1. Introduction
Acromegaly is a rare, chronic systemic disorder caused by prolonged exposure to excess growth hormone (GH) and its downstream effector insulin-like growth factor-1 (IGF-1), most often resulting from a pituitary somatotroph adenoma. The resulting endocrine milieu drives progressive multisystem involvement – classically soft-tissue overgrowth, organomegaly, connective-tissue remodeling, and contributes to a wide range of metabolic and cardiovascular comorbidities [
1]. Despite the breadth of recognized sequelae, the female reproductive tract, particularly the uterus and cervix, has not been comprehensively characterized in acromegaly with available data limited to isolated case reports and small, heterogeneous series.
Two converging lines of contemporary evidence support targeted assessment of uterine and, especially, cervical morphometry in acromegaly. First, GH/IGF-1 signaling exerts potent trophic effects on fibroblasts and extracellular matrix (ECM) biology. In human pelvic floor models, IGF-1 enhances fibroblast growth and modulates collagen metabolism via MAPK and NF-κB pathways; parallel findings across connective-tissue systems demonstrate that IGF-1 promotes matrix synthesis (notably collagen I) and influences tissue repair dynamics [
2]. These matrix-directed actions provide a mechanistic rationale for stromal hypertrophy under conditions of sustained endocrine excess
Second, the cervix is a predominantly ECM-based organ. Contemporary reviews of cervical biology emphasize that ECM accounts for the vast majority of cervical tissue mass (commonly estimated at ~85 – 90%), whereas cellular elements – including a relatively small smooth-muscle fraction – compose the minority. Cervical dimensions and biomechanics are governed by collagen architecture, cross-linking, and glycosaminoglycan-mediated hydration [
3]. In contrast, the uterine corpus is dominated by smooth muscle (myometrium) with a comparatively lower ECM fraction. From this contrast arises a biologically coherent expectation: chronic GH/IGF-1 excess should disproportionately influence collagen-rich, hydrophilic connective tissues such as the cervix, potentially more than the myometrial uterus.
Clinically, reproductive manifestations in acromegaly – menstrual irregularities, subfertility, and pregnancy-related considerations – are typically interpreted through endocrine mechanisms (hyperprolactinemia, hypogonadotropic hypogonadism, treatment effects). These explanations, however, do not preclude a structural contribution from GH/IGF-1–driven stromal remodeling within the uterus–cervix unit. The published gynecologic literature in acromegaly remains sparse and heterogeneous. A recent 2024 systematic review on uterine fibroids (leiomyomas) in acromegaly reported inconsistent findings across small observational studies and highlighted methodological and confounding issues, leaving unresolved questions regarding the somatotropic axis and uterine disease burden [
4]. Notably, virtually no studies have prospectively or systematically quantified cervical size, despite its ECM-rich composition and theoretical susceptibility to GH/IGF-1 trophic effects.
Ultrasonography provides a pragmatic avenue to address this knowledge gap. Current practice guidelines recognize transvaginal (and, when indicated, transabdominal) sonography as a reproducible method for assessing cervical dimensions and detecting disease-related changes in length and caliber. Standardized technique (e.g., minimizing probe pressure, mid-sagittal alignment) measurement improves reliability, while ratio-based indices can help normalize inter-individual variation [
5]. Although uterine sonographic metrics are well established in gynecologic imaging, disease-specific cervical reference patterns outside obstetric contexts remain underdeveloped – precisely where acromegaly may contribute a distinct morphological phenotype.
Accordingly, we conducted a retrospective case–control study to compare morphometric parameters of the uterine corpus and cervix in women with a history of acromegaly against two comparators: age-matched healthy controls and women with adenomyosis (without acromegaly). Rather than testing a single predefined hypothesis, we systematically measured and contrasted uterine and cervical characteristics across cohorts. Our exploratory finding is biologically plausible given contemporary endocrine and tissue-engineering insights: (i) GH/IGF-1 augments fibroblast activity and ECM synthesis in human connective tissues; (ii) cervical structure and mechanics are governed primarily by collagen-rich ECM and tissue hydration; and (iii) matrix-dominant organs would be expected to manifest the stromal imprint of acromegaly more readily than muscle-dominant ones [
2].
Defining whether acromegaly is associated with a reproducible, noninvasive cervical morphometric signature has meaningful clinical implications. A validated imaging pattern (e.g., enlarged cervix with preserved uterine body, altered uterus-to-cervix proportionality) could: (a) serve as an auxiliary marker of systemic GH/IGF-1 tissue effects; (b) inform planning for transcervical procedures in reproductive medicine and gynecology, where dimensions and viscoelasticity influence instrumentation and success; and (c) generate hypotheses about obstetric and pelvic floor outcomes that warrant prospective evaluation. By comparing women with acromegaly to both healthy controls and gynecologic disease cohorts, our study aims to distinguish a somatotropic, connective-tissue–specific phenotype from nonspecific pelvic pathology.
In summary, integrating current insights into GH/IGF-1-ECM biology and contemporary sonographic methodology, we test the hypothesis that acromegaly is characterized by a selective, ECM-driven cervical hypertrophy – an underrecognized visceral manifestation with potential diagnostic and procedural relevance.
2. Materials and Methods
We performed a retrospective case–control study including three cohorts drawn from reproductive-age women with available pelvic ultrasound measurements: acromegaly (n = 33), defined by a confirmed diagnosis; healthy controls (n = 45), with no known endocrine disease; and adenomyosis (n = 44), with no history of acromegaly.
Analyses were conducted on a complete-case basis for the prespecified ultrasound outcomes. Eligibility criteria are summarized in
Table 1.
Ultrasound Acquisition
Pelvic ultrasound was performed transvaginal or transabdominal according to clinical indication. Measurements were obtained on orthogonal planes following routine gynecologic imaging practice: uterine body measured in mid-sagittal and transverse views, avoiding excessive probe pressure. Cervix measured with the endocervical canal centered, from external to internal os for length, and at maximal orthogonal diameters for width and anteroposterior (AP) thickness. All linear dimensions were recorded in centimeters. Uterine and cervical dimensions (length, width, and anteroposterior size) were measured via transabdominal or transvaginal ultrasound, depending on clinical indications. Volumes were calculated using the ellipsoid formula: V = (L × W × H) 3/ 60.79, where L = length, W = width, and H = anteroposterior dimension. Ultrasound acquisition and morphometric variables used in the analysis are summarized in
Table 2.
Statistical Analysis
Continuous variables were summarized as median [IQR]. Because distributions were non-parametric, Mann–Whitney U-tests were used for pairwise comparisons (acromegaly vs controls; acromegaly vs adenomyosis). Chi-square/Fisher’s exact tests were used for categorical data if applicable. Two-sided p<0.05 (or p<0.025 for Bonferroni-adjusted pairwise tests) indicated statistical significance. Analyses were performed on complete cases only.
3. Results
3.1. Cohort Overview
A total of 122 women were included: 33 with history of acromegaly, 45 healthy controls, and 44 with adenomyosis. Groups were age-matched by design. Ultrasound approach (transvaginal and transabdominal) followed clinical indications across cohorts.
3.2. Uterine Body Size and Volume
Across the three cohorts, no significant differences were observed in uterine body morphometry. Uterine length, width, anteroposterior (AP) dimension, and calculated uterine volume were statistically indistinguishable between women with acromegaly and both comparison groups (all p > 0.1). Thus, within the predominantly myometrial uterine corpus, acromegaly did not demonstrate measurable enlargement under our imaging protocol.
3.3. Cervical Dimensions and Volume
In contrast, all three cardinal cervical measurements were significantly increased in the acromegaly cohort relative to healthy controls:
Cervical length: p = 0.000041
Cervical width: p = 0.000002
Cervical AP thickness: p = 0.000021
These differences translated into a marked elevation of cervical volume in the acromegaly group (p = 0.000005). The consistency of findings across all measurement planes supports a global hypertrophic effect rather than a shape-specific artifact. Compared with controls, women with acromegaly demonstrated larger cervical linear dimensions and a higher cervical volume, while uterine body dimensions were comparatively less affected. This pattern yielded a consistently increased cervix-to-uterus ratio, suggesting a preferential “cervical phenotype” rather than a diffuse enlargement of the whole uterus.
3.4. Proportionality of Uterus and Cervix
The uterine-to-cervical (U:C) volume ratio differed significantly between acromegaly and healthy controls (p = 0.001091). This ratio shift reflects a disproportionate cervical enlargement with relative preservation of uterine body size. Clinically, this disproportion may be more informative than any single absolute metric, as it normalizes for patient-level body size and inter-scanner variation. Descriptive statistics for all ultrasound parameters are summarized in
Table 3.
3.5. Disease Comparator Analyses
To probe specificity, we compared acromegaly to women with adenomyosis. While uterine body dimensions again did not differ, cervical width (p = 0.00003), cervical AP thickness (p = 0.000126), and U:C size-ratio (p = 0.000152) remained significantly altered in acromegaly. These findings suggest that the cervical phenotype is not a non-specific byproduct of pelvic pain disorders, chronic inflammation, or hormonal treatments typical of adenomyosis care, but rather a pattern distinctive of acromegaly-related connective tissue remodeling.
3.6. Sensitivity Considerations
The consistency of results across three orthogonal cervical axes argues against measurement bias from a single view. The significant ratio-based differences further mitigate concerns about inter-individual variability and imaging approach. As uterine body metrics did not differ across groups, the acromegaly-associated signal appears anatomically localized to the cervix rather than reflecting generalized uterine enlargement. Taken together, these data support the presence of a previously underrecognized acromegalic uteropathy characterized by disproportionate cervical hypertrophy with preserved uterine corpus dimensions.
4. Discussion
This study provides, to our knowledge, the first targeted ultrasonographic evidence that acromegaly in women is associated with selective cervical enlargement, while uterine body dimensions remain comparable to both healthy controls and a gynecologic disease comparator (adenomyosis). The consistent shift in internal proportionality (reduced U:C volume ratio) reinforces the concept that the cervix–rather than the uterine body–is preferentially involved.
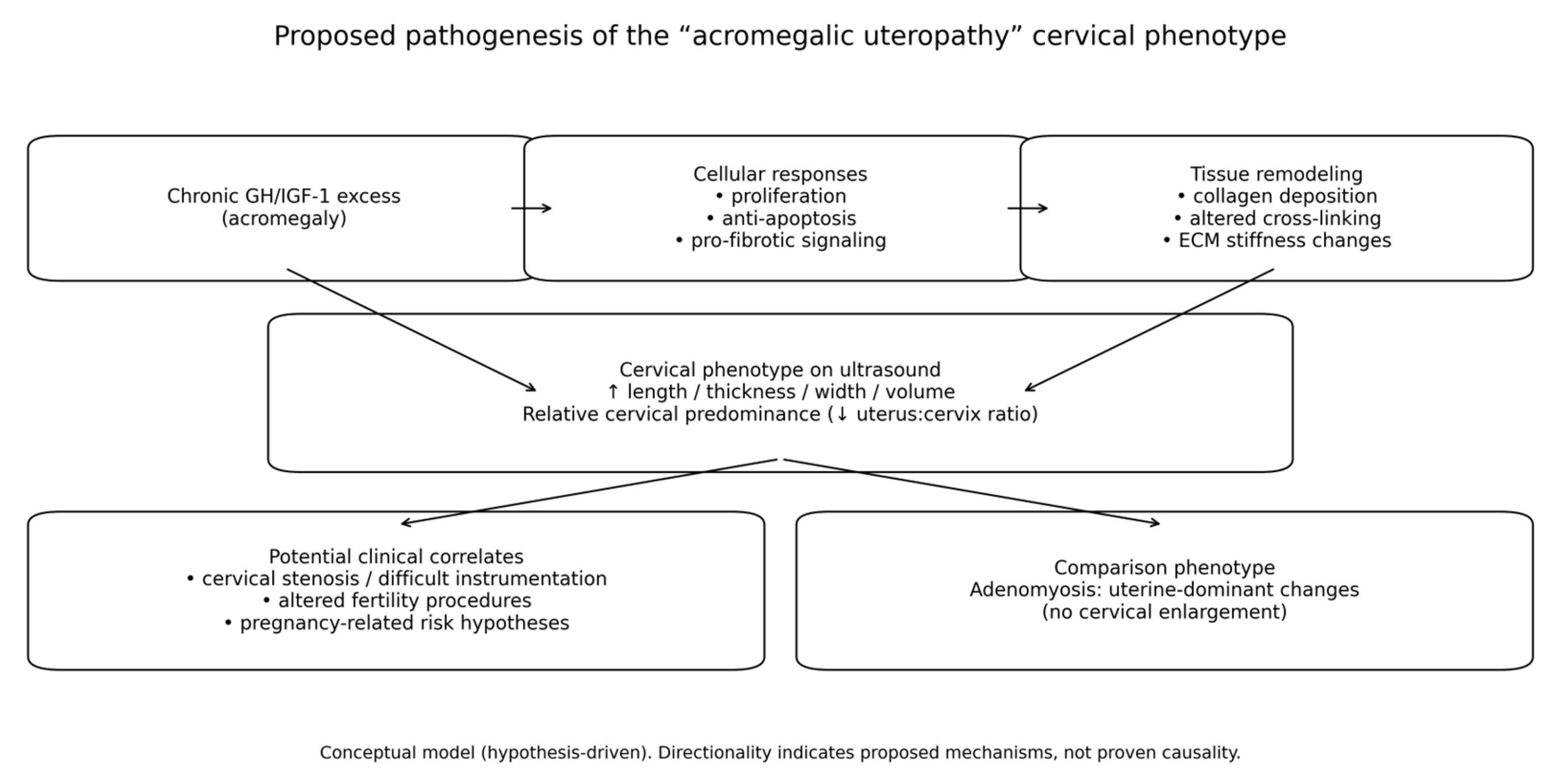
The observed pattern is biologically coherent. The cervix is a matrix-dominant organ. As mentioned earlier about 80–85% of its stroma is collagen-rich connective tissue, with a relatively small smooth muscle component. GH/IGF-1 signaling is known to stimulate fibroblast activation, enhance collagen synthesis, and expand ECM mass and hydration–mechanisms that underlie classic soft-tissue features of acromegaly (e.g., skin thickening, tendon enlargement, ligamentous hypertrophy) [
6]. Contemporary structural and microstructural work confirms that cervical size, biomechanics, and viscoelasticity are governed by collagen architecture, cross-linking, and glycosaminoglycan-mediated hydration [
7]. In a tissue whose bulk reflects ECM quantity and hydration more than muscle mass, sustained trophic drive from GH/IGF-1 would be expected to produce diffuse hypertrophy across all cervical axes—precisely the pattern observed here. By contrast, the uterine corpus is myometrium-predominant (smooth muscle), where size and stiffness are less directly dictated by collagen-rich ECM. If GH/IGF-1 excess preferentially augments fibroblast/ECM programs (via PI3K/AKT–MAPK pathways, among others), a matrix-heavy organ should exhibit the stromal imprint of acromegaly more readily than a muscle-heavy one [
8]. Proposed pathophysiological model of “acromegalic uteropathy” with preferential cervical stromal expansion are summarized in
Figure 1.
From a mechanistic standpoint, acromegaly is characterized by chronic exposure of connective tissues to GH/IGF-1 excess, which is known to activate stromal fibroblasts and to increase collagen turnover and extracellular matrix remodeling. A prospective biomarker study in active acromegaly reported higher circulating fibroblast activation protein (FAP) levels that declined after biochemical control, in parallel with markers of collagen turnover [
9]. Together with broader consensus documents on acromegaly comorbidities and outcomes [
10], these data support the biological plausibility that the cervix—an organ rich in collagenous stroma and proteoglycans—could enlarge predominantly through extracellular matrix expansion, altered hydration and cross-linking, and changes in vascular permeability.
Bioengineering studies further demonstrate that measurable cervical dimensions and mechanics are emergent properties of ECM microarchitecture and hydration, supporting the plausibility that sonographic enlargement reflects underlying ECM remodeling rather than mere geometric variation [
11]. Broader translational literature also confirms IGF-1 as a key facilitator of type I collagen synthesis and ECM turnover [
10]. ECM hydration driven by glycosaminoglycan content modulates tissue turgor and viscoelasticity and could further contribute to increased cervical volume [
13].
Historically, gynecologic discussions in acromegaly focused on uterine fibroids, with inconsistent results likely driven by small samples and confounding [
4]. Fibroids, as discrete smooth-muscle tumors, do not model diffuse stromal responses, and our findings suggest that generalized uterine enlargement is not a prominent feature of acromegaly. Rather, the cervix appears to embody the connective-tissue responsiveness of the somatotropic axis.
The comparison group with adenomyosis is informative: adenomyosis classically affects the uterine body and the junctional zone, with ultrasound definitions refined in recent years through the revised MUSA consensus [
14]. Our data show that although adenomyosis may increase uterine volume, it does not reproduce the same magnitude of cervical enlargement or cervix/uterus ratio elevation observed in acromegaly. This distinction strengthens the concept of “acromegalic uteropathy” as a separate imaging phenotype and helps avoid misattribution of cervical changes to adenomyosis alone.
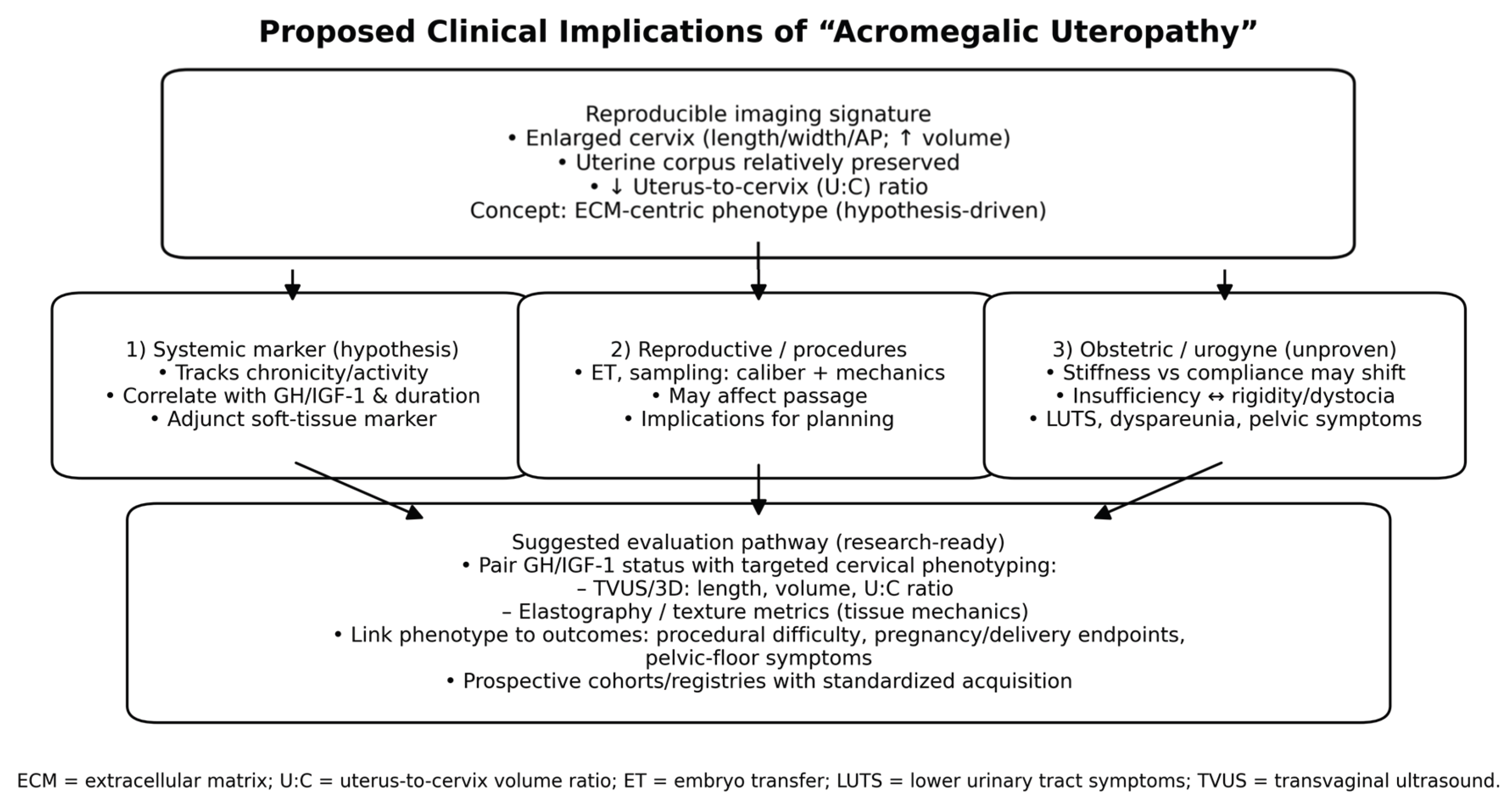
Potential Clinical Implications.
First, a reproducible imaging signature (enlarged cervix with preserved uterine body; altered U:C ratio) might serve as an auxiliary marker of systemic disease activity or chronicity in women with acromegaly, analogous to heel-pad thickness or soft-tissue measurements elsewhere.
Second, cervical enlargement could intersect with reproductive care. Transcervical procedures (e.g., embryo transfer, endometrial sampling) rely on predictable cervical dimensions and tissue mechanics. Cervical ECM content and hydration influence viscoelasticity and instrument passage; in other gynecologic contexts, sonographic cervical texture has been associated with procedure difficulty [
15]. Whether the acromegalic cervix alters procedure planning, analgesia, or device choice deserves prospective evaluation.
Third, obstetric and urogynecologic sequelae are plausible but unproven. The balance between collagen content, cross-linking, and hydration determines cervical stiffness and compliance. If GH/IGF-1 excess increases collagen turnover and matrix mass, the net effect on competence in pregnancy (insufficiency vs. rigidity) is not self-evident and may depend on disease control and parity. Current reviews of pregnancy in acromegaly [
16]) reports generally reassuring maternal–fetal outcomes but highlights heterogeneous, observational data and a lack of standardized, cervix-specific phenotyping. In other words, current evidence says pregnancy is often safe overall, but it has not asked the question our data raise: whether acromegaly-related cervical enlargement – an ECM-centric phenotype – modifies risks relevant to pregnancy and delivery (e.g., cervical insufficiency vs. dystocia), or affects procedure planning in reproductive care [
16]. Prospective registries should therefore pair GH/IGF-1 control with targeted cervical assessments (length/volume and uterus-to-cervix ratio by TVUS/3D, plus elastography or texture metrics) to define mechanics and outcomes; similar protocols could clarify potential urogynecologic implications (pelvic floor symptoms, lower urinary tract complaints, dyspareunia) in relation to cervical bulk and tissue properties. These hypotheses require targeted, longitudinal studies.
Methodologically, our approach leveraged standard pelvic ultrasound and simple derived indices that are widely available and inexpensive. The use of ratio metrics (U:C volume) adds robustness by internal normalization. Ultrasonographic volume estimation by ellipsoid approximation remains routine in pelvic imaging, providing a pragmatic surrogate for true 3D anatomy [
17]. Potential clinical implications of acromegalic uteropathy are summarized in
Figure 2.
Future Directions: Standardized Imaging and Prospective Validation
Future work should standardize pelvic ultrasound acquisition and reporting using contemporary practice parameters and explicit measurement conventions (including ellipsoid-derived volumes), to improve reproducibility of cervical phenotyping across centers [
18,
19]. For differential diagnosis and comparator phenotyping, adenomyosis should be reported using revised MUSA definitions and complementary ratio-based ultrasound metrics (e.g., myometrial–cervical ratio), with emerging severity-grading approaches to support reproducible stratification [
14,
21,
22]. Prospective registries could then pair GH/IGF-1 status (using contemporary consensus criteria and management guidance) with targeted cervical phenotyping and clinically meaningful endpoints, including technical outcomes of embryo transfer/other transcervical procedures and pregnancy-related outcomes, where current evidence is generally reassuring but heterogeneous [
16,
23,
24,
25,
26,
27,
28,
29,
30].
Limitations and Strengths
Limitations include the retrospective design and lack of granular covariates that affect pelvic anatomy (parity, menopausal status, hormonal therapy, prior cervical procedures). Imaging route (transvaginal vs. transabdominal) was clinically determined, although the direction and strength of differences across all cervical axes mitigate concerns about systematic bias. Importantly, we did not correlate morphometry with contemporaneous GH/IGF-1 or disease duration, precluding dose–response inference.
Strengths include two comparator groups (healthy and disease), consistent directionality across axes, and a proportionality metric that internalizes normalization. The biological rationale is anchored in current GH/IGF-1–ECM science and modern cervical ECM characterization.
5. Conclusions
We propose that cervical enlargement is an under-recognized visceral manifestation of acromegaly, reflecting an ECM-centric response to GH/IGF-1. This acromegalic uteropathy–disproportionate cervical hypertrophy with preserved uterine body size–may carry diagnostic and procedural relevance across gynecologic and reproductive care. Next steps are to (i) validate these findings prospectively with standardized transvaginal protocols and 3D/elasticity measures; (ii) establish reference ranges for cervical volume and U:C across reproductive stages; (iii) correlate cervical morphometry with GH/IGF-1 control and disease chronicity; and (iv) assess clinical outcomes in pregnancy and assisted reproduction.
Author Contributions
Conceptualization, I.S., S.V.; methodology, I.S., S.V., E.G.; validation, A.A., Yu.F., U.S. and A.B.; formal analysis, S.V., L.D., E.P., G.M. and N.M.; data curation, A.A., Yu.F. and A.B.; writing—review and editing, A.A, S.V., L.D., E.P. and G.M; supervision, I.S and U.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of the Endocrinology Research Centre (Approval #12/23.November.2023).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The datasets used and analyzed during this study are available from the corresponding author upon reasonable request.
Acknowledgments
The authors thank the clinical and ultrasound teams of the Endocrinology Research Centre and the Kursk Region Scientific and Clinical Center named after G. E. Ostroverkhov for their assistance with retrieval of ultrasound data and verification of measurements. The authors have reviewed and take full responsibility for the content of this publication.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| 3D |
three-dimensional |
| AKT |
protein kinase B |
| AP |
anteroposterior |
| ECM |
extracellular matrix |
| GH |
growth hormone |
| IGF-1 |
insulin-like growth factor 1 |
| IQR |
interquartile range |
| MAPK |
mitogen-activated protein kinase |
| NF-κB |
nuclear factor kappa B |
| PI3K |
phosphoinositide 3-kinase |
| TVUS |
transvaginal ultrasound |
| U:C |
uterus-to-cervix |
References
- Fleseriu, Maria; et al. Acromegaly: pathogenesis, diagnosis, and management. The Lancet Diabetes & Endocrinology 2022, Volume 10(Issue 11), 804–826. [Google Scholar] [CrossRef]
- Yin, Y; Han, Y; Shi, C; Xia, Z. IGF-1 regulates the growth of fibroblasts and extracellular matrix deposition in pelvic organ prolapse. Open Med (Wars) 2020, 15(1), 833–840. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Félix, J; Bartosch, C; Matias, A. Unlocking the Cervix: Biological Mechanisms and Research Gaps in Preterm Birth. Cureus 2024, 16(11), e72835. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Górski, K; Zgliczyński, S; Stelmachowska-Banaś, M; Czajka-Oraniec, I; Zgliczyński, W; Ciebiera, M; Zgliczyńska, M. Uterine fibroids in women diagnosed with acromegaly: a systematic review. Rev Endocr Metab Disord 2024, 25(4), 773–781. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Coutinho, CM; Sotiriadis, A; Odibo, A; Khalil, A; D'Antonio, F; Feltovich, H; Salomon, LJ; Sheehan, P; Napolitano, R; Berghella, V; da Silva Costa, F. ISUOG Practice Guidelines: role of ultrasound in the prediction of spontaneous preterm birth. Ultrasound Obstet Gynecol 2022, 60(3), 435–456. [Google Scholar] [CrossRef] [PubMed]
- House, M; Kaplan, DL; Socrate, S. Relationships between mechanical properties and extracellular matrix constituents of the cervical stroma during pregnancy. Semin Perinatol 2009, 33(5), 300–7. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wu, W.; Sun, Z.; Gao, H.; et al. Whole cervix imaging of collagen, muscle, and cellularity in term and preterm pregnancy. Nat Commun 2024, 15, 5942. [Google Scholar] [CrossRef] [PubMed]
- Khan, MZ; Zugaza, JL; Torres Aleman, I. The signaling landscape of insulin-like growth factor 1. J Biol Chem. 2025, 301(1), 108047. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Arlien-Søborg, M. C.; Grøndahl, C.; Bæk, A.; Dal, J.; Alle Madsen, M.; Høgild, M. L.; Pedersen, S. B.; Bjerre, M.; Jørgensen, J. O. L. Fibroblast activation protein is a GH target: A prospective study of patients with acromegaly before and after treatment. Journal of Clinical Endocrinology and Metabolism 2020, 105(1), 106–115. [Google Scholar] [CrossRef]
- Fleseriu, M; Biller, BMK; Freda, PU; Gadelha, MR; Giustina, A; Katznelson, L; Molitch, ME; Samson, SL; Strasburger, CJ; van der Lely, AJ; Melmed, S. A Pituitary Society update to acromegaly management guidelines. Pituitary 2021, 24(1), 1–13. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yoshida, Kyoko. Bioengineering and the cervix: The past, current, and future for addressing preterm birth. Current Research in Physiology 2023, Volume 6, 100107. [Google Scholar] [CrossRef]
- Miescher, I.; Rieber, J.; Calcagni, M.; Buschmann, J. In Vitro and In Vivo Effects of IGF-1 Delivery Strategies on Tendon Healing: A Review. Int. J. Mol. Sci. 2023, 24, 2370. [Google Scholar] [CrossRef] [PubMed]
- Berdiaki, A; Neagu, M; Tzanakakis, P; Spyridaki, I; Pérez, S; Nikitovic, D. Extracellular Matrix Components and Mechanosensing Pathways in Health and Disease. Biomolecules 2024, 14(9), 1186. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Harmsen, MJ; Van den Bosch, T; de Leeuw, RA; Dueholm, M; Exacoustos, C; Valentin, L; Hehenkamp, WJK; Groenman, F; De Bruyn, C; Rasmussen, C; Lazzeri, L; Jokubkiene, L; Jurkovic, D; Naftalin, J; Tellum, T; Bourne, T; Timmerman, D; Huirne, JAF. Consensus on revised definitions of Morphological Uterus Sonographic Assessment (MUSA) features of adenomyosis: results of modified Delphi procedure. Ultrasound Obstet Gynecol 2022, 60(1), 118–131. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Stanziano, A; Caringella, AM; Cantatore, C; Trojano, G; Caroppo, E; D'Amato, G. Evaluation of the cervix tissue homogeneity by ultrasound elastography in infertile women for the prediction of embryo transfer ease: a diagnostic accuracy study. Reprod Biol Endocrinol 2017, 15(1), 64. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bandeira, DB; Olivatti, TOF; Bolfi, F; Boguszewski, CL; Dos Santos Nunes-Nogueira, V. Acromegaly and pregnancy: a systematic review and meta-analysis. Pituitary 2022, 25(3), 352–362. [Google Scholar] [CrossRef] [PubMed]
- Karaca-Saygili, O; Taskin, MI; Keyik, BY; Sackes, M; Ozcan, E; Kus, I. Comparison of Three Methods for Estimating Volume of the Uterine Layers in Healthy Women: A Stereological Study. International Journal of Morphology 2018, 36(2), 614–622. [Google Scholar] [CrossRef]
- AIUM Practice Parameter for the Performance of Ultrasound of the Female Pelvis: 2024 Revision. J Ultrasound Med. 2024, 43(11), E56–E64. [CrossRef]
- Gao, H.; Liu, D.E.; Li, Y.; Tang, J.; Hu, S.; Wu, X.; Tian, Z.; Tan, H. Uterine size and volume are associated with a higher clinical pregnancy rate in patients undergoing assisted reproduction technology: A longitudinal study (STROBE-compliant). Medicine (Baltimore) 2019, 98(8), e14366. [Google Scholar] [CrossRef] [PubMed]
- McCaughey, T.; Cheng, W.; Liscano, J.; et al. The use of the myometrial–cervical ratio in the ultrasound diagnosis of adenomyosis. Ultrasound Obstet Gynecol. 2022, 60(3), 420–426. [Google Scholar] [CrossRef]
- Trommelen, LM; De Leeuw, RA; Van den Bosch, T; Huirne, JAF. Grading Sonographic Severity of Adenomyosis: A Pilot Study Assessing Feasibility and Interobserver Reliability. J Ultrasound Med. 2025, 44(3), 495–506. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gallo, R; Kamberaj, L; Baroni, A; De Cicco Nardone, A; Scambia, G; Masciullo, V. Advances in non-invasive diagnosis of uterine adenomyosis: a narrative review. Gynecol Pelvic Med 2025, 8, 13. [Google Scholar] [CrossRef]
- D'Angelo, A; Panayotidis, C; Alteri, A; Mcheik, S; Veleva, Z. Evidence and consensus on technical aspects of embryo transfer. Hum Reprod Open 2022, 2022(4), hoac038. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Schoolcraft, WB. Importance of embryo transfer technique in maximizing assisted reproductive outcomes. Fertil Steril 2016, 105(4), 855–60. [Google Scholar] [CrossRef] [PubMed]
- Nazzaro, G; Saccone, G; Miranda, M; Crocetto, F; Zullo, F; Locci, M. Cervical elastography using E-cervix for prediction of preterm birth in singleton pregnancies with threatened preterm labor. J Matern Fetal Neonatal Med. 2022, 35(2), 330–335. [Google Scholar] [CrossRef] [PubMed]
- Giustina, A; Biermasz, N; Casanueva, FF; Fleseriu, M; Mortini, P; Strasburger, C; van der Lely, AJ; Wass, J; Melmed, S; Acromegaly Consensus Group. Consensus on criteria for acromegaly diagnosis and remission. Pituitary Erratum in: Pituitary. 2024 Feb;27(1):88. doi: 10.1007/s11102-023-01373-w. PMID: 37923946; PMCID: PMC10837217. 2024, 27(1), 7–22. [Google Scholar] [CrossRef]
- Melmed, S; di Filippo, L; Fleseriu, M; Mercado, M; Karavitaki, N; Gurnell, M; Salvatori, R; Tsagarakis, S; Losa, M; Maffei, P; Pereira, AM; Geer, EB; Katznelson, L; van der Lely, AJ; Bollerslev, J; Esposito, D; Webb, SM; Zatelli, MC; Valassi, E; Neggers, S; Chanson, P; Ho, KKY; Ioachimescu, AG; Biller, BMK; Samson, SL; Kaiser, UB; Schilbach, K; Luque, RM; Casanueva, FF; Shimon, I; Boguszewski, CL; Biermasz, N; Colao, A; Pirchio, R; Lamberts, SWJ; Kadioglu, P; Buchfelder, M; Frara, S; Chiloiro, S; Petersenn, S; Gadelha, MR; Puig-Domingo, M; Luger, A; Brue, T; Beckers, A; Ferone, D; Clemmons, DR; Greenman, Y; Marazuela, M; Mortini, P; Strasburger, CJ; Giustina, A. Consensus on acromegaly therapeutic outcomes: an update. Nat Rev Endocrinol 2025, 21(11), 718–737. [Google Scholar] [CrossRef] [PubMed]
- Tönjes, A; Würfel, M; Quinkler, M; Knappe, UJ; Honegger, J; Krause-Joppig, N; Bacher, K; Deutschbein, T; Störmann, S; Schopohl, J; Meyhöfer, SM. participants of the German Acromegaly Registry. Pregnancy and acromegaly: clinical outcomes of retrospectively analysed data from the German acromegaly registry. Reprod Biol Endocrinol 2024, 22(1), 48. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jiao, R; Ju, J; Wang, L; Yang, H; Yao, Y; Deng, K; Zhu, H; Duan, L. Safety of pregnancy in acromegaly patients and maternal and infant outcomes after pregnancy: single-center experience from China and review of the literature. BMC Endocr Disord. 2023, 23(1), 104. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Abucham, J; Bronstein, MD; Dias, ML. MANAGEMENT OF ENDOCRINE DISEASE: Acromegaly and pregnancy: a contemporary review. Eur J Endocrinol 2017, 177(1), R1–R12. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).