Submitted:
28 January 2026
Posted:
29 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
Rational
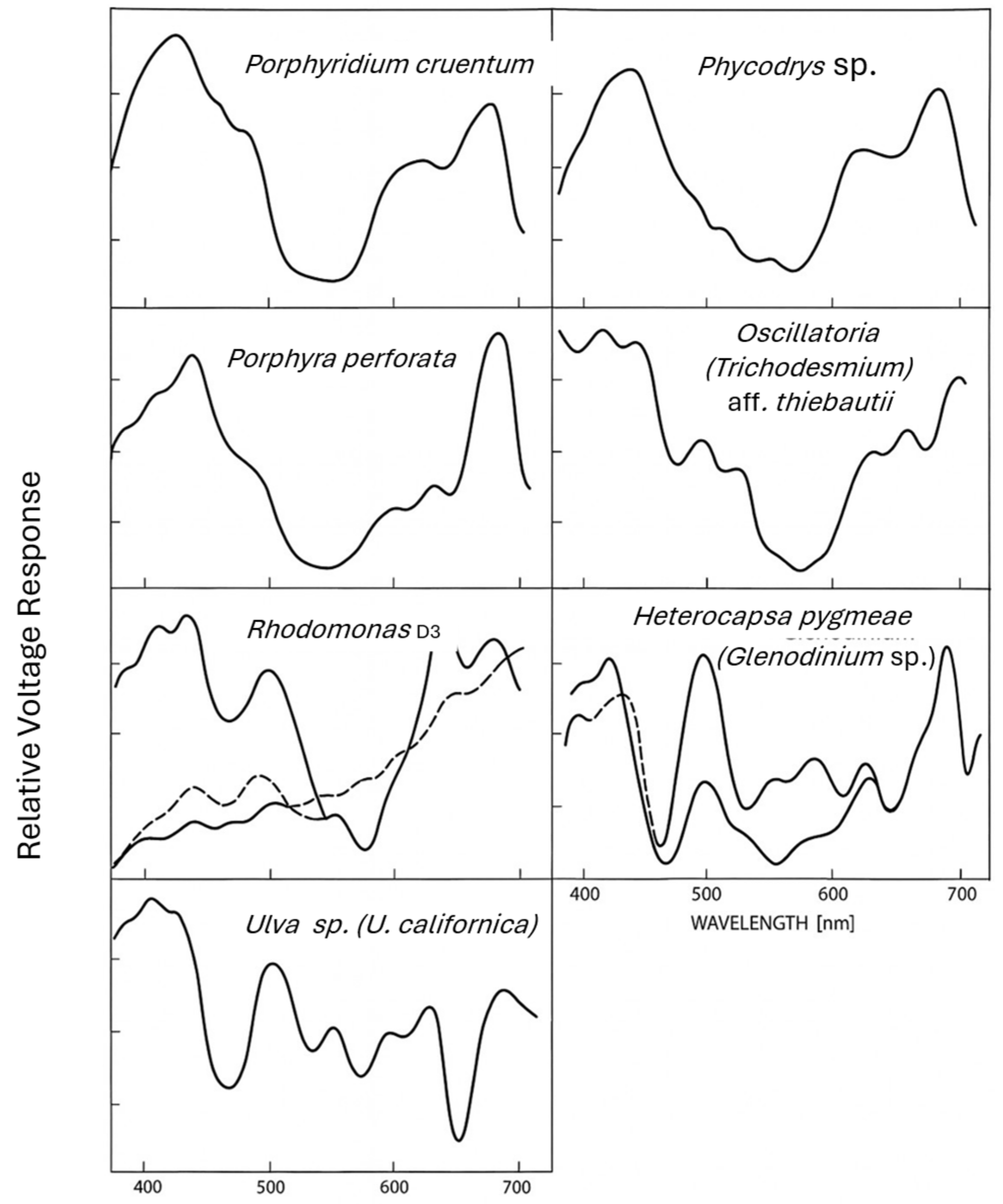
2. Results
| Genus | In vivo absorption maxima (nm) | Pigment |
|---|---|---|
|
Porphyra perforata abci (Rhodophyta) |
436, 677 493, 546, 565 617 |
Chlorophyll a R-phycoerythrin R-phycocyanin |
|
Phycodrys dc (Rhodophyta) |
438, 625, 680 498, 540, 566 625 |
Chlorophyll a R-phycoerythrin T-phycocyanin |
|
Rhodomonas bdfij (Cryptophyta) |
435, 638, 676 460, 632 545, 565 460, 495 |
Chlorophyll a Chlorophyll c2 Phycoerythrin type I Alloxanthin |
|
Glenodinium p (Pyrrophyta) |
437, 590, 622, 676 462, 590, 632 525 (broad) 497 |
Chlorophyll a Chlorophyll c2 Peridinin β-Carotene and minor xanthophylls |
|
Ulva abqr (Chlorophyta) |
437, 678 475, 652 475 |
Chlorophyll a Chlorophyll b Lutein and other carotenoids |
3. Discussion
3.1. Rhodophytes and Cyanobacteria
3.2. Chlorophytes
3.3. Cryptomonads
3.4. Dinoflagellates
3.5. Comparative Interpretation Between Species
4. Materials and Methods
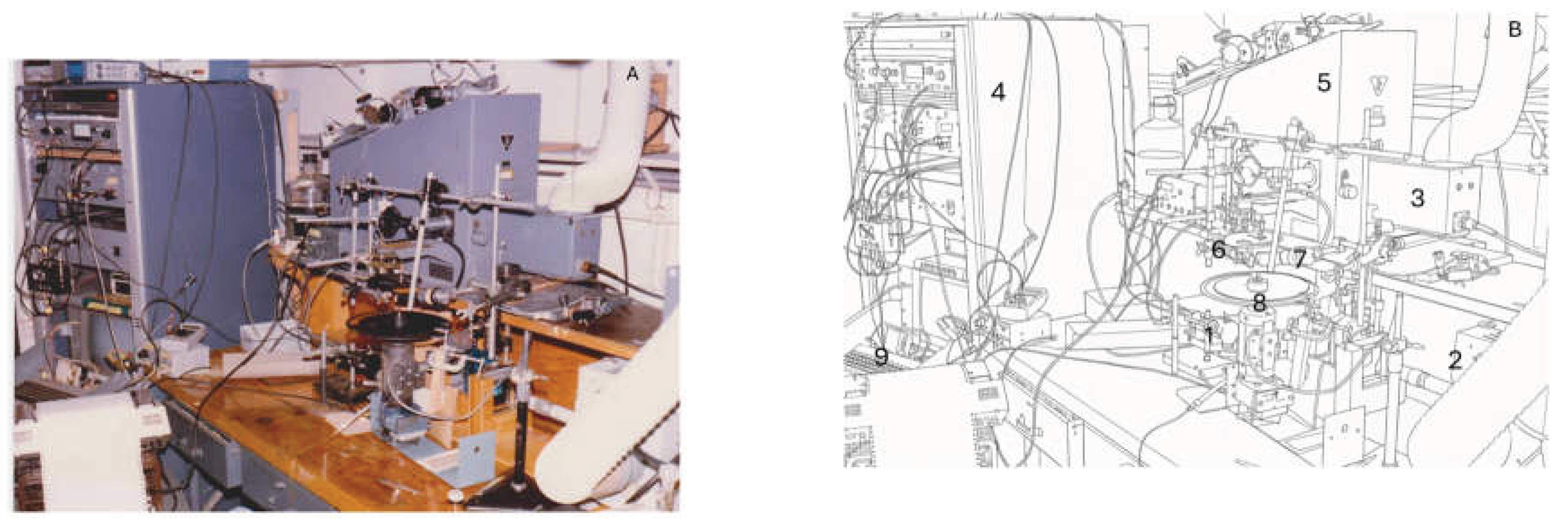
4.1. Measurement of Enhancement Spectra
- Background beam: A broadband continuous light source was applied at an intensity Ib sufficient to elicit a strong yet linear oxygen-evolution response. Its spectral distribution (referred to as radiation II) was preferentially absorbed by accessory pigments associated with PSII and overlapped with wavelengths corresponding to regions of maximal quantum yield in the PSII action spectrum of each alga.
- Measuring beam: This modulated beam of intensity Im had the same spectral characteristics as the higher-intensity background beam (radiation II). A 10 Herz modulation frequency produced the highest-amplitude alternating-current oxygen-evolution signal, consistent with findings from later independent studies [92].
- Scanning beam: A weak, continuous, monochromatic beam with a five-nanometer half-bandwidth was generated by the computer-controlled monochromator and used to scan the spectral range (radiation I). The instrument automatically maintained a constant quantum flux to the sample across all wavelengths.
4.2. Algal Material
| Species | n | Background Beam | Measuring Beam | Scanning Beam |
|---|---|---|---|---|
| Porphyridium cruentum | 2 | 150.5 1+3 | 10.5 2+5 | 10.5 7 |
| (7.0) | (0.5) | (0.5) | ||
| Porphyra perforata | 1 | 60.5 1+3 | 2.5 2+3 | 6.5 7 |
| (3.0) | (0.12) | (0.3) | ||
| Phycodrys sp. | 1 | 60.5 1+3 | 5.5 2+3 | 3.5 7 |
| (3.0) | (0.25) | (0.15) | ||
|
Oscillatoria (trichodesmium) thiebautii |
3 | 5.5 3 | 15.5 2+3 | 2.5 7 |
| (0.25) | (0.7) | (0.12) | ||
| Ulva sp. | 3 | 40.0 1+4 | 4.5 2+4 | 20.5 6 |
| (1.6) | (0.15) | (0.8) |
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Govindjee, G. On the Evolution of the Concept of Two Light Reactions and Two Photosystems for Oxygenic Photosynthesis: A Personal Perspective. Photosynthetica 2023, 61, 37–47. [CrossRef]
- Emerson, R.; Rabinowitch, E. Red Drop and Role of Auxiliary Pigments in Photosynthesis. Plant Physiology 1960, 35, 477–485.
- Evans, J.R. A Quantitative Analysis of Light Distribution between the Two Photosystems, Considering Variation in Both the Relative Amounts of the Chlorophyll-Protein Complexes and the Spectral Quality of Light. Photobiochemistry and Photobiophysics 1986, 10, 135–147.
- Falkowski, P.; Owens, T.G. Light-Shade Adaptation. Plant Physiol. 1980, 66, 592–595.
- Malkin, S.; Herbert, S.K.; Fork, D.C. Light Distribution, Transfer and Utilization in the Marine Red Alga Porphyra perforata: From Photoacoustic Energy-Storage Measurements. Biochimica et Biophysica Acta (BBA) - Bioenergetics 1990, 1016, 177–189. [CrossRef]
- Gallagher, J.C.; Wood, A.M.; Alberte, R.S. Ecotypic Differentiation in the Marine Diatom Skeletonema costatum: Influence of Light Intensity on the Photosynthetic Apparatus. Mar. Biol. 1984, 82, 121–134. [CrossRef]
- Stadnichuk, I.N.; Novikova, T.M.; Miniuk, G.S.; Boichenko, V.A.; Bolychevtseva, Yu.V.; Gusev, E.S.; Lukashev, E.P. Phycoerythrin Association with Photosystem II in the Cryptophyte Alga Rhodomonas salina: Biochemistry Moscow 2020, 85, 679–688. [CrossRef]
- Neori, A.; Vernet, M.; Holm-Hansen, O.; Haxo, F.T. Comparison of Chlorophyll Far-Red and Red Fluorescence Excitation Spectra with Photosynthetic Oxygen Action Spectra for Photosystem II in Algae. Marine Ecology Progress Series 1988, 44, 297–302.
- Neori, A. Excitation Spectra of Chlorophyll a Fluorescence in vivo : Their Correlation with Photosynthetic Action Spectra and Their Use in the Study of Algal Photoadaptation, University of California, San Diego, 1986.
- Mitchell, B. g.; Kiefer, D. a. Determination of Absorption and Fluorescence Excitation Spectra for Phytoplankton. In Marine Phytoplankton and Productivity; American Geophysical Union (AGU), 1984; pp. 157–169 ISBN 978-1-118-66951-8.
- Neori, A. The Use of Fluorescence Excitation Spectra of Chlorophyll a in Phytoplankton to Estimate Photoadaptation and Its Relation to Photosynthesis Action Spectra. In Proceedings of the Group for Aquatic Productivity GAP II International Workshop, Haifa, Israel; 1984.
- Falkowski, P.G.; LaRoche, J. Acclimation to Spectral Irradiance in Algae. Journal of Phycology 1991, 27, 8–14. [CrossRef]
- Iglesias-Prieto, R.; Trench, R.K. Acclimation and Adaptation to Irradiance in Symbiotic Dinoflagellates. II. Response of Chlorophyll–Protein Complexes to Different Photon-Flux Densities. Marine Biology 1997, 130, 23–33. [CrossRef]
- Lawrenz, E.; Richardson, T.L. Differential Effects of Changes in Spectral Irradiance on Photoacclimation, Primary Productivity and Growth in Rhodomonas salina (Cryptophyceae) and Skeletonema costatum (Bacillariophyceae) in Simulated Blackwater Environments. Journal of Phycology 2017, 53, 1241–1254. [CrossRef]
- Richardson, K.; Beardall, J.; Raven, J.A. Adaptation of Unicellular Algae to Irradiance: An Analysis of Strategies. New Phytologist 1983, 93, 157–191. [CrossRef]
- Larkum, A.W.D.; Barrett, J. Light-Harvesting Processes in Algae. In Advances in Botanical Research; Woolhouse, H.W., Ed.; Academic Press, 1983; Vol. 10, pp. 1–219.
- Fujita, Y.; Murakami, A. Regulation of Electron Transport Composition in Cyanobacterial Photosynthetic System: Stoichiometry among Photosystem I and II Complexes and Their Light-Harvesting Antennae and Cytochrome B6/f Complex. Plant and Cell Physiology 1987, 28, 1547–1553. [CrossRef]
- Machalek, K.M.; Davison, I.R.; Falkowski, P.G. Thermal Acclimation and Photoacclimation of Photosynthesis in the Brown Alga Laminaria saccharina. Plant, Cell & Environment 1996, 19, 1005–1016. [CrossRef]
- Satoh, A.; Kurano, N.; Senger, H.; Miyachi, S. Regulation of Energy Balance in Photosystems in Response to Changes in CO2 Concentrations and Light Intensities during Growth in Extremely-High-CO2 -Tolerant Green Microalgae. Plant Cell Physiol 2002, 43, 440–451. [CrossRef]
- Ueno, Y.; Aikawa, S.; Kondo, A.; Akimoto, S. Adaptation of Light-Harvesting Functions of Unicellular Green Algae to Different Light Qualities. Photosynth Res 2019, 139, 145–154. [CrossRef]
- Santabarbara, S.; Villafiorita Monteleone, F.; Remelli, W.; Rizzo, F.; Menin, B.; Casazza, A.P. Comparative Excitation-Emission Dependence of the FV/FM Ratio in Model Green Algae and Cyanobacterial Strains. Physiologia Plantarum 2019, 166, 351–364. [CrossRef]
- Dubinsky, Z.; Falkowski, P.G.; Wyman, K. Light Harvesting and Utilization by Phytoplankton. Plant Cell Physiol. 1986, 27(7), 1335–1349.
- Dring, M.J.; Lüning, K. Emerson Enhancement Effect and Quantum Yield of Photosynthesis for Marine Macroalgae in Stimulated Underwater Light Fields. Mar. Biol. 1985, 87, 109–117.
- Jerlov, N.G. Marine Optics; Elsevier Oceanography Series; 2nd ed.; Elsevies: Amsterdam, 1976;
- McCree, K.J. Significance of Enhancement for Calculations Based on the Action Spectrum for Photosynthesis 1. Plant Physiology 1972, 49, 704–706. [CrossRef]
- French, C.S.; Fork, D.C. Computer Solutions for Photosynthesis Rates from a Two Pigment Model. Biophysical Journal 1961, 1, 669–681. [CrossRef]
- Ried, A.; Hessenberg, B.; Metzler, H.; Ziegler, R. Distribution of Excitation Energy among Photosystem I and Photosystem II in Red Algae. I. Action Spectra of Light Reactions I and II. Biochimica et Biophysica Acta (BBA) - Bioenergetics 1977, 459, 175–186. [CrossRef]
- Joliot, P.; Joliot, A. A Polarographic Method for Detection of Oxygen Production and Reduction of Hill Reagent by Isolated Chloroplasts. Biochimica et Biophysica Acta (BBA) - Bioenergetics 1968, 153, 625–634. [CrossRef]
- Joliot, P.; Joliot, A.; Kok, B. Analysis of the Interactions between the Two Photosystems in Isolated Chloroplasts. Biochimica et Biophysica Acta (BBA) - Bioenergetics 1968, 153, 635–652. [CrossRef]
- Wang, R.T.; Myers, J. On the Distribution of Excitation Energy to Two Photoreactions of Photosynthesis. Photochemistry and Photobiology 1976, 23, 405–410. [CrossRef]
- Wang, R.T.; Stevens, C.L.R.; Myers, J. Action Spectra for Photoreactions I and II of Photosynthesis in the Blue-Green Alga Anacystis nidulans. Photochemistry and Photobiology 1977, 25, 103–108. [CrossRef]
- Wang, T.; Myers, J. Simultaneous Measurment of Action Spectra for Photoreactions I and II of Photosynthesis. Photochemistry and Photobiology. 1976, 23, 411–414.
- Neori, A.; Vernet, M.; Holm-Hansen, O.; Haxo, F.T. Relationship between Action Spectra for Chlorophyll a Fluorescence and Photosynthetic O2 Evolution in Algae. J Plankton Res 1986, 8, 537–548. [CrossRef]
- Ley, A.C.; Butler, W.L. Energy Transfer from Photosystem II to Photosystem I in Porphyridium cruentum. Biochimica et Biophysica Acta (BBA) - Bioenergetics 1977, 462, 290–294. [CrossRef]
- Fork, D. c. Observations on the Function of Chl a and Accessory Pigments in Photosynthesis. In Photosynthetic Mechanisms of Green Plants; Kok, B., Jagendorf, A.T., Eds.; Nat. Acad. Sci. Nat. Res. Council: Washington D.C., 1963; pp. 352–361.
- Ludlow, C.J.; Park, R.B. Action Spectra for Photosystems I and II in Formaldehyde Fixed Algae 1. Plant Physiology 1969, 44, 540–543. [CrossRef]
- Hamada, E. a. M.; Dowiadar, S.M.A.; Punnet, T. Influence of Spectral Range and Carbon and Nitrogen Sources on Oxygen Evolution and Emerson Enhancement in Chlamydomonas reinhardtii. Biologia Plantarum 2003, 3, 389–397. [CrossRef]
- Ried, A. Improved Action Spectra of Light Reaction I And II. In Proceedings of the Photosynthesis, two centuries after its discovery by Joseph Priestley; Forti, G., Avron, M., Melandri, A., Eds.; Springer Netherlands: Dordrecht, 1972; pp. 763–772.
- Kramer, H.J.M.; Westerhuis, W.H.J.; Amesz, J. Low Temperature Spectroscopy of Intact Algae. Physiol. vég. (Paris) 1985, 23, 535–543.
- Kok, B.; Gott, W. Activation Spectra of 700 Mμ Absorption Change in Photosynthesis. Plant Physiology 1960, 35, 802. [CrossRef]
- Joliot, P. Cinétiques Des Réactions Liées a l’émission d’oxygène Photosynthétique. Biochimica et Biophysica Acta (BBA) - Biophysics including Photosynthesis 1965, 102, 116–134. [CrossRef]
- Myers, J.; Graham, J.-R. Enhancement in Chlorella. Plant Physiol. 1963, 38, 1–5. [CrossRef]
- Itagaki, T.; Nakayama, K.; Okada, M. Chlorophyll-Protein Complexes Associated with Photosystem I Isolated from the Green Alga, Bryopsis maxima. Plant Cell Physiol 1986, 27, 1241–1247. [CrossRef]
- Laisk, A.; Oja, V.; Eichelmann, H.; Dall’Osto, L. Action Spectra of Photosystems II and I and Quantum Yield of Photosynthesis in Leaves in State 1. Biochimica et Biophysica Acta (BBA) - Bioenergetics 2014, 1837, 315–325. [CrossRef]
- Chapman, D.J. Studies on the Carotenoids of the Flagellate Cryptophyceae, and the Chloroplast Pigments of the Endosymbiotic Algae in Cyanophora paradoxa and Glaucocystis nostochinearum. Ph.D. Dissertation, University of California, San Diego, Calif .: San Diego, 1966.
- Pennington, F.C.; Haxo, F.T.; Borch, G.; Liaaen-Jensen, S. Carotenoids of Cryptophyceae. Biochemical Systematics and Ecology 1985, 13, 215–219. [CrossRef]
- Vernet, M.; Neori, A.; Haxo, F.T. Role of Carotenoid Pigments in Photosynthesis of Cryptophyceae. In Proceedings of the Transactions American Geophysical Union; Washington D.C., 1985; Vol. 66 (51), p. 1302.
- Haxo, F.T.; Blinks, L.R. Photosynthetic Action Spectra of Marine Algae. J Gen Physiol 1950, 33, 389–422. [CrossRef]
- Haxo, F.; O’hEocha, C.O.; Norris, P. Comparative Studies of Chromatographically Separated Phycoerythrins and Phycocyanins. Archives of Biochemistry and Biophysics 1955, 54, 162–173. [CrossRef]
- O’Carra, P.; O’hEocha, C. Algal Biliproteins and Phycobilins. In Chemistry and biochemistry of plant pigments; Goodwin, T.W., Ed.; Academic Press: New York, 1965; pp. 328–376 ISBN 978-0-12-289902-7.
- Haxo, F.T.; Fork, D.C. Photoreactive Pigments in Flagellates: Photosynthetically Active Accessory Pigments of Cryptomonads. Nature 1959, 184, 1051–1052. [CrossRef]
- MacColl, R.; Berns, D.S.; Gibbons, O. Characterization of Cryptomonad Phycoerythrin and Phycocyanin. Archives of Biochemistry and Biophysics 1976, 177, 265–275. [CrossRef]
- Ingram, K.; Hiller, R.G. Isolation and Characterization of a Major Chlorophyll Alc2 Light-Harvesting Protein from a Chroomonas Species (Cryptophyceae). Biochimica et Biophysica Acta (BBA) - Bioenergetics 1983, 722, 310–319. [CrossRef]
- Mann, J.E.; Myers, J. Photosynthetic Enhancement in the Diatom Phaeodactylum tricornutum. Plant Physiol 1968, 43, 1991–1995. [CrossRef]
- Goedheer, J.C. Chlorophyll a Forms in Phaeodactylum tricornutum: Comparison with Other Diatoms and Brown Algae. Biochimica et Biophysica Acta (BBA) - Bioenergetics 1973, 314, 191–201. [CrossRef]
- Prézelin, B.B.; Ley, A.C.; Haxo, F.T. Effects of Growth Irradiance on the Photosynthetic Action Spectra of the Marine Dinoflagellate, Glenodinium Sp. Planta 1976, 130, 251–256. [CrossRef]
- Vidaver, W. Separate Action Spectra for the Two Photochemical Systems of Photosynthesis. Plant Physiology 1966, 41, 87–89.
- Johansen, J.E.; Svec, W.A.; Liaaen-Jensen, S.; Haxo, F.T. Carotenoids of the Dinophyceae. Phytochemistry 1974, 13, 2261–2271. [CrossRef]
- Boczar, B.A.; Prézelin, B.B.; Markwell, J.P.; Thornber, J.P. A Chlorophyll c -containing Pigment—Protein Complex from the Marine Dinoflagellate, Glenodinium Sp. FEBS Letters 1980, 120, 243–247. [CrossRef]
- Butler, W.L. On the Primary Nature of Fluorescence Yield Changes Associated with Photosynthesis. Proceedings of the National Academy of Sciences 1972, 69, 3420–3422. [CrossRef]
- Murata, N. The Discovery of State Transitions in Photosynthesis 40 Years Ago. Photosynth Res 2009, 99, 155–160. [CrossRef]
- Lichtlé, C.; Jupin, H.; Duval, J.C. Energy Transfers from Photosystem II to Photosystem I in Cryptomonas rufescens (Cryptophyceae). Biochimica et Biophysica Acta (BBA) - Bioenergetics 1980, 591, 104–112. [CrossRef]
- Bruce, D.; Biggins, J.; Steiner, T.; Thewalt, M. Excitation Energy Transfer in the Cryptophytes. Fluorescence Excitation Spectra and Picosecond Time-Resolved Emission Spectra of Intact Algae at 77 K. Photochemistry and Photobiology 1986, 44, 519–525. [CrossRef]
- Snyder, U.K.; Biggins, J. Excitation-Energy Redistribution in the Cryptomonad Alga Cryptomonas ovata. Biochimica et Biophysica Acta (BBA) - Bioenergetics 1987, 892, 48–55. [CrossRef]
- Krausz, E.; Hughes, J.L.; Smith, P.J.; Pace, R.J.; Årsköld, S.P. Assignment of the Low-Temperature Fluorescence in Oxygen-Evolving Photosystem II. Photosynth Res 2005, 84, 193–199. [CrossRef]
- Mohanty, P.; Allakhverdiev, S.I.; Murata, N. Application of Low Temperatures during Photoinhibition Allows Characterization of Individual Steps in Photodamage and the Repair of Photosystem II. Photosynth Res 2007, 94, 217–224. [CrossRef]
- Romero, E.; van Stokkum, I.H.M.; Novoderezhkin, V.I.; Dekker, J.P.; van Grondelle, R. Two Different Charge Separation Pathways in Photosystem II. Biochemistry 2010, 49, 4300–4307. [CrossRef]
- Luimstra, V.M.; Schuurmans, J.M.; Verschoor, A.M.; Hellingwerf, K.J.; Huisman, J.; Matthijs, H.C.P. Blue Light Reduces Photosynthetic Efficiency of Cyanobacteria through an Imbalance between Photosystems I and II. Photosynth Res 2018, 138, 177–189. [CrossRef]
- Agami, M.; Beer., S.; Waisel, Y. Seasonal Variations in the Growth Capacity of Najas marina L. as a Function of Various Water Depths at the Yarkon Springs, Israel.. Aquatic Botany. 1984, 19, 45–51.
- Agami, M.; Beer, S.; Waisel, Y. Growth and Photosynthesis of Najas marina L. as Affected by Light Intensity. Aquatic Botany 1980, 9, 285–289. [CrossRef]
- Frenette, J.-J.; Massicotte, P.; Lapierre, J.-F. Colorful Niches of Phytoplankton Shaped by the Spatial Connectivity in a Large River Ecosystem: A Riverscape Perspective. PLOS ONE 2012, 7, e35891. [CrossRef]
- Barber, J. Photosynthetic Energy Conversion: Natural and Artificial. Chemical Society Reviews 2009, 38, 185–196. [CrossRef]
- Govindjee; Björn, L.O.; Nickelsen, K. Evolution of the Z-Scheme of Electron Transport in Oxygenic Photosynthesis. In Proceedings of the Photosynthesis Research for Food, Fuel and the Future; Kuang, T., Lu, C., Zhang, L., Eds.; Springer: Berlin, Heidelberg, 2013; pp. 827–833.
- Neori, A.; Haxo, F.T. Effect of Photoadaptation on Action Spectra of Photosynthetic Enhancement in Rhodomonas D3 and in Glenodinium Sp. Photosynthetica (Praha) 1990, 24, 573–585.
- Zhang, Y.-Z.; Li, K.; Qin, B.-Y.; Guo, J.-P.; Zhang, Q.-B.; Zhao, D.-L.; Chen, X.-L.; Gao, J.; Liu, L.-N.; Zhao, L.-S. Structure of Cryptophyte Photosystem II–Light-Harvesting Antennae Supercomplex. Nat Commun 2024, 15, 4999. [CrossRef]
- Siefermann-Harms, D. Carotenoids in Photosynthesis. I. Location in Photosynthetic Membranes and Light-Harvesting Function. Biochimica et Biophysica Acta (BBA) - Reviews on Bioenergetics 1985, 811, 325–355. [CrossRef]
- Kaňa, R.; Kotabová, E.; Sobotka, R.; Prášil, O. Non-Photochemical Quenching in Cryptophyte Alga Rhodomonas Salina Is Located in Chlorophyll a/c Antennae. PLOS ONE 2012, 7, e29700. [CrossRef]
- Staleva, H.; Komenda, J.; Shukla, M.K.; Šlouf, V.; Kaňa, R.; Polívka, T.; Sobotka, R. Mechanism of Photoprotection in the Cyanobacterial Ancestor of Plant Antenna Proteins. Nat Chem Biol 2015, 11, 287–291. [CrossRef]
- Prézelin, B.B.; Alberte, R.S. Photosynthetic Characteristics and Organization of Chlorophyll in Marine Dinoflagellates. Proceedings of the National Academy of Sciences 1978, 75, 1801–1804. [CrossRef]
- Herbert, S.K.; Fork, D.C.; Malkin, S. Photoacoustic Measurements in Vivo of Energy Storage by Cyclic Electron Flow in Algae and Higher Plants 1. Plant Physiology 1990, 94, 926–934. [CrossRef]
- Larkum, A.W.D.; Grossman, A.R.; Raven, J.A. Photosynthesis in Algae: Biochemical and Physiological Mechanisms; Advances in Photosynthesis and Respiration; Springer International Publishing: Cham, 2020; Vol. 45; ISBN 978-3-030-33396-6.
- Johnsen, G.; Sakshaug, E. Biooptical Characteristics of PSII and PSI in 33 Species (13 Pigment Groups) of Marine Phytoplankton, and the Relevance for Pulse-Amplitude-Modulated and Fast-Repetition-Rate Fluorometry. Journal of Phycology 2007, 43, 1236–1251. [CrossRef]
- Johnsen, G.; Sakshaug, E. Bio-Optical Characteristics and Photoadaptive Responses in the Toxic and Bloom-Forming Dinoflagellates Gyrodinium aureolum, Gymnodinium galatheanum, and Two Strains of Prorocentrum minimum. Journal of Phycology 1993, 29, 627–642. [CrossRef]
- Zhen, S.; Bugbee, B. Far-Red Photons Have Equivalent Efficiency to Traditional Photosynthetic Photons: Implications for Redefining Photosynthetically Active Radiation. Plant, Cell & Environment 2020, 43, 1259–1272. [CrossRef]
- Oliveira, C.Y.B.; Abreu, J.L.; Santos, E.P.; Matos, Â.P.; Tribuzi, G.; Oliveira, C.D.L.; Veras, B.O.; Bezerra, R.S.; Müller, M.N.; Gálvez, A.O. Light Induces Peridinin and Docosahexaenoic Acid Accumulation in the Dinoflagellate Durusdinium glynnii. Appl Microbiol Biotechnol 2022, 106, 6263–6276. [CrossRef]
- Millie, D.F.; Kirkpatrick, G.J.; Vinyard, B.T. Relating Photosynthetic Pigments and in Vivo Optical Density Spectra to Irradiance for the Florida Red-Tide Dinoflagellate Gymnodinium breve. Marine Ecology Progress Series 1995, 120, 65–75.
- Warner, M.E.; Berry-Lowe, S. Differential Xanthophyll Cycling and Photochemical Activity in Symbiotic Dinoflagellates in Multiple Locations of Three Species of Caribbean Coral. Journal of Experimental Marine Biology and Ecology 2006, 339, 86–95. [CrossRef]
- Vernet, M.; Neori, A.; Haxo, F.T. Spectral Properties and Photosynthetic Action in Red-Tide Populations of Prorocentrum micans and Gonyaulax polyedra. Mar. Biol. 1989, 103, 365–371. [CrossRef]
- Dionisio-sese, M.L. Aquatic Microalgae As Potential Sources Of UV-Screening Compounds. Philippine Journal of Science 2010, 139, 5–16.
- Rastogi, R.P.; Richa; Sinha, R.P.; Singh, S.P.; Häder, D.-P. Photoprotective Compounds from Marine Organisms. Journal of Industrial Microbiology and Biotechnology 2010, 37, 537–558. [CrossRef]
- López-Rosales, L.; Gallardo-Rodríguez, J.J.; Sánchez-Mirón, A.; Cerón-García, M.D.C.; Belarbi, E.H.; García-Camacho, F.; Molina-Grima, E. Simultaneous Effect of Temperature and Irradiance on Growth and Okadaic Acid Production from the Marine Dinoflagellate Prorocentrum belizeanum. Toxins 2014, 6, 229–253. [CrossRef]
- Martín-Girela, I.; Curt, M.D.; Fernández, J. Flashing Light Effects on CO2 Absorption by Microalgae Grown on a Biofilm Photobioreactor. Algal Research 2017, 25, 421–430. [CrossRef]
- Healey, F.P. The Carotenoids of Four Blue-Green Algae. Journal of Phycology 1968, 4, 126–129. [CrossRef]
- Loeblich, A.R., III; Schmidt, R.J.; Sherley, J.L. Scanning Electron Microscopy of Heterocapsa pygmaea Sp. Nov., and Evidence for Polyploidy as a Speciation Mechanism in Dinoflagellates. Journal of Plankton Research 1981, 3, 67–79. [CrossRef]
- Loeblich, A.R. A Seawater Medium for Dinoflagellates and the Nutrition of Cachonina niei. Journal of Phycology 1975, 11, 80–86. [CrossRef]
- Haxo, F.T.; Lewin, R.A.; Lee, K.W.; Li, M.-R. Fine Structure and Pigments of Oscillatoria(Trichodesmium) Aff. thiebautii (Cyanophyta) in Culture. Phycologia 1987, 26, 443–456. [CrossRef]
- McCarthy, J.J.; Carpenter, E.J. Oscillatoria(Trichodesmium) thiebautii (Cyanophyta) in the Central North Atlantic Ocean. Journal of Phycology 1979, 15, 75–82. [CrossRef]
- Duysens, L. Transfer of Excitation Energy in Photosynthesis, 1952.
| Variable | Definition | Units |
|---|---|---|
| AC | Signal of modulated photosynthetic oxygen evolution resulting from the modulated measuring light beam of intensity Iₘ. | Arbitrary (voltage) |
| Iₘ | Intensity of the modulated measuring radiant beam. | µmol photons·m-²·s-¹ |
| Ib | Intensity of the continuous background radiant beam. | µmol photons·m-²·s-¹ |
| Is | A weak scanning beam | µmol photons·m-²·s-¹ |
| U | Fraction of open Photosystem II reaction centers. | Dimensionless (range: 0-1) |
| Ub | Intensity of a background beam that controls the opening state of Photosystem II reaction centers. | Dimensionless (range: 0-1) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




