Submitted:
22 January 2026
Posted:
22 January 2026
You are already at the latest version
Abstract
Keywords:
MSC: 92C37, 91A80, 92B05, 91A22, 37N25, 92D25
1. Introduction
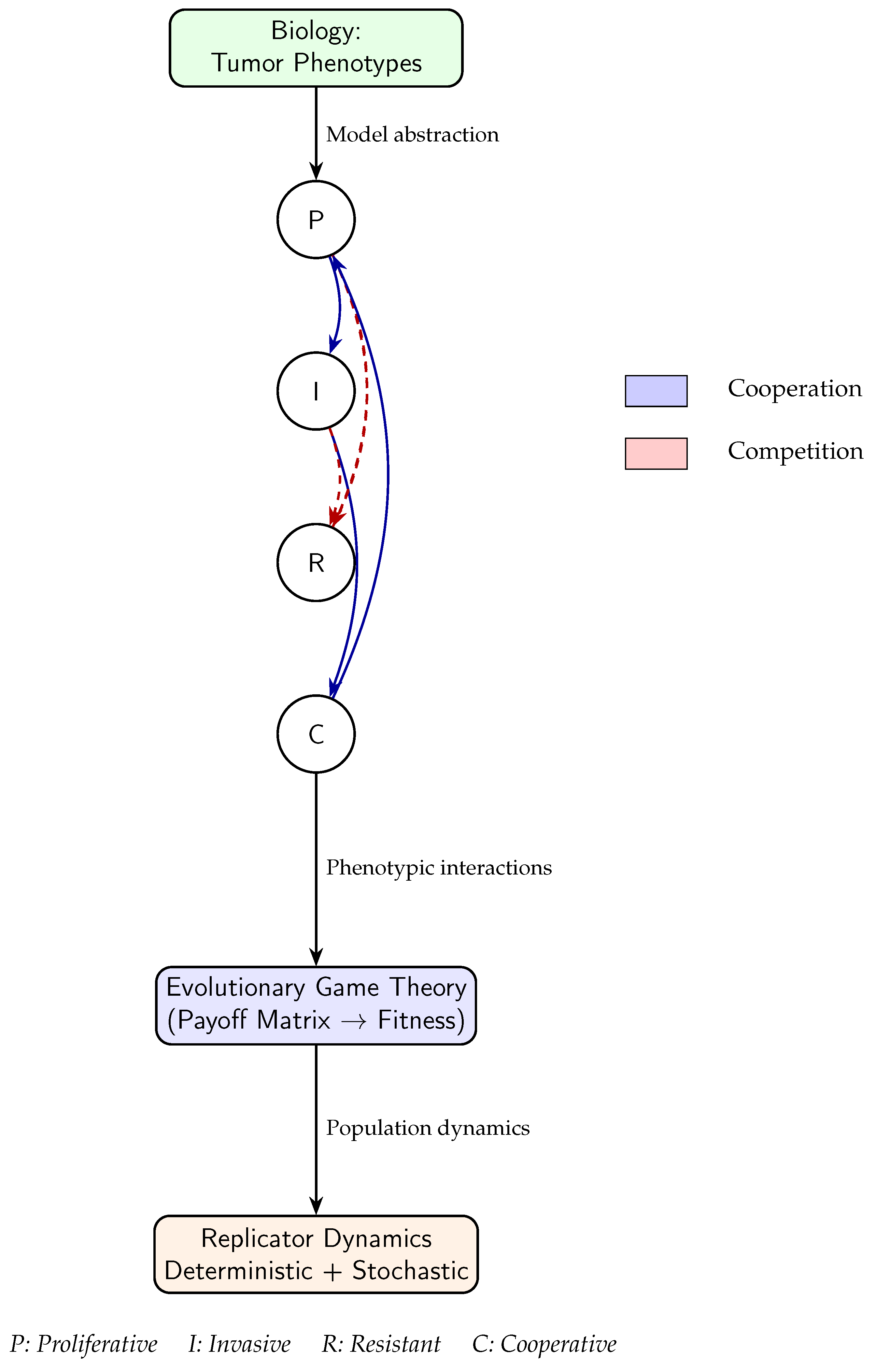
- Proliferative cells (P) — rapidly dividing but resource-sensitive,
- Invasive cells (I) — motile and adaptive to spatial gradients,
- Resistant cells (R) — tolerant to therapeutic stress, and
- Cooperative cells (C) — producers of shared public goods such as growth factors or extracellular modifiers.
2. Background and Motivation
2.1. Basics of Game Theory
2.2. Tumor Heterogeneity and Evolution
3. Game-Theoretic Models in Tumor Biology
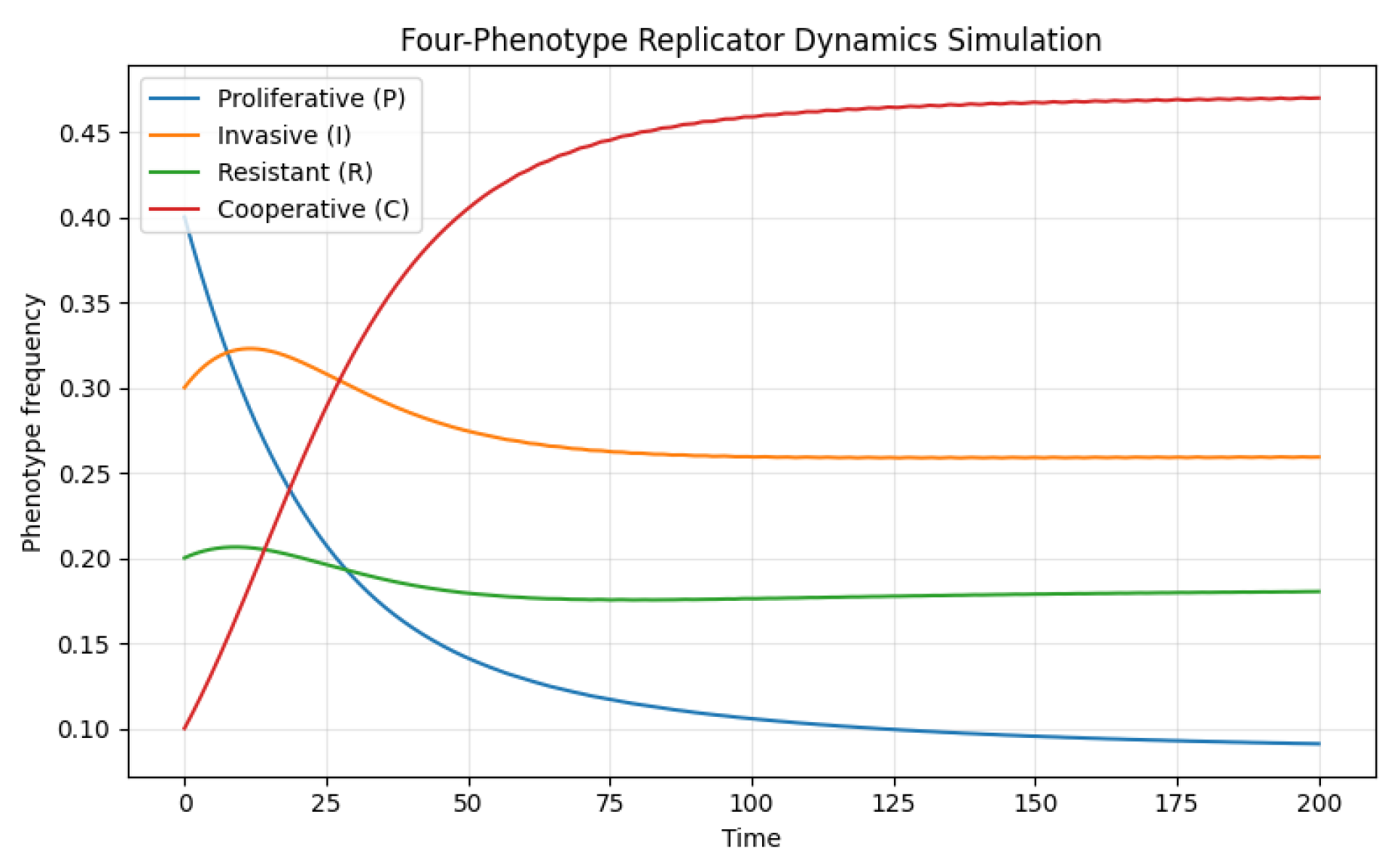
4. Numerical Simulation and Phase Analysis
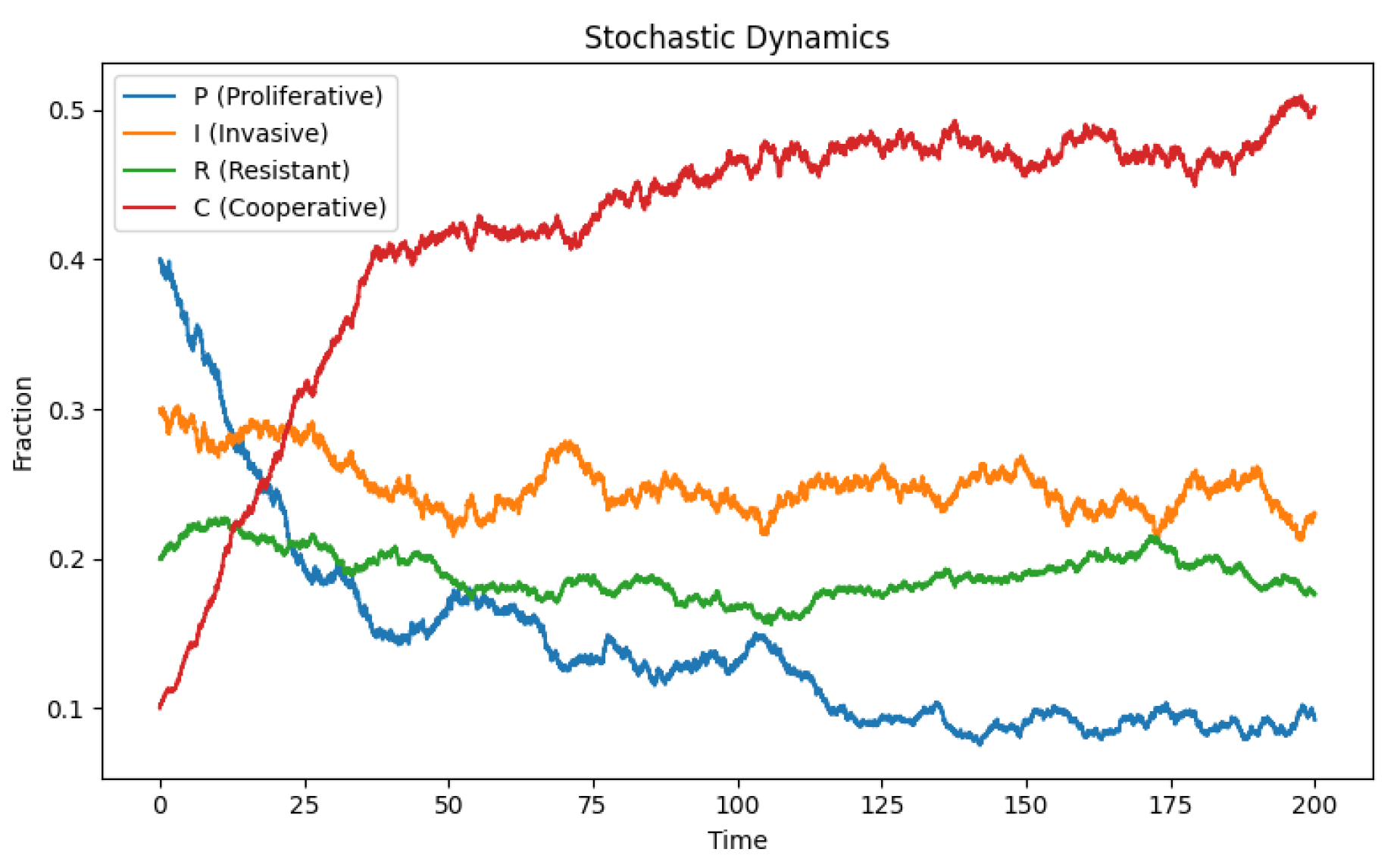
5. Integrating Stochastic Dynamics into the Four-Phenotype Model
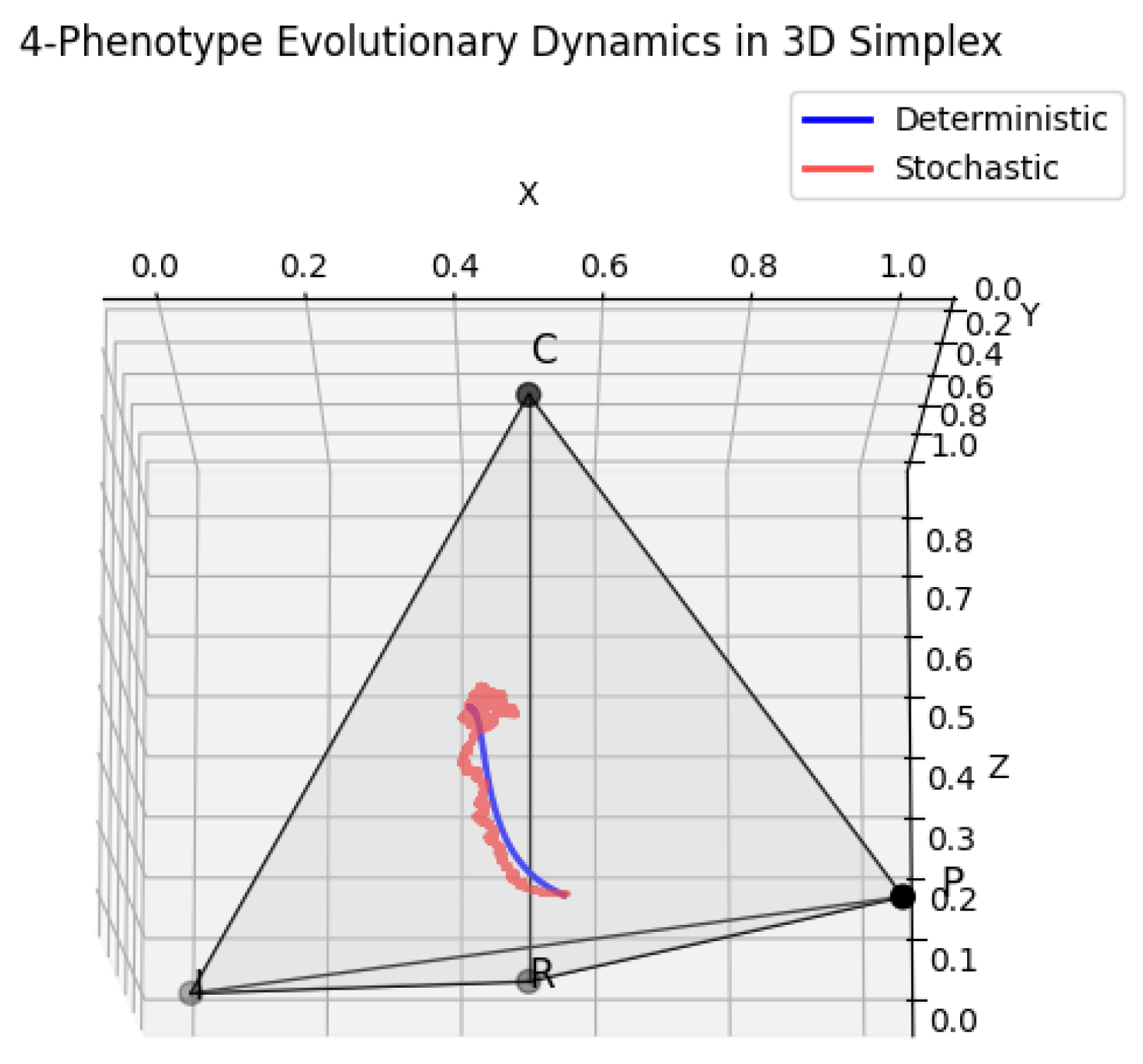
5.1. 3D Simplex Visualization of Stochastic and Deterministic Dynamics
6. Deterministic vs Stochastic Dynamics
7. Case Study: Environmental Modulation of Tumor Dynamics
7.1. Comparative Framework
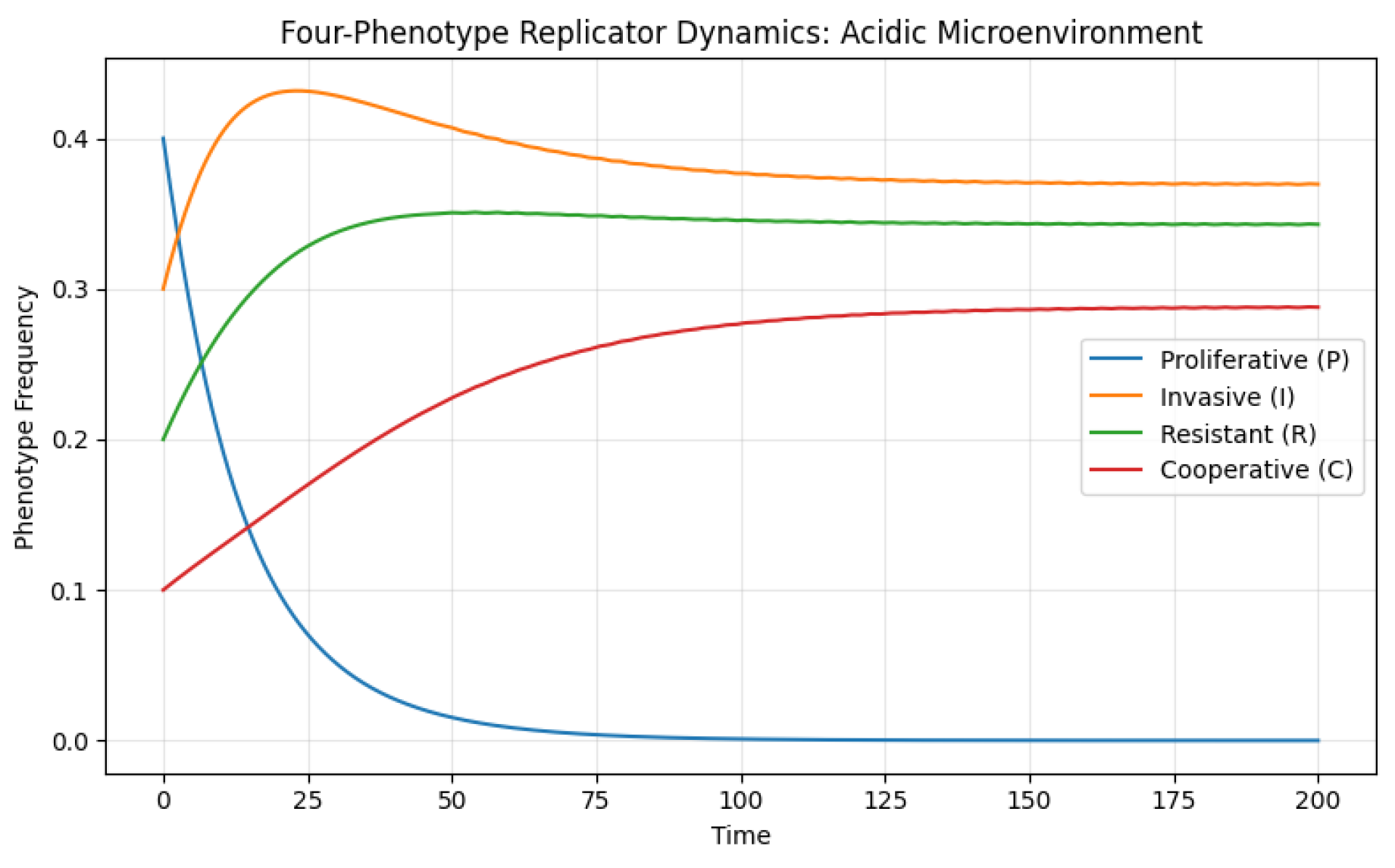
7.2. Acidic Microenvironment
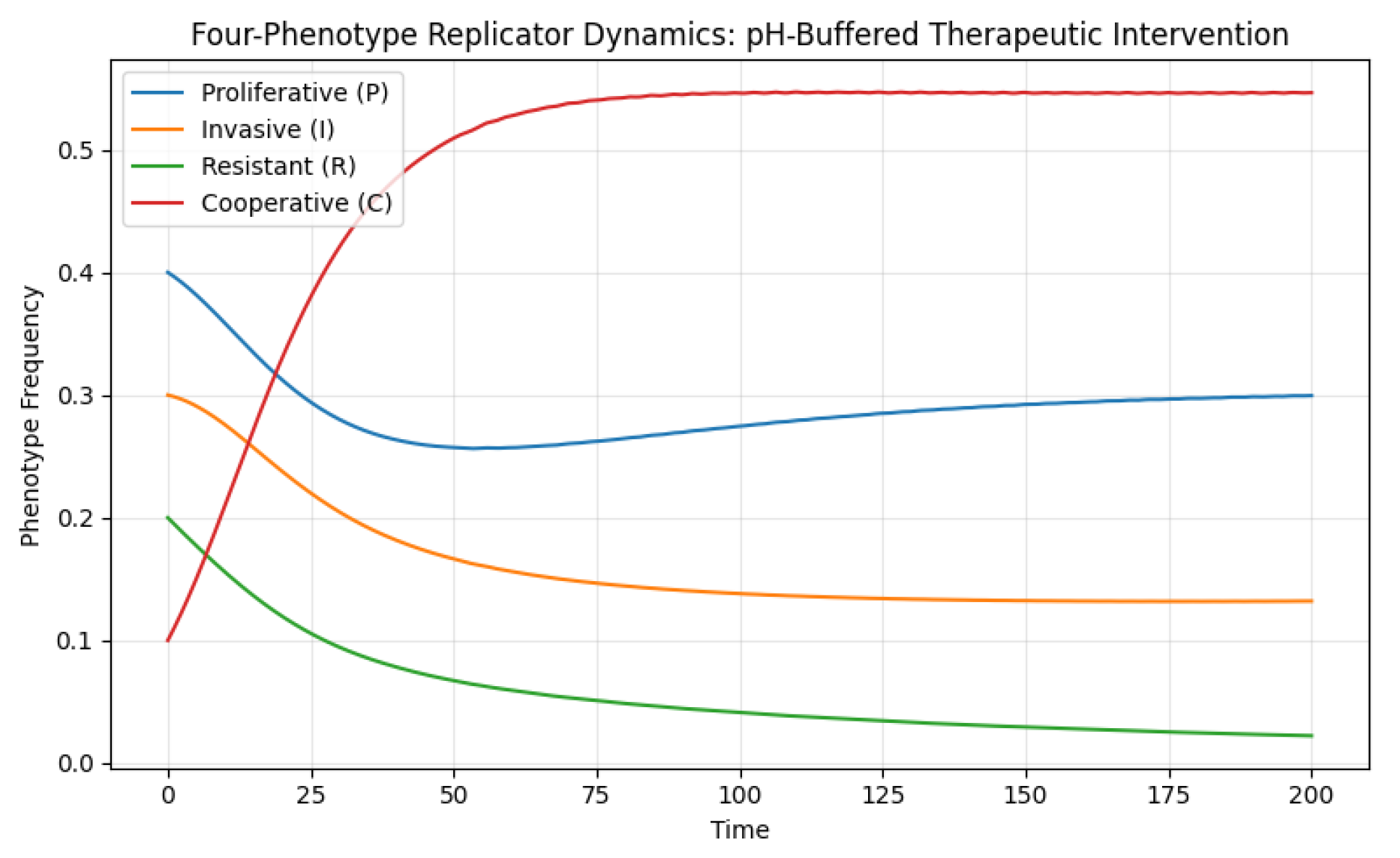
7.3. pH-Buffered Therapeutic Intervention
7.4. Interpretation and Implications
8. Conclusions
Acknowledgments
Conflicts of Interest
Appendix A. Replicator Equation Derivations, Simplified Forms, and Stability Calculations
Appendix A.1. Notation and Basic Replicator Equation
Appendix A.2. Derivation of the One-Dimensional Reduction
Appendix A.3. Mapping the Two-Strategy Reduction to the Four-Phenotype Model
Example.
Appendix A.4. Three-Strategy Interior Equilibrium
Appendix A.5. Jacobian and Linear Stability
Two-strategy case.
Appendix A.6. How to Reproduce the Algebra for the Manuscript’s Equations
- 1.
- Identify the equation numbers in the manuscript corresponding to simplified replicator forms (e.g., Eqs. (6), (13), (18)).
- 2.
- For each, determine which phenotypes are active and extract the appropriate or submatrix of A.
- 3.
- Substitute the submatrix entries into the formulas in Sections A.2–A.4.
Example substitution.
Appendix A.7. Stochastic Extension
References
- Maynard Smith, J.; Price, G.R. The logic of animal conflict. Nature 1973, 246, 15–18. [Google Scholar] [CrossRef]
- Hardin, G. The Tragedy of the Commons. Science 1968, 162, 1243–1248. [Google Scholar] [CrossRef]
- Nowak, M.A. Five rules for the evolution of cooperation. Science 2006, 314, 1560–1563. [Google Scholar] [CrossRef]
- Jarrett, A.M.; Lima, E.A.B.F.; Hormuth, D.A.; McKenna, M.T.; Feng, X.; Ekrut, D.A.; Yankeelov, T.E. Mathematical models of tumor cell proliferation: A review of the literature. Expert Review of Anticancer Therapy 2018, 18, 1271–1286. [Google Scholar] [CrossRef] [PubMed]
- von Neumann, J.; Morgenstern, O. Theory of Games and Economic Behavior; Princeton University Press, 1944. [Google Scholar]
- Basanta, D.; Anderson, A.R.A. Exploiting ecological principles to better understand cancer progression and treatment. Interface Focus 2013, 3, 20130020. [Google Scholar] [CrossRef] [PubMed]
- Archetti, M. Evolutionary game theory of growth factor production: implications for tumor heterogeneity and resistance to therapies. British Journal of Cancer 2013, 109, 1056–1062. [Google Scholar] [CrossRef]
- Gatenby, R.A.; Vincent, T.L. An evolutionary model of carcinogenesis. Cancer Research 2003, 63, 6212–6220. [Google Scholar]
- Tomlinson, I.P.M.; Bodmer, W.F. Modelling the consequences of interactions between tumour cells. British Journal of Cancer 1997, 75, 157–160. [Google Scholar] [CrossRef]
- Nowak, M.A.; Sigmund, K. Evolutionary dynamics of biological games. Science 2004, 303, 793–799. [Google Scholar] [CrossRef] [PubMed]
- McFarland, C.D.; Mirny, L.A.; Korolev, K.S. Tug-of-war between driver and passenger mutations in cancer and other adaptive processes. Proceedings of the National Academy of Sciences 2014, 111, 15138–15143. [Google Scholar] [CrossRef]
- Archetti, M.; Ferraro, D.A.; Christofori, G. Heterogeneity for IGF-II production maintained by public goods dynamics in neuroendocrine pancreatic cancer. Proceedings of the National Academy of Sciences 2015, 112, 1833–1838. [Google Scholar] [CrossRef]
- Dingli, D.; Chalub, F.A.C.C.; Santos, F.C.; Van Segbroeck, S.; Pacheco, J.M. Cancer phenotype as the outcome of an evolutionary game between normal and malignant cells. British Journal of Cancer 2009, 101, 1130–1136. [Google Scholar] [CrossRef]
- Basanta, D.; Hatzikirou, H.; Deutsch, A. Studying the emergence of invasiveness in tumours using game theory. European Physical Journal B 2008, 63, 393–397. [Google Scholar] [CrossRef]
- Orlando, P.A.; Gatenby, R.A.; Brown, J.S. Cancer treatment as a game: integrating evolutionary game theory into the optimal control of chemotherapy. Physical Biology 2012, 9, 065007. [Google Scholar] [CrossRef] [PubMed]
- Marusyk, A.; Almendro, V.; Polyak, K. Intra-tumour heterogeneity: a looking glass for cancer? Nature Reviews Cancer 2012, 12, 323–334. [Google Scholar] [CrossRef] [PubMed]
- Dagogo-Jack, I.; Shaw, A.T. Tumour heterogeneity and resistance to cancer therapies. Nature Reviews Clinical Oncology 2018, 15, 81–94. [Google Scholar] [CrossRef] [PubMed]
- Greaves, M.; Maley, C.C. Clonal evolution in cancer. Nature 2012, 481, 306–313. [Google Scholar] [CrossRef]
- Nowell, P.C. The clonal evolution of tumor cell populations. Science 1976, 194, 23–28. [Google Scholar] [CrossRef]
- Archetti, M. Cooperation among cancer cells: applying game theory to tumour metabolism. Nature Reviews Cancer 2015, 15, 558–566. [Google Scholar]
- Bach, L.A.; Bentzen, S.M.; Alsner, J.; Christiansen, F.B. An evolutionary-game model of tumour-cell interactions: possible relevance to gene therapy. European Journal of Cancer 2001, 37, 2116–2120. [Google Scholar] [CrossRef]
- Gatenby, R.A.; Brown, J.; Vincent, T. Lessons from Applied Ecology: Cancer Control Using an Evolutionary Double Bind. Cancer Research 2009, 69, 7499–7502. [Google Scholar] [CrossRef]
- Gallaher, J.A.; Enriquez-Navas, P.M.; Luddy, K.A.; Gatenby, R.A.; Anderson, A.R.A. Spatial heterogeneity and evolutionary dynamics modulate time to recurrence in continuous and adaptive cancer therapies. Cancer Research 2018, 78, 2127–2139. [Google Scholar] [CrossRef] [PubMed]
- Michor, F.; Iwasa, Y.; Nowak, M.A. Dynamics of cancer progression. Nature Reviews Cancer 2004, 4, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Waclaw, B.; Bozic, I.; Pittman, M.E.; Hruban, R.H.; Vogelstein, B.; Nowak, M.A. A spatial model predicts that dispersal and cell turnover limit intratumour heterogeneity. Nature 2015, 525, 261–264. [Google Scholar] [CrossRef] [PubMed]
- Anderson, A.R.A.; Weaver, A.M.; Cummings, P.T.; Quaranta, V. Tumor morphology and phenotypic evolution driven by selective pressure from the microenvironment. Cell 2006, 127, 905–915. [Google Scholar] [CrossRef]
- Hanahan, D. Hallmarks of cancer: new dimensions. Cancer Discovery 2022, 12, 31–46. [Google Scholar] [CrossRef]
- Kareva, I. Prisoner’s Dilemma in Cancer Metabolism. PLoS ONE 2011, 6, e28576. [Google Scholar] [CrossRef]
- Gerlee, P.; Anderson, A.R.A. The evolution of carrying capacity in constrained and expanding tumour cell populations. PLoS Computational Biology 2015, 11, e1004359. [Google Scholar] [CrossRef]
- Hofbauer, J.; Sigmund, K. Evolutionary Games and Population Dynamics; Cambridge University Press: Cambridge, UK, 1998. [Google Scholar]
- Szabó, G.; Fáth, G. Evolutionary games on graphs. Physics Reports 2007, 446, 97–216. [Google Scholar] [CrossRef]
- Archetti, M.; Pienta, K.J. Cooperation among cancer cells: applying game theory to cancer. Nature Reviews Cancer 2019, 19, 110–117. [Google Scholar] [CrossRef]
- Rodrigues, J.A. Stochastic Graph-Based Models of Tumor Growth and Cellular Interactions. AppliedMath 2025, 5, 62. [Google Scholar] [CrossRef]
- Ao, P. Potential in stochastic differential equations: novel construction. Journal of Physics A: Mathematical and General 2004, 37, L25. [Google Scholar] [CrossRef]
- Brock, A.; Chang, H.; Huang, S. Non-genetic heterogeneity—a mutation-independent driving force for cancer cell plasticity and drug resistance. Nature Reviews Genetics 2009, 10, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.B.; et al. Stochastic state transitions give rise to phenotypic equilibrium in populations of cancer cells. Cell 2011, 146, 633–644. [Google Scholar] [CrossRef] [PubMed]
- Kaznatcheev, A.; et al. Cancer treatment scheduling and dynamic heterogeneity in social dilemmas of tumor acidity and vasculature. Nature Ecology & Evolution 2019, 3, 460–468. [Google Scholar]
- Archetti, M.; Pienta, K.J. Cooperation among cancer cells: applying game theory to cancer. Philosophical Transactions of the Royal Society B 2019, 374, 20180145. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



