1. Introduction
Recuperative heat transfer is one of the key elements influencing the performance of mixed-refrigerant Joule-Thomson (MR JT) cryocoolers, used to obtain low temperatures in the cryogenic range and often deployed due to their simplicity, which results in high reliability. Therefore, in order to avoid undesirable heat transfer scenarios, such as the presence of pinch points, an accurate and robust modeling framework utilizing single- and two-phase heat transfer correlations needs to be developed. Additionally, modeling results must be validated with trustful experimental data, especially in terms of mixed-refrigerant composition.
Due to their low cost, high reliability, wide capacity range and suitable operational range, brazed plate heat exchangers (BPHE) have become widely used in MR JT cryocoolers [
1,
2]. Effective recuperation usually requires the application of a few BPHE units in series [
1,
2], which provides sufficient thermal length. However, it can trigger issues related to two-phase flow, especially at the heat exchanger inlet ports. Currently, data on heat transfer effectiveness during recuperation in MR JT cryocoolers using BPHE is limited, especially for systems with precooling [
1,
3]. Precooled MR JT cryocoolers have been reported to be more efficient than single-stage MR JT systems, but also more demanding in terms of heat transfer effectiveness in the recuperation section [
4].
The authors of [
2] realized an experimental survey on the recuperation heat transfer performance of middle BPHE (second out of 3 BPHEs connected in series) and validated it against developed model. The LMTD-based model takes into account numerous boiling and condensation heat transfer correlations specifically for pure refrigerants. In total, 40 correlation combinations were tested, of which the authors recommended two which agree +- 30% with the experimental data. The error in the composition of the circulating MR is reported as +-4.3%, but it is not clear how it affects the validation procedure. It is also not clear how often and when exactly the circulating MR composition was measured. This information is crucial as it was recently shown by [
5] that the composition of circulating MR varies significantly with thermal load or the progress of the cooldown process.
The modeling of complete recuperation heat transfer process for tube-in-tube heat exchanger is presented in[
6]. The authors did not consider different boiling and condensation correlations. The modeling results were validated against the experimental data; however, it is reported that the circulating MR composition was not always measured, and it was assumed to be the same as the charged one. The measurement error of the MR composition is not reported.
While [
2,
6] has investigated the MR of hydrocarbons, the work of [
7] is focused on the argon-freon MRs. The recuperator was a customized microchannel heat exchanger (MCHE). The application of MCHE allows one to minimize the MR charge, which, in general, is desired but makes the system sensitive to probing [
5]. The authors validated the LMTD-based model against the experimental data. Gas chromatography was used to measure the MR composition; however, it is stated that it was used to confirm charged compositions, which will likely differ from the circulating one. The authors examined 8 condensation and 8 evaporation heat transfer correlations .
Tests in the section incorporated into the dedicated test stand, including the GM cryocooler, have been performed in [
8]. Such an approach enabled to precisely simulate the conditions of MR in the test section but, on the other hand, limited the measurement to flow boiling phenomena. The authors compared experimental data with two single-phase correlations that reported significant deviations for qualities ranging from 0.4 to 0.8. The study focused on MRs consisting of nitrogen and hydrocarbons. Circulating MR composition was frequently measured via gas chromatography.
Experimental tests aimed at obtaining the thermodynamic and hydraulic behavior of the tube-in-tube heat exchanger have been performed in [
9]. The authors measured the composition of the MR, consisting of R14, methane, ethane, propane, isobutane, isopentane, nitrogen and neon, as well as temperatures and pressures along the HX. The heat transfer characteristics of the heat exchanger were concluded to be very sensitive to the mixture used, which means that circulating MR composition measurement is crucial for reliable modeling of this heat transfer case. The works of [
10,
11] led to very similar conclusions for tube-in-tube HX and MR consisting of nitrogen and hydrocarbons.
The most questionable point of most studies is the measurement and later consideration of circulating MR composition. The available studies do not address the topic of the exact measurement moment, which according to recent findings from [
5], significantly affects the measured circulating MR composition. Secondly, most studies report measurement errors related to MR composition, but without considering the impact of this issue on the results, even if a significant impact of MR composition was concluded [
9].
Successful validation of the modeling results requires complete information concerning heat transfer conditions, including the circulating composition of the mixed refrigerant. According to numerous works, it cannot be simply determined based on the charge composition of the mixed refrigerant [
3,
9,
11,
12,
13,
14,
15] and will depend greatly on the specific operational conditions and architecture of the system. Credible validation would require many measurements of the MR composition during the test and in case of application of the invasive method (requiring probing of the MR sample for further investigation ) will affect the remaining MR composition [
5]. To solve this issue, the following study employed a novel noninvasive method to measure MR composition that is described in detail in [
5]. Moreover, it was investigated how uncertainties of the MR composition measurement affect the credibility of results, which was not performed in any of the studies cited above.
In this work, a series of measurements were performed on MR JT cryocooler, using binary mixtures of methane and ethane in various concentrations. Inlet temperatures for the low pressure and high pressure streams, obtained in recuperation section measurements of the system, were used as input to the proposed numerical model, which utilizes the e-NTU method and produces a temperature distribution along the heat exchanger length as an output. A wide variety of single- and two-phase correlations for ideal heat transfer were investigated. Selected correlations were also used with available correction factors taken into account to further improve computational accuracy. All results were compared with the experimental data and evaluated.
4. Numerical Model
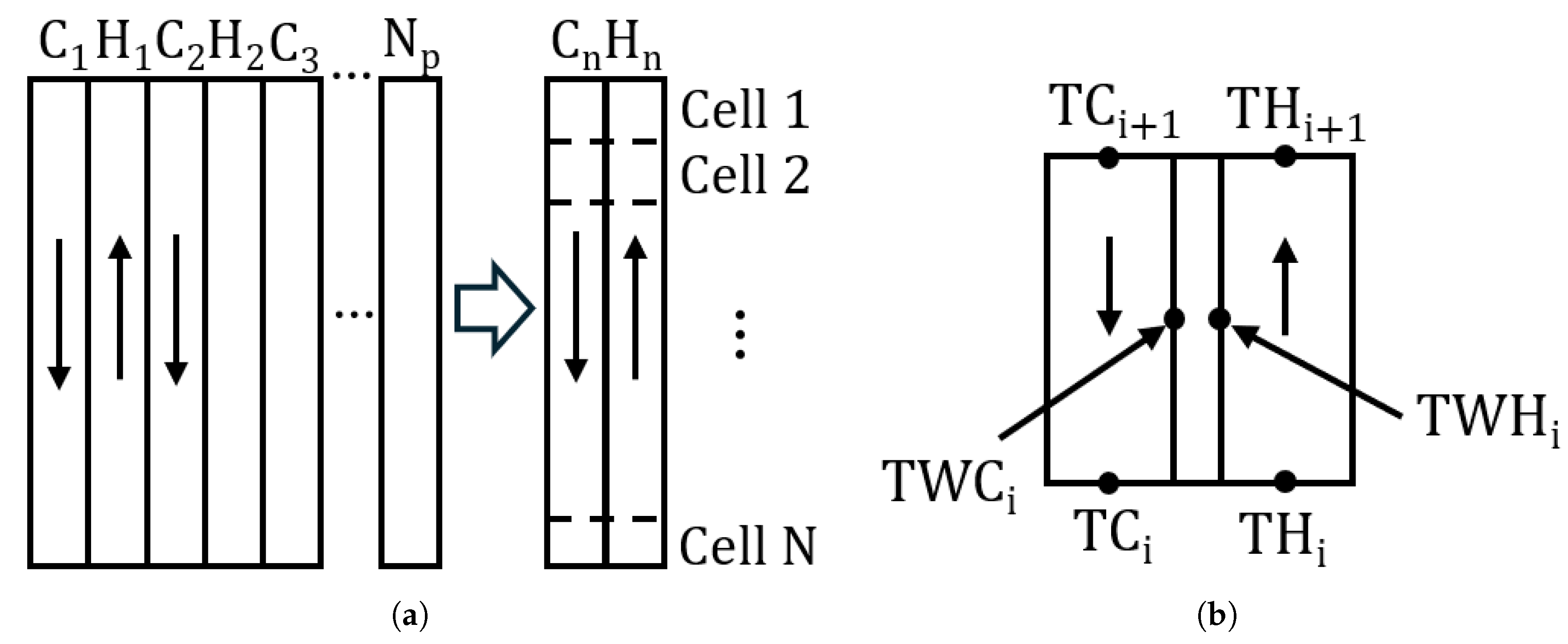
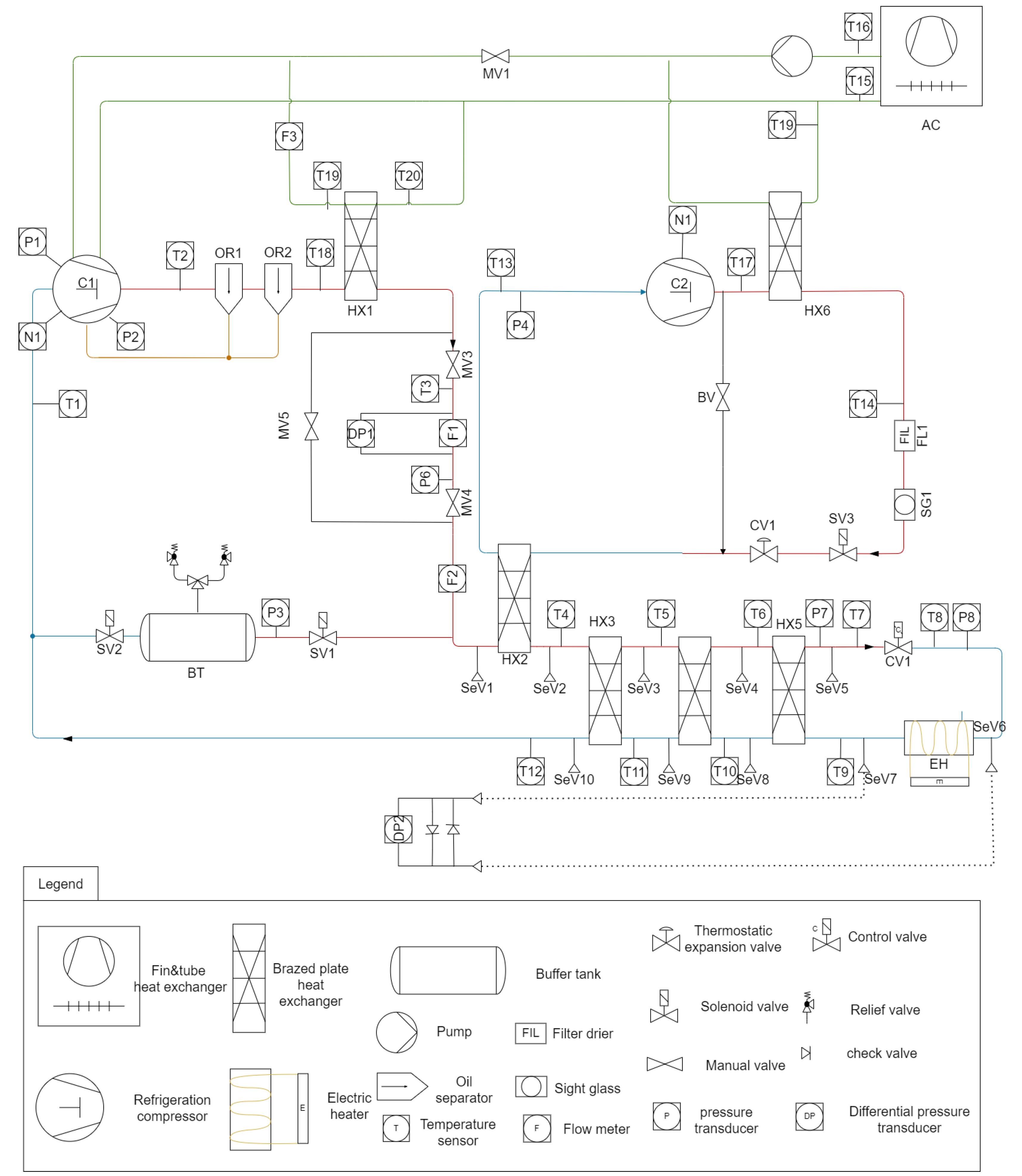
A steady-state numerical model was formulated to predict the outlet temperatures in a brazed plate heat exchanger assembly when all inlet conditions are known. Three plate heat exchangers operating in series were represented as a single equivalent HX. Within each HX, the physical stack of plates was simplified to one plate channel whose heat transfer area is equal to the total exchange surface. The model does not take the pressure drop along the HXs into account.
Figure 4.
Numerical model - discretization scheme. (a) HX geometry simplification (b) Temperatures used by the model (one cell).
Figure 4.
Numerical model - discretization scheme. (a) HX geometry simplification (b) Temperatures used by the model (one cell).
The equivalent surface of HX was divided along the length of the HX into N computational elements (cells) of equal area, for . An initial guess for the temperature distribution of both streams was provided. The number of cells in this study is set to after an investigation of mesh sensitivity has been performed. The difference in total power of the heat exchanger Q between 400 and 800 cells was, on average, 1%. The initial temperature distribution was assumed to be linear, with the difference between the inlet and outlet for both channels set at 5 K. The outlet of element i served as the inlet of element .
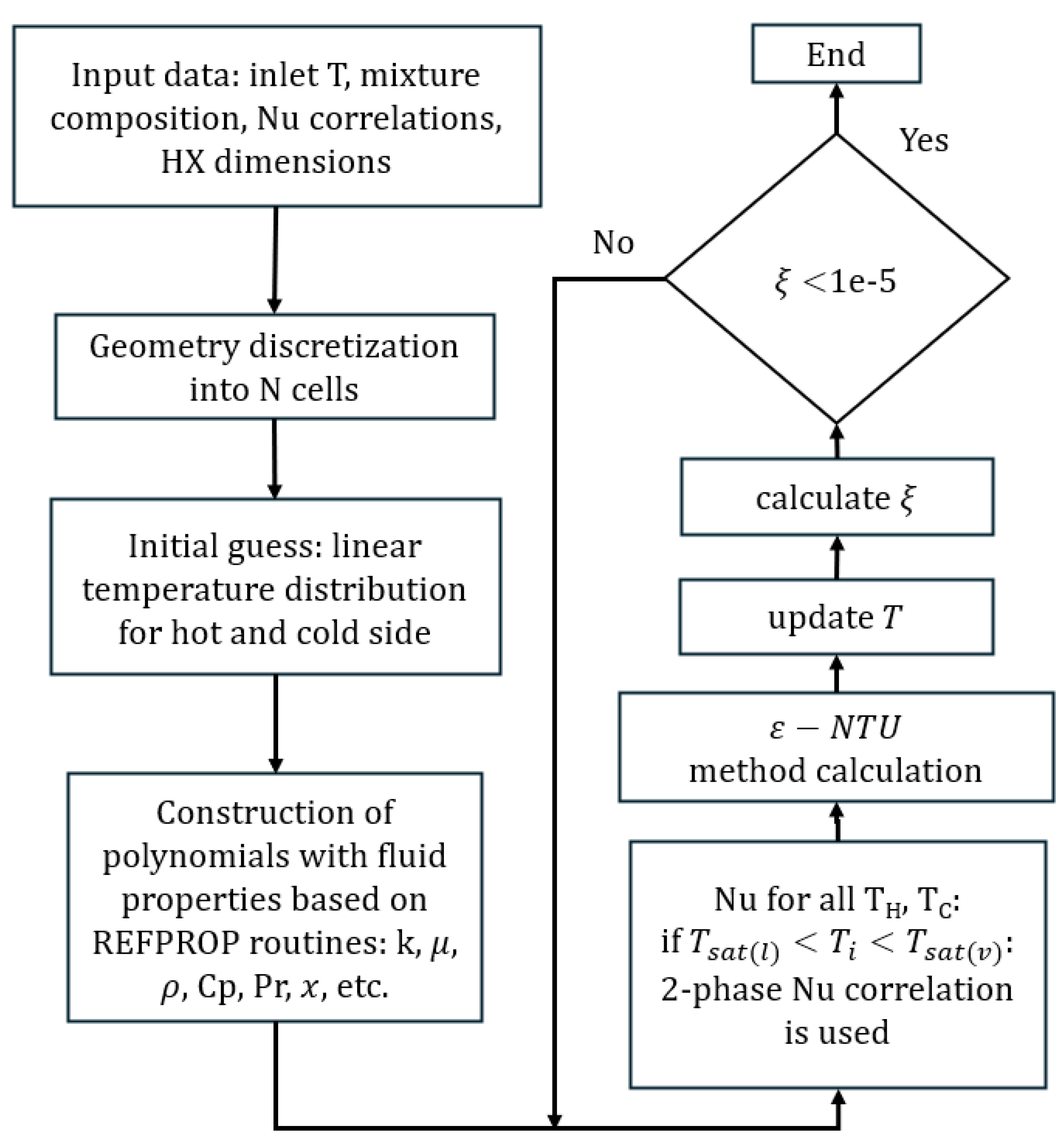
The block diagram of the numerical model is shown in
Figure 5. After the initial conditions were defined and heat transfer correlations selected for a particular case, the local properties of the working fluid mixtures were obtained from REFPROP 10 [
16]. For each element
i, quantities such as heat capacity
, density
, viscosity
, and thermal conductivity
k were obtained for both hot and cold streams in the cells. The properties in one element are calculated for the inlet of the cell. In the case of the two-phase regime, vapor quality is used to obtain estimated properties using Eq.
1. Heat transfer was computed for each cell using the effectiveness-NTU method. Heat capacity rates were formed for both streams (Eq.
2), and then the minimum and maximum capacity rates were found, (Eq.
3 and
4). The NTU per element was consequently calculated (Eq.
5), where
denotes the local overall heat transfer coefficient. The effectiveness of the heat exchanger
was then obtained from the
for the BPHE counter-flow arrangement. Lastly, using Eq.
6, temperatures were updated using the energy balance for both streams and all cells (Eq.
7). The iteration error for one cell
was defined as the relative difference between the new (k+1) and old (k) temperature values (Eq.
8). The global error
is expressed as the average error in all cells. The solving process was completed when
, where the tolerance was
.
5. Results and Discussion
Temperature, pressure, and mass flow measurements were performed in the recuperation section of the MR JT cooler while operating in a two-phase regime. Experimental data (input temperatures, pressures, composition and mass flow) were used as input to model the heat transfer in the recuperation section using heat transfer correlation sets. Additionally, calculations for all data points were performed for the range of possible mixture compositions, i.e. percentage points from the measured value, with an increment of one percentage point. In this range, the composition with the lowest error for the considered case is selected and used for further analysis. This is later referred to as "composition correction", not to be confused with the evaporation correction of the heat transfer coefficient.
There were 22 data points from the experiment. Taking into account the 15 possible mixture compositions and 96 correlation combinations in total, this yields a total of 31680 computational cases. Correlations used are presented in
Table 3.
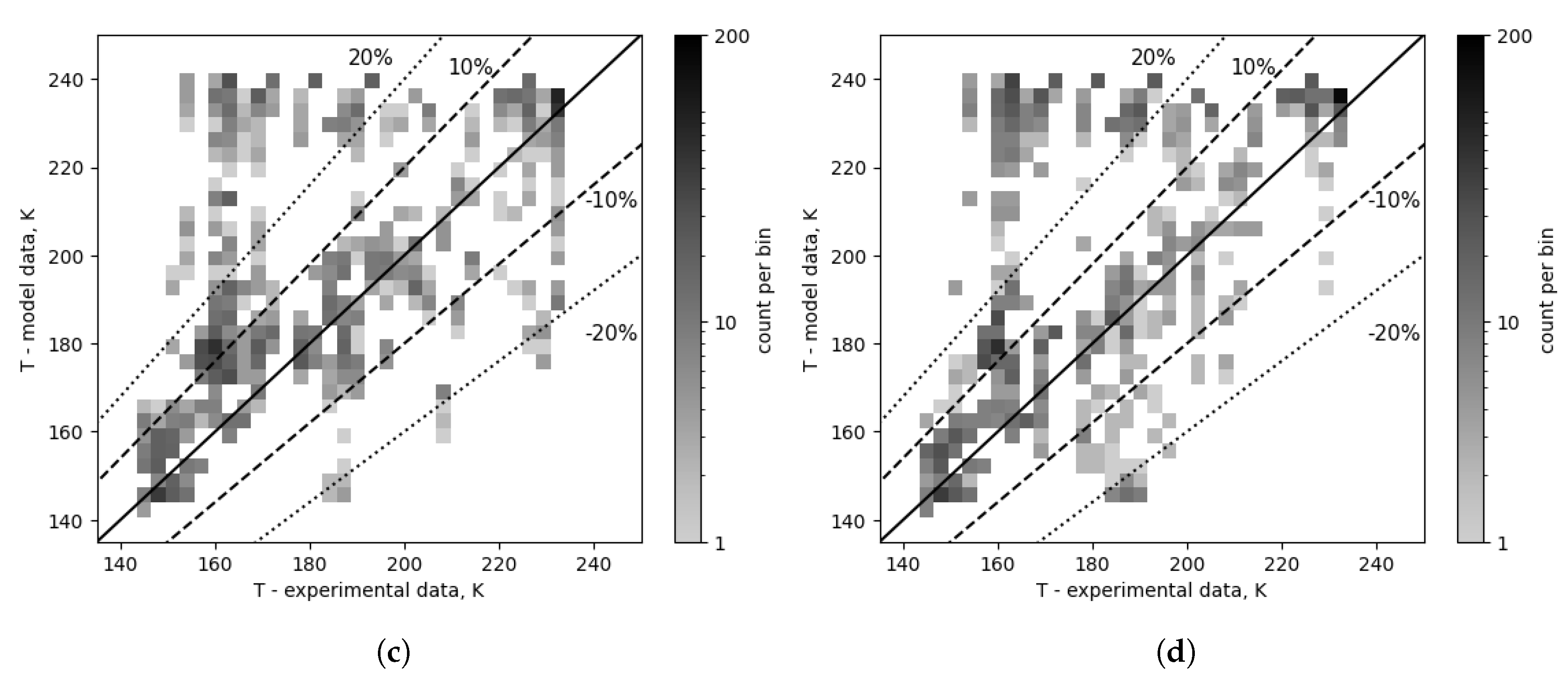
It is important to note that the plots in
Figure 12,
Figure 13,
Figure 14,
Figure 11,
Figure 10 and
Figure 15 have a logarithmic scale of point density to capture both high point concentrations and outliers. All data points have been mapped on the 2D histogram, and the data range has been established to be 135 to 245 K for both axes. This range was then divided into 30 bins, and based on the point count per bin, the density map was created for the data subsets used in small scale comparisons and the overall results.
In this study, the main error definition used is MAE (Mean Absolute Error). Detailed statistical analysis can be found later in this chapter.
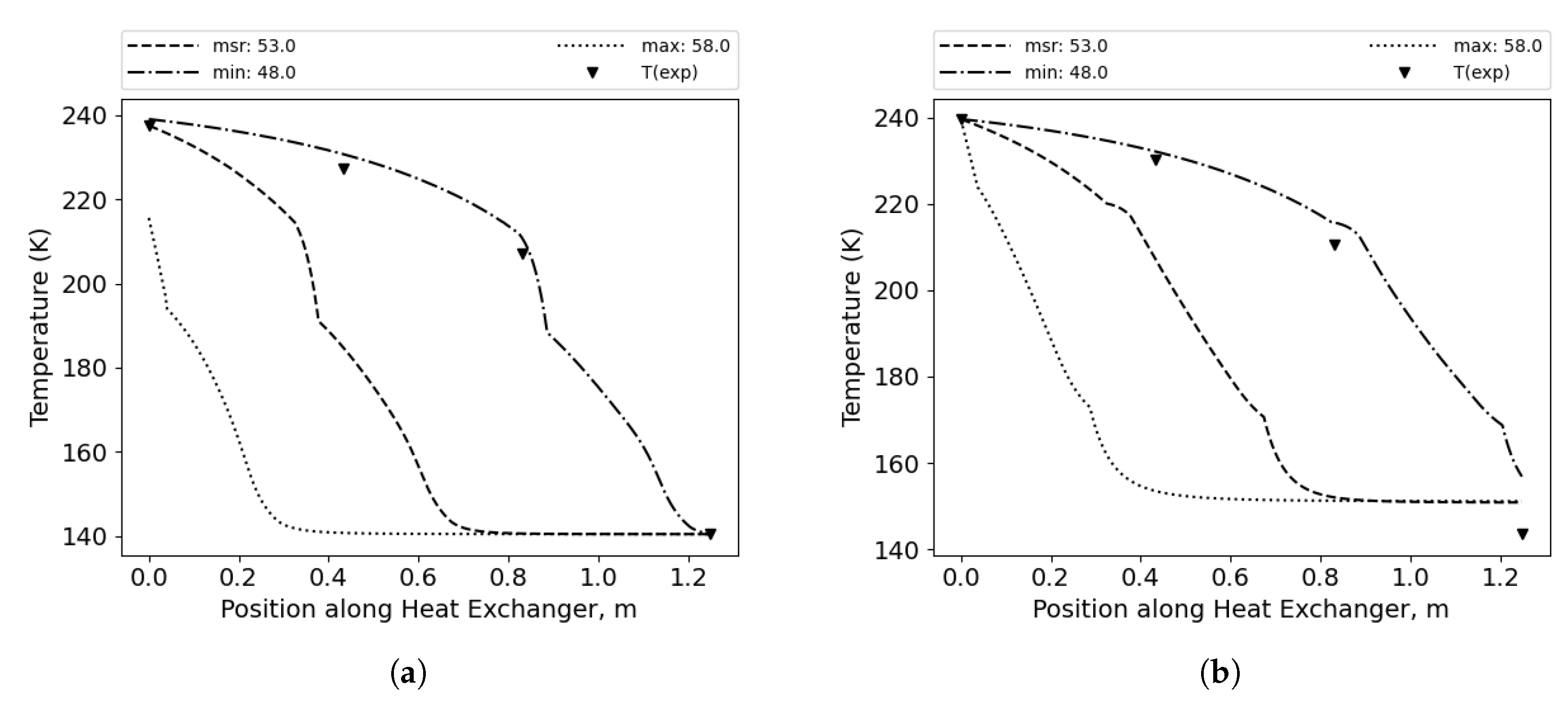
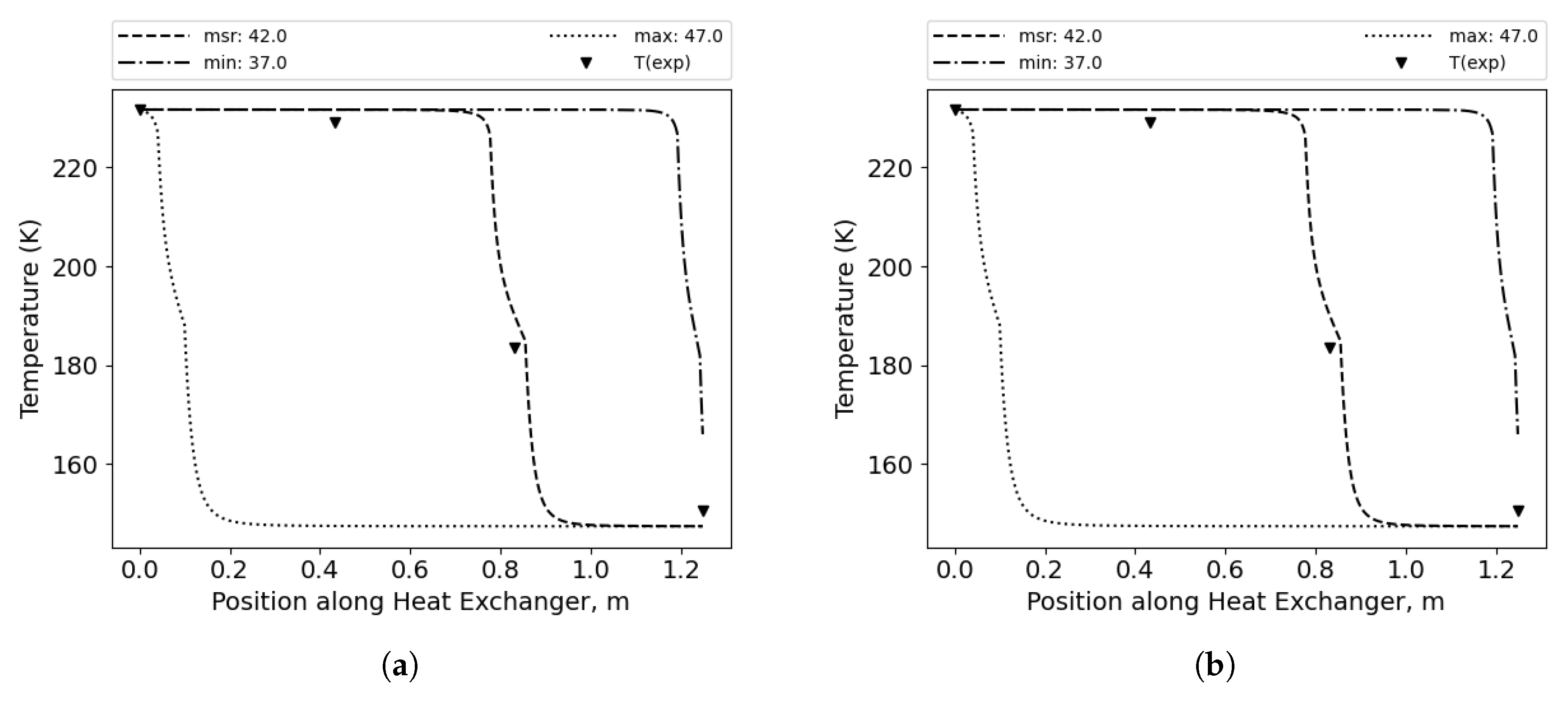
Due to the high number of correlation combinations (96) and the wide range of possible mixture compositions (15 percentage points), the calculated temperature distributions in some considered cases exhibit the lowest error for the measured values, while in other instances, the lowest error is achieved for minimum values, and in some cases the lowest error is achieved for the maximum possible concentration values, as shown in
Figure 7,
Figure 8, and
Figure 9. This observation emphasizes the importance of the discussion regarding the impact of mixture composition on results in similar studies, especially with binary mixtures that differ significantly in boiling points; this effect may not be as pronounced with mixtures that have more components. [
11]
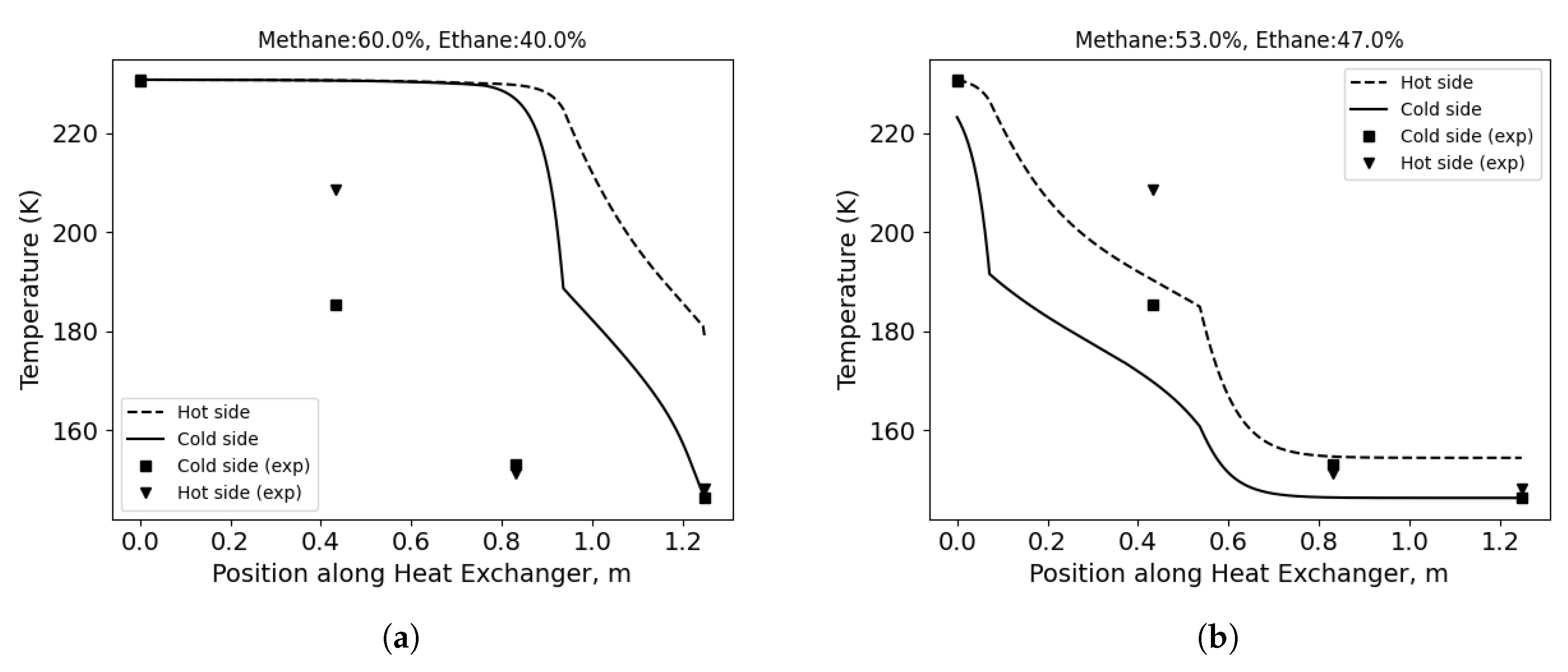
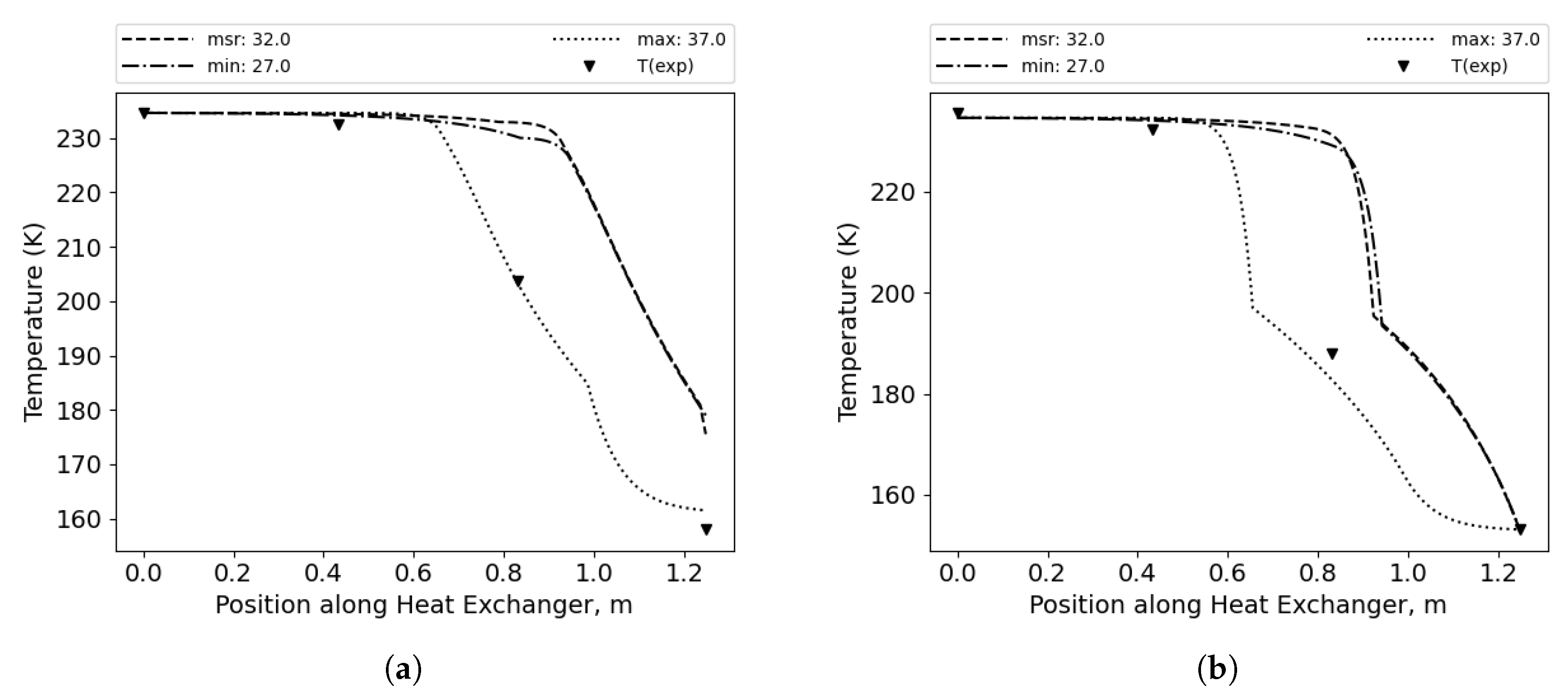
Figure 6 shows the exemplary temperature distributions for the overall best pair of correlations and the effect of composition correction. In
Figure 6a, the results for the measured mixture composition are presented.
Figure 6b shows the temperature distribution for the corrected composition; in this case, the methane concentration is decreased by 7 percentage points. As a result, the error decreased from 57.1 K to 7.64 K.
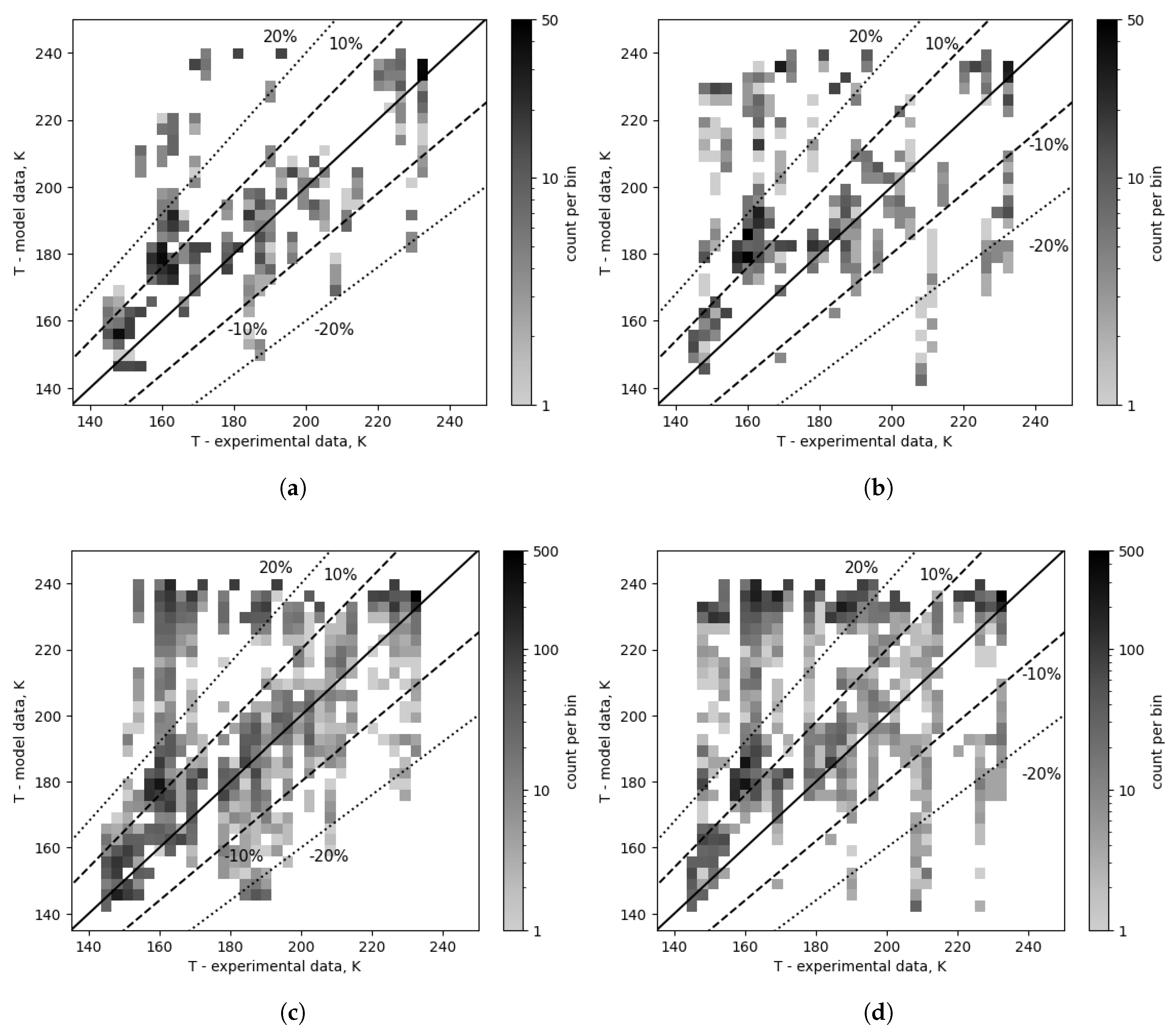
A comparison of the corrected and uncorrected cases is presented in
Figure 10, and their distribution in
Table 4. The majority (73.9%) of all modeling results before composition correction were within a range of ±30% of experimental values, 59.3% in the range of ±20% and 36.3% within ±10% of experimental values. After the composition correction, these values increased to 84.4%, 75.9% and 58.9%, respectively. Overall, the two-phase heat transfer coefficients ranged from 0.13 to 2.90
for evaporation with correction, and 0.585 to 36.1
for evaporation without correction. The condensation heat transfer coefficient values ranged from 0.36 to 8.17
. Single phase heat transfer, depending on the phase, ranged from 1.77 to 8.71
for the liquid phase and from 0.53 to 17.8
for the vapor phase. The combination of correlations with the lowest overall error was Kuo for condensation, Hsieh and Lin for evaporation with the Fujita correction factor, and Wanniarachchi for single phase heat transfer.
A high concentration of points can be observed in the range of model data from 220 to 240 K and from 145 to 190 K for experimental temperatures (referred to as high error band in
Table 5 and
Table 4) visible in
Figure 10c,d. This area is likely the source of high error values in the overall results, since when all correlation combinations are taken into account, the composition correction decreases the percentage of points in this zone from 29.7% of all points to 15.1%. The number of outliers (single points or small concentrations of points with high error values) is also significantly decreased, as the number of points outside the -20% range decreased from 3.6% to 0.38% of data points. The number of points beyond the 20% range decreases from 38.4% to 22.5%. Considering the top 15 combinations temperature map in
Figure 10a,b, composition correction decreased the number of points in the high error band from 13.6% to 2.7% of all data points, reduced the number of points outside the -20% range from 1.85% to 1.55%, and resulted in a decrease in the overall number of points in the ranges of ±30% (84.6% vs 92.5%), ±20% (73.3% vs 88.4%), and ±10% (45.5% vs 66.6%). Compared to the overall results, the percentage of high error points is very small for the top 15, and the distribution is more symmetrical.
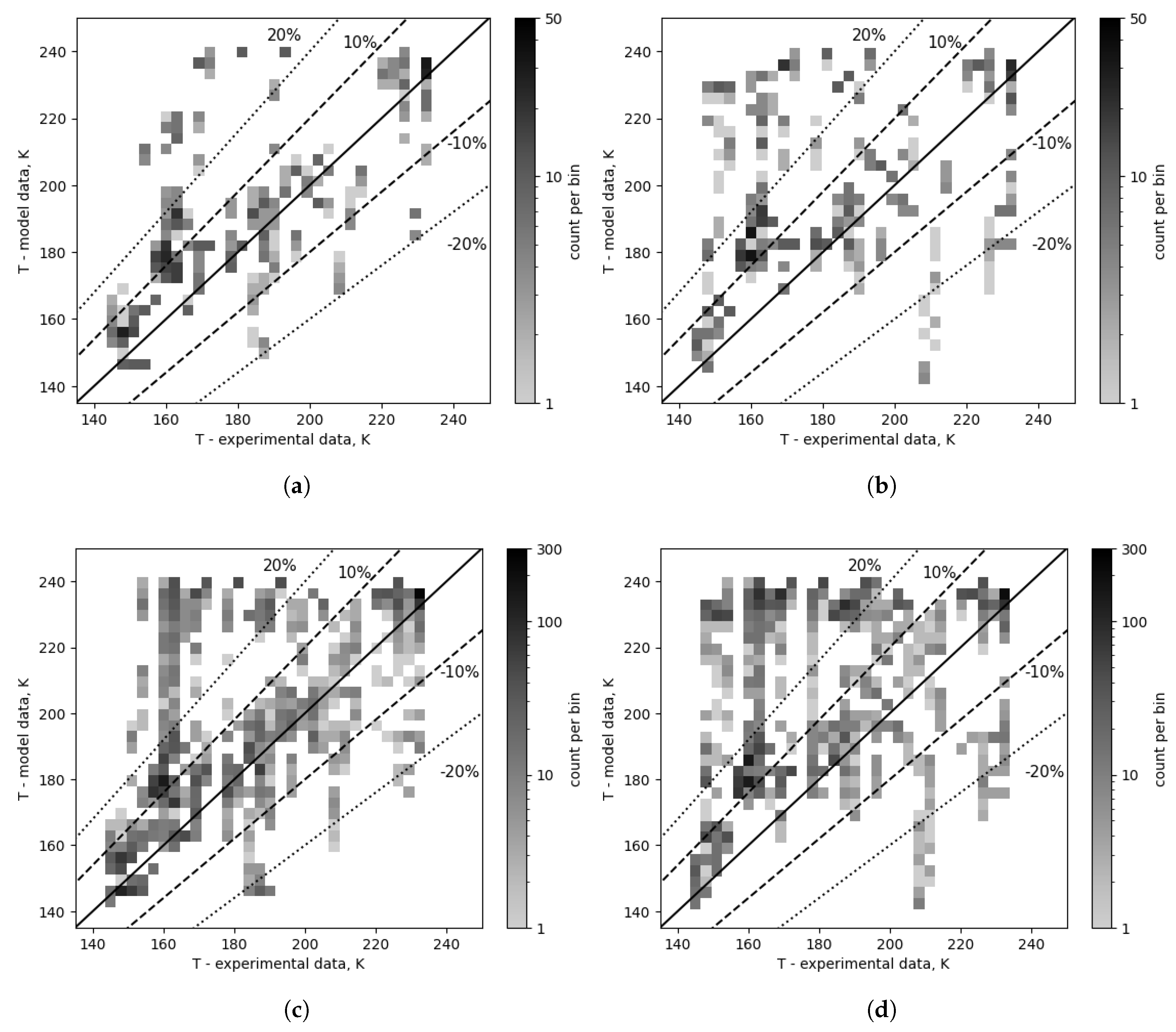
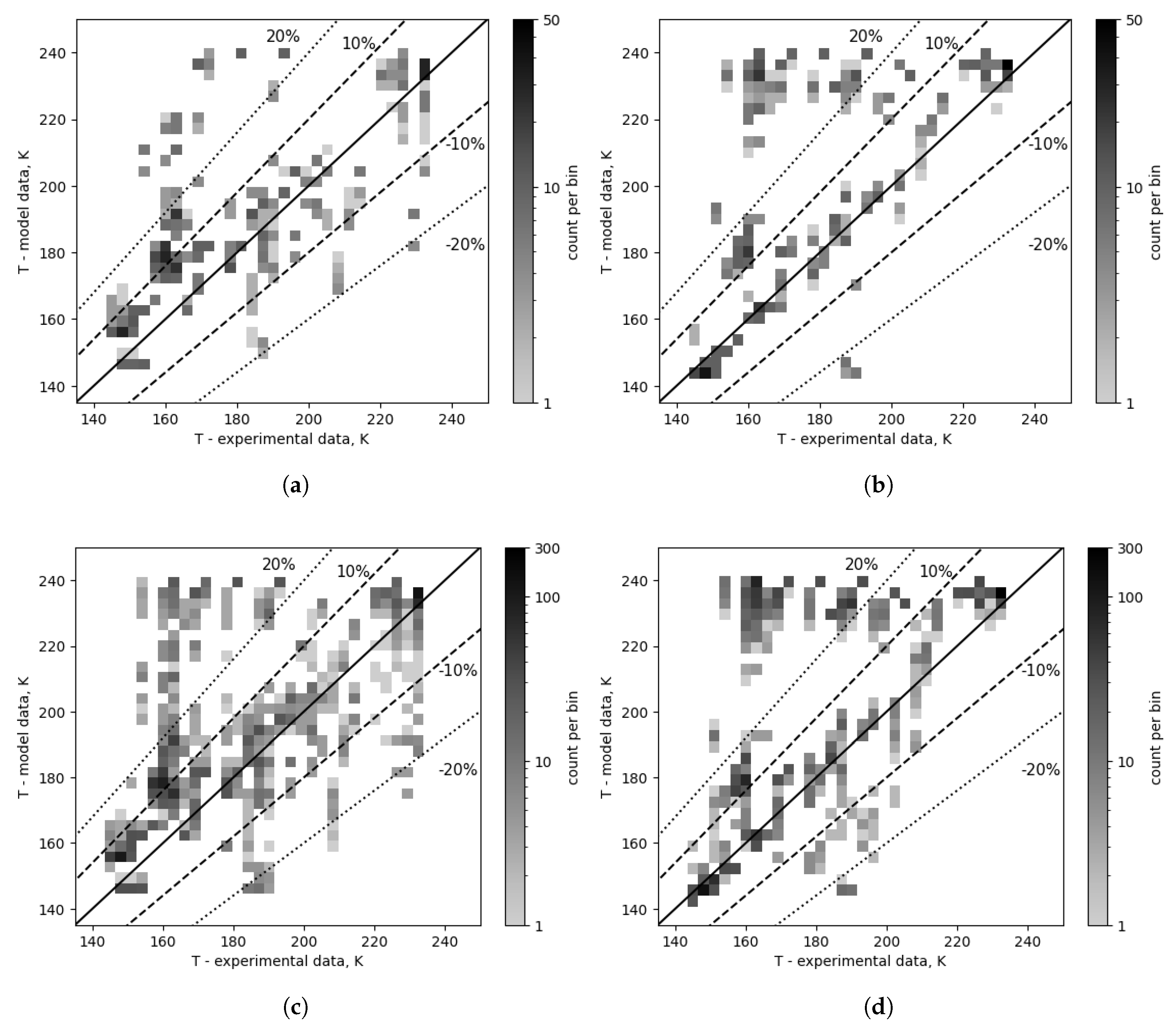
The composition correction results in an analogous change in one correlation, as shown in
Figure 11 and
Table 5, where the chosen Hsieh & Lin evaporation correlation results, both without and with composition correction, are compared. Consequently, this yields an increased overall amount of points in the ranges of ±30% (76.7% vs 87.3%), ±20% (62.5% vs 79.2%), and ±10% (37.7% vs 60.8%), as well as a reduction in the number of points in the high error band (26.3% vs 11.6%).
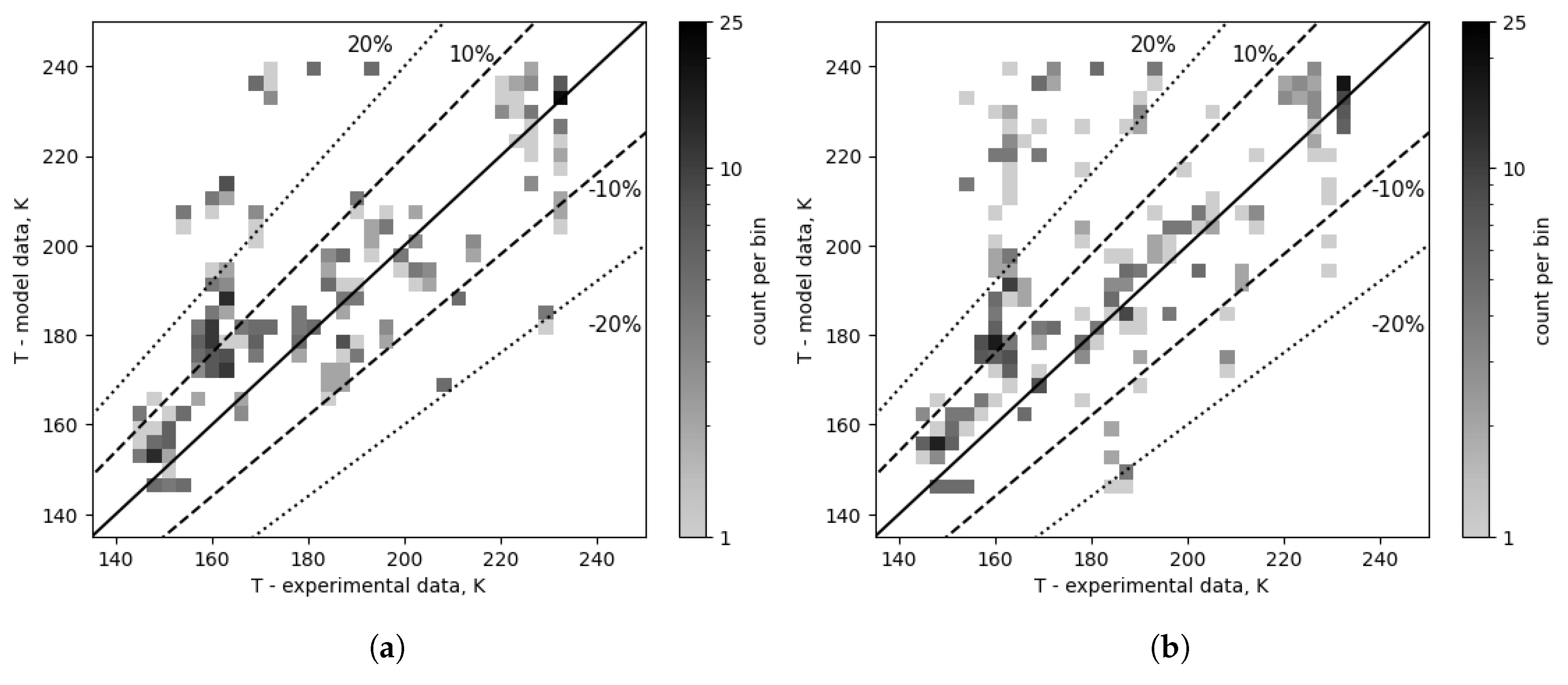
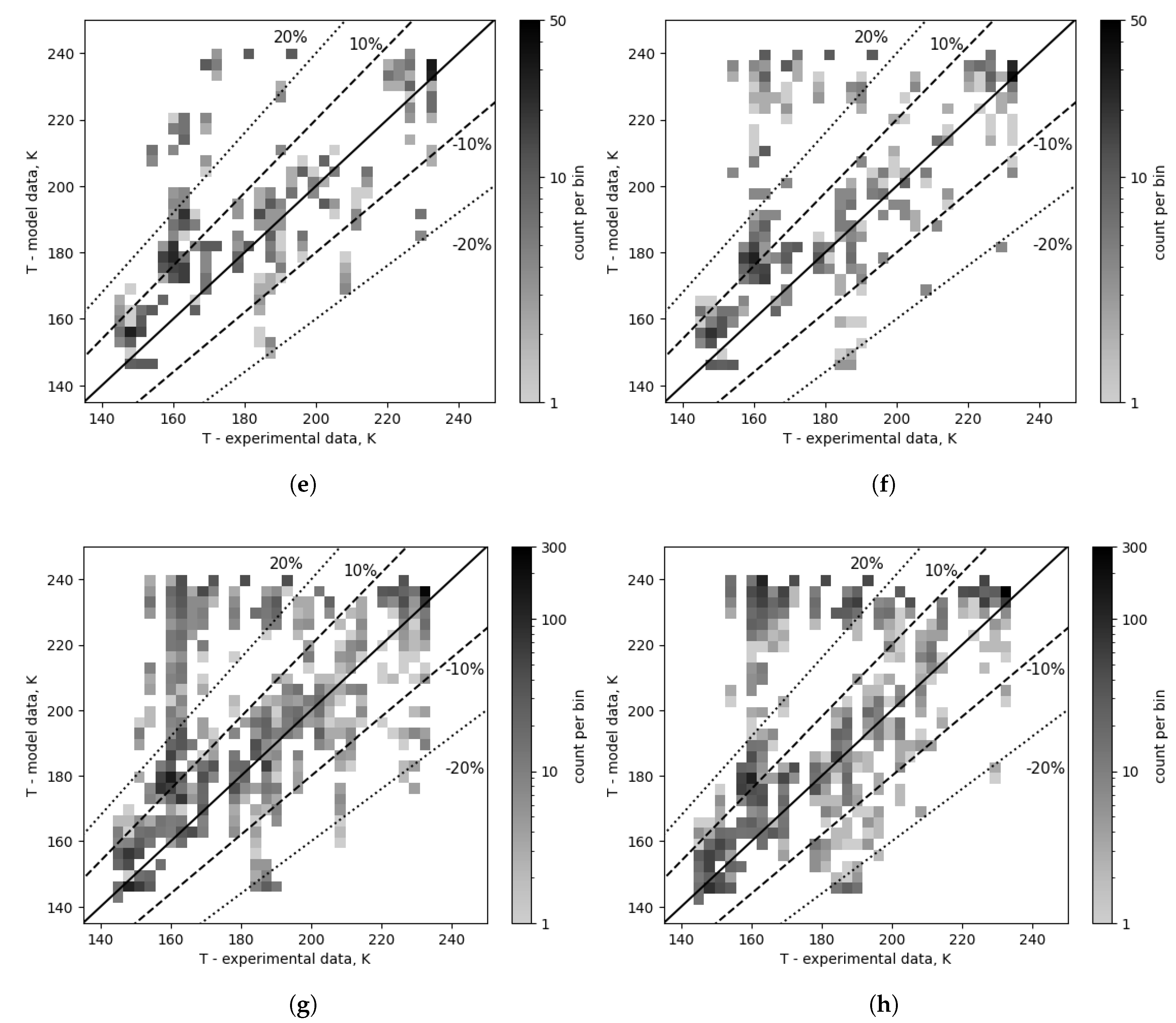
Figure 12 shows the comparison between the experimental and modeled outlet temperature data after the composition correction for two selected pairs of condensation correlations, chosen based on the lowest and highest error values: Kuo and Thonon, respectively. In
Figure 12c,d, the 220-240 K model temperature band contains a significant portion of all points, i.e., 10% for Kuo and 17% for Thonon, similar to the previous comparisons. There are significant differences in data distribution between Kuo and Thonon in ranges: ±30% (88.9% vs 82.7%), ±20% (82.1% vs 73.5%), and ±10% (62.4% vs 56.8%), as well as a reduction in the number of points in the high error band (26.3% vs 11.6%). As shown in
Figure 12a, the high error band is much less discernible if the analyzed correlation combinations have a lower overall error value, as in this instance where the top 5 is taken into account; In the case of the top 5, the high error band contains 2.5% and 7.2% of the data points, and the point distribution changed as follows: range of ±30% (92.5% vs 90.6%), ±20% (89.3% vs 83.6%), and ±10% (67.3% vs 63.8%). Kuo condensation also exhibited the highest spread of values among all the condensation correlations, with HTC values going as low as 0.355
, while the others had their lowest values in the order of 4
.
Analogous observations can be made regarding the correlations of evaporation with low and high error values. In
Figure 13, two evaporation correlations are compared in the same manner as in
Figure 12. Considering the overall results, 11.6% of points for Hsieh & Lin and 18.5% for Han fall within the high error band. A noticeable gap in the 10-30% range for experimental temperature values from 160 K to 190 K can be observed. The overall distribution of points for Hsieh & Lin and Han is as follows: range of ±30% (87.3% vs 81.5%), ±20% (79.2% vs 72.6%), and ±10% (60.8% vs 56.9%). For the top 10 correlation combinations, the distribution is: range of ±30% (92.3% vs 88.9%), ±20% (88.1% vs 82.5%), and ±10% (66.8% vs 64.0%).
Evaporation correction also seems to have the most significant effect on the results. In
Figure 14, a comparison between uncorrected and corrected evaporation heat transfer is presented. The overall distribution is as follows: range of ±30% (90.9% vs 78.9%), ±20% (83.1% vs 68.9%), and ±10% (63.0% vs 52.9%); for the top 10: range of ±30% (92.7% vs 81.8%), ±20% (88.2% vs 71.7%), and ±10% (66.8% vs 56.1%). In the high error band, overall there are 7.35% and 22.6% of all points, and for the top 10 combinations, the distribution is 2.6% and 22.6%, respectively. Interestingly, evaporation correction increases the percentage of points in the high error band the most among all the comparisons presented.
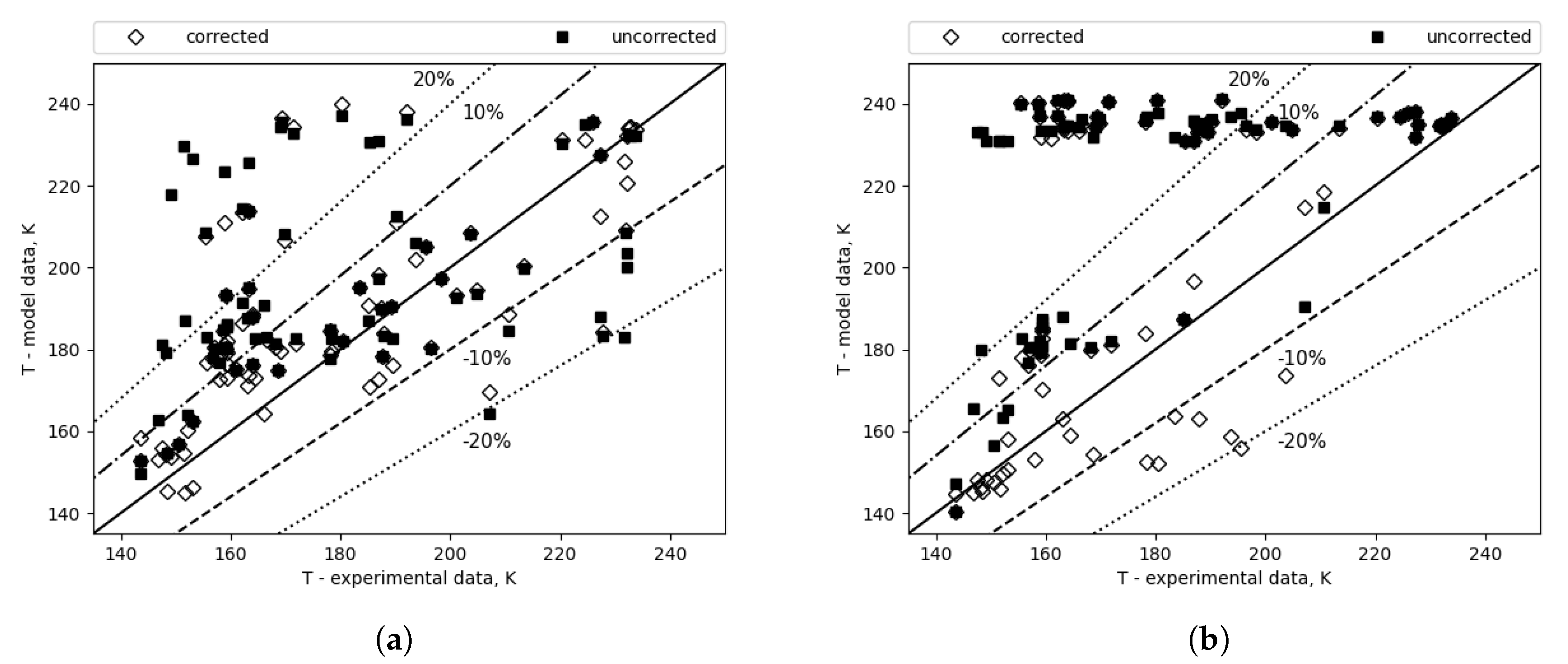
Correction seems to have less of an effect if the correlation combination itself has a low error, as shown in
Figure 15, which depicts the comparison of outlet temperatures for the combination with the lowest calculated error (
Figure 15a) and the highest (
Figure 15b). In the case of the combination with the highest error, the correction yielded approximately a 40 K reduction in error, and for the best overall combination, the composition correction resulted in approximately a 5 K decrease.
The cause of the high error band, or more broadly speaking, the strong asymmetry in the distribution of model temperatures, is most likely due to a general overestimation of the overall heat transfer coefficient values in this study. An example can be seen in
Figure 6a.
The assumption of no pressure drop may influence the results by shifting the phase change temperatures, especially in the cold stream, where a pressure drop of 0.2 bar can change the evaporation temperature by 3 K. However, the impact of the composition range is substantial (an 8 K change) and will not be negated by the introduction of pressure drop to the analysis.
Figure 6.
Temperature distribution along the HX length for the overall best combination of correlations. Single phase: Wanniarachchi, condensation: Kuo, evaporation: Hsieh & Lin and no evaporation correction. (a) MR composition as measured.(b) Corrected composition.
Figure 6.
Temperature distribution along the HX length for the overall best combination of correlations. Single phase: Wanniarachchi, condensation: Kuo, evaporation: Hsieh & Lin and no evaporation correction. (a) MR composition as measured.(b) Corrected composition.
Figure 7.
Temperature distributions along the HX length for minimum, maximum and measured composition of the MR. Best fit for minimum possible methane concentration. (a) Cold side. (b) Hot side.
Figure 7.
Temperature distributions along the HX length for minimum, maximum and measured composition of the MR. Best fit for minimum possible methane concentration. (a) Cold side. (b) Hot side.
Figure 8.
Temperature distributions along the HX length for minimum, maximum and measured composition of the MR. Best fit for measured methane concentration. (a) Cold side. (b) Hot side.
Figure 8.
Temperature distributions along the HX length for minimum, maximum and measured composition of the MR. Best fit for measured methane concentration. (a) Cold side. (b) Hot side.
Figure 9.
Temperature distributions along the HX length for minimum, maximum and measured composition of the MR. Best fit for measured methane concentration. (a) Cold side. (b) Hot side.
Figure 9.
Temperature distributions along the HX length for minimum, maximum and measured composition of the MR. Best fit for measured methane concentration. (a) Cold side. (b) Hot side.
Figure 10.
Outlet temperature comparison for all cases and combinations - composition corrected vs as measured. (a) Corrected cases - top 15. (b) Cases with composition as measured - top 15. (c) Composition corrected cases - overall (top 96). (d) Cases with composition as measured - overall (top 96).
Figure 10.
Outlet temperature comparison for all cases and combinations - composition corrected vs as measured. (a) Corrected cases - top 15. (b) Cases with composition as measured - top 15. (c) Composition corrected cases - overall (top 96). (d) Cases with composition as measured - overall (top 96).
Figure 11.
Outlet temperature comparison for a selected evaporation combination (number 1: Hsieh & Lin) - composition corrected vs as measured. (a) Composition corrected cases - top 10. (b) Cases with composition as measured - top 10. (c) Composition corrected cases - top 96. (d) Cases with composition as measured - top 96.
Figure 11.
Outlet temperature comparison for a selected evaporation combination (number 1: Hsieh & Lin) - composition corrected vs as measured. (a) Composition corrected cases - top 10. (b) Cases with composition as measured - top 10. (c) Composition corrected cases - top 96. (d) Cases with composition as measured - top 96.
Figure 12.
Outlet temperature comparison for selected condensation correlations, Kuo and Thonon. (a) Condensation: Kuo (number 2), top 5. (b) Condensation: Thonon (number 1), top 5. (c) Condensation: Kuo (number 2), overall (top 24). (d) Condensation: Thonon (number 1), overall (top 24).
Figure 12.
Outlet temperature comparison for selected condensation correlations, Kuo and Thonon. (a) Condensation: Kuo (number 2), top 5. (b) Condensation: Thonon (number 1), top 5. (c) Condensation: Kuo (number 2), overall (top 24). (d) Condensation: Thonon (number 1), overall (top 24).
Figure 13.
Outlet temperature comparison for selected evaporation correlations. (a) Evaporation: Hsieh & Lin (number 1), top 5. (b) Evaporation: Han (number 0), top 5. (c) Evaporation: Hsieh & Lin (number 1), overall (top 48). (d) Evaporation: Han (number 0), overall (top 48).
Figure 13.
Outlet temperature comparison for selected evaporation correlations. (a) Evaporation: Hsieh & Lin (number 1), top 5. (b) Evaporation: Han (number 0), top 5. (c) Evaporation: Hsieh & Lin (number 1), overall (top 48). (d) Evaporation: Han (number 0), overall (top 48).
Figure 14.
Outlet temperature comparison of corrected and uncorrected evaporation. (a) Evaporation correction: Fujita (number 2), top 5. (b) Evaporation correction: none (number 0), top 5. (c) Evaporation correction: Fujita (number 2), overall (top 32). (d) Evaporation correction: none (number 0), overall (top 32).
Figure 14.
Outlet temperature comparison of corrected and uncorrected evaporation. (a) Evaporation correction: Fujita (number 2), top 5. (b) Evaporation correction: none (number 0), top 5. (c) Evaporation correction: Fujita (number 2), overall (top 32). (d) Evaporation correction: none (number 0), overall (top 32).
Figure 15.
Outlet temperature comparison - correlation combinations with the lowest and highest error, respectively. (a) Lowest MAE (top 1). (b) Highest MAE (top 96).
Figure 15.
Outlet temperature comparison - correlation combinations with the lowest and highest error, respectively. (a) Lowest MAE (top 1). (b) Highest MAE (top 96).
5.1. Statistical Analysis
Beside graphical interpretation of the results, statistical analysis has been applied to quantify the validation of the model and possibly capture some unclear trends and impacts. Differences among heat-transfer correlations were evaluated (
,
,
,
,
) and their combinations using a blocked, repeated-measures design at the level of a case (one experiment with fixed conditions). For each case and each correlation combination, the model reproduced outlet temperatures and point-wise errors and then case-level mean absolute error (MAE; Eq.
9) and root-mean-square error (RMSE; Eq.
10) were computed.
Because the composition of the mixture was found to significantly affect the thermal profile, the analyzes were performed in two modes: (i) the measured composition (AM; = 0) and (ii) the corrected composition (CC; = best), where for each (case times combination) the composition shift was selected minimizing the MAE for summary analyzes. Unless stated otherwise, all inferences are blocked by case.
For each heat transfer mechanism, marginal effects (case-wise averaging over other factors) were computed and ranked all 96 combinations by case-average MAE (and RMSE) (
Table 6,
Table 7,
Table 8,
Table 9,
Table 10 and
Table 11). Global differences among
levels were tested with the Friedman test, and two-level contrasts with the Wilcoxon signed-rank test; pairwise post-hoc comparisons used Wilcoxon tests with
Holm correction for multiplicity. Along with p-values we report effect sizes (median differences with 95% bootstrap CIs) and, where applicable, Cohen’s
and
Kendall’s W.
All analyzes were performed in Python (v3.8.8) using NumPy, pandas (v1.2.4), and SciPy (v1.6.2) for data handling and statistical testing. The bootstrap confidence intervals used the NumPy random generator with a fixed seed (42) for reproducibility.
5.2. Ranking and Marginal Effects
The Kuo et al. condensation correlation ranked first in both modes and differed significantly from the alternatives (Friedman, ; post-hoc Holm-adjusted Wilcoxon, ). For evaporation, Hsieh & Lin outperformed Han (AM: K; CC: K; both ). Among evaporation-correction correlations, Fujita et al. achieved the lowest marginal . Notably, with Sun et al. the inclusion of the boiling-heat-transfer correction yielded higher errors than without correction (all pairwise ). For single-phase heat transfer (hot/cold streams analyzed separately), Wanniarachchi consistently performed best, irrespective of stream or mode. The best overall combination (AM) achieved K, lower than the best single-factor marginal MAE (e.g., K), indicating that no single mechanism dominates and that joint selection of levels across mechanisms provides synergistic gains.
5.3. Effect of Composition Correction
Across all case × combination pairs, composition-corrected mode (CC) reduced errors relative to AM (Wilcoxon in =CC-AM): median K (95% CI), mean K, ; improvement occurred in 90.5% of comparisons. The optimal composition shift had median pp (Wilcoxon vs 0: ), suggesting a systematic positive bias in the measured composition.
Despite the reductions in absolute error, the rankings were largely preserved between modes (Kendall’s , ); 10/15 top combinations overlapped. Thus, composition uncertainty did not alter the choice of best-performing correlations in this dataset. CC estimates is regarded as an upper bound on achievable accuracy, since composition correction was tuned on the same data.