Submitted:
30 January 2026
Posted:
30 January 2026
You are already at the latest version
Abstract
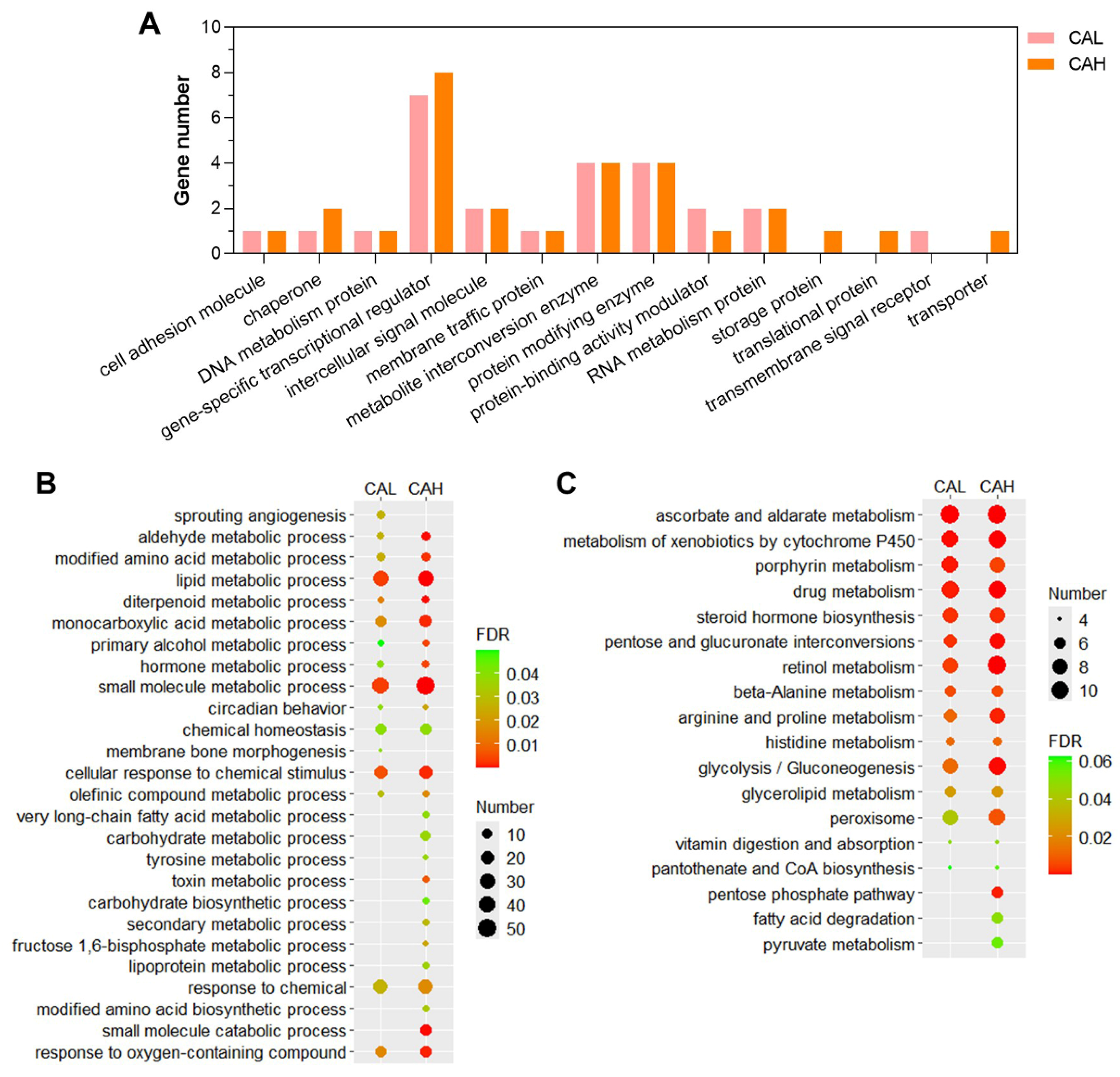
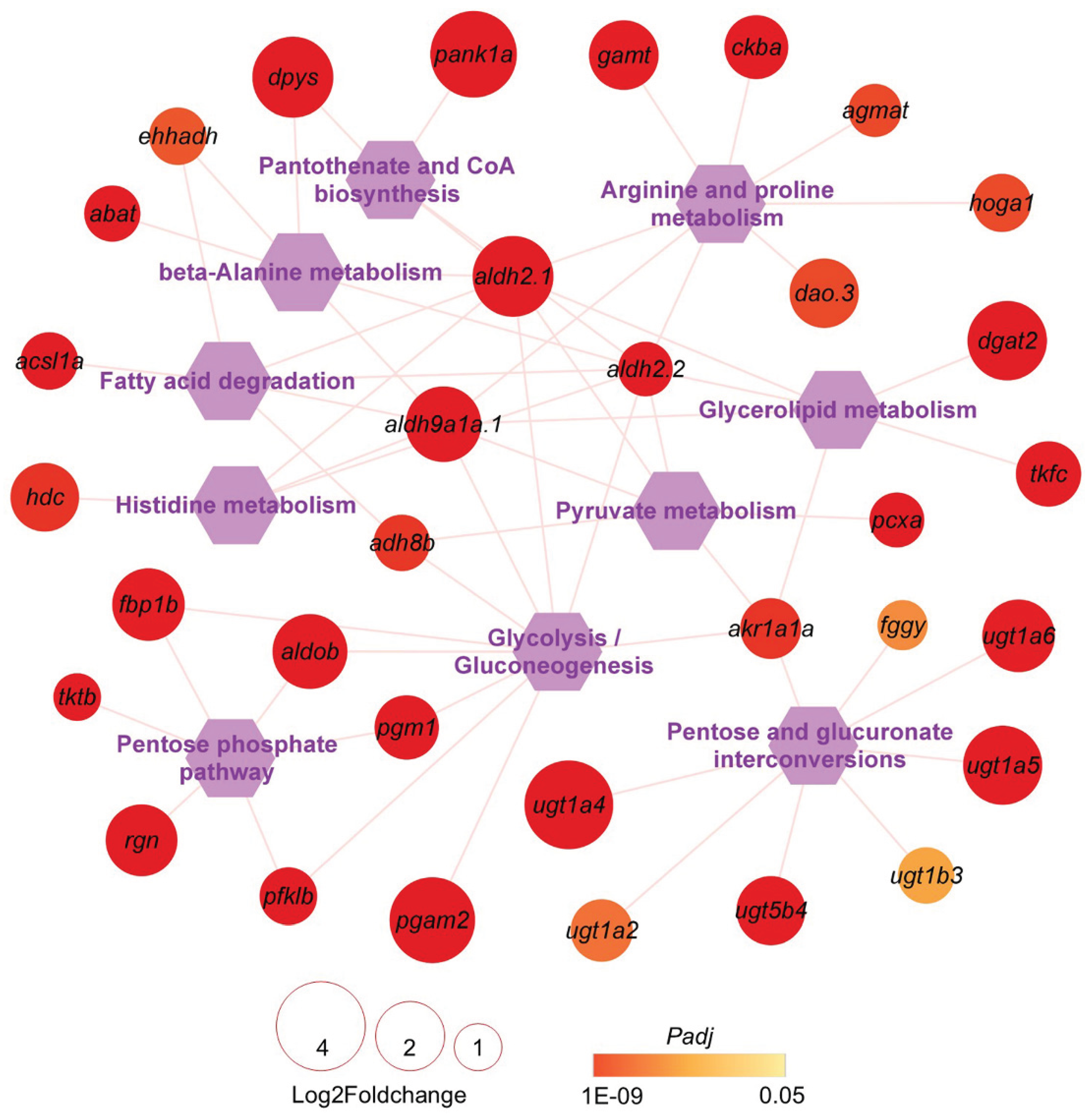
The global prevalence of obesity continues to rise, posing serious risks to human health largely because obesity itself leads to metabolic disorders of carbohydrate and lipids. Currently, effective and healthy interventions for lowering blood glucose, reducing blood lipids, and promoting weight loss remain limited due to the complexity of obesity development. Lactobacillus plantarum (GDMCC 1.140) was shown to promote catabolic process and reduce hepatic lipid accumulation in largemouth bass fed with high-starch feed (HSF) in our previous study; however, molecular mechanisms underlying the function of this probiotic remain unclear. Here, we evaluated the effects of L-carnosine, one of metabolites from Lactobacillus plantarum, on carbohydrate and lipid metabolisms in an obesity model of zebrafish, which was induced by HSF. Histopathological analyses of livers from different groups indicated that dietary supplement with L-carnosine can alleviate hepatic impairment and reduce lipid accumulation in the hepatocytes of obese zebrafish. Transcriptomic analyses revealed that L-carnosine supplementation can reverse the expression of about 70 HSF-induced genes, mainly gene-specific transcription regulators and metabolite interconversion enzymes. Furthermore, about 250 HSF-inhibited genes were found to be up-regulated by L-carnosine toward the levels in normal-starch feed (NSF) zebrafish. These L-carnosine-targeted and HSF-inhibited genes are highly enriched in GO terms such as lipid metabolic process, small molecule metabolic process, and cellular response to chemical stimulus, followed by monocarboxylic acid metabolic process, modified amino acid metabolic process and aldehyde metabolic process, and in KEGG pathways of carbohydrate, lipid, and amino acid metabolisms, such as pentose and glucuronate interconversions, glycolysis/gluconeogenesis, glycerolipid metabolism, pentose phosphate pathways, fatty acid degradation, beta-alanine metabolism and arginine and proline metabolism. These findings provide functional and molecular evidence that L-carnosine can ameliorate HSF-induced disorders of carbohydrate and lipid metabolisms.
Keywords:
1. Introduction
2. Results
2.1. L-carnosine Supplementation Alleviates Hepatic Steatosis in Zebrafish
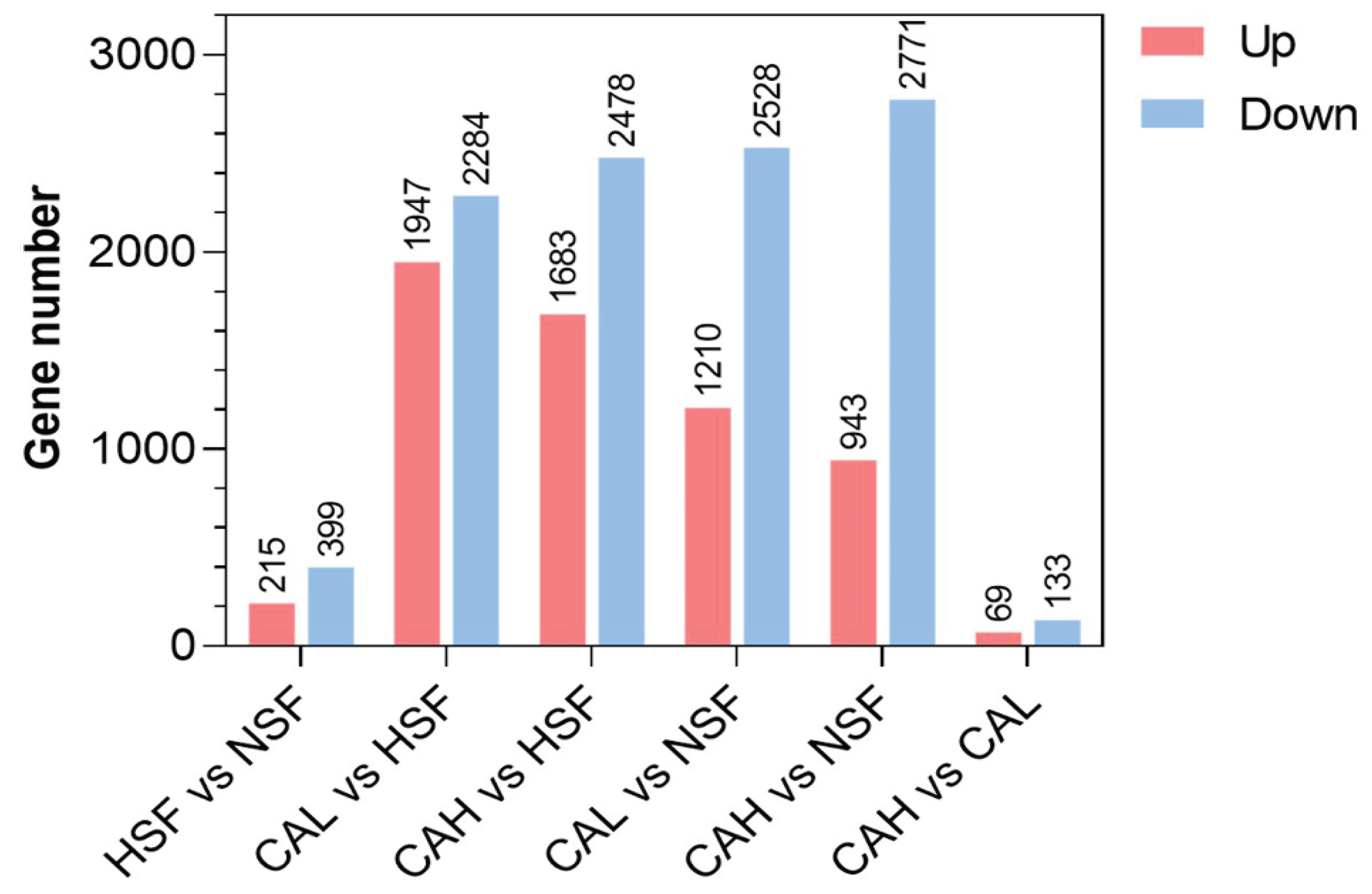
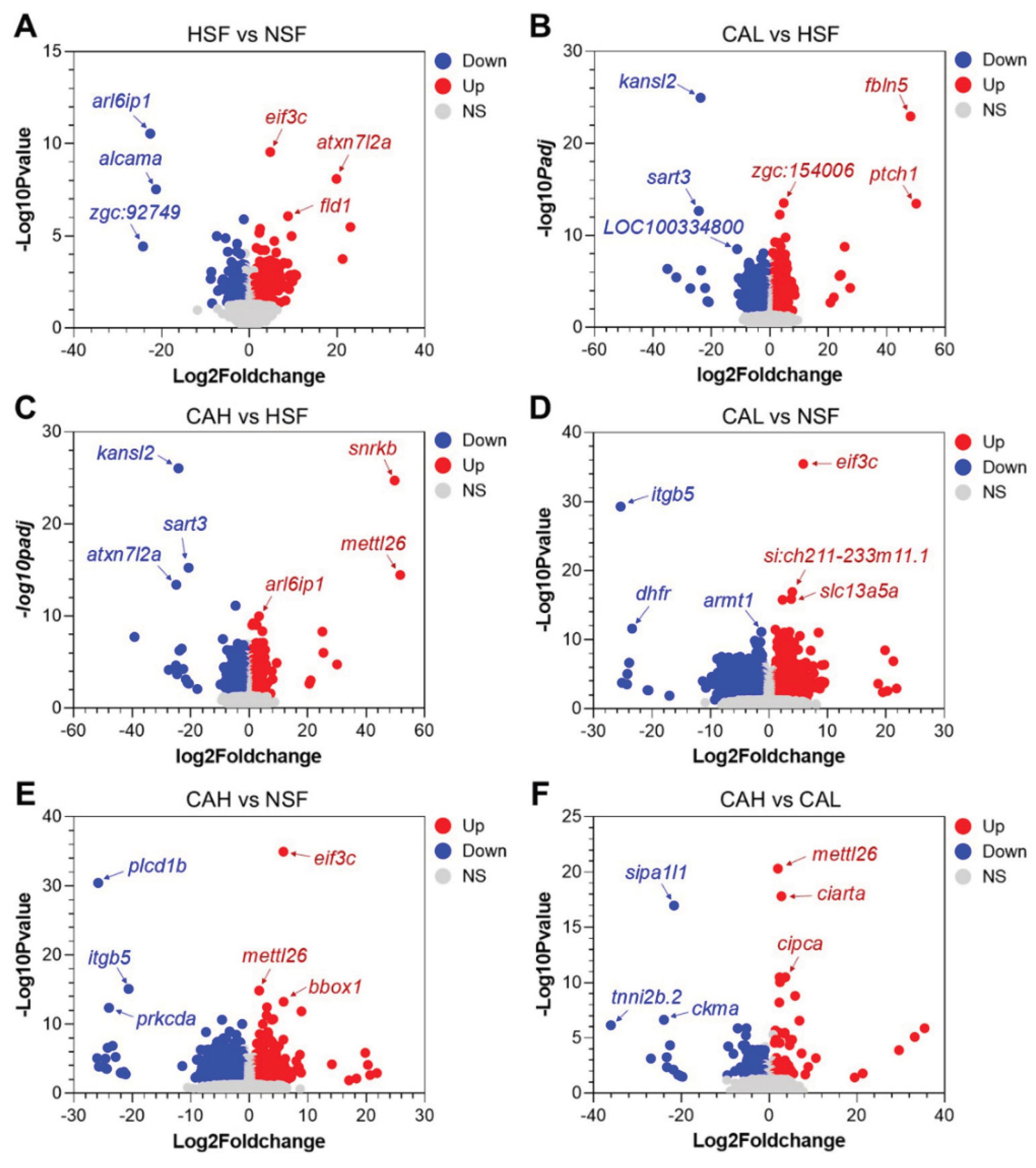
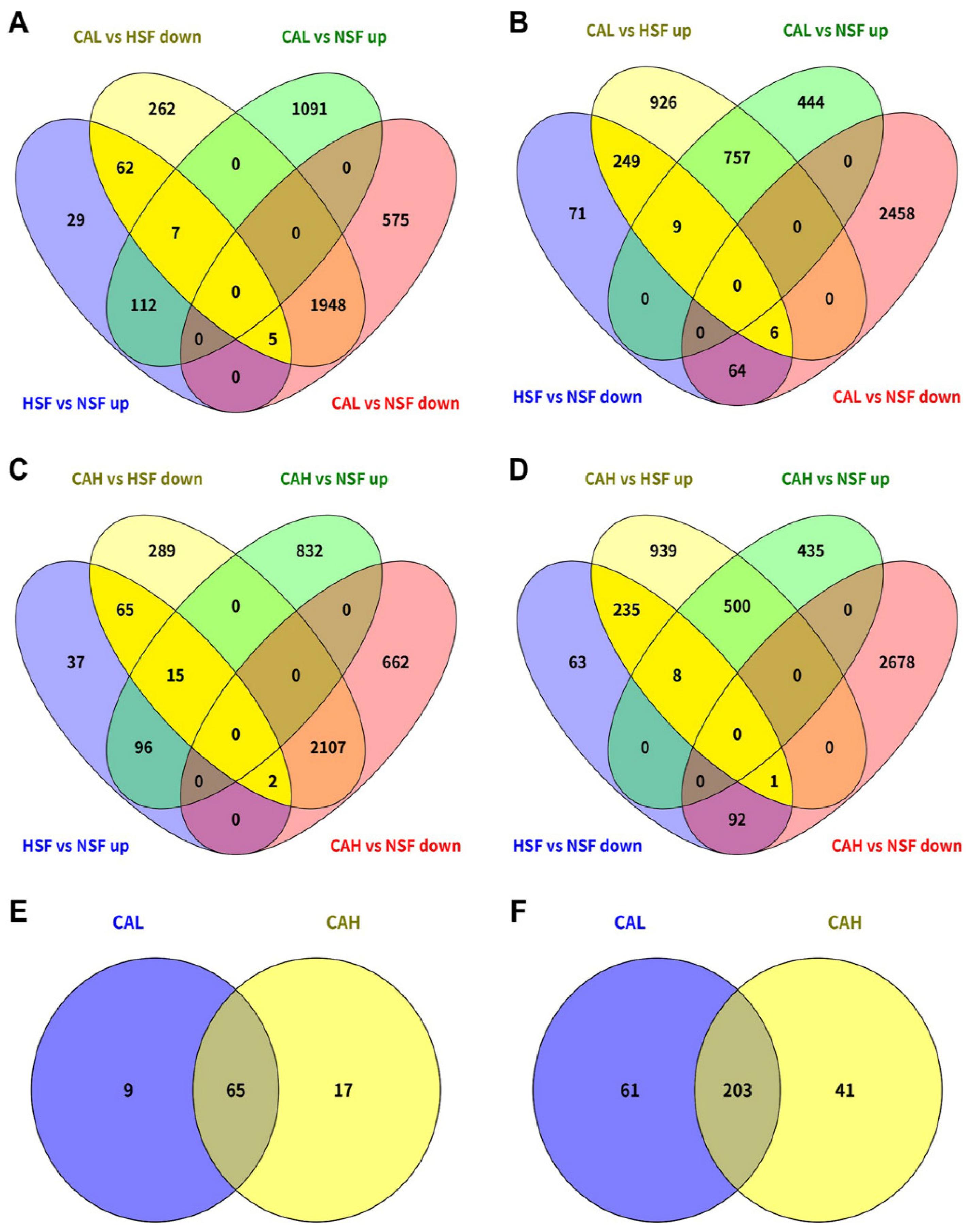
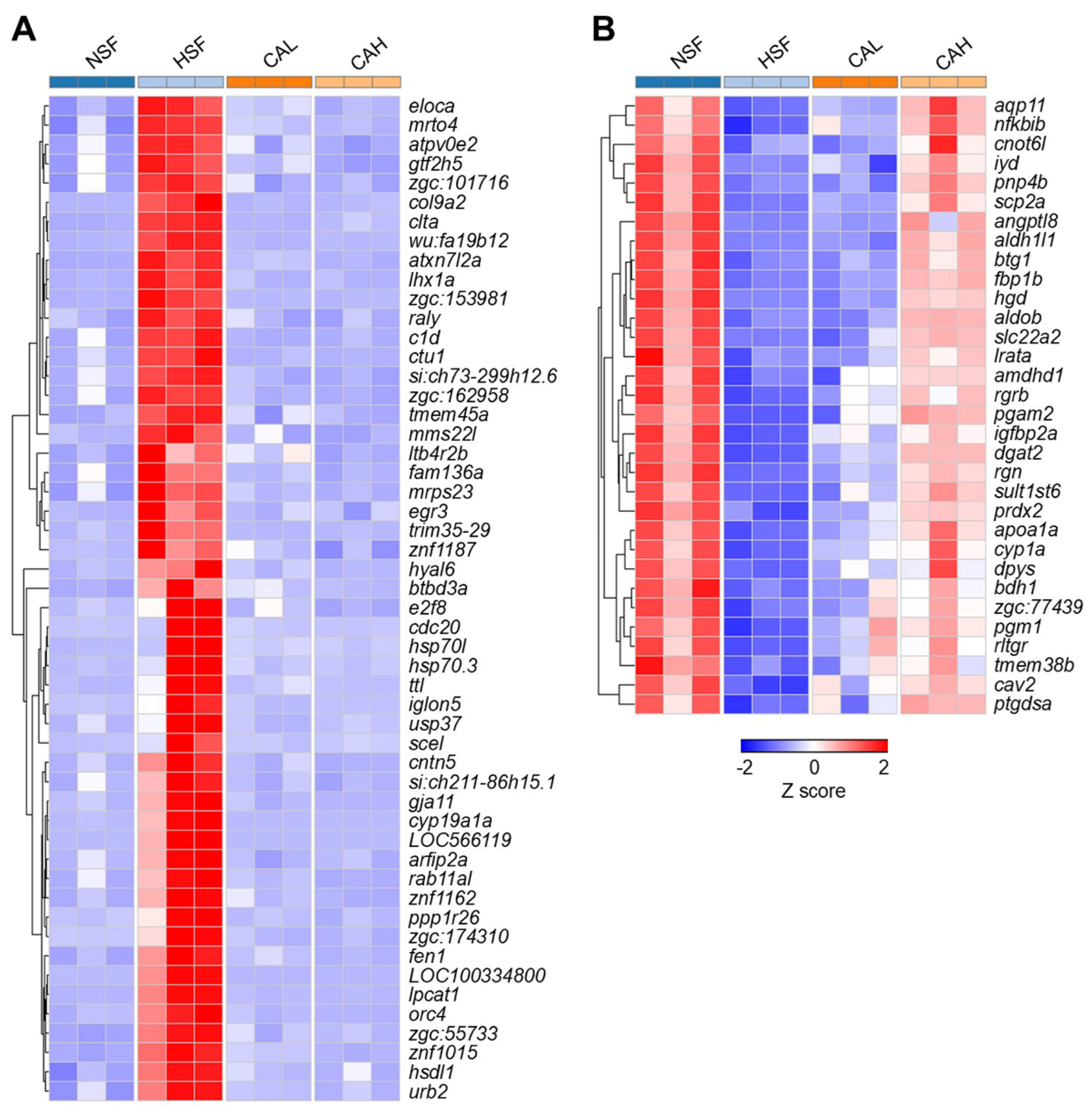
2.2. Effects of L-carnosine Treatments on Gene Expression in Zebrafish Liver
2.3. Identification of Genes Underlying the Protective Effects of L-carnosine in Zebrafish Liver
2.4. Functional Enrichments of the L-carnosine Targeted Genes
2.5. Energy Metabolism Pathways and Associated the Genes Targeted by L-carnosine
3. Discussion
4. Materials and Methods
4.1. Ethics Statement
4.2. Experimental Diet Formulation and Preparation
4.3. Establishment of Zebrafish Model with Glucose and Lipid Metabolism Disorders
4.4. Anesthesia and Sampling
4.5. Histopathology Analysis
4.6. RNA Sequencing and Data Analysis
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shafiei-Jahani, P.; Hurrell, B. P.; Galle-Treger, L.; Helou, D. G.; Howard, E.; Painter, J.; Lo, R.; Lewis, G.; Soroosh, P.; Akbari, O. DR3 stimulation of adipose resident ILC2s ameliorates type 2 diabetes mellitus. Nature Communications 2020, 11, 4718. [Google Scholar] [CrossRef]
- Linton, M. F.; Yancey, P. G.; Leuthner, Z. M.; Brown, J. D. The FoxOs are in the ApoM house. Journal of Clinical Investigation 2022, 132, e158471. [Google Scholar] [CrossRef]
- Zeybel, M.; Arif, M.; Li, X.; Altay, O.; Yang, H.; Shi, M.; Akyildiz, M.; Saglam, B.; Gonenli, M. G.; Yigit, B.; Ulukan, B.; Ural, D.; Shoaie, S.; Turkez, H.; Nielsen, J.; Zhang, C.; Uhlén, M.; Borén, J.; Mardinoglu, A. Multiomics Analysis Reveals the Impact of Microbiota on Host Metabolism in Hepatic Steatosis. Advanced Science 2022, 9, e2104373. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Wang, L.; Liu, Z.; Long, K.; Kong, M.; Ye, D.; Chen, X.; Wang, K.; Wu, K. K. L.; Fan, M.; Song, E.; Wang, C.; Hoo, R. L. C.; Hui, X.; Hallenborg, P.; Piao, H.; Xu, A.; Cheng, K. K. Y. Hepatic MDM2 Causes Metabolic Associated Fatty Liver Disease by Blocking Triglyceride-VLDL Secretion via ApoB Degradation. Advanced Science 2022, 9, 2200742. [Google Scholar] [CrossRef]
- Zhang, Y.; Yan, T.; Wang, T.; Liu, X.; Hamada, K.; Sun, D.; Sun, Y.; Yang, Y.; Wang, J.; Takahashi, S.; Wang, Q.; Krausz, K. W.; Jiang, C.; Xie, C.; Yang, X.; Gonzalez, F. J. Crosstalk between CYP2E1 and PPARα substrates and agonists modulate adipose browning and obesity. Acta Pharmaceutica Sinica B 2022, 12, 2224–2238. [Google Scholar] [CrossRef] [PubMed]
- Gregg, E.; Basto-Abreu, A.; Braverman-Bronstein, A.; Camacho-García-Formentí, D.; Zepeda-Tello, R.; Popkin, B. M.; Rivera-Dommarco, J.; Hernández-Ávila, M.; Barrientos-Gutiérrez, T. Expected changes in obesity after reformulation to reduce added sugars in beverages: A modeling study. PLOS Medicine 2018, 15, e100266, Erratum in PLOS Medicine2019, 16, e1002743.. [Google Scholar] [CrossRef] [PubMed]
- Ge, X.; He, X.; Liu, J.; Zeng, F.; Chen, L.; Xu, W.; Shao, R.; Huang, Y.; Farag, M. A.; Capanoglu, E.; El-Seedi, H. R.; Zhao, C.; Liu, B. Amelioration of type 2 diabetes by the novel 6, 8-guanidyl luteolin quinone-chromium coordination via biochemical mechanisms and gut microbiota interaction. Journal of Advanced Research 2023, 46, 173–188. [Google Scholar] [CrossRef]
- Zhang, Y.; Liang, X.-f.; He, S.; Wang, J.; Li, L.; Zhang, Z.; Li, J.; Chen, X.; Li, L.; Alam, M. S. Metabolic responses of Chinese perch (Siniperca chuatsi) to different levels of dietary carbohydrate. Fish Physiology and Biochemistry 2021, 47, 1449–1465. [Google Scholar] [CrossRef]
- Shi, Y.; Zhong, L.; Fan, Y.; Zhang, J.; Zhong, H.; Liu, X.; Shao, C.; Hu, Y. The Protective Effect of Mulberry Leaf Flavonoids on High-Carbohydrate-Induced Liver Oxidative Stress, Inflammatory Response and Intestinal Microbiota Disturbance in Monopterus albus. Antioxidants 2022, 11, 976. [Google Scholar] [CrossRef]
- Liu, H.; Pan, L.; Shen, J.; Tan, B.; Dong, X.; Yang, Q.; Chi, S.; Zhang, S. Effects of Carbohydrase Supplementation on Growth Performance, Intestinal Digestive Enzymes and Flora, Glucose Metabolism Enzymes, and glut2 Gene Expression of Hybrid Grouper (Epinephelus fuscoguttatus♀ × E. lanceolatus♂) Fed Different CHO/L Ratio Diets. Metabolites 2023, 13, 98. [Google Scholar] [CrossRef]
- Abdelsalam, M.; Abdel-Gaber, R.; Mahmoud, M. A.; Mahdy, O. A.; Khafaga, N. I. M.; Warda, M. Morphological, molecular and pathological appraisal of Callitetrarhynchus gracilis plerocerci (Lacistorhynchidae) infecting Atlantic little tunny (Euthynnusalletteratus) in Southeastern Mediterranean. Journal of Advanced Research 2016, 7, 317–326. [Google Scholar] [CrossRef] [PubMed]
- Jia, R.; Cao, L.-P.; Du, J.-L.; He, Q.; Gu, Z.-Y.; Jeney, G.; Xu, P.; Yin, G.-J. Effects of High-Fat Diet on Steatosis, Endoplasmic Reticulum Stress and Autophagy in Liver of Tilapia (Oreochromis niloticus). Frontiers in Marine Science 2020, 7. [Google Scholar] [CrossRef]
- Zhang, X.-J.; Zhou, L.; Lu, W.-J.; Du, W.-X.; Mi, X.-Y.; Li, Z.; Li, X.-Y.; Wang, Z.-W.; Wang, Y.; Duan, M.; Gui, J.-F. Comparative transcriptomic analysis reveals an association of gibel carp fatty liver with ferroptosis pathway. BMC Genomics 2021, 22, 328. [Google Scholar] [CrossRef]
- Deng, Y.; Zhang, W.; Yang, Z.; Kong, Q.; Liu, P.; Liao, H.; Cui, Z.; Tang, H. Dietary Lactobacillus plantarum can alleviate high starch diet-induced liver lipid deposition, tissue damage and oxidative stress in largemouth bass (Micropterus salmoides). Aquaculture Reports 2024, 35, 101955. [Google Scholar] [CrossRef]
- Ma hootchi, E.; Kleppe, R; Winge, I; Hegvik, T-A.; Megias-perez, R.; Totland, C.; Mogavero, F.; Baumann, A.; Glennon, J-C.; Miletic, H.; Kursula, K.; Haavik, J. GADL1 is a multifunctional decarboxylase with tissue-specific roles in β-alanine and carnosine production. Science Advances 2020, 6, eabb3713. [Google Scholar] [CrossRef] [PubMed]
- Boldyrev, A. A.; Aldini, G.; Derave, W. Physiology and Pathophysiology of Carnosine. Physiological Reviews 2013, 93, 1803–1845. [Google Scholar] [CrossRef]
- Matthews, J. J.; Dolan, E.; Swinton, P. A.; Santos, L.; Artioli, G. G.; Turner, M. D.; Elliott-Sale, K. J.; Sale, C. The effect of carnosine or β-alanine supplementation on markers of glycaemic control and insulin resistance in human and animal studies: a protocol for a systematic review and meta-analysis. Systematic Reviews 2020, 9, 2216–2231. [Google Scholar] [CrossRef]
- Kawahara, M.; Tanaka, K.-i.; Kato-Negishi, M. Zinc, Carnosine, and Neurodegenerative Diseases. Nutrients 2018, 10, 147. [Google Scholar] [CrossRef]
- Li, X.; Yang, K.; Gao, S.; Zhao, J.; Liu, G.; Chen, Y.; Lin, H.; Zhao, W.; Hu, Z.; Xu, N. Carnosine Stimulates Macrophage-Mediated Clearance of Senescent Skin Cells Through Activation of the AKT2 Signaling Pathway by CD36 and RAGE. Frontiers in Pharmacology 2020, 11, 593832. [Google Scholar] [CrossRef]
- Jain, S.; Kim, E.-S.; Kim, D.; Burrows, D.; De Felice, M.; Kim, M.; Baek, S.-H.; Ali, A.; Redgrave, J.; Doeppner, T. R.; Gardner, I.; Bae, O.-N.; Majid, A. Comparative Cerebroprotective Potential of d- and l-Carnosine Following Ischemic Stroke in Mice. International Journal of Molecular Sciences 2020, 21, 3053. [Google Scholar] [CrossRef]
- Feng, M.; Luo, J.; Wan, Y.; Zhang, J.; Lu, C.; Wang, M.; Dai, L.; Cao, X.; Yang, X.; Wang, Y. Polystyrene Nanoplastic Exposure Induces Developmental Toxicity by Activating the Oxidative Stress Response and Base Excision Repair Pathway in Zebrafish (Danio rerio). ACS Omega 2022, 7, 32153–32163. [Google Scholar] [CrossRef]
- Sugasawa, T.; Komine, R.; Manevich, L.; Tamai, S.; Takekoshi, K.; Kanki, Y. Gene Expression Profile Provides Novel Insights of Fasting-Refeeding Response in Zebrafish Skeletal Muscle. Nutrients 2022, 14, 2239. [Google Scholar] [CrossRef]
- Ribas, L.; Piferrer, F. The zebrafish (Danio rerio) as a model organism, with emphasis on applications for finfish aquaculture research. Reviews in Aquaculture 2013, 6, 209–240. [Google Scholar] [CrossRef]
- Ulloa, P. E.; Iturra, P.; Neira, R.; Araneda, C. Zebrafish as a model organism for nutrition and growth: towards comparative studies of nutritional genomics applied to aquacultured fishes. Reviews in Fish Biology and Fisheries 2011, 21, 649–666. [Google Scholar] [CrossRef]
- Bellia, F.; Vecchio, G.; Cuzzocrea, S.; Calabrese, V.; Rizzarelli, E. Neuroprotective features of carnosine in oxidative driven diseases. Molecular Aspects of Medicine 2011, 32((4-6)), 258–266. [Google Scholar] [CrossRef] [PubMed]
- Peters, V.; Schmitt, C. P.; Zschocke, J.; Gross, M.-L.; Brismar, K.; Forsberg, E. Carnosine treatment largely prevents alterations of renal carnosine metabolism in diabetic mice. Amino Acids 2011, 42, 2411–2416. [Google Scholar] [CrossRef] [PubMed]
- de Courten, B.; Jakubova, M.; de Courten, M. P. J.; Kukurova, I. J.; Vallova, S.; Krumpolec, P.; Valkovic, L.; Kurdiova, T.; Garzon, D.; Barbaresi, S.; Teede, H. J.; Derave, W.; Krssak, M.; Aldini, G.; Ukropec, J.; Ukropcova, B. Effects of carnosine supplementation on glucose metabolism: Pilot clinical trial. Obesity 2016, 24, 1027–1034. [Google Scholar] [CrossRef] [PubMed]
- Dai, Z.; Lu, X.-Y.; Zhu, W.-L.; Liu, X.-Q.; Li, B.-Y.; Song, L.; Liu, H.-F.; Cai, W.-W.; Deng, Y.-X.; Xu, T.-T.; Wang, Q.; Zhang, S.-J. Carnosine ameliorates age-related dementia via improving mitochondrial dysfunction in SAMP8 mice. Food & Function 2020, 11, 2489–2497. [Google Scholar] [CrossRef]
- Bingül, İ.; Yılmaz, Z.; Aydın, A. F.; Çoban, J.; Doğru-Abbasoğlu, S.; Uysal, M. Antiglycation and anti-oxidant efficiency of carnosine in the plasma and liver of aged rats. Geriatrics & Gerontology International 2017, 17, 2610–2614. [Google Scholar]
- Al-Sawalha, N. A.; Alshogran, O. Y.; Awawdeh, M. S.; Almomani, B. A. The effects of l-Carnosine on development of metabolic syndrome in rats. Life Sciences 2019, 237, 116905. [Google Scholar] [CrossRef]
- Anderson, E. J.; Vistoli, G.; Katunga, L. A.; Funai, K.; Regazzoni, L.; Monroe, T. B.; Gilardoni, E.; Cannizzaro, L.; Colzani, M.; De Maddis, D.; Rossoni, G.; Canevotti, R.; Gagliardi, S.; Carini, M.; Aldini, G. A carnosine analog mitigates metabolic disorders of obesity by reducing carbonyl stress. Journal of Clinical Investigation 2018, 128, 5280–5293. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Zhang, A.-h.; Song, Q.; Fang, H.; Liu, X.-y.; Su, J.; Yang, L.; Yu, M.-d.; Wang, X.-j. Functional metabolomics discover pentose and glucuronate interconversion pathways as promising targets for Yang Huang syndrome treatment with Yinchenhao Tang. RSC Advances 2018, 8, 36831–36839. [Google Scholar] [CrossRef]
- Zhang, C.; Zhong, H.; Chen, H.; Yalimaimaiti, N.; Liang, J.; Duan, J.; Yang, Y.; Zhang, S.; Yadav, V.; Zhou, X.; Wu, X.; Zhang, F.; Hao, J. ‘Sultanina’ leaves increase their trehalose content in response to grapevine brown leaf spot infection by regulating the pentose and glucuronate interchange pathway. Plant Stress 2024, 13, 100524. [Google Scholar] [CrossRef]
- Zhang, T.; Zhang, N.; Xing, J.; Zhang, S.; Chen, Y.; Xu, D.; Gu, J. UDP-glucuronate metabolism controls RIPK1-driven liver damage in nonalcoholic steatohepatitis. Nature Communications 2023, 14, 2715. [Google Scholar] [CrossRef]
- Zeng, W.; Liu, X.; Wu, Y.; Cai, Y.; Li, Z.; Ye, F.; Sun, Y.; Li, F.; Xing, H.; Wang, S. Dysregulated hepatic UDP-glucuronosyltransferases and flavonoids glucuronidation in experimental colitis. Frontiers in Pharmacology 2022, 13, 100524. [Google Scholar] [CrossRef]
- Serra, D.; Mera, P.; Malandrino, M. I.; Mir, J. F.; Herrero, L. Mitochondrial Fatty Acid Oxidation in Obesity. Antioxidants & Redox Signaling 2013, 19, 269–284. [Google Scholar]
- Thomas, A. S.; Sassi, M.; Angelini, R.; Morgan, A. H.; Davies, J. S. Acylation, a Conductor of Ghrelin Function in Brain Health and Disease. Frontiers in Physiology 2022, 13, 831641. [Google Scholar] [CrossRef]
- Dierendonck, X. A. M. H. v.; Vrieling, F.; Smeehuijzen, L.; Deng, L.; Boogaard, J. P.; Croes, C.-A.; Temmerman, L.; Wetzels, S.; Biessen, E.; Kersten, S.; Stienstra, R. Triglyceride breakdown from lipid droplets regulates the inflammatory response in macrophages. Proceedings of the National Academy of Sciences 2022, 199, e2114739119. [Google Scholar] [CrossRef]
- Fletcher, J. A.; Deja, S.; Satapati, S.; Fu, X.; Burgess, S. C.; Browning, J. D. Impaired ketogenesis and increased acetyl-CoA oxidation promote hyperglycemia in human fatty liver. JCI Insight 2019, 5, e127737. [Google Scholar] [CrossRef] [PubMed]
- Abo Alrob, O.; Lopaschuk, Gary D. Role of CoA and acetyl-CoA in regulating cardiac fatty acid and glucose oxidation. Biochemical Society Transactions 2014, 42, 1043–1051. [Google Scholar] [CrossRef] [PubMed]
- Okamura, A.; Koyanagi, S.; Dilxiat, A.; Kusunose, N.; Chen, J. J.; Matsunaga, N.; Shibata, S.; Ohdo, S. Bile Acid-regulated Peroxisome Proliferator-activated Receptor-α (PPARα) Activity Underlies Circadian Expression of Intestinal Peptide Absorption Transporter PepT1/Slc15a1. Journal of Biological Chemistry 2014, 289, 25296–25305. [Google Scholar] [CrossRef] [PubMed]
- Grandini, N. A.; Costa, M. R.; Gregolin, C. S.; Siqueira, J. S.; Vieira, T. A.; Togneri Ferron, A. J.; Francisqueti-Ferron, F. V.; Romualdo, G. R.; Lúcia dos Anjos Ferreira, A.; Aldini, G.; Corrêa, C. R.; Moreto, F. Effects of carnosine supplementation on markers for the pathophysiological development of metabolic dysfunction-associated steatotic liver disease in a diet-induced model. Molecular and Cellular Endocrinology 2024, 582, 112138. [Google Scholar] [CrossRef]
- Seo, Y. S.; Kim, J. H.; Jo, N. Y.; Choi, K. M.; Baik, S. H.; Park, J. J.; Kim, J. S.; Byun, K. S.; Bak, Y. T.; Lee, C. H.; Kim, A.; Yeon, J. E. PPAR agonists treatment is effective in a nonalcoholic fatty liver disease animal model by modulating fatty-acid metabolic enzymes. Journal of Gastroenterology and Hepatology 2007, 23, 102–109, Erratum inJournal of Gastroenterology and Hepatology2009, 24, 932; Erratum in Journal of Gastroenterology and Hepatology 2009, 24, 1310.. [Google Scholar] [CrossRef]
- Mao, Z.; Feng, M.; Li, Z.; Zhou, M.; Xu, L.; Pan, K.; Wang, S.; Su, W.; Zhang, W. ETV5 Regulates Hepatic Fatty Acid Metabolism Through PPAR Signaling Pathway. Diabetes 2021, 70, 214–226. [Google Scholar] [CrossRef]
- Xi, L.; Lu, Q.; Liu, Y.; Gong, Y.; Liu, H.; Jin, J.; Zhang, Z.; Yang, Y.; Zhu, X.; Han, D.; Xie, S.; Xu, H. Study on Carbohydrate Metabolism in Adult Zebrafish (Danio rerio). Aquaculture Nutrition 2023, 2023, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Qiao, H.; Peng, L.; Meng, Y.; Song, G.; Luo, C.; Long, Y. Influence of High Temperature and Ammonia and Nitrite Accumulation on the Physiological, Structural, and Genetic Aspects of the Biology of Largemouth Bass (Micropterus salmoides). Antioxidants (Basel) 2025, 14, 495. [Google Scholar] [CrossRef]
- Chen, S.; Zhou, Y.; Chen, Y.; Gu, J. fastp: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 2018, 34, i884–i890. [Google Scholar] [CrossRef] [PubMed]
- Patro, R.; Duggal, G.; Love, M. I.; Irizarry, R. A.; Kingsford, C. Salmon provides fast and bias-aware quantification of transcript expression. Nat Methods 2017, 14, 417–419. [Google Scholar] [CrossRef]
- Love, M. I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 2014, 15, 550. [Google Scholar] [CrossRef]
- Maere, S.; Heymans, K.; Kuiper, M. BiNGO: a Cytoscape plugin to assess overrepresentation of gene ontology categories in biological networks. Bioinformatics 2005, 21, 3448–3449. [Google Scholar] [CrossRef]
- Bindea, G.; Mlecnik, B.; Hackl, H.; Charoentong, P.; Tosolini, M.; Kirilovsky, A.; Fridman, W. H.; Pages, F.; Trajanoski, Z.; Galon, J. ClueGO: a Cytoscape plug-in to decipher functionally grouped gene ontology and pathway annotation networks. Bioinformatics 2009, 25, 1091–1093. [Google Scholar] [CrossRef] [PubMed]
- Supek, F.; Bosnjak, M.; Skunca, N.; Smuc, T. REVIGO summarizes and visualizes long lists of gene ontology terms. PLoS One 2011, 6, e21800. [Google Scholar] [CrossRef] [PubMed]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N. S.; Wang, J. T.; Ramage, D.; Amin, N.; Schwikowski, B.; Ideker, T. Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res 2003, 13, 2498–2504. [Google Scholar] [CrossRef] [PubMed]
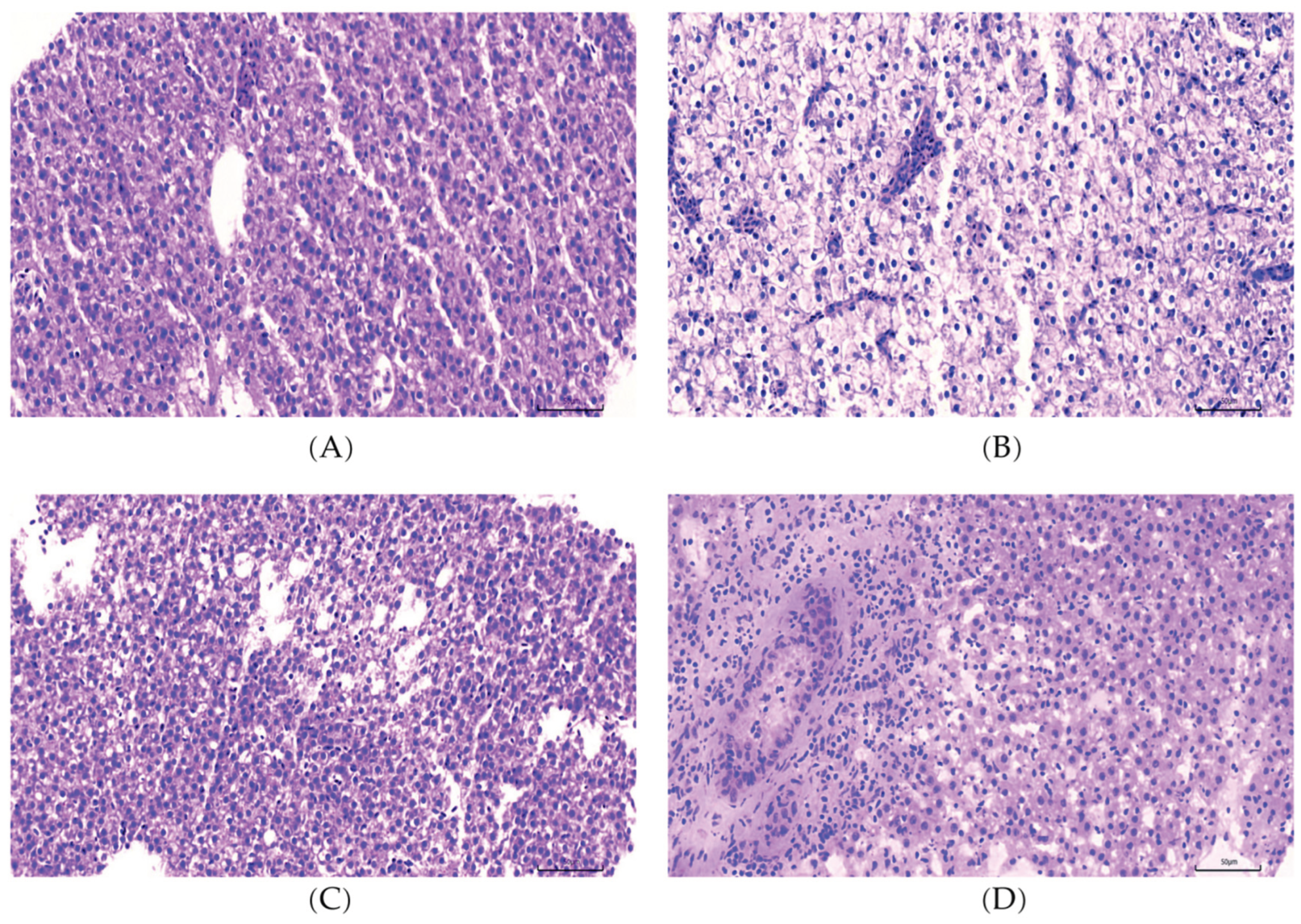
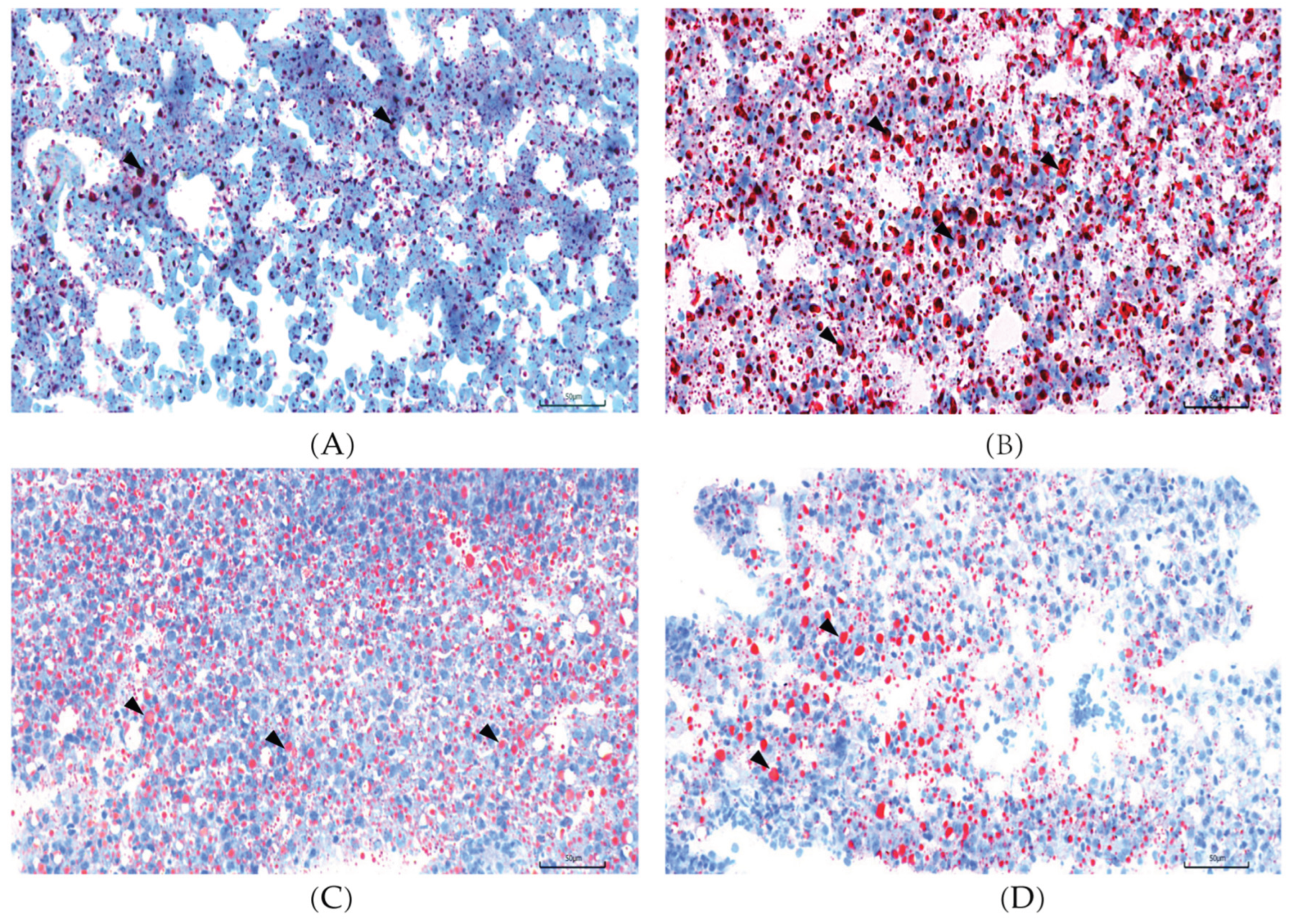
| Items | NSF | HSF | CAL | CAH |
| Body weight (mg) | 340 ± 5.5 | 396 ± 9.2* | 390 ± 12.4* | 370 ± 17.8 |
| Body length (mm) | 336 ± 6.7 | 342 ± 7.3 | 330 ± 8.9 | 338 ± 5.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




