Submitted:
28 January 2026
Posted:
28 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Experimental Materials
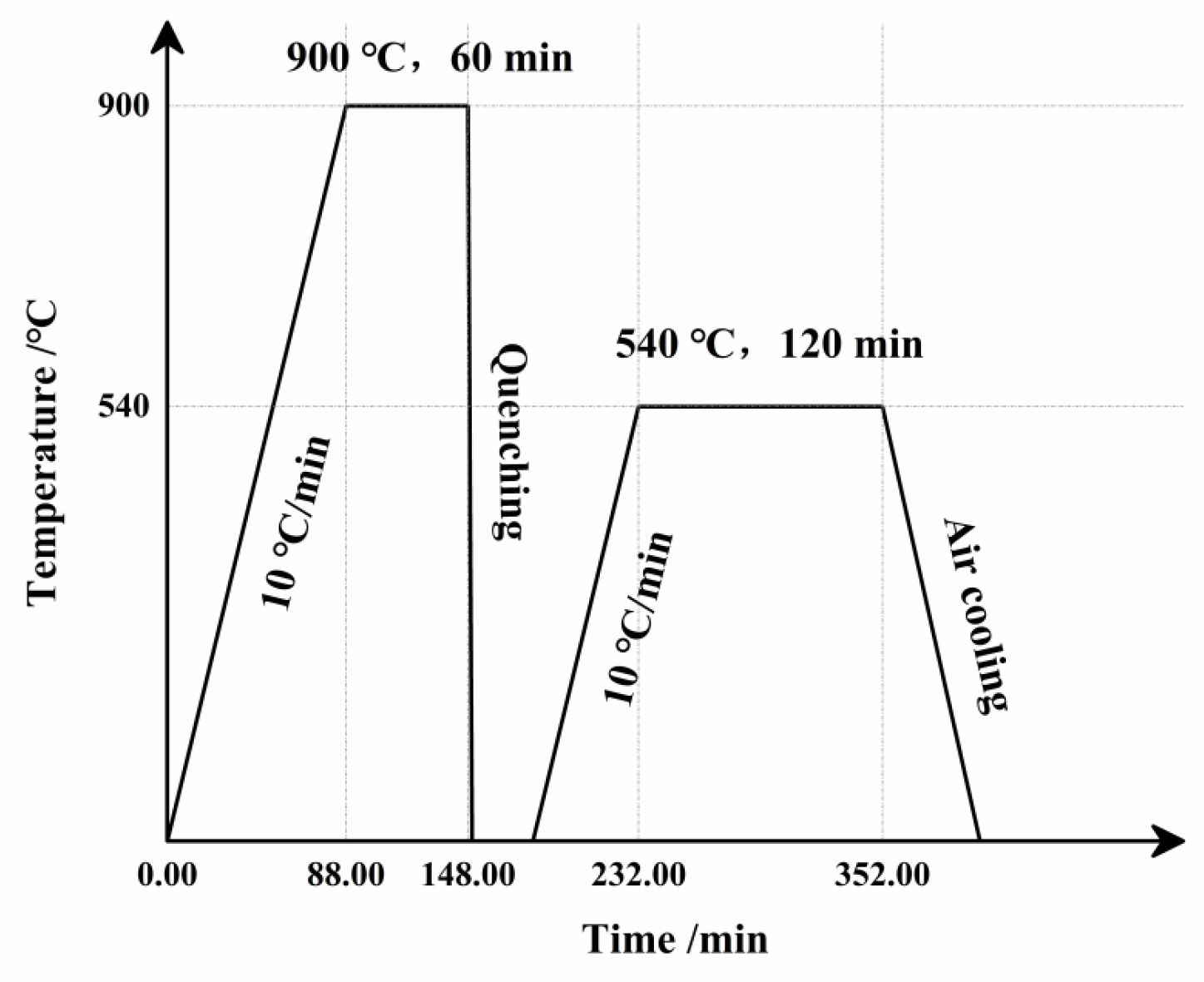
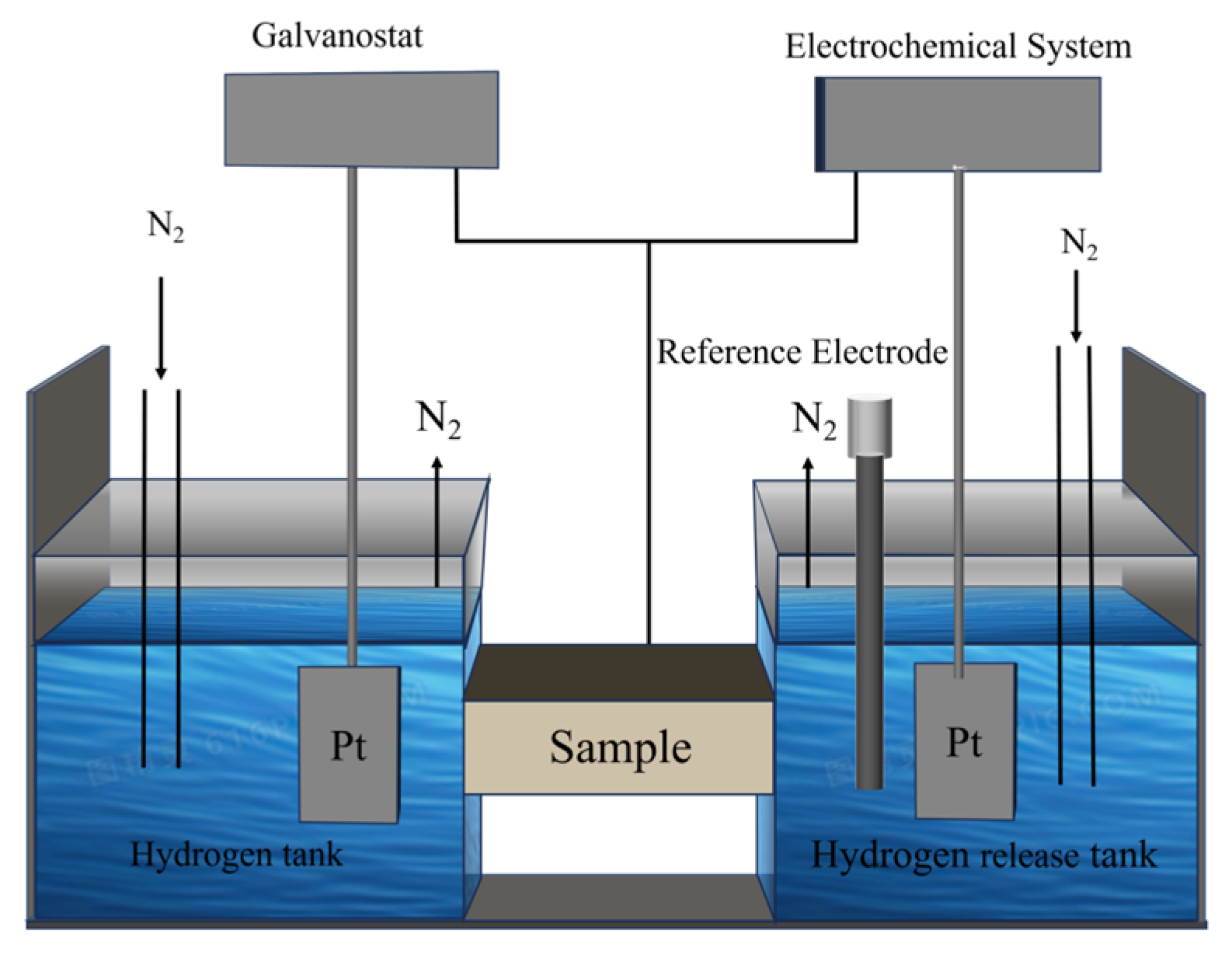
2.2. Experimental Methods
3. Results
3.1. Microstructure
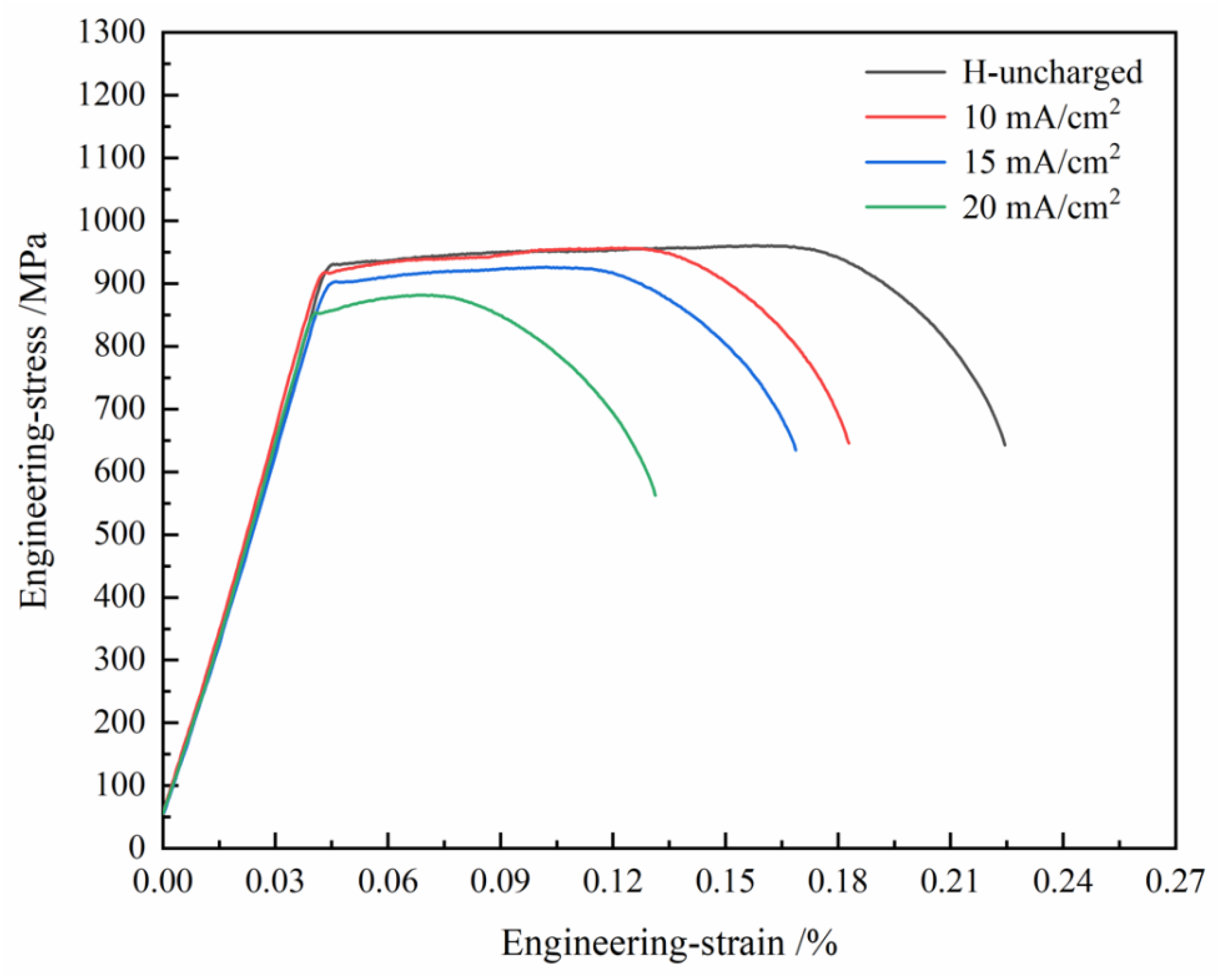
3.2. Mechanical Properties and Hydrogen Embrittlement Sensitivity
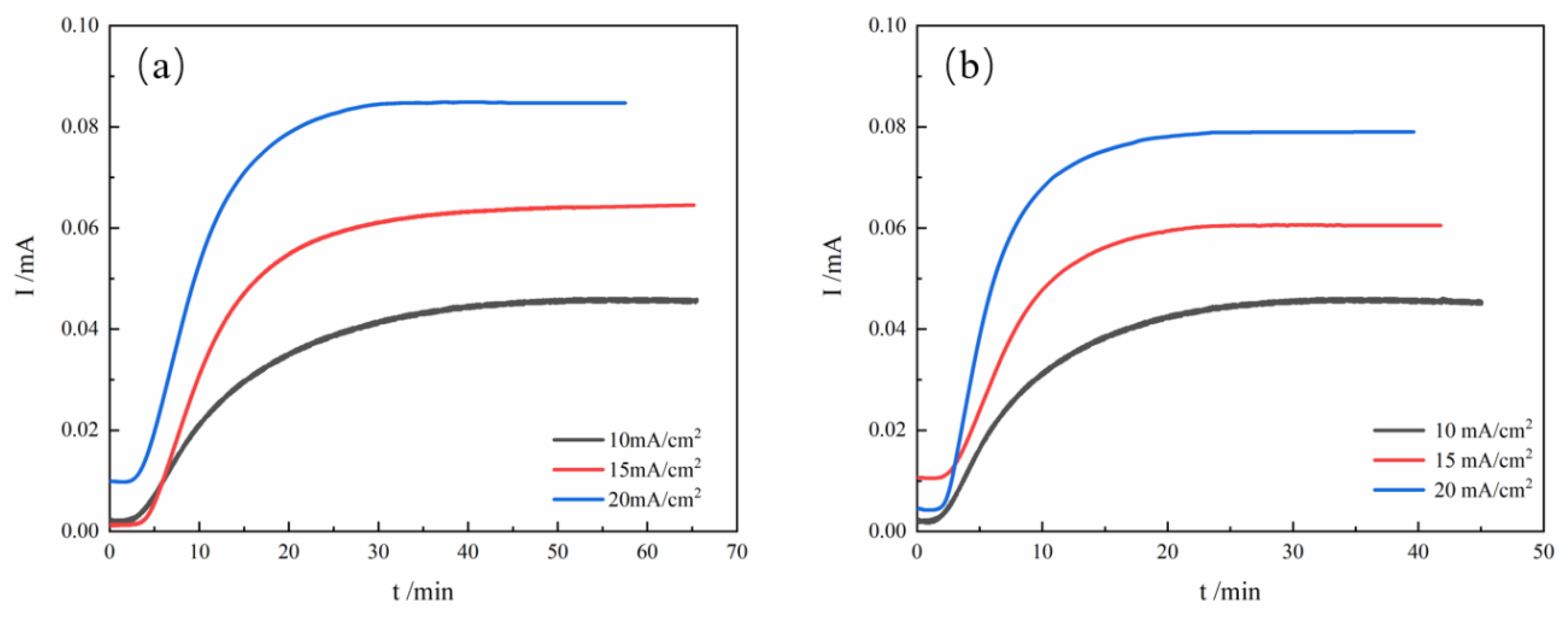
3.3. Hydrogen Diffusion Behavior
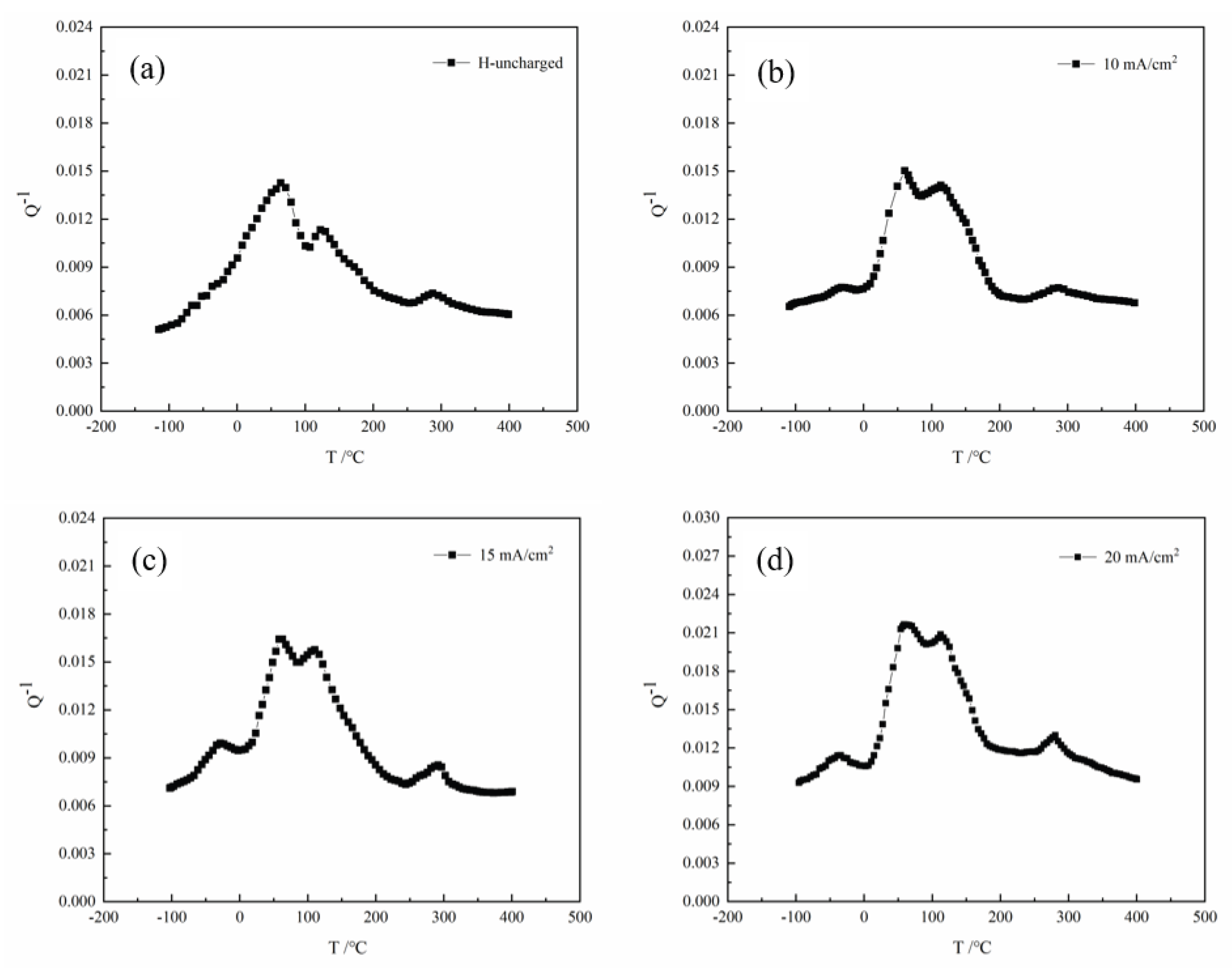
3.4. Internal Friction Behavior
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lirong, D; Zhixin, W; Jianjun, W. Analysis of the world oil and gas exploration situation in 2021[J]. Petroleum Exploration and Development Online 2022, 49(5), 1195–1209. [Google Scholar]
- Fu, E; He, W. The development and utilization of shale oil and gas resources in China and economic analysis of energy security under the background of global energy crisis[J]. Journal of Petroleum Exploration and Production Technology 2024, 14(8-9), 2315–2341. [Google Scholar]
- Rui, L; Li, L; Fuhui, W. The role of hydrostatic pressure on the metal corrosion in simulated deep-sea environments — a review[J]. Journal of Materials Science & Technology (prepublish). 2021. [Google Scholar]
- Hong, Heting; et al. Research of Cu-bearing stainless steel with inhibition function of biofouling formation in freshwater environment. Journal of Materials Research and Technology 2024, 28, 2943–2954. [Google Scholar]
- Jae, Y J; On, S K; Sung, J P. Strong and ductile Fe-24Mn-3Cr alloy resistant against erosion-corrosion[J]. npj Materials Degradation 2021, 5(1). [Google Scholar]
- Spiryagin, V V; Kravchenko, N I; Kuznetsov, A Y. Review of International Experience in Substantiating the Technology of Smelting Copper Sulfide-Containing Concentrates Taking into Account the Criteria of Efficiency, Cost Effectiveness, and Environmental Friendliness[J]. Surface Engineering and Applied Electrochemistry 2025, 61(2), 192–201. [Google Scholar] [CrossRef]
- HUANG, Yun-hua; et al. Influence of nanosized precipitate on the corrosion behavior of high-strength low-alloy steels: A review. Chinese Journal of Engineering 2021, 43.3, 321–331. [Google Scholar]
- Sobola, D; Dallaev, R. Exploring Hydrogen Embrittlement: Mechanisms, Consequences, and Advances in Metal Science[J]. Energies 2024, 17(12), 2972–2972. [Google Scholar] [CrossRef]
- Tuhin, D.; Brahimi, S.V.; Song, J.; Yue, S. A fast fracture approach to assess hydrogen embrittlement (HE) susceptibility and mechanism(s) of high strength martensitic steels[J]. Corrosion Science 2021, (prepublish), 109701. [Google Scholar]
- Li, Q; Ghadiani, H; Jalilvand, V. Hydrogen Impact: A Review on Diffusibility, Embrittlement Mechanisms, and Characterization[J]. Materials 2024, 17(4), 965. [Google Scholar]
- Ahad, T M; Bhuiyan, H M M; Sakib, N A. An Overview of Challenges for the Future of Hydrogen[J]. Materials 2023, 16(20). [Google Scholar] [CrossRef]
- Harris, D Z; Somerday, P B. Hydrogen embrittlement of steels: Mechanical properties in gaseous hydrogen[J]. International Materials Reviews 2025, 70(5), 394–421. [Google Scholar] [CrossRef]
- Krella, A. Hydrogen-Induced Degradation of Metallic Materials—A Short Review[J]. Materials 2025, 18(3), 597–597. [Google Scholar] [CrossRef]
- Seo, H J; Heo, Y U; Kim, J N; et al. Effect of V/Mo ratio on the evolution of carbide precipitates and hydrogen embrittlement of tempered martensitic steel[J]. Corrosion Science 2020, 176, 108929. [Google Scholar] [CrossRef]
- Momotani, Y; Shibata, A; Yonemura, T; et al. Effect of initial dislocation density on hydrogen accumulation behavior in martensitic steel[J]. Scripta Materialia 2020, 178, 318–323. [Google Scholar] [CrossRef]
- Solheim, K G; Solberg, J K; Walmsley, J; et al. The role of retained austenite in hydrogen embrittlement of supermartensitic stainless steel[J]. Engineering Failure Analysis 2013, 34, 140–149. [Google Scholar] [CrossRef]
- Bechtle, S; Kumar, M; Somerday, B P; et al. Grain-boundary engineering markedly reduces susceptibility to intergranular hydrogen embrittlement in metallic materials[J]. Acta materialia 2009, 57(14), 4148–4157. [Google Scholar] [CrossRef]
- Martin, M; Weber, S; Theisen, W; et al. Effect of alloying elements on hydrogen environment embrittlement of AISI type 304 austenitic stainless steel[J]. International journal of hydrogen energy 2011, 36(24), 15888–15898. [Google Scholar] [CrossRef]
- Wu, X; Song, Z; Tan, M. Hydrogen-induced failure mechanism of X80 pipeline steel welded joints based on macro-and micro-scale experimental analysis: Embrittlement enhancement effect caused by high hydrogen trap density[J]. Engineering Failure Analysis 2026, 183110190–110190. [Google Scholar] [CrossRef]
- Ehsan, E; González, L J V; M.A., M. Experimental observations of nucleation and crack growth paths of hydrogen-induced cracking in pipeline steel[J]. Engineering Failure Analysis 2023, 154. [Google Scholar]
- Yu, H; Díaz, A; Lu, X. Hydrogen Embrittlement as a Conspicuous Material Challenge-Comprehensive Review and Future Directions.[J]. Chemical reviews 2024, 124(10), 6271–6392. [Google Scholar] [CrossRef] [PubMed]
- Shiqi, Z; Wanqi, C; Feng, H. The significant effect of tantalum on the hydrogen-induced cracking of pipeline steel: Morphology, hydrogen permeation, and theoretical studies[J]. Corrosion Science 2022, (prepublish), 110213. [Google Scholar]
- Dan, J W; Shi, H; Tang, W C. Effects of Electrochemical Hydrogen Charging Parameters on the Mechanical Behaviors of High-Strength Steel[J]. Materials 2024, 17(17), 4290–4290. [Google Scholar] [CrossRef]
- Xu, C; Wang, F; Li, J. The Influence of Hydrogen-Charging Current Density and Temperature on Hydrogen Permeation and Hydrogen Embrittlement Susceptibility of 4130X Steel[J]. Materials 2025, 18(15), 3448–3448. [Google Scholar] [CrossRef]
- L.B., P; D, A.; J.M., A. Hydrogen uptake and diffusion kinetics in a quenched and tempered low carbon steel: experimental and numerical study[J]. International Journal of Hydrogen Energy 2023, 48(90), 35347–35365. [Google Scholar]
- Du, Y; Gao, X; Lan, L. Hydrogen embrittlement behavior of high strength low carbon medium manganese steel under different heat treatments[J]. International Journal of Hydrogen Energy 2019, 44(60), 32292–32306. [Google Scholar] [CrossRef]
- Qinying, W; Xiaojia, J; Xingshou, Z. Effect of hydrogen charging current density on hydrogen damage sensitivity of laser-peened 316L stainless steel[J]. Anti-Corrosion Methods and Materials 2025, 72(4), 615–627. [Google Scholar]
- Takano, N; Yokka, Y; Terasaki, F. The effect of copper precipitation on hydrogen embrittlement in iron[J]. Materials Science & Engineering A 2003, 387428–432. [Google Scholar]
- Gu, J; Jiang, S; Qi, Y. Effect of Copper Alloying on Hydrogen Embrittlement of Fe-28Mn-10Al-1C Austenitic Low-Density Steel[J]. Materials 2025, 18(17), 4139–4139. [Google Scholar] [CrossRef]
- Kwon, J Y; Lee, T; Lee, J. Role of Cu on hydrogen embrittlement behavior in Fe-Mn-C-Cu TWIP steel[J]. International Journal of Hydrogen Energy 2015, 40(23), 7409–7419. [Google Scholar] [CrossRef]


| C | Cu | Ni | Mn | Cr | Si | Ti |
|---|---|---|---|---|---|---|
| 0.036 | 6.01 | 2.70 | 0.97 | 0.48 | 0.20 | 0.0089 |
| Hydrogen Charging Conditions | Yield Strength /MPa | Tensile Strength /MPa | Elongation /% | Hydrogen Embrittlement Sensitivity /% |
|---|---|---|---|---|
| Uncharged | 916 | 960 | 22.8 | - |
| 10 mA/cm2 | 909 | 956 | 17.3 | 24.1 |
| 15 mA/cm2 | 888 | 926 | 16.8 | 26.3 |
| 20 mA/cm2 | 840 | 861 | 13.1 | 42.5 |
| Parameter | 10 mA/cm2 | 15 mA/cm2 | 20 mA/cm2 | |||
|---|---|---|---|---|---|---|
| 1st Permeation | 2nd Permeation | 1st Permeation | 2nd Permeation | 1st Permeation | 2nd Permeation | |
| tlag(s) | 862.2 | 558.35 | 745.8 | 443.4 | 603.6 | 367.2 |
| Jss(mol/cm2/s) | 6.7×10-8 | 6.57×10-8 | 9.34×10-8 | 8.76×10-8 | 1.23×10-7 | 1.15×10-7 |
| Deff(cm2/s) | 1.93×10-6 | 3.33×10-6 | 2.23×10-6 | 3.76×10-6 | 2.76×10-6 | 4.54×10-6 |
| C0(mol/cm3) | 3.47×10-3 | 1.97×10-3 | 4.19×10-3 | 2.33×10-3 | 4.46×10-3 | 2.5×10-3 |
| NT(cm-3) | 3.36×1022 | 3.09×1022 | 3.5×1022 | 1.13×1022 | 8.97×1022 | 4.21×1022 |
| Nir(cm-3) | 2.7×1022 | 2.37×1022 | 4.76×1022 | |||
| Hydrogen charging current density I /(mA/cm2) | Internal friction peak | Peak temperature Tm /℃ | Frequency Fm /Hz | Activation energy H /eV |
|---|---|---|---|---|
| Uncharged | P2 | 64.1 | 1.88 | 0.871 |
| P3 | 120.5 | 1.868 | 1.021 | |
| P4 | 297.2 | 1.824 | 1.519 | |
| 10 | P1 | -29.6 | 1.91 | 0.68 |
| P2 | 62.4 | 1.849 | 0.868 | |
| P3 | 117.2 | 1.841 | 1.014 | |
| P4 | 291.9 | 1.796 | 1.506 | |
| 15 | P1 | -33.2 | 1.669 | 0.621 |
| P2 | 61.7 | 1.648 | 0.868 | |
| P3 | 114.5 | 1.633 | 1.013 | |
| P4 | 286.6 | 1.599 | 1.498 | |
| 20 | P1 | -39.3 | 1.779 | 0.598 |
| P2 | 59.8 | 1.771 | 0.862 | |
| P3 | 112.2 | 1.761 | 1.002 | |
| P4 | 280.5 | 1.715 | 1.477 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




