Submitted:
23 January 2026
Posted:
27 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.1.1. Spent LCO Black Mass
2.1.2. Reagents
2.1.3. Microorganism
2.2. Characterization of Black Mass
2.2.1. X-Ray Fluorescence (XRF)
2.2.2. X-Ray Diffraction (XRD)
2.2.3. Scanning Electron Microscopy (SEM) and Energy Dispersive Spectroscopy (EDS)
2.3. Cultivation of Acidithiobacillus ferrooxidans
2.4. Bioleaching Medium and Experimental Design
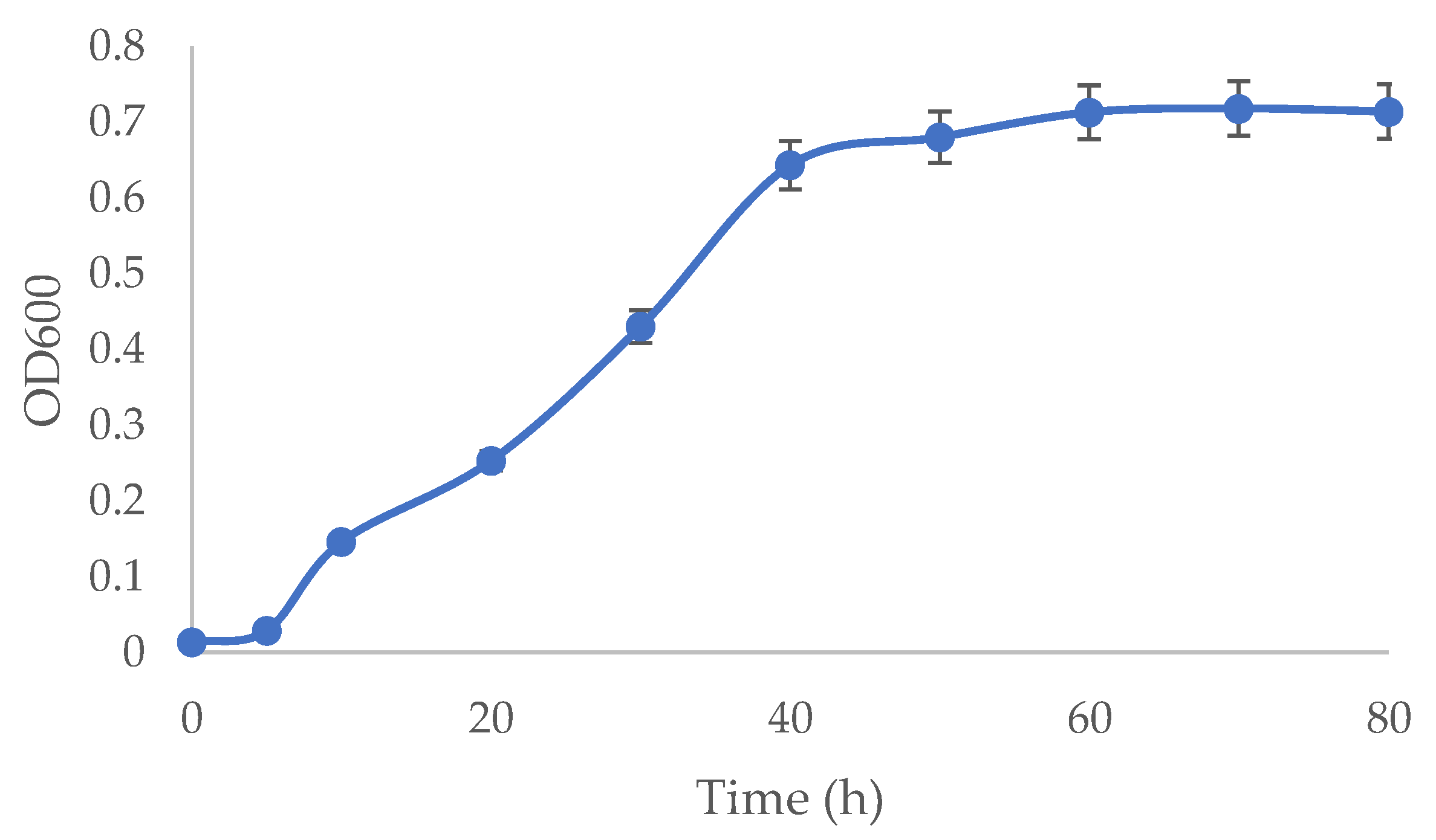
2.5. Microbial Growth Monitoring
2.6. Bioleaching Experiments and Sampling
2.7. Post-Leaching Solid Characterization
3. Results and Discussion
3.1. Chemical Composition and Microstructure of the Black Mass
3.1.1. AAS Quantification
3.1.2. XRF Results
3.1.3. XRD Analysis of the Black Mass
3.1.4. SEM-EDS Microstructural Analysis
3.2. Growth Behaviour of A. ferrooxidans and Implications for Bioleaching
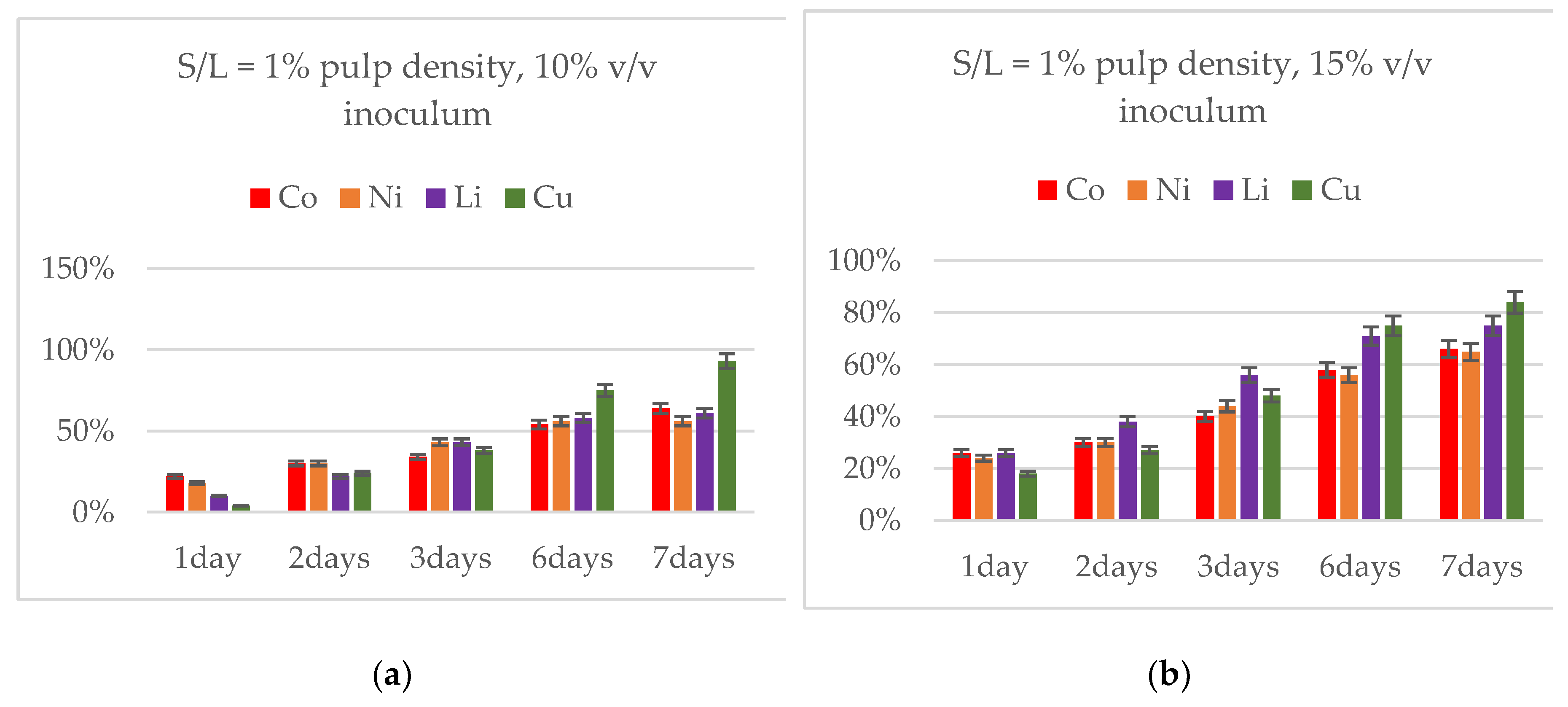
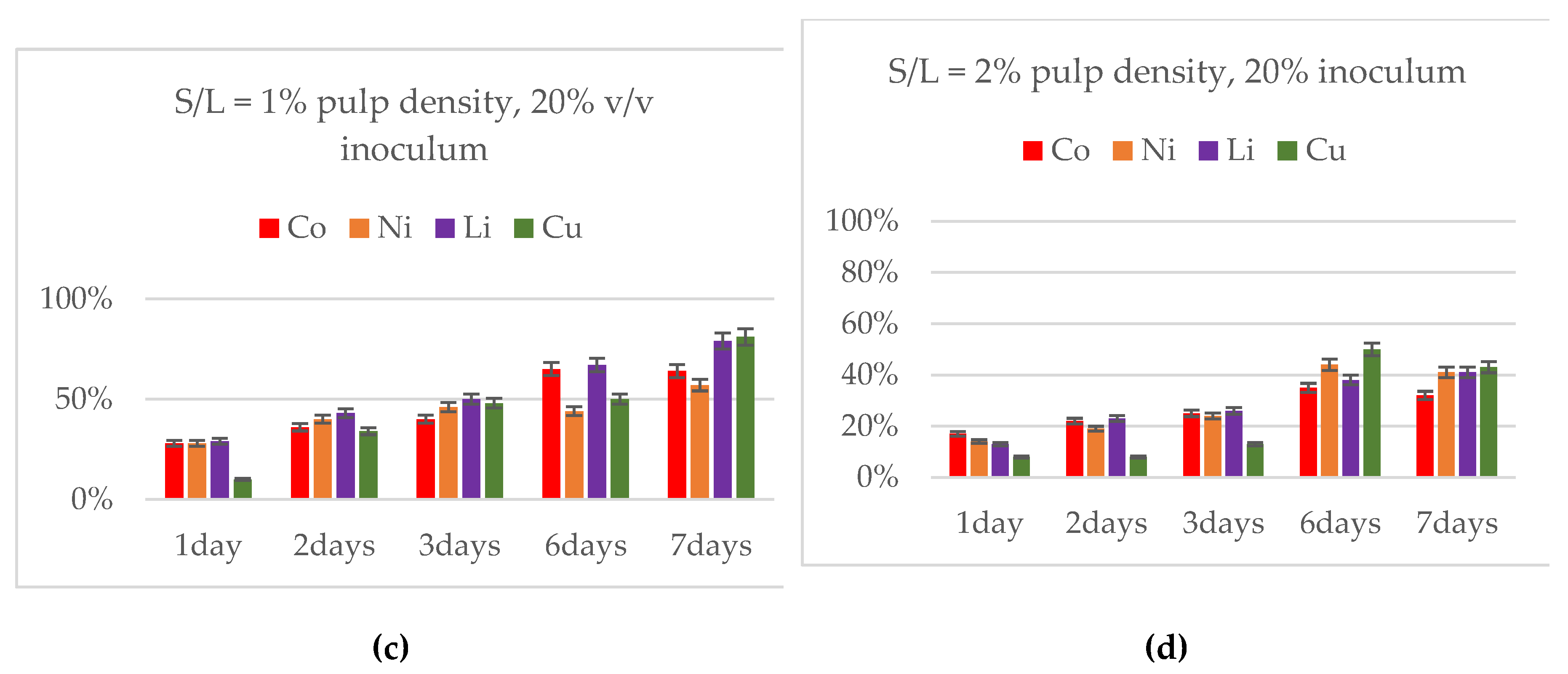
3.3. Effect of Inoculum Volume at 1 % Pulp Density
3.4. Effect of Pulp Density
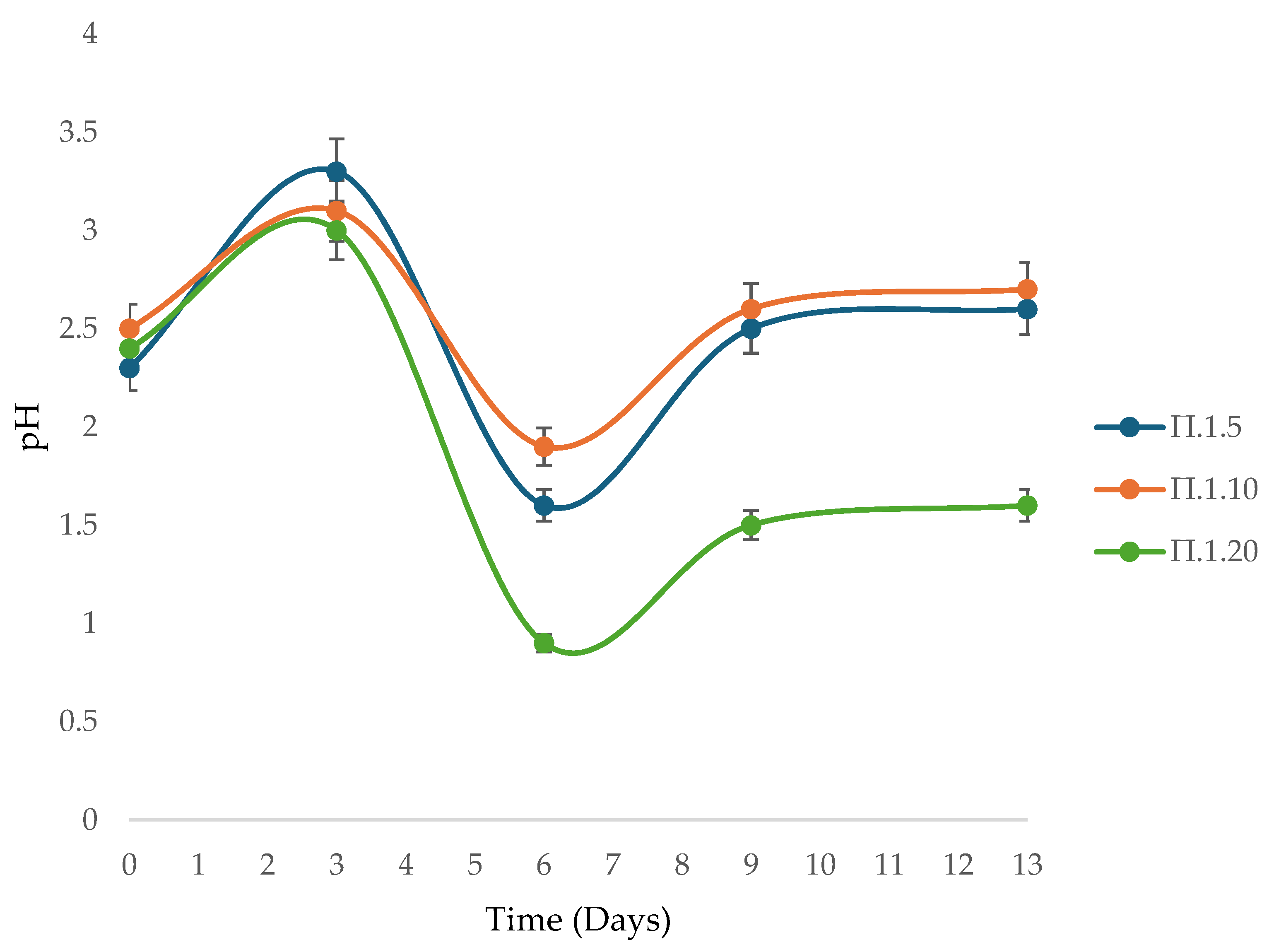
3.5. Influence of Initial and Dynamic pH Evolution on Bioleaching Performance
3.6. Linking pH Evolution, Microbial Activity and Dissolution Kinetics
3.7. Post-Leaching Residue Characterization
3.8. Comparison with Previous Bioleaching Studies and Process Implications
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Abbreviation | Definition |
| AAS | Atomic Absorption Spectroscopy |
| CEJ | Chemical Engineering Journal |
| Co | Cobalt |
| CRM | Critical Raw Material |
| Cu | Copper |
| DoE | Design of Experiments |
| DSMZ | Deutsche Sammlung von Mikroorganismen und Zellkulturen (German Collection of Microorganisms and Cell Cultures) |
| EDS | Energy Dispersive Spectroscopy |
| ED-XRF | Energy-Dispersive X-ray Fluorescence |
| EU | European Union |
| Fe | Iron |
| HPLC | High-Performance Liquid Chromatography |
| LIB | Lithium-Ion Battery |
| LCO | Lithium Cobalt Oxide |
| Li | Lithium |
| LOI | Loss on Ignition |
| Mn | Manganese |
| Ni | Nickel |
| NTUA | National Technical University of Athens |
| OD₆₀₀ | Optical Density at 600 nm |
| SEM | Scanning Electron Microscopy |
| UV–Vis | Ultraviolet–Visible (spectrophotometry) |
| WEEE | Waste Electrical and Electronic Equipment |
| XRD | X-ray Diffraction |
| XRF | X-ray Fluorescence |
| Zn | Zinc |
References
- Rautela, R.; Yadav, B.R.; Kumar, S. A Review on Technologies for Recovery of Metals from Waste Lithium-Ion Batteries. J. Power Sources 2023, 580, 233428. [Google Scholar] [CrossRef]
- Kiskira, K.; Plakantonaki, S.; Gerolimos, N.; Kalkanis, K.; Sfyroera, E.; Coelho, F.; Priniotakis, G. Life Cycle Optimization of Circular Industrial Processes: Advances in By-Product Recovery for Renewable Energy Applications. Clean Technol. 2026, 8, 5. [Google Scholar] [CrossRef]
- Ma, X.; Meng, Z.; Bellonia, M.V.; Spangenberger, J.; Harper, G.; Gratz, E.; Olivetti, E.; Arsenault, R.; Wang, Y. The Evolution of Lithium-Ion Battery Recycling. Nat. Rev. Clean Technol. 2025, 1, 75–94. [Google Scholar] [CrossRef]
- Zhang, X.; Li, L.; Fan, E.; Xue, Q.; Bian, Y.; Wu, F.; Chen, R. Toward Sustainable and Systematic Recycling of Spent Rechargeable Batteries. Chem. Soc. Rev. 2018, 47, 7239–7302. [Google Scholar] [CrossRef]
- Psomopoulos, C.S.; Kalkanis, K.; Chatzistamou, E.D.; Kiskira, K.; Ioannidis, G.Ch.; Kaminaris, S.D. End of Life Treatment of Photovoltaic Panels: Expected Volumes up to 2045 in E.U. AIP Conf. Proc. 2022, 2437, 020084. [Google Scholar] [CrossRef]
- Mansur, M.B.; Guimarães, A.S.; Petraniková, M. An Overview on the Recovery of Cobalt from End-of-Life Lithium-Ion Batteries. Miner. Process. Extr. Metall. Rev. 2022, 43, 489–509. [Google Scholar] [CrossRef]
- Mishra, G.; Jha, R.; Meshram, A.; Singh, K.K. A Review on Recycling of Lithium-Ion Batteries to Recover Critical Metals. J. Environ. Chem. Eng. 2022, 10, 108534. [Google Scholar] [CrossRef]
- Takahashi, V.C.I.; Botelho Junior, A.B.; Espinosa, D.C.R.; Tenório, J.A.S. Enhancing Cobalt Recovery from Li-Ion Batteries Using Grinding Treatment Prior to the Leaching and Solvent Extraction Process. J. Environ. Chem. Eng. 2020, 8, 103801. [Google Scholar] [CrossRef]
- Meshram, P.; Virolainen, S.; Abhilash; Sainio, T. Solvent Extraction for Separation of 99.9% Pure Cobalt and Recovery of Li, Ni, Fe, Cu, Al from Spent LIBs. Metals 2022, 12, 1056. [Google Scholar] [CrossRef]
- Aboulaich, A.; Yaden, A.; Elhalya, N.; Taylour, M.; Aqil, M.; Hdidou, L.; Dahbi, M.; Alami, J. Synthesis and Recyclability of Sheet-like Cobalt Carbonate Recovered from Spent Li-Ion Batteries Using a Simple Hydrometallurgy Process. Sustainability 2022, 14, 2552. [Google Scholar] [CrossRef]
- Cornelio, A.; Zanoletti, A.; Bontempi, E. Recent Progress in Pyrometallurgy for the Recovery of Spent Lithium-Ion Batteries: A Review of State-of-the-Art Developments. Curr. Opin. Green Sustain. Chem. 2024, 46, 100881. [Google Scholar] [CrossRef]
- Holzer, A.; Windisch-Kern, S.; Ponak, C.; Raupenstrauch, H. A Novel Pyrometallurgical Recycling Process for Lithium-Ion Batteries and Its Application to the Recycling of LCO and LFP. Metals 2021, 11, 1–22. [Google Scholar] [CrossRef]
- Bourtsalas, A.C.; Papadatos, P.E.; Kiskira, K.; Kalkanis, K.; Psomopoulos, C.S. Ecodesign for Industrial Furnaces and Ovens: A Review of the Current Environmental Legislation. Sustainability 2023, 15, 9436. [Google Scholar] [CrossRef]
- Botelho Junior, A.B.; Stopic, S.; Friedrich, B.; Tenório, J.A.S.; Espinosa, D.C.R. Cobalt Recovery from Li-Ion Battery Recycling: A Critical Review. Metals 2021, 11, 1999. [Google Scholar] [CrossRef]
- Ochsenkühn-Petropoulou, M.; Tsakanika, L.A.; Lymperopoulou, T.; Ochsenkühn, K.M.; Hatzilyberis, K.; Georgiou, P.; Stergiopoulos, C.; Serifi, O.; Tsopelas, F. Efficiency of Sulfuric Acid Leaching for the Recovery of Critical Metals from Greek Bauxite Residue. Metals 2018, 8, 915. [Google Scholar] [CrossRef]
- Strauss, M.L.; Diaz, L.A.; McNally, J.; Klaehn, J.; Lister, T.E. Separation of Cobalt, Nickel, and Manganese in Leach Solutions of Waste Lithium-Ion Batteries Using Dowex M4195 Ion Exchange Resin. Hydrometallurgy 2021, 206, 105757. [Google Scholar] [CrossRef]
- Álvial-Hein, G.; Mahandra, H.; Ghahreman, A. Separation and Recovery of Cobalt and Nickel from End-of-Life Products via Solvent Extraction Technique: A Review. J. Clean. Prod. 2021, 297, 126592. [Google Scholar] [CrossRef]
- Hatzilyberis, K.; Tsakanika, L.-A.; Lymperopoulou, T.; Georgiou, P.; Kiskira, K.; Tsopelas, F.; Ochsenkühn, K.-M.; Ochsenkühn-Petropoulou, M. Design of an Advanced Hydrometallurgy Process for the Intensified and Optimized Industrial Recovery of Scandium from Bauxite Residue. Chem. Eng. Process. Process Intensif. 2020, 155, 108015. [Google Scholar] [CrossRef]
- Tsakanika, L.-A.; Panagiotatos, G.; Lymperopoulou, T.; Chatzitheodoridis, E.; Ochsenkühn, K.; Ochsenkühn-Petropoulou, M. Direct Phosphoric Acid Leaching of Bauxite Residue for Selective Scandium Extraction. Metals 2022, 12, 228. [Google Scholar] [CrossRef]
- Arnold, S.; Ruthes, J.G.A.; Kim, C.; Presser, V. Electrochemical Recycling of Lithium-Ion Batteries: Advancements and Future Directions. EcoMat 2024, 6, e12494. [Google Scholar] [CrossRef]
- Yu, L.; Bai, Y.; Belharouak, I. Recycling of Lithium-Ion Batteries via Electrochemical Recovery: A Mini-Review. Batteries 2024, 10, 337. [Google Scholar] [CrossRef]
- Roy, J.J.; Cao, B.; Madhavi, S. A Review on the Recycling of Spent Lithium-Ion Batteries (LIBs) by the Bioleaching Approach. Chemosphere 2021, 282, 130944. [Google Scholar] [CrossRef] [PubMed]
- Kiskira, K.; Lymperopoulou, T.; Tsakanika, L.-A.; Pavlopoulos, C.; Papadopoulou, K.; Ochsenkühn, K.-M.; Lyberatos, G.; Ochsenkühn-Petropoulou, M. Study of Microbial Cultures for the Bioleaching of Scandium from Alumina Industry By-Products. Metals 2021, 11, 951. [Google Scholar] [CrossRef]
- Kiskira, K.; Lymperopoulou, T.; Lourentzatos, I.; et al. Bioleaching of Scandium from Bauxite Residue Using Fungus Aspergillus niger. Waste Biomass Valorization 2023, 14, 3377–3390. [Google Scholar] [CrossRef]
- Alipanah, M.; Jin, H.; Zhou, Q.; Barboza, C.; Gazzo, D.; Thompson, V.; Fujita, Y.; Liu, J.; Anderko, A.; Reed, D. Sustainable Bioleaching of Lithium-Ion Batteries for Critical Metal Recovery: Process Optimization through Design of Experiments and Thermodynamic Modeling. Resour. Conserv. Recycl. 2023, 199, 107293. [Google Scholar] [CrossRef]
- Kiskira, K.; Tsakanika, L.A.; Kritikos, A.; Ntakanas, C.; Chatzitheodoridis, E.; Papadopoulou, K.; Lyberatos, G.; Ochsenkühn-Petropoulou, M. Bioleaching of critical metals from spent lithium cobalt oxide batteries using Acidithiobacillus ferrooxidans. In Proceedings of the 14th International Conference on Instrumental Methods of Analysis: Modern Trends and Applications, Kefalonia, Greece, 14-17 September 2025. [Google Scholar] [CrossRef]
- Panda, S.; Dembele, S.; Mishra, S.; Akcil, A.; Agcasulu, İ.; Hazrati, E.; Tuncuk, A.; Malavasi, P.; Gaydardzhiev, S. Small-Scale and Scale-Up Bioleaching of Li, Co, Ni and Mn from Spent Lithium-Ion Batteries. J. Chem. Technol. Biotechnol. 2024. [Google Scholar] [CrossRef]
- Biswal, B.K.; Balasubramanian, R. Recovery of Valuable Metals from Spent Lithium-Ion Batteries Using Microbial Agents for Bioleaching: A Review. Front. Microbiol. 2023, 14, 1197081. [Google Scholar] [CrossRef]
- Tonietti, L.; Esposito, M.; Cascone, M.; Barosa, B.; Fiscale, S.; Muscari Tomajoli, M.T.; Sbaffi, T.; Santomartino, R.; Covone, G.; Cordone, A.; Rotundi, A.; Giovannelli, D. Unveiling the Bioleaching Versatility of Acidithiobacillus ferrooxidans. Microorganisms 2024, 12, 2407. [Google Scholar] [CrossRef]
- Naseri, T.; Mousavi, S.M. Improvement of Li and Mn Bioleaching from Spent Lithium-Ion Batteries Using Step-Wise Addition of Biogenic Sulfuric Acid by Acidithiobacillus thiooxidans. Heliyon 2024, e37447. [Google Scholar] [CrossRef] [PubMed]
- Kazemian, Z.; Larypoor, M.; Marandi, R. Evaluation of Myco-Leaching Potential of Valuable Metals from Spent Lithium Battery by Penicillium chrysogenum and Aspergillus niger. Int. J. Environ. Anal. Chem. 2023, 103, 514–527. [Google Scholar] [CrossRef]
- Li, J.; Xu, T.; Liu, J.; et al. Bioleaching metals from waste electrical and electronic equipment (WEEE) by Aspergillus niger: a review. Environ Sci Pollut Res 2021, 28, 44622–44637. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.; Nie, Y.; Li, Z.; Yang, K.; An, L.; Jiang, H.; Han, X.; Fu, Z.; Zhao, X. Efficient Bioleaching of Li from Waste Lithium-Iron Phosphate Batteries by Acidophilic Bacterial Consortium: Enrichment Condition, Li Recovery, and Brief Carbon Footprint Analysis. Chem. Eng. J. 2025, 161450. [Google Scholar] [CrossRef]
- Heydarian, A.; Mousavi, S.M.; Vakilchap, F.; Baniasadi, M. Application of a Mixed Culture of Adapted Acidophilic Bacteria in Two-Step Bioleaching of Spent Lithium-Ion Laptop Batteries. J. Power Sources 2018, 380, 105–116. [Google Scholar] [CrossRef]
- Premathilake, D.S.; Botelho Junior, A.B.; Tenório, J.A.S.; Espinosa, D.C.R.; Vaccari, M. Designing of a Decentralized Pretreatment Line for EOL-LIBs Based on Recent Literature of LIB Recycling for Black Mass. Metals 2023, 13, 374. [Google Scholar] [CrossRef]
- Rudolph, M.; van den Boogaart, K.G.; et al. Mineralogy and Material Characterization of Black Mass from Industrial LIB Recycling-Implications for Process Optimization. Minerals Engineering 2021, 171, 107080. [Google Scholar] [CrossRef]
- Nazerian, M.; Bahaloo-Horeh, N.; Mousavi, S.M. Enhanced Bioleaching of Valuable Metals from Spent Lithium-Ion Batteries Using Ultrasonic Treatment. Korean J. Chem. Eng. 2023, 40, 584–593. [Google Scholar] [CrossRef]
- Dash, J.; Ojha, R.; Pradhan, D. Progress in Bioleaching and Its Mechanism: A Short Review. Discov. Environ. 2025, 3, 238. [Google Scholar] [CrossRef]
- Morell, J.; Lao-Luque, C.; Solé-Sardans, M.; Ribas, D.; Guimerà, X.; Dorado, A.D. Closed-Loop Bioleaching for Mobile Phones from the Bioregeneration of the Leaching Agent to the Recovery of Metal: A Review. J. Ind. Eng. Chem. 2025, xx, 104020. [Google Scholar] [CrossRef]
- Garcia, L.; Morell, J.; Lao, C.; Solé-Sardans, M.; Dorado, A.D. Metal Recovery from Discarded Lithium-Ion Batteries by Bioleaching Coupled with Minimal Mechanical Pre-Treatment. Minerals 2025, 15, 566. [Google Scholar] [CrossRef]
- Mandl, M.M.; Lerchbammer, R.; Gerold, E. Bioleaching of Lithium-Ion Battery Black Mass: A Comparative Study on Gluconobacter oxydans and Acidithiobacillus thiooxidans. Metals 2025, 15, 1112. [Google Scholar] [CrossRef]
- Chacana-Olivares, J.; Peceño, B.; Grageda, M.; et al. Lithium-Ion Battery Recycling: A Perspective on Key Challenges and Opportunities. npj Mater. Sustain. 2025, 3, 38. [Google Scholar] [CrossRef]
- Zhang, X.; Shi, H.; Tan, N.; Zhu, M.; Tan, W.; Daramola, D.; Gu, T. Advances in Bioleaching of Waste Lithium Batteries under Metal Ion Stress. Bioresour. Bioprocess. 2023, 10, 19. [Google Scholar] [CrossRef] [PubMed]
- Elander, B.E.; Jiang, M.; Fahrenbruch, M.; Li, W.; Momeni, B.; Wang, D. Recycling Li-Ion Battery Cathode Materials in Iron-Fueled, Low-Sulfate Cultures of Acidithiobacillus ferrooxidans. ACS Sustainable Resour. Manage. 2025, 2, 1760–1768. [Google Scholar] [CrossRef]
- Kim, J.; Nwe, H.H.; Yoon, C.S. Enhanced Bioleaching of Spent Li-Ion Batteries Using A. ferrooxidans by Application of External Magnetic Field. J. Environ. Manag. 2024, 367, 122012. [Google Scholar] [CrossRef]
- Hong, J.-H.; Kim, J.; Han, E.; Yang, S.-M.; Kim, H.-S.; Kim, J.; Yoon, C.S. Magnetic Field-Assisted Bioleaching of Cathode Materials from Spent Li-Ion Batteries Using Acidithiobacillus ferrooxidans. Chemosphere 2025, 376, 144303. [Google Scholar] [CrossRef]
- Kiskira, K.; Papirio, S.; Mascolo, M.C.; Fourdrin, C.; Pechaud, Y.; van Hullebusch, E.D.; Esposito, G. Mineral Characterization of the Biogenic Fe(III)(Hydr)oxides Produced during Fe(II)-Driven Denitrification with Cu, Ni and Zn. Sci. Total Environ. 2019, 687, 401–412. [Google Scholar] [CrossRef] [PubMed]

| Element (Metal Oxide) | Element Concentration (%) | Metal Oxide Concentration (%) | |
|---|---|---|---|
| Co (Co3O4) | 19.1 | 26.3 | |
| Ni (NiO) | 4.9 | 7.2 | |
| Cu (CuO) | 0.8 | 1.0 | |
| Fe (Fe2O3) | 0.8 | 1.2 | |
| Al (Al2O3) | 0.6 | 1.0 | |
| Mn (MnO) | 0.4 | 0.6 | |
| LOI | 40% | ||
| Study | Microorganism | Feed material | Key operating conditions | Co (%) | Ni (%) | Li (%) | Cu (%) |
| Heydarian et al., 2018 [34] | Mixed acidophilic culture | Laptop LIBs (mixed cathodes) | 1% pulp, pH 1.8-2.0, 30 °C, two-step | ~65 | ~55 | — | — |
| Nazerian et al., 2023 [37] | A. ferrooxidans (+ ultrasound) | Spent LIB black mass | 2% pulp, pH 2.0, 30 °C, ultrasound-assisted | 68 | 62 | 71 | — |
| Alipanah et al., 2023 [25] | A. ferrooxidans | Spent LIBs (mixed cathodes) | 1% pulp, pH 2.0, 30 °C, optimized via DoE | ~70 | ~60 | ~65 | — |
| Panda et al., 2024 [27] | Mixed bacterial consortium | Industrial LIB black mass | 1-2% pulp, pH 2.0, 30 °C, scale-up tests | 60-68 | 55-63 | 50-65 | — |
| Kim et al., 2024 [45] | A. ferrooxidans + magnetic field | Spent LIB cathode material | 3% pulp, pH 2.0, 30 °C, magnetic field | >80 | >80 | — | — |
| This work | A. ferrooxidans | Pyrolyzed industrial LCO black mass | 1% pulp, pH 2.0 / non-adjusted, 30 °C | 64-70 | 57-72 | 52-79 | 81-100 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




