Submitted:
26 January 2026
Posted:
27 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Physicochemical Context: Water Stability Across the Urban Water Cycle
2.1.1. Harare Physicochemical Baseline and Operational Trends (2021)
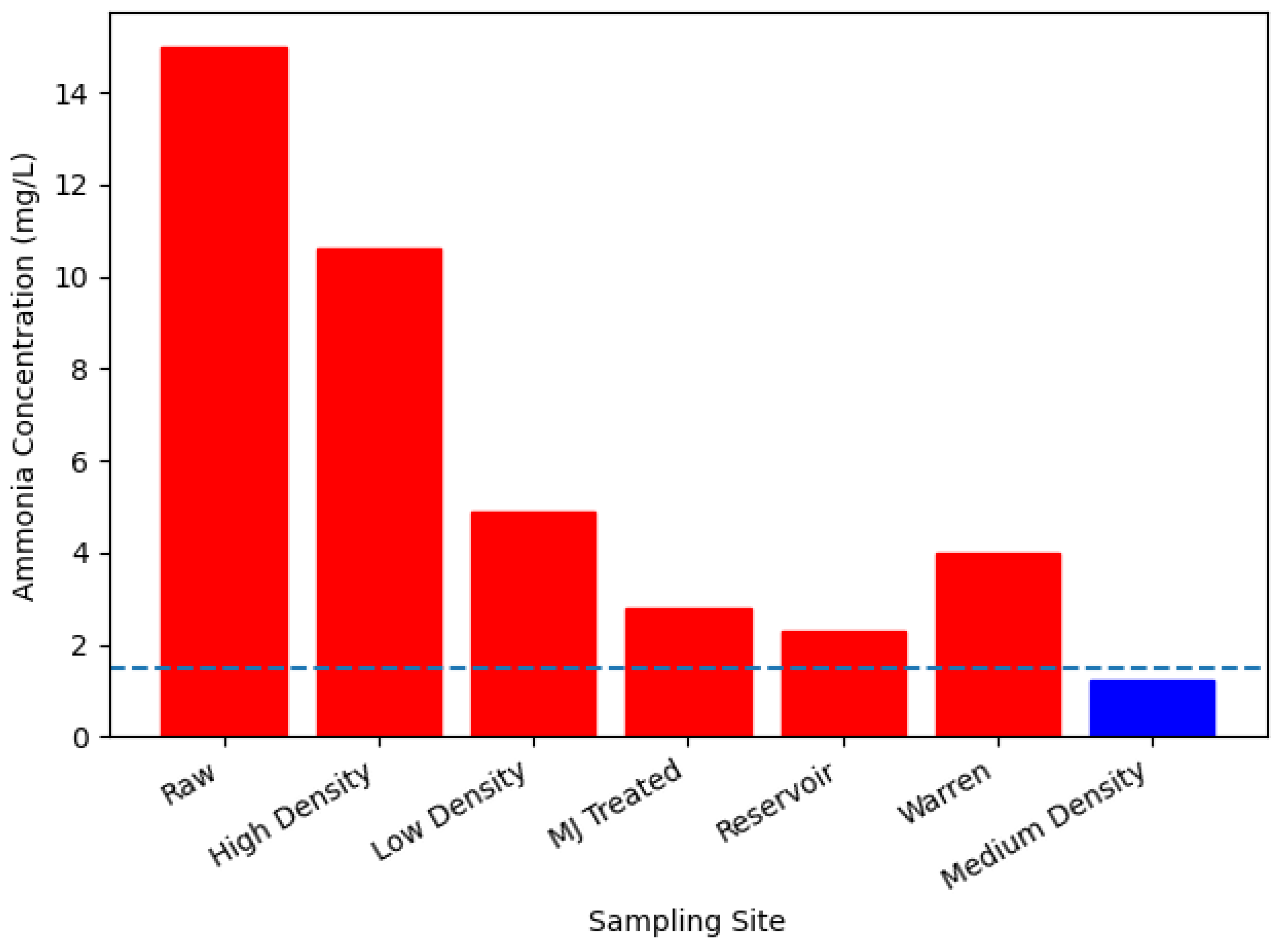
| Sampling site | pH | Turbidity (NTU) | Colour (Hazen) | Ammonia (mg/L) | Conductivity (μS/cm) | Iron (mg/L) | Aluminium (mg/L) |
|---|---|---|---|---|---|---|---|
| Raw | 8.0±0.2 | 6.7±6.5 | 43.6±9.67 | 15±3.86 | 188±21 | 13±9.0 | 0.01±0.04 |
| High | 7.1±0.9 | 2.3±0.40 | 13±8.67 | 10.6±3.88 | 656±48 | 0.1±0.089 | 0.43±0.1 |
| Low | 6.9±0.08 | 2.1±0.28 | 15.3±9.51 | 4.9±3.13 | 623±47 | 0.14±0.15 | 0.23±0.06 |
| Warren | 7.1±0.18 | 2.1±0.32 | 8.8±2.77 | 4±1 | 616±36 | 0.1±0.07 | NIL |
| MJ treated | 7.0±0.1 | 2.2±0.43 | 6.5±4.46 | 2.8±0.75 | 618±61 | 0.18±0.07 | 0.1±0.08 |
| Medium | 7.1±0.19 | 5.1±2.6 | 8±2.82 | 1.2±1.16 | 640±36 | 0.23±0.12 | 0.2±0.12 |
| Reservoir | 7.2±0.1 | 6.9±0.12 | 16.3±2.6 | 2.3±1.55 | 466±18 | 0.05±0.05 | 0.1±0.08 |
| WHO Guideline | 6.5 - 8.5 | ≤ 5 | ≤ 15 | ≤ 1.5 | 3000 | ≤ 0.5 | ≤ 0.2 |
| Parameter | MJWTP (after chlorination) | Warren Control | Reservoirs | Medium Density | Low Density | High Density | WHO Guideline |
|---|---|---|---|---|---|---|---|
| Free residual chlorine (mg L−1) | 0.12–0.46 0.30 ± 0.17 |
0.10–0.46 0.20 ± 0.17 |
0.00–0.11 0.02 ± 0.03 |
0.00–0.11 0.05 ± 0.03 |
0.06–0.70 0.25 ± 0.19 |
0.05–0.42 0.15 ± 0.11 |
0.20–0.50 |
| Total residual chlorine (mg L−1) | 0.25–0.95 0.60 ± 0.35 |
0.15–0.95 0.50 ± 0.35 |
0.01–0.21 0.09 ± 0.05 |
0.01–0.21 0.10 ± 0.05 |
0.15–1.31 0.57 ± 0.34 |
0.10–1.06 0.34 ± 0.29 |
2.1.2. Synthesis of Physicochemical Controls on Downstream Risk
2.2.1. Baseline Evidence of Antibiotic Presence (2020 Screening)
2.2.2. Occurrence and Behaviour of Pharmaceutical and Agrochemical Compounds in Urban Aquatic Systems (2021 Screening)
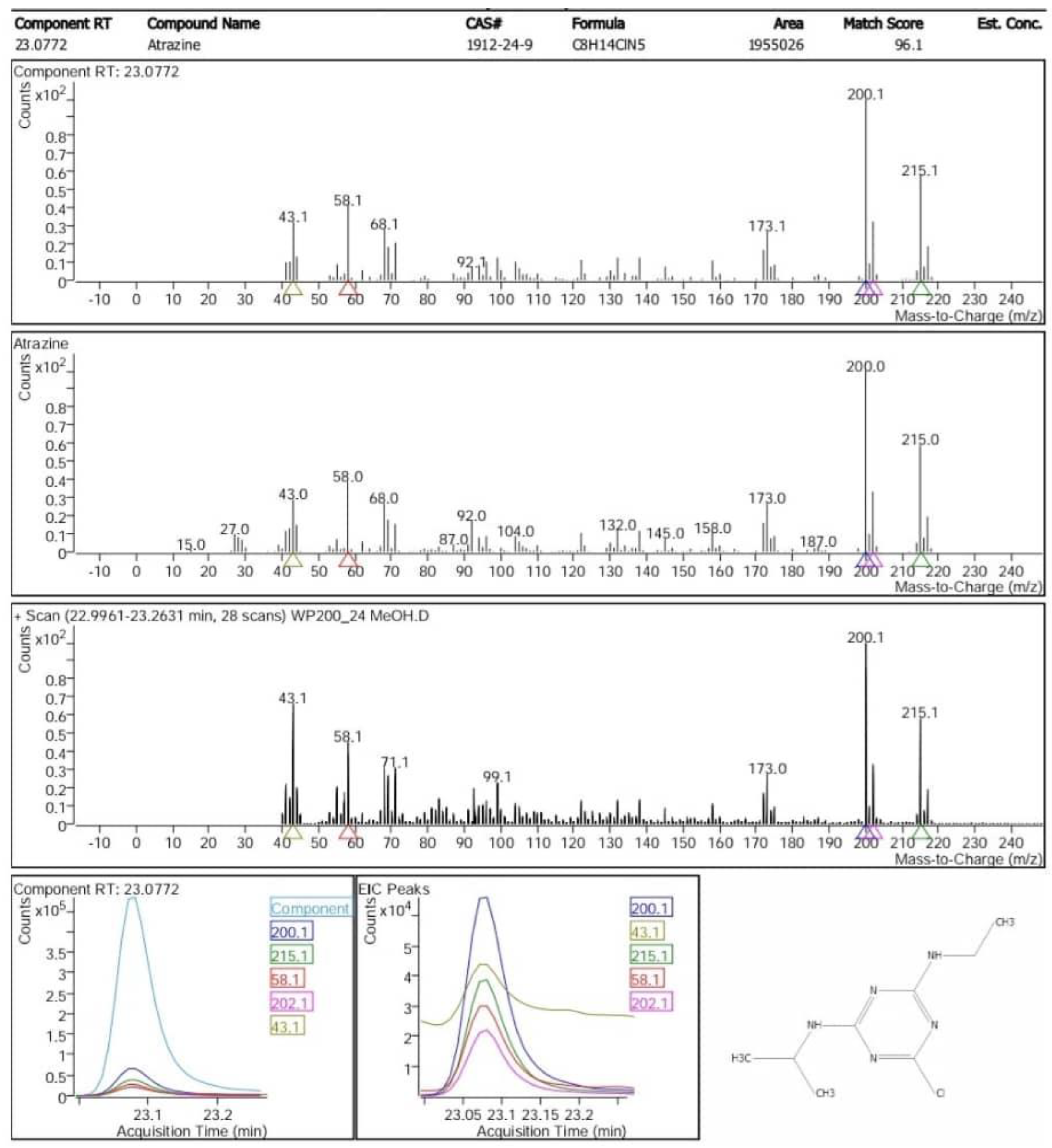
2.2.3. GC– MS Identification of Organic Micropollutants
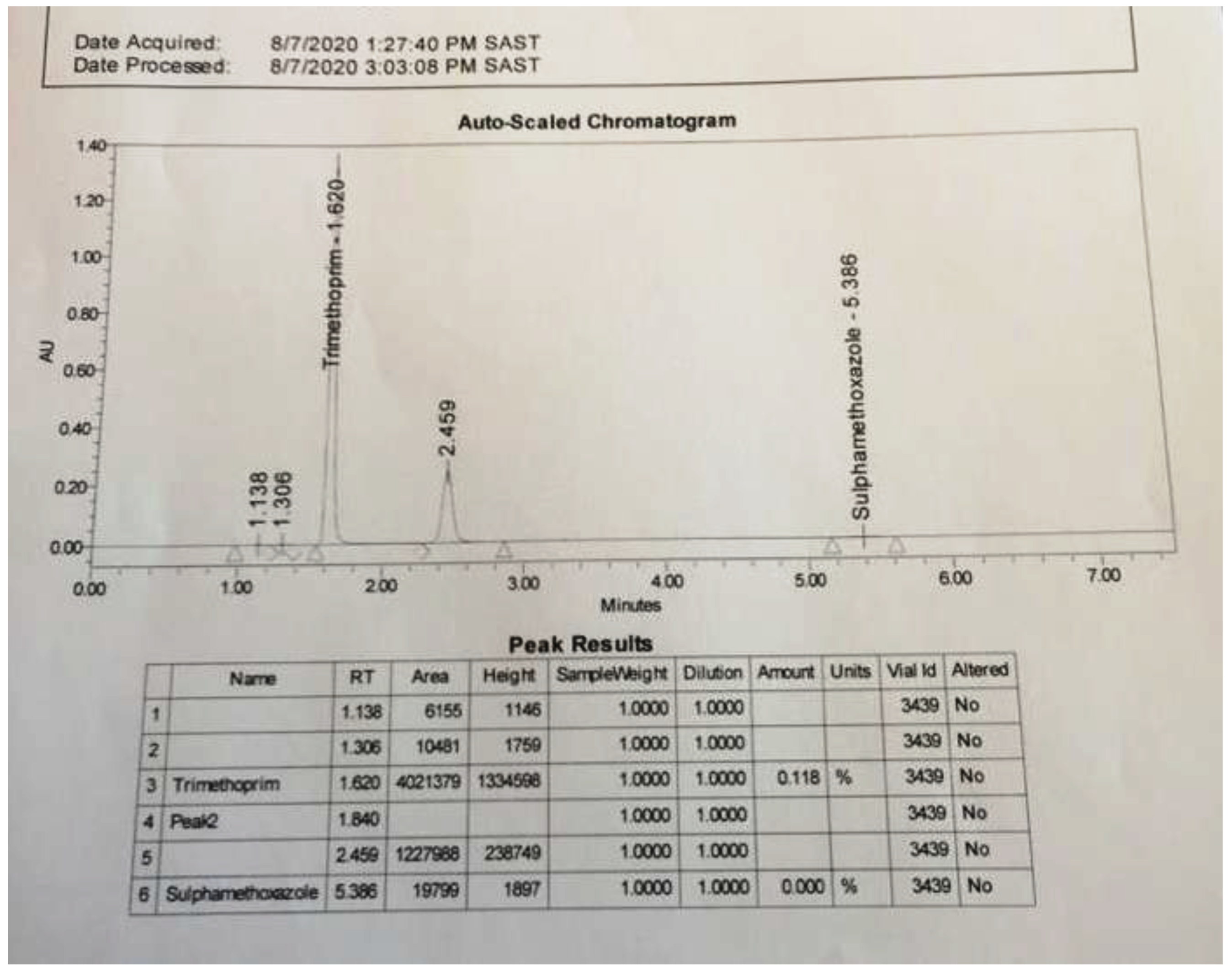
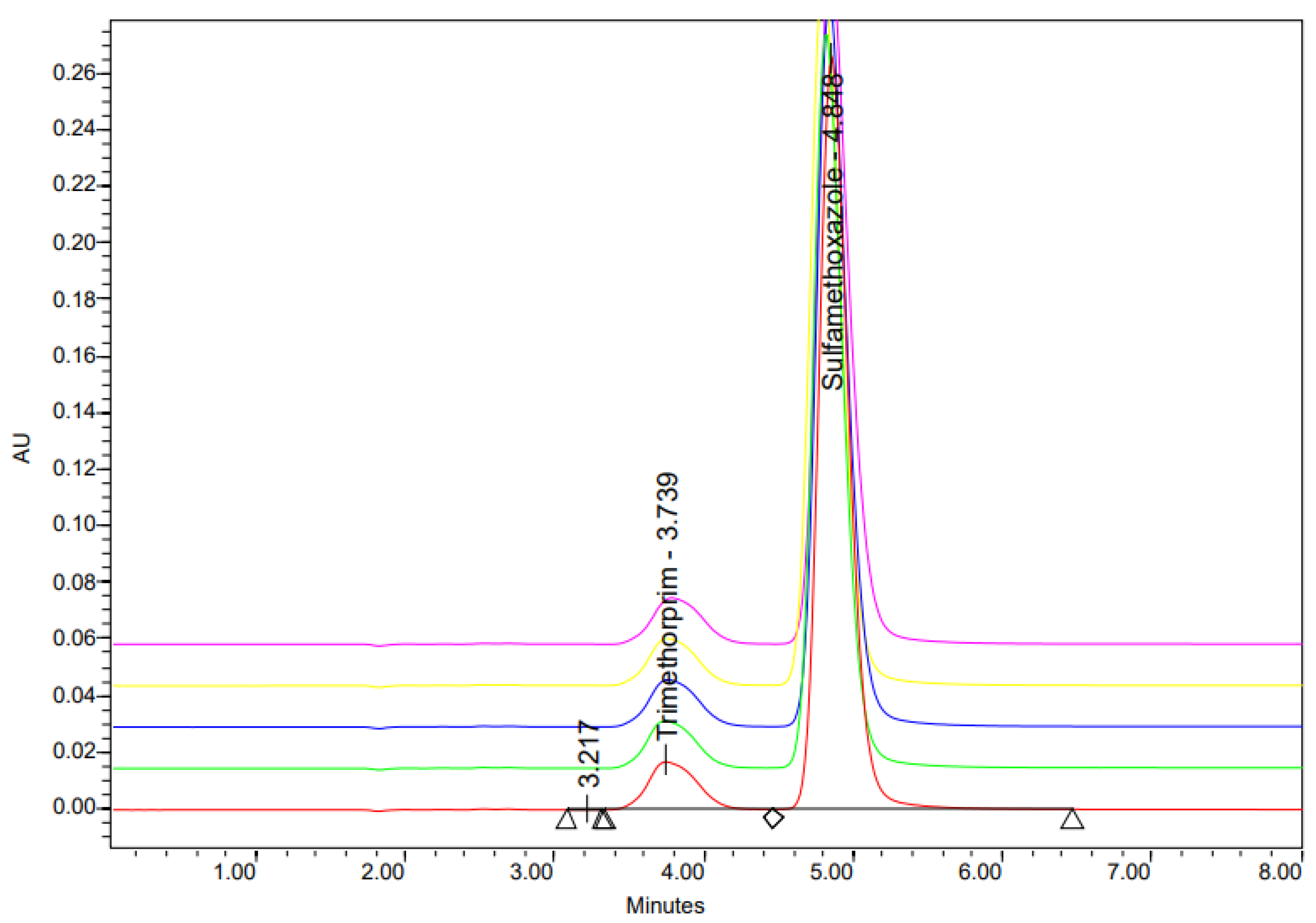
2.2.4. Occurrence of Sulfamethoxazole and Trimethoprim in Wastewater (2024)
2.3. Microbial Contamination and Environmental Exposure
2.3.1. Comparison of Microbial Contamination Levels Across Sampling Sites
2.3.2. Identification of Indicator and Potentially Pathogenic Bacteria
2.3.3. Public Health Relevance of Identified Bacteria (2021)
2.4. Antibiotic Susceptibility of Environmentally Relevant Bacterial Isolates
3. Discussion
3.1. Analytical Validation and Methodological Rigor
3.2. Pharmaceutical Persistence and Mechanistic Role of Co-Selective Stressors
3.3. Longitudinal Synthesis: The Evolutionary Reactor and Systemic Vulnerability
3.3.1. The Instability-Driven Emergence Hypothesis
3.3.2. One Health Conclusion
3.4. Microbial Proliferation, Regrowth, and Environmental Exposure Pathways
3.5. Chemical–Biological Interplay, Adaptive Resistance and Future Research Directions Across the Urban Water Cycle
4. Materials and Methods
4.1. Study Design and Temporal Framework
4.2. Longitudinal Integration and Taxonomic Continuity
4.3. Sampling Campaigns and Temporal Framework
4.4. Sample Collection, Containers, and Preservation
4.5. Physicochemical Water Quality Analysis
4.6. Microbiological Procedures
4.7. Antibiotic Susceptibility Testing and Environmental AMR Assessment
4.8. Organic Extraction of Organic from Lake Water and Treatedwaster Effluent
4.8.1. Lake Water Samples
4.8.2. Treated Wastewater Effluent
4.8. Statistical Analysis
4.9. Data Availability and Ethics
5. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Category | Abbreviation | Full Term | |||||
| Institutional & Regulatory | BUSE | Bindura University of Science Education | |||||
| CLSI | Clinical and Laboratory Standards Institute | ||||||
| EMA | Environmental Management Agency (Zimbabwe) | ||||||
| EPA | United States Environmental Protection Agency | ||||||
| ISP | International Programme in the Chemical Sciences | ||||||
| MCAZ | Medicines Control Authority of Zimbabwe | ||||||
| WHO | World Health Organization | ||||||
| Systems & Infrastructure | FWTP | Firle Wastewater Treatment Plant | |||||
| WWTP | Wastewater Treatment Plant | ||||||
| MJWTP | Morton Jaffray Water Treatment Plant | ||||||
| POE / POU | Point-of-Entry / Point-of-Use | ||||||
| STW | Sewage Treatment Works | ||||||
| Analytical & Physicochemical | BDL | Below Detection Limit | |||||
| BOD5 / COD | Biochemical Oxygen Demand (5-day) / Chemical Oxygen Demand | ||||||
| DO / TDS | Dissolved Oxygen / Total Dissolved Solids | ||||||
| DPD | N,N-Diethyl-p-phenylenediamine (chlorine method) | ||||||
| EC | Electrical Conductivity | ||||||
| GC–MS | Gas Chromatography–Mass Spectrometry | ||||||
| HPLC | High-Performance Liquid Chromatography | ||||||
| MSPD | Matrix Solid-Phase Dispersion | ||||||
| SPE | Solid Phase Extraction | ||||||
| tr | Retention time | ||||||
| IC | Ion Chromatography | ||||||
| LLE | Liquid–Liquid Extraction | ||||||
| LOD / LOQ | Limit of Detection / Limit of Quantitation | ||||||
| MQL | Method Quantitation Limit | ||||||
| ND | Non-Detect (or Not Detected) | ||||||
| NTU | Nephelometric Turbidity Units | ||||||
| TA / TH | Total Alkalinity / Total Hardness | ||||||
| UV | Ultraviolet | ||||||
| Microbiological & AMR | AMR | Antimicrobial Resistance | |||||
| CFU | Colony Forming Units | ||||||
| EMB | Eosin Methylene Blue (Agar) | ||||||
| H2S | Hydrogen Sulphide (Test) | ||||||
| HPC | Heterotrophic Plate Count | ||||||
| MDR | Multidrug Resistance | ||||||
| AR | Antibiotic Resistance | ||||||
| MAC | MacConkey (Agar) | ||||||
| NA | Nutrient Agar | ||||||
| TSI | Triple Sugar Iron (Test) | ||||||
| Chemical Targets | TMP | Trimethoprim | |||||
| SMX | Sulfamethoxazole | ||||||
| PhACs | Pharmaceutical Active Compounds | ||||||
| Statistics | ANOVA | Analysis of Variance | |||||
| PAST | Paleontological Statistics (Software) | ||||||
| STATA | Data Analysis and Statistical Software | ||||||
| RSD | Relative Standard Deviation | ||||||
References
- Yohannes, L; Begna, BB; Yirdaw, AA; Sergindo, MT; Bezie, AE; Weldegebriel, F; et al. Bacteriological quality of drinking water and associated factors in Alemwach refugee camp in Dabat District, Northwest Ethiopia. Sci Rep. Nature Research 2025, 15. [Google Scholar] [CrossRef] [PubMed]
- Water for prosperity and peace; United Nations Educational, Scientific and Cultural Organization, 2024.
- Water Overview_ Development news, research, data _ World Bank.
- Shemer, H; Wald, S; Semiat, R. Challenges and Solutions for Global Water Scarcity. In Membranes (Basel). MDPI; 2023. [Google Scholar] [CrossRef]
- Matengo, T; Mwedzi, T; Mbetu, J. Spatial and temporal patterns in water quality analysis near effluent dump-sites in Harare, Zimbabwe. Journal of Water Sanitation and Hygiene for Development. IWA Publishing 2025, 15, 206–14. [Google Scholar] [CrossRef]
- Ward, FA. Addressing Global Water Challenges in 2025: an Integrated Framework for Research, Policy, and Resource Management. Water Resources Management [Internet] 2025, 39, 7885–918. [Google Scholar] [CrossRef]
- Brooke, A; Fenner, RA. Improving urban water management and building water supply resilience in the city of Harare, Zimbabwe–a systems view. In Civil Engineering and Environmental Systems; Taylor and Francis Ltd., 2023; Volume 40, pp. 195–228. [Google Scholar] [CrossRef]
- Nduli, S; Tekere, M; Masindi, V; Foteinis, S. Assessment of the water quality and microbial regrowth in drinking water treatment plants and the distribution network. Water Resour Ind. Elsevier B.V 2025, 33. [Google Scholar] [CrossRef]
- Water Security Financing Report 2024.
- Nhongo, K; Dinka, MO. Assessing the vulnerability of districts to waterborne diseases in Harare, Zimbabwe. Journal of Water Sanitation and Hygiene for Development. IWA Publishing 2025, 15, 443–53. [Google Scholar] [CrossRef]
- Olatunji, G; Kokori, E; Moradeyo, A; Olatunji, D; Ajibola, F; Otolorin, O; et al. A Perspective on the 2023 Cholera Outbreaks in Zimbabwe: Implications, Response Strategies, and Policy Recommendations. In J Epidemiol Glob Health; Springer Science and Business Media B.V., 2024; Volume 14, pp. 243–8. [Google Scholar] [CrossRef]
- Global development [Internet]. Available online: https://www.theguardian.com/global-development/2023/oct/12/zimbabwe-bans-large-gatherings-as-threat-of-cholera-outbreak-grows.
- Funding Overview and Partnerships.
- Nduli, S; Tekere, M; Masindi, V; Foteinis, S. Assessment of the water quality and microbial regrowth in drinking water treatment plants and the distribution network. Water Resour Ind. Elsevier B.V 2025, 33. [Google Scholar] [CrossRef]
- Bangia, S; Bangia, R; Daverey, A. Pharmaceutically active compounds in aqueous environment: recent developments in their fate, occurrence and elimination for efficient water purification. In Environ. Monit. Assess.; Springer Science and Business Media Deutschland GmbH, 2023. [Google Scholar] [CrossRef]
- Bruno, A; Agostinetto, G; Fumagalli, S; Ghisleni, G; Sandionigi, A. It’s a Long Way to the Tap: Microbiome and DNA-Based Omics at the Core of Drinking Water Quality. Int. J. Environ. Res. Public Health. MDPI 2022. [Google Scholar] [CrossRef] [PubMed]
- Lunghi, C; Valetto, MR; Caracciolo, AB; Bramke, I; Caroli, S; Bottoni, P; et al. Call to action: Pharmaceutical residues in the environment: threats to ecosystems and human health. In Drug Saf. Adis; 2025; pp. 315–20. [Google Scholar] [CrossRef]
- Ashiwaju, BI; Uzougbo, CG; Orikpete, OF. Environmental Impact of Pharmaceuticals: A Comprehensive Review. Matrix Science Pharma. Medknow 2023, 7, 85–94. [Google Scholar] [CrossRef]
- Pärnänen, KMM; Narciso-Da-Rocha, C; Kneis, D; Berendonk, TU; Cacace, D; Do, TT; et al. Antibiotic resistance in European wastewater treatment plants mirrors the pattern of clinical antibiotic resistance prevalence. In Sci Adv; American Association for the Advancement of Science, 2019; p. 5. [Google Scholar] [CrossRef]
- Alem, K; Dagnew, M; Gizachew, M; Gelaw, B; Moges, F. Environmental Antimicrobial Resistance: Key Drivers, Hotspots, Innovative Strategies, and Challenges in the Fight Against Superbugs. In Microbiologyopen; John Wiley and Sons Inc, 2025. [Google Scholar] [CrossRef]
- Takawira, H; Mbanga, J. Occurrence of multidrug-resistant Escherichia coli and antibiotic resistance genes in a wastewater treatment plant and its associated river water in Harare, Zimbabwe. Water SA. South African Water Research Commission 2023, 49, 396–403. [Google Scholar] [CrossRef]
- Akhter, S; Bhat, MA; Ahmed, S; Siddiqui, WA. Antibiotic residue contamination in the aquatic environment, sources and associated potential health risks. In Environ. Geochem. Health; Springer, 2024. [Google Scholar] [CrossRef]
- Marti, E; Jofre, J; Balcazar, JL. Prevalence of Antibiotic Resistance Genes and Bacterial Community Composition in a River Influenced by a Wastewater Treatment Plant. PLoS One 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Occurrence of pharmaceuticals in aquatic environments_ A review and potential impacts in South Africa.
- Malíková, P; Chromíková, J; Biskup, D. Removal Efficiency of Pharmaceuticals from Municipal Wastewater by Advanced Treatment Methods. In AEE 2025 [Internet]; MDPI: Basel Switzerland, 2025. [Google Scholar] [CrossRef]
- Thakur, R; Dhar, H; Wozniak, TM; Mathew, S. Addressing the overlooked frontier in AMR research and surveillance. In Front. Public Health; Frontiers Media SA, 2025. [Google Scholar] [CrossRef]
- Zhang, H; Liu, J; Wang, L; Zhai, Z. Glyphosate escalates horizontal transfer of conjugative plasmid harboring antibiotic resistance genes. In Bioengineered; Bellwether Publishing, Ltd., 2021; Volume 12, pp. 63–9. [Google Scholar] [CrossRef]
- Cocker, D; Mwapasa, T; Grabic, R; Grabicová, K; Staňová, AV; Chidziwisano, K; et al. Environmental hazards from pollution of antibiotics and resistance-driving chemicals in an urban river network from Malawi. npj Antimicrobials and Resistance [Internet] 2025, 3, 85. [Google Scholar] [CrossRef] [PubMed]
- Matengo, T; Mwedzi, T; Mbetu, J. Spatial and temporal patterns in water quality analysis near effluent dump-sites in Harare, Zimbabwe. Journal of Water Sanitation and Hygiene for Development. IWA Publishing 2025, 15, 206–14. [Google Scholar] [CrossRef]
- Chirenda, TG; Srinivas, SC; Tandlich, R. Microbial water quality of treated water and raw water sources in the Harare area, Zimbabwe. Water SA. South African Water Research Commission 2015, 41, 691–7. [Google Scholar] [CrossRef]
- Kativhu, CC. POTABILITY OF BOREHOLE, BOTTLED, AND MUNICIPAL WATER IN HARARE; 2013. [Google Scholar]
- Antimicrobial Resistance and the Water Sector Current Landscape and Recommendations.
- Ngqwala, NP; Muchesa, P. Occurrence of pharmaceuticals in aquatic environments: A review and potential impacts in South Africa. S. Afr. J. Sci. Academy of Science of South Africa 2020. [Google Scholar] [CrossRef] [PubMed]
- Ajayi, AO; Odeyemi, AT; Akinjogunla, OJ; Adeyeye, AB; Ayo-ajayi, I. Review of antibiotic-resistant bacteria and antibiotic resistance genes within the one health framework. In Infect. Ecol. Epidemiol.; Taylor and Francis Ltd., 2024. [Google Scholar] [CrossRef]
- Garland, J; Franklin, A; Bagley, M; Keely, S; Brinkman, N; Jahne, M; et al. EPA Office of Research & Development Initiatives to Monitor Environmental AMR and Model Risk.
- Antimicrobial resistance in surface waters — developing environmental monitoring for better risk management _ Publications _ European Environment Agency (EEA).
- Mudenda, S; Chabalenge, B; Daka, V; Mfune, RL; Salachi, KI; Mohamed, S; et al. Global Strategies to Combat Antimicrobial Resistance: A One Health Perspective. In Pharmacology & Pharmacy; Scientific Research Publishing, Inc., 2023; Volume 14, pp. 271–328. [Google Scholar] [CrossRef]
- Guidelines for Drinking-water Quality FOURTH EDITION INCORPORATING THE FIRST ADDENDUM.
- Dzomba, P; Kugara, J; Zaranyika, MF. African Journal of Pharmacy and Pharmacology Extraction of tetracycline antimicrobials from river water and sediment: a comparative study of three solid phase extraction methods. 2015, 9, 523–31. [Google Scholar] [CrossRef]
- Religia, A. Title Ecotoxicogenomics study combined with Cytochrome P450 mutagenesis on sublethal effects of the model herbicide atrazine in Daphnia magna. In Citation 大阪大学 [Internet]; 2021. [Google Scholar] [CrossRef]
- Atrazine Formula: C H ClN [Internet]. 2018. Available online: https://webbook.nist.gov/cgi/cbook.cgi?ID=C1912249&Units=SI&Mask=FFF#Mass-Spec.
- Epa U, of Pesticide Program O. DER – Atrazine & Other Analytes in Soil – MRID 49537101.
- M100 Performance Standards for Antimicrobial Susceptibility Testing A CLSI supplement for global application. Available online: www.clsi.org.
- Abbasi, Z; Ghasemi, SM; Ahmadi, Y; Shokri, D. Isolation and Identification of Effective Probiotics on Drug-Resistant Acinetobacter baumannii Strains and Their Biofilms. In Canadian Journal of Infectious Diseases and Medical Microbiology; Hindawi Limited, 2024. [Google Scholar] [CrossRef]
- Tadesse, A; Sharew, B; Tilahun, M; Million, Y. Isolation and antimicrobial susceptibility profile of Salmonella species from slaughtered cattle carcasses and abattoir personnel at Dessie, municipality Abattoir, Northeast Ethiopia. In BMC Microbiol; BioMed Central Ltd., 2024. [Google Scholar] [CrossRef]
- Sung, K; Nawaz, M; Park, M; Chon, J; Khan, SA; Alotaibi, K; et al. Comprehensive Genomic Analysis of Uropathogenic E. coli: Virulence Factors, Antimicrobial Resistance, and Mobile Genetic Elements. In Pathogens; Multidisciplinary Digital Publishing Institute (MDPI), 2024. [Google Scholar] [CrossRef]
- Stedman, E; Molin, A; Oglesby, V; Torpey, E; Spivack, S; Sullivan, K V.; et al. High Rates of Bacteremia and Fluoroquinolone Resistance During an Outbreak of Shigellosis Among People Experiencing Homelessness and Opioid Use Disorder in Philadelphia, Pennsylvania. In Open Forum Infect Dis; Oxford University Press, 2025; p. 12. [Google Scholar] [CrossRef]
- Kurenbach, B; Marjoshi, D; Amábile-Cuevas, CF; Ferguson, GC; Godsoe, W; Gibson, P; et al. Sublethal exposure to commercial formulations of the herbicides dicamba, 2,4-dichlorophenoxyacetic acid, and Glyphosate cause changes in antibiotic susceptibility in Escherichia coli and Salmonella enterica serovar Typhimurium. In mBio; American Society for Microbiology, 2015; p. 6. [Google Scholar] [CrossRef]
- Glyphosate escalates horizontal transfer of conjugative plasmid harboring antibiotic resistance genes.
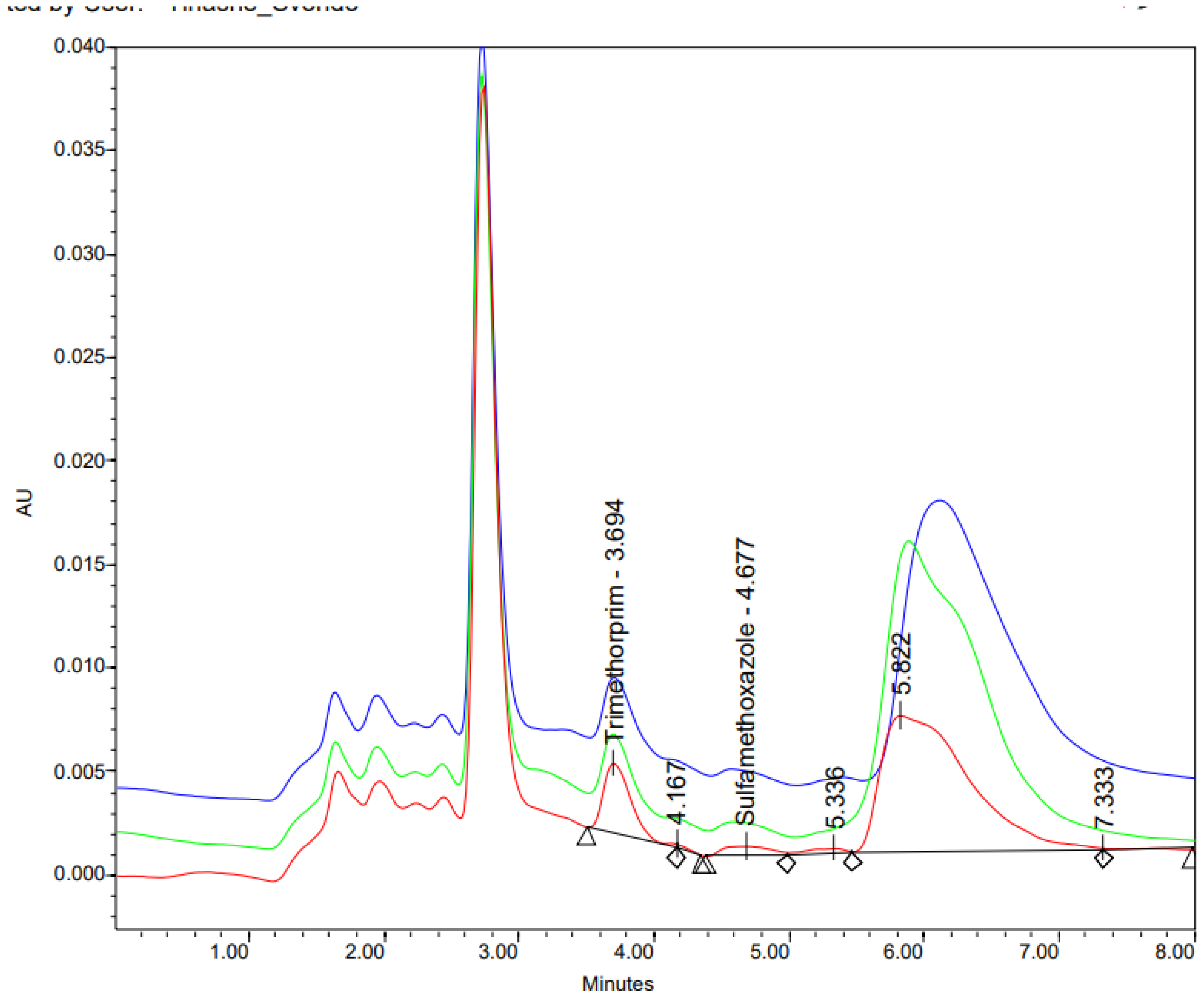
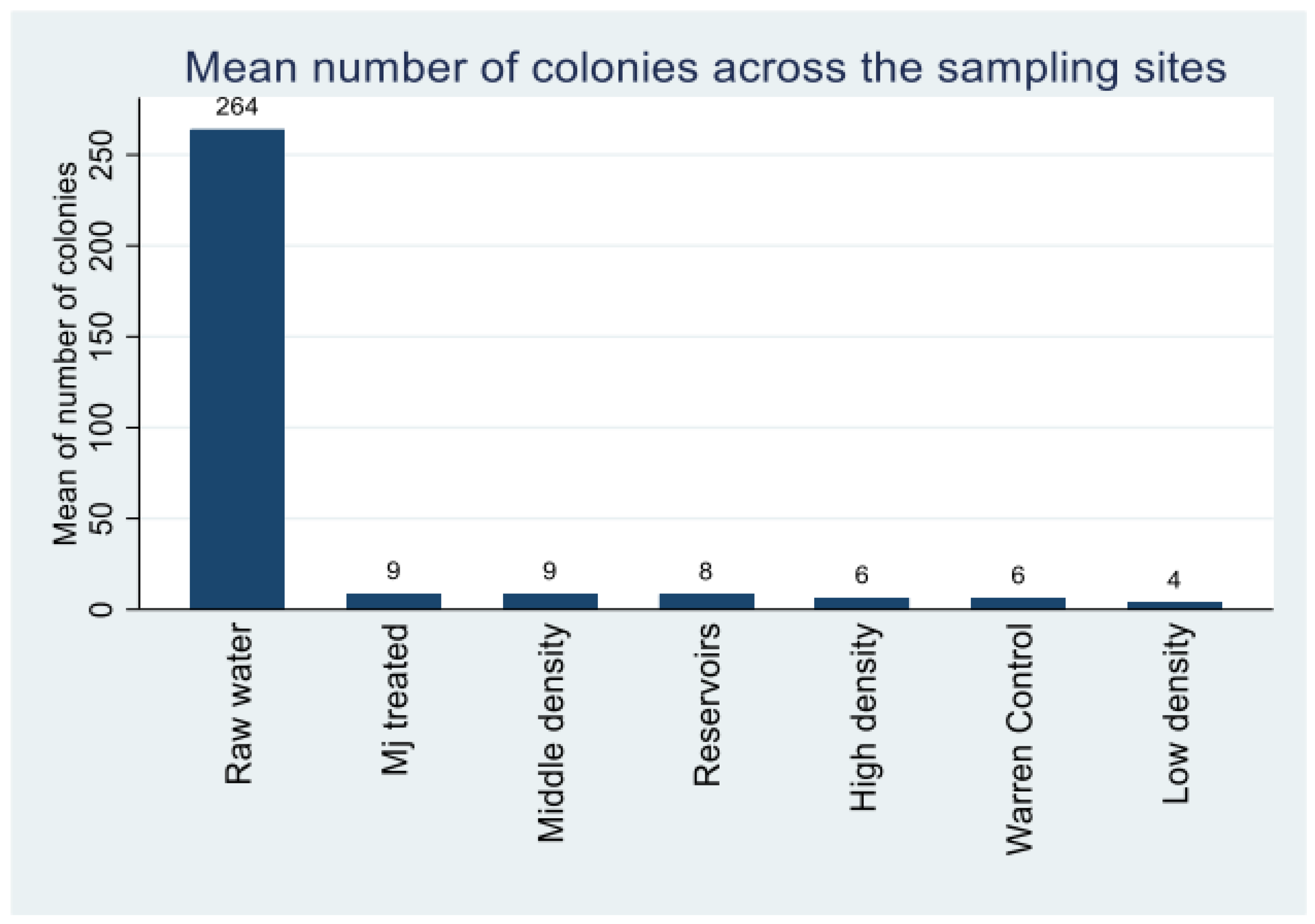
| Bacteria | Health significance | Persistence in water supply | Resistance to chlorine | Relative infectivity |
|---|---|---|---|---|
| E.coli Enterohaemorrhagic | High | Moderate | Low | High |
| Shigella spp | High | Short | Low | Low |
| Proteus spp | Moderate | Short | Low | High |
| Klebsiella spp | High | Moderate | Low | Moderate |
| Other | High | May multiply | low | Low |
| Salmonella | Health significance | Persistence in water supply | Resistance to chlorine | Relative infectivity |
| Bacteria | High | Moderate | Low | High |
| E.coli Enterohaemorrhagic | High | Short | Low | Low |
| Sampling Site | E. coli | Shigella spp | Salmonella | Klebsiella spp | Proteus spp |
|---|---|---|---|---|---|
| Lake Chivero | + | + | + | + | + |
| MJ (Treated) | - | - | - | - | - |
| Warren Control | - | - | - | - | - |
| Reservoirs | - | - | - | - | - |
| Medium Density (Avg) | + | - | - | - | - |
| High Density (Avg) | + | - | + | - | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




