.; Sobala, W.; Hanke, W. Phthalate exposure and child development: The Polish Mother and Child Cohort Study. Early Hum. Dev. 2014, 90, 477–485. [CrossRef]
- Praveena, S.M.; Munisvaradass, R.; Masiran, R.; Rajendran, R.K.; Lin, C.-C.; Kumar, S. Phthalates exposure and attention-deficit/hyperactivity disorder in children: a systematic review of epidemiological literature. Environ. Sci. Pollut. Res. 2020, 27, 44757–44770. [CrossRef]
- Linghu, D.; Zhu, Z.; Zhang, D.; Luo, Y.; Ma, J.; Li, T.; Sun, Z.; Xie, Z.; Sun, J.; Cao, C. Diethylhexyl phthalate induces immune dysregulation and is an environmental immune disruptor. J. Hazard. Mater. 2024, 480, 136244. [CrossRef]
- Boissiere-O’nEill, T.; Lazarevic, N.; Sly, P.D.; Ponsonby, A.-L.; Chen, A.; Azad, M.B.; Braun, J.M.; Brook, J.R.; Burgner, D.; Lanphear, B.P.; et al. Phthalates and bisphenols early-life exposure, and childhood allergic conditions: a pooled analysis of cohort studies. J. Expo. Sci. Environ. Epidemiology 2025, 35, 965–980. [CrossRef]
- Li, R.; Zheng, B.; Zhang, Y.; He, L.; Ren, C.; Guan, L.; Yang, H.; Tian, J.; Chen, X.; Shi, D.; et al. The impact of phthalates on asthma and chronic obstructive pulmonary disease: a comprehensive analysis based on network toxicology and molecular docking. Front. Pharmacol. 2025, 16, 1566965. [CrossRef]
- Ahern, T.P.; Broe, A.; Lash, T.L.; Cronin-Fenton, D.P.; Ulrichsen, S.P.; Christiansen, P.M.; Cole, B.F.; Tamimi, R.M.; Sørensen, H.T.; Damkier, P. Phthalate Exposure and Breast Cancer Incidence: A Danish Nationwide Cohort Study. J. Clin. Oncol. 2019, 37, 1800–1809. [CrossRef]
- Attina, T.M.; Trasande, L. Association of Exposure to Di-2-Ethylhexylphthalate Replacements With Increased Insulin Resistance in Adolescents From NHANES 2009–2012. J. Clin. Endocrinol. Metab. 2015, 100, 2640–2650. [CrossRef]
- Dales, R.E.; Kauri, L.M.; Cakmak, S. The associations between phthalate exposure and insulin resistance, β-cell function and blood glucose control in a population-based sample. Sci. Total. Environ. 2018, 612, 1287–1292. [CrossRef]
- Tan, Y.; Guo, Z.; Yao, H.; Liu, H.; Fu, Y.; Luo, Y.; He, R.; Liu, Y.; Li, P.; Nie, L.; et al. Association of phthalate exposure with type 2 diabetes and the mediating effect of oxidative stress: A case-control and computational toxicology study. Ecotoxicol. Environ. Saf. 2024, 274, 116216. [CrossRef]
- Wu, Q.; Li, G.; Zhao, C.-Y.; Na, X.-L.; Zhang, Y.-B. Association between phthalate exposure and obesity risk: A meta-analysis of observational studies. Environ. Toxicol. Pharmacol. 2023, 102, 104240. [CrossRef]
- Mérida, D.M.; Moreno-Franco, B.; Marquès, M.; León-Latre, M.; Laclaustra, M.; Guallar-Castillón, P. Phthalate exposure and the metabolic syndrome: A systematic review and meta-analysis. Environ. Pollut. 2023, 333, 121957. [CrossRef]
- Kosmas, C.E.; Bousvarou, M.D.; Kostara, C.E.; Papakonstantinou, E.J.; Salamou, E.; Guzman, E. Insulin resistance and cardiovascular disease. J Int Med Res. 2023, 51, 3000605231164548.
- Kobiyama, K.; Ley, K. Atherosclerosis. Circ Res. 2018, 123, 1118-1120.
- Lind, P.M.; Lind, L. Circulating levels of bisphenol A and phthalates are related to carotid atherosclerosis in the elderly. Atherosclerosis 2011, 218, 207–213. [CrossRef]
- Olkowska, E. Permeability of Dimethyl Phthalate Through Human Skin Models – Health Risk Assessment. Expo. Heal. 2023, 16, 369–376. [CrossRef]
- Lagarde, F.; Beausoleil, C.; Belcher, S.M.; Belzunces, L.P.; Emond, C.; Guerbet, M.; Rousselle, C. Non-monotonic dose-response relationships and endocrine disruptors: a qualitative method of assessment. Environ. Heal. 2015, 14, 13–13. [CrossRef]
- EFSA Scientific Committee; More, S.; Benford, D.; Bennekou, S.H.; Bampidis, V.; Bragard, C.; Halldorsson, T.; Hernandez-Jerez, A.; Koutsoumanis, K.; Lambré, C.; et al. Opinion on the impact of non-monotonic dose responses on EFSA′s human health risk assessments. EFSA J. 2021, 19, e06877. [CrossRef]
- Lind, L.; Andersson, J.; Rönn, M.; Gustavsson, T. The echogenecity of the intima–media complex in the common carotid artery is closely related to the echogenecity in plaques. Atherosclerosis 2007, 195, 411–414. [CrossRef]
- Andersson, J.; Sundström, J.; Gustavsson, T.; Hulthe, J.; Elmgren, A.; Zilmer, K.; Zilmer, M.; Lind, L. Echogenecity of the carotid intima-media complex is related to cardiovascular risk factors, dyslipidemia, oxidative stress and inflammation: the Prospective Investigation of the Vasculature in Uppsala Seniors (PIVUS) study. Atherosclerosis. 2009, 204, 612-618.
- Olsén, L.; Lind, L.; Lind, P.M. Associations between circulating levels of bisphenol A and phthalate metabolites and coronary risk in the elderly. Ecotoxicol. Environ. Saf. 2012, 80, 179–183. [CrossRef]
- Al-Shamsi, S. Performance of the Framingham coronary heart disease risk score for predicting 10-year cardiac risk in adult United Arab Emirates nationals without diabetes: a retrospective cohort study. BMC Fam. Pr. 2020, 21, 1–7. [CrossRef]
- Wiberg, B.; Lind, P.M.; Lind, L. Serum levels of monobenzylphthalate (MBzP) is related to carotid atherosclerosis in the elderly. Environ. Res. 2014, 133, 348–352. [CrossRef]
- Herrero, Ó.; Planelló, R.; Morcillo, G. The plasticizer benzyl butyl phthalate (BBP) alters the ecdysone hormone pathway, the cellular response to stress, the energy metabolism, and several detoxication mechanisms in Chironomus riparius larvae. Chemosphere 2015, 128, 266–277. [CrossRef]
- Lin, C.-Y.; Hsieh, C.-J.; Lo, S.-C.; Chen, P.-C.; Torng, P.-L.; Hu, A.; Sung, F.-C.; Su, T.-C. Positive association between concentration of phthalate metabolites in urine and microparticles in adolescents and young adults. Environ. Int. 2016, 92-93, 157–164. [CrossRef]
- Feng, S.; Chen, J.W.; Shu, X.Y.; Aihemaiti, M.; Quan, J.W.; Lu, L.; Zhang, R.Y.; Yang, C.D.; Wang, X.Q. Endothelial microparticles: A mechanosensitive regulator of vascular homeostasis and injury under shear stress. Front. Cell Dev. Biol. 2022, 10, 980112. [CrossRef]
- Strukel, S.; Teshome, B.; Rai, V. The Multifaceted Role of Platelets in Atherosclerosis and Ischemic Disease: Pathogenesis, Inflammation, and Therapeutic Opportunities. Life 2025, 15, 1656. [CrossRef]
- Olson, N.C.; Koh, I.; Reiner, A.P.; Judd, S.E.; Irvin, M.R.; Howard, G.; Zakai, N.A.; Cushman, M. Soluble CD14, Ischemic Stroke, and Coronary Heart Disease Risk in a Prospective Study: The REGARDS Cohort. J. Am. Hear. Assoc. 2020, 9, e014241. [CrossRef]
- Ban, J.-B.; Fan, X.-W.; Huang, Q.; Li, B.-F.; Chen, C.; Zhang, H.-C.; Xu, S.-Q. Mono-(2-Ethylhexyl) Phthalate Induces Injury in Human Umbilical Vein Endothelial Cells. PLOS ONE 2014, 9, e97607. [CrossRef]
- Choy, J.C.; Granville, D.J.; Hunt, D.W.; McManus, B.M. Endothelial Cell Apoptosis: Biochemical Characteristics and Potential Implications for Atherosclerosis. J. Mol. Cell. Cardiol. 2001, 33, 1673–1690. [CrossRef]
- Su, T.-C.; Hwang, J.-S.; Torng, P.-L.; Wu, C.; Lin, C.-Y.; Sung, F.-C. Phthalate exposure increases subclinical atherosclerosis in young population. Environ. Pollut. 2019, 250, 586–593. [CrossRef]
- Lin, C.-Y.; Lee, H.-L.; Hwang, Y.-T.; Wang, C.; Hsieh, C.-J.; Wu, C.; Sung, F.-C.; Su, T.-C. The association between urine di-(2-ethylhexyl) phthalate metabolites, global DNA methylation, and subclinical atherosclerosis in a young Taiwanese population. Environ. Pollut. 2020, 265, 114912. [CrossRef]
- Hsueh, Y.-M.; Chen, W.-J.; Lee, H.-L.; Huang, Y.-L.; Shiue, H.-S.; Hsu, S.-L.; Chen, H.-H.; Lin, Y.-C. Global DNA methylation and the association between metal exposure and chronic kidney disease. Front. Public Heal. 2023, 11, 1104692. [CrossRef]
- Willmer, T.; Mabasa, L.; Sharma, J.; Muller, C.J.F.; Johnson, R. Blood-Based DNA Methylation Biomarkers to Identify Risk and Progression of Cardiovascular Disease. Int. J. Mol. Sci. 2025, 26, 2355. [CrossRef]
- Agha, G.; Mendelson, M.M.; Ward-Caviness, C.K.; Joehanes, R.; Huan, T.; Gondalia, R.; Salfati, E.; Brody, J.A.; Fiorito, G.; Bressler, J.; et al. Blood Leukocyte DNA Methylation Predicts Risk of Future Myocardial Infarction and Coronary Heart Disease. Circulation 2019, 140, 645–657. [CrossRef]
- Farmakis, D.; Richter, D.; Chronopoulou, G.; Goumas, G.; Kountouras, D.; Mastorakou, A.; Papingiotis, G.; Hahalis, G.; Tsioufis, K. High-sensitivity cardiac troponin I for cardiovascular risk stratification in apparently healthy individuals. Hell. J. Cardiol. 2023, 75, 74–81. [CrossRef]
- Taggart, C.; Wereski, R.; Mills, N.L.; Chapman, A.R. Diagnosis, Investigation and Management of Patients with Acute and Chronic Myocardial Injury. J. Clin. Med. 2021, 10, 2331. [CrossRef]
- Su, T.-C.; Hwang, J.-J.; Sun, C.-W.; Wang, S.-L. Urinary phthalate metabolites, coronary heart disease, and atherothrombotic markers. Ecotoxicol. Environ. Saf. 2019, 173, 37–44. [CrossRef]
- Hernández-Díaz, S.; Su, Y.-C.; Mitchell, A.A.; Kelley, K.E.; Calafat, A.M.; Hauser, R. Medications as a potential source of exposure to phthalates among women of childbearing age. Reprod. Toxicol. 2013, 37, 1–5. [CrossRef]
- Swastini, D.A.; Wiryanthini, I.A.D.; Ariastuti, N.L.P.; Muliantara, A. Atherosclerosis Prediction with High Sensitivity C-Reactive Protein (hs-CRP) and Related Risk Factor in Patient with Dyslipidemia. Open Access Maced. J. Med Sci. 2019, 7, 3887–3890. [CrossRef]
- Surma, S.; Banach, M. Fibrinogen and Atherosclerotic Cardiovascular Diseases—Review of the Literature and Clinical Studies. Int. J. Mol. Sci. 2021, 23, 193. [CrossRef]
- Gong, P.; Yang, S.-H.; Li, S.; Luo, S.-H.; Zeng, R.-X.; Zhang, Y.; Guo, Y.-L.; Zhu, C.-G.; Xu, R.-X.; Li, J.-J. Plasma d-Dimer as a Useful Marker Predicts Severity of Atherosclerotic Lesion and Short-Term Outcome in Patients With Coronary Artery Disease. Clin. Appl. Thromb. Hemost. 2016, 22, 633–640. [CrossRef]
- Nosalski, R.; Siedlinski, M.; Neves, K.B.; Monaco, C. Editorial: The interplay between oxidative stress, immune cells and inflammation in cardiovascular diseases. Front. Cardiovasc. Med. 2024, 11, 1385809. [CrossRef]
- Wu, X.; Jiang, L.; Sun, X.; Yao, X.; Bai, Y.; Liu, X.; Liu, N.; Zhai, X.; Wang, S.; Yang, G. Mono(2-ethylhexyl) phthalate induces autophagy-dependent apoptosis through lysosomal-mitochondrial axis in human endothelial cells. Food Chem. Toxicol. 2017, 106, 273–282. [CrossRef]
- Liu, N.; Jiang, L.; Sun, X.; Yao, X.; Zhai, X.; Liu, X.; Wu, X.; Bai, Y.; Wang, S.; Yang, G. Mono-(2-ethylhexyl) phthalate induced ROS-dependent autophagic cell death in human vascular endothelial cells. Toxicol. Vitr. 2017, 44, 49–56. [CrossRef]
- Wen, W.; Liu, Z.; Li, L.; Qiu, F.; Zhang, H.; Cao, Y.; Li, J.; Huang, X.; Huang, Y. Multimodal mechanisms of di-(2-ethylhexyl) phthalate-induced cardiovascular–kidney–metabolic syndrome. Ecotoxicol Environ Saf. 2025, 309, 119515.
- Fan, X.; Gu, C.; Shen, L.; Gao, Z.; Yang, X.; Bian, Y.; Wang, F.; Jiang, X. Theoretical insights into the binding of mono/di-ethyl phthalates to superoxide dismutase and associated structural changes impairing antioxidant activity: A coupled molecular docking and dynamics simulation approach. Sci. Total. Environ. 2025, 983, 179667. [CrossRef]
- Mariana, M.; Cairrao, E. Phthalates Implications in the Cardiovascular System. J. Cardiovasc. Dev. Dis. 2020, 7, 26. [CrossRef]
- Amara, I.; Timoumi, R.; Annabi, E.; Neffati, F.; Najjar, M.F.; Bouaziz, C.; Abid-Essefi, S. Di (2-ethylhexyl) phthalate induces cardiac disorders in BALB/c mice. Environ. Sci. Pollut. Res. 2019, 26, 7540–7549. [CrossRef]
- Zhao, J.-F.; Hsiao, S.-H.; Hsu, M.-H.; Pao, K.-C.; Kou, Y.R.; Shyue, S.-K.; Lee, T.-S. Di-(2-ethylhexyl) phthalate accelerates atherosclerosis in apolipoprotein E-deficient mice. Arch. Toxicol. 2014, 90, 181–190. [CrossRef]
- Cui, J.G.; Zhang, H.; Zhang, Y.Q.; Sun, B.; Li, X.N.; Zhao, Y.; Li, J.L. New insights into DEHP-induced cardiotoxicity: Pyroptosis via sphingolipid metabolism disruption. J Hazard Mater. 2025, 499, 140148.
- Chi, Z.; Lin, H.; Wang, X.; Meng, X.; Zhou, J.; Xiang, L.; Cao, G.; Wu, P.; Cai, Z.; Zhao, X. Dimethyl phthalate induces blood immunotoxicity through oxidative damage and caspase-dependent apoptosis. Sci. Total. Environ. 2022, 838, 156047. [CrossRef]
- Schwendt, A.; Chammas, J.B.; Chalifour, L.E. Acute phthalate exposure during post-myocardial infarction recovery enhances inflammasome activation. Toxicol Appl Pharmacol. 2022, 440, 115954.
- Wang, J.-X.; Zhao, Y.; Chen, M.-S.; Zhang, H.; Cui, J.-G.; Li, J.-L. Heme-oxygenase-1 as a target for phthalate-induced cardiomyocytes ferroptosis. Environ. Pollut. 2022, 317, 120717. [CrossRef]
- Liang, X.; Wu, Y.; Feng, Q.; Zhu, D.; Huang, Q.; Wei, Z.; Ma, P.; Yang, X.; Bao, C.; Bao, X. Dibutyl phthalate synergizes with high-fat diet to aggravate cardiac fibrosis. Ecotoxicol Environ Saf. 2025, 302, 118708.
- Yamaguchi, R.; Sakamoto, A.; Yamaguchi, R.; Haraguchi, M.; Narahara, S.; Sugiuchi, H.; Katoh, T.; Yamaguchi, Y. Di-(2-ethylhexyl) phthalate promotes tissue factor–bearing microparticle release from macrophages. Am J Med Sci. 2019, 357, 492–506.
- Shi, X.; Zhang, Y.; Li, Y.; Yu, F.; Li, Z.; Wang, L.; Tu, X.; Zhou, S.; Lu, Q. Phthalate exposure, oxidative stress and hypertension risk: Exploring associations and mediation effects in a Chinese general population. Ecotoxicol. Environ. Saf. 2025, 303, 118832. [CrossRef]
- Su, T.-C.; Hwang, J.-J.; Sun, C.-W.; Wang, S.-L. Urinary phthalate metabolites, coronary heart disease, and atherothrombotic markers. Ecotoxicol. Environ. Saf. 2019, 173, 37–44. [CrossRef]
- Wallace, D.C. Mitochondrial DNA Variation in Human Radiation and Disease. Cell 2015, 163, 33–38. [CrossRef]
- Ashar, F.N.; Zhang, Y.; Longchamps, R.J.; Lane, J.; Moes, A.; Grove, M.L.; Mychaleckyj, J.C.; Taylor, K.D.; Coresh, J.; Rotter, J.I.; et al. Association of Mitochondrial DNA Copy Number With Cardiovascular Disease. JAMA Cardiol. 2017, 2, 1247–1255. [CrossRef]
- Cheng, Q.; Liu, Q.Q.; Lu, C.(. A state-of-the-science review of using mitochondrial DNA copy number as a biomarker for environmental exposure. Environ. Pollut. 2024, 346, 123642. [CrossRef]
- Dorn, G.W.; Vega, R.B.; Kelly, D.P. Mitochondrial biogenesis and dynamics in the heart. Genes Dev. 2015, 29, 1981–1991.
- Chen, X.J.; Butow, R.A. The organization and inheritance of the mitochondrial genome. Nat. Rev. Genet. 2005, 6, 815–825. [CrossRef]
- Wallace, D.C. A Mitochondrial Paradigm of Metabolic and Degenerative Diseases, Aging, and Cancer: A Dawn for Evolutionary Medicine. Annu. Rev. Genet. 2005, 39, 359–407. [CrossRef]
- Shokolenko, I.N.; Alexeyev, M.F. Mitochondrial DNA: A disposable genome? Biochim Biophys Acta. 2015, 1852, 1805–1809.
- Yang, H.M. Mitochondrial dysfunction in cardiovascular diseases. Int J Mol Sci. 2025, 26, 1917.
- Wang, L.; Zhang, Q.; Yuan, K.; Yuan, J. mtDNA in cardiovascular disease pathogenesis. Dis Markers. 2021, 2021, 7157109.
- Hu, H.; Lin, Y.; Xu, X.; Lin, S.; Chen, X.; Wang, S. Mitochondrial DNA alterations in coronary heart disease. Exp Mol Pathol. 2020, 114, 104412.
- Fan, X.; Zhang, D.; Hou, T.; Zhang, Q.; Wang, Z. Mitochondrial toxicity of dibutyl phthalate in zebrafish cells. Chemosphere. 2023, 326, 138510.
- Fan, X.; Zhang, D.; Hou, T.; Zhang, Q.; Tao, L.; Bian, C.; Wang, Z. mtDNA stress–mediated toxicity of dibutyl phthalate in zebrafish. Environ Sci Technol. 2024, 58, 7731–7742.
- Fu, X.; Zhao, Y.; Ke, Y.; Gao, Y.; Wang, M.; Chen, Y.; Huo, W.; Wang, L.; Zhang, W.; Wu, Y.; et al. Mitochondrial DNA copy number and risk of cardiovascular disease and all-cause mortality: a systematic review and meta-analysis of observational studies. Qjm: Int. J. Med. 2024, 118, 5–15. [CrossRef]
- Dor, Y.; Cedar, H. Principles of DNA methylation and their implications for biology and medicine. Lancet 2018, 392, 777–786. [CrossRef]
- Wei, J.W.; Huang, K.; Yang, C.; Kang, C.S. Non-coding RNAs as regulators in epigenetics. Oncol Rep. 2017, 37, 3–9.
- Damiani, I.; Solberg, E.H.; Iyer, M.; Cheng, P.; Weldy, C.S.; Kim, J.B. Environmental pollutants and atherosclerosis: Epigenetic mechanisms linking genetic risk and disease. Atherosclerosis 2025, 404, 119131. [CrossRef]
- Wen, Y.; Kong, Y.; Cao, G.; Xu, Y.; Zhang, C.; Zhang, J.; Xiao, P.; Wang, Y. Di-n-butyl phthalate regulates vascular smooth muscle cells phenotypic switching by MiR-139–5p-MYOCD pathways. Toxicology 2022, 477, 153279. [CrossRef]
- Kong, Y.; Wen, Y.; Cao, G.; Xu, Y.; Zhang, C.; Tang, C.; Zhang, J.; Wang, Y. Di-n-butyl phthalate promotes monocyte recruitment via miR-137-3p-SP1-MCP-1 pathway. Ecotoxicol. Environ. Saf. 2022, 236, 113491. [CrossRef]
- Liu, C.; Qin, Q.; Xu, J.; Li, X.; Cong, H. Phthalate promotes atherosclerosis through interacting with long-non coding RNA and induces macrophage foam cell formation and vascular smooth muscle damage. Chemosphere 2022, 308, 136383. [CrossRef]
- Wang, K.; Li, M.; Sartor, M.A.; Colacino, J.A.; Dolinoy, D.C.; Svoboda, L.K. Perinatal Exposure to Lead or Diethylhexyl Phthalate in Mice: Sex-Specific Effects on Cardiac DNA Methylation and Gene Expression across Time. Environ. Heal. Perspect. 2025, 133. [CrossRef]
- Svoboda, L.K.; Wang, K.; Cavalcante, R.G.; Neier, K.; A Colacino, J.; A Sartor, M.; Dolinoy, D.C. Sex-Specific Programming of Cardiac DNA Methylation by Developmental Phthalate Exposure. Epigenetics Insights 2020, 13. [CrossRef]
- Wu, W.; Wang, Z.; Yang, W.; Feng, X.; Yang, Y.; Xu, M.; Li, B.; Wu, P.; Cao, Z. Association between prenatal phthalate exposure and preschoolers’ blood pressure: Mediating role of DNA methylation in hypertension-related genes. Environ. Pollut. 2025, 390, 127450. [CrossRef]
- Lin, C.-Y.; Chen, C.-W.; Lee, H.-L.; Wu, C.; Wang, C.; Sung, F.-C.; Su, T.-C. Global DNA methylation mediates the association between urine mono-2-ethylhexyl phthalate and serum apoptotic microparticles in a young Taiwanese population. Sci. Total. Environ. 2022, 808, 152054. [CrossRef]
- Bai, C.; Yang, H.; Zhao, L.; Liu, L.; Guo, W.; Yu, J.; Li, M.; Liu, M.; Lai, X.; Zhang, X.; et al. The mediating role of plasma microRNAs in the association of phthalates exposure with arterial stiffness: A panel study. Environ. Res. 2022, 212, 113469. [CrossRef]
- Palombo, C.; Kozakova, M. Arterial stiffness, atherosclerosis and cardiovascular risk: Pathophysiologic mechanisms and emerging clinical indications. Vasc. Pharmacol. 2016, 77, 1–7. [CrossRef]
- Ference, B.A.; Graham, I.; Tokgozoglu, L.; Catapano, A.L. Impact of lipids on cardiovascular health: JACC health promotion series. J Am Coll Cardiol. 2018, 72, 1141–1156.
- Yin, L.; Yu, K.S.; Lu, K.; Yu, X. Benzyl butyl phthalate promotes adipogenesis in 3T3-L1 preadipocytes: A High Content Cellomics and metabolomic analysis. Toxicol. Vitr. 2016, 32, 297–309. [CrossRef]
- Meruvu, S.; Zhang, J.; Choudhury, M. Butyl Benzyl Phthalate Promotes Adipogenesis in 3T3-L1 Cells via the miRNA-34a-5p Signaling Pathway in the Absence of Exogenous Adipogenic Stimuli. Chem. Res. Toxicol. 2021, 34, 2251–2260. [CrossRef]
- Schaedlich, K.; Schmidt, J.; Kwong, W.Y.; Sinclair, K.D.; Kurz, R.; Jahnke, H.; Fischer, B. Impact of di-ethylhexylphthalate exposure on metabolic programming in P19 ECC-derived cardiomyocytes. J. Appl. Toxicol. 2014, 35, 861–869. [CrossRef]
- Kokai, D.; Filipovic, J.M.; Opacic, M.; Ivelja, I.; Banjac, V.; Stanic, B.; Andric, N. In vitro and in vivo exposure of endothelial cells to dibutyl phthalate promotes monocyte adhesion. Food Chem. Toxicol. 2024, 188, 114663. [CrossRef]
- Sui, Y.; Meng, Z.; Chen, J.; Liu, J.; Hernandez, R.; Gonzales, M.B.; Gwag, T.; Morris, A.J.; Zhou, C. Effects of Dicyclohexyl Phthalate Exposure on PXR Activation and Lipid Homeostasis in Mice. Environ. Heal. Perspect. 2021, 129. [CrossRef]
- Golestanzadeh, M.; Riahi, R.; Kelishadi, R. Association of exposure to phthalates with cardiometabolic risk factors in children and adolescents: a systematic review and meta-analysis. Environ. Sci. Pollut. Res. 2019, 26, 35670–35686. [CrossRef]
- Harley, K.G.; Berger, K.; Rauch, S.; Kogut, K.; Henn, B.C.; Calafat, A.M.; Huen, K.; Eskenazi, B.; Holland, N. Association of prenatal urinary phthalate metabolite concentrations and childhood BMI and obesity. Pediatr. Res. 2017, 82, 405–415. [CrossRef]
- Wu, H.-T.; Liao, C.-C.; Peng, C.-F.; Lee, T.-Y.; Liao, P.-H. Exploring the application of machine learning to identify the correlations between phthalate esters and disease: enhancing nursing assessments. Heal. Inf. Sci. Syst. 2024, 13, 1–12. [CrossRef]
- Jiang, H.; Lee, T.; Ha, S.; Hwang, J.; Shin, J.; Kim, Y.-P.; Jung, H.-I. Machine Learning-Driven Electrochemical Aptasensing Platform for Highly Accurate Prediction of Phthalate Concentration in Multiple River Sites. BioChip J. 2025, 19, 133–141. [CrossRef]
- Parsa, S.; Shah, P.; Doijad, R.; Rodriguez, F. Artificial Intelligence in Ischemic Heart Disease Prevention. Curr. Cardiol. Rep. 2025, 27, 1–11. [CrossRef]
- Ward, A.; Sarraju, A.; Chung, S.; Li, J.; Harrington, R.; Heidenreich, P.; Palaniappan, L.; Scheinker, D.; Rodriguez, F. Machine learning and atherosclerotic cardiovascular disease risk prediction in a multi-ethnic population. npj Digit. Med. 2020, 3, 1–7. [CrossRef]
- Sarraju, A.; Ward, A.; Chung, S.; Li, J.; Scheinker, D.; Rodríguez, F. Machine learning approaches improve risk stratification for secondary cardiovascular disease prevention in multiethnic patients. Open Hear. 2021, 8, e001802. [CrossRef]
- Alireza, Z.; Maleeha, M.; Kaikkonen, M.; Fortino, V. Enhancing prediction accuracy of coronary artery disease through machine learning-driven genomic variant selection. J. Transl. Med. 2024, 22, 1–14. [CrossRef]
- Nurmohamed, N.S.; Pereira, J.P.B.; Hoogeveen, R.M.; Kroon, J.; Kraaijenhof, J.M.; Waissi, F.; Timmerman, N.; Bom, M.J.; Hoefer, I.E.; Knaapen, P.; et al. Targeted proteomics improves cardiovascular risk prediction in secondary prevention. Eur. Hear. J. 2022, 43, 1569–1577. [CrossRef]
- Wu, J.; Giles, C.; Dakic, A.; Beyene, H.B.; Huynh, K.; Wang, T.; Meikle, T.; Olshansky, G.; Salim, A.; Duong, T.; et al. Lipidomic Risk Score to Enhance Cardiovascular Risk Stratification for Primary Prevention. JACC 2024, 84, 434–446. [CrossRef]
- Treskesm R,W.; van Winden, L.A.M.; van Keulen, N.; van der Velde, E.T.; Beeres, S.L.M.A.; Atsma, D.E.; Schalij, M.J. Effect of Smartphone-Enabled Health Monitoring Devices vs regular follow-up on blood pressure control among patients after myocardial infarction. JAMA Netw Open. 2020, 3, e202165.
- Shufelt, C.L.; Kim, A.; Joung, S.; Barsky, L.; Arnold, C.; Cheng, S.; Dhawan, S.; Fuller, G.; Speier, W.; Lopez, M.; et al. Biometric and Psychometric Remote Monitoring and Cardiovascular Risk Biomarkers in Ischemic Heart Disease. J. Am. Hear. Assoc. 2020, 9. [CrossRef]
- Hughes, J.W.; Tooley, J.; Soto, J.T.; Ostropolets, A.; Poterucha, T.; Christensen, M.K.; Yuan, N.; Ehlert, B.; Kaur, D.; Kang, G.; et al. A deep learning-based electrocardiogram risk score for long term cardiovascular death and disease. npj Digit. Med. 2023, 6, 1–9. [CrossRef]
- Awasthi, S.; Sachdeva, N.; Gupta, Y.; Anto, A.G.; Asfahan, S.; Abbou, R.; Bade, S.; Sood, S.; Hegstrom, L.; Vellanki, N.; et al. Identification and risk stratification of coronary disease by artificial intelligence-enabled ECG. eClinicalMedicine 2023, 65, 102259. [CrossRef]
- Naghavi, M.; Reeves, A.P.; Atlas, K.; Zhang, C.; Atlas, T.; Henschke, C.I.; Yankelevitz, D.F.; Budoff, M.J.; Li, D.; Roy, S.K.; et al. Artificial intelligence applied to coronary artery calcium scans (AI-CAC) significantly improves cardiovascular events prediction. npj Digit. Med. 2024, 7, 1–9. [CrossRef]
- Eslami, P.; Parmar, C.; Foldyna, B.; Scholtz, J.-E.; Ivanov, A.; Zeleznik, R.; Lu, M.T.; Ferencik, M.; Vasan, R.S.; Baltrusaitis, K.; et al. Radiomics of Coronary Artery Calcium in the Framingham Heart Study. Radiol. Cardiothorac. Imaging 2020, 2, e190119. [CrossRef]
- Zou, L.-M.; Xu, K.-T.; Wang, Y.-N. Research advances and applications of artificial intelligence in cardiac CT. Meta-Radiology 2024, 2. [CrossRef]
- Choi, A.D.; Marques, H.; Kumar, V.; Griffin, W.F.; Rahban, H.; Karlsberg, R.P.; Zeman, R.K.; Katz, R.J.; Earls, J.P. CT Evaluation by Artificial Intelligence for Atherosclerosis, Stenosis and Vascular Morphology (CLARIFY): A Multi-center, international study. J. Cardiovasc. Comput. Tomogr. 2021, 15, 470–476. [CrossRef]
- Arnett, D.K.; Blumenthal, R.S.; Albert, M.A.; Buroker, A.B.; Goldberger, Z.D.; Hahn, E.J.; Himmelfarb, C.D.; Khera, A.; Lloyd-Jones, D.; McEvoy, J.W.; et al. 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease: Executive Summary: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 2019, 140. [CrossRef]
- Upton, R.; Mumith, A.; Beqiri, A.; Parker, A.; Hawkes, W.; Gao, S.; Porumb, M.; Sarwar, R.; Marques, P.; Markham, D.; et al. Automated Echocardiographic Detection of Severe Coronary Artery Disease Using Artificial Intelligence. JACC: Cardiovasc. Imaging 2022, 15, 715–727. [CrossRef]
- Barzilai, D.H.; Cohen-Shelly, M.; Sorin, V.; Zimlichman, E.; Massalha, E.; Allison, T.G.; Klang, E. Machine learning in cardiac stress test interpretation: a systematic review. Eur. Hear. J. - Digit. Heal. 2024, 5, 401–408. [CrossRef]
- Pieszko, K.; Shanbhag, A.D.; Singh, A.; Hauser, M.T.; Miller, R.J.H.; Liang, J.X.; Motwani, M.; Kwieciński, J.; Sharir, T.; Einstein, A.J.; et al. Time and event-specific deep learning for personalized risk assessment after cardiac perfusion imaging. npj Digit. Med. 2023, 6, 1–11. [CrossRef]
- Meder, B.; Asselbergs, F.W.; Ashley, E. Artificial intelligence to improve cardiovascular population health. Eur. Hear. J. 2025, 46, 1907–1916. [CrossRef]
- Muse, E.D.; Topol, E.J. Transforming the cardiometabolic disease landscape: Multimodal AI-powered approaches in prevention and management. Cell Metab. 2024, 36, 670–683. [CrossRef]
- Fontanelli, L.; Castronovo, A.; Ferri, C.; Vozzi, F.; Recchia, F.A.; Borghini, A. iPSC-Derived Endothelial Cells as Experimental Models for Predictive and Personalized Strategies in Cardiovascular and Cerebrovascular Disease. Int. J. Mol. Sci. 2026, 27, 780. [CrossRef]
| Country | Regulation | Phthalate | Reference |
| European Union | 1999/815/EC: temporary restriction on children’s toys |
BBP, DEHP, DINP, DIDP, DnOP | [48] |
| European Union | Directive 2005/84/EC: permanent restriction on children’s toys and articles |
BBzP, DEHP, DnBP | [51] |
| European Union | Commission Regulation (EU) 2018/2005: maximum concentration of 0.1% by weight in plasticized material in all plastic products | BBP, DBP, DEHP, DIBP | [52] |
| European Union | Question numbers: EFSA-Q-2017-00588/-00589/-00590, EFSA-Q-2018-00800/-00801: TDI of 0.05 mg/kg body weight per day | BBP, DBP, DEHP, DINP | [53] |
| European Union | EU Regulation No. 2023/2482: restrictions to the use of DEHP in medical devices | DEHP | [55] |
| United States | EPA-HQ-OPPT-2018-0435; EPA-HQ-OPPT-2024-0073: established risk for reproductive toxicity in female workers | DIDP | [57] |
| United States | EPA-HQ-OPPT-2018-0436; EPA-HQ-OPPT-2024-0073: established risk for developmental toxicity, hepatotoxicity, and cancer at high exposure | DINP | [58] |
| China | GB 26572-2025: maximum concentration of 0.1% by weight in electrical and electronic products | BBP, DBP, DEHP, DIDP | [59] |
| Clues | Reference | Pitfalls | Reference |
| Serum MMP levels significantly related to the number of plaques in an inverted U-shaped manner | [89] | No significant association between circulating MEP levels and plaque GSM | [89] |
| Serum MMP and MBzP levels significantly and inversely associated with IMT | [89,97] | No significant association of MEHP, MEP, and MiBP in serum with IMT | [89] |
| MBzP, MiBP, and MMP in serum significantly and positively associated with IM-GSM and plaque GSM | [89,97] | No significant association between serum levels of MEP, MEHP, MiBP, and MMP and FRS | [95] |
| MEP in serum significantly and positively associated with IM-GSM | [89] | Serum MBzP levels not significantly related to plaque prevalence | [97] |
| Serum MEHP concentration inversely related to IM-GSM and plaque GSM | [89] | Cross-sectional design | [32,89,95,97,99,105,106] |
| Circulating levels of MEHP and MMP associated with LDL- cholesterol, MEP with diastolic blood pressure, and MiBP with fasting glucose |
[95] | Single-spot urine measurement | [32,99,105,106,112] |
| Urinary MEHP concentration significantly and positively associated with serum levels of CD31+/CD42a−, CD31+/CD42a+, and CD14 |
[99] | Study performed exclusively on Caucasian individuals aged 70 | [89,95,97] |
| Urinary MEHP, MnBP, and ∑DEHP levels significantly and positively associated with both maximal and mean values of carotid IMT as both continuous and categorical variables | [105] | No significant association of urinary MEHHP and MEHOP levels with EMPs and PMPs | [99] |
| Urinary MEHP levels significantly and positively correlated with 5mdC/dG and carotid IMT | [106] | Study conducted on adolescents and young adults with abnormal urinalysis | [99] |
| Significantly positive association of urinary levels of ∑DEHP, ∑LMWP, ∑HMWP, MEP, and MECPP significantly with hs-cTnI | [32] | No adjustment of associations for medications, genetics and other occupational environmental factors (e.g., bisphenol A, perfluorooctane sulfate, air pollutants) | [99,105,106,112] |
| Phthalate co-exposure significantly associated with an increase in hs-cTnI percentage | [32] | Urinary concentration of MEHHP and MEHOP not significantly associated with carotid IMT | [105] |
| Highest tertiles of urinary MEHP and MiBP significantly associated with increased risk of CHD | [112] | MEHHP and MEHOP not significantly associated either with carotid IMT or 5mdC/dG | [106] |
| Increased serum concentration of hsCRP and D-dimer significantly and positively associated across tertiles of urinary ∑DEHP, MEHP, MEHHP, and MEHP | [112] | Study performed exclusively on young adults | [106] |
| Increased serum concentration of fibrinogen significantly and positively associated across tertiles of urinary ∑DEHP, MEHHP, and MEHP |
[112] | No measurement of methylation at specific gene loci | [106] |
| No significant association of urinary concentration of MEHP, MEHHP, MEHOP, MCPP, MBzP, MMP, MBP, and MiBP, with hs-cTnI | [32] | ||
| Underestimation of hs-cTnI due to its degradation if long stored under -80°C | [32] | ||
| No significant association of urinary MEHHP, MEHOP, ΣDEHP, MMP, MnBP, MBzP, MiBP with increased risk of CHD |
[112] |
| Oxidative stress, inflammation, and apoptosis | ||||
|
Identified/ Described mechanisms |
Biological meaning | Models and treatments | Methods | References |
| MEHP → ↑ intracellular ROS, ↓ GSH, ↑ MDA → mitochondrial dysfunction → apoptosis | Oxidative stress–mediated lipid peroxidation and mitochondrial damage leading to activation of the intrinsic apoptotic pathway in endothelial cells |
In vitro model: Human umbilical vein endothelial cells (HUVEC) Treatment: MEHP exposure at 0, 6.25, 12.5, 25, 50, and 100 μM; 24 h) |
• Intracellular ROS measurement (DCFH-DA fluorescence assay) • GSH content determination (colorimetric assay) • Lipid peroxidation assessment via MDA levels (TBARS assay) • Mitochondrial membrane potential (ΔΨm) analysis (JC-1 staining) • Cytochrome c release (Western blot) • Apoptosis assessment (Annexin V/PI flow cytometry) • Caspase-9 and caspase-3 activation (Western blot / activity assays) |
[103] |
| MEHP → autophagy activation → lysosomal destabilization → cathepsin B release → mitochondrial dysfunction → intrinsic apoptosis | Autophagy-dependent lysosomal–mitochondrial crosstalk promotes endothelial apoptosis via activation of the intrinsic apoptotic pathway |
In vitro model: Human endothelial cells (HUVEC) Treatment: MEHP exposure at 0, 25, 50, 100, and 200 μM; 6, 12, and 24 h |
• Autophagy assessment (LC3-I/LC3-II conversion, Beclin-1 expression; Western blot) • Autophagosome formation (GFP-LC3 fluorescence microscopy) • Lysosomal membrane permeabilization (LysoTracker staining) • Cathepsin B release and activity (Western blot and enzymatic assay) • Mitochondrial membrane potential (ΔΨm) analysis (JC-1 staining) • Cytochrome c release (Western blot) • Apoptosis quantification (Annexin V/PI flow cytometry) • Caspase-9 and caspase-3 activation (Western blot) • Pharmacological inhibition of autophagy and cathepsin B to confirm pathway involvement |
[118] |
| MEHP → ↑ ROS → Akt1 pathway inhibition → autophagy activation → autophagic cell death | ROS-mediated Akt1 signaling disruption promotes excessive autophagy and autophagic cell death in endothelial cells |
In vitro model: Human vascular endothelial cells (EA.hy926) Treatment: MEHP exposure at 0, 25, 50, 100, and 200 μM; 24 h |
• Intracellular ROS measurement (DCFH-DA fluorescence assay) • Autophagy markers analysis (LC3-I/LC3-II conversion, Beclin-1 expression; Western blot) • Autophagosome formation (transmission electron microscopy; fluorescence microscopy) • Akt1 pathway analysis (Akt1 phosphorylation status; Western blot) • Pharmacological modulation of ROS (antioxidant pretreatment) • Cell viability and autophagic cell death assays (MTT assay, LDH release) |
[119] |
| DEHP → ↑ ROS, ↓ antioxidant defenses → PTGS2 (COX-2) upregulation → inflammation → apoptosis | Oxidative stress–driven inflammatory signaling promotes cardiomyocyte injury and apoptotic cell death |
In vitro model: primary rat cardiomyocytes Treatment: DEHP exposure at 0, 25, 50, 100, and 150 μM; 24, 48 h |
• Intracellular ROS measurement (fluorescent ROS probes) • Antioxidant system evaluation (GSH content, antioxidant enzyme activity assays) • Lipid peroxidation assessment (MDA levels) • Inflammatory pathway analysis (PTGS2/COX-2 expression; Western blot and qPCR) • Pro-inflammatory cytokine measurement (ELISA) • Apoptosis assessment (Annexin V/PI staining; caspase-3 activation) • Cell viability assays (CCK-8 / MTT) |
[120] |
| DEP/MEP → direct binding to SOD → structural destabilization → ↓ SOD catalytic activity → ↑ oxidative stress susceptibility | Direct inhibition of antioxidant enzyme activity compromises cellular redox homeostasis, favoring oxidative stress |
In silico model: Human superoxide dismutase (SOD) structure Treatment: Molecular interaction with DEP and its metabolite MEP |
• Molecular docking analysis (DEP–SOD and MEP–SOD binding affinity and interaction sites) • Molecular dynamics simulations to assess protein structural stability • Analysis of conformational changes (RMSD, RMSF, radius of gyration) • Evaluation of catalytic site perturbation and enzyme flexibility |
[121] |
| DEHP → ↑ cardiac ROS, ↑ lipid peroxidation → dyslipidemia → ↑ atherogenic index | Oxidative stress–induced cardiac damage and lipid metabolism alteration increase atherosclerosis and cardiovascular risk |
Animal model: BALB/c mice (male) Treatment: intraperitoneal administration of DEHP at 5, 50, and 200 mg/kg body weight daily for 30 consecutive days |
• Cardiac oxidative stress evaluation (MDA levels; protein carbonyl content assays) • Lipid peroxidation assessment (TBARS assay) • Serum lipid profile analysis (total cholesterol, triglycerides, LDL-C, HDL-C) • Atherogenic index calculation • Histopathological analysis of cardiac tissue |
[123] |
| DEHP → cholesterol homeostasis disruption + endothelial inflammation → accelerated atherosclerosis | Chronic DEHP exposure promotes atherogenesis via lipid dysregulation and vascular inflammation |
Animal model: Apolipoprotein E–deficient mice (ApoE⁻/⁻, male) Treatment: oral administration of DEHP at 1,500 mg/kg/day via drinking water for 4 weeks |
• Serum lipid profile (total cholesterol, LDL-C, HDL-C) • Atherosclerotic lesion analysis (Oil Red O staining of aortic root and en face aorta) • Endothelial inflammation markers (VCAM-1, ICAM-1; Western blot, immunohistochemistry) • Macrophage infiltration in plaques • Oxidative stress and inflammatory signaling evaluation |
[124] |
| DEHP/MEHP → SPHK1/S1PR2 sphingolipid pathway activation → cardiomyocyte pyroptosis → cell death | Disruption of sphingolipid metabolism triggers inflammatory programmed cell death, leading to myocardial injury |
Animal model: Male C57BL/6J mice Treatment: oral administration of DEHP at 500 mg/kg/day via gavage for 28 consecutive days |
• Histopathology (H&E staining; assessment of disorganized myocardial fibers, collagen deposition) • Serum cardiac injury markers (CK-MB, CRP; ELISA) • Pyroptosis assessment (caspase-1 activation; GSDMD cleavage; Western blot) • Sphingolipid pathway analysis (SPHK1, S1PR2 expression; qPCR and Western blot) • Pharmacological inhibition of SPHK1/S1PR2 to confirm pathway involvement |
[125] |
| DMP → ↑ ROS → intrinsic (Bax/Bcl-2) & extrinsic (Fas/FasL) apoptotic pathways → caspase-dependent cardiomyocyte apoptosis | Oxidative stress–mediated activation of multiple apoptotic cascades promotes cardiomyocyte death |
Animal model: Sprague-Dawley rats (male) Treatment: intragastric (oral gavage) administration of DMP at 0, 50, 100, and 200 mg/kg/day |
• Transcriptomic analysis for apoptotic gene expression (Bcl-2 family, Fas/FasL) • Flow cytometry for apoptosis quantification (Annexin V/PI) • Caspase activity assays (caspase-3, caspase-9) • ROS measurement (DCFH-DA fluorescence assay) • Evaluation of antioxidant status (GSH, SOD activity) |
[126] |
| Phthalates → NLRP3 inflammasome activation → ↑ IL-1β, ↑ IL-18 → neutrophil & macrophage infiltration → cardiac inflammation | Pro-inflammatory signaling and innate immune activation exacerbate myocardial injury |
Animal model: Male C57BL/6N mice Treatment: acute exposure to DEHP via intraperitoneal injection at 30 mg/kg/day for 7 days during the recovery period following surgically-induced myocardial infarction |
• NLRP3 inflammasome assessment (NLRP3, ASC, caspase-1 expression; Western blot) • IL-1β and IL-18 quantification (ELISA) • Histology & immunohistochemistry for neutrophil and macrophage infiltration • Cardiac injury evaluation (histopathology, infarct size measurement) |
[127] |
| DEHP → ↑ lipid peroxidation + mitochondrial damage → Nrf2/HO-1 pathway activation → ferroptosis in cardiomyocytes | Oxidative stress–induced iron-dependent cell death contributes to cardiomyocyte injury |
Animal model: Male C57BL/6J mice Treatment: oral gavage administration of DEHP at 50, 200, and 500 mg/kg body weight per day for 28 consecutive days. |
• Lipid peroxidation assessment (MDA assay; TBARS) • Mitochondrial damage evaluation (TEM; mitochondrial membrane potential assay) • Ferroptosis markers (GPX4 expression, ACSL4, iron accumulation; Western blot, biochemical assays) • Nrf2/HO-1 pathway analysis (Western blot, qPCR, immunohistochemistry) • ROS measurement (DCFH-DA fluorescence) |
[128] |
| DBP + high-fat diet → ↑ ROS, ↑ MDA, ↓ GSH → membrane & mitochondrial damage → activation of NLRP3 inflammasome, caspase-1, GSDMD → inflammatory + pyroptotic pathways → disrupted lipid metabolism & endocrine homeostasis → cardiac fibrosis & dysfunction | Oxidative stress, inflammation, pyroptosis, and metabolic/endocrine disruption collectively promote structural myocardial damage and cardiotoxicity |
Animal model: Sprague-Dawley rats (male) Treatment: oral gavage administration of DBP at 0.01, 1, and 50 mg/kg/day for 12 weeks combined with high-fat diet for sub-chronic period |
• Oxidative stress measurement (ROS assay, MDA/TBARS, GSH levels) • Mitochondrial function assays (mitochondrial membrane potential, morphology via TEM) • Inflammatory & pyroptosis markers (NLRP3, caspase-1, GSDMD; Western blot, immunohistochemistry) • Lipid profile & endocrine parameter evaluation (serum cholesterol, triglycerides, hormones) • Cardiac histopathology (fibrosis assessment; Masson’s trichrome staining) • Functional assessment (echocardiography, if included) |
[129] |
| DEHP → TGF-β1/Smad/PAI-1 pathway activation in M1 macrophages → ↑ tissue factor–bearing microparticle release → procoagulant activity | Vascular inflammation and enhanced thrombogenic potential contribute to CVD progression |
In vitro model: Human M1 macrophages Treatment: DEHP at 0, 10, 50, and 100 μM; 24 h |
• Microparticle isolation and quantification (flow cytometry, annexin V labeling) • Tissue factor expression analysis (Western blot, qPCR) • Signaling pathway assessment (TGF-β1/Smad/PAI-1; Western blot, inhibitor studies) • Functional coagulation assays (procoagulant activity of microparticles) |
[130] |
| Urinary phthalate metabolites (MEP, MBP, MiBP, MBzP, sum of six) → ↑ oxidative stress biomarkers (8-OHdG, MDA) → ↑ blood pressure & hypertension prevalence | Phthalate-induced oxidative stress contributes to cardiovascular risk and elevated blood pressure in humans |
Human cohort: >1000 individuals Treatment: No experimental treatment; observational study |
• Urinary phthalate metabolite quantification (LC-MS/MS) • Oxidative stress biomarkers in urine/plasma (8-OHdG, MDA; ELISA/HPLC) • Blood pressure measurement (systolic/diastolic) • Statistical correlation analysis (regression models, adjustment for confounders) |
[131] |
| Urinary MEHP → ↑ MDA, ↓ SOD → oxidative stress associated with CHD | Oxidative stress contributes to CHD pathology |
Human case-control study: CHD patients and matched controls Treatment: No experimental treatment; observational study |
• Urinary MEHP quantification (LC-MS/MS) • Oxidative stress markers (MDA, SOD activity; ELISA/biochemical assays) • Correlation/statistical analysis with CHD status |
[35] |
| Urinary MEHP/DEHP metabolites → ↑ endothelial (CD31+/CD42a-) & platelet (CD31+/CD42a+) microparticles → activation of monocytes, macrophages, neutrophils (CD14) | Microparticle release reflects apoptosis and inflammation contributing to atherosclerosis |
Human cohort: Adolescents and young adults Treatment: No experimental treatment; observational study |
• Urinary phthalate metabolite measurement (LC-MS/MS) • Microparticle identification & quantification (flow cytometry: CD31, CD42a, CD14 markers) • Correlation/statistical analysis between urinary metabolites and microparticle levels |
[99] |
| Urinary DEHP metabolites → ↑ hs-CRP, fibrinogen, D-dimer → vascular inflammation & thrombosis | Phthalate-induced oxidative/inflammatory stress promotes pro-atherothrombotic state in CHD patients |
Human case-control study: CHD patients Treatment: No experimental treatment; observational study |
• Urinary DEHP metabolite measurement (LC-MS/MS) • Serum inflammatory and thrombotic marker quantification (hs-CRP, fibrinogen, D-dimer; ELISA/clinical assays) • Statistical correlation analysis |
[132] |
| Phthalate metabolites → binding to PI3K-Akt, JAK-STAT, BCL2, PIK3CA → oxidative stress, inflammatory signaling, apoptosis | Oxidative stress, inflammation, and apoptosis drive cardiovascular toxicity in cardiac and vascular cells |
In silico + human data: NHANES 2005–2018 dataset; computational network toxicology analysis Treatment: No experimental treatment; observational / computational study |
• Pathway enrichment analysis (PI3K-Akt, JAK-STAT, BCL2, PIK3CA) • Protein-protein interaction network construction • Molecular docking simulations (binding affinity between phthalates and target proteins) • Integration with NHANES biomarker/exposure data |
[33] |
| Mitochondrial DNA alterations and dysfunction | ||||
|
Identified/ Described mechanisms |
Biological meaning | Models and treatments | Methods | References |
| DEHP → ↑ mitochondrial ROS → oxidative damage to membranes, proteins, mtDNA; disrupted mitochondrial dynamics & biogenesis → defective oxidative phosphorylation, ↓ mtDNA copy number | Mitochondrial redox imbalance and structural/functional impairment contribute to cardiomyocyte dysfunction and CVD risk |
In vitro models: human endothelial cells and cardiomyocytes Treatment: DEHP from 10 to 100 μM; 24, 48 h Animal model: C57BL/6 mice Treatment: DEHP by oral gavage at 50, 200, and 500 mg/kg/day for 28 days |
• ROS measurement (MitoSOX, DCFDA) • Antioxidant enzyme activity (SOD, GSH) • Mitochondrial membrane integrity (JC-1, TMRE) • mtDNA copy number quantification (qPCR) • Mitochondrial dynamics & biogenesis markers (Western blot, immunofluorescence) • Oxidative phosphorylation assessment (Seahorse XF, ATP assays) |
[34] |
|
In vitro model: primary human vascular endothelial cells Treatment: DEHP at 10, 50, and 100 μM; 24 h Animal model: Sprague-Dawley rats Treatment: DEHP at 100 and 400 mg/kg/day via oral gavage for 12 weeks |
[30] | |||
| DBP → mitochondrial fragmentation, ↓ ΔΨm, ↓ ATP synthesis | Collapse of mitochondrial bioenergetic capacity, contributing to cardiomyocyte/vascular cell dysfunction |
In vitro model: zebrafish (Danio rerio) ZF4 cells Treatment: DBP exposure at 0, 10, 50, and 100 μM; 24 h |
• Mitochondrial morphology (fluorescence microscopy, Mitotracker staining) • Mitochondrial membrane potential (ΔΨm; JC-1/TMRE) • ATP content measurement (bioluminescence assay) |
[143] |
| DBP → ↑ mtDNA lesions, ↓ mtDNA copy number → mtDNA release → cGAS-STING pathway activation | mtDNA damage links mitochondrial dysfunction to cellular inflammation and contributes to cardiovascular risk |
Animal model: zebrafish (Danio rerio) embryos and larvae Treatment: DBP at 0.1, 1, and 10 μM in the aquatic medium; up to 7 days post-fertilization |
• mtDNA damage assessment (long-amplicon qPCR, lesion quantification) • mtDNA copy number quantification (qPCR) • Cytosolic mtDNA measurement (PCR, imaging) • cGAS-STING pathway activation (Western blot, immunofluorescence for cGAS, STING, downstream cytokines) |
[144] |
| ↓ mtDNA copy number → ↑ risk of atherosclerosis and major cardiovascular events | Reduced mtDNA integrity is linked to higher susceptibility to CHD and CVD |
Human cohort: Leukocytes from >1000 individuals Treatment: Observational study, no experimental treatment |
• mtDNA copy number quantification (qPCR) • Statistical association with cardiovascular outcomes |
[134] |
| ↓ mtDNA copy number → ↑ risk of CVD, CHD, HF, stroke, all-cause mortality | mtDNA copy number is a robust biomarker of mitochondrial dysfunction and adverse cardiovascular outcomes |
Human observational studies: Multiple cohorts included in meta-analysis Treatment: Observational, no experimental intervention |
• Meta-analysis of observational studies • Risk ratio (RR) calculation for CVD, CHD, HF, stroke, all-cause mortality |
[145] |
| DNA methylation and non-coding RNAs | ||||
|
Identified/ Described mechanisms |
Biological meaning | Models and treatments | Methods | References |
| DBP → ↑ miR-139-5p → ↓ myocardin (MYOCD) → VSMC phenotypic switch (contractile → synthetic) | Promotion of atherosclerotic plaque formation and instability |
In vitro model: s A7r5 rat vascular smooth muscle cells (VSMCs) Treatment: DBP at 10 μM; 24 h |
• miRNA expression (qPCR) • MYOCD protein quantification (Western blot) • VSMC phenotypic markers (immunofluorescence, α-SMA, SM22α for contractile; OPN, vimentin for synthetic) • Functional assays for cell proliferation/migration (scratch assay, transwell) |
[149] |
| DBP → ↓ miR-137-3p → ↑ SP1 → ↑ MCP-1 | Enhanced monocyte recruitment to endothelium, promoting early atherogenesis |
In vitro model: EA.hy926 human vascular endothelial cells Treatment: DBP at 10 μM; 24 h |
• miRNA expression (qPCR) • SP1 and MCP-1 protein levels (Western blot, ELISA) • Monocyte adhesion assay (fluorescent-labeled monocytes) |
[150] |
| DEHP → ↑ GAS5 (lncRNA) → sequestration of miR-145-5p → ↑ VSMC proliferation + ↑ apoptosis | Promotion of vascular damage, plaque instability, and risk of coronary artery disease (CAD) |
In vitro model: RAW264.7 murine macrophages and rat vascular smooth muscle cells (VSMCs) Treatment: DEHP at 100 μM; 24 h |
• lncRNA and miRNA expression (qPCR) • VSMC proliferation assay (BrdU, Ki-67 staining) • Apoptosis assay (Annexin V/PI staining, caspase activity) • Protein markers of VSMC phenotype (Western blot, α-SMA, SM22α) |
[151] |
| perinatal DEHP exposure → ↑ Differentially methylated regions (DMRs) and ↑ differentially expressed genes (DEGs) → persistent alteration of cardiac cell-type proportion and composition | Early-life epigenetic reprogramming of the heart that persistently modifies gene expression patterns and cardiac cellular architecture, predisposing to increased cardiovascular risk later in life |
Animal model: C57BL/6J mice (both sexes) Treatment: DEHP at 25 mg/kg chow or control chow, starting 2 weeks prior to mating and continuing through pregnancy and lactation until weaning at postnatal day 21 |
• Genome-wide DNA methylation analysis (RRBS) • Transcriptomic profiling (RNA-seq) • Cell-type proportion inference • Longitudinal epigenetic and gene expression analysis |
[152] |
| DEHP (developmental exposure) → ↑ DMRs → persistent cardiac epigenetic reprogramming (sex-specific) | Developmental DEHP exposure programs long-lasting, sex-dependent alterations in cardiac gene regulation, potentially increasing later-life CVD risk |
Animal model: C57BL/6J mice (both sexes) Treatment: oral, DEHP at 25 mg/kg chow beginning 2 weeks prior to mating, continuing through pregnancy and lactation until weaning at postnatal day 21 |
• RRBS (DNA methylation profiling) • RNA-seq (gene expression) • DMR and DEG analysis (sex-stratified) |
[153] |
| Prenatal phthalates → ↑ DNA methylation changes (ECE1, SCNN1G) → ↑ blood pressure in offspring | Prenatal phthalate exposure epigenetically programs hypertension risk, linking early-life exposure to later cardiovascular dysfunction |
Human study: pregnant women (n = 198) during 3rd trimester and their preschool-age children Treatment: No experimental treatment; observational exposure to phthalates (MMP, MEP, MEcPP) |
• Urinary phthalate metabolites: LC–MS/MS • Blood pressure measurement in children: standardized sphygmomanometry • DNA methylation analysis: bisulfite conversion + targeted methylation assays (hypertension-related genes, e.g. ECE1, SCNN1G) • Mediation analysis: statistical modeling |
[154] |
| MEHP → ↑ global DNA methylation → ↑ carotid intima-media thickness | Phthalate exposure is associated with epigenetic alterations that may contribute to early atherosclerotic changes |
Human study: young individuals, Taiwanese Treatment: No experimental treatment; cross-sectional observational study |
• Urinary DEHP metabolites quantified via LC–MS/MS • Global DNA methylation measured in blood samples (ELISA-based 5-methylcytosine quantification) • Carotid intima-media thickness (CIMT) assessed by ultrasound imaging • Statistical correlation and regression analyses |
[106] |
| MEHP → ↑ global DNA methylation → ↑ apoptotic microparticles (CD31+/CD42a-, CD14+) | Phthalate exposure induces vascular cell apoptosis via epigenetic modulation, linking DNA methylation changes to subclinical vascular injury |
Human study: young individuals, Taiwanese Treatment: No experimental treatment; cross-sectional observational study |
• Urinary MEHP quantification: LC–MS/MS • Serum apoptotic microparticles (CD31+/CD42a-, CD14+) measured by flow cytometry • Global DNA methylation: ELISA-based 5-methylcytosine quantification • Mediation analysis linking MEHP, DNA methylation, and microparticle levels |
[155] |
| MMP/MBP → ↑ miR-146a → ↑ arterial stiffness | Phthalate exposure modulates cardiovascular-related miRNAs, promoting subclinical vascular dysfunction linked to IHD risk | Human study: adults Treatment: No experimental treatment; panel observational study | • Urinary phthalate metabolites quantified via LC–MS/MS • Plasma miRNAs (miR-146a and others) quantified by qRT-PCR • Arterial stiffness measured via pulse wave velocity (PWV) • Statistical mediation analysis linking phthalates, miRNAs, and PWV |
[156] |
| MEHP → ↑ miR-155 / ↑ miR-208a → ↑ vascular inflammation / endothelial dysfunction | Phthalate exposure modulates cardiovascular-related miRNAs, promoting atherogenesis and CHD risk |
Human study: CHD patients vs healthy controls Treatment: No experimental treatment; case-control study) |
• Urinary MEHP quantified via LC–MS/MS • Plasma/serum miRNAs (miR-155, miR-208a) measured by qRT-PCR • Statistical comparison between CHD patients and controls |
[35] |
| Lipid accumulation | ||||
|
Identified/ Described mechanisms |
Biological meaning | Models and treatments | Methods | References |
| BBP → ↑ C/EBPα & ↑ PPARγ → ↑ adipogenesis / lipid accumulation | Phthalate promotes preadipocyte differentiation and metabolic reprogramming, leading to enhanced lipid storag | In vitro model: 3T3-L1 preadipocytes Treatment: BBP, 0.1–100 μM; 24 - 96 h | • Lipid droplet visualization: Oil Red O staining, high-content cellomics imaging • Gene/protein expression: qRT-PCR & Western blot for C/EBPα, PPARγ, downstream targets • Metabolomics: LC–MS-based profiling; pathway analysis (glyceroneogenesis, fatty acid synthesis) |
[159] |
| BBP → ↑ miR-34a-5p → ↑ adipogenesis / lipid accumulation | Phthalate promotes preadipocyte differentiation through miRNA-mediated signaling, enhancing lipid storage even without exogenous adipogenic stimuli |
In vitro model: 3T3-L1 preadipocytes Treatment: BBP, 1–50 μM; 48, 96 h |
• Lipid accumulation: Oil Red O staining • miRNA expression: qRT-PCR for miR-34a-5p • Adipogenic markers (C/EBPα, PPARγ) assessed by Western blot and qRT-PCR |
[160] |
| DEHP → ↑ PPARγ → ↑ Fabp4 → ↑ lipid accumulation | Phthalate exposure promotes metabolic reprogramming and lipid storage during cardiomyogenic differentiation | In vitro model: P19 embryonal carcinoma cells differentiating into cardiomyocytes Treatment: DEHP, 10–100 μM; 7 days | • Gene/protein expression: qRT-PCR and Western blot for PPARγ and Fabp4 • Lipid accumulation: Oil Red O staining • Cardiomyocyte differentiation monitored via immunostaining for cardiac markers |
[161] |
| DBP → ↑ adhesion molecules / ↑ chemokines → ↑ monocyte adhesion | Phthalate promotes endothelial activation, facilitating initiation and progression of atherosclerosis |
In vitro model: Human umbilical vein endothelial cells (HUVEC) Treatment: DBP, 1–50 μM; 24, 48 h Animal model: Male C57BL/6 mice Treatment: DBP, 50 mg/kg/day, oral gavage; 7 days |
• Monocyte adhesion assay: fluorescently labeled monocytes co-cultured with endothelial cells • Gene/protein expression: qRT-PCR & Western blot for adhesion molecules and chemokines • Histological analysis in mice: immunostaining of vascular tissue for adhesion molecules |
[162] |
| DEHP → ↑ oxLDL uptake → ↑ foam cell formation | Phthalate promotes macrophage lipid accumulation and accelerates atherosclerotic plaque development |
In vitro model: Human THP-1 macrophages Treatment: DEHP, 10–50 μM; 24–72 h Animal model: ApoE-deficient mice, male, 8 weeks Treatment: DEHP, 50 mg/kg/day, oral gavage; 12 weeks |
• Foam cell formation: Oil Red O staining, microscopy • Lipid uptake: fluorescently labeled oxLDL assay • Lipid profile in mice: serum cholesterol, triglycerides, LDL/HDL measurement • Histology: aortic plaque assessment via Oil Red O and immunostaining |
[151] |
| DCHP → ↑ PXR activation → ↑ lipogenic & ceramide genes | Phthalate promotes lipid synthesis and alters lipid homeostasis via nuclear receptor signaling |
Animal model: C57BL/6 mice, male Treatment: DCHP, 50 mg/kg/day, oral gavage; 4 weeks |
• Gene expression: qRT-PCR for PXR target genes (lipogenesis, ceramide synthesis) • Protein expression: Western blot for PXR and downstream targets • Lipid profiling: serum and hepatic lipid quantification • Histology: liver tissue staining for lipid accumulation |
[163] |
| Phthalate metabolites (MBP, MBzP, MEHP, MMP, MEOHP) → ↑ BMI / ↑ waist circumference / ↑ LDL-C / ↑ triglycerides | Phthalate exposure associated with increased cardiometabolic risk and lipid accumulation in pediatric population |
Human cohort: Children and adolescents Treatment: No experimental treatment; observational study |
• Anthropometric measurements: BMI, waist circumference • Blood biochemistry: LDL-C, triglycerides quantification • Statistical analysis: correlation between urinary phthalate metabolite levels and cardiometabolic markers |
[164] |
| Phthalate metabolites (MEHP, MBzP, MiBP, MMP) → ↑ risk of metabolic syndrome | Phthalate exposure linked to dysregulated lipid homeostasis, central obesity, insulin resistance, ↑ triglycerides; established risk factors for atherosclerosis and IHD | Human cohort: 25,365 individuals across 9 cross-sectional observational studies Treatment: No experimental treatment; observational | • Anthropometric measures: waist circumference, BMI • Blood biochemistry: triglycerides, fasting glucose, lipid profile • Statistical analysis: association between urinary phthalate metabolites and prevalence of metabolic syndrome |
[73] |
| Maternal urinary phthalate metabolites (DEP, DBP, DEHP) → ↑ BMI / ↑ risk of childhood overweight/obesity | Prenatal phthalate exposure linked to metabolic disorders in offspring, promoting higher adiposity and obesity risk | Human cohort: Mother-child pairs Treatment: No experimental treatment; prospective observational study | Treatment: No experimental treatment; prospective observational study • Maternal urinary phthalate metabolite quantification (DEP, DBP, DEHP) • Child anthropometrics: BMI, weight-for-age, height-for-age • Statistical analysis: association between maternal phthalate levels and child BMI/obesity outcomes |
[165] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.





