Submitted:
29 January 2026
Posted:
30 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Gut Microbiome Alterations in HFpEF:
1.2. Metabolomic Consequences:
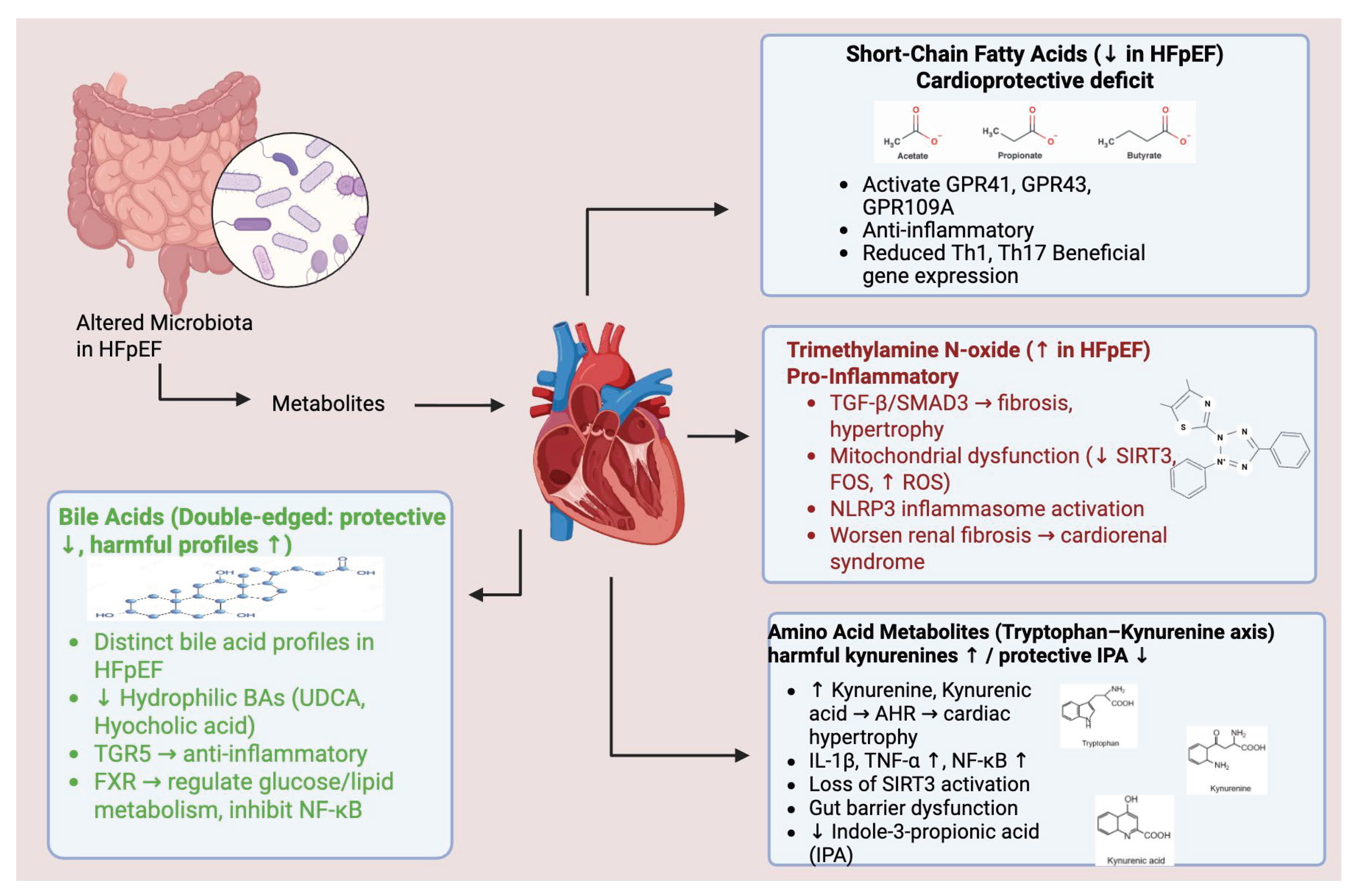
2. Microbiome-Derived Metabolites: Key Players in HFpEF Pathogenesis
2.1. Short-Chain Fatty Acids: The Cardioprotective Deficit
2.2. Trimethylamine N-Oxide: The Inflammatory Culprit
2.3. Amino Acid Metabolites: The Tryptophan-Kynurenine Axis
2.4. Bile Acids: The Double-Edged Metabolites
3. Endothelial Dysfunction in the Gut-Heart Axis
- TMAO-Induced Endothelial Damage: Trimethylamine N-oxide directly impairs endothelial function through several mechanisms. It enhances endothelial cell apoptosis and inflammatory activation, promotes oxidative stress, and reduces nitric oxide bioavailability. Furthermore, TMAO facilitates foam cell formation, accelerates atherosclerosis, and impairs endothelium-dependent vasodilation, all contributing to endothelial dysfunction. [43,44].
- LPS-Mediated Endothelial Activation: Bacterial lipopolysaccharides, crossing the compromised gut barrier, activate endothelial cells via Toll-like receptor 4 signaling. This activation leads to the upregulation of adhesion molecules (ICAM-1, VCAM-1) and chemokines, increasing vascular permeability and inflammatory cell recruitment. This process ultimately contributes to a pro-thrombotic endothelial phenotype and activates complement and coagulation cascades. [45,46,47].
- SCFA Deficiency Effects: A reduction in beneficial short-chain fatty acid levels also contributes to endothelial dysfunction. This deficiency results in decreased G-protein coupled receptor-mediated protective signaling in endothelial cells, impairs endothelial barrier function leading to increased permeability, and reduces the production of anti-inflammatory mediators. Consequently, it compromises endothelial repair and regeneration mechanisms, exacerbating dysfunction. [48,49].
4. Clinical Implications of Endothelial Dysfunction and Therapeutic Targets
5. Therapeutic Interventions Targeting the Gut-Heart Axis in HFpEF
5.1. Probiotic Interventions: Targeted Microbial Supplementation
5.2. Metabolite-Based Therapies and Precision Medicine Approach
6. Future Directions and Research Opportunities:
6.1. Novel Therapeutic Targets
6.2. Advanced Diagnostic Approaches:
6.3. Therapeutic Innovation:
| Proposed Study (Acronym) | Objective | Proposed Design | Primary Endpoints | Secondary Endpoints | Key Innovation |
|---|---|---|---|---|---|
| Precision Microbiome Medicine for HFpEF | Develop and validate a precision medicine platform for personalized microbiome interventions in HFpEF patients | Multi-center RCT; personalized interventions vs. standard care; n=1,000 | Composite CV outcomes, QoL, exercise capacity | Microbiome restoration, metabolite profiles, inflammatory biomarkers | ML-guided real-time microbiome monitoring for treatment optimization |
| Gut–Heart Metabolite Intervention Trial | Assess efficacy/safety of combined SCFA + IPA replacement vs. placebo in metabolite-deficient HFpEF | Phase II/III double-blind RCT; n=500 | Peak VO2, NT-proBNP, diastolic function | Endothelial function, inflammation, gut barrier integrity | First precision metabolite replacement therapy trial in CVD |
| Early HFpEF Detection Through Microbiome Analysis | Develop microbiome biomarkers for early HFpEF detection in at-risk individuals | Prospective cohort; 5-year follow-up; n=2,000 | Time to HFpEF diagnosis, predictive accuracy | Intermediate phenotypes, cost-effectiveness | First large-scale microbiome-based CV prediction study |
| Microbiome-Targeted Combination Therapy | Evaluate synergy of FMT, probiotics, and metabolite supplementation in refractory HFpEF | Adaptive randomized trial; n=300 | Clinical improvement, HF hospitalization rates, QoL | Microbiome restoration, safety, biomarkers | First systematic combination microbiome therapy trial in HFpEF |
| Pediatric-to-Adult Microbiome Trajectory Study | Determine early-life microbiome influence on adult HFpEF risk | 20-year longitudinal cohort; n=1,000 | Adult CV risk profile, HFpEF incidence | Metabolic health, inflammatory profiles, intervention windows | First lifespan microbiome–cardiovascular link study |
| Environmental Modulation of Gut–Heart Axis | Investigate environmental factors affecting gut-heart axis and HFpEF risk | Multi-cohort; varied exposures; n=5,000 | Environmental associations with microbiome disruption and HFpEF | Mechanistic pathways, reversibility | First comprehensive study of environmental determinants of CV-relevant microbiome changes |
| Artificial Intelligence–Guided Microbiome Therapy | Develop AI algorithms for real-time microbiome therapy optimization | Single-arm pilot; wearable and microbiome monitoring; n=100 | Feasibility, clinical improvement | Algorithm performance, patient acceptability, cost-effectiveness | First AI-driven real-time microbiome therapy approach in CVD |
| Microbiome–Heart Device Integration Study | Test whether microbiome optimization enhances device-based HFpEF therapy efficacy | RCT: device therapy vs. device + microbiome optimization; n=200 | Device efficacy enhancement, clinical outcomes | Inflammatory profiles, device complications, mechanisms | First integration of microbiome therapy with device-based HFpEF treatment |
6.4. Population Health Implications:
7. Conclusion
- (a)
- establishing precision microbiome medicine platforms;
- (b)
- validating combination microbiome therapies;
- (c)
- developing microbiome-based systems for early disease detection;
- (d)
- integrating environmental determinants into treatment planning; and
- (e)
- leveraging artificial intelligence to optimize outcomes.
Funding
Acknowledgments
Conflicts of Interest
References
- Clark, K; Velazquez, EJ. Heart failure with preserved ejection fraction. JAMA 2020, 324(15), 1506. [Google Scholar] [CrossRef] [PubMed]
- Guo, X; Huang, C; Zhang, L; Hu, G; Du, Y; Chen, X; et al. Lymphatic endothelial branched-chain amino acid catabolic defects undermine cardiac lymphatic integrity and drive HFpEF. Circulation 2025, 151(23), 1651–1666. [Google Scholar] [CrossRef] [PubMed]
- Tah, S; Valderrama, M; Afzal, M; Iqbal, J; Farooq, A; Lak, MA; et al. Heart failure with preserved ejection fraction: an evolving understanding. Cureus 2023, 15(7), e46152. [Google Scholar] [CrossRef] [PubMed]
- Lucà, F; Oliva, F; Abrignani, MG; Fusco, SAD; Gori, M; Giubilato, S; et al. Heart failure with preserved ejection fraction: how to deal with this chameleon. J Clin Med. 2024, 13(5), 1375. [Google Scholar] [CrossRef]
- Vaz-Salvador, P; Adão, R; Vasconcelos, I; Leite-Moreira, A; Brás-Silva, C. Heart failure with preserved ejection fraction: a pharmacotherapeutic update. Cardiovasc Drugs Ther. 2022, 37(4), 815–832. [Google Scholar] [CrossRef]
- Boulet, J; Sridhar, VS; Bouabdallaoui, N; Tardif, JC; White, M. Inflammation in heart failure: pathophysiology and therapeutic strategies. Inflamm Res. 2024, 73(5), 709–723. [Google Scholar] [CrossRef]
- Clemenza, F; Citarrella, R; Patti, AM; Rizzo, M. Obesity and HFpEF. J Clin Med. 2022, 11(13), 3858. [Google Scholar] [CrossRef]
- Lin, Y; Fu, S; Yao, Y; Li, Y; Zhao, Y; Luo, L. Heart failure with preserved ejection fraction based on aging and comorbidities. J Transl Med. 2021, 19(1), 291. [Google Scholar] [CrossRef]
- Inácio, JM; Cristo, F; Pinheiro, M; Vasques-Nóvoa, F; Saraiva, F; Nunes, MM; et al. Myocardial RNA sequencing reveals new potential therapeutic targets in heart failure with preserved ejection fraction. Biomedicines 2023, 11(8), 2131. [Google Scholar] [CrossRef]
- Zhou, S; Zhou, X; Zhang, P; et al. The gut microbiota-inflammation-HFpEF axis: deciphering the role of gut microbiota dysregulation in the pathogenesis and management of HFpEF. Front Cell Infect Microbiol. 2025, 15, 1537576. [Google Scholar] [CrossRef]
- Huang, Z; Mei, X; Jiang, Y; Chen, T; Zhou, Y. Gut microbiota in heart failure patients with preserved ejection fraction (GUMPTION Study). Front Cardiovasc Med. 2022, 8, 803744. [Google Scholar] [CrossRef] [PubMed]
- Trøseid, M; Andersen, GØ; Broch, K; Hov, JR. The gut microbiome in coronary artery disease and heart failure: current knowledge and future directions. EBioMedicine 2020, 52, 102649. [Google Scholar] [CrossRef] [PubMed]
- Andreadou, I; Ghigo, A; Nikolaou, PE; et al. Immunometabolism in heart failure. Nat Rev Cardiol. Published online. 22 June 2025. [CrossRef]
- Shen, S; Tian, B; Zhang, H; Wang, YC; Li, T; Cao, Y. Heart failure and gut microbiota: what is cause and effect? Research (Wash D C) 2025, 8, 0610. [Google Scholar] [CrossRef] [PubMed]
- Beale, A; O’Donnell, J; Nakai, M; Nanayakkara, S; Vizi, D; Carter, K; et al. The gut microbiome of heart failure with preserved ejection fraction. J Am Heart Assoc. 2021, 10(13), e020654. [Google Scholar] [CrossRef]
- Perticone, M; Gigliotti, S; Shehaj, E; Maio, R; Suraci, E; Miceli, S; et al. Gut permeability and immune-mediated inflammation in heart failure. Biomedicines 2024, 12(6), 1217. [Google Scholar] [CrossRef]
- Yukino-Iwashita, M; Nagatomo, Y; Kawai, A; Taruoka, A; Yumita, Y; Kagami, K; et al. Short-chain fatty acids in gut-heart axis: their role in the pathology of heart failure. J Pers Med. 2022, 12(11), 1805. [Google Scholar] [CrossRef]
- Liu, J; Ge, P; Luo, Y; Sun, Z; Luo, X; Li, H; et al. Decoding TMAO in the gut-organ axis: from biomarkers and cell death mechanisms to therapeutic horizons. Drug Des Devel Ther. 2025, 19, 3363–3393. [Google Scholar] [CrossRef]
- Zhou, XD; Xu, CF; Chen, QF; Shapiro, MD; Lip, GYH; Chen, LL; et al. Serum bile acid profiles are associated with heart failure with preserved ejection fraction in patients with metabolic dysfunction-associated fatty liver disease: an exploratory study. Diabetes Obes Metab. 2024, 26(9), 3684–3695. [Google Scholar] [CrossRef]
- Palm, CL; Nijholt, KT; Bakker, BM; Westenbrink, BD. Short-chain fatty acids in the metabolism of heart failure: rethinking the fat stigma. Front Cardiovasc Med. 2022, 9, 915102. [Google Scholar] [CrossRef]
- Hatahet, J; Cook, TM; Bonomo, RR; Elshareif, N; Gavini, CK; White, CR; et al. Fecal microbiome transplantation and tributyrin improves early cardiac dysfunction and modifies the BCAA metabolic pathway in a diet-induced pre-HFpEF mouse model. Front Cardiovasc Med. 2023, 10, 1105581. [Google Scholar] [CrossRef]
- Dong, Z; Zheng, S; Shen, Z; Luo, Y; Hai, X. Trimethylamine N-oxide is associated with heart failure risk in patients with preserved ejection fraction. Lab Med. 2021, 52(4), 346–351. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Giménez, R; Ahmed-Khodja, W; Molina-Castro, Y; Peiró, ÓM; Bonet, G; Carrasquer, A; et al. Gut microbiota-derived metabolites and cardiovascular disease risk: a systematic review of prospective cohort studies. Nutrients 2022, 14(13), 2654. [Google Scholar] [CrossRef] [PubMed]
- Roncal, C; Martínez-Aguilar, E; Orbe, J; Ravassa, S; Fernández-Montero, A; Saenz-Pipaon, G; et al. Trimethylamine N-oxide predicts cardiovascular mortality in peripheral artery disease. Sci Rep. 2019, 9(1), 15580. [Google Scholar] [CrossRef] [PubMed]
- Almer, G; Semeraro, MD; Meinitzer, A; Enko, D; Blanco, GR; Gallè, B; et al. Impact of long-term high dietary fat intake and regular exercise on serum TMAO and microbiome composition in female rats. Nutr Healthy Aging 2023, 8(1), 157–170. [Google Scholar] [CrossRef]
- Hsu, C; Lu, P; Lo, M; Lin, I; Chang-Chien, G; Lin, S; et al. Gut microbiota-dependent trimethylamine N-oxide pathway associated with cardiovascular risk in children with early-stage chronic kidney disease. Int J Mol Sci. 2018, 19(12), 3699. [Google Scholar] [CrossRef]
- Liu, L; Lin, Y; Zhuang, J; Ren, J; Jiang, X; Chen, M; et al. Analysis of serum metabolite profiles in syphilis patients by untargeted metabolomics. J Eur Acad Dermatol Venereol. 2019, 33(7), 1378–1385. [Google Scholar] [CrossRef]
- James, K; Gertz, E; Kirschke, CP; Allayee, H; Huang, L; Kable, ME; et al. Trimethylamine N-oxide response to a mixed macronutrient tolerance test in a cohort of healthy United States adults. Int J Mol Sci. 2023, 24(3), 2074. [Google Scholar] [CrossRef]
- Wang, Q; Sun, Y; Zhou, T; Jiang, C; A, L; Xu, W. Gut microbiota-dependent trimethylamine N-oxide pathway contributes to the bidirectional relationship between intestinal inflammation and periodontitis. Front Cell Infect Microbiol. 2022, 12, 1125463. [Google Scholar] [CrossRef]
- Dong, Z; Liang, Z; Guo, M; Hu, S; Shen, Z; Hai, X. The association between plasma levels of trimethylamine N-oxide and the risk of coronary heart disease in Chinese patients with or without type 2 diabetes mellitus. Dis Markers 2018, 2018, 1578320. [Google Scholar] [CrossRef]
- Andraos, S; Jones, B; Lange, K; Clifford, S; Thorstensen, EB; Kerr, JA; et al. Trimethylamine N-oxide (TMAO) is not associated with cardiometabolic phenotypes and inflammatory markers in children and adults. Curr Dev Nutr. 2021, 5(1), nzaa179. [Google Scholar] [CrossRef]
- Hahn, VS; Petucci, C; Kim, M; Bedi, K; Wang, H; Mishra, S; et al. Myocardial metabolomics of human heart failure with preserved ejection fraction. Circulation 2023, 147(15), 1147–1161. [Google Scholar] [CrossRef]
- Benítez, TM; Vanderwoude, E; Han, Y; Byun, J; Konje, VC; Gillespie, BW; et al. Kynurenine pathway metabolites predict subclinical atherosclerotic disease and new cardiovascular events in chronic kidney disease. Clin Kidney J. 2022, 15(10), 1952–1965. [Google Scholar] [CrossRef] [PubMed]
- Hakan, H; Kurhan, F; Akbay, Hİ. Predictive value of kynurenine pathway metabolites in the severity of patients with obsessive-compulsive disorder. Psychiatry Clin Neurosci. 2025, 79(2), 13819. [Google Scholar] [CrossRef]
- Lewkowicz, J; Tankiewicz-Kwedlo, A; Pawlak, D; Kiluk, M; Lagoda, K; Kowalska, I. Kynurenines in heart failure with preserved ejection fraction: an influence of type 2 diabetes. Res Square [Preprint] 2024. [Google Scholar] [CrossRef] [PubMed]
- Regan, JA; Truby, LK; Tahir, UA; Katz, DH; Nguyen, M; Kwee, LC; et al. Protein biomarkers of cardiac remodeling and inflammation associated with HFpEF and incident events. Sci Rep. 2022, 12(1), 20488. [Google Scholar] [CrossRef]
- Tian, Y; Cai, J; Gui, W; Nichols, RG; Koo, I; Zhang, J; et al. Berberine directly affects the gut microbiota to promote intestinal farnesoid X receptor activation. Drug Metab Dispos. 2019, 47(2), 86–93. [Google Scholar] [CrossRef]
- Pathak, P; Xie, C; Nichols, RG; Ferrell, JM; Boehme, S; Krausz, KW; et al. Intestine farnesoid X receptor agonist and the gut microbiota activate G-protein bile acid receptor-1 signaling to improve metabolism. Hepatology 2018, 68(4), 1574–1588. [Google Scholar] [CrossRef]
- Kim, H; Fang, S. Crosstalk between FXR and TGR5 controls glucagon-like peptide 1 secretion to maintain glycemic homeostasis. Lab Anim Res. 2018, 34(4), 140–147. [Google Scholar] [CrossRef]
- Li, Y; Hou, H; Wang, X; Dai, X; Zhang, W; Tang, Q; et al. Diammonium glycyrrhizinate ameliorates obesity through modulation of gut microbiota-conjugated BAS-FXR signaling. Front Pharmacol. 2021, 12, 796590. [Google Scholar] [CrossRef]
- Ding, L; Zhang, E; Yang, Q; Jin, L; Sousa, KM; Dong, B; et al. Vertical sleeve gastrectomy confers metabolic improvements by reducing intestinal bile acids and lipid absorption in mice. Proc Natl Acad Sci U S A 2021, 118(6), e2019388118. [Google Scholar] [CrossRef]
- Zhang, C; Fang, R; Lu, X; Zhang, Y; Yang, M; Su, Y; et al. Lactobacillus reuteri J1 prevents obesity by altering the gut microbiota and regulating bile acid metabolism in obese mice. Food Funct. 2022, 13(12), 6688–6701. [Google Scholar] [CrossRef]
- Lee, S; Han, A; Kim, B; Sung, M; Hong, S. Lactococcus lactis-fermented spinach juice suppresses LPS-induced expression of adhesion molecules and inflammatory cytokines through the NF-κB pathway in HUVECs. Exp Ther Med. 2022, 23(6), 393. [Google Scholar] [CrossRef]
- Kim, D; Unenkhuu, B; Kim, G; Kim, S; Kim, H. Cynarin attenuates LPS-induced endothelial inflammation via upregulation of the negative regulator MKP-3. Anim Cells Syst (Seoul) 2022, 26(3), 119–128. [Google Scholar] [CrossRef] [PubMed]
- Gram, A; Kowalewski, M. Molecular mechanisms of lipopolysaccharide (LPS)-induced inflammation in an immortalized ovine luteal endothelial cell line (OLENDO). Vet Sci. 2022, 9(3), 99. [Google Scholar] [CrossRef] [PubMed]
- Tsioti, I; Steiner, B; Escher, P; Zinkernagel, M; Benz, P; Kokona, D. Endothelial toll-like receptor 4 is required for microglia activation in the murine retina after systemic lipopolysaccharide exposure. J Neuroinflammation 2023, 20(1), 162. [Google Scholar] [CrossRef] [PubMed]
- Cai, G; Yang, Z; Guo, D; Hu, C; Yan, M; Yan, J. Macrophages enhance lipopolysaccharide-induced apoptosis via ANG1 and NF-κB pathways in human umbilical vein endothelial cells. Sci Rep. 2021, 11(1), 3024. [Google Scholar] [CrossRef]
- Kutikhin, A; Shishkova, D; Velikanova, E; Sinitsky, M; Sinitskaya, A; Markova, V. Endothelial dysfunction in the context of blood-brain barrier modeling. J Evol Biochem Physiol. 2022, 58(3), 781–806. [Google Scholar] [CrossRef]
- Wong, E; Xu, F; Joffre, J; Nguyen, N; Wilhelmsen, K; Hellman, J. ERK1/2 has divergent roles in LPS-induced microvascular endothelial cell cytokine production and permeability. Shock. 2020, 55(3), 349–356. [Google Scholar] [CrossRef]
- Robles-Vera, I; Toral, M; Visitación, N; Aguilera-Sánchez, N; Redondo, J; Duarte, J. Protective effects of short-chain fatty acids on endothelial dysfunction induced by angiotensin II. Front Physiol. 2020, 11, 277. [Google Scholar] [CrossRef]
- Oh, J; Vasquez, R; Kim, S; Hwang, I; Song, J; Park, J; et al. Multispecies probiotics alter fecal short-chain fatty acids and lactate levels in weaned pigs by modulating gut microbiota. J Anim Sci Technol. 2021, 63(5), 1142–1158. [Google Scholar] [CrossRef]
- Colombo, A; Sadler, R; Llovera, G; Singh, V; Roth, S; Heindl, S; et al. Microbiota-derived short chain fatty acids modulate microglia and promote Aβ plaque deposition. Elife 2021, 10, e59826. [Google Scholar] [CrossRef]
- Xu, X; Jin, H; Li, X; Yan, C; Zhang, Q; Yu, X; et al. Fecal microbiota transplantation regulates blood pressure by altering gut microbiota composition and intestinal mucosal barrier function in spontaneously hypertensive rats. Res Square [Preprint] 2024. [Google Scholar] [CrossRef]
- Paramsothy, S; Nielsen, S; Kamm, MA; Deshpande, N; Faith, JJ; Clemente, JC; et al. Specific bacteria and metabolites associated with response to fecal microbiota transplantation in patients with ulcerative colitis. Gastroenterology 2019, 156(5), 1440–1454.e2. [Google Scholar] [CrossRef] [PubMed]
- Guirro, M; Costa, A; Gual-Grau, A; Herrero, P; Torrell-Galceran, H; Canela-Canela, N; et al. Effects from diet-induced gut microbiota dysbiosis and obesity can be ameliorated by fecal microbiota transplantation: a multiomics approach. PLoS One 2019, 14(9), e0218143. [Google Scholar] [CrossRef] [PubMed]
- Huang, X; Zhang, Y; Huang, J; Gao, W; Xie, Y; Zeng, C; et al. The effect of FMT and vitamin C on immunity-related genes in antibiotic-induced dysbiosis in mice. PeerJ. 2023, 11, e15356. [Google Scholar] [CrossRef] [PubMed]
- Cammarota, G; Ianiro, G; Kelly, C; Mullish, BH; Allegretti, JR; Kassam, Z; et al. International consensus conference on stool banking for faecal microbiota transplantation in clinical practice. Gut 2019, 68(12), 2111–2121. [Google Scholar] [CrossRef]
- Sun, L; Shang, B; Lv, S; Liu, G; Qiu, W; Geng, Y. Effects of semaglutide on metabolism and gut microbiota in high-fat diet-induced obese mice. Front Pharmacol. 2025, 16, 1562896. [Google Scholar] [CrossRef]
- Dupas, T; Pelé, T; Dhot, J; Burban, M; Persello, A; Aillerie, V; et al. The endothelial dysfunction could be a cause of heart failure with preserved ejection fraction development in a rat model. Oxid Med Cell Longev. 2022, 2022, 7377877. [Google Scholar] [CrossRef]
- Drekonja, D; Shaukat, A; Zhang, JH; Reinink, AR; Nugent, S; Dominitz, JA; et al. Microbiota or placebo after antimicrobial therapy for recurrent Clostridioides difficile at home: a clinical trial with novel home-based enrollment. Clin Trials 2021, 18(5), 622–629. [Google Scholar] [CrossRef]
- Ooijevaar, RE; Terveer, EM; Verspaget, HW; Kuijper, EJ; Keller, JJ. Clinical application and potential of fecal microbiota transplantation. Annu Rev Med. 2019, 70, 335–351. [Google Scholar] [CrossRef]
- Szulińska, M; Łoniewski, I; van Hemert, S; Sobieska, M; Bogdański, P. Dose-dependent effects of multispecies probiotic supplementation on the lipopolysaccharide (LPS) level and cardiometabolic profile in obese postmenopausal women: a 12-week randomized clinical trial. Nutrients 2018, 10(6), 773. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z; Chen, X; Sheng, H; Shen, X; Sun, X; Yan, Y; et al. Engineering probiotics as living diagnostics and therapeutics for improving human health. Microb Cell Fact. 2020, 19(1), 56. [Google Scholar] [CrossRef] [PubMed]
- Grumet, L; Tromp, Y; Stiegelbauer, V. The development of high-quality multispecies probiotic formulations: from bench to market. Nutrients 2020, 12(8), 2453. [Google Scholar] [CrossRef] [PubMed]
- Przerwa, F; Kukowka, A; Kotrych, K; Uzar, I. Probiotics in prevention and treatment of cardiovascular diseases. Herba Pol. 2021, 67(4), 77–85. [Google Scholar] [CrossRef]
- Khan, S; Ahmad, F; Khalid, N. Applications of strain-specific probiotics in the management of cardiovascular diseases: a systematic review. Mol Nutr Food Res. 2024, 68(8), e2300675. [Google Scholar] [CrossRef]
- Dixon, A; Robertson, K; Yung, A; Que, MS; Randall, H; Wellalagodage, T; et al. Efficacy of probiotics in patients at cardiovascular disease risk: a systematic review and meta-analysis. Curr Hypertens Rep. 2020, 22(9), 66. [Google Scholar] [CrossRef]
- Singh, RP; Shadan, A; Ma, Y. Biotechnological applications of probiotics: a multifarious weapon to disease and metabolic abnormality. Probiotics Antimicrob Proteins 2022, 14(6), 1184–1210. [Google Scholar] [CrossRef]
- Suez, J; Zmora, N; Elinav, E. Probiotics in the next-generation sequencing era. Gut Microbes 2020, 11(1), 77–93. [Google Scholar] [CrossRef]
- Videja, M; Sevostjanovs, E; Upmale-Engela, S; Liepinsh, E; Konrāde, I; Dambrova, M. Fasting-mimicking diet reduces trimethylamine N-oxide levels and improves serum biochemical parameters in healthy volunteers. Nutrients 2022, 14(5), 1093. [Google Scholar] [CrossRef]
- Goldiș, A; Dragomir, R; Mercioni, MA; Goldiș, C; Sirca, D; Enătescu, I; et al. Personalized microbiome modulation to improve clinical outcomes in pediatric inflammatory bowel disease: a multi-omics and interventional approach. Microorganisms 2025, 13(5), 1047. [Google Scholar] [CrossRef]
- Zamparette, CP; Teixeira, BL; Cruz, GNF; Filho, VB; Oliveira, LFV. Individual effect of diet on postprandial glycemic response and its relationship with gut microbiome profile in healthy subjects: protocol for a series of randomized N-of-1 trials. medRxiv [Preprint] 2023. [Google Scholar] [CrossRef]
- Shoer, S; Shilo, S; Godneva, A; Ben-Yacov, O; Rein, M; Wolf, BC; et al. Impact of dietary interventions on pre-diabetic oral and gut microbiome, metabolites and cytokines. Nat Commun. 2023, 14(1), 5716. [Google Scholar] [CrossRef] [PubMed]
- Iorra, FQ; Rodrigues, PG; Bock, PM; Guahnon, MP; Eller, S; Oliveira, TF; et al. Gut microbiota metabolite TMAO and adolescent cardiometabolic health: a cross-sectional analysis. J Endocr Soc. 2025, 9(5), bvaf055. [Google Scholar] [CrossRef] [PubMed]
- Hill, EB; Chen, L; Bailey, MT; Khalsa, AS; Maltz, R; Kelleher, KJ; et al. Facilitating a high-quality dietary pattern induces shared microbial responses linking diet quality, blood pressure, and microbial sterol metabolism in caregiver-child dyads. Gut Microbes 2022, 14(1), 2150502. [Google Scholar] [CrossRef]
- Saxena, R; Sharma, V; Saxena, AR; Patel, A. Harnessing AI and gut microbiome research for precision health. J Artif Intell Gen Sci. 2024, 3(1), 74–88. [Google Scholar] [CrossRef]
- Karwowska, Z; Szczerbiak, P; Kościółek, T. Microbiome time series data reveal predictable patterns of change. Microbiol Spectr. 2024, 12(10), e04109-23. [Google Scholar] [CrossRef]
- Mendes-Soares, H; Raveh-Sadka, T; Azulay, S; Edens, K; Ben-Shlomo, Y; Cohen, Y; et al. Assessment of a personalized approach to predicting postprandial glycemic responses to food among individuals without diabetes. JAMA Netw Open. 2019, 2(2), e188102. [Google Scholar] [CrossRef]
- Ye, Y; More, SK; Hamade, H; Alexeev, EE; Shimodaira, Y; Blackwood, AY; et al. TL1A overexpression in Crohn’s disease and mice alters Paneth cells and microbiota promoting ileal inflammation. bioRxiv [Preprint] 2025. [Google Scholar] [CrossRef]
- Fu, Y; Mei, Q; Yin, N; Huang, Z; Li, B; Luo, S; et al. Paneth cells protect against acute pancreatitis via modulating gut microbiota dysbiosis. mSystems 2022, 7(3), e01507-21. [Google Scholar] [CrossRef]
- Lee, VH; Gulati, A. Implications of Paneth cell dysfunction on gastrointestinal health and disease. Curr Opin Gastroenterol. 2022, 38(6), 535–540. [Google Scholar] [CrossRef]
- Du, W; Wang, J; Kuo, T; Wang, L; McKimpson, WM; Son, J; et al. Pharmacological conversion of gut epithelial cells into insulin-producing cells lowers glycemia in diabetic animals. J Clin Invest. 2022, 132(24), e162720. [Google Scholar] [CrossRef] [PubMed]
- Wang, J; Tian, F; Wang, P; Zheng, H; Zhang, Y; Tian, H; et al. Gut microbiota as a modulator of Paneth cells during parenteral nutrition in mice. JPEN J Parenter Enteral Nutr. 2018, 42(8), 1280–1287. [Google Scholar] [CrossRef] [PubMed]
- Sun, S; Hodel, M; Wang, X; Vicente, J; Haritunians, T; Debebe, A; et al. Macrophage LRRK2 hyperactivity impairs autophagy and induces Paneth cell dysfunction. Sci Immunol. 2024, 9(101), eadi7907. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H; Zhou, S; Gillilland, M; Li, J; Lee, A; Gao, J; et al. Bile acid toxicity in Paneth cells contributes to gut dysbiosis induced by high-fat feeding. JCI Insight 2020, 5(20), e138881. [Google Scholar] [CrossRef]
- Ahlawat, S; Asha, A; Sharma, KK. Gut–organ axis: A microbial outreach and networking. Lett Appl Microbiol. 2021, 72(6), 636–668. [Google Scholar] [CrossRef]
- Srugo, SA; Bloise, E; Nguyen, T; Connor, KL. Impact of maternal malnutrition on gut barrier defense: implications for pregnancy health and fetal development. Nutrients 2019, 11(6), 1375. [Google Scholar] [CrossRef]
- Rivera, K; González, L; Bravo, L; Manjarres, L; Andía, ME. The gut–heart axis: molecular perspectives and implications for myocardial infarction. Int J Mol Sci. 2024, 25(22), 12465. [Google Scholar] [CrossRef]
- Jaimez-Alvarado, S; López-Tenorio, II; Santos, J; Bello-Vega, DC; Gómez, FJR; Amedei, A; et al. Gut-heart axis: microbiome involvement in restrictive cardiomyopathies. Biomedicines 2025, 13(1), 144. [Google Scholar] [CrossRef]
- Kessler, EL; Oerlemans, MI; van den Hoogen, P; Yap, C; Sluijter, JPG; de Jager, SC. Immunomodulation in heart failure with preserved ejection fraction: current state and future perspectives. J Cardiovasc Transl Res. 2021, 14(1), 63–74. [Google Scholar] [CrossRef]
- Shi, Y; Perez-Bonilla, P; Chen, X; Tam, K; Marshall, M; Morin, J; et al. Metabolic syndrome nonalcoholic steatohepatitis male mouse with adeno-associated viral renin as a novel model for heart failure with preserved ejection fraction. J Am Heart Assoc. Published online. 2024. [CrossRef]
- Doiron, JE; Xia, H; Yu, X; Nevins, A; LaPenna, KB; Sharp, TE; et al. Adjunctive therapy with an oral H2S donor provides additional therapeutic benefit beyond SGLT2 inhibition in cardiometabolic heart failure with preserved ejection fraction. Br J Pharmacol. 2024, 181(21), 4294–4310. [Google Scholar] [CrossRef]
- Tong, D; Schiattarella, GG; Jiang, N; Altamirano, F; Szweda, PA; Elnwasany, A; et al. NAD+ repletion reverses heart failure with preserved ejection fraction. Circ Res. 2021, 128(11), 1629–1641. [Google Scholar] [CrossRef] [PubMed]
- Parajuli, P; Lara-Garcia, O; Regmi, MR; Skoza, W; Bhattarai, M; Kulkarni, A; et al. Heart failure drug class effects on 30-day readmission rates in patients with heart failure with preserved ejection fraction: a retrospective single-center study. Medicines (Basel) 2020, 7(5), 30. [Google Scholar] [CrossRef] [PubMed]
- Zhou, G; Chen, J; Wu, C; Jiang, P; Wang, Y; Zhang, Y; et al. Deciphering the protein, modular connections and precision medicine for heart failure with preserved ejection fraction and hypertension based on TMT quantitative proteomics and molecular docking. Front Physiol. 2021, 12, 607089. [Google Scholar] [CrossRef] [PubMed]
- Aimo, A; Senni, M; Barison, A; Panichella, G; Passino, C; Bayés-Genís, A; et al. Management of heart failure with preserved ejection fraction: from neurohormonal antagonists to empagliflozin. Heart Fail Rev. Published online. 2022. [CrossRef]
- Herisson, FM; Cluzel, G; Llopis-Grimalt, MA; O’Donovan, AN; Koç, F; Karnik, K; et al. Targeting the gut-heart axis improves cardiac remodeling in a clinical scale model of cardiometabolic syndrome. JACC Basic Transl Sci. 2025, 10(1), 1–15. [Google Scholar] [CrossRef]
- Withaar, C; Meems, LMG; Markousis-Mavrogenis, G; Boogerd, CJ; Silljé, HH; Schouten, EM; et al. The effects of liraglutide and dapagliflozin on cardiac function and structure in a multi-hit mouse model of heart failure with preserved ejection fraction. Cardiovasc Res. 2021, 117(9), 2108–2124. [Google Scholar] [CrossRef]
- Momot, K; Krauz, K; Czarzasta, K; Zarębiński, M; Puchalska, L; Wojciechowska, M. Evaluation of nitrosative/oxidative stress and inflammation in heart failure with preserved and reduced ejection fraction. Int J Mol Sci. 2023, 24(21), 15944. [Google Scholar] [CrossRef]
- Guivala, SJ; Bode, KA; Okun, JG; Kartal, E; Pohl, LV; Werner, S; et al. Interactions between the gut microbiome, associated metabolites and the manifestation and progression of heart failure with preserved ejection fraction in ZSF1 rats. Res Square [Preprint] 2024. [Google Scholar] [CrossRef]
- Gaćeša, R; Kurilshikov, A; Vila, AV; Sinha, T; Klaassen, MAY; Bolte, LA; et al. The Dutch Microbiome Project defines factors that shape the healthy gut microbiome. bioRxiv [Preprint] 2020. [Google Scholar] [CrossRef]
- Wagenaar, CA; van de Put, M; Bisschops, M; Walrabenstein, W; de Jonge, CS; Herrema, H; et al. The effect of dietary interventions on chronic inflammatory diseases in relation to the microbiome: a systematic review. Nutrients 2021, 13(9), 3208. [Google Scholar] [CrossRef]
- Krüger-Genge, A; Jung, F; Hufert, F; Küpper, J; Storsberg, J. Effects of gut microbial metabolite trimethylamine N-oxide (TMAO) on platelets and endothelial cells. Clin Hemorheol Microcirc. 2020, 76(2), 309–316. [Google Scholar] [CrossRef]
- Joishy, TK; Jha, AR; Oudah, M; Das, S; Adak, A; Deb, D; et al. Human gut microbes associated with systolic blood pressure. Int J Hypertens. 2022, 2022, 2923941. [Google Scholar] [CrossRef]
- Valencia, S; Zuluaga, M; Pérez, M; Montoya-Quintero, KF; Candamil-Cortés, MS; Robledo, S. Human gut microbiome: a connecting organ between nutrition, metabolism, and health. Int J Mol Sci. 2025, 26(9), 4112. [Google Scholar] [CrossRef]
- Jama, H; Dona, MS; Dinakis, E; Nakai, M; Paterson, M; Shihata, W; et al. Maternal diet and gut microbiota influence predisposition to cardiovascular disease in the offspring. bioRxiv [Preprint] 2022. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




