Submitted:
27 January 2026
Posted:
28 January 2026
You are already at the latest version
Abstract
Keywords:
I. Introduction
1.1. Toll-like Receptors: Mechanisms and Potential as Vaccine Adjuvants
1.2. Innate Immune Signaling as a Determinant of Adaptive Immune Outcomes
1.3. Molecular Determinants of TLR Activation and Their Immunological Consequences
1.4. Vaccine Development and the Significance of Adjuvants
1.4.1. Progression of Vaccine Adjuvant Development and Innate Immune Understanding
1.4.2. Mechanistic and Clinical Considerations in Next-Generation Vaccine Platforms
1.5. Mechanistic and Translational Synthesis of TLR Agonist–Based Adjuvant Research
II. Molecular and Immunological Basis of Toll-like Receptors and Their Agonists
2.1. Toll-like Receptors (TLRs)
- A. Domain Organization and Signaling Mechanisms of Toll-Like Receptors
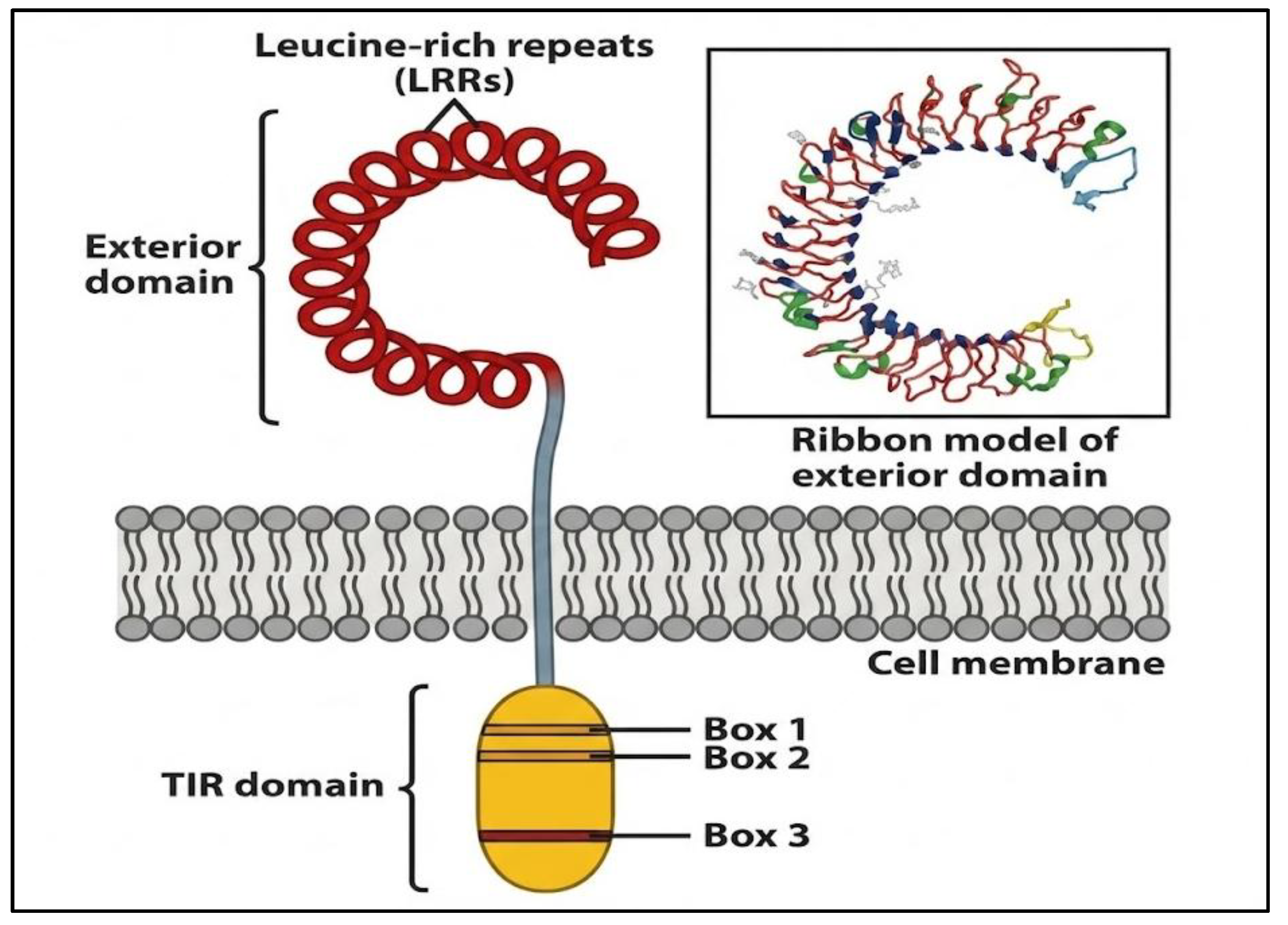
- B. Divergent Signaling Pathways of TLRs: MyD88 vs. TRIF
2.2. Toll-like Receptor Agonists
2.2.1. TLR Agonists’ Molecular Activation and Mechanism
2.3. TLRs in the Immune System
2.3.1. TLRs on Dendritic Cells (DCs)
2.3.2. TLRs on Macrophages
2.3.3. TLRs on B-Cells
2.4. Host-Dependent Modulation of Toll-like Receptor Activity
2.5. Specific Involvement of TLR Agonists on Vaccine Development
| Vaccine | Target Pathogen | TLR Agonist / Adjuvant | Formulation | Key Findings |
| Cervarix | HPV | TLR4 agonist: MPL | AS04 adjuvant | Enhanced immune response |
| Fendrix | Hepatitis B | TLR4 agonist: MPL | AS04 adjuvant | Enhanced immune response |
| Experimental Mucosal Vaccine | DQ-OVA (respiratory model antigen) | TLR5 agonist: Flagellin (FLA-ST) | Intranasal protein subunit vaccine | Strong activation of neonatal lung APCs |
| Heplisav-B | Hepatitis B | TLR9 agonist: CpG-1018 | Subunit vaccine | Enables two-dose schedule |
| 1790GAHB | Shigella sonnei | OMVs (GMMA) with inherent TLR agonists | OMV-based nanoparticle vaccine | Phase 1 trial: safe, strong humoral & cellular responses |
2.6. Nanotechnology and Formulation Strategies in TLR-Based Vaccines
III. Systematic Classification of Toll-like Receptors Relevant to Immune Adjuvant Development
3.1. Receptor Family: Pattern Recognition Receptors (PRRs)
3.1.1. Receptor Subgroup: Toll-like Receptors (TLRs)
3.1.1.1. Structural and Functional Evolution of Toll-like Receptors
3.2. TLRs’ Ligand Recognition and Structural Characteristics
3.3. Classification of TLRs and Their Agonists
3.3.1. Agonist-Specific TLR Signaling and Downstream Immunological Effects
| TLR | Agonist | Key Signaling Adaptors | Primary Ligands | Functional Role in Adjuvanticity | Dominant Immune Responses |
| TLR2 | Pam3CSK4 (synthetic), MALP-2 (natural) | MyD88-dependent pathway |
Bacterial lipoproteins and lipopeptides | Enhances innate immune activation and antigen presentation | Pro-inflammatory cytokine production |
| TLR3 | Poly I:C, Ampligen, ARNAX | TRIF-dependent pathway | Double-stranded RNA (dsRNA) | Promotes robust antiviral immunity | Strong type I interferon (IFN-α/β) production |
| TLR4 | Monophosphoryl lipid A (MPLA) | MyD88-dependent pathway | Single-stranded RNA (ssRNA) | Potent immune activation; high adjuvant efficacy | Robust pro-inflammatory cytokine production |
| TLR5 | Flagellin (natural or recombinant) | MyD88-dependent pathway |
Bacterial flagellin | Promotes DC maturation and | Pro-inflammatory cytokine production |
| TLR7/8 | Imiquimod, Resiquimod (R848) | MyD88-dependent pathway | Single-stranded RNA (ssRNA) | Enhancement of antiviral immunity | Strong antiviral responses; type I IFN production |
| TLR 9 | CpG oligodeoxynucleotides | MyD88-dependent pathway | Single-stranded RNA (ssRNA) | Promotes cellular and humoral immune responses | Th1-biased immunity; B-cell activation and antibody enhancement |
IV. Spatial and Cellular Distribution of Toll-like Receptors
4.1. Tissue- and Cell-Specific Distribution of Toll-like Receptors
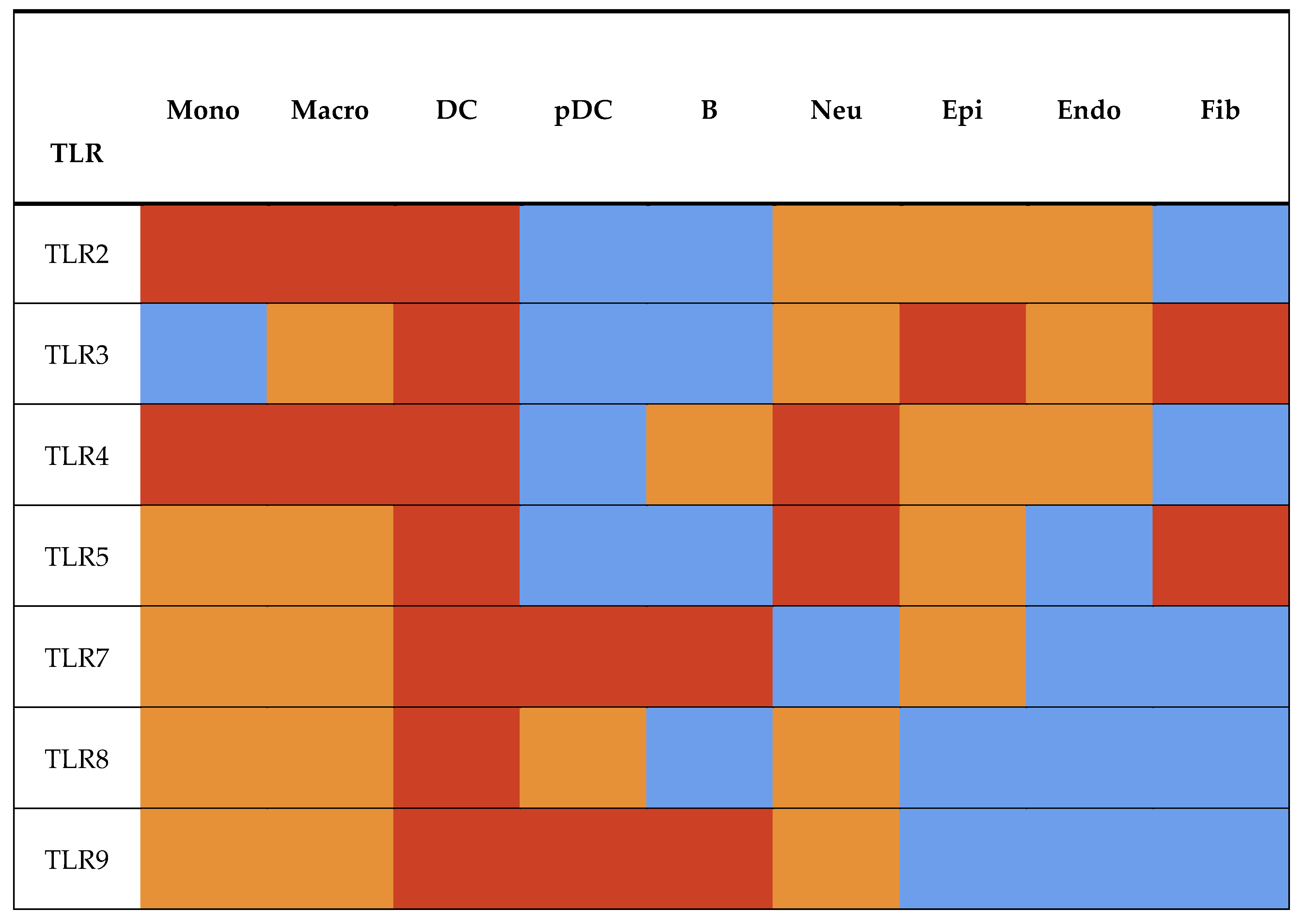
4.1.1. Structural Divergence of mccTLRs and sccTLRs and Its Impact on Tissue-Specific TLR Expression
4.2. TLR Expression in Immune Cells
| TLR | Historical Group | Tissue and Organ Distribution | Dominant Cellular Distribution | Cellular Lineage | Function |
| TLR2 | single cysteine cluster (sccTLRs) | Barrier tissues; lymphoid organs | Myeloid cells; barrier-associated non-immune cells | Immune and Non-immune Cells | Senses bacterial lipoproteins, resulting in pro-inflammatory innate signaling |
| TLR3 | single cysteine cluster (sccTLRs) | Barrier and lymphoid tissues | Dendritic cells; stromal cells | Immune and Non-immune cells | Detects dsRNA, leading to antiviral/type I IFN responses |
| TLR4 | single cysteine cluster (sccTLRs) | Systemic organs | Myeloid antigen-presenting cells | Immune and Non-immune cells | Functions in systemic inflammatory responses |
| TLR5 | single cysteine cluster (sccTLRs) | Barrier tissues; mucosal surfaces | Monocytes; dendritic cells; epithelial cells | Immune and non-immune cells | Activation of innate antibacterial immunity |
| TLR7/8 | single cysteine cluster (sccTLRs) | Lymphoid and mucosal tissues | pDCs, B cells; and myeloid cells | Immune cells | Initiates antiviral immune responses |
| TLR9 | single cysteine cluster (sccTLRs) | Hematopoietic and lymphoid tissues | B cells; plasmacytoid DCs | Immune cells | Regulates both the hematopoietic and immune systems |
4.3. Precision Immunization Through TLR Subtype Targeting
V. Translational Applications and Strategies of TLR Agonists in Vaccine Development and Immune Regulation
5.1. Trends in Increasing Use of TLR Agonists in Adjuvant Study
5.1.1. Monophosphoryl Lipid A (MPLA) as Clinically Approved TLR4-Based Vaccine Adjuvant
5.1.2. Flagellin-Mediated TLR5 Activation Enhancing Antigen-Specific Immunity in Vaccine Models
5.1.3. TLR7 and TLR8 Agonists as Complementary Adjuvants in TLR4-Based Vaccine Strategies
5.1.4. TLR9 Agonists as Clinically Validated Adjuvants Supporting TLR4-Focused Vaccines
| Target TLR | Representative Agonist(s) |
Primary Application |
Immune Effect | Year and Status |
| TLR3 | ARNAX, Poly ICLC (Hiltonol), HSPPC-96 |
Cancer | Immune modulation enhances checkpoint therapy ; improves antiviral & antibacterial responses | 2018-2024, Experimental |
| TLR4 | MPL | Hepatitis B, Multivalent vaccines |
Enhanced cellular and humoral immune responses |
2025, Licensed |
| TLR5 | Flagellin | Influenza, bacterial vaccines, mucosal vaccines |
Potent innate activation; enhanced antigen presentation | 202 5 Experimental |
| TLR7&8 | Synthetic imidazoquinoline, lipid-modified | Influenza, Cancer |
Strong Th1/Th17 polarization and cytotoxic T-cell activation | 2020-2023, Experimental |
| TLR9 | CpG-ODN 1018 |
Hepatitis B | Higher seroprotection rates than alum-adjuvanted vaccines | 2015, Licensed |
| TLR4 + TLR7&8 | MPLA + lipid-modified TLR 7&8 | SARS-CoV-2, Influenza |
Synergistic, stronger, and sustained immune responses at reduced doses |
2023, Experimental |
| TLR3, TLR4, TLR9 |
Synthetic agonists |
Cancer, Infectious diseases |
Immune modulation; TME alteration enhance checkpoint therapy | 2018-2024, Experimental |
5.2. Combination and Emerging TLR Agonist Formulations
5.3. Therapeutic and Immunological Applications of Toll-like Receptor Agonists
5.3.1. Immunotherapy
VI. Conceptual and Translational Limitations in TLR Agonist–Based Adjuvant Development
6.1. Evolving Perspectives on the Complexity of Toll-like Receptor Signaling
6.1.1. Barriers to Experimental Integration of In Silico Modeling and TLR-Mediated Immune Responses
6.1.2. Underutilization of Multi-Agonist Approaches in Adjuvant Development
VII. Conclusion and Future Directions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Ethics Statement
References
- Pulendran, B; S. Arunachalam, P; O’Hagan, DT. Emerging concepts in the science of vaccine adjuvants. Nature reviews Drug discovery 2021, 20(6), 454–75. [Google Scholar] [CrossRef]
- Kayesh, MEH; Kohara, M; Tsukiyama-Kohara, K. TLR agonists as vaccine adjuvants in the prevention of viral infections: an overview. Front Microbiol. 2023, 14, 1249718. [Google Scholar] [CrossRef]
- Ou, BS; Baillet, J; Filsinger Interrante, MV; Adamska, JZ; Zhou, X; Saouaf, OM; et al. Saponin nanoparticle adjuvants incorporating Toll-like receptor agonists drive distinct immune signatures and potent vaccine responses. Sci Adv. 2024, 10(32), eadn7187. [Google Scholar] [CrossRef]
- Zhang, P; Yang, M; Chen, C; Liu, L; Wei, X; Zeng, S. Toll-like receptor 4 (TLR4)/opioid receptor pathway crosstalk and impact on opioid analgesia, immune function, and gastrointestinal motility. Front Immunol. 2020, 11, 1455. [Google Scholar] [CrossRef] [PubMed]
- Luo, L; Lucas, RM; Liu, L; Stow, JL. Signalling, sorting and scaffolding adaptors for Toll-like receptors. J Cell Sci. 2020, 133(5), jcs239194. [Google Scholar] [CrossRef]
- Ko, KH; Cha, SB; Lee, SH; Bae, HS; Ham, CS; Lee, MG; Kim, DH; Han, SH. A novel defined TLR3 agonist as an effective vaccine adjuvant. Frontiers in Immunology 2023, 14, 1075291. [Google Scholar] [CrossRef] [PubMed]
- Maisonneuve, C; Bertholet, S; Philpott, DJ; De Gregorio, E. Unleashing the potential of NOD-and Toll-like agonists as vaccine adjuvants. Proceedings of the National Academy of Sciences 2014, 111(34), 12294–9. [Google Scholar] [CrossRef]
- Ozverel, CS; Erdag, E. Investigation of the molecular interactions of vaccine adjuvants: can a strategic trio of toll-like receptor agonists enhance efficacy in a multifaceted approach? Biomed Biotechnol Res J 2024, 8(1), 27–36. [Google Scholar] [CrossRef]
- Kadivella, M; Varma, VP; Cp, J; Kavela, S; Azam, S; Faisal, SM. Adjuvant activity of a small molecule TLR4 agonist discovered via structure-based virtual screening. Commun Biol. 2025, 8(1), 1382. [Google Scholar] [CrossRef] [PubMed]
- Knisely, JM; Erbelding, E. Vaccines for global health: progress and challenges. J Infect Dis. 2025, 232(1), 25–27. [Google Scholar] [CrossRef]
- Cui, Y; Ho, M; Hu, Y; Shi, Y. Vaccine adjuvants: current status, research and development, licensing, and future opportunities. J Mater Chem B 2024, 12(17), 4118–4137. [Google Scholar] [CrossRef]
- Cao, LM; Yu, YF; Li, ZZ; Zhong, NN; Wang, GR; Xiao, Y; et al. Adjuvants for cancer mRNA vaccines in the era of nanotechnology: strategies, applications, and future directions. J Nanobiotechnology. 2024, 22(1), 308. [Google Scholar] [CrossRef]
- Do, KTH; Willenzon, S; Ristenpart, J; Janssen, A; Volz, A; Sutter, G; et al. The effect of Toll-like receptor agonists on the immunogenicity of MVA-SARS-2-S vaccine after intranasal administration in mice. Front Cell Infect Microbiol. 2023, 13, 1259822. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Tejada, A; Tan, DS; Gin, DY. Development of improved vaccine adjuvants based on the saponin natural product QS-21 through chemical synthesis. Accounts of Chemical Research 2016, 49(9), 1741–56. [Google Scholar] [CrossRef]
- Moni, SS; Abdelwahab, SI; Jabeen, A; Elmobark, ME; Aqaili, D; Gohal, G; Oraibi, B; Farasani, AM; Jerah, AA; Alnajai, MM; Mohammad Alowayni, AM. Advancements in vaccine adjuvants: the journey from alum to nano formulations. Vaccines 2023, 11(11), 1704. [Google Scholar] [CrossRef]
- Zhao, T; Cai, Y; Jiang, Y; He, X; Wei, Y; Yu, Y; Tian, X. Vaccine adjuvants: mechanisms and platforms. Signal transduction and targeted therapy 2023, 8(1), 283. [Google Scholar] [CrossRef] [PubMed]
- Manicassamy, S; Pulendran, B. Modulation of adaptive immunity with Toll-like receptors. In InSeminars in immunology; Academic Press, 1 Aug 2009; Vol. 21, No. 4, pp. 185–193. [Google Scholar] [CrossRef]
- Oboge, H; Riitho, V; Nyamai, M; Omondi, GP; Lacasta, A; Githaka, N; et al. Safety and efficacy of toll-like receptor agonists as therapeutic agents and vaccine adjuvants for infectious diseases in animals: a systematic review. Front Vet Sci. 2024, 11, 1428713. [Google Scholar] [CrossRef] [PubMed]
- Temchura, V; Wagner, JT; Damm, D. Immunogenicity of recombinant lipid-based nanoparticle vaccines: danger signal vs. helping hand. Pharmaceutics 2023, 16(1), 24. [Google Scholar] [CrossRef]
- Behzadi, P.; García-Perdomo, H. A.; Karpiński, T. M. Toll-like receptors: general molecular and structural biology. Journal of Immunology Research 2021, 2021(1), 9914854. [Google Scholar] [CrossRef]
- Xun, Y.; Yang, H.; Kaminska, B.; You, H. Toll-like receptors and toll-like receptor-targeted immunotherapy against glioma. Journal of hematology & oncology 2021, 14(1), 176. Available online: https://link.springer.com/article/10.1186/s13045-021-01191-2.
- Shi, M.; Chen, X.; Ye, K.; Yao, Y.; Li, Y. Application potential of toll-like receptors in cancer immunotherapy: Systematic review. Medicine 2016, 95(25), e3951. [Google Scholar] [CrossRef]
- Luchner, M.; Reinke, S.; Milicic, A. TLR agonists as vaccine adjuvants targeting cancer and infectious diseases. Pharmaceutics 2021, 13(2), 142. [Google Scholar] [CrossRef]
- Tiwari, U.; Das, S.; Tandon, M.; Ramachandran, V.; Saha, R. Vaccines for fungal infections . The National Medical Journal of India 2015, 28(1), 14–19. Available online: https://www.researchgate.net/publication/280561624_Vaccines_for_fungal_infections.
- Ouyang, MZ; Zhou, D; Zhu, Y; Zhang, M; Li, L. The inhibition of MyD88 and TRIF signaling serve equivalent roles in attenuating myocardial deterioration due to acute severe inflammation. Int J Mol Med. 2018, 41(1), 399–408. [Google Scholar] [CrossRef]
- Androulidaki, A; Wachsmuth, L; Polykratis, A; Pasparakis, M. Differential role of MyD88 and TRIF signaling in myeloid cells in the pathogenesis of autoimmune diabetes. PLoS One 2018, 13(3), e0194048. [Google Scholar] [CrossRef] [PubMed]
- Pang, Z; Junkins, RD; Raudonis, R; MacNeil, AJ; McCormick, C; Cheng, Z; Lin, TJ. Regulator of calcineurin 1 differentially regulates TLR-dependent MyD88 and TRIF signaling pathways. PLoS One 2018, 13(5), e0197491. [Google Scholar] [CrossRef] [PubMed]
- Rink, L; Hebel, T; Fukumot, J. Sepsis and signal transduction pathway: cross-talk TLR4/MyD88/TRIF. Am J Biomed Sci. 2015, 3(4), 150–163. [Google Scholar] [CrossRef]
- Yang, J. X.; Tseng, J. C.; Yu, G. Y.; Luo, Y.; Huang, C. Y. F.; Hong, Y. R.; Chuang, T. H. Recent advances in the development of toll-like receptor agonist-based vaccine adjuvants for infectious diseases. Pharmaceutics 2022, 14(2), 423. [Google Scholar] [CrossRef]
- Fitzgerald, K. A.; Kagan, J. C. Toll-like receptors and the control of immunity. Cell 2020, 180(6), 1044–1066. Available online: https://www.cell.com/cell/fulltext/S0092-8674(20)30218-X. [CrossRef]
- Anthoney, N.; Foldi, I.; Hidalgo, A. Toll and Toll-like receptor signalling in development. Development 2018, 145(9), dev156018. [Google Scholar] [CrossRef]
- Seya, T.; Takeda, Y.; Matsumoto, M. A Toll-like receptor 3 (TLR3) agonist ARNAX for therapeutic immunotherapy. Advanced Drug Delivery Reviews 2019, 147, 37–43. [Google Scholar] [CrossRef]
- Owen, A. M.; Fults, J. B.; Patil, N. K.; Hernandez, A.; Bohannon, J. K. TLR agonists as mediators of trained immunity: mechanistic insight and immunotherapeutic potential to combat infection. Frontiers in immunology 2021, 11, 622614. [Google Scholar] [CrossRef]
- Buchta, C. M.; Bishop, G. A. Toll-like receptors and B cells: functions and mechanisms. Immunologic research 2014, 59(1-3), 12–22. [Google Scholar] [CrossRef]
- Mancini, F.; Rossi, O.; Necchi, F.; Micoli, F. OMV vaccines and the role of TLR agonists in immune response. International journal of molecular sciences 2020, 21(12), 4416. [Google Scholar] [CrossRef]
- Fischinger, S; Boudreau, CM; Butler, AL; Streeck, H; Alter, G. Sex differences in vaccine-induced humoral immunity. Semin Immunopathol 2019, 41(2), 239–249. [Google Scholar] [CrossRef] [PubMed]
- Rio, P; Caldarelli, M; Chiantore, M; Ocarino, F; Candelli, M; Gasbarrini, A; et al. Immune cells, gut microbiota, and vaccines: a gender perspective. Cells 2024, 13(6), 526. [Google Scholar] [CrossRef]
- Martínez-García, MÁ; Ojeda-Ojeda, M; Rodríguez-Martín, E; Insenser, M; Moncayo, S; Álvarez-Blasco, F; et al. TLR2 and TLR4 surface and gene expression in white blood cells after fasting and oral glucose, lipid and protein challenges: influence of obesity and sex hormones. Biomolecules 2020, 10(1), 111. [Google Scholar] [CrossRef]
- Sharma, P; Levy, O; Dowling, DJ. The TLR5 agonist flagellin shapes phenotypical and functional activation of lung mucosal antigen-presenting cells in neonatal mice. Front Immunol. 2020, 11, 171. [Google Scholar] [CrossRef] [PubMed]
- Yang, JX; Tseng, JC; Yu, GY; Luo, Y; Huang, CYF; Hong, YR; Chuang, TH. Recent advances in the development of toll-like receptor agonist-based vaccine adjuvants for infectious diseases. Pharmaceutics 2022, 14(2), 423. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, S; Ye, J; Wang, H; Sun, M; Zhang, Y; Sang, X; Zhuang, Z. Application of toll-like receptors (TLRs) and their agonists in cancer vaccines and immunotherapy. Front Immunol. 2023, 14, 1227833. [Google Scholar] [CrossRef]
- Díaz-Dinamarca, DA; Salazar, ML; Castillo, BN; Manubens, A; Vasquez, AE; Salazar, F; Becker, MI. Protein-based adjuvants for vaccines as immunomodulators of the innate and adaptive immune response: current knowledge, challenges, and future opportunities. Pharmaceutics 2022, 14(8), 1671. [Google Scholar] [CrossRef]
- Takeuchi, O; Akira, S. Pattern recognition receptors and inflammation. Cell. 2016, 164(6), 1233–1247. [Google Scholar] [CrossRef] [PubMed]
- Brennan, JJ; Gilmore, TD. Evolutionary Origins of Toll-like Receptor Signaling. Mol Biol Evol 2018, 35(7), 1576-1587. Available online: https://academic.oup.com/mbe/article/35/7/1576/4994247. [CrossRef] [PubMed]
- Li, H; Li, X; Zhu, Y; Xu, J; Zhao, Q; Chen, X; et al. Systematic review of structural and immunological features of mollusk toll-like receptors in aquaculture context. Front Immunol 2025, 16, 1671434. Available online: https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2025.1671434/full. [CrossRef]
- Liu, G; Zhang, H; Zhao, C; Zhang, H. Evolutionary History of the Toll-Like Receptor Gene Family across Vertebrates. Genome Biol Evol 2020, 12(1), 3615-3634. Available online: https://pubmed.ncbi.nlm.nih.gov/31800025/. [CrossRef] [PubMed] [PubMed Central]
- Gay, NJ; Symmons, MF; Gangloff, M; Bryant, CE. Assembly and localization of Toll-like receptor signalling complexes. Nat Rev Immunol. 2018, 18(9), 546–558. [Google Scholar] [CrossRef]
- Patra, MC; Choi, S. Recent progress in the development of Toll-like receptor (TLR) agonists. Expert Opin Ther Pat. 2016, 26(6), 719–730. [Google Scholar] [CrossRef]
- Afzal, H; Murtaza, A; Cheng, LT. Structural engineering of flagellin as vaccine adjuvant: quest for the minimal domain of flagellin for TLR5 activation. Mol Biol Rep. 2025, 52(1), 104. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- De Gregorio, E; Caproni, E; Ulmer, JB. Vaccine adjuvants: mode of action. Front Immunol. 2013, 4, 214. [Google Scholar] [CrossRef] [PubMed]
- Pergolizzi, S; Fumia, A; D’Angelo, R; Mangano, A; Lombardo, GP; Giliberti, A; et al. Expression and function of toll-like receptor 2 in vertebrate. Acta Histochemica 2023, 125(3), 152028. [Google Scholar] [CrossRef] [PubMed]
- Zhang, SY; Herman, M; Ciancanelli, MJ; Pérez de Diego, R; Sancho-Shimizu, V; Abel, L; et al. TLR3 immunity to infection in mice and humans. Current Opinion in Immunology 2013, 25(1), 19–33. [Google Scholar] [CrossRef]
- Duan, T; Du, Y; Xing, C; Wang, HY; Wang, RF. Toll-Like Receptor Signaling and Its Role in Cell-Mediated Immunity. Frontiers in Immunology 2022, 13. [Google Scholar] [CrossRef] [PubMed]
- Abarca-Merlin, DM; Martínez-Durán, JA; Medina-Pérez, JD; Rodríguez-Santos, G; Alvarez-Arellano, L. From Immunity to Neurogenesis: Toll-like Receptors as Versatile Regulators in the Nervous System. International Journal of Molecular Sciences 2024, 25(11), 5711. [Google Scholar] [CrossRef]
- Mukherjee, S; Karmakar, S; Babu, SPS. TLR2 and TLR4 mediated host immune responses in major infectious diseases: a review. The Brazilian Journal of Infectious Diseases 2016, 20(2), 193–204. [Google Scholar] [CrossRef]
- Chen, Y; Lin, J; Zhao, Y; Ma, X; Yi, H. Toll-like receptor 3 (TLR3) regulation mechanisms and roles in antiviral innate immune responses. Journal of Zhejiang University-SCIENCE B 2021, 22(8), 609–32. [Google Scholar] [CrossRef]
- Lim, JS; Jeon, EJ; Go, HS; Kim, HJ; Kim, KY; Nguyen, TQT; et al. Mucosal TLR5 activation controls healthspan and longevity. Nature Communications 2024, 15(1). [Google Scholar] [CrossRef] [PubMed]
- Pellegrino, P; Falvella, FS; Cheli, S; Perrotta, C; Clementi, E; Radice, S. The role of Toll-like receptor 4 polymorphisms in vaccine immune response. Pharmacogenomics J 2016, 16(1), 96–101. [Google Scholar] [CrossRef] [PubMed]
- Skevaki, C; Pararas, M; Kostelidou, K; Tsakris, A; Routsias, JG. Single nucleotide polymorphisms of Toll-like receptors and susceptibility to infectious diseases. Clin Exp Immunol. 2015, 180(2), 165–177. [Google Scholar] [CrossRef]
- Jeon, D; Hill, E; McNeel, DG. Toll-like receptor agonists as cancer vaccine adjuvants. Human Vaccines & Immunotherapeutics 2023, 20(1). [Google Scholar] [CrossRef]
- Lian, S; Yang, W; Zeng, Y; Tang, R; Wang, K. Targeted nano-drug delivery systems for tumor immunotherapy. Journal of Pharmaceutical Analysis 2025, 101408. [Google Scholar] [CrossRef]
- Liew, C. W.; Polanco, L. F. C.; Manalang, K.; Kurt, R. A.; others. An experimental and computational approach to unraveling interconnected TLR signaling cascades. Informatics in Medicine Unlocked 30 2022, 100939. [Google Scholar] [CrossRef]
- Carter, D; De La Rosa, G; Garçon, N; Moon, HM; Nam, HJ; Skibinski, DAG. The success of toll-like receptor 4 based vaccine adjuvants. Vaccine 2025, 61, 127413. [Google Scholar] [CrossRef]
- Choi, H; Lee, S; Kim, H; Bae, SH; Jo, S; Kim, J; et al. Integration of TLR7/8 agonists into lipid nanoparticles enhances antigen-specific immune responses to N1-methyl-Ψ-modified mRNA-LNP vaccines. Journal of Biological Engineering 2025, 19(1). [Google Scholar] [CrossRef] [PubMed]
- Afzal, H; Hoa, NT; Murtaza, A; Doan, TD; Chung, YC; Ke, GM; et al. Conserved domains of flagellin as adjuvants for an inactivated vaccine against porcine epidemic diarrhea virus. The Veterinary Journal 2025, 313, 106393. [Google Scholar] [CrossRef] [PubMed]
- Li, X; Cao, Y; Mou, M; Li, J; Huang, S; Zhang, E; et al. Enhanced TLR5-dependent migration and activation of antigen-loaded airway dendritic cells by flagellin. Journal of Leukocyte Biology 2023, 113(6), 567–76. [Google Scholar] [CrossRef] [PubMed]
- Pang, S; Liu, M; Wang, L; Shao, M; Zhu, G; Duan, Q. Differential Adjuvant Activity by Flagellins from Escherichia coli, Salmonella enterica Serotype Typhimurium, and Pseudomonas aeruginosa. Vaccines 2024, 12(11), 1212. [Google Scholar] [CrossRef]
- Wang, K; Huang, H; Zhan, Q; Ding, H; Li, Y. Toll-like receptors in health and disease. MedComm 2024, 5(5). [Google Scholar] [CrossRef]
- Crouse, B; Miller, SM; Muelken, P; Hicks, L; Vigliaturo, JR; Marker, CL; et al. A TLR7/8 agonist increases efficacy of anti-fentanyl vaccines in rodent and porcine models. npj Vaccines 2023, 8(1). [Google Scholar] [CrossRef]
- Dowling, DJ. Recent Advances in the Discovery and Delivery of TLR7/8 Agonists as Vaccine Adjuvants. ImmunoHorizons 2018, 2(6), 185–97. [Google Scholar] [CrossRef]
- Janssen, JM; Jackson, S; Heyward, WL; Janssen, RS. Immunogenicity of an investigational hepatitis B vaccine with a toll-like receptor 9 agonist adjuvant (HBsAg-1018) compared with a licensed hepatitis B vaccine in subpopulations of healthy adults 18–70 years of age. Vaccine 2015, 33(31), 3614–8. [Google Scholar] [CrossRef]
- Siram, K; Lathrop, SK; Abdelwahab, WM; Tee, R; Davison, CJ; Partlow, HA; et al. Co-Delivery of Novel Synthetic TLR4 and TLR7/8 Ligands Adsorbed to Aluminum Salts Promotes Th1-Mediated Immunity against Poorly Immunogenic SARS-CoV-2 RBD. Vaccines 2023, 12(1), 21. [Google Scholar] [CrossRef]
- Miller, SM; Cybulski, V; Whitacre, M; Bess, LS; Livesay, MT; Walsh, L; et al. Novel Lipidated Imidazoquinoline TLR7/8 Adjuvants Elicit Influenza-Specific Th1 Immune Responses and Protect Against Heterologous H3N2 Influenza Challenge in Mice. Frontiers in Immunology 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Kuai, R; Sun, X; Yuan, W; Ochyl, LJ; Xu, Y; Hassani Najafabadi, A; et al. Dual TLR agonist nanodiscs as a strong adjuvant system for vaccines and immunotherapy. Journal of Controlled Release 2018, 282, 131–9. [Google Scholar] [CrossRef] [PubMed]
- Omange, R. W.; Kim, J.; Kolhatkar, P. P.; Plott, D. M.; Van Trump, J. I.; Zhang, L.; O’Donnell, T. A.; Chen, N.–H.; Hosny, A.; Wiest, J. S.; Barry, A. H.; Addiego, E. N.; Mengistu, A.; Odorizzi, P. M.; Cai, Y.; Jacobson, R. M.; Wallin, J. J. AI discovery of TLR agonist-driven phenotypes reveals unique immune responses in people living with HIV. Frontiers in Immunology 16 2025, Article 1541152. [Google Scholar] [CrossRef]
- Manček-Keber, M.; Jerala, R. Postulates for validating TLR4 agonists. European Journal of Immunology 2015, 45(6), 1510–1516. [Google Scholar] [CrossRef] [PubMed]
- Shanmugam, A.; Rajoria, S.; George, A. L.; Mittelman, A.; Suriano, R.; Tiwari, R. K. Synthetic toll-like receptor 4 agonist peptides as a novel class of adjuvants. PLOS ONE 2012, 7(3), e30839. [Google Scholar] [CrossRef]
- Nouri-Shirazi, M.; Tamjidi, S.; Nourishirazi, E.; Guinet, E. Combination of TLR8 and TLR4 agonists reduces the degrading effects of nicotine on DC-NK mediated effector T cell generation. International immunopharmacology 2018, 61, 54–63. [Google Scholar] [CrossRef]
- Abiri, A.; Rezaei, M.; Zeighami, M. H.; Vaezpour, Y.; Dehghan, L.; KhorramGhahfarokhi, M. Discovery of new TLR7 agonists by a combination of statistical learning-based QSAR, virtual screening, and molecular dynamics. Informatics in medicine unlocked 2021, 27, 100787. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




