Submitted:
03 February 2026
Posted:
03 February 2026
You are already at the latest version
Abstract
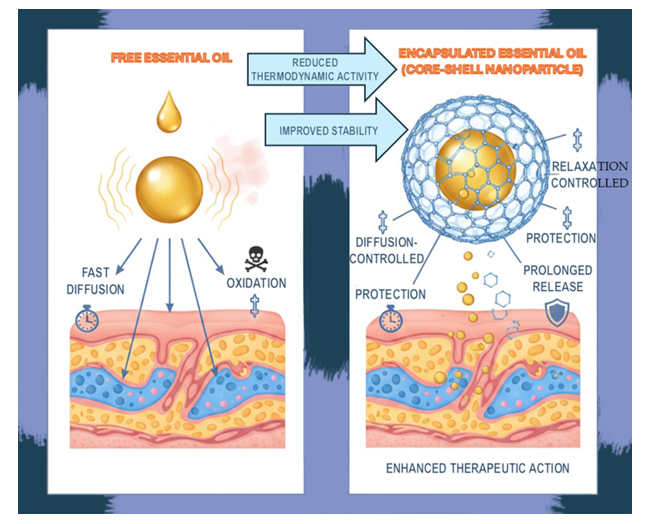
Keywords:
1. Introduction
2. Essential Oils for Acne and Sebum Regulation
2.1. Antimicrobial Properties Against Acne-Causing Bacteria
2.1.1. Cutibacterium acnes and Staphylococcus epidermidis
2.1.2. Evidence of Antibacterial, Anti-Inflammatory, and Antioxidant Effects
2.2. Sebum-Modulating and Anti-Inflammatory Effects
2.3. Challenges of Direct Essential Oils Application
3. Core–Shell Nanoformulations: Concepts and Advantages
3.1. Definition and Structural Features
3.2. Eco-Friendly and Biodegradable Components
3.2.1. Natural Polymers
3.2.2. Green Synthesis Approaches
3.3. Benefits for Essential Oil Delivery
3.3.1. Enhanced Stability and Sustained Release
3.3.2. Improved Skin Penetration and Reduced Irritation
4. Types of Eco-Friendly Core–Shell Nanoformulations
4.1. Polymeric Nanocapsules
4.2. Lipid-Based Nanocarriers
4.3. Polysaccharide-Based Nanocarriers
4.4. Hybrid and Bioinspired Nanostructures
5. Recent Advances and Applications in Acne Management
5.1. In Vitro Studies
5.1.1. Antimicrobial Activity
5.1.2. Effects on Sebum-Producing Cells
5.2. In Vivo and Clinical Findings
5.2.1. Skin Compatibility.
5.2.2. Clinical Improvement in Acne Symptoms
5.3. Comparative Analysis of Essential Oils Nanoformulations and Conventional Treatments
6. Safety, Toxicity, and Regulatory Considerations
6.1. Biocompatibility of Eco-Friendly Shell Materials
6.2. Irritation Potential Reductions via Encapsulation
6.3. Regulatory Hurdles for Natural Nano-Dermatological Products
7. Sustainability and Environmental Impact
7.1. Biodegradability of Materials
7.2. Green Chemistry in Synthesis
7.3. Reduced Reliance on Synthetic Antimicrobials
8. Challenges and Future Perspectives
8.1. Scaling Up Green Synthesis Techniques
8.2. Standardizing Essential Oils Chemical Profiles
8.3. Integration with Smart-Release and Stimuli-Responsive Technologies
8.4. Potential for Personalized Dermatological Care
9. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AgNPs | Silver NPs |
| AuNPs | Gold NPs |
| DLS | Dynamic light scattering |
| DSC | Differential scanning calorimetry |
| FTIR | Fourier transform infrared spectroscopy |
| GC-MS | Gas Chromatography-Mass Spectrometry |
| HNSs | Hollow nanospheres |
| MBC | Minimum bactericidal concentration |
| MIC | Minimum inhibitory concentration |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| NLC | Nanostructured lipid carriers |
| NPs | NPs |
| PCL | Poly(ε-caprolactone) |
| PPAR | Peroxisome proliferator-activated receptor |
| SEM | Scanning electron microscopy |
| SLN | Solid lipid NPs |
| TEM | Transmission electron microscopy |
| TGA | Thermogravimetric Analysis |
| UV-Vis | Ultra-visible spectroscopy |
| XRD | X-ray diffraction |
References
- Pappas, A.; Johnsen, S.; Liu, J.C.; Eisinger, M. Sebum analysis of individuals with and without acne. Dermato-endocrinology 2009, 1, 157–161. [Google Scholar] [CrossRef]
- Zouboulis, C.C.; Eady, A.; Philpott, M.; Goldsmith, L.A.; Orfanos, C.; Cunliffe, W.C.; Rosenfield, R. What is the pathogenesis of acne? Experimental Dermatology 2005, 14, 143–143. [Google Scholar] [CrossRef]
- Yaqoubi, W.; Touby, S.; Hossain, M.A. Laboratory investigations of liver function and lipid profiles tests before and after oral isotretinoin treatment among Acne valgaris clients at Ibri Polyclinic: A retrospective study. Toxicology reports 2024, 13, 101799. [Google Scholar] [CrossRef] [PubMed]
- Abdelhamed, A.; Ezz El-Dawla, R.; Karadag, A.S.; Agamia, N.F.; Melnik, B.C. The impact of isotretinoin on the pituitary-ovarian axis: An interpretative review of the literature. Reproductive toxicology 2021, 104, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Mangalagiri, N.P.; Panditi, S.K.; Jeevigunta, N.L.L. Antimicrobial activity of essential plant oils and their major components. Heliyon 2021, 7, e06835. [Google Scholar] [CrossRef] [PubMed]
- Szweda, P.; Kot, B. Bee Products and Essential Oils as Alternative Agents for Treatment of Infections Caused by S. aureus. 2017. [Google Scholar] [CrossRef]
- Ganosi, E.; Barda, C.; Grafakou, M.-E.; Rallis, M.C.; Skaltsa, H. An In-Depth Stability Study of the Essential Oils from Mentha × piperita, Mentha spicata, Origanum vulgare, and Thymus vulgaris: The Impact of Thermal and Storage Conditions. Separations 2023, 10, 488. [Google Scholar] [CrossRef]
- Fakhariha, M.; Rafati, A.A.; Garmakhany, A.D.; Asl, A.Z. Nanoencapsulation enhances stability, release behavior, and antimicrobial properties of Sage and Thyme essential oils. Scientific reports 2025, 15, 18373. [Google Scholar] [CrossRef]
- Silva-Flores, P.G.; Galindo-Rodriguez, S.A.; Perez-Lopez, L.A.; Alvarez-Roman, R. Development of Essential Oil-Loaded Polymeric Nanocapsules as Skin Delivery Systems: Biophysical Parameters and Dermatokinetics Ex Vivo Evaluation. Molecules 2023, 28. [Google Scholar] [CrossRef]
- Krzak, M.; Tabor, Z.; Nowak, P.; Warszyński, P.; Karatzas, A.; Kartsonakis, I.A.; Kordas, G.C. Water diffusion in polymer coatings containing water-trapping particles. Part 2. Experimental verification of the mathematical model. Progress in Organic Coatings 2012, 75, 207–214. [Google Scholar] [CrossRef]
- Hou, H.S.; Bonku, E.M.; Zhai, R.; Zeng, R.; Hou, Y.L.; Yang, Z.H.; Quan, C. Extraction of essential oil from Citrus reticulate Blanco peel and its antibacterial activity against Cutibacterium acnes (formerly Propionibacterium acnes). Heliyon 2019, 5, e02947. [Google Scholar] [CrossRef]
- Liao, S.; Yang, G.; Ou, Y.; Huang, S.; Li, B.; Li, A.; Kan, J. Inhibitory impacts of essential oil (Zanthoxylum schinifolium Sieb. et Zucc) on the growth of Staphylococcus epidermidis. Food Bioscience 2022, 49, 101906. [Google Scholar] [CrossRef]
- Pekacar, S.; Göçmen İlhan, I.E.; Kürkçüoğlu, M.; Özçelik, B.; Orhan, D.D. GC-MS based comparative phytochemical profiling with anti-acne and antidermatophytic activities of commercial and organic tea tree oils. Journal of Herbal Medicine 2025, 54, 101064. [Google Scholar] [CrossRef]
- Yang, J.; Lee, S.Y.; Jang, S.K.; Kim, K.J.; Park, M.J. Anti-Inflammatory Effects of Essential Oils from the Peels of Citrus Cultivars. Pharmaceutics 2023, 15. [Google Scholar] [CrossRef] [PubMed]
- Sidiropoulou, E.; Marugan-Hernandez, V.; Skoufos, I.; Giannenas, I.; Bonos, E.; Aguiar-Martins, K.; Lazari, D.; Papagrigoriou, T.; Fotou, K.; Grigoriadou, K.; et al. In Vitro Antioxidant, Antimicrobial, Anticoccidial, and Anti-Inflammatory Study of Essential Oils of Oregano, Thyme, and Sage from Epirus, Greece. Life 2022, 12. [Google Scholar] [CrossRef] [PubMed]
- Abdelhamed, F.M.; Abdeltawab, N.F.; ElRakaiby, M.T.; Shamma, R.N.; Moneib, N.A. Antibacterial and Anti-Inflammatory Activities of Thymus vulgaris Essential Oil Nanoemulsion on Acne Vulgaris. Microorganisms 2022, 10. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.X.; Xiao, W.L.; Zhang, X.J. Essential oil of Saposhnikovia divaricata mitigates Cutibacterium acnes-induced inflammatory acne via Nrf2 pathway activation and NF-kappaB pathway inhibition. Journal of ethnopharmacology 2026, 355, 120647. [Google Scholar] [CrossRef]
- Mohanta, O.; Das, P.K.; Panda, S.S.; Sahoo, A.; Jena, S.; Ray, A.; Nayak, S.; Panda, P.C. Anti-inflammatory activity of Leonotis nepetifolia leaf essential oil in LPS-stimulated RAW 264.7 cells and its molecular mechanism of action. Scientific reports 2025, 15, 32978. [Google Scholar] [CrossRef]
- Prashar, A.; Locke, I.C.; Evans, C.S. Cytotoxicity of lavender oil and its major components to human skin cells. Cell proliferation 2004, 37, 221–229. [Google Scholar] [CrossRef]
- Ren, G.; Ke, G.; Huang, R.; Pu, Q.; Zhao, J.; Zheng, Q.; Yang, M. Study of the volatilization rules of volatile oil and the sustained-release effect of volatile oil solidified by porous starch. Scientific reports 2022, 12, 8153. [Google Scholar] [CrossRef]
- Peng, X.; Ren, W.; Jia, M.; Zhou, Q.; Li, B.; Li, G.; Xie, Y.; Dai, X.; Cao, H.; Shi, X. Multi-scale study on the volatility of essential oil and its modulation methods. Journal of Drug Delivery Science and Technology 2025, 112, 107227. [Google Scholar] [CrossRef]
- Natrajan, D.; Srinivasan, S.; Sundar, K.; Ravindran, A. Formulation of essential oil-loaded chitosan-alginate nanocapsules. Journal of food and drug analysis 2015, 23, 560–568. [Google Scholar] [CrossRef]
- Homayoonfal, M.; Mousavi, S.M.; Kiani, H.; Askari, G.; Desobry, S.; Arab-Tehrany, E. Encapsulation of Berberis vulgaris Anthocyanins into Nanoliposome Composed of Rapeseed Lecithin: A Comprehensive Study on Physicochemical Characteristics and Biocompatibility. Foods 2021, 10. [Google Scholar] [CrossRef] [PubMed]
- Hasanin, M.S.; Ayoob, F.A.; Hashem, A.H.; Emam, M. Synthesis of Chitosan based nanoemulsions and their characterization and antifungal activity toward fungi causing mucormycosis. Scientific reports 2025, 15, 20326. [Google Scholar] [CrossRef]
- How, Y.-H.; Lim, E.M.-Y.; Kong, I.; Kee, P.-E.; Pui, L.-P. Development of carboxymethyl cellulose–chitosan based antibacterial films incorporating a Persicaria minor Huds. essential oil nanoemulsion. Sustainable Food Technology 2024, 2, 400–414. [Google Scholar] [CrossRef]
- Pitterou, I.; Kalogeropoulou, F.; Tzani, A.; Tsiantas, K.; Gatou, M.A.; Pavlatou, E.; Batrinou, A.; Fountzoula, C.; Kriebardis, A.; Zoumpoulakis, P.; et al. Development of Alginate Hydrogels Incorporating Essential Oils Loaded in Chitosan NPs for Biomedical Applications. Molecules 2024, 29. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Hernandez, E.; Santiago-Aliste, A.; Correa-Guimaraes, A.; Martin-Gil, J.; Gavara-Clemente, R.J.; Martin-Ramos, P. Carvacrol Encapsulation in Chitosan-Carboxymethylcellulose-Alginate Nanocarriers for Postharvest Tomato Protection. Int J Mol Sci 2024, 25. [Google Scholar] [CrossRef] [PubMed]
- Moosavy, M.H.; de la Guardia, M.; Mokhtarzadeh, A.; Khatibi, S.A.; Hosseinzadeh, N.; Hajipour, N. Green synthesis, characterization, and biological evaluation of gold and silver NPs using Mentha spicata essential oil. Scientific reports 2023, 13, 7230. [Google Scholar] [CrossRef]
- Vilas, V.; Philip, D.; Mathew, J. Essential oil mediated synthesis of silver nanocrystals for environmental, anti-microbial and antioxidant applications. Materials science & engineering. C, Materials for biological applications 2016, 61, 429–436. [Google Scholar] [CrossRef]
- Ahmadi, S.; Fazilati, M.; Nazem, H.; Mousavi, S.M. Green Synthesis of Magnetic NPs Using Satureja hortensis Essential Oil toward Superior Antibacterial/Fungal and Anticancer Performance. BioMed research international 2021, 2021, 8822645. [Google Scholar] [CrossRef]
- Zhang, M.; Li, M.; Zhang, D.; Yu, Y.; Zhu, K.; Zang, X.; Liu, D. Preparation and Investigation of Sustained-Release Nanocapsules Containing Cumin Essential Oil for Their Bacteriostatic Properties. Foods 2024, 13. [Google Scholar] [CrossRef]
- Yang, J.; Kok, C.R.; Ciftci, D.; Hutkins, R.; Ciftci, O.N. Hollow solid lipid particles loaded with essential oils via supercritical carbon dioxide to develop novel antimicrobial lipids with controlled release for food applications. Innovative Food Science & Emerging Technologies 2025, 102, 103980. [Google Scholar] [CrossRef]
- Manzoor, A.; Asif, M.; Khalid, S.H.; Ullah Khan, I.; Asghar, S. Nanosizing of Lavender, Basil, and Clove Essential Oils into Microemulsions for Enhanced Antioxidant Potential and Antibacterial and Antibiofilm Activities. ACS omega 2023, 8, 40600–40612. [Google Scholar] [CrossRef]
- Lavender essential oil nanoemulsion gel as skin lightener: green formulation, full characterization, anti-melanogenesis effect, and in-vitro/in-vivo safety profile assessment. [CrossRef]
- Drais, H.K. Development and Evaluation Essential Oils Nanoemulgel as Human Skin Sanitizer Using Novel Method. Turkish journal of pharmaceutical sciences 2024, 21, 456–462. [Google Scholar] [CrossRef]
- Aman, R.M.; Abu, H., II; Meshali, M.M. Novel Clove Essential Oil Nanoemulgel Tailored by Taguchi's Model and Scaffold-Based Nanofibers: Phytopharmaceuticals with Promising Potential as Cyclooxygenase-2 Inhibitors in External Inflammation. International journal of nanomedicine 2020, 15, 2171–2195. [Google Scholar] [CrossRef]
- Granata, G.; Stracquadanio, S.; Leonardi, M.; Napoli, E.; Consoli, G.M.L.; Cafiso, V.; Stefani, S.; Geraci, C. Essential oils encapsulated in polymer-based nanocapsules as potential candidates for application in food preservation. Food chemistry 2018, 269, 286–292. [Google Scholar] [CrossRef]
- Granata, G.; Riccobene, C.; Napoli, E.; Geraci, C. Polymeric Nanocapsules Containing Fennel Essential Oil: Their Preparation, Physicochemical Characterization, Stability over Time and in Simulated Gastrointestinal Conditions. Pharmaceutics 2022, 14. [Google Scholar] [CrossRef] [PubMed]
- Pinto, E.P.; Menezes, R.P.; de, S.T.W.; Ferreira, A.M.; Sousa, F.F.O.; Araujo da Silva, G.; Zamora, R.R.M.; Araujo, R.S.; de Souza, T.M. Copaiba essential oil loaded-nanocapsules film as a potential candidate for treating skin disorders: preparation, characterization, and antibacterial properties. International journal of pharmaceutics 2023, 633, 122608. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Reza, R.M.; Hernandez-Sanchez, H.; Zambrano-Zaragoza, M.L.; Gutierrez-Lopez, G.F.; Del Real, A.; Quintanar-Guerrero, D.; Velasco-Bejarano, B. Influence of Stabilizing and Encapsulating Polymers on Antioxidant Capacity, Stability, and Kinetic Release of Thyme Essential Oil Nanocapsules. Foods 2020, 9. [Google Scholar] [CrossRef]
- Negi, A.; Kesari, K.K. Chitosan Nanoparticle Encapsulation of Antibacterial Essential Oils. Micromachines 2022, 13. [Google Scholar] [CrossRef]
- Ahmad Nasrollahi, S.; Koohestani, F.; Naeimifar, A.; Samadi, A.; Vatanara, A.; Firooz, A. Preparation and evaluation of adapalene nanostructured lipid carriers for targeted drug delivery in acne. Dermatologic therapy 2021, 34, e14777. [Google Scholar] [CrossRef] [PubMed]
- Raza, K.; Singh, B.; Singal, P.; Wadhwa, S.; Katare, O.P. Systematically optimized biocompatible isotretinoin-loaded solid lipid NPs (SLNs) for topical treatment of acne. Colloids Surf B Biointerfaces 2013, 105, 67–74. [Google Scholar] [CrossRef] [PubMed]
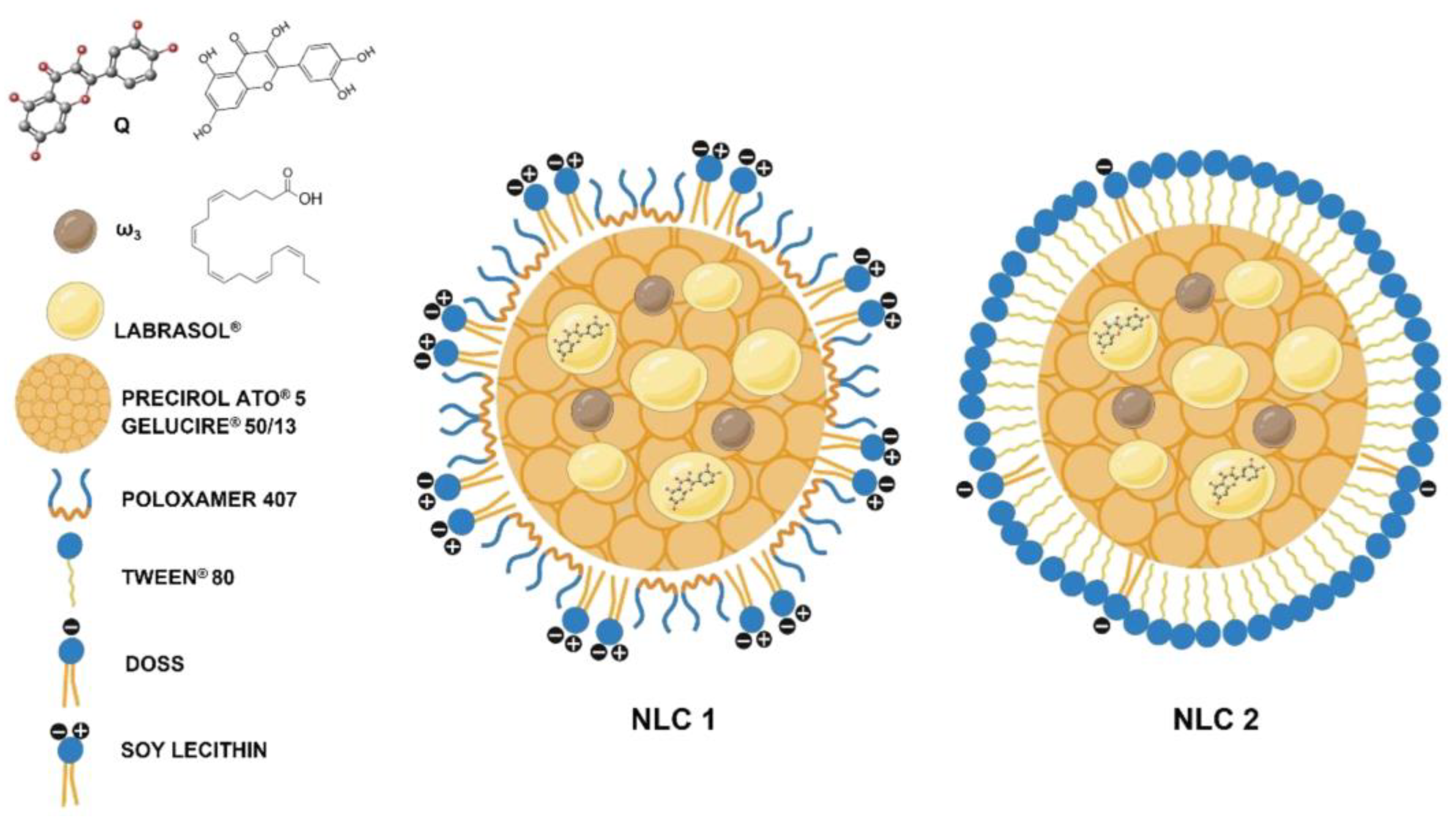
- Lucio, M.; Giannino, N.; Barreira, S.; Catita, J.; Goncalves, H.; Ribeiro, A.; Fernandes, E.; Carvalho, I.; Pinho, H.; Cerqueira, F.; et al. Nanostructured Lipid Carriers Enriched Hydrogels for Skin Topical Administration of Quercetin and Omega-3 Fatty Acid. Pharmaceutics 2023, 15. [Google Scholar] [CrossRef]
- Atapour-Mashhad, H.; Tayarani-Najaran, Z.; Golmohammadzadeh, S. Preparation and characterization of novel nanostructured lipid carriers (NLC) and solid lipid NPs (SLN) containing coenzyme Q10 as potent antioxidants and antityrosinase agents. Heliyon 2024, 10, e31429. [Google Scholar] [CrossRef]
- Nguyen, H.T.; Munnier, E.; Souce, M.; Perse, X.; David, S.; Bonnier, F.; Vial, F.; Yvergnaux, F.; Perrier, T.; Cohen-Jonathan, S.; et al. Novel alginate-based nanocarriers as a strategy to include high concentrations of hydrophobic compounds in hydrogels for topical application. Nanotechnology 2015, 26, 255101. [Google Scholar] [CrossRef]
- Friedman, A.J.; Phan, J.; Schairer, D.O.; Champer, J.; Qin, M.; Pirouz, A.; Blecher-Paz, K.; Oren, A.; Liu, P.T.; Modlin, R.L.; et al. Antimicrobial and anti-inflammatory activity of chitosan-alginate NPs: a targeted therapy for cutaneous pathogens. The Journal of investigative dermatology 2013, 133, 1231–1239. [Google Scholar] [CrossRef]
- Amante, C.; Andretto, V.; Rosso, A.; Augusti, G.; Marzocco, S.; Lollo, G.; Del Gaudio, P. Alginate-pectin microparticles loaded with nanoemulsions as nanocomposites for wound healing. Drug delivery and translational research 2023, 13, 1343–1357. [Google Scholar] [CrossRef]
- Nguyen, T.T.T.; Tran, N.T.K.; Le, T.Q.; Nguyen, T.T.A.; Nguyen, L.T.M.; Tran, T.V. Passion fruit peel pectin/chitosan based antibacterial films incorporated with biosynthesized silver NPs for wound healing application. Alexandria Engineering Journal 2023, 69, 419–430. [Google Scholar] [CrossRef]
- Saini, A.; Verma, R.; Tiwari, R.; Jain, A.; Dandia, A.; Gour, V.S.; Lamba, N.P.; Srivastava, S.C.; Chauhan, M.S. Green synthesis of silver nanoparticle for catalytic applications and priming study by seed germination. Scientific reports 2024, 14, 20744. [Google Scholar] [CrossRef] [PubMed]
- Gul, F.; Ullah, Z.; Iqbal, J.; Abbasi, B.A.; Ali, S.; Kanwal, S.; Uddin, J.; Kazi, M.; Mahmood, T. Ecofriendly synthesis characterization and biological activities of Eruca sativa mediated silver oxide NPs. Scientific reports 2025, 15, 13466. [Google Scholar] [CrossRef]
- Mohammadi, A.; Hashemi, N.; Ghassabzadeh, M.; Sharafi, A.; Yazdinezhad, A.; Danafar, H. Green synthesis and toxicological evaluation of zinc oxide NPs utilizing Punica granatum fruit Peel extract: an eco-friendly approach. Scientific reports 2025, 15, 20853. [Google Scholar] [CrossRef]
- Naiel, B.; Fawzy, M.; Halmy, M.W.A.; Mahmoud, A.E.D. Green synthesis of zinc oxide NPs using Sea Lavender (Limonium pruinosum L. Chaz.) extract: characterization, evaluation of anti-skin cancer, antimicrobial and antioxidant potentials. Scientific reports 2022, 12, 20370. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Jang, J.H.; Kim, H.R.; Cho, J.H. Novel-designed antimicrobial peptides with dual antimicrobial and anti-inflammatory actions against Cutibacterium acnes for acne vulgaris therapy. Biochemical pharmacology 2025, 232, 116708. [Google Scholar] [CrossRef]
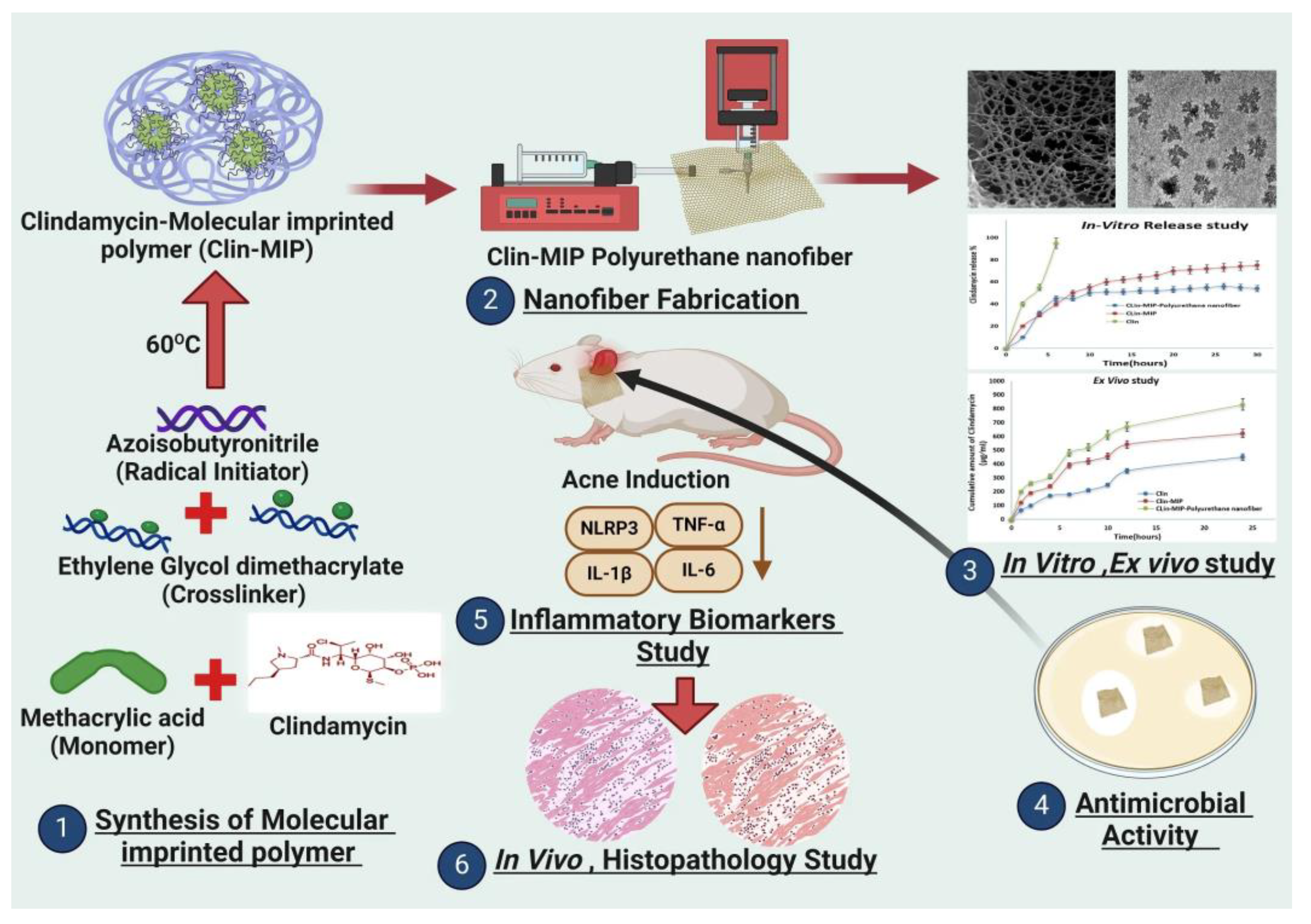
- Elhabal, S.F.; Abdelmonem, R.; El Nashar, R.M.; Elrefai, M.F.M.; Hamdan, A.M.E.; Safwat, N.A.; Shoela, M.S.; Hassan, F.E.; Rizk, A.; Kabil, S.L.; et al. Enhanced Antibacterial Activity of Clindamycin Using Molecularly Imprinted Polymer NPs Loaded with Polyurethane Nanofibrous Scaffolds for the Treatment of Acne Vulgaris. Pharmaceutics 2024, 16. [Google Scholar] [CrossRef] [PubMed]
- Dong, Q.; Wang, S.; Miao, Y.; Luo, H.; Weng, Z.; Yu, L. 2023. [CrossRef]
- Cao, W.Y.; Zhao, H.Q.; Gu, X.; Shao, W.J.; Zheng, L.J.; Zeng, Q.Y.; Wang, Z.K.; Chen, M.L.; Ma, T.; Li, Y.Y.; et al. Light-responsive antibacterial dissolving microneedles loaded with 5-aminolevulinic acid and silver NPs for the treatment of acne. International journal of pharmaceutics 2024, 667, 124961. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Luo, X.H.; Liu, Y.F.; Zouboulis, C.C.; Shi, G. Emodin exhibits anti-acne potential by inhibiting cell growth, lipogenesis, and inflammation in human SZ95 sebocytes. Scientific reports 2023, 13, 21576. [Google Scholar] [CrossRef]
- Kitano, T.; Koiwai, T.; Fujikawa, K.; Mori, S.; Matsumoto, T.; Sato, T. Ozenoxacin suppresses sebum production by inhibiting mTORC1 activation in differentiated hamster sebocytes. The Journal of dermatology 2024, 51, 1187–1198. [Google Scholar] [CrossRef]
- Zhang, J.; He, L.; Chen, X.; Tu, Y.; Kostov, B.; Cabedo, J.; Baykal, T.; Garcia Gil, E. Efficacy and Safety of Sarecycline in Chinese Patients with Moderate-to-Severe Acne Vulgaris: Randomized Phase 3 Clinical Trial with Open-Label Follow-Up. Dermatology and therapy 2025, 15, 3285–3300. [Google Scholar] [CrossRef]
- Le, Y.; Yang, W.; Lu, J.; Jiang, X.; Guo, Q.; Huang, C.; Sun, Q.; Wang, H.; Bi, M.; Wang, H.; et al. Efficacy and safety of topical minocycline foam (FMX101 4%) in treatment of Chinese subjects with moderate-to-severe facial acne vulgaris: A phase 3, multi-centre, randomized, double-blind, vehicle-controlled study. Journal of the European Academy of Dermatology and Venereology: JEADV 2025, 39, 967–975. [Google Scholar] [CrossRef]
- Saki, N.; Mohammadi, F.; Parvizi, M.M.; Kamali, M.; Saleem, S. Efficacy of Intramuscular Pantothenic Acid in the Treatment of Acne Vulgaris: A Single Blind Randomized Clinical Trial. Dermatologic therapy 2025, 2025. [Google Scholar] [CrossRef]
- Rimon, A.; Rakov, C.; Lerer, V.; Sheffer-Levi, S.; Oren, S.A.; Shlomov, T.; Shasha, L.; Lubin, R.; Zubeidat, K.; Jaber, N.; et al. Topical phage therapy in a mouse model of Cutibacterium acnes-induced acne-like lesions. Nature communications 2023, 14, 1005. [Google Scholar] [CrossRef]
- Uddin, M.K.; Yesmin, S. Evaluation of the effectiveness of the combination of doxycycline and trifarotene for managing acne vulgaris of moderate to severe severity: a study in a tertiary care hospital. International Journal of Research in Dermatology 2024, 10, 66–70. [Google Scholar] [CrossRef]
- Hebert, A.; Thiboutot, D.; Stein Gold, L.; Cartwright, M.; Gerloni, M.; Fragasso, E.; Mazzetti, A. Efficacy and Safety of Topical Clascoterone Cream, 1%, for Treatment in Patients With Facial Acne: Two Phase 3 Randomized Clinical Trials. JAMA dermatology 2020, 156, 621–630. [Google Scholar] [CrossRef]
- Taleb, M.H.; Abdeltawab, N.F.; Shamma, R.N.; Abdelgayed, S.S.; Mohamed, S.S.; Farag, M.A.; Ramadan, M.A. Origanum vulgare L. Essential Oil as a Potential Anti-Acne Topical Nanoemulsion-In Vitro and In Vivo Study. Molecules 2018, 23. [Google Scholar] [CrossRef]
- Sasan, S.; Molavi, A.M.; Moqadam, K.H.; Farrokhi, N.; Oroojalian, F. Enhanced wound healing properties of biodegradable PCL/alginate core-shell nanofibers containing Salvia abrotanoides essential oil and ZnO NPs. International journal of biological macromolecules 2024, 279, 135152. [Google Scholar] [CrossRef] [PubMed]
- Shamszadeh, S.; Akrami, M.; Asgary, S. Size-dependent bioactivity of electrosprayed core-shell chitosan-alginate particles for protein delivery. Scientific reports 2022, 12, 20097. [Google Scholar] [CrossRef]
- Flekka, K.; Dimaki, V.D.; Mourelatou, E.; Avgoustakis, K.; Lamari, F.N.; Hatziantoniou, S. Stability and Retention of Nanoemulsion Formulations Incorporating Lavender Essential Oil. Cosmetics 2024, 11, 65. [Google Scholar] [CrossRef]
- Fytianos, G.; Rahdar, A.; Kyzas, G.Z. Nanomaterials in Cosmetics: Recent Updates. Nanomaterials (Basel) 2020, 10. [Google Scholar] [CrossRef]
- Karamanidou, T.; Bourganis, V.; Gatzogianni, G.; Tsouknidas, A. A Review of the EU’s Regulatory Framework for the Production of Nano-Enhanced Cosmetics. Metals 2021, 11, 455. [Google Scholar] [CrossRef]
- Oliveira, C.; Coelho, C.; Teixeira, J.A.; Ferreira-Santos, P.; Botelho, C.M. Nanocarriers as Active Ingredients Enhancers in the Cosmetic Industry-The European and North America Regulation Challenges. Molecules 2022, 27. [Google Scholar] [CrossRef]
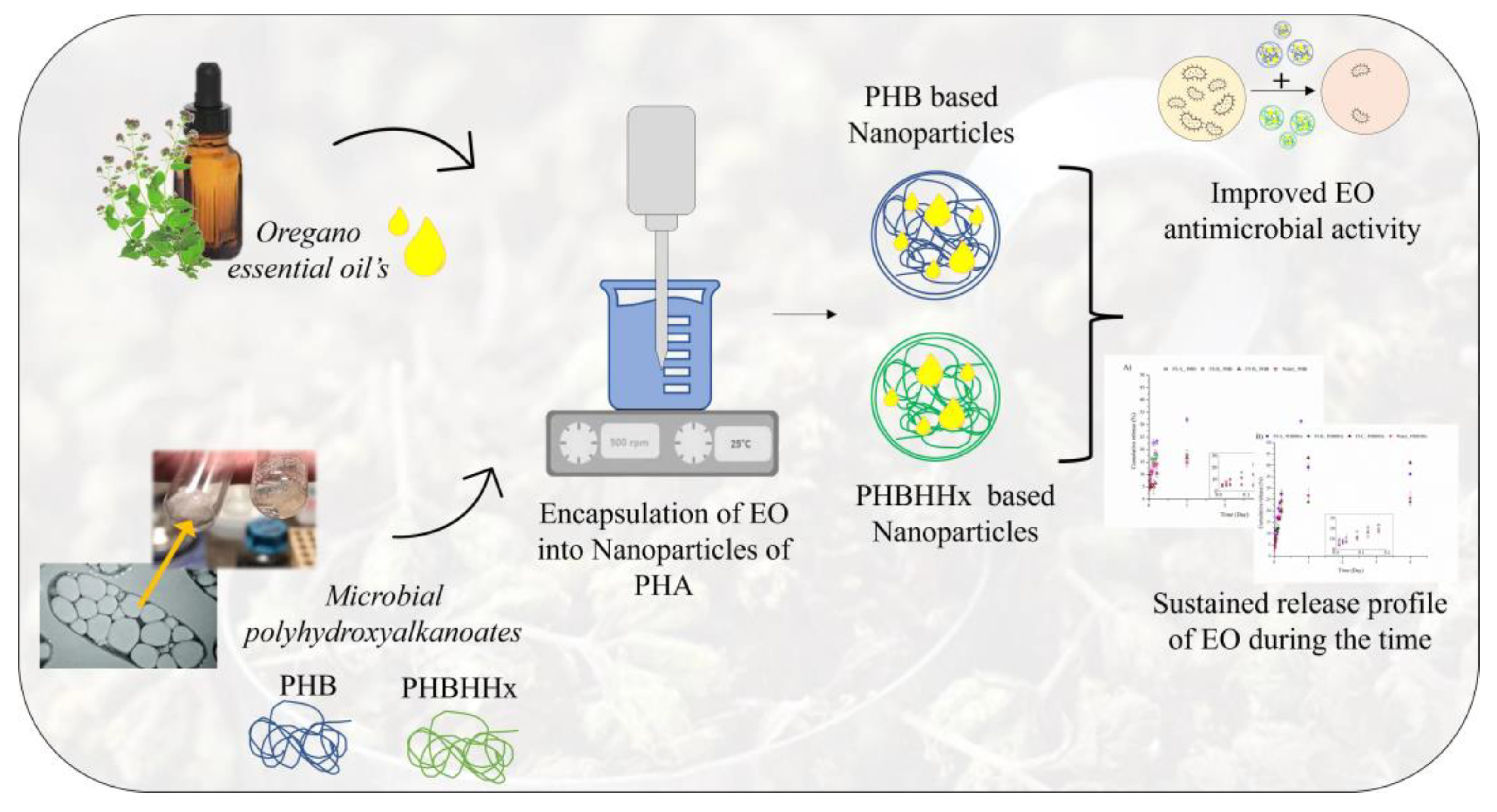
- Corrado, I.; Di Girolamo, R.; Regalado-Gonzalez, C.; Pezzella, C. Polyhydroxyalkanoates-Based NPs as Essential Oil Carriers. Polymers (Basel) 2022, 14. [Google Scholar] [CrossRef] [PubMed]
- Gheorghita Puscaselu, R.; Lobiuc, A.; Sirbu, I.O.; Covasa, M. The Use of Biopolymers as a Natural Matrix for Incorporation of Essential Oils of Medicinal Plants. Gels 2022, 8. [Google Scholar] [CrossRef]
- Golzarnezhad, F.; Allahdou, M.; Mehravaran, L.; Naderi, S. Green synthesis of ZnO NPs from the extract of Cymbopogon olivieri and investigation of their antimicrobial and anticancer effects. Discover Applied Sciences 2025, 7. [Google Scholar] [CrossRef]
- Al-Ogaidi, I.; Aguilar, Z.P.; Lay, J.O., Jr. Development of Biodegradable/Biocompatible Nanoliposome-Encapsulated Antimicrobial Essential Oils for Topical Creams and Gels. ACS omega 2022, 7, 23875–23889. [Google Scholar] [CrossRef]
- Gacem, M.A.; Abd-Elsalam, K.A. Strategies for scaling up of green-synthesized nanomaterials: Challenges and future trends 2022, 669–698. [CrossRef]
- Gupta, D.; Boora, A.; Thakur, A.; Gupta, T.K. Green and sustainable synthesis of nanomaterials: Recent advancements and limitations. Environmental research 2023, 231, 116316. [Google Scholar] [CrossRef] [PubMed]
- Ayub, A.; Wani, A.K.; Malik, S.M.; Ayub, M.; Singh, R.; Chopra, C.; Malik, T. Green nanoscience for healthcare: Advancing biomedical innovation through eco-synthesized nanoparticle. Biotechnology reports 2025, 47, e00913. [Google Scholar] [CrossRef]
- Bernal, F.A.; Matulevich, J.A.; Corredor, J.A.; Coy-Barrera, E. GC/MS-Based Fingerprinting Reveals Two Chemotypes in the Leaf Essential Oils from Magnolia grandiflora Trees within The Urban Forestry of a Colombian Andean Plateau. Chemistry & biodiversity 2022, 19, e202200448. [Google Scholar] [CrossRef]
- Ratnasekhar, C.H.; Khan, S.; Rai, A.K.; Mishra, H.; Verma, A.K.; Lal, R.K.; Ananda Kumar, T.M.; Elliott, C.T. Rapid metabolic fingerprinting meets machine learning models to identify authenticity and detect adulteration of essential oils with vegetable oils: Mentha and Ocimum study. Food chemistry 2025, 471, 142709. [Google Scholar] [CrossRef] [PubMed]
- Deng, S.; Gigliobianco, M.R.; Mijit, E.; Minicucci, M.; Cortese, M.; Campisi, B.; Voinovich, D.; Battistelli, M.; Salucci, S.; Gobbi, P.; et al. Dually Cross-Linked Core-Shell Structure Nanohydrogel with Redox-Responsive Degradability for Intracellular Delivery. Pharmaceutics 2021, 13. [Google Scholar] [CrossRef] [PubMed]
- Ertugral-Samgar, E.G.; Ozmen, A.M.; Gok, O. Thermo-Responsive Hydrogels Encapsulating Targeted Core-Shell NPs as Injectable Drug Delivery Systems. Pharmaceutics 2023, 15. [Google Scholar] [CrossRef]
- Tan, I.J.; Podwojniak, A.; Parikh, A.; Cohen, B.A. Precision Dermatology: A Review of Molecular Biomarkers and Personalized Therapies. Current issues in molecular biology 2024, 46, 2975–2990. [Google Scholar] [CrossRef]
- Kim, J. Smart Nanocarriers in Cosmeceuticals Through Advanced Delivery Systems. Biomimetics 2025, 10, 217. [Google Scholar] [CrossRef] [PubMed]

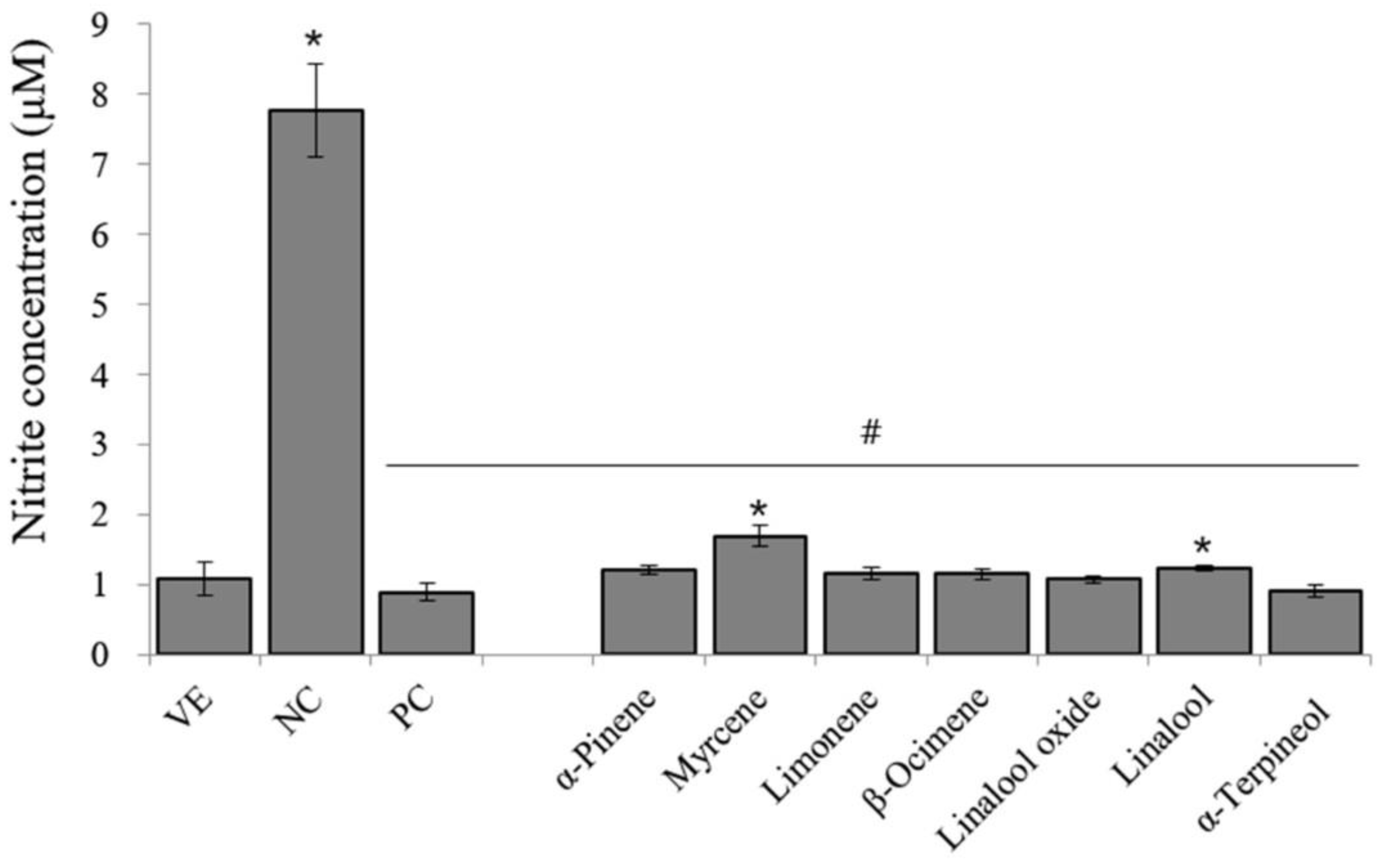
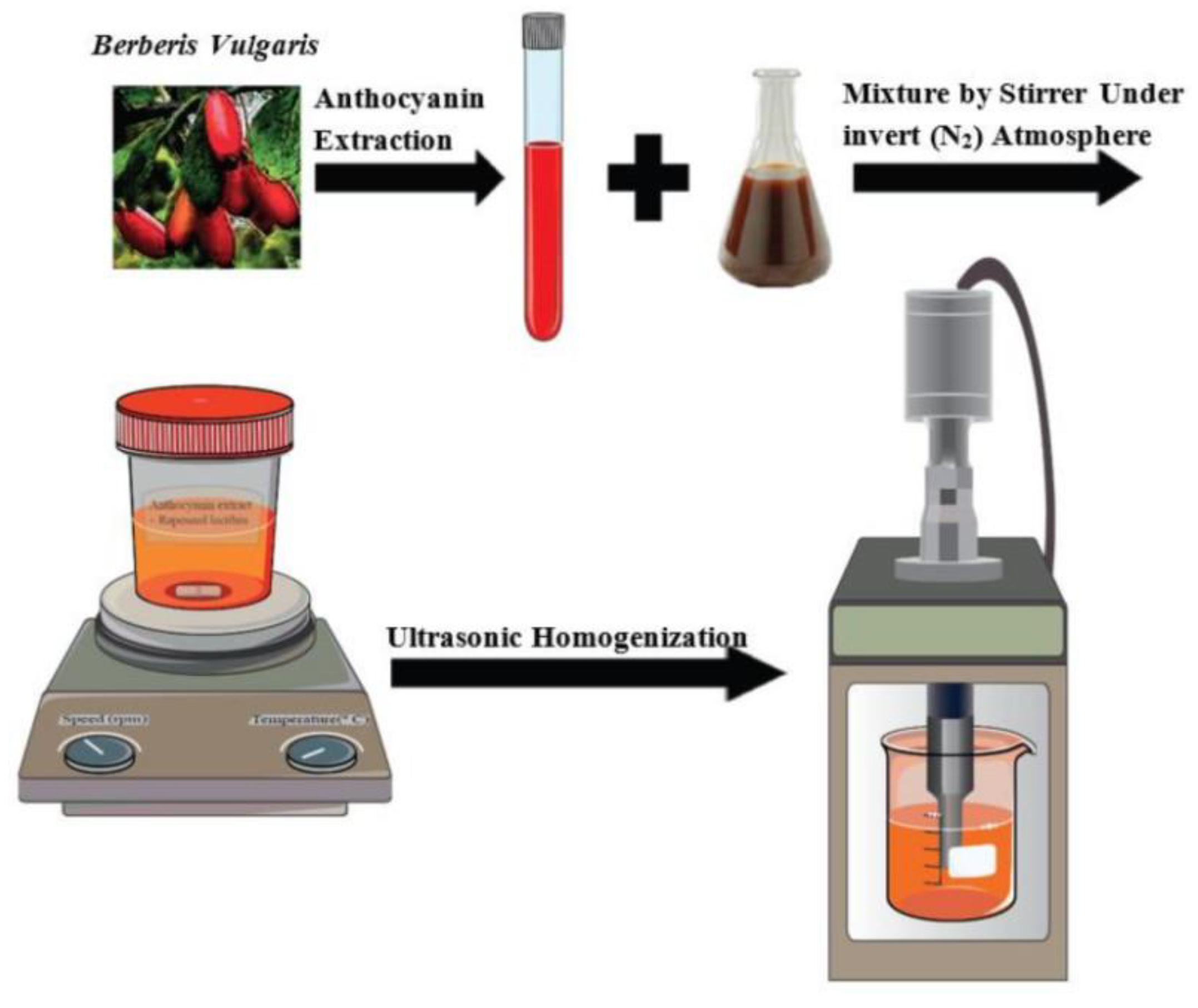

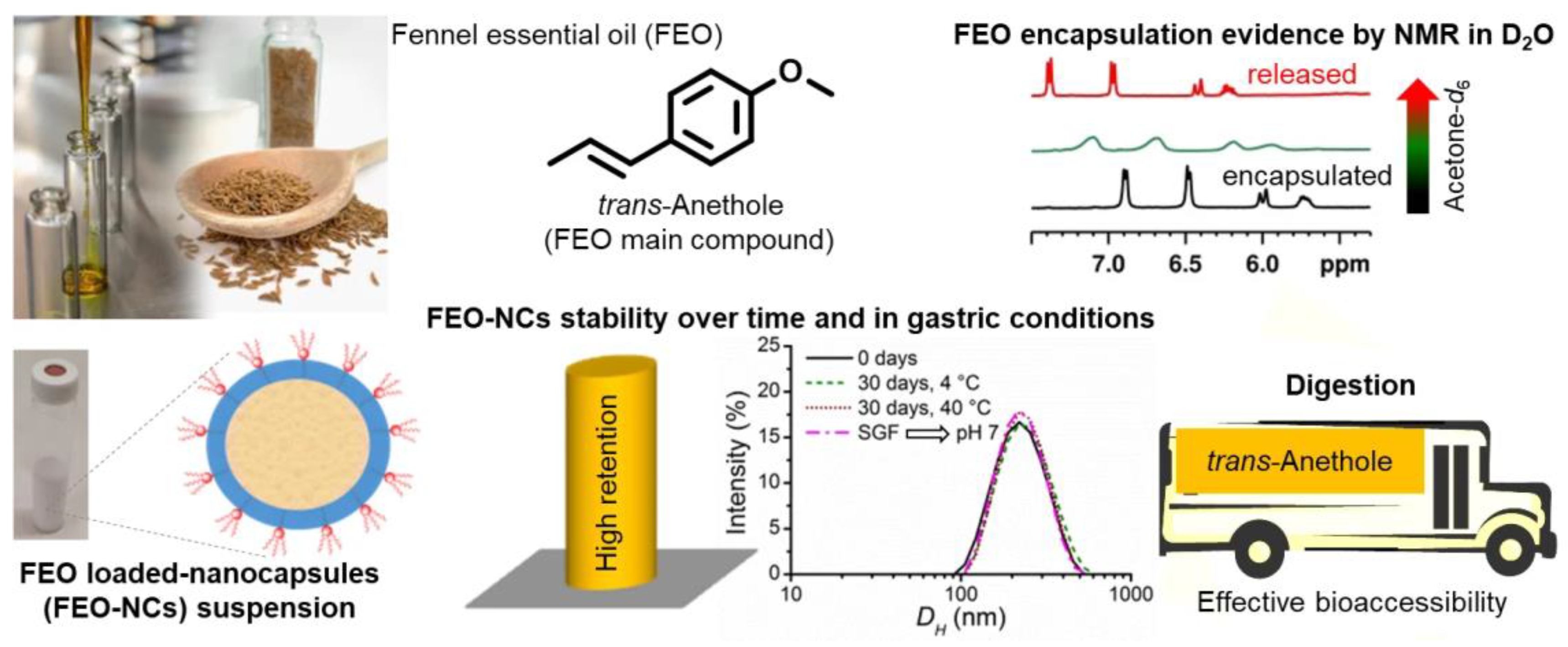

| Time Point | Pretreatment | 100 μg mL−1 | 50 μg mL−1 | 20 μg mL−1 | 5 μg mL−1 |
|---|---|---|---|---|---|
| 2 hpi | OEO | 82.8 ± 6.95 | 15.5 ± 37.1 | 0 ± 0 | 0 ± 0 |
| TEO | 81.3 ± 14.1 | 62.8 ± 24.6 | 62.9 ± 20.3 | 24.1 ± 57.1 | |
| SEO | 72.2 ± 18.4 * | 33.0 ± 44.8 | 11.6 ± 35.0 | 5.25 ± 14.6 | |
| 24 hpi | OEO | 92.9 ± 6.9 | 81.5 ± 25.6 | 38.1 ± 13.1 | 33.3 ± 66.7 |
| TEO | 90.8 ± 17.9 | 73.1 ± 1.6 | 49.6 ± 54.9 | 67.4 ± 18.5 | |
| SEO | 89.6 ± 9.4 | 32.5 ± 65.1 | 31.6 ± 63.3 | 38.5 ± 42.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




