Submitted:
26 January 2026
Posted:
27 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. The Aged Muscle Stem Cell Niche and the Fragility of Early Satellite Cell Activation
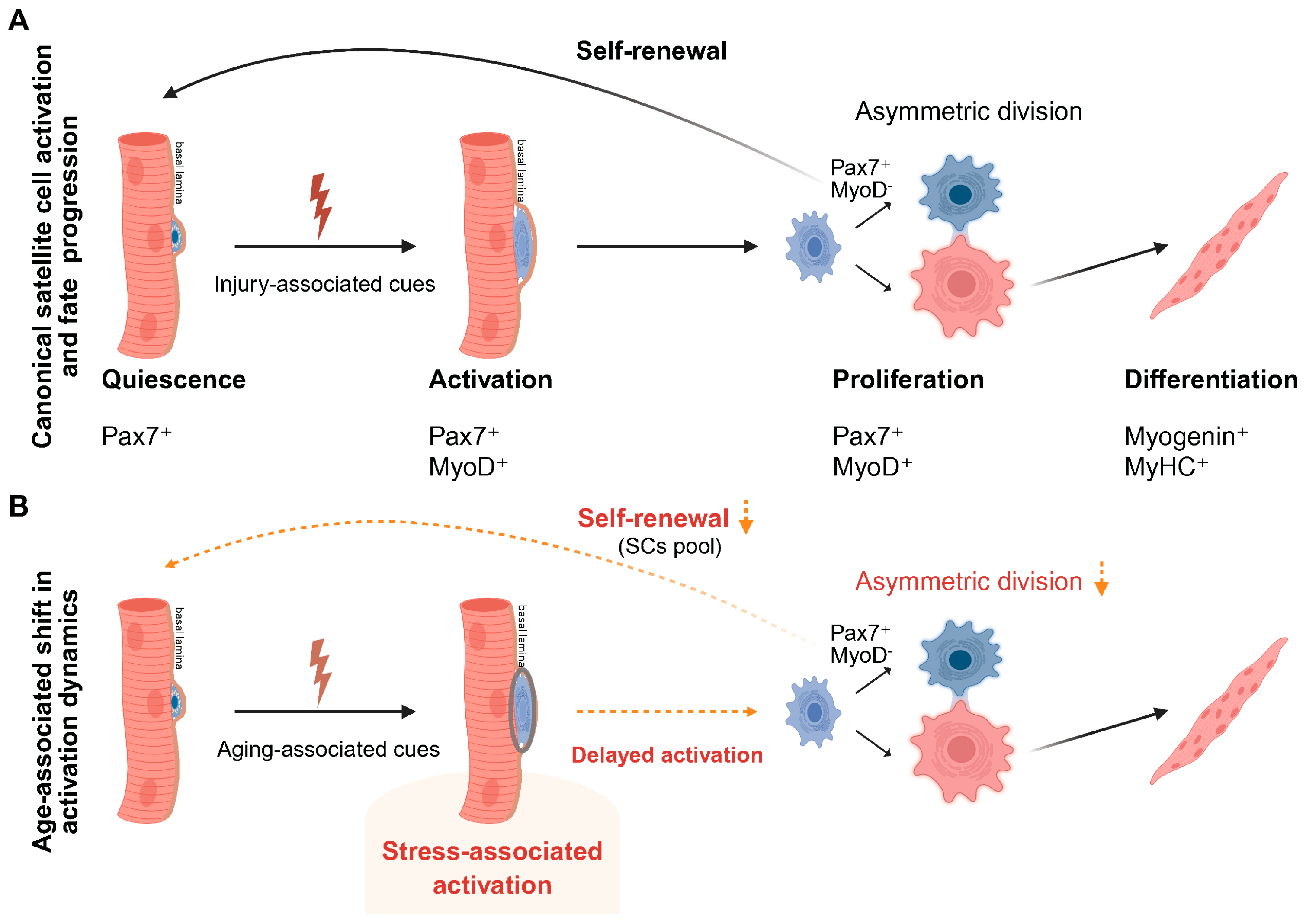
2.1. Classical Satellite Cell States Revisited in Aging
2.2. Early Activation as a Stress-Sensitive Transitional Checkpoint in Aging
2.3. The Aged Muscle Stem Cell Niche Constrains Early Activation and Introduces Delayed Activation Kinetics
3. Early Activation as a Vulnerable Checkpoint in Aged Muscle
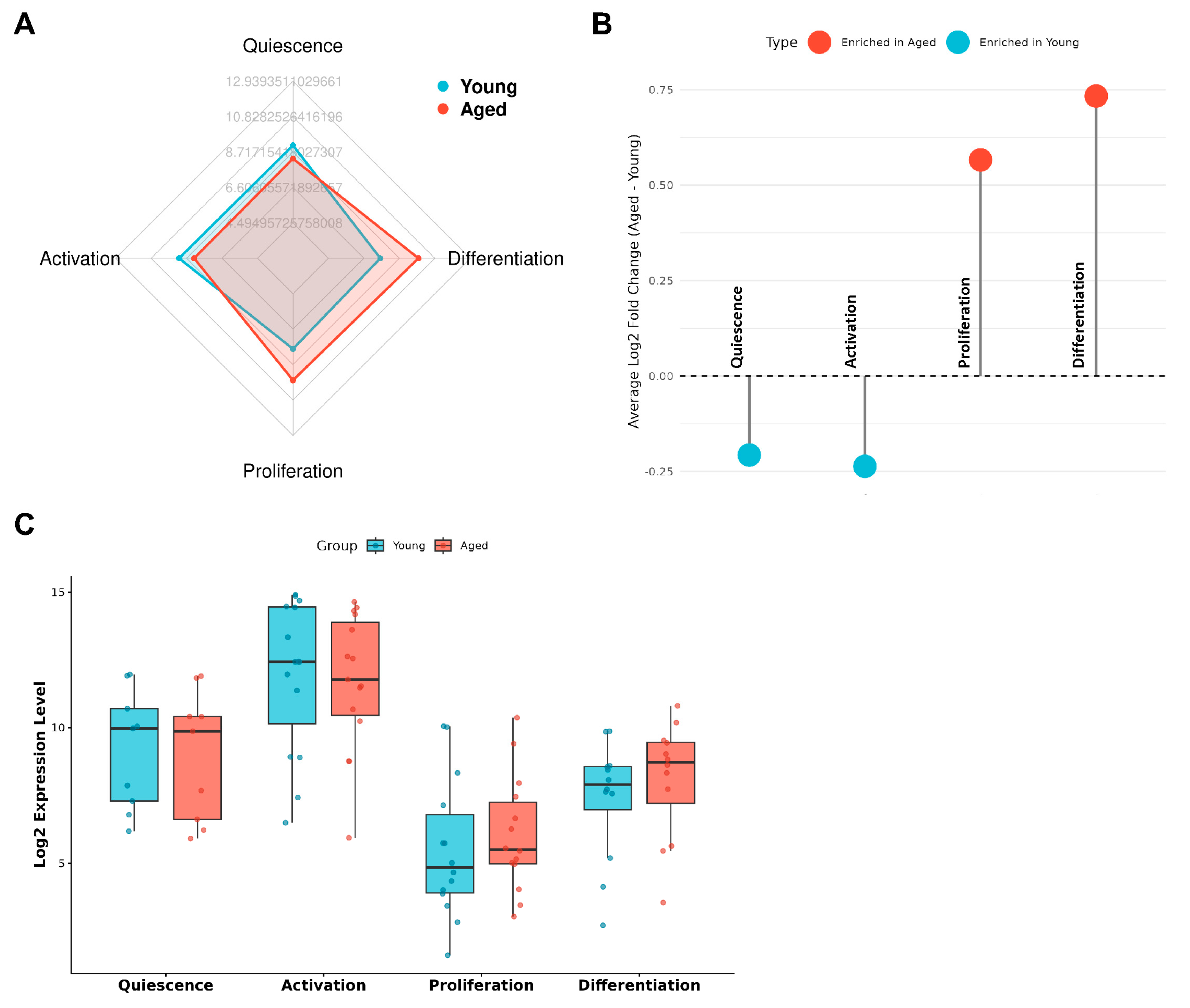
3.1. The Aged Niche Selectively Burdens Early Activation
3.2. Transcriptomic Evidence for a Stage-Selective Defect in Aged Satellite Cells
3.3. Compensatory Proliferation Masks Early Failure But Accelerates Long-Term Attrition
4. MG53: Beyond Membrane Repair
4.1. Canonical Roles of MG53 in Skeletal Muscle
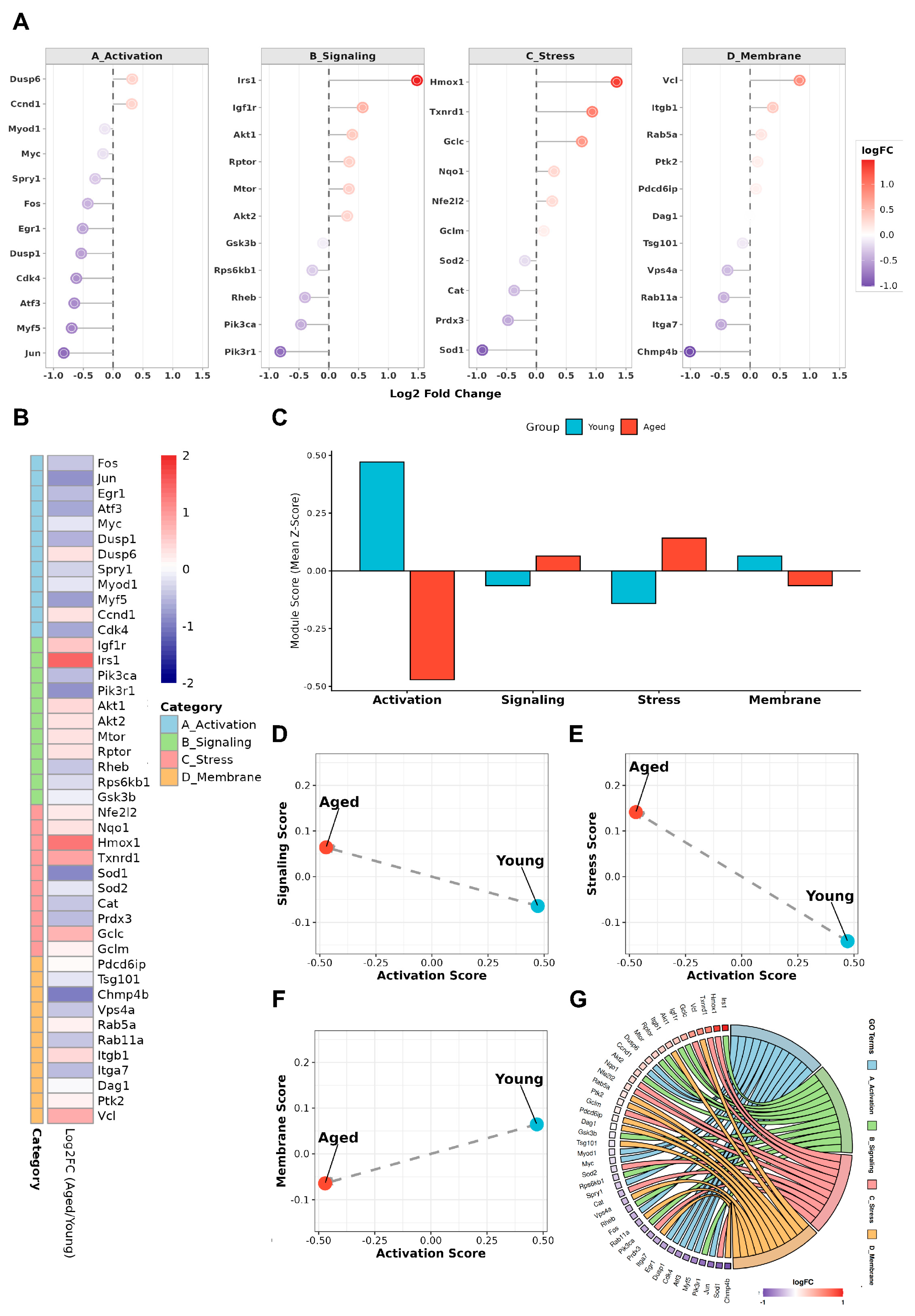
4.2. Activation-Associated stress, Membrane Remodeling, and Signaling Imbalance in Aged Satellite Cells
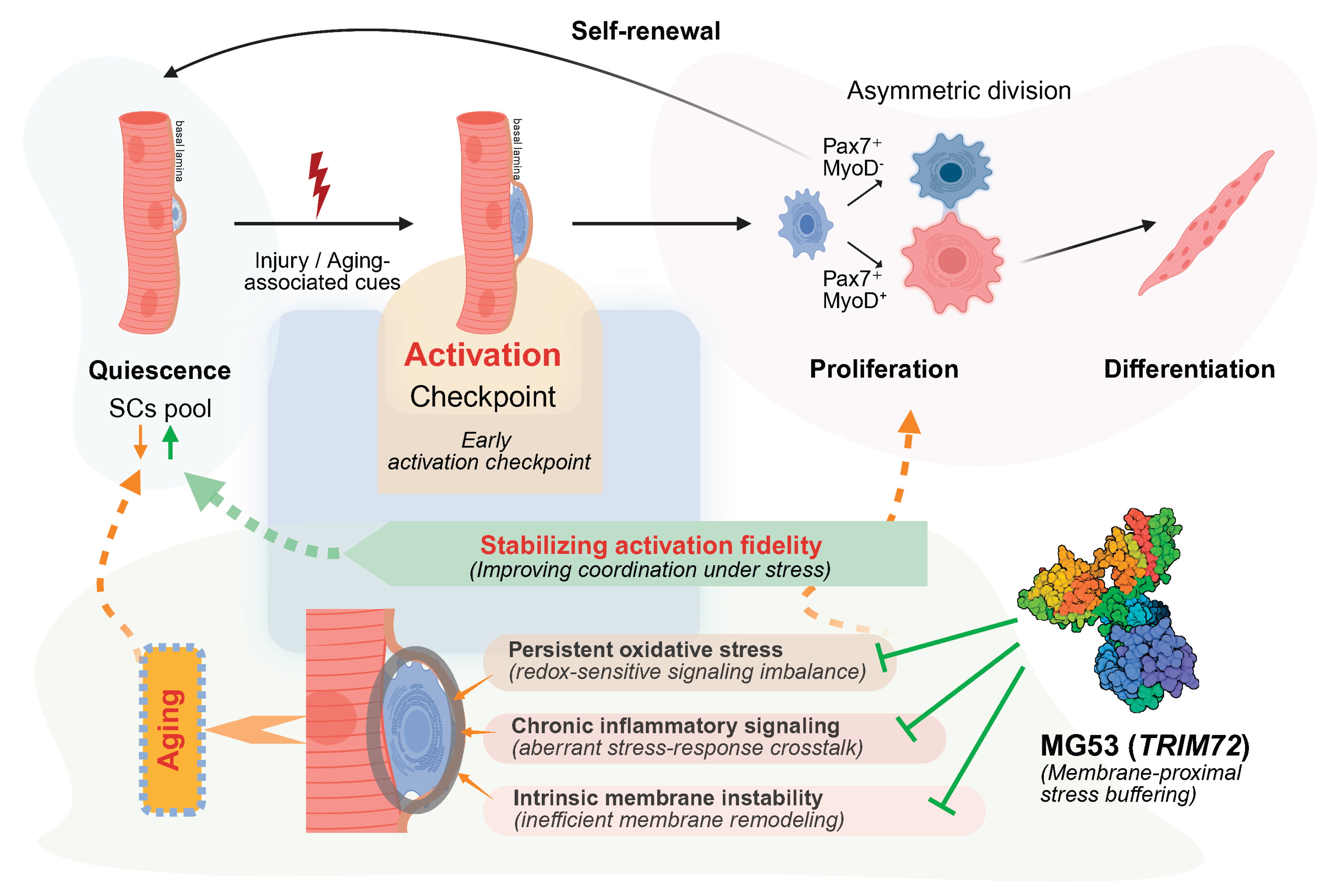
4.3. MG53 as a Permissive Regulator at the Early Activation Checkpoint
4.3.1. MG53-Mediated Buffering of Oxidative and Mitochondrial Stress During Early Activation
4.3.2. MG53 Modulation of Inflammatory Signaling and Stress Amplification
4.3.3. MG53-Dependent Membrane Stabilization at the Activation Checkpoint
4.4. Early Activation Fidelity as a Determinant of Regenerative Aging
5. Implications for Aged Muscle Regeneration and Therapeutic Perspectives
6. Open Questions and Future Directions
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Fu, X.; Wang, H.; Hu, P. Stem Cell Activation in Skeletal Muscle Regeneration. Cell Mol Life Sci 2015, 72, 1663–1677. [Google Scholar] [CrossRef]
- Mauro, A. Satellite Cell of Skeletal Muscle Fibers. J Biophys Biochem Cytol 1961, 9, 493–495. [Google Scholar] [CrossRef]
- Almada, A.E.; Wagers, A.J. Molecular Circuitry of Stem Cell Fate in Skeletal Muscle Regeneration, Ageing and Disease. Nat Rev Mol Cell Biol 2016, 17, 267–279. [Google Scholar] [CrossRef] [PubMed]
- Brack, A.S.; Rando, T.A. Tissue-Specific Stem Cells: Lessons from the Skeletal Muscle Satellite Cell. Cell Stem Cell 2012, 10, 504–514. [Google Scholar] [CrossRef] [PubMed]
- Arpke, R.W.; Shams, A.S.; Collins, B.C.; Larson, A.A.; Lu, N.; Lowe, D.A.; Kyba, M. Preservation of Satellite Cell Number and Regenerative Potential with Age Reveals Locomotory Muscle Bias. Skelet Muscle 2021, 11, 22. [Google Scholar] [CrossRef] [PubMed]
- Bernet, J.D.; Doles, J.D.; Hall, J.K.; Kelly Tanaka, K.; Carter, T.A.; Olwin, B.B. P38 MAPK Signaling Underlies a Cell-Autonomous Loss of Stem Cell Self-Renewal in Skeletal Muscle of Aged Mice. Nat Med 2014, 20, 265–271. [Google Scholar] [CrossRef]
- Kimmel, J.C.; Hwang, A.B.; Scaramozza, A.; Marshall, W.F.; Brack, A.S. Aging Induces Aberrant State Transition Kinetics in Murine Muscle Stem Cells. Development 2020, 147, dev183855. [Google Scholar] [CrossRef]
- Cosgrove, B.D.; Gilbert, P.M.; Porpiglia, E.; Mourkioti, F.; Lee, S.P.; Corbel, S.Y.; Llewellyn, M.E.; Delp, S.L.; Blau, H.M. Rejuvenation of the Muscle Stem Cell Population Restores Strength to Injured Aged Muscles. Nat Med 2014, 20, 255–264. [Google Scholar] [CrossRef]
- Sousa-Victor, P.; Gutarra, S.; García-Prat, L.; Rodriguez-Ubreva, J.; Ortet, L.; Ruiz-Bonilla, V.; Jardí, M.; Ballestar, E.; González, S.; Serrano, A.L.; et al. Geriatric Muscle Stem Cells Switch Reversible Quiescence into Senescence. Nature 2014, 506, 316–321. [Google Scholar] [CrossRef]
- Rodgers, J.T.; King, K.Y.; Brett, J.O.; Cromie, M.J.; Charville, G.W.; Maguire, K.K.; Brunson, C.; Mastey, N.; Liu, L.; Tsai, C.-R.; et al. mTORC1 Controls the Adaptive Transition of Quiescent Stem Cells from G0 to GAlert. Nature 2014, 510, 393–396. [Google Scholar] [CrossRef]
- Machado, L.; de Lima, J.E.; Fabre, O.; Proux, C.; Legendre, R.; Szegedi, A.; Varet, H.; Ingerslev, L.R.; Barrès, R.; Relaix, F.; et al. In Situ Fixation Redefines Quiescence and Early Activation of Skeletal Muscle Stem Cells. Cell Reports 2017, 21, 1982–1993. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.; Price, F.; Rudnicki, M.A. Satellite Cells and the Muscle Stem Cell Niche. Physiological Reviews 2013, 93, 23–67. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Cánoves, P.; Neves, J.; Sousa-Victor, P. Understanding Muscle Regenerative Decline with Aging: New Approaches to Bring Back Youthfulness to Aged Stem Cells. FEBS J 2020, 287, 406–416. [Google Scholar] [CrossRef] [PubMed]
- Weisleder, N.; Takizawa, N.; Lin, P.; Wang, X.; Cao, C.; Zhang, Y.; Tan, T.; Ferrante, C.; Zhu, H.; Chen, P.-J.; et al. Recombinant MG53 Protein Modulates Therapeutic Cell Membrane Repair in Treatment of Muscular Dystrophy. Sci Transl Med 2012, 4, 139ra85. [Google Scholar] [CrossRef]
- Cai, C.; Masumiya, H.; Weisleder, N.; Matsuda, N.; Nishi, M.; Hwang, M.; Ko, J.-K.; Lin, P.; Thornton, A.; Zhao, X.; et al. MG53 Nucleates Assembly of Cell Membrane Repair Machinery. Nat Cell Biol 2009, 11, 56–64. [Google Scholar] [CrossRef]
- Cai, C.; Masumiya, H.; Weisleder, N.; Pan, Z.; Nishi, M.; Komazaki, S.; Takeshima, H.; Ma, J. MG53 Regulates Membrane Budding and Exocytosis in Muscle Cells. J Biol Chem 2009, 284, 3314–3322. [Google Scholar] [CrossRef]
- Wang, X.; Li, X.; Ong, H.; Tan, T.; Park, K.H.; Bian, Z.; Zou, X.; Haggard, E.; Janssen, P.M.; Merritt, R.E.; et al. MG53 Suppresses NF-κB Activation to Mitigate Age-Related Heart Failure. JCI Insight 2021, 6, e148375. [Google Scholar] [CrossRef]
- Sermersheim, M.; Kenney, A.D.; Lin, P.-H.; McMichael, T.M.; Cai, C.; Gumpper, K.; Adesanya, T.M.A.; Li, H.; Zhou, X.; Park, K.-H.; et al. MG53 Suppresses Interferon-β and Inflammation via Regulation of Ryanodine Receptor-Mediated Intracellular Calcium Signaling. Nat Commun 2020, 11, 3624. [Google Scholar] [CrossRef]
- Bian, Z.; Wang, Q.; Zhou, X.; Tan, T.; Park, K.H.; Kramer, H.F.; McDougal, A.; Laping, N.J.; Kumar, S.; Adesanya, T.M.A.; et al. Sustained Elevation of MG53 in the Bloodstream Increases Tissue Regenerative Capacity without Compromising Metabolic Function. Nat Commun 2019, 10, 4659. [Google Scholar] [CrossRef]
- Hwang, M.; Ko, J.-K.; Weisleder, N.; Takeshima, H.; Ma, J. Redox-Dependent Oligomerization through a Leucine Zipper Motif Is Essential for MG53-Mediated Cell Membrane Repair. Am J Physiol Cell Physiol 2011, 301, C106–114. [Google Scholar] [CrossRef]
- CHARGÉ, S.B.P.; RUDNICKI, M.A. Cellular and Molecular Regulation of Muscle Regeneration. Physiological Reviews 2004, 84, 209–238. [Google Scholar] [CrossRef]
- Cheung, T.H.; Quach, N.L.; Charville, G.W.; Liu, L.; Park, L.; Edalati, A.; Yoo, B.; Hoang, P.; Rando, T.A. Maintenance of Muscle Stem-Cell Quiescence by microRNA-489. Nature 2012, 482, 524–528. [Google Scholar] [CrossRef] [PubMed]
- Kuang, S.; Kuroda, K.; Grand, F.L.; Rudnicki, M.A. Asymmetric Self-Renewal and Commitment of Satellite Stem Cells in Muscle. Cell 2007, 129, 999–1010. [Google Scholar] [CrossRef] [PubMed]
- Evano, B.; Khalilian, S.; Carrou, G.L.; Almouzni, G.; Tajbakhsh, S. Dynamics of Asymmetric and Symmetric Divisions of Muscle Stem Cells In Vivo and on Artificial Niches. Cell Reports 2020, 30, 3195–3206.e7. [Google Scholar] [CrossRef] [PubMed]
- Blau, H.M.; Cosgrove, B.D.; Ho, A.T.V. The Central Role of Muscle Stem Cells in Regenerative Failure with Aging. Nat Med 2015, 21, 854–862. [Google Scholar] [CrossRef]
- Chakkalakal, J.V.; Jones, K.M.; Basson, M.A.; Brack, A.S. The Aged Niche Disrupts Muscle Stem Cell Quiescence. Nature 2012, 490, 355–360. [Google Scholar] [CrossRef]
- van Velthoven, C.T.J.; de Morree, A.; Egner, I.M.; Brett, J.O.; Rando, T.A. Transcriptional Profiling of Quiescent Muscle Stem Cells In Vivo. Cell Reports 2017, 21, 1994–2004. [Google Scholar] [CrossRef]
- Dell’Orso, S.; Juan, A.H.; Ko, K.-D.; Naz, F.; Perovanovic, J.; Gutierrez-Cruz, G.; Feng, X.; Sartorelli, V. Single Cell Analysis of Adult Mouse Skeletal Muscle Stem Cells in Homeostatic and Regenerative Conditions. Development 2019, 146, dev174177. [Google Scholar] [CrossRef]
- Lukjanenko, L.; Jung, M.J.; Hegde, N.; Perruisseau-Carrier, C.; Migliavacca, E.; Rozo, M.; Karaz, S.; Jacot, G.; Schmidt, M.; Li, L.; et al. Loss of Fibronectin from the Aged Stem Cell Niche Affects the Regenerative Capacity of Skeletal Muscle in Mice. Nat Med 2016, 22, 897–905. [Google Scholar] [CrossRef]
- Brack, A.S.; Muñoz-Cánoves, P. The Ins and Outs of Muscle Stem Cell Aging. Skelet Muscle 2016, 6, 1. [Google Scholar] [CrossRef]
- Pala, F.; Di Girolamo, D.; Mella, S.; Yennek, S.; Chatre, L.; Ricchetti, M.; Tajbakhsh, S. Distinct Metabolic States Govern Skeletal Muscle Stem Cell Fates during Prenatal and Postnatal Myogenesis. J Cell Sci 2018, 131, jcs212977. [Google Scholar] [CrossRef] [PubMed]
- Oprescu, S.N.; Yue, F.; Qiu, J.; Brito, L.F.; Kuang, S. Temporal Dynamics and Heterogeneity of Cell Populations during Skeletal Muscle Regeneration. iScience 2020, 23, 100993. [Google Scholar] [CrossRef] [PubMed]
- Moyle, L.A.; Cheng, R.Y.; Liu, H.; Davoudi, S.; Ferreira, S.A.; Nissar, A.A.; Sun, Y.; Gentleman, E.; Simmons, C.A.; Gilbert, P.M. Three-Dimensional Niche Stiffness Synergizes with Wnt7a to Modulate the Extent of Satellite Cell Symmetric Self-Renewal Divisions. Mol Biol Cell 2020, 31, 1703–1713. [Google Scholar] [CrossRef] [PubMed]
- Uezumi, A.; Ikemoto-Uezumi, M.; Zhou, H.; Kurosawa, T.; Yoshimoto, Y.; Nakatani, M.; Hitachi, K.; Yamaguchi, H.; Wakatsuki, S.; Araki, T.; et al. Mesenchymal Bmp3b Expression Maintains Skeletal Muscle Integrity and Decreases in Age-Related Sarcopenia. J Clin Invest 2021, 131. [Google Scholar] [CrossRef]
- Park, S.H.; Han, J.; Jeong, B.-C.; Song, J.H.; Jang, S.H.; Jeong, H.; Kim, B.H.; Ko, Y.-G.; Park, Z.-Y.; Lee, K.E.; et al. Structure and Activation of the RING E3 Ubiquitin Ligase TRIM72 on the Membrane. Nat Struct Mol Biol 2023, 30, 1695–1706. [Google Scholar] [CrossRef]
- Ma, Y.; Ding, L.; Li, Z.; Zhou, C. Structural Basis for TRIM72 Oligomerization during Membrane Damage Repair. Nat Commun 2023, 14, 1555. [Google Scholar] [CrossRef]
- Lee, C.S.; Yi, J.-S.; Jung, S.-Y.; Kim, B.-W.; Lee, N.-R.; Choo, H.-J.; Jang, S.-Y.; Han, J.; Chi, S.-G.; Park, M.; et al. TRIM72 Negatively Regulates Myogenesis via Targeting Insulin Receptor Substrate-1. Cell Death Differ 2010, 17, 1254–1265. [Google Scholar] [CrossRef]
- Yi, J.-S.; Park, J.S.; Ham, Y.-M.; Nguyen, N.; Lee, N.-R.; Hong, J.; Kim, B.-W.; Lee, H.; Lee, C.-S.; Jeong, B.-C.; et al. MG53-Induced IRS-1 Ubiquitination Negatively Regulates Skeletal Myogenesis and Insulin Signalling. Nat Commun 2013, 4, 2354. [Google Scholar] [CrossRef]
- Brack, A.S.; Rando, T.A. Intrinsic Changes and Extrinsic Influences of Myogenic Stem Cell Function during Aging. Stem Cell Rev 2007, 3, 226–237. [Google Scholar] [CrossRef]
- Cooper, S.T.; McNeil, P.L. Membrane Repair: Mechanisms and Pathophysiology. Physiol Rev 2015, 95, 1205–1240. [Google Scholar] [CrossRef]
- McNeil, P.L.; Steinhardt, R.A. Plasma Membrane Disruption: Repair, Prevention, Adaptation. Annu Rev Cell Dev Biol 2003, 19, 697–731. [Google Scholar] [CrossRef]
- Duann, P.; Li, H.; Lin, P.; Tan, T.; Wang, Z.; Chen, K.; Zhou, X.; Gumpper, K.; Zhu, H.; Ludwig, T.; et al. MG53-Mediated Cell Membrane Repair Protects against Acute Kidney Injury. Sci Transl Med 2015, 7, 279ra36. [Google Scholar] [CrossRef]
- Gumpper-Fedus, K.; Park, K.H.; Ma, H.; Zhou, X.; Bian, Z.; Krishnamurthy, K.; Sermersheim, M.; Zhou, J.; Tan, T.; Li, L.; et al. MG53 Preserves Mitochondrial Integrity of Cardiomyocytes during Ischemia Reperfusion-Induced Oxidative Stress. Redox Biol 2022, 54, 102357. [Google Scholar] [CrossRef]
- Wang, Q.; Park, K.H.; Geng, B.; Chen, P.; Yang, C.; Jiang, Q.; Yi, F.; Tan, T.; Zhou, X.; Bian, Z.; et al. MG53 Inhibits Necroptosis Through Ubiquitination-Dependent RIPK1 Degradation for Cardiac Protection Following Ischemia/Reperfusion Injury. Front. Cardiovasc. Med. 2022, 9. [Google Scholar] [CrossRef]
- Ma, H.; Zhou, X.; Wang, X.; Wu, J.; Gumpper, K.; Tan, T.; Adesanya, T.A.; Yang, C.; Zheng, Y.; Chandler, H.; et al. MG53 Interacts with Cardiolipin to Protect Mitochondria from Ischemia-Reperfusion Induced Oxidative Stress. Biophysical Journal 2017, 112, 102a. [Google Scholar] [CrossRef]
- Kenney, A.D.; Aron, S.L.; Gilbert, C.; Kumar, N.; Chen, P.; Eddy, A.; Zhang, L.; Zani, A.; Vargas-Maldonado, N.; Speaks, S.; et al. Influenza Virus Replication in Cardiomyocytes Drives Heart Dysfunction and Fibrosis. Sci Adv 2022, 8, eabm5371. [Google Scholar] [CrossRef]
- Kenney, A.D.; Li, Z.; Bian, Z.; Zhou, X.; Li, H.; Whitson, B.A.; Tan, T.; Cai, C.; Ma, J.; Yount, J.S. Recombinant MG53 Protein Protects Mice from Lethal Influenza Virus Infection. Am J Respir Crit Care Med 2021, 203, 254–257. [Google Scholar] [CrossRef]
- Zhu, H.; Hou, J.; Roe, J.L.; Park, K.H.; Tan, T.; Zheng, Y.; Li, L.; Zhang, C.; Liu, J.; Liu, Z.; et al. Amelioration of Ischemia-Reperfusion-Induced Muscle Injury by the Recombinant Human MG53 Protein. Muscle Nerve 2015, 52, 852–858. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



