Core Tip: This study from western Kenya confirms diabetes as a key risk factor for severe COVID-19. It provides novel evidence suggesting that acute metabolic decompensation may be an important intermediary pathway to severe disease. This highlights a direct clinical target, advocating for prioritized metabolic management in diabetic patients with COVID-19 within resource-constrained environments.
Introduction
The COVID-19 pandemic, precipitated by the SARS-CoV-2 virus, has unveiled critical syndemic interactions with non-communicable diseases (NCDs), significantly shaping global mortality patterns [
1]. Diabetes mellitus (DM) has been consistently identified as a primary comorbidity, potentiating the risk of severe respiratory and systemic complications [
2,
3]. International cohorts and recent meta-analyses have demonstrated that individuals with diabetes experience disproportionately higher rates of intensive care admission and death [
4,
5,
6,
7]. However, the COVID-19 pandemic has also placed immense strain on healthcare systems globally, with Sub-Saharan African (SSA) health systems facing unique vulnerabilities, including workforce shortages, limited infrastructure, and a high preexisting burden of communicable and non-communicable diseases [
8]. This strain exacerbates the syndemic interaction between COVID-19 and diabetes, where biological and socio-contextual factors converge to worsen outcomes.
The pathophysiological link between DM and COVID-19 severity is multifactorial, involving chronic inflammation, immune dysfunction, endothelial damage, and a dysregulated cytokine response [
9,
10]. Chronic hyperglycemia leads to a state of low-grade inflammation and an impaired immune response, making the host more susceptible to severe viral illness [
11]. Conversely, SARS-CoV-2 infection can directly exacerbate metabolic dysregulation through cytokine-driven insulin resistance and potential viral injury to pancreatic β-cells [
12]. Much of this evidence, however, originates from high-income settings with older populations, advanced critical care capacity, and different burdens of comorbid disease. The relative contribution of these chronic inflammatory pathways versus acute, infection-induced metabolic crises in driving severe outcomes remains unclear, particularly in regions with younger populations, constrained healthcare resources, and potentially differing background glycemic control.
Sub-Saharan Africa faces an increasing double burden of communicable and non-communicable diseases [
13]. Kenya has an estimated diabetes prevalence of 7.1%, yet the local interaction between DM and COVID-19 is critically understudied [
14]. Within the complex landscape of a strained health system, understanding the dominant, modifiable pathways to severe disease is critical for optimizing scarce clinical resources [
15,
16]. Distinct contexts—including genetic background, age structure, prevalence of other infections, and healthcare system resilience—may modulate the diabetes–COVID-19 interaction in ways not captured by global models [
17]. In settings where ICU beds and ventilators are scarce, identifying the most immediate and treatable pathways to deterioration is a clinical necessity.
Therefore, this study was designed to achieve two primary objectives within a tertiary care setting in western Kenya: first, to quantify the independent association between diabetes and COVID-19 severity and mortality; and second, to investigate the hypothesis that acute metabolic decompensation serves as a key mediating mechanism for severe disease in this population. By doing so, we aim to provide evidence to inform more precise clinical management and health system preparedness.
Materials and Methods
Study Design and Setting
We conducted a retrospective cohort study at the COVID-19 isolation centre of Jaramogi Oginga Odinga Teaching and Referral Hospital (JOOTRH), the primary tertiary care and referral facility for Kisumu County and the wider western Kenya region. The study period (May 2020 to May 2021) encompassed Kenya’s initial pandemic waves prior to the widespread availability of SARS-CoV-2 vaccines.
Study Population and Data Sources
The study population included all patients aged ≥ 18 years admitted with RT-PCR-confirmed SARS-CoV-2 infection. Patient records were identified from hospital medical records and isolation unit admission registries.
Sample Size and Participant Selection
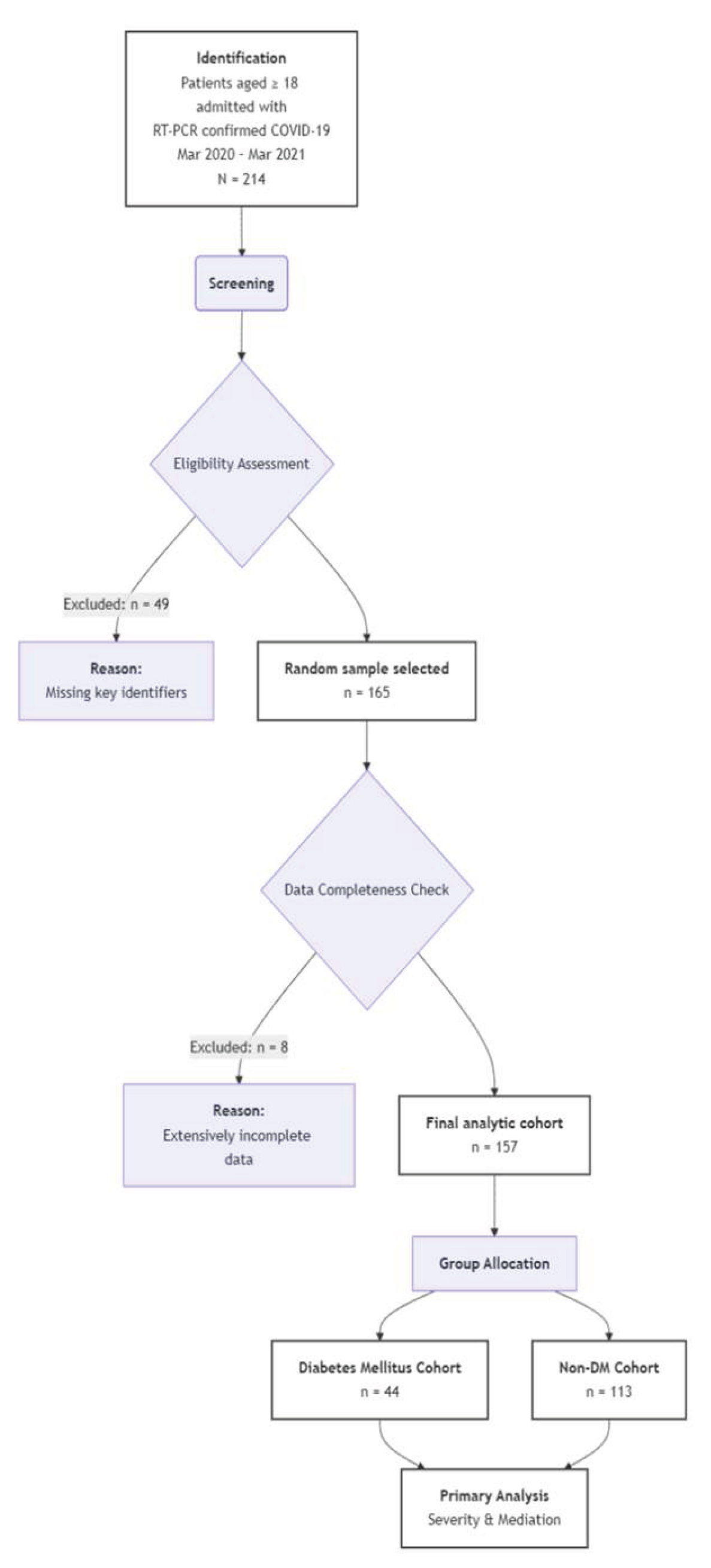
A minimum sample size of 150 was calculated a priori using G*Power 3.1.9.7 software, assuming 80% power, a two-sided alpha of 0.05, a proportion of severe disease in non-diabetics of 20%, and an odds ratio of 2.5 for patients with DM. We aimed for complete enumeration of all 214 eligible admission records identified during the study period. After rigorous screening, 57 records were excluded due to incomplete core data (missing exposure status, primary outcome, or essential covariates like age). The final analytic cohort comprised 157 participants.
Data Collection and Variables
Data was extracted by two trained clinical officers using a standardized, pre-piloted electronic data-collection tool (KoboToolbox). Inter-rater reliability was assessed on a randomly selected 10% sample; any discrepancies were resolved through review and consensus by a third senior investigator (F.P.O.).
Exposure variable: DM status was defined by a documented preexisting diagnosis, history of anti-diabetic medication use, or patient self-report of a prior physician diagnosis. To ensure the stability of the exposure group and avoid confounding by transient stress-induced hyperglycemia, only patients with a confirmed history of diabetes were included in the DM cohort.
Primary outcome: COVID-19 disease severity at the point of worst clinical status during admission, categorized according to the Kenyan Ministry of Health COVID-19 Clinical Management Guidelines (3rd Edition, July 2020):
1. Mild/Moderate: Asymptomatic or symptomatic (e.g., fever, cough) without evidence of severe pneumonia, and peripheral oxygen saturation (SpO2) ≥94% on room air.
2. Severe/Critical: Clinical signs of severe pneumonia (respiratory rate >30 breaths/min, severe respiratory distress) or SpO2 <94% on room air or requirement for supplemental oxygen to maintain SpO2 ≥94% or critical illness (acute respiratory distress syndrome (ARDS), sepsis, septic shock, or requiring intensive care for mechanical ventilation or vasopressor support).
Secondary outcomes included in-hospital mortality (all-cause), development of any major COVID-19 complication (e.g., ARDS, acute kidney injury, thromboembolism), and the occurrence of an acute diabetic complication.
Potential mediator: Acute metabolic decompensation was defined as a physician-documented diagnosis of DKA or HHS during the admission. Hypoglycemic crises were excluded from the mediation model to focus specifically on the hypermetabolic pathway to deterioration.
Covariates: Extracted data included age, sex, and the presence of key comorbidities (hypertension, cardiovascular disease, chronic kidney disease (CKD), chronic lung disease, HIV, cancer). Data on HIV status were available, but prevalence was low (n=8, 5.1%) and did not differ significantly between groups (P=0.99); it was therefore not included as a covariate in the primary model. Data on body mass index (BMI) and glycated hemoglobin (HbA1c) were not consistently available in the clinical records and are acknowledged as a key limitation.
Statistical Analysis
Data were analysed using Stata/MP 18.0 (StataCorp LP, College Station, TX, USA). Descriptive statistics are presented as medians with interquartile ranges (IQR) for non-normally distributed continuous variables and as counts with percentages for categorical variables. Group comparisons utilized the Mann-Whitney U test, Chi-square test, or Fisher’s exact test, as appropriate.
Multivariable logistic regression models were constructed to determine the independent association between DM and (a) severe/critical COVID-19 and (b) in-hospital mortality. Covariate selection followed a two-part strategy to balance clinical relevance with model stability, given the sample size. Age and sex were included a priori. Other comorbidities were considered for inclusion if they demonstrated an association with the primary outcome at P < 0.1 in bivariate analysis; hypertension and cardiovascular disease met this criterion. Although CKD and chronic lung disease were more prevalent in the DM group, they did not meet the pre-specified threshold for the primary severity model. A sensitivity analysis was conducted to adjust for these additional comorbidities.
To investigate the secondary aim—the potential mediating role of acute metabolic decompensation—we employed a formal causal mediation analysis framework using the paramed command in Stata. This analysis estimates the natural direct effect (the effect of DM not operating through the mediator) and the natural indirect effect (the effect of DM operating through the mediator) of the total effect on the outcome. The analysis adjusted for the same confounders (age, sex, hypertension, cardiovascular disease) and provided an estimate of the proportion mediated with 95% confidence intervals derived from 1000 non-parametric bootstrap replications. The interpretation of these results is predicated on the assumption that there is no unmeasured confounding of the exposure-mediator and mediator-outcome relationships.
To assess the robustness of the primary exposure-outcome association to potential unmeasured confounding, we calculated the E-value. The E-value quantifies the minimum strength of association that an unmeasured confounder would need to have with both the exposure and the outcome to explain away the observed effect fully.
A two-sided P-value <0.05 was considered statistically significant. A post hoc power calculation confirmed that the mortality analysis was underpowered.
Ethical Considerations
The Maseno University Scientific and Ethics Review Committee (MUSERC) granted ethical approval for this study (Ref: MUSERC/01109/21). A waiver of individual informed consent was authorised for this retrospective analysis of de-identified patient data. All research procedures were performed in accordance with the ethical standards of the Declaration of Helsinki
Patient and Public Involvement
Due to the retrospective and urgent nature of this research conducted during a pandemic surge, patients or the public were not involved in the design, conduct, reporting, or dissemination plans of this study.
Results
Characteristics of the Study Cohort
Figure 1 presents the participant flow diagram. The final analytic cohort of 157 patients had a median age of 57 years (IQR: 45, 68) and was predominantly male (89, 56.7%). Forty-four patients (28.0%) were classified as having DM. Patients with DM were significantly older and had a higher prevalence of cardiovascular disease, chronic kidney disease (P=0.049), and chronic lung disease (P=0.021) (
Table 1)
Association Between DM and COVID-19 Severity and Outcomes
The primary analysis focused on disease severity. In unadjusted analysis, a significantly higher proportion of patients with DM presented with or progressed to severe/critical COVID-19 (50.0% vs. 23.0%, P < 0.001). After adjustment for age, sex, hypertension, and cardiovascular disease, DM remained a strong, independent predictor of severe/critical disease (adjusted Odds Ratio [aOR]: 3.15, 95% CI: 1.52-6.54, P=0.002). In a sensitivity analysis that additionally adjusted for CKD and chronic lung disease, the association persisted (aOR: 2.97, 95% CI: 1.40-6.31, P=0.005). The E-value for the primary aOR of 3.15 was 5.45, suggesting considerable robustness to potential unmeasured confounding.
Regarding mortality, the unadjusted rate was higher in the DM group (22.7% vs 15.9%), but the adjusted association was not statistically significant (aOR: 1.48). Post-hoc power analysis confirmed the study was substantially underpowered (31.5%) to detect an effect size for mortality; therefore, this finding is strictly exploratory and should not be interpreted as a lack of association.
Discussion
This hospital-based cohort study from western Kenya provides robust evidence that DM is a powerful, independent predictor of severe or critical COVID-19, conferring a three-fold increase in adjusted odds of severe disease. This central finding aligns with and reinforces the global evidence base linking DM to adverse COVID-19 outcomes across diverse geographical settings [
4,
5,
6,
7,
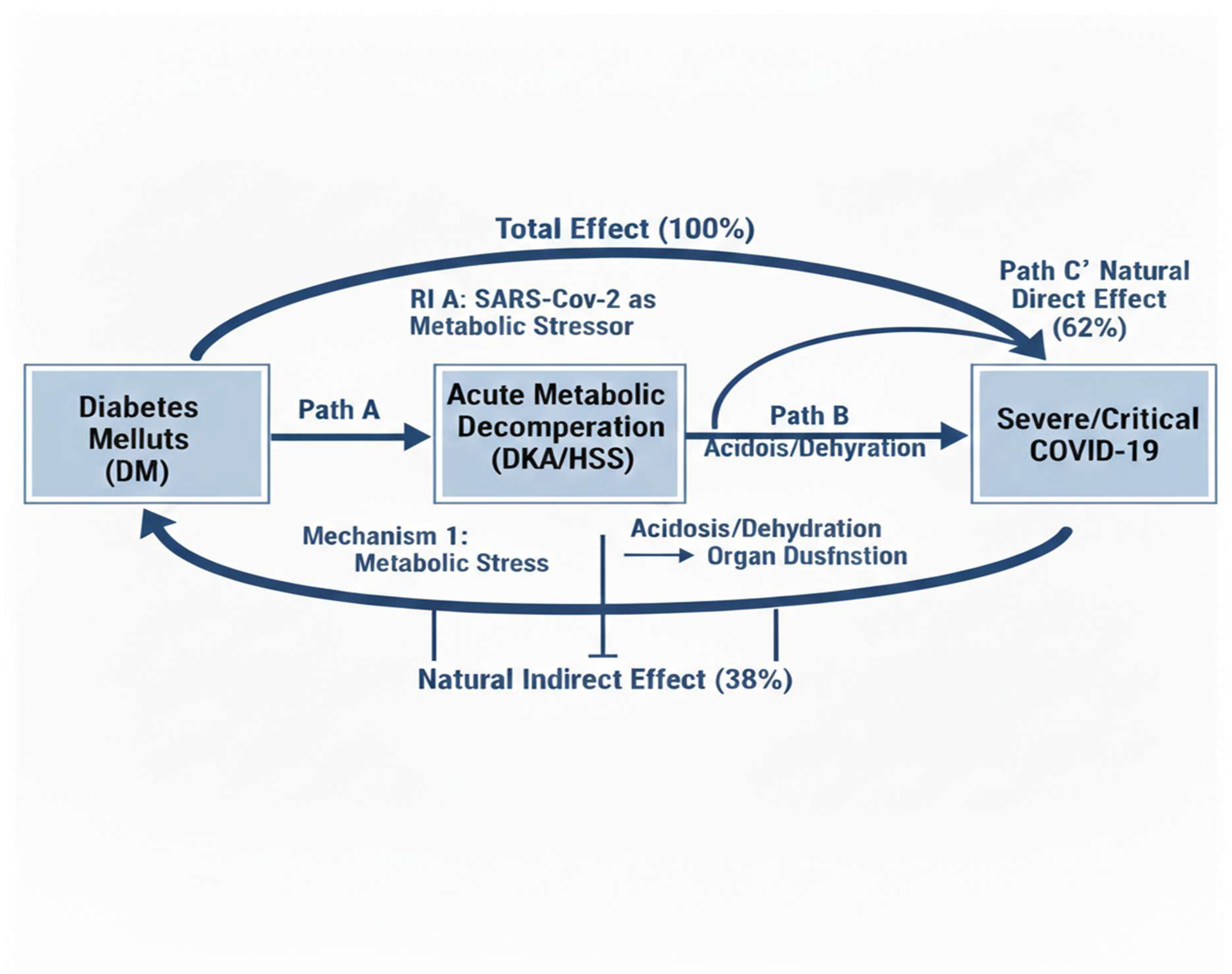
18]. However, our study extends this understanding in a manner particularly pertinent to resource-limited healthcare systems in SSA. Our exploratory mediation analysis yielded novel evidence statistically consistent with a substantial mediating role for acute metabolic decompensation—specifically DKA and HHS—accounting for an estimated 38% of the observed total effect of DM on disease severity. This mechanistic insight, while requiring cautious interpretation, offers a crucial perspective on a potential pathway to deterioration.
The proposition that a significant proportion of the diabetes–severe COVID-19 association is statistically consistent with mediation by acute metabolic crises is transformative for clinical practice in similar settings. Much of the global pathophysiological discourse has emphasized a model in which chronic hyperglycaemia, inflammation, and immune dysfunction in DM create a substrate predisposing to a dysregulated host response and progressive respiratory failure [
9,
10]. While this chronic inflammatory pathway is undoubtedly relevant, our data suggest that in our setting, a more immediate and clinically overt metabolic crisis may be a primary driver of rapid clinical deterioration for a substantial subset of patients. This supports a plausible mechanistic sequence: SARS-CoV-2 infection acts as a potent metabolic stressor, inducing rapid insulin resistance and the release of catabolic hormones [
12]. In individuals with underlying beta-cell dysfunction or insulin deficiency, this can precipitate DKA or HHS, which, in turn, induces profound physiological insults—severe acidosis, dehydration, electrolyte imbalances, and a prothrombotic and proinflammatory state—that can directly exacerbate respiratory decline and multiorgan dysfunction [
15]. While observational, this model positions acute metabolic decompensation not merely as a comorbidity, but as a key, clinically actionable link in the causal pathway to severe COVID-19 within similar resource-constrained environments.
It is critical to underscore that the analysis of in-hospital mortality was underpowered. The observed elevated but non-significant adjusted odds ratio (aOR=1.48) should be interpreted strictly as an exploratory finding. It should not be construed as evidence against an association between DM and COVID-19 mortality, a link firmly established in larger, adequately powered studies [
5,
7]. Any interpretation beyond the acknowledged issue of inadequate statistical power would be speculative.
The strengths of this study include its focus on an underrepresented population in the global COVID-19 literature, the application of advanced causal mediation methods to probe a specific mechanistic hypothesis, and the use of quantitative bias analysis (E-value) to transparently assess the robustness of our primary finding to unmeasured confounding. The E-value of 5.45 for our primary association indicates that an unmeasured confounder would need to be strongly associated with both DM and severe COVID-19 (a risk ratio of ~5.45 for each) to explain the observed association fully. However, we must acknowledge that factors such as obesity and poor long-term glycemic control (HbA1c), which were unmeasured in our study, are strongly linked to both DM and severe COVID-19[
11,
20], and could potentially have effect magnitudes in this range. Therefore, while the E-value is reassuring, residual confounding by these factors cannot be ruled out entirely.
The study has several limitations. First, the lack of data on BMI and HbA1c is a significant gap; these factors are potent confounders of both diabetes and COVID-19 severity. While the E-value of 5.45 suggests our findings are robust, residual confounding cannot be ruled out. Second, the modest sample size resulted in a wide confidence interval for the mediation estimate (12%–75%), reflecting uncertainty. Finally, the mortality analysis was explicitly underpowered and should be interpreted with extreme caution.
Notwithstanding these limitations, our findings have direct implications for managing COVID-19 and similar respiratory pandemics in regions with a growing DM burden but constrained critical care resources. Clinically, they argue for a paradigm shift in the early inpatient care algorithm for diabetic patients presenting with COVID-19. Protocols should prioritize aggressive, protocol-driven metabolic screening (for ketosis, acidosis, and severe hyperglycemia) and proactive management of impending or established metabolic crises. In settings where ventilators are scarce, preventing metabolic decompensation may be as vital as managing its respiratory consequences.
Future research should employ larger, prospective, multicenter designs with comprehensive metabolic phenotyping (including HbA1c, C-peptide, and ketone monitoring) to validate the potential mediating role of acute metabolic crises. Furthermore, operational research investigating how pre-hospital diabetes management, health literacy, and health system factors (such as drug stock-outs) influence the risk of metabolic decompensation following viral infection would be particularly valuable for designing effective public health interventions in this context.
Conclusion
In western Kenya, DM is a strong predictor of severe COVID-19. Our findings underscore the importance of integrating aggressive frontline glycemic crisis management into COVID-19 care, as preventing metabolic decompensation may be as vital as managing respiratory failure in resource-limited settings.
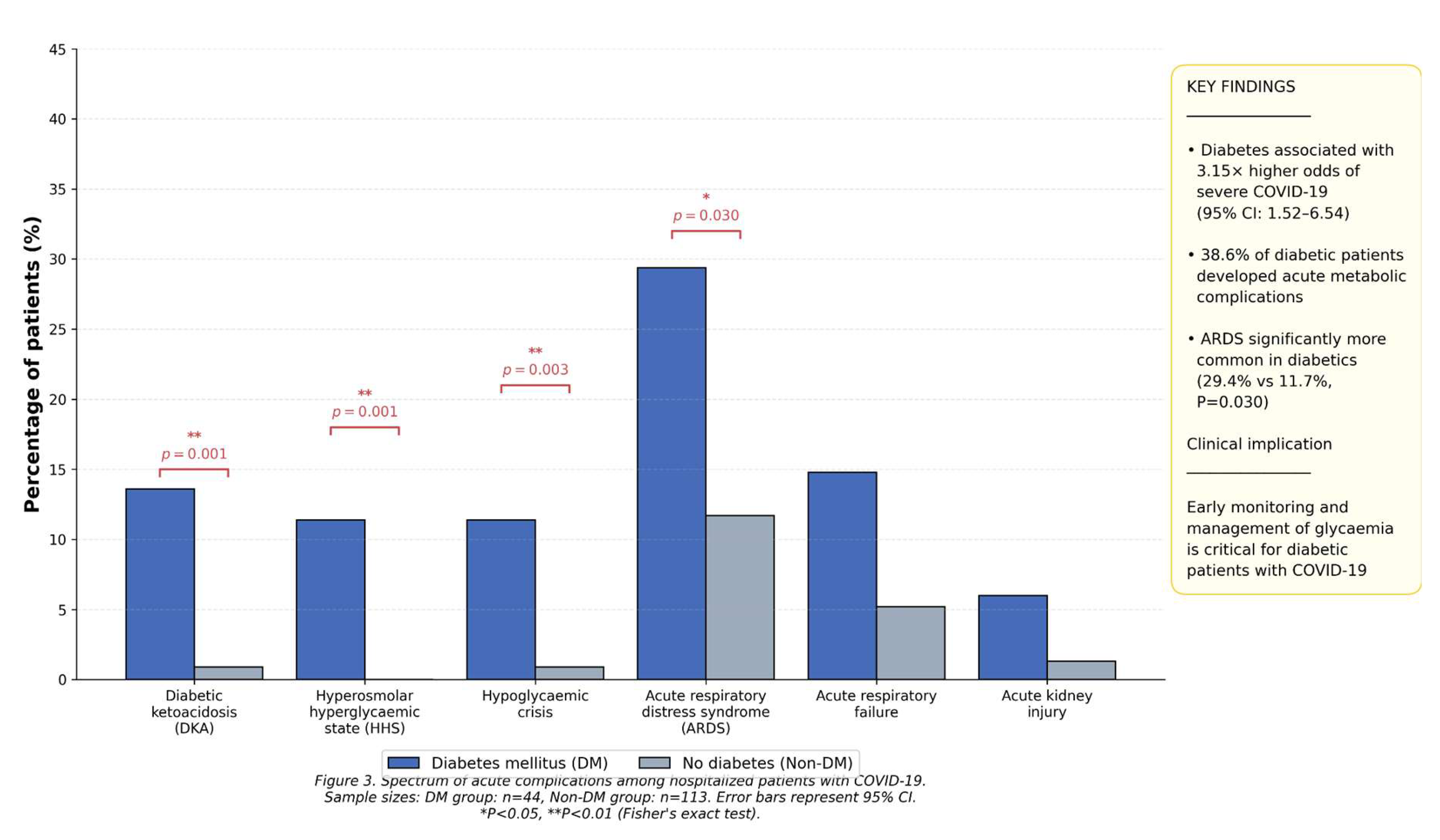
Figure 3.
Spectrum of acute complications by diabetes status. This bar chart compares the prevalence of key acute complications between patients with and without diabetes mellitus, highlighting the significant differences in metabolic crises and ARDS. DKA: Diabetic Ketoacidosis; HHS: Hyperosmolar Hyperglycemic State; ARDS: Acute Respiratory Distress Syndrome. Data presented as percentage within each diabetes status group (DM: n=44; Non-DM: n=113 for DKA/HHS/Hypo; for other complications, percentages are based on the subset with any complication: DM n=34, Non-DM n=77).
Figure 3.
Spectrum of acute complications by diabetes status. This bar chart compares the prevalence of key acute complications between patients with and without diabetes mellitus, highlighting the significant differences in metabolic crises and ARDS. DKA: Diabetic Ketoacidosis; HHS: Hyperosmolar Hyperglycemic State; ARDS: Acute Respiratory Distress Syndrome. Data presented as percentage within each diabetes status group (DM: n=44; Non-DM: n=113 for DKA/HHS/Hypo; for other complications, percentages are based on the subset with any complication: DM n=34, Non-DM n=77).
Table 2.
Disease severity, complications, and outcomes by diabetes status.
Table 2.
Disease severity, complications, and outcomes by diabetes status.
| Outcome |
Total (N=157) |
DM (n=44) |
Non-DM (n=113) |
p-value |
Adjusted odds ratio (95% CI)† |
p-value† |
| Disease severity, n (%) <0.001 |
| Mild/moderate disease |
109 (69.4) |
22 (50.0) |
87 (77.0) |
|
1.0 (Reference) |
|
| Severe/critical disease |
48 (30.6) |
22 (50.0) |
26 (23.0) |
|
3.15 (1.52-6.54) |
0.002 |
| Any COVID-19 complication, n % |
111 (70.7) |
34 (77.3) |
77 (68.1) |
0.259 |
1.42 (0.63-3.21) |
0.402 |
| Acute diabetic complications n/N (%) |
| Diabetic ketoacidosis |
7/157 (4.5) |
6/44 (13.6) |
1/113 (0.9) |
0.001 |
- |
- |
| Hyperosmolar hyperglycemic state (HHS) |
5/157 (3.2) |
5/44 (11.4) |
0/113 (0.0) |
<0.001 |
- |
- |
| Clinical outcomes, n (%) |
| Recovered |
129 (82.2) |
34 (77.3) |
95 (84.1) |
0.311 |
1.0 (Reference) |
|
| Died |
28 (17.8) |
10 (22.7) |
18 (15.9) |
|
1.48 (0.62 - 3.55) |
0.380 |
Table 3.
Specific COVID-19-related complications by diabetes mellitus status.
Table 3.
Specific COVID-19-related complications by diabetes mellitus status.
| Type of COVID-19 complication |
Total (n=111) |
DM (n=34) |
Non-DM (n=77) |
p-value* |
| Severe pneumonia, n (%) |
90 (81.1) |
27 (79.4) |
63 (81.8) |
0.757 |
| Acute respiratory distress syndrome (ARDS), n (%) |
19 (17.1) |
10 (29.4) |
9 (11.7) |
0.030 |
| Acute respiratory failure (%) |
9 (8.1) |
5 (14.7) |
4 (5.2) |
0.128 |
| Acute kidney injury, n (%) |
3 (2.7) |
2 (5.9) |
1(1.3) |
0.202 |
| Thromboembolic events (DVT & PE), n (%) |
8 (7.2) |
2 (5.9) |
6 (7.8) |
1.000 |
| Acute cardiac injury, n (%) |
7 (6.3) |
2 (5.9) |
5 (6.5) |
1.000 |
Author Contributions
Omullo FP and Kamau AW designed the study and wrote the original draft; Omullo FP, Kamau AW, and Kipsang I were responsible for developing the methodology and data curation; Otsyulah HF, Abdimajid AO, and Wachiye WA participated in the formal analysis and investigation; Muraria KM, Oweke LA, and Morema C participated in the review and editing. All authors have read and approved the final manuscript.
References
- Fronteira, I; Sidat, M; Magalhães, JP; de Barros, FPC; Delgado, AP; Correia, T; Daniel-Ribeiro, CT; Ferrinho, P. The SARS-CoV-2 pandemic: A syndemic perspective. One Health 2021, 12, 100228. [Google Scholar] [CrossRef] [PubMed]
- Guo, W; Li, M; Dong, Y; et al. Diabetes is a risk factor for the progression and prognosis of COVID-19. Diabetes Metab Res Rev. 2020, 36(7), e3319. [Google Scholar] [CrossRef] [PubMed]
- Yu, H; Liu, J; He, X. From Glucotoxicity to Lung Injury: Emerging Perspectives on Diabetes-Associated Respiratory Complications. Lung 2025, 203, 80. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F; Yu, T; Du, R; Fan, G; Liu, Y; Liu, Z; Xiang, J; Wang, Y; Song, B; Gu, X; Guan, L; Wei, Y; Li, H; Wu, X; Xu, J; Tu, S; Zhang, Y; Chen, H; Cao, B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef] [PubMed]
- Onder, G; Rezza, G; Brusaferro, S. Case-Fatality Rate and Characteristics of Patients Dying in Relation to COVID-19 in Italy. JAMA 2020, 323, 1775–1776. [Google Scholar] [CrossRef] [PubMed]
- Stokes, EK; Zambrano, LD; Anderson, KN; Marder, EP; Raz, KM; El Burai Felix, S; Tie, Y; Fullerton, KE. Coronavirus Disease 2019 Case Surveillance - United States, January 22-May 30, 2020. MMWR Morb Mortal Wkly Rep 2020, 69, 759–765. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A; Arora, A; Sharma, P; Anikhindi, SA; Bansal, N; Singla, V; Khare, S; Srivastava, A. Is diabetes mellitus associated with mortality and severity of COVID-19? A meta-analysis. Diabetes Metab Syndr 2020, 14, 535–545. [Google Scholar] [CrossRef] [PubMed]
- Hussain, A; Bhowmik, B; do Vale Moreira, NC. COVID-19 and diabetes: Knowledge in progress. Diabetes Res Clin Pract 2020, 162, 108142. [Google Scholar] [CrossRef] [PubMed]
- Bornstein, SR; Dalan, R; Hopkins, D; Mingrone, G; Boehm, BO. Endocrine and metabolic link to coronavirus infection. Nat Rev Endocrinol 2020, 16, 297–298. [Google Scholar] [CrossRef] [PubMed]
- Yang, JK; Lin, SS; Ji, XJ; Guo, LM. Binding of SARS coronavirus to its receptor damages islets and causes acute diabetes. Acta Diabetol 2010, 47, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Muniyappa, R; Gubbi, S. COVID-19 pandemic, coronaviruses, and diabetes mellitus. Am J Physiol Endocrinol Metab 2020, 318, E736–E741. [Google Scholar] [CrossRef] [PubMed]
- Pal, R; Bhadada, SK. Managing common endocrine disorders amid COVID-19 pandemic. Diabetes Metab Syndr. 2020, 14(5), 767–771. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z; McGoogan, JM. Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72 314 Cases From the Chinese Center for Disease Control and Prevention. JAMA 2020, 323(13), 1239–1242. [Google Scholar] [CrossRef] [PubMed]
- Lone, SA; Ahmad, A. COVID-19 pandemic - an African perspective. Emerg Microbes Infect 2020, 9, 1300–1308. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, SF; Mwangi, M; Mutua, MK; Kibachio, J; Hussein, A; Ndegwa, Z; Owondo, S; Asiki, G; Kyobutungi, C. Prevalence and factors associated with pre-diabetes and diabetes mellitus in Kenya: results from a national survey. BMC Public Health 2018, 18, 1215. [Google Scholar] [CrossRef] [PubMed]
- Nkengasong, JN; Ndembi, N; Tshangela, A; Raji, T. COVID-19 vaccines: how to ensure Africa has access. Nature 2020, 586, 197–199. [Google Scholar] [CrossRef] [PubMed]
- Haider, N; Osman, AY; Gadzekpo, A; Akipede, GO; Asogun, D; Ansumana, R; Lessells, RJ; Khan, P; Hamid, MMA; Yeboah-Manu, D; Mboera, L; Shayo, EH; Mmbaga, BT; Urassa, M; Musoke, D; Kapata, N; Ferrand, RA; Kapata, PC; Stigler, F; Czypionka, T; Zumla, A; Kock, R; McCoy, D. Lockdown measures in response to COVID-19 in nine sub-Saharan African countries. BMJ Glob Health 2020, 5, e003319. [Google Scholar] [CrossRef] [PubMed]
- Sardu, C; D’Onofrio, N; Balestrieri, ML; Barbieri, M; Rizzo, MR; Messina, V; Maggi, P; Coppola, N; Paolisso, G; Marfella, R. Outcomes in Patients With Hyperglycemia Affected by COVID-19: Can We Do More on Glycemic Control? Diabetes Care 2020, 43, 1408–1415. [Google Scholar] [CrossRef] [PubMed]
- Agyemang, C; Richters, A; Jolani, S; et al. Ethnic minority status as a determinant of COVID-19 vaccine hesitancy and a target for intervention strategies in the Netherlands: findings from the HELIUS study. Lancet Reg Health Eur. 2022, 17, 100372. [Google Scholar]
- Mbunge, E; Akinnuwesi, B; Fashoto, SG; et al. A critical review of emerging technologies for tackling COVID-19 pandemic. Hum Behav Emerg Technol. 2021, 3(1), 25–39. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |