Submitted:
23 January 2026
Posted:
27 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. ECM and Immune Cells Bidirectional Interactions
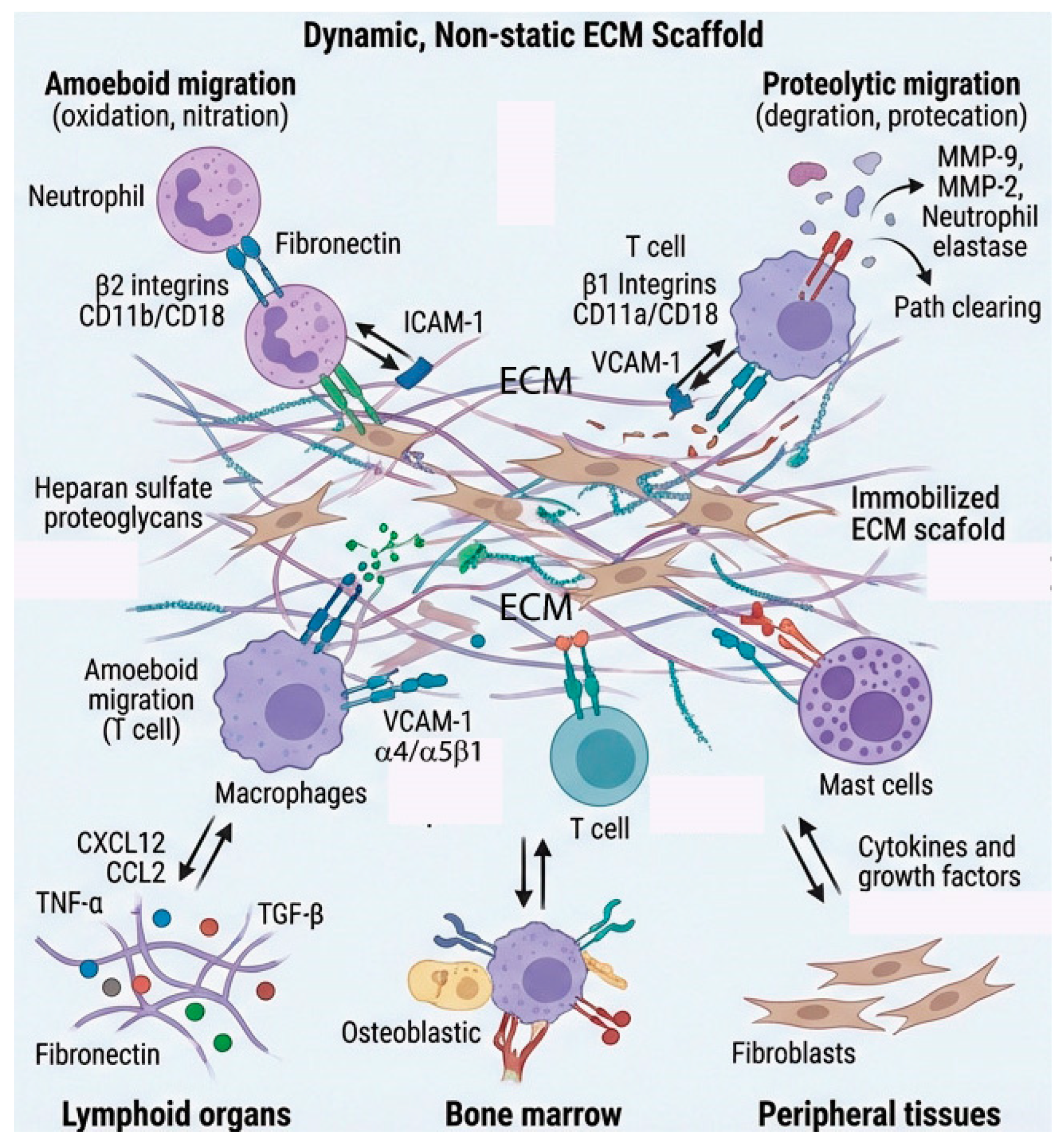
2.1. Integrin-Mediated Immune Cell-ECM Interactions
2.2. Immune Cell Migration Through ECM
2.3. ECM-Sequestered Factors and Immune Regulation
2.4. Immune Cell-Mediated ECM Remodeling
2.5. ECM in Immune Cell Niche Formation
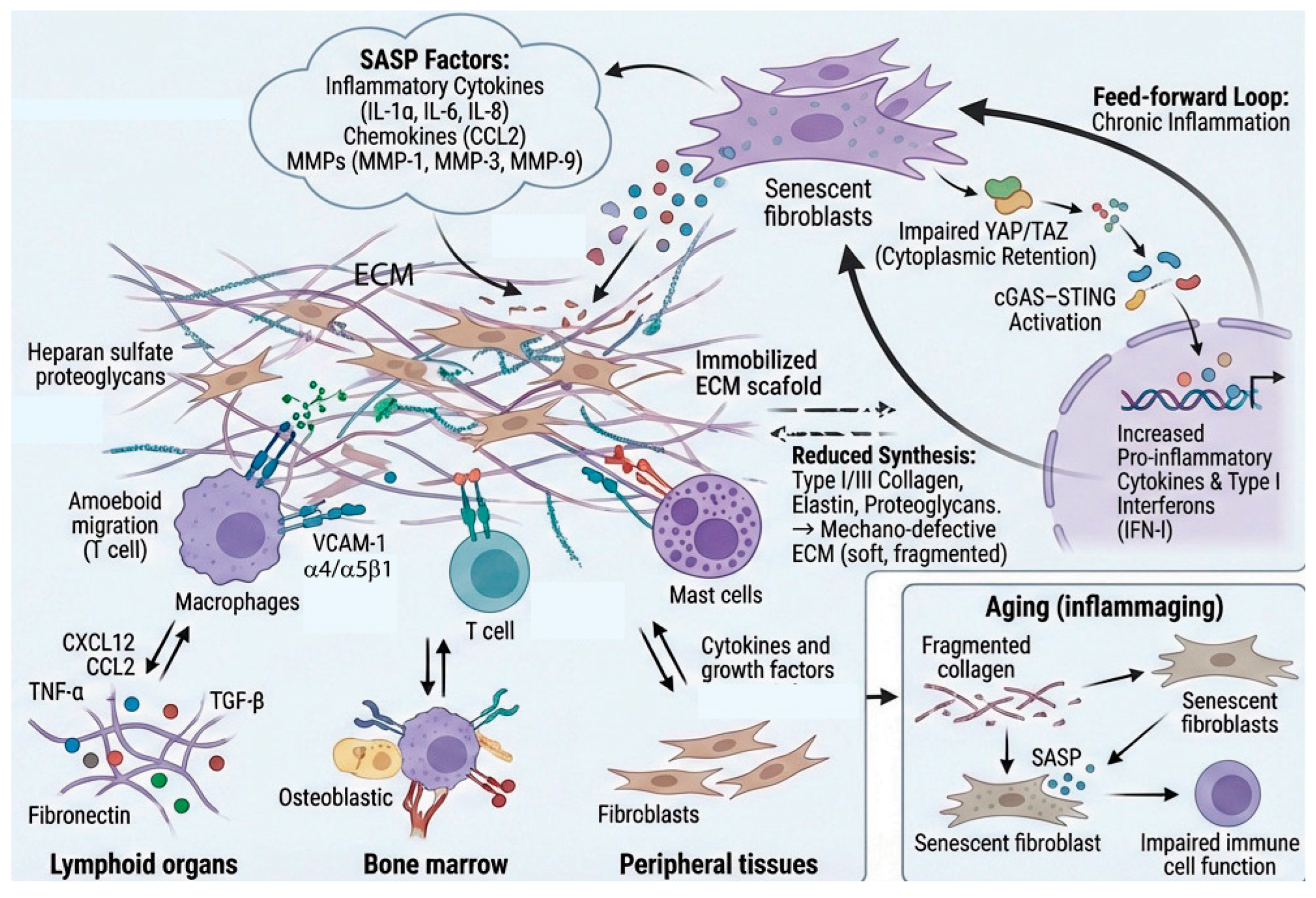
2.6. Pathological Immune Cell-ECM Interactions
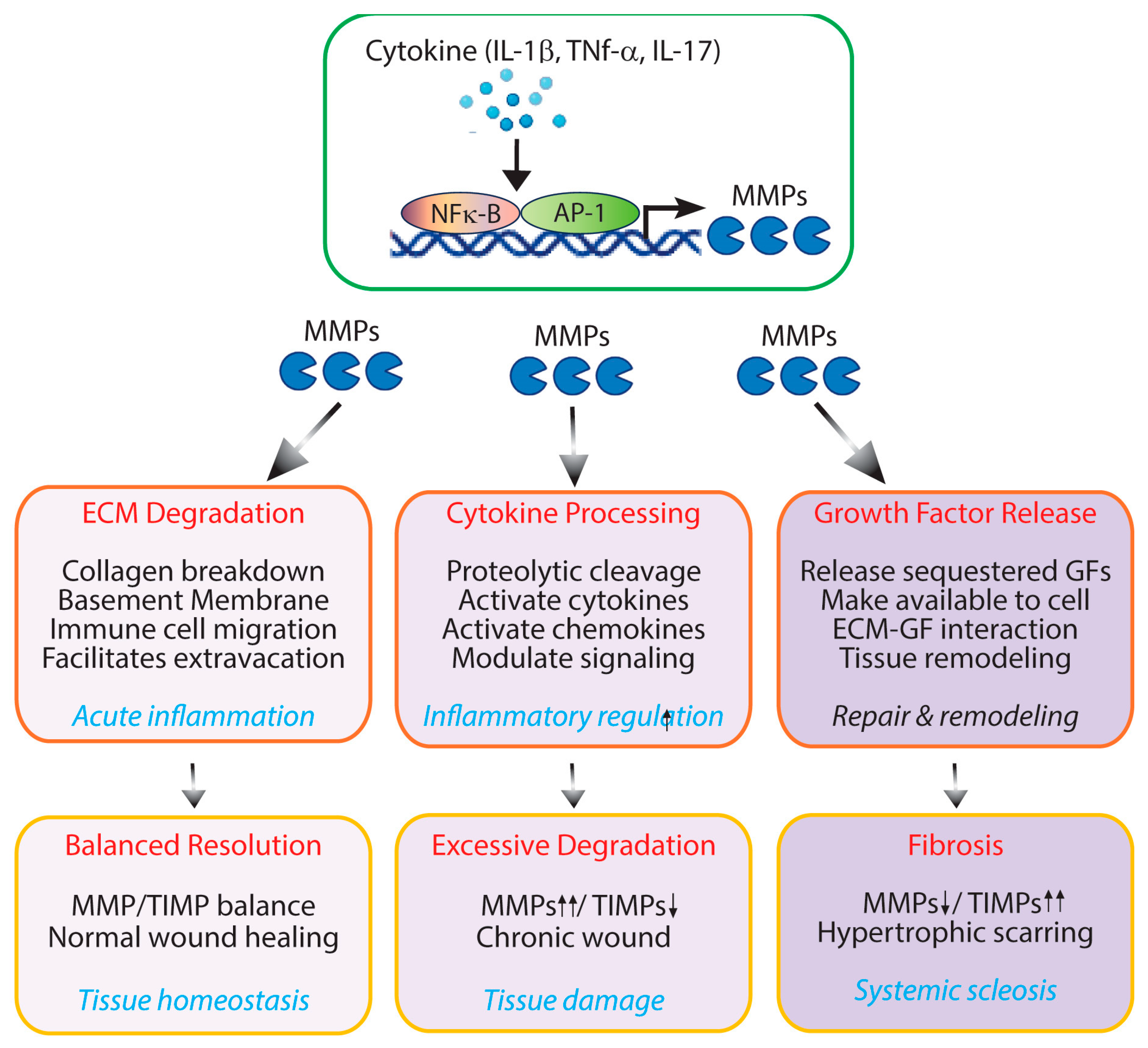
3. Matrisome-Associated Proteins: MMPs as Inflammatory Modulators
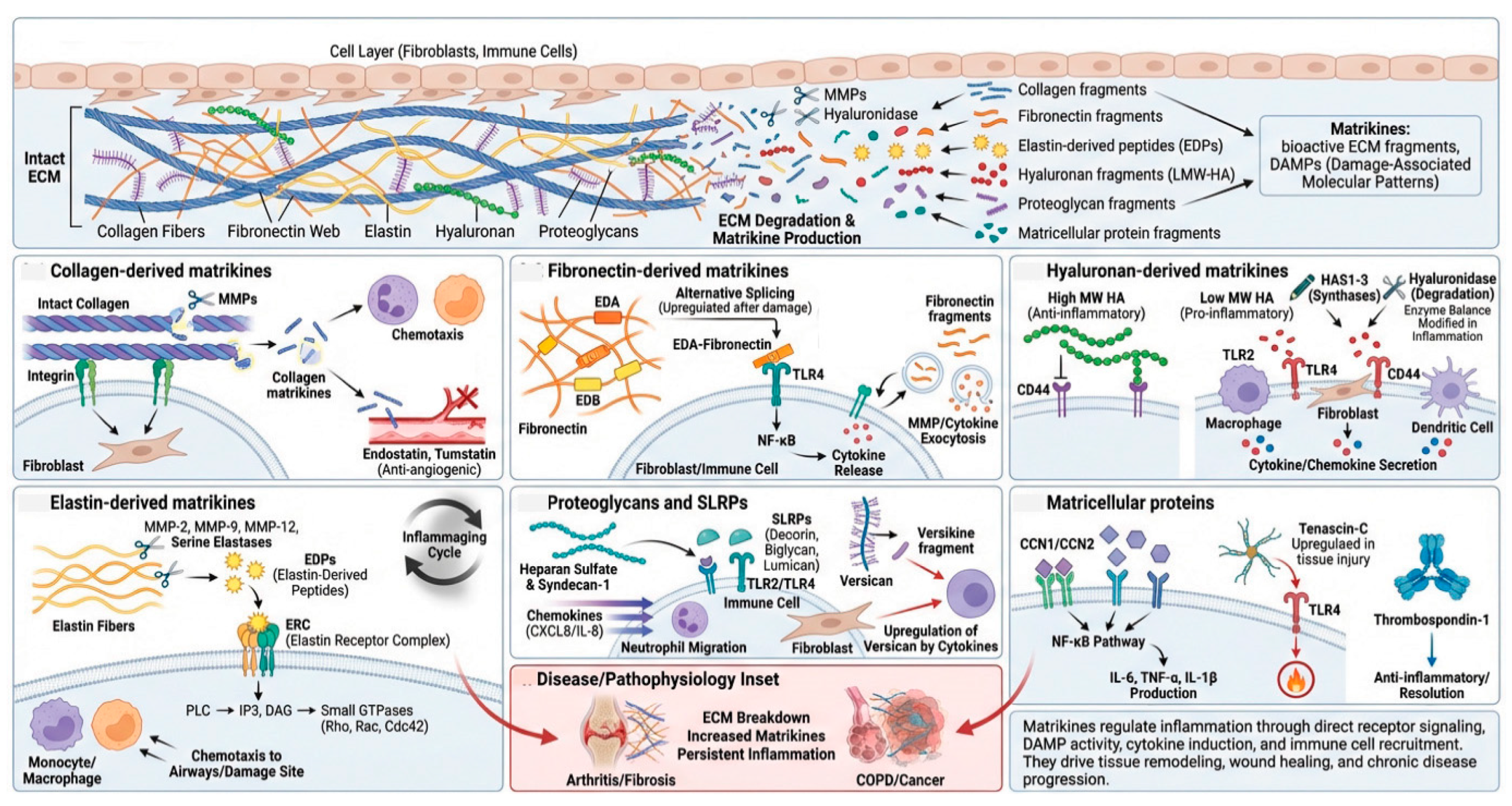
4. Matrikines and Matrisome-Associated Proteins as Inflammatory Modulators
4.1. Collagen-Derived Matrikines
4.2. Fibronectin-Derived Matrikines
4.3. Hyaluronan-Derived Matrikines
4.4. Elastin-Derived Matrikines
4.5. Proteoglycans
4.6. Matricellular Proteins
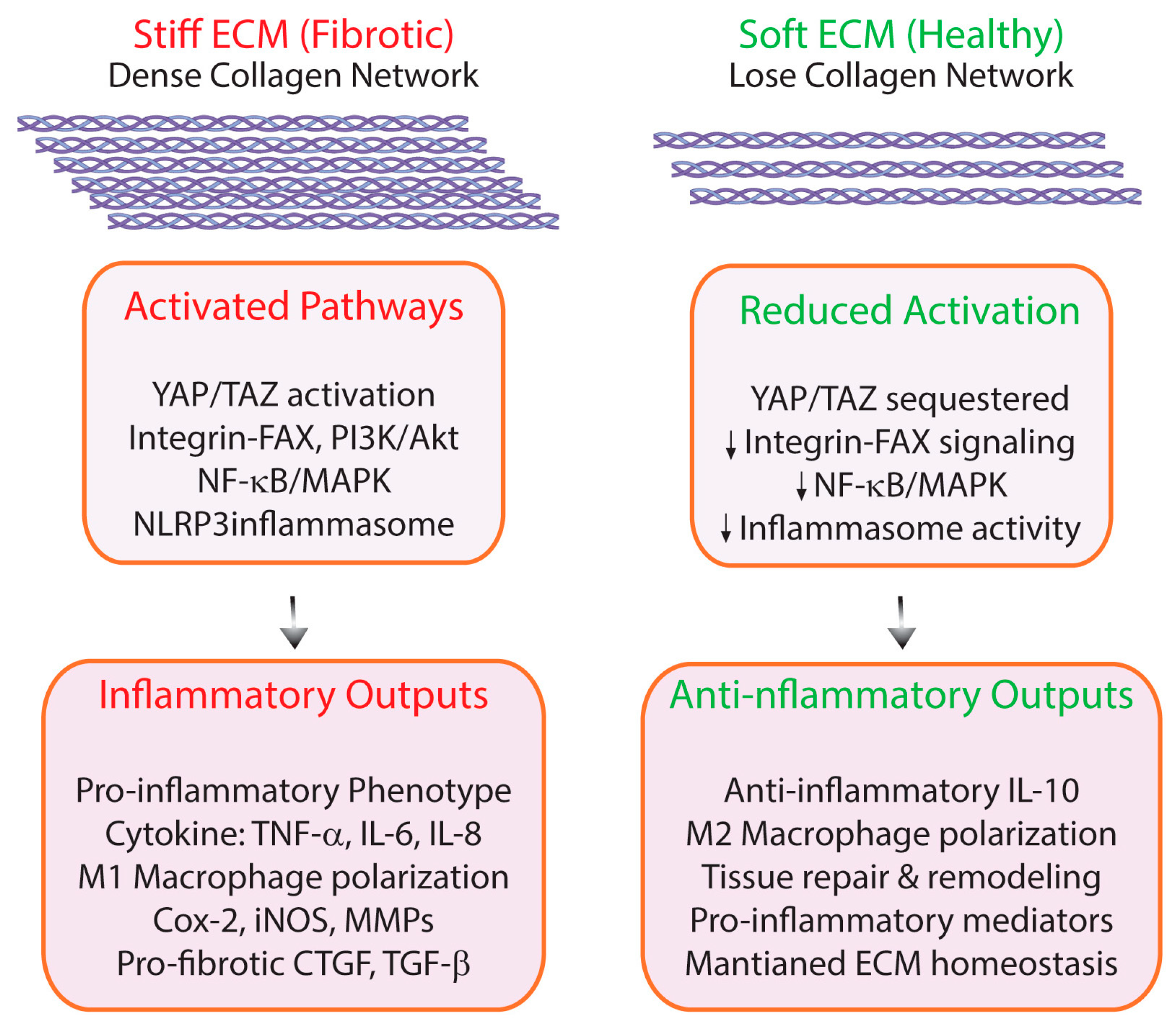
5. ECM Mechanical Properties as Inflammatory Modulators
6. Matrisome-Related Senescence-Associated Secretory Phenotype (SASP) Factors as Inflammatory Modulators
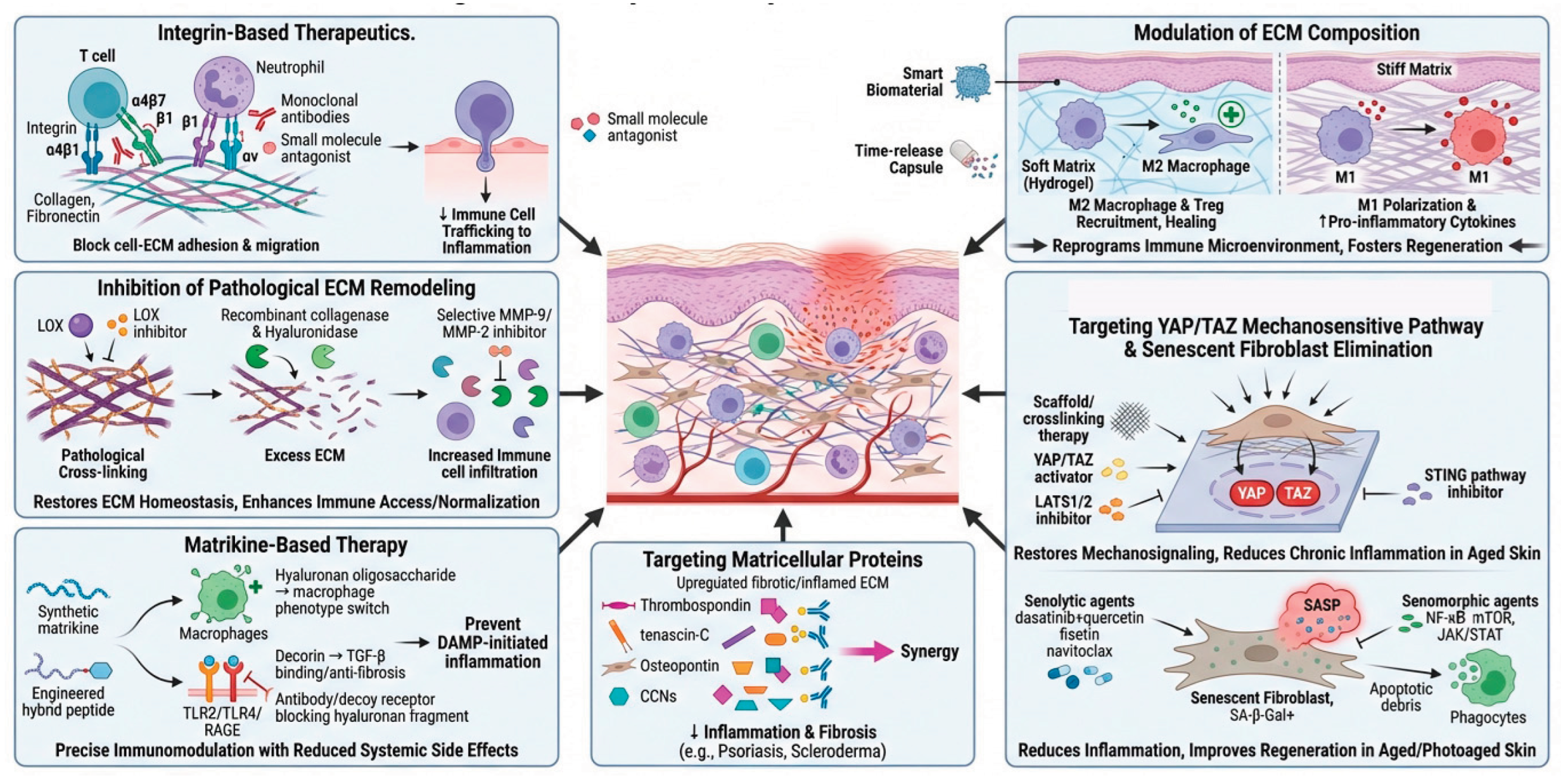
7. Matrisome-Targeted Therapeutic Implications
7.1. Integrin-Based Therapeutics: Disrupting Immune Cell-ECM Adhesion
7.2. Targeting ECM Composition to Modulate Immune Cell Behavior
7.3. Targeting Pathological ECM Remodeling and Mechanical Properties
7.4. Matricellular Proteins as Therapeutic Targets
7.5. Matrikine-Based Therapeutics
7.6. Targeting YAP/TAZ Mechanosensitive Signaling Pathway
7.7. Elimination of Senescent Fibroblasts
8. Future Directions
8.1. High-Resolution Mapping of Matrisome-Immune Interactions
8.2. Mechanobiology of Inflammation
8.3. Matrikine Signaling Networks
9. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sutherland, T.E.; Dyer, D.P.; Allen, J.E. The extracellular matrix and the immune system: A mutually dependent relationship. Science 2023, 379, eabp8964. [Google Scholar] [CrossRef] [PubMed]
- Bonnans, C.; Chou, J.; Werb, Z. Remodelling the extracellular matrix in development and disease. Nat Rev Mol Cell Biol 2014, 15, 786–801. [Google Scholar] [CrossRef]
- Henderson, N.C.; Rieder, F.; Wynn, T.A. Fibrosis: From mechanisms to medicines. Nature 2020, 587, 555–566. [Google Scholar] [CrossRef]
- Hynes, R.O.; Naba, A. Overview of the matrisome--an inventory of extracellular matrix constituents and functions. Cold Spring Harb Perspect Biol 2012, 4, a004903. [Google Scholar] [CrossRef]
- Naba, A.; Clauser, K.R.; Ding, H.; Whittaker, C.A.; Carr, S.A.; Hynes, R.O. The extracellular matrix: Tools and insights for the "omics" era. Matrix Biol 2016, 49, 10–24. [Google Scholar] [CrossRef]
- Naba, A.; Hoersch, S.; Hynes, R.O. Towards definition of an ecm parts list: An advance on go categories. Matrix Biol 2012, 31, 371–372. [Google Scholar] [CrossRef]
- Plikus, M.V.; Wang, X.; Sinha, S.; Forte, E.; Thompson, S.M.; Herzog, E.L.; Driskell, R.R.; Rosenthal, N.; Biernaskie, J.; Horsley, V. Fibroblasts: Origins, definitions, and functions in health and disease. Cell 2021, 184, 3852–3872. [Google Scholar] [CrossRef]
- Tracy, L.E.; Minasian, R.A.; Caterson, E.J. Extracellular matrix and dermal fibroblast function in the healing wound. Adv Wound Care (New Rochelle) 2016, 5, 119–136. [Google Scholar] [CrossRef]
- Hynes, R.O. The extracellular matrix: Not just pretty fibrils. Science 2009, 326, 1216–1219. [Google Scholar] [CrossRef] [PubMed]
- Maquart, F.X.; Pasco, S.; Ramont, L.; Hornebeck, W.; Monboisse, J.C. An introduction to matrikines: Extracellular matrix-derived peptides which regulate cell activity - implication in tumor invasion. Crit Rev Oncol Hemat 2004, 49, 199–202. [Google Scholar] [CrossRef] [PubMed]
- Sorokin, L. The impact of the extracellular matrix on inflammation. Nature Reviews Immunology 2010, 10, 712–723. [Google Scholar] [CrossRef]
- Frevert, C.W.; Felgenhauer, J.; Wygrecka, M.; Nastase, M.V.; Schaefer, L. Danger-associated molecular patterns derived from the extracellular matrix provide temporal control of innate immunity. J Histochem Cytochem 2018, 66, 213–227. [Google Scholar] [CrossRef]
- Boyd, D.F.; Thomas, P.G. Towards integrating extracellular matrix and immunological pathways. Cytokine 2017, 98, 79–86. [Google Scholar] [CrossRef]
- Quan, T.; Xia, W.; He, T.; Calderone, K.; Bou-Gharios, G.; Voorhees, J.J.; Dlugosz, A.A.; Fisher, G.J. Matrix metalloproteinase-1 expression in fibroblasts accelerates dermal aging and promotes papilloma development in mouse skin. J Invest Dermatol 2023, 143, 1700–1707 e1701. [Google Scholar] [CrossRef]
- Quan, T. Molecular insights of human skin epidermal and dermal aging. J Dermatol Sci 2023, 112, 48–53. [Google Scholar] [CrossRef]
- McCabe MC, H.R.; Calderone, K; Cui, Y; Yan, Y; Quan, T; Fisher, GJ; Hansen, KC. Alterations in extracellular matrix composition during aging and photoaging of the skin. Matrix Biology Plus, in press 2020. [Google Scholar]
- Pfisterer, K.; Shaw, L.E.; Symmank, D.; Weninger, W. The extracellular matrix in skin inflammation and infection. Front Cell Dev Biol 2021, 9, 682414. [Google Scholar] [CrossRef]
- Bale, S.; Verma, P.; Varga, J.; Bhattacharyya, S. Extracellular matrix-derived damage-associated molecular patterns (damp): Implications in systemic sclerosis and fibrosis. Journal of Investigative Dermatology 2023, 143, 1877–1885. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, O.; Ayyangar, U.; Kurbet, A.S.; Ashok, D.; Raghavan, S. Unraveling the ecm-immune cell crosstalk in skin diseases. Frontiers in Cell and Developmental Biology 2019, 7. [Google Scholar] [CrossRef] [PubMed]
- Fisher, G.J.; Sachs, D.L.; Voorhees, J.J. Ageing: Collagenase-mediated collagen fragmentation as a rejuvenation target. Br J Dermatol 2014, 171, 446–449. [Google Scholar] [CrossRef] [PubMed]
- Jacob, M.P. Extracellular matrix remodeling and matrix metalloproteinases in the vascular wall during aging and in pathological conditions. Biomed Pharmacother 2003, 57, 195–202. [Google Scholar] [CrossRef]
- Gould, L.; Abadir, P.; Brem, H.; Carter, M.; Conner-Kerr, T.; Davidson, J.; DiPietro, L.; Falanga, V.; Fife, C.; Gardner, S.; et al. Chronic wound repair and healing in older adults: Current status and future research. J Am Geriatr Soc 2015, 63, 427–438. [Google Scholar] [CrossRef]
- Khalid, K.A.; Nawi, A.F.M.; Zulkifli, N.; Barkat, M.A.; Hadi, H. Aging and wound healing of the skin: A review of clinical and pathophysiological hallmarks. Life (Basel) 2022, 12. [Google Scholar] [CrossRef]
- Dotto, G.P. Multifocal epithelial tumors and field cancerization: Stroma as a primary determinant. J Clin Invest 2014, 124, 1446–1453. [Google Scholar] [CrossRef]
- Wolf, K.; Friedl, P. Extracellular matrix determinants of proteolytic and non-proteolytic cell migration. Trends Cell Biol 2011, 21, 736–744. [Google Scholar] [CrossRef]
- Moreno-Layseca, P.; Icha, J.; Hamidi, H.; Ivaska, J. Integrin trafficking in cells and tissues. Nature Cell Biology 2019, 21, 122–132. [Google Scholar] [CrossRef]
- Hogg, N.; Patzak, I.; Willenbrock, F. The insider's guide to leukocyte integrin signalling and function. Nature Reviews Immunology 2011, 11, 416–426. [Google Scholar] [CrossRef] [PubMed]
- Blythe, E.N.; Weaver, L.C.; Brown, A.; Dekaban, G.A. Β2 integrin cd11d/cd18: From expression to an emerging role in staged leukocyte migration. Front Immunol 2021, 12, 775447. [Google Scholar] [CrossRef] [PubMed]
- Bednarczyk, M.; Stege, H.; Grabbe, S.; Bros, M. Β2 integrins-multi-functional leukocyte receptors in health and disease. Int J Mol Sci 2020, 21. [Google Scholar] [CrossRef] [PubMed]
- Nordenfelt, P.; Moore, T.I.; Mehta, S.B.; Kalappurakkal, J.M.; Swaminathan, V.; Koga, N.; Lambert, T.J.; Baker, D.; Waters, J.C.; Oldenbourg, R.; et al. Direction of actin flow dictates integrin lfa-1 orientation during leukocyte migration. Nature Communications 2017, 8, 2047. [Google Scholar] [CrossRef]
- Shi, H.; Shao, B. Lfa-1 activation in t-cell migration and immunological synapse formation. Cells 2023, 12, 1136. [Google Scholar] [CrossRef]
- Evans, R.; Patzak, I.; Svensson, L.; De Filippo, K.; Jones, K.; McDowall, A.; Hogg, N. Integrins in immunity. Journal of Cell Science 2009, 122, 215–225. [Google Scholar] [CrossRef]
- Chan, P.Y.; Aruffo, A. Vla-4 integrin mediates lymphocyte migration on the inducible endothelial cell ligand vcam-1 and the extracellular matrix ligand fibronectin. J Biol Chem 1993, 268, 24655–24664. [Google Scholar] [CrossRef] [PubMed]
- Hauzenberger, D.; Klominek, J.; Sundqvist, K.G. Functional specialization of fibronectin-binding beta 1-integrins in t lymphocyte migration. The Journal of Immunology 1994, 153, 960–971. [Google Scholar] [CrossRef]
- Zhu, Y.; Huang, Y.; Ji, Q.; Fu, S.; Gu, J.; Tai, N.; Wang, X. Interplay between extracellular matrix and neutrophils in diseases. J Immunol Res 2021, 2021, 8243378. [Google Scholar] [CrossRef]
- Ong, C.W.; Elkington, P.T.; Brilha, S.; Ugarte-Gil, C.; Tome-Esteban, M.T.; Tezera, L.B.; Pabisiak, P.J.; Moores, R.C.; Sathyamoorthy, T.; Patel, V.; et al. Neutrophil-derived mmp-8 drives ampk-dependent matrix destruction in human pulmonary tuberculosis. PLoS Pathog 2015, 11, e1004917. [Google Scholar] [CrossRef] [PubMed]
- Dean, R.A.; Cox, J.H.; Bellac, C.L.; Doucet, A.; Starr, A.E.; Overall, C.M. Macrophage-specific metalloelastase (mmp-12) truncates and inactivates elr+ cxc chemokines and generates ccl2, -7, -8, and -13 antagonists: Potential role of the macrophage in terminating polymorphonuclear leukocyte influx. Blood 2008, 112, 3455–3464. [Google Scholar] [CrossRef]
- Esparza, J.; Vilardell, C.; Calvo, J.; Juan, M.; Vives, J.; Urbano-Márquez, A.; Yagüe, J.; Cid, M.C. Fibronectin upregulates gelatinase b (mmp-9) and induces coordinated expression of gelatinase a (mmp-2) and its activator mt1-mmp (mmp-14) by human t lymphocyte cell lines. A process repressed through ras/map kinase signaling pathways. Blood 1999, 94, 2754–2766. [Google Scholar] [CrossRef] [PubMed]
- Wolf, K.; Müller, R.; Borgmann, S.; Bröcker, E.B.; Friedl, P. Amoeboid shape change and contact guidance: T-lymphocyte crawling through fibrillar collagen is independent of matrix remodeling by mmps and other proteases. Blood 2003, 102, 3262–3269. [Google Scholar] [CrossRef]
- Ullo, M.F.; D’Amico, A.E.; Lavenus, S.B.; Logue, J.S. The amoeboid migration of monocytes in confining channels requires the local remodeling of the cortical actin cytoskeleton by cofilin-1. Sci Rep-Uk 2024, 14, 10241. [Google Scholar] [CrossRef]
- Crijns, H.; Vanheule, V.; Proost, P. Targeting chemokine-glycosaminoglycan interactions to inhibit inflammation. Front Immunol 2020, 11, 483. [Google Scholar] [CrossRef]
- McQuitty, C.E.; Williams, R.; Chokshi, S.; Urbani, L. Immunomodulatory role of the extracellular matrix within the liver disease microenvironment. Frontiers in Immunology 2020, 11. [Google Scholar] [CrossRef]
- Wynn, T.A.; Vannella, K.M. Macrophages in tissue repair, regeneration, and fibrosis. Immunity 2016, 44, 450–462. [Google Scholar] [CrossRef] [PubMed]
- Papayannopoulos, V. Neutrophil extracellular traps in immunity and disease. Nature Reviews Immunology 2018, 18, 134–147. [Google Scholar] [CrossRef]
- Murray, P.J.; Allen, J.E.; Biswas, S.K.; Fisher, E.A.; Gilroy, D.W.; Goerdt, S.; Gordon, S.; Hamilton, J.A.; Ivashkiv, L.B.; Lawrence, T.; et al. Macrophage activation and polarization: Nomenclature and experimental guidelines (vol 41, pg 14, 2014). Immunity 2014, 41, 339–340. [Google Scholar] [CrossRef]
- Wynn, T.A. Type 2 cytokines: Mechanisms and therapeutic strategies. Nature Reviews Immunology 2015, 15, 271–282. [Google Scholar] [CrossRef]
- Tchougounova, E.; Lundequist, A.; Fajardo, I.; Winberg, J.O.; Åbrink, M.; Pejler, G. A key role for mast cell chymase in the activation of pro-matrix metalloprotease-9 and pro-matrix metalloprotease-2. Journal of Biological Chemistry 2005, 280, 9291–9296. [Google Scholar] [CrossRef]
- Hussain, T.; Tan, B.; Yin, Y.; Blachier, F.; Tossou, M.C.B.; Rahu, N. Oxidative stress and inflammation: What polyphenols can do for us? Oxidative Medicine and Cellular Longevity 2016, 2016, 7432797. [Google Scholar] [CrossRef]
- Lu, P.; Weaver, V.M.; Werb, Z. The extracellular matrix: A dynamic niche in cancer progression. J Cell Biol 2012, 196, 395–406. [Google Scholar] [CrossRef]
- Link, A.; Vogt, T.K.; Favre, S.; Britschgi, M.R.; Acha-Orbea, H.; Hinz, B.; Cyster, J.G.; Luther, S.A. Fibroblastic reticular cells in lymph nodes regulate the homeostasis of naive t cells. Nature Immunology 2007, 8, 1255–1265. [Google Scholar] [CrossRef] [PubMed]
- Morrison, S.J.; Scadden, D.T. The bone marrow niche for haematopoietic stem cells. Nature 2014, 505, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Heissig, B.; Hattori, K.; Dias, S.; Friedrich, M.; Ferris, B.; Hackett, N.R.; Crystal, R.G.; Besmer, P.; Lyden, D.; Moore, M.A.S.; et al. Recruitment of stem and progenitor cells from the bone marrow niche requires mmp-9 mediated release of kit-ligand. Cell 2002, 109, 625–637. [Google Scholar] [CrossRef]
- Stier, S.; Ko, Y.; Forkert, R.; Lutz, C.; Neuhaus, T.; Grünewald, E.; Cheng, T.; Dombkowski, D.; Calvi, L.M.; Rittling, S.R.; et al. Osteopontin is a hematopoietic stem cell niche component that negatively regulates stem cell pool size. Journal of Experimental Medicine 2005, 201, 1781–1791. [Google Scholar] [CrossRef]
- Chiquet-Ehrismann, R.; Orend, G.; Chiquet, M.; Tucker, R.P.; Midwood, K.S. Tenascins in stem cell niches. Matrix Biology 2014, 37, 112–123. [Google Scholar] [CrossRef]
- Di Vito, A.; Donato, A.; Bria, J.; Conforti, F.; La Torre, D.; Malara, N.; Donato, G. Extracellular matrix structure and interaction with immune cells in adult astrocytic tumors. Cell Mol Neurobiol 2024, 44. [Google Scholar] [CrossRef]
- Huse, M. Mechanical forces in the immune system. Nature Reviews Immunology 2017, 17, 679–690. [Google Scholar] [CrossRef] [PubMed]
- Wynn, T.A.; Ramalingam, T.R. Mechanisms of fibrosis: Therapeutic translation for fibrotic disease. Nat Med 2012, 18, 1028–1040. [Google Scholar] [CrossRef]
- Pakshir, P.; Hinz, B. The big five in fibrosis: Macrophages, myofibroblasts, matrix, mechanics, and miscommunication. Matrix Biology 2018, 68-69, 81-93. [Google Scholar] [CrossRef] [PubMed]
- McInnes, I.B.; Schett, G. Mechanisms of disease the pathogenesis of rheumatoid arthritis. New Engl J Med 2011, 365, 2205–2219. [Google Scholar] [CrossRef]
- Trouw, L.A.; Rispens, T.; Toes, R.E.M. Beyond citrullination: Other post-translational protein modifications in rheumatoid arthritis. Nature Reviews Rheumatology 2017, 13, 331–339. [Google Scholar] [CrossRef] [PubMed]
- Hinshaw, D.C.; Shevde, L.A. The tumor microenvironment innately modulates cancer progression. Cancer Research 2019, 79, 4557–4566. [Google Scholar] [CrossRef]
- Salmon, H.; Franciszkiewicz, K.; Damotte, D.; Dieu-Nosjean, M.C.; Validire, P.; Trautmann, A.; Mami-Chouaib, F.; Donnadieu, E. Matrix architecture defines the preferential localization and migration of t cells into the stroma of human lung tumors. Journal of Clinical Investigation 2012, 122, 899–910. [Google Scholar] [CrossRef]
- Chitty, J.L.; Yam, M.; Perryman, L.; Parker, A.L.; Skhinas, J.N.; Setargew, Y.F.I.; Mok, E.T.Y.; Tran, E.; Grant, R.D.; Latham, S.L.; et al. A first-in-class pan-lysyl oxidase inhibitor impairs stromal remodeling and enhances gemcitabine response and survival in pancreatic cancer. Nat Cancer 2023, 4, 1326–+. [Google Scholar] [CrossRef]
- Levental, K.R.; Yu, H.M.; Kass, L.; Lakins, J.N.; Egeblad, M.; Erler, J.T.; Fong, S.F.T.; Csiszar, K.; Giaccia, A.; Weninger, W.; et al. Matrix crosslinking forces tumor progression by enhancing integrin signaling. Cell 2009, 139, 891–906. [Google Scholar] [CrossRef]
- Sladitschek-Martens, H.L.; Guarnieri, A.; Brumana, G.; Zanconato, F.; Battilana, G.; Xiccato, R.L.; Panciera, T.; Forcato, M.; Bicciato, S.; Guzzardo, V.; et al. Yap/taz activity in stromal cells prevents ageing by controlling cgas-sting. Nature 2022, 607, 790–798. [Google Scholar] [CrossRef]
- Quintero-Fabian, S.; Arreola, R.; Becerril-Villanueva, E.; Torres-Romero, J.C.; Arana-Argaez, V.; Lara-Riegos, J.; Ramirez-Camacho, M.A.; Alvarez-Sanchez, M.E. Role of matrix metalloproteinases in angiogenesis and cancer. Front Oncol 2019, 9, 1370. [Google Scholar] [CrossRef] [PubMed]
- Nissinen, L.; Kähäri, V.M. Matrix metalloproteinases in inflammation. Bba-Gen Subjects 2014, 1840, 2571–2580. [Google Scholar] [CrossRef] [PubMed]
- Wells, J.M.; Gaggar, A.; Blalock, J.E. Mmp generated matrikines. Matrix Biol 2015, 44-46, 122–129. [Google Scholar] [CrossRef]
- Köhrmann, A.; Kammerer, U.; Kapp, M.; Dietl, J.; Anacker, J. Expression of matrix metalloproteinases (mmps) in primary human breast cancer and breast cancer cell lines: New findings and review of the literature. Bmc Cancer 2009, 9. [Google Scholar] [CrossRef]
- de Almeida, L.G.N.; Thode, H.; Eslambolchi, Y.; Chopra, S.; Young, D.; Gill, S.; Devel, L.; Dufour, A. Matrix metalloproteinases: From molecular mechanisms to physiology, pathophysiology, and pharmacology. Pharmacological Reviews 2022, 74, 714–770. [Google Scholar] [CrossRef] [PubMed]
- Quan, T.; Qin, Z.; Xia, W.; Shao, Y.; Voorhees, J.J.; Fisher, G.J. Matrix-degrading metalloproteinases in photoaging. J Investig Dermatol Symp Proc 2009, 14, 20–24. [Google Scholar] [CrossRef]
- Siwik, D.A.; Chang, D.L.F.; Colucci, W.S. Interleukin-1β and tumor necrosis factor-α decrease collagen synthesis and increase matrix metalloproteinase activity in cardiac fibroblasts in vitro. Circulation Research 2000, 86, 1259–1265. [Google Scholar] [CrossRef]
- Dasu, M.R.; Barrow, R.E.; Spies, M.; Herndon, D.N. Matrix metalloproteinase expression in cytokine stimulated human dermal fibroblasts. Burns 2003, 29, 527–531. [Google Scholar] [CrossRef]
- Cortez, D.M.; Feldman, M.D.; Mummidi, S.; Valente, A.J.; Steffensen, B.; Vincenti, M.; Barnes, J.L.; Chandrasekar, B. Il-17 stimulates mmp-1 expression in primary human cardiac fibroblasts via p38 mapk- and erk1/2-dependent c/ebp-beta, nf-kappab, and ap-1 activation. Am J Physiol Heart Circ Physiol 2007, 293, H3356–3365. [Google Scholar] [CrossRef] [PubMed]
- Kessenbrock, K.; Plaks, V.; Werb, Z. Matrix metalloproteinases: Regulators of the tumor microenvironment. Cell 2010, 141, 52–67. [Google Scholar] [CrossRef]
- Becker-Pauly, C.; Rose-John, S. Tnfα cleavage beyond tace/adam17: Matrix metalloproteinase 13 is a potential therapeutic target in sepsis and colitis. EMBO Molecular Medicine 2013, 5, EMMM201302899. [Google Scholar] [CrossRef] [PubMed]
- Elkington, P.T.; O'Kane, C.M.; Friedland, J.S. The paradox of matrix metalloproteinases in infectious disease. Clin Exp Immunol 2005, 142, 12–20. [Google Scholar] [CrossRef]
- Liu, J.; Khalil, R.A. Matrix metalloproteinase inhibitors as investigational and therapeutic tools in unrestrained tissue remodeling and pathological disorders. Prog Mol Biol Transl Sci 2017, 148, 355–420. [Google Scholar]
- Kazemi, A.; Fathy, M.; Jahanian, A.; Khanali, J.; Ostadi, Y.; Babajani, A.; Tayebi, T.; Niknejad, H. The role of mmps and timps in regenerative medicine: From pathological ecm remodeling to therapeutic applications. Biomed Pharmacother 2025, 191, 118457. [Google Scholar] [CrossRef] [PubMed]
- Caley, M.P.; Martins, V.L.; O'Toole, E.A. Metalloproteinases and wound healing. Adv Wound Care (New Rochelle) 2015, 4, 225–234. [Google Scholar] [CrossRef]
- Leong, E.; Bezuhly, M.; Marshall, J.S. Distinct metalloproteinase expression and functions in systemic sclerosis and fibrosis: What we know and the potential for intervention. Front Physiol 2021, 12, 727451. [Google Scholar] [CrossRef]
- Jariwala, N.; Ozols, M.; Bell, M.; Bradley, E.; Bradley, E.; Gilmore, A.; Debelle, L.; Sherratt, M.J. Matrikines as mediators of tissue remodelling. Adv Drug Deliver Rev 2022, 185. [Google Scholar] [CrossRef] [PubMed]
- Adair-Kirk, T.L.; Senior, R.M. Fragments of extracellular matrix as mediators of inflammation. Int J Biochem Cell B 2008, 40, 1101–1110. [Google Scholar] [CrossRef]
- Ricard-Blum, S.; Vallet, S.D. Fragments generated upon extracellular matrix remodeling: Biological regulators and potential drugs. Matrix Biology 2019, 75-76, 170-189. [Google Scholar] [CrossRef]
- Kadler, K.E.; Baldock, C.; Bella, J.; Boot-Handford, R.P. Collagens at a glance. Journal of Cell Science 2007, 120, 1955–1958. [Google Scholar] [CrossRef]
- Quan, T.; Qin, Z.; He, T.; Fisher, G.J. Integrin alpha11beta1 as a key collagen receptor in human skin dermis: Insight into fibroblast function and skin dermal aging. J Invest Dermatol 2025, 145, 2449–2463. [Google Scholar] [CrossRef] [PubMed]
- Senior, R.M.; Hinek, A.; Griffin, G.L.; Pipoly, D.J.; Crouch, E.C.; Mecham, R.P. Neutrophils show chemotaxis to type-iv collagen and its 7s domain and contain a 67-kd type-iv collagen binding-protein with lectin properties. Am J Resp Cell Mol 1989, 1, 479–487. [Google Scholar] [CrossRef]
- Jensen, C.; Sinkeviciute, D.; Madsen, D.H.; Önnerfjord, P.; Hansen, M.; Schmidt, H.; Karsdal, M.A.; Svane, I.M.; Willumsen, N. Granzyme b degraded type iv collagen products in serum identify melanoma patients responding to immune checkpoint blockade. Cancers 2020, 12, 2786. [Google Scholar] [CrossRef] [PubMed]
- Alexdottir, M.S.; Pehrsson, M.; Domislovic, V.; Godskesen, L.E.; Krag, A.; Kjeldsen, J.; Brinar, M.; Barisic, A.; Bay-Jensen, A.-C.; Karsdal, M.A.; et al. Neutrophil-mediated type iv collagen degradation is elevated in patients with mild endoscopic ulcerative colitis reflecting early mucosal destruction. Sci Rep-Uk 2024, 14, 1641. [Google Scholar] [CrossRef]
- Walia, A.; Yang, J.F.; Huang, Y.H.; Rosenblatt, M.I.; Chang, J.H.; Azar, D.T. Endostatin's emerging roles in angiogenesis, lymphangiogenesis, disease, and clinical applications. Biochim Biophys Acta 2015, 1850, 2422–2438. [Google Scholar] [CrossRef]
- Okamura, Y.; Watari, M.; Jerud, E.S.; Young, D.W.; Ishizaka, S.T.; Rose, J.; Chow, J.C.; Strauss, J.I.I.I. The extra domain a of fibronectin activates toll-like receptor 4. Journal of Biological Chemistry 2001, 276, 10229–10233. [Google Scholar] [CrossRef]
- Lasarte, J.J.; Casares, N.; Gorraiz, M.; Hervás-Stubbs, S.; Arribillaga, L.; Mansilla, C.; Durantez, M.; Llopiz, D.; Sarobe, P.; Borrás-Cuesta, F.; et al. The extra domain a from fibronectin targets antigens to tlr4-expressing cells and induces cytotoxic t cell responses in vivo. Journal of Immunology 2007, 178, 748–756. [Google Scholar] [CrossRef]
- White, E.S.; Muro, A.F. Fibronectin splice variants: Understanding their multiple roles in health and disease using engineered mouse models. IUBMB Life 2011, 63, 538–546. [Google Scholar] [CrossRef]
- Gondokaryono, S.P.; Ushio, H.; Niyonsaba, F.; Hara, M.; Takenaka, H.; Jayawardana, S.T.M.; Ikeda, S.; Okumura, K.; Ogawa, H. The extra domain a of fibronectin stimulates murine mast cells via toll-like receptor 4. J Leukocyte Biol 2007, 82, 657–665. [Google Scholar] [CrossRef]
- Lefebvre, J.S.; Lévesque, T.; Picard, S.; Paré, G.; Gravel, A.; Flamand, L.; Borgeat, P. Extra domain a of fibronectin primes leukotriene biosynthesis and stimulates neutrophil migration through activation of toll-like receptor 4. Arthritis Rheum 2011, 63, 1527–1533. [Google Scholar] [CrossRef]
- Pankov, R.; Yamada, K.M. Fibronectin at a glance. Journal of Cell Science 2002, 115, 3861–3863. [Google Scholar] [CrossRef] [PubMed]
- Barilla, M.L.; Carsons, S.E. Fibronectin fragments and their role in inflammatory arthritis. Semin Arthritis Rheu 2000, 29, 252–265. [Google Scholar] [CrossRef] [PubMed]
- Grinnell, F.; Zhu, M. Fibronectin degradation in chronic wounds depends on the relative levels of elastase, alpha1-proteinase inhibitor, and alpha2-macroglobulin. J Invest Dermatol 1996, 106, 335–341. [Google Scholar] [CrossRef]
- Grinnell, F.; Ho, C.H.; Wysocki, A. Degradation of fibronectin and vitronectin in chronic wound fluid: Analysis by cell blotting, immunoblotting, and cell adhesion assays. J Invest Dermatol 1992, 98, 410–416. [Google Scholar] [CrossRef]
- Tavianatou, A.G.; Caon, I.; Franchi, M.; Piperigkou, Z.; Galesso, D.; Karamanos, N.K. Hyaluronan: Molecular size-dependent signaling and biological functions in inflammation and cancer. 2019, 286, 2883–2908. [Google Scholar] [CrossRef]
- Petrey, A.C.; de la Motte, C.A. Hyaluronan, a crucial regulator of inflammation. Front Immunol 2014, 5, 101. [Google Scholar] [CrossRef] [PubMed]
- Scheibner, K.A.; Lutz, M.A.; Boodoo, S.; Fenton, M.J.; Powell, J.D.; Horton, M.R. Hyaluronan fragments act as an endogenous danger signal by engaging tlr2. J Immunol 2006, 177, 1272–1281. [Google Scholar] [CrossRef]
- Žádníková, P.; Šínová, R.; Pavlík, V.; Šimek, M.; Šafránková, B.; Hermannová, M.; Nešporová, K.; Velebný, V. The degradation of hyaluronan in the skin. Biomolecules 2022, 12, 251. [Google Scholar] [CrossRef]
- Terazawa, S.; Nakajima, H.; Tobita, K.; Imokawa, G. The decreased secretion of hyaluronan by older human fibroblasts under physiological conditions is mainly associated with the down-regulated expression of hyaluronan synthases but not with the expression levels of hyaluronidases. Cytotechnology 2015, 67, 609–620. [Google Scholar] [CrossRef] [PubMed]
- Jiang, D.; Liang, J.; Noble, P.W. Hyaluronan as an immune regulator in human diseases. Physiological Reviews 2011, 91, 221–264. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.K.; Meng, X.G.; Guo, Z.K. Elastin structure, synthesis, regulatory mechanism and relationship with cardiovascular diseases. Frontiers in Cell and Developmental Biology 2021, 9. [Google Scholar] [CrossRef]
- Duca, L.; Floquet, N.; Alix, A.J.P.; Haye, B.; Debelle, L. Elastin as a matrikine. Crit Rev Oncol Hemat 2004, 49, 235–244. [Google Scholar] [CrossRef]
- Le Page, A.; Khalil, A.; Vermette, P.; Frost, E.H.; Larbi, A.; Witkowski, J.M.; Fulop, T. The role of elastin-derived peptides in human physiology and diseases. Matrix Biology 2019, 84, 81–96. [Google Scholar] [CrossRef]
- Tembely, D.; Henry, A.; Vanalderwiert, L.; Toussaint, K.; Bennasroune, A.; Blaise, S.; Sartelet, H.; Jaisson, S.; Gales, C.; Martiny, L.; et al. The elastin receptor complex: An emerging therapeutic target against age-related vascular diseases. Front Endocrinol 2022, 13. [Google Scholar] [CrossRef] [PubMed]
- Mochizuki, S.; Brassart, B.; Hinek, A. Signaling pathways transduced through the elastin receptor facilitate proliferation of arterial smooth muscle cells. Journal of Biological Chemistry 2002, 277, 44854–44863. [Google Scholar] [CrossRef]
- Scandolera, A.; Odoul, L.; Salesse, S.; Guillot, A.; Blaise, S.; Kawecki, C.; Maurice, P.; El Btaouri, H.; Romier-Crouzet, B.; Martiny, L.; et al. The elastin receptor complex: A unique matricellular receptor with high anti-tumoral potential. Frontiers in Pharmacology 2016, 7. [Google Scholar] [CrossRef]
- Cantor, J. Elastin peptides as a potential disease vector in the pathogenesis of pulmonary emphysema: An investigation of this hypothesis. Life 2025, 15, 356. [Google Scholar] [CrossRef]
- Mecham, R.P. Elastin in lung development and disease pathogenesis. Matrix Biol 2018, 73, 6–20. [Google Scholar] [CrossRef]
- Yi, J.Z.; Wang, Y.X.; Sui, H.R.; Chen, Z.C.; Ye, T.N.; Zhong, Y.L.; Qian, J.Y.; Wu, B.B.; Huang, J.Y.; Tian, T.; et al. Elastin-derived extracellular matrix fragments drive aging through innate immune activation. Nature Aging 2025. [Google Scholar] [CrossRef]
- Jin, S.M.; Elisseeff, J.H. Aging as a glitch in the matrix. In Nature Aging; 2025. [Google Scholar]
- Gill, S.; Wight, T.N.; Frevert, C.W. Proteoglycans: Key regulators of pulmonary inflammation and the innate immune response to lung infection. Anat Rec (Hoboken) 2010, 293, 968–981. [Google Scholar] [CrossRef] [PubMed]
- Rajarathnam, K.; Desai, U.R. Structural insights into how proteoglycans determine chemokine-cxcr1/cxcr2 interactions: Progress and challenges. Frontiers in Immunology 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Hayashida, K.; Parks, W.C.; Park, P.W. Syndecan-1 shedding facilitates the resolution of neutrophilic inflammation by removing sequestered cxc chemokines. Blood 2009, 114, 3033–3043. [Google Scholar] [CrossRef] [PubMed]
- Nastase, M.V.; Janicova, A.; Roedig, H.; Hsieh, L.T.; Wygrecka, M.; Schaefer, L. Small leucine-rich proteoglycans in renal inflammation: Two sides of the coin. J Histochem Cytochem 2018, 66, 261–272. [Google Scholar] [CrossRef]
- Merline, R.; Moreth, K.; Beckmann, J.; Nastase, M.V.; Zeng-Brouwers, J.; Tralhão, J.G.; Lemarchand, P.; Pfeilschifter, J.; Schaefer, R.M.; Iozzo, R.V.; et al. Signaling by the matrix proteoglycan decorin controls inflammation and cancer through pdcd4 and microrna-21. Sci Signal 2011, 4, ra75. [Google Scholar] [CrossRef]
- Nastase, M.V.; Young, M.F.; Schaefer, L. Biglycan: A multivalent proteoglycan providing structure and signals. J Histochem Cytochem 2012, 60, 963–975. [Google Scholar] [CrossRef]
- Lohr, K.; Sardana, H.; Lee, S.; Wu, F.; Huso, D.L.; Hamad, A.R.; Chakravarti, S. Extracellular matrix protein lumican regulates inflammation in a mouse model of colitis. Inflamm Bowel Dis 2012, 18, 143–151. [Google Scholar] [CrossRef]
- Wight, T.N.; Kang, I.; Merrilees, M.J. Versican and the control of inflammation. Matrix Biol 2014, 35, 152–161. [Google Scholar]
- Wight, T.N.; Kang, I.; Evanko, S.P.; Harten, I.A.; Chang, M.Y.; Pearce, O.M.T.; Allen, C.E.; Frevert, C.W. Versican-a critical extracellular matrix regulator of immunity and inflammation. Frontiers in Immunology 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Eun, K.; Kim, A.Y.; Ryu, S. Matricellular proteins in immunometabolism and tissue homeostasis. Bmb Rep 2024, 57, 400–416. [Google Scholar] [CrossRef]
- Holbourn, K.P.; Acharya, K.R.; Perbal, B. The ccn family of proteins: Structure–function relationships. Trends in Biochemical Sciences 2008, 33, 461–473. [Google Scholar] [CrossRef]
- Leask, A.; Abraham, D.J. All in the ccn family: Essential matricellular signaling modulators emerge from the bunker. Journal of Cell Science 2006, 119, 4803–4810. [Google Scholar] [CrossRef]
- Kular, L.; Pakradouni, J.; Kitabgi, P.; Laurent, M.; Martinerie, C. The ccn family: A new class of inflammation modulators? Biochimie 2011, 93, 377–388. [Google Scholar] [CrossRef]
- Jun, J.I.; Lau, L.F. Taking aim at the extracellular matrix: Ccn proteins as emerging therapeutic targets. Nat Rev Drug Discov 2011, 10, 945–963. [Google Scholar] [CrossRef] [PubMed]
- Jiang, R.Q.; Tang, J.F.; Zhang, X.H.; He, Y.J.; Yu, Z.Q.; Chen, S.H.; Xia, J.F.; Lin, J.P.; Ou, Q.S. Ccn1 promotes inflammation by inducing il-6 production α6β1/pi3k/akt/nf-κb pathway in autoimmune hepatitis. Frontiers in Immunology 2022, 13. [Google Scholar] [CrossRef]
- Bai, T.; Chen, C.C.; Lau, L.F. Matricellular protein ccn1 activates a proinflammatory genetic program in murine macrophages. J Immunol 2010, 184, 3223–3232. [Google Scholar] [CrossRef]
- Leask, A. Conjunction junction, what’s the function? Ccn proteins as targets in fibrosis and cancers. Am J Physiol-Cell Ph 2020, 318, C1046–C1054. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; Li, L.; Lu, Q.; Wang, L.H.; Liu, C.Y.; Lei, Q.; Guan, K.L. Angiomotin is a novel hippo pathway component that inhibits yap oncoprotein. Genes Dev 2011, 25, 51–63. [Google Scholar] [CrossRef]
- Imanaka-Yoshida, K. Tenascin-c in heart diseases-the role of inflammation. Int J Mol Sci 2021, 22. [Google Scholar] [CrossRef]
- Midwood, K.; Sacre, S.; Piccinini, A.M.; Inglis, J.; Trebaul, A.; Chan, E.; Drexler, S.; Sofat, N.; Kashiwagi, M.; Orend, G.; et al. Tenascin-c is an endogenous activator of toll-like receptor 4 that is essential for maintaining inflammation in arthritic joint disease. Nat Med 2009, 15, 774–780. [Google Scholar] [CrossRef]
- Midwood, K.S.; Orend, G. The role of tenascin-c in tissue injury and tumorigenesis. J Cell Commun Signal 2009, 3, 287–310. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Olonisakin, T.F.; Xiong, Z.; Hulver, M.; Sayeed, S.; Yu, M.T.; Gregory, A.D.; Kochman, E.J.; Chen, B.B.; Mallampalli, R.K.; et al. Thrombospondin-1 restrains neutrophil granule serine protease function and regulates the innate immune response during klebsiella pneumoniae infection. Mucosal Immunology 2015, 8, 896–905. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.W.; Liu, C.; Li, Y.J.; Wang, W.F.; Dou, Q.L. The dual role of thrombospondin-1 in inflammatory regulation during acute respiratory distress syndrome: A mini-review. Frontiers in Immunology 2025, 16. [Google Scholar] [CrossRef]
- Di, X.; Gao, X.; Peng, L.; Ai, J.; Jin, X.; Qi, S.; Li, H.; Wang, K.; Luo, D. Cellular mechanotransduction in health and diseases: From molecular mechanism to therapeutic targets. Signal Transduction and Targeted Therapy 2023, 8, 282. [Google Scholar] [CrossRef]
- Yusko, E.C.; Asbury, C.L. Force is a signal that cells cannot ignore. Mol Biol Cell 2014, 25, 3717–3725. [Google Scholar] [CrossRef] [PubMed]
- Mai, Z.; Lin, Y.; Lin, P.; Zhao, X.; Cui, L. Modulating extracellular matrix stiffness: A strategic approach to boost cancer immunotherapy. Cell Death Dis 2024, 15, 307. [Google Scholar] [CrossRef]
- Jiang, T.; Zheng, M.T.; Li, R.M.; Ouyang, N.J. The effects of matrix stiffness on immune cells in bone biology. Mechanobiol Med 2024, 2. [Google Scholar] [CrossRef]
- Tiskratok, W.; Chuinsiri, N.; Limraksasin, P.; Kyawsoewin, M.; Jitprasertwong, P. Extracellular matrix stiffness: Mechanotransduction and mechanobiological response-driven strategies for biomedical applications targeting fibroblast inflammation. Polymers 2025, 17, 822. [Google Scholar] [CrossRef] [PubMed]
- Saha, S.; Müller, D.; Clark, A.G. Mechanosensory feedback loops during chronic inflammation. Front Cell Dev Biol 2023, 11, 1225677. [Google Scholar] [CrossRef]
- Mei, F.; Guo, Y.; Wang, Y.; Zhou, Y.; Heng, B.C.; Xie, M.; Huang, X.; Zhang, S.; Ding, S.; Liu, F.; et al. Matrix stiffness regulates macrophage polarisation via the piezo1-yap signalling axis. Cell Prolif 2024, 57, e13640. [Google Scholar] [CrossRef] [PubMed]
- Gruber, E.; Heyward, C.; Cameron, J.; Leifer, C. Toll-like receptor signaling in macrophages is regulated by extracellular substrate stiffness and rho-associated coiled-coil kinase (rock1/2). Int Immunol 2018, 30, 267–278. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.S.; Dolgova, N.V.; Chancellor, T.J.; Acharya, A.P.; Karpiak, J.V.; Lele, T.P.; Keselowsky, B.G. The effect of cyclic mechanical strain on activation of dendritic cells cultured on adhesive substrates. Biomaterials 2013, 34, 9063–9070. [Google Scholar] [CrossRef]
- Lee, M.G.; Du, H.X.; Winer, D.A.; Clemente-Casares, X.; Tsai, S. Mechanosensing in macrophages and dendritic cells in steady-state and disease. Frontiers in Cell and Developmental Biology 2022, 10. [Google Scholar] [CrossRef]
- Du, H.; Bartleson, J.M.; Butenko, S.; Alonso, V.; Liu, W.F.; Winer, D.A.; Butte, M.J. Tuning immunity through tissue mechanotransduction. Nature Reviews Immunology 2023, 23, 174–188. [Google Scholar] [CrossRef]
- Mammoto, A.; Mammoto, T.; Ingber, D.E. Mechanosensitive mechanisms in transcriptional regulation. J Cell Sci 2012, 125, 3061–3073. [Google Scholar] [CrossRef]
- Dupont, S.; Morsut, L.; Aragona, M.; Enzo, E.; Giulitti, S.; Cordenonsi, M.; Zanconato, F.; Le Digabel, J.; Forcato, M.; Bicciato, S.; et al. Role of yap/taz in mechanotransduction. Nature 2011, 474, 179–183. [Google Scholar] [CrossRef]
- Hansen, C.G.; Moroishi, T.; Guan, K.L. Yap and taz: A nexus for hippo signaling and beyond. Trends Cell Biol 2015, 25, 499–513. [Google Scholar] [CrossRef]
- Meli, V.S.; Veerasubramanian, P.K.; Downing, T.L.; Wang, W.; Liu, W.F. Mechanosensation to inflammation: Roles for yap/taz in innate immune cells. Sci Signal 2023, 16, eadc9656. [Google Scholar] [CrossRef]
- Liu, F.; Lagares, D.; Choi, K.M.; Stopfer, L.; Marinković, A.; Vrbanac, V.; Probst, C.K.; Hiemer, S.E.; Sisson, T.H.; Horowitz, J.C.; et al. Mechanosignaling through yap and taz drives fibroblast activation and fibrosis. Am J Physiol Lung Cell Mol Physiol 2015, 308, L344–357. [Google Scholar] [CrossRef]
- Piersma, B.; Bank, R.A.; Boersema, M. Signaling in fibrosis: Tgf-β, wnt, and yap/taz converge. Front Med (Lausanne) 2015, 2, 59. [Google Scholar] [CrossRef]
- Qin, Z.; He, T.; Guo, C.; Quan, T. Age-related downregulation of ccn2 is regulated by cell size in a yap/taz-dependent manner in human dermal fibroblasts: Impact on dermal aging. JID Innov 2022, 2, 100111. [Google Scholar] [CrossRef]
- Chu, C.-Q.; Quan, T. Fibroblast yap/taz signaling in extracellular matrix homeostasis and tissue fibrosis. J Clin Med 2024, 13, 3358. [Google Scholar]
- Pirri, C. Piezo channels in mechano-inflammation: Gatekeepers of neuroimmune crosstalk. Diseases 2025, 13. [Google Scholar] [CrossRef]
- Gao, W.N.; Hasan, H.; Anderson, D.E.; Lee, W.S. The role of mechanically-activated ion channels piezo1, piezo2, and trpv4 in chondrocyte mechanotransduction and mechano-therapeutics for osteoarthritis. Frontiers in Cell and Developmental Biology 2022, 10. [Google Scholar] [CrossRef] [PubMed]
- Dogra, C.; Changotra, H.; Wergedal, J.E.; Kumar, A. Regulation of phosphatidylinositol 3-kinase (pi3k)/akt and nuclear factor-kappa b signaling pathways in dystrophin-deficient skeletal muscle in response to mechanical stretch. J Cell Physiol 2006, 208, 575–585. [Google Scholar] [CrossRef] [PubMed]
- Tu, P.C.; Pan, Y.L.; Liang, Z.Q.; Yang, G.L.; Wu, C.J.; Zeng, L.; Wang, L.N.; Sun, J.; Liu, M.M.; Yuan, Y.F.; et al. Mechanical stretch promotes macrophage polarization and inflammation via the rhoa-rock/nf-κb pathway. Biomed Res Int 2022, 2022, 6871269. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kim, S.K.; Park, H.; Lee, Y.J.; Park, S.H.; Lee, K.J.; Lee, D.G.; Kang, H.; Kim, J.E. Contribution of autophagy-notch1-mediated nlrp3 inflammasome activation to chronic inflammation and fibrosis in keloid fibroblasts. Int J Mol Sci 2020, 21. [Google Scholar] [CrossRef]
- Ershaid, N.; Sharon, Y.; Doron, H.; Raz, Y.; Shani, O.; Cohen, N.; Monteran, L.; Leider-Trejo, L.; Ben-Shmuel, A.; Yassin, M.; et al. Nlrp3 inflammasome in fibroblasts links tissue damage with inflammation in breast cancer progression and metastasis. Nat Commun 2019, 10, 4375. [Google Scholar] [CrossRef] [PubMed]
- Artlett, C.M.; Sassi-Gaha, S.; Rieger, J.L.; Boesteanu, A.C.; Feghali-Bostwick, C.A.; Katsikis, P.D. The inflammasome activating caspase 1 mediates fibrosis and myofibroblast differentiation in systemic sclerosis. Arthritis Rheum 2011, 63, 3563–3574. [Google Scholar] [CrossRef]
- Weinberg, J.B. Nitric oxide synthase 2 and cyclooxygenase 2 interactions in inflammation. Immunol Res 2000, 22, 319–341. [Google Scholar] [CrossRef] [PubMed]
- Seed, M.P.; Willoughby, D.A. Cox-2, ho no! Cyclooxygenase-2, heme oxygenase and nitric oxide synthase: Their role and interactions in inflammation. Inflamm Res 1996, 46, 279–281. [Google Scholar] [CrossRef]
- Wang, B.; Han, J.; Elisseeff, J.H.; Demaria, M. The senescence-associated secretory phenotype and its physiological and pathological implications. Nat Rev Mol Cell Bio 2024, 25, 958–978. [Google Scholar] [CrossRef]
- Coppé, J.P.; Desprez, P.Y.; Krtolica, A.; Campisi, J. The senescence-associated secretory phenotype: The dark side of tumor suppression. Annu Rev Pathol 2010, 5, 99–118. [Google Scholar] [CrossRef]
- Nguyen, T.Q.T.; Cho, K.A. Targeting immunosenescence and inflammaging: Advancing longevity research. Experimental & Molecular Medicine 2025, 57, 1881–1892. [Google Scholar] [CrossRef]
- Mavrogonatou, E.; Papadopoulou, A.; Pratsinis, H.; Kletsas, D. Senescence-associated alterations in the extracellular matrix: Deciphering their role in the regulation of cellular function. Am J Physiol-Cell Ph 2023, 325, C633–C647. [Google Scholar] [CrossRef]
- Xiang, Y.; Qin, Z.; Yang, Y.; Fisher, G.J.; Quan, T. Age-related elevation of hgf is driven by the reduction of fibroblast size in a yap/taz/ccn2 axis-dependent manner. J Dermatol Sci 2021, 102, 36–46. [Google Scholar] [CrossRef]
- Chu, C.Q.; Quan, T.H. Fibroblast yap/taz signaling in extracellular matrix homeostasis and tissue fibrosis. J Clin Med 2024, 13. [Google Scholar] [CrossRef] [PubMed]
- Karsdal, M.; Cox, T.R.; Parker, A.L.; Willumsen, N.; Sand, J.M.B.; Jenkins, G.; Hansen, H.H.; Oldenburger, A.; Geillinger-Kaestle, K.E.; Larsen, A.T.; et al. Advances in extracellular matrix-associated diagnostics and therapeutics. J Clin Med 2025, 14. [Google Scholar] [CrossRef]
- Pang, X.; He, X.; Qiu, Z.; Zhang, H.; Xie, R.; Liu, Z.; Gu, Y.; Zhao, N.; Xiang, Q.; Cui, Y. Targeting integrin pathways: Mechanisms and advances in therapy. Signal Transduct Target Ther 2023, 8, 1. [Google Scholar] [CrossRef]
- Ley, K.; Rivera-Nieves, J.; Sandborn, W.J.; Shattil, S. Integrin-based therapeutics: Biological basis, clinical use and new drugs. Nat Rev Drug Discov 2016, 15, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Selewski, D.T.; Shah, G.V.; Segal, B.M.; Rajdev, P.A.; Mukherji, S.K. Natalizumab (tysabri). AJNR Am J Neuroradiol 2010, 31, 1588–1590. [Google Scholar] [CrossRef] [PubMed]
- Khoy, K.; Mariotte, D.; Defer, G.; Petit, G.; Toutirais, O.; Le Mauff, B. Natalizumab in multiple sclerosis treatment: From biological effects to immune monitoring. Front Immunol 2020, 11, 549842. [Google Scholar] [CrossRef]
- Klotz, L.; Berger, T.; Brownlee, W.J.; Chan, A.; Lycke, J.; Oreja-Guevara, C.; Palavra, F.; Sacca, F.; Sejbaek, T.; Weber, M.S.; et al. Twenty years of natalizumab in multiple sclerosis: Lessons learned and future outlook. Ther Adv Neurol Diso 2025, 18. [Google Scholar] [CrossRef]
- Lebwohl, M.; Tyring, S.K.; Hamilton, T.K.; Toth, D.; Glazer, S.; Tawfik, N.H.; Walicke, P.; Dummer, W.; Wang, X.; Garovoy, M.R.; et al. A novel targeted t-cell modulator, efalizumab, for plaque psoriasis. New Engl J Med 2003, 349, 2004–2013. [Google Scholar] [CrossRef] [PubMed]
- Slack, R.J.; Macdonald, S.J.F.; Roper, J.A.; Jenkins, R.G.; Hatley, R.J.D. Emerging therapeutic opportunities for integrin inhibitors. Nat Rev Drug Discov 2022, 21, 60–78. [Google Scholar] [CrossRef] [PubMed]
- O'Reilly, P.J.; Gaggar, A.; Blalock, J.E. Interfering with extracellular matrix degradation to blunt inflammation. Curr Opin Pharmacol 2008, 8, 242–248. [Google Scholar] [CrossRef]
- Closset, L.; Gultekin, O.; Salehi, S.; Sarhan, D.; Lehti, K.; Gonzalez-Molina, J. The extracellular matrix-immune microenvironment crosstalk in cancer therapy: Challenges and opportunities. Matrix Biology 2023, 121, 217–228. [Google Scholar] [CrossRef]
- Assunção, M.; Dehghan-Baniani, D.; Yiu, C.H.K.; Später, T.; Beyer, S.; Blocki, A. Cell-derived extracellular matrix for tissue engineering and regenerative medicine. Front Bioeng Biotechnol 2020, 8, 602009. [Google Scholar] [CrossRef]
- Qu, F.; Guilak, F.; Mauck, R.L. Cell migration: Implications for repair and regeneration in joint disease. Nature Reviews Rheumatology 2019, 15, 167–179. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.N.; Zeng, X.Y.; Deng, X.K.; Yang, F.L.; Ma, X.Y.; Gao, W. Smart biomaterials: As active immune modulators to shape pro-regenerative microenvironments. Frontiers in Cell and Developmental Biology 2025, 13. [Google Scholar] [CrossRef]
- Coser, C.; Ghaemmaghami, A.M.; Yang, J. Soft tissue-mimicking hydrogel stiffness modulates polarisation of human monocyte-derived macrophages. Biomater Sci-Uk 2025, 13, 6637–6651. [Google Scholar] [CrossRef] [PubMed]
- Butenko, S.; Nagalla, R.R.; Guerrero-Juarez, C.F.; Palomba, F.; David, L.-M.; Nguyen, R.Q.; Gay, D.; Almet, A.A.; Digman, M.A.; Nie, Q.; et al. Hydrogel crosslinking modulates macrophages, fibroblasts, and their communication, during wound healing. Nature Communications 2024, 15, 6820. [Google Scholar] [CrossRef] [PubMed]
- Luo, T.; Tan, B.; Zhu, L.; Wang, Y.; Liao, J. A review on the design of hydrogels with different stiffness and their effects on tissue repair. Front Bioeng Biotechnol 2022, 10, 817391. [Google Scholar] [CrossRef]
- Da, L.C.; Huang, Y.Z.; Xie, H.Q.; Zheng, B.H.; Huang, Y.C.; Du, S.R. Membranous extracellular matrix-based scaffolds for skin wound healing. Pharmaceutics 2021, 13. [Google Scholar] [CrossRef]
- Romero, D.J.; Hussey, G.; Capella-Monsonís, H. Immune response to extracellular matrix bioscaffolds: A comprehensive review. Biologics 2025, 5, 28. [Google Scholar] [CrossRef]
- Hsu, C.-H.; Chen, J.; Lee, K.-J.; Donahue, L.R.; Kacaj, D.; McDonough, S.P.; White, A.C. Therapy-induced ecm remodeling creates a transient immune barrier in residual melanoma. Advanced Science 2025, 12, e08451. [Google Scholar] [CrossRef]
- Perryman, L.; Findlay, A.; Baskar, J.; Charlton, B.; Foot, J.; Hamilton, R.; Hamprecht, D.; Joshi, A.; Stolp, J.; Turner, C.; et al. The small molecule loxl2 inhibitor snt-5382 reduces cardiac fibrosis and achieves strong clinical target engagement. Sci Rep-Uk 2025, 15, 22653. [Google Scholar] [CrossRef]
- Mascharak, S.; Guo, J.L.; Griffin, M.; Berry, C.E.; Wan, D.C.; Longaker, M.T. Modelling and targeting mechanical forces in organ fibrosis. Nat Rev Bioeng 2024, 2, 305–323. [Google Scholar]
- Abyaneh, H.S.; Regenold, M.; McKee, T.D.; Allen, C.; Gauthier, M.A. Towards extracellular matrix normalization for improved treatment of solid tumors. Theranostics 2020, 10, 1960–1980. [Google Scholar] [CrossRef]
- Najafi, M.; Farhood, B.; Mortezaee, K. Extracellular matrix (ecm) stiffness and degradation as cancer drivers. J Cell Biochem 2019, 120, 2782–2790. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.; Lucas, M.C.; Leonte, L.E.; Garcia-Montolio, M.; Singh, L.B.; Findlay, A.D.; Deodhar, M.; Foot, J.S.; Jarolimek, W.; Timpson, P.; et al. Pre-clinical evaluation of small molecule loxl2 inhibitors in breast cancer. Oncotarget 2017, 8, 26066–26078. [Google Scholar] [CrossRef]
- Burchard, P.R.; Ruffolo, L.I.; Ullman, N.A.; Dale, B.S.; Dave, Y.A.; Hilty, B.K.; Ye, J.; Georger, M.; Jewell, R.; Miller, C.; et al. Pan-lysyl oxidase inhibition disrupts fibroinflammatory tumor stroma, rendering cholangiocarcinoma susceptible to chemotherapy. Hepatology Communications 2024, 8, e0502. [Google Scholar] [CrossRef]
- Badarau, E.; Wang, Z.; Rathbone, D.L.; Costanzi, A.; Thibault, T.; Murdoch, C.E.; El Alaoui, S.; Bartkeviciute, M.; Griffin, M. Development of potent and selective tissue transglutaminase inhibitors: Their effect on tg2 function and application in pathological conditions. Chem Biol 2015, 22, 1347–1361. [Google Scholar] [PubMed]
- Vandenbroucke, R.E.; Libert, C. Is there new hope for therapeutic matrix metalloproteinase inhibition? Nature Reviews Drug Discovery 2014, 13, 904–927. [Google Scholar] [CrossRef] [PubMed]
- Fingleton, B. Mmps as therapeutic targets--still a viable option? Semin Cell Dev Biol 2008, 19, 61–68. [Google Scholar] [CrossRef]
- Winer, A.; Adams, S.; Mignatti, P. Matrix metalloproteinase inhibitors in cancer therapy: Turning past failures into future successes. Molecular Cancer Therapeutics 2018, 17, 1147–1155. [Google Scholar] [CrossRef]
- Coates-Park, S.; Rich, J.A.; Stetler-Stevenson, W.G.; Peeney, D. The timp protein family: Diverse roles in pathophysiology. Am J Physiol-Cell Ph 2024, 326, C917–C934. [Google Scholar]
- Leask, A. Ccn1: A saspy protein that plays multifaceted roles in fibrogenesis. Matrix Biology 2026, 143, 14–17. [Google Scholar] [CrossRef]
- Feng, D.; Gerarduzzi, C. Emerging roles of matricellular proteins in systemic sclerosis. International Journal of Molecular Sciences 2020, 21, 4776. [Google Scholar] [CrossRef] [PubMed]
- Quan, T.; Johnston, A.; Gudjonsson, J.E.; Fisher, G.J. Cyr61/ccn1: A novel mediator of epidermal hyperplasia and inflammation in psoriasis? J Invest Dermatol 2015, 135, 2562–2564. [Google Scholar] [CrossRef]
- Kasprzycka, M.; Hammarström, C.; Haraldsen, G. Tenascins in fibrotic disorders-from bench to bedside. Cell Adhes Migr 2015, 9, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Frenzel, D.F.; Borkner, L.; Scheurmann, J.; Singh, K.; Scharffetter-Kochanek, K.; Weiss, J.M. Osteopontin deficiency affects imiquimod-induced psoriasis-like murine skin inflammation and lymphocyte distribution in skin, draining lymph nodes and spleen. Exp Dermatol 2015, 24, 305–307. [Google Scholar] [CrossRef]
- Sirois, J.P.; Heinz, A. Matrikines in the skin: Origin, effects, and therapeutic potential. Pharmacol Therapeut 2024, 260. [Google Scholar] [CrossRef]
- Tolg, C.; Telmer, P.; Turley, E. Specific sizes of hyaluronan oligosaccharides stimulate fibroblast migration and excisional wound repair. PLoS One 2014, 9, e88479. [Google Scholar] [CrossRef]
- Baghy, K.; Szakadáti, H.; Kovalszky, I. Decorin the antifibrotic proteoglycan and its progression in therapy. Am J Physiol Cell Physiol 2025, 328, C1853–c1865. [Google Scholar] [CrossRef]
- Avenoso, A.; Bruschetta, G.; D'Ascola, A.; Scuruchi, M.; Mandraffino, G.; Gullace, R.; Saitta, A.; Campo, S.; Campo, G.M. Hyaluronan fragments produced during tissue injury: A signal amplifying the inflammatory response. Arch Biochem Biophys 2019, 663, 228–238. [Google Scholar] [CrossRef]
- Patel, D.F.; Snelgrove, R.J. The multifaceted roles of the matrikine pro-gly-pro in pulmonary health and disease. Eur Respir Rev 2018, 27. [Google Scholar] [CrossRef] [PubMed]
- Jha, A.; Moore, E. Yigsr, a laminin-derived peptide, dictates a concentration-dependent impact on macrophage phenotype response. Cell Mol Bioeng 2024, 17, 423–440. [Google Scholar] [CrossRef] [PubMed]
- Thau, H.; Gerjol, B.P.; Hahn, K.; von Gudenberg, R.W.; Knoedler, L.; Stallcup, K.; Emmert, M.Y.; Buhl, T.; Wyles, S.P.; Tchkonia, T.; et al. Senescence as a molecular target in skin aging and disease. Ageing Research Reviews 2025, 105. [Google Scholar] [CrossRef] [PubMed]
- Saliev, T.; Singh, P.B. Targeting senescence: A review of senolytics and senomorphics in anti-aging interventions. Biomolecules 2025, 15. [Google Scholar] [CrossRef]
- Kirkland, J.L.; Tchkonia, T. Senolytic drugs: From discovery to translation. Journal of Internal Medicine 2020, 288, 518–536. [Google Scholar] [CrossRef]
- Zhang, L.; Pitcher, L.E.; Prahalad, V.; Niedernhofer, L.J.; Robbins, P.D. Targeting cellular senescence with senotherapeutics: Senolytics and senomorphics. The FEBS Journal 2023, 290, 1362–1383. [Google Scholar] [CrossRef]
- Gerdes, E.O.W.; Misra, A.; Netto, J.M.E.; Tchkonia, T.; Kirkland, J.L. Strategies for late phase preclinical and early clinical trials of senolytics. Mechanisms of Ageing and Development 2021, 200. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



