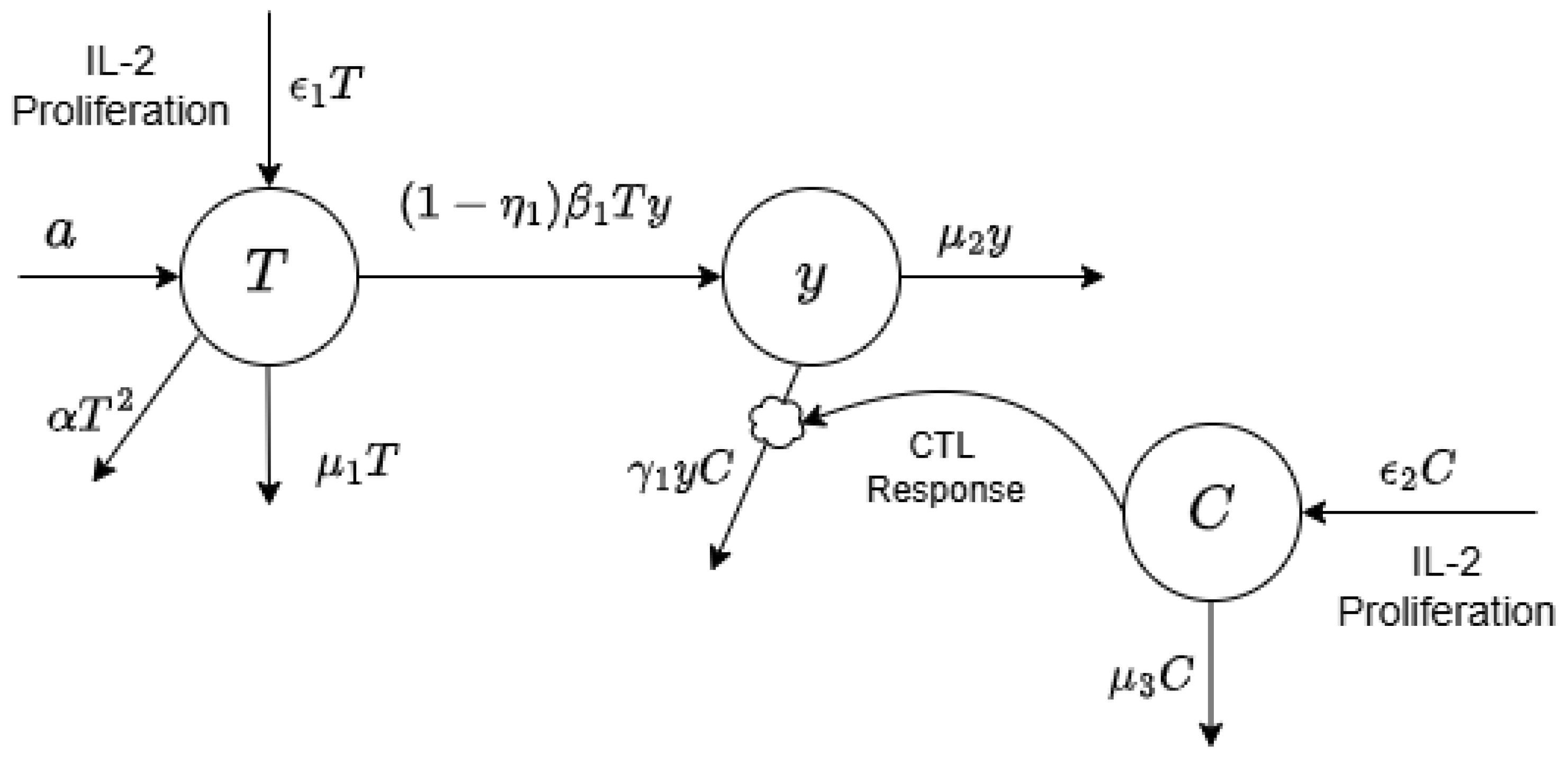
1. Introduction
AIDS (Acquired Immunodeficiency Syndrome) can develop from HIV (Human Immunodeficiency Virus), a chronic infection that targets the immune system, if treatment is not received. Effective treatment enables people with HIV to live long, healthy lives even though there is no cure [
1]. Significant advancements in long-acting treatments and cure-focused research are highlighted by recent HIV research. Clinical trials are showing promising results in both prevention and potential cures, and scientists are moving beyond daily pills to treatments that can be administered only twice a year [
2]. Researchers have made great strides in 2025 with long-acting injectables and innovative PrEP options, which lessen the burden of daily medication and enhance adherence for HIV-positive individuals. Significant advancements in long-acting injectables and innovative PrEP options, which lessen the burden of daily medication and increase adherence for individuals living with HIV, have been reported by researchers in 2025. Data on a investigational twice-yearly regimen that combined lenacapavir with broadly neutralizing antibodies (bNAbs) was presented by Gilead Sciences. The regimen met its primary endpoint in Phase 2 trials and was designated as a breakthrough therapy [
3]. The first HIV cure clinical trial, which was carried out in South Africa, also showed promising early results, highlighting the global movement toward functional cures. These advancements are significant because they improve quality of life and advance the scientific community’s long-term objective of HIV eradication [
4].
HIV transmission dynamics emphasize the critical role of Highly Active Antiretroviral Therapy (HAART) in reducing viral load and thereby lowering the probability of transmission. Mathematical models consistently show that HAART suppresses viral replication, driving the basic reproduction number (
) below unity and stabilizing the system toward a disease-free equilibrium [
5,
6]. Interleukin-2 (IL-2), a cytokine that promotes
T-cell proliferation, has been investigated as an adjunct to HAART. Clinical trials demonstrated that IL-2 administration increases
counts, enhancing immune reconstitution, though without significant improvement in long-term clinical outcomes compared to HAART alone [
7,
8]. Thus, while HAART directly reduces transmission risk by lowering viral load, IL-2 contributes indirectly by bolstering immune recovery, highlighting a complementary but limited role in transmission dynamics.
Mathematical modeling of viral dynamics provides a rigorous framework for designing therapeutic strategies and assessing the efficacy of antiviral interventions. Extensive research has examined the global stability of models that capture the progression of viral infections, particularly in the context of HIV. The host immune response is a critical determinant in limiting viral proliferation, and its modulation significantly influences disease outcomes. Modern antiretroviral agents are capable of suppressing viral replication and reducing immune activation, thereby mitigating apoptosis. Nevertheless, while Highly Active Antiretroviral Therapy (HAART) has proven effective in controlling viral load, it only partially restores immune function. Complete eradication of HIV through HAART alone remains unattainable, as viral rebound typically occurs upon cessation of therapy. Consequently, there is growing interest in complementary therapeutic approaches, such as the administration of interleukin-2 (IL-2), which may enhance immune reconstitution and support long-term viral control [
9,
10,
11].
Hopf bifurcation analysis is an important mathematical characteristic [
12]. It is helpfun in understanding the oscillatory behavior of human diseases such as HIV infection dynamics [
13]. Such oscillations, captured through Hopf bifurcation, provide insight into how treatment delays, cure rates, or immune saturation effects influence the persistence or suppression of HIV. Recent studies have rigorously examined these dynamics, showing that bifurcation thresholds mark transitions between stable infection states and oscillatory regimes, thereby offering valuable guidance for therapeutic strategies and long-term disease management [
14].
Interleukin-2 (IL-2) is a well-established cytokine that functions as a critical growth factor for T cells, regulating both their proliferation and differentiation across the entire T-cell compartment. Following antigenic stimulation, IL-2 is secreted by
and
T-cell subsets within peripheral lymphoid organs such as the spleen and lymph nodes. In the context of HIV infection, intermittent administration of highly active antiretroviral therapy (HAART) in combination with immune modulators like IL-2 has been proposed as a therapeutic strategy to suppress viral replication. Mechanistically, IL-2 enhances
T-cell activation, which can transiently reactivate latent HIV reservoirs. This reactivation allows cytotoxic T lymphocytes (CTLs) specific to HIV to recognize and eliminate infected cells, thereby contributing to viral clearance [
12].
To better understand these complex interactions, mathematical models have been developed to simulate the effects of drug therapy in HIV patients. These models provide insights into viral dynamics, the emergence of drug resistance, and the decay kinetics of HIV-infected cellular compartments [
15]. Furthermore, they have been applied to study thymic reconstitution in HIV-1 patients undergoing HAART, as well as to design optimal control strategies that integrate both HAART and IL-2 therapy [
16]. Collectively, such modeling approaches highlight the potential of combining immunotherapy with antiretroviral treatment to improve long-term outcomes in HIV management.
Optimal control theory provides a rigorous mathematical framework for the design of drug administration strategies in HIV treatment, enabling researchers to balance therapeutic efficacy against the minimization of adverse side effects and economic costs [
17]. By embedding drug scheduling protocols into dynamical models of HIV infection, one can formulate control functions that simultaneously suppress viral replication and preserve immune competence [
18,
19]. Within this context, the Pontryagin Maximum Principle (PMP) supplies necessary conditions for optimality, allowing the derivation of adjoint equations and the construction of the Hamiltonian function that encapsulates system dynamics, control variables, and associated cost functionals [
20,
21,
22]. The Hamiltonian formalism facilitates the identification of optimal trajectories for antiretroviral therapy (ART), guiding both dosage and timing decisions.
Recent studies have extended these models to incorporate compartments for remission, latent reservoirs, and patient adherence, thereby reflecting the complex biological and behavioral dimensions of HIV treatment [
23]. Such analyses demonstrate that optimal control strategies, derived through PMP and Hamiltonian methods, can significantly improve long-term outcomes by reducing viral load while maintaining immune system integrity [
18,
19]. Boukary et al. [
24] developed a mathematical model considering vertical transmission and applied optimal control to reduce the basic reproduction number and stabilize disease-free equilibria. Similarly, Rana et al. [
25] introduced a deterministic SHATR model (Susceptible–HIV infected–AIDS infected–Treatment–Recovered) and used nonlinear stability analysis with control strategies to evaluate rehabilitation and treatment outcomes. More recently, Kumar et al. [
26] emphasized stochastic elements in HIV spread, showing how optimal control can adapt drug interventions under uncertainty. These approaches demonstrate that mathematical optimization not only refines drug application design but also enhances long-term treatment planning, offering insights into personalized therapy schedules and public health strategies.
In previous studies, the focus has primarily been on stability analysis and optimal control, while phenomena such as periodic oscillations, limit cycles, and Hopf bifurcations have not been adequately captured by mathematical models. Although investigations incorporating time delays have addressed these aspects, models without delay have not yet examined Hopf bifurcations, stability properties, or the direction of bifurcating periodic solutions.
In this study, we undertake a comprehensive investigation of the reconstitution dynamics of T cells and their regulatory influence on cytotoxic T lymphocytes (CTLs) in HIV-infected individuals undergoing Highly Active Antiretroviral Therapy (HAART) in conjunction with interleukin-2 (IL-2) administration. The proposed mathematical modeling framework is designed to elucidate the interactions among distinct T lymphocyte subsets during HIV progression and to determine optimal conditions for immune-based therapeutic strategies associated with HAART. Moreover, the framework integrates principles of optimal control theory to inform the rational scheduling of IL-2 as an adjuvant therapy alongside HAART, with the overarching objective of enhancing immune restoration, suppressing viral persistence, and ultimately advancing toward a functional cure.
3. Basic Properties
We now analyze the qualitative properties of the system (
1) with respect to existence, uniqueness, and positivity of solutions.
3.1. Existence and Uniqueness of Solutions
The right-hand side of system (
1) is a polynomial in
and hence belongs to
. Therefore, it is locally Lipschitz continuous on
. By the Picard–Lindelöf Theorem, for any initial condition
, there exists a unique local solution. We emphasize that while uniqueness and existence are guaranteed, closed-form explicit solutions are not generally available.
3.2. Positive Invariance
We now show that the nonnegative orthant is positively invariant under the dynamics.
From the second equation of (
1),
which is linear in
y. Thus, we have
If
, then
. If
, then
for all
t in the interval of existence. Hence
for all
. Similarly, the third equation of (
1) is linear in
C:
yielding
Thus for all whenever .
For the nonnegativity of
T, we consider the first equation of (
1),
We note that, on the boundary , , so the vector field points inward. Also, the quadratic and bilinear terms are non-positive for , ensuring that cannot cross into negative values. Therefore for all whenever .
3.3. Boundedness of the System
The right-hand side of system (
1) consists of smooth polynomial functions in the variables
T,
y, and
C, together with the system parameters. Therefore, local existence, uniqueness, and continuity of solutions are guaranteed. In this subsection, we establish that the solutions of the system are uniformly bounded under suitable parameter conditions.
Theorem 1.
Let be a solution of (1) with nonnegative initial data. If and , then is uniformly bounded for all .
Proof. Firstly, we consider the first equation of (
1) to obtain the inequality
This is a linear differential inequality. By the comparison Theorem [
29],
is bounded above by the solution of
Solving this equation, we get
For the boundedness of
, we recall
which implies
Since and are bounded and continuous, the integral remains finite for all , so is bounded.
For the boundedness of
, we consider the third equation of (
1)
Since , the coefficient of C is bounded above by . Since we already proved is bounded, the growth rate of C is bounded, and hence itself is bounded.
Combining these results, we conclude that is uniformly bounded for all if and . □
3.4. Equilibrium Analysis
We now determine the equilibrium points of system (
1). The model (
1) admits three equilibria, namely the followings:
- (i)
the disease-free equilibrium
where
- (ii)
-
the CTL-free endemic equilibrium
, where
is feasible if .
- (iii)
the endemic equilibrium
where
and
.
is biologically feasible if
and
.
3.5. The Basic Reproduction Number
In this section, we derive the basic reproduction number using the next-generation matrix (NGM) method. We follow the standard framework in which is defined as the spectral radius of , where F collects the rates of appearance of new infections and V collects the transition terms out of infected compartments.
Here, we note that the DFE is
, where
Now, we choose the infected compartments. The only epidemiologically infected compartment is the population of infected T cells, y. CTLs (C) mediate immune response but are not infected states; T represents susceptible/uninfected T cells. Therefore, the infected subsystem is one-dimensional with state y.
We write the differential equation for
y as follows
At the DFE
, the linearization of
F and
V with respect to
y is the followings:
Since the infected subsystem is one-dimensional, the next-generation matrix
reduces to the scalar
This expression makes explicit how RTI efficacy () and IL-2 augmentation () modulate the infection potential through the susceptible pool at the DFE.
Theorem 2.
By the NGM theory[30,31], the DFE is locally asymptotically stable if and unstable if .
Remark 1.
A small introductions of infected T cells die out, and the system returns to the DFE when . The immune system successfully eliminates the initial viral challenge, preventing further spread to uninfected cells. When , the DFE becomes unstable and an endemic equilibrium with exist (subject to feasibility conditions identified in the equilibrium analysis). In this situation, the immune response is insufficient to eradicate the virus before additional cells become infected, thereby sustaining the infection.
4. Stability of Equilibria
For local stability at a state , linearize the system with the Jacobian . The equilibrium is locally asymptotically stable if and only if all eigenvalues of J have negative real parts. For a system, use the Routh–Hurwitz conditions on the characteristic polynomial.
(i) The Jacobian at disease-free equilibrium,
is
At
, the eigenvalues are
For the stability of the equilibrium, we require the conditions as:
(ii) At
the Jacobian matrix is determined as
At
one eigenvalue is
Other two eigenvalues satisfy
where
Thus the equilibrium is stable if
(iii) Local stability at a endemic equilibrium point
: The Jacobian matrix at
is
with
The characteristic equation of
J at
be
where,
The Routh–Hurwitz conditions for all roots of
to have negative real parts are
Therefore, the local asymptotic stability conditions at the point
are
Now, we study the local Hopf bifurcation of . Any of the parameters of the model may be a bifurcation parameter. Thus we assume, as the generic bifurcating parameter of the system.
Theorem 3.
The system (1), undergoes Hopf bifurcation around the endemic equilibrium point whenever the critical parameter value contained in the following domain
Proof. Using the condition,
, the characteristic equation (
6) becomes
which has three roots namely
and
. Therefore, a pair of purely imaginary eigenvalues exists for
.
Now we verify the transversality condition. For this, we differentiate the characteristic equation (
6) with respect to
to obtain
Thus Hopf bifurcation occurs at □
4.1. Stability and Direction of Hopf bifurcation
We follow the center–manifold and normal–form method of Hassard, Kazarinoff, and Wan [
32] to determine the stability and direction of the Hopf bifurcation.
Let us denote
,
, and
, and write the system near
in vector form
where
J is the Jacobian at
and
collects all nonlinear terms. Denote the components of the vector field by
, i.e.,
and expand
into a Taylor series up to cubic order:
Equivalently, using multi-index notation,
so that
.
Suppose that at
the characteristic polynomial satisfies
with
and
, so that
J has a simple pair of purely imaginary eigenvalues
with
and a third eigenvalue
. Let
and
be the right and left eigenvectors associated with
:
normalized by
, where
is the standard complex inner product. On the two-dimensional center manifold, the solution can be represented as
where
is tangent to the stable subspace. Projecting onto
gives the scalar amplitude
. The reduced dynamics for
z has the normal form
where the complex coefficients
are computed from the quadratic and cubic terms of
F and the eigenvectors
.
Let us define the symmetric bilinear form
and the symmetric trilinear form
by
where “sym” indicates symmetrization over arguments and the sums are formed from the second- and third-order partial derivatives at
. In practice one computes
B and
C directly from the Taylor coefficients
.
The leading normal-form coefficients are (Hassard’s formulas)
Here denote the inverses restricted to the stable subspace (they exist since are simple eigenvalues).
We introduce the following quantities:
The
first Lyapunov coefficient is
If the Hopf crossing is transversal, then can write
- (i)
If , the Hopf bifurcation is supercritical: a stable limit cycle is born for parameter values on the side where the equilibrium becomes unstable.
- (ii)
If , the Hopf bifurcation is subcritical: an unstable limit cycle exists on the side where the equilibrium is still stable.
For completeness, in the notation often used in Hassard [
32], one defines
where
denotes the eigenvalue branch with
. These parameters encode, respectively, the direction of the bifurcation (
), the orbital stability of periodic solutions (
), and the period variation (
).
We have the following Theorem.
Theorem 4.
The coefficients in (17) determine the qualitative behavior:
- (i)
If , the bifurcating periodic orbits exist for ; if , they exist for .
- (ii)
The periodic orbits are orbitally stable if and unstable if .
- (iii)
The period varies according to : it increases if and decreases if .
Equivalently, in terms of the first Lyapunov coefficient, the Hopf bifurcation is supercritical if and subcritical if .
5. System with Optimal Drug Dosing
The primary objective of this section is to design an optimal control strategy that reduces the population of infected T cells while simultaneously minimizing the overall cost associated with drug administration. In addition, the formulation seeks to maximize the concentration of healthy T cells. To achieve this, the following two bounded control inputs are introduced:
- (i)
: dosage of reverse transcriptase inhibitor (RTI),
- (ii)
: dosage of interleukin-2 (IL-2) therapy,
with
for
(maximal dosing corresponds to
, and no treatment to
[
27]). RTI reduces the infection rate by the factor
, where
is the efficacy. IL-2 enriches uninfected T cell and CTL responses; its effect enters linearly via
multiplying the respective endogenous expansion rates
.
The reverse transcriptase inhibitor (RTI) is assumed to reduce the infection rate by a factor of
, where
denotes the efficacy of the drug and
is the control input. Similarly, interleukin-2 (IL-2) therapy enhances the proliferation of uninfected T cells and cytotoxic T lymphocyte (CTL) responses, modeled by
, where
represents the efficacy of IL-2 and
is the corresponding control input. Incorporating these controls, the modified system (
1) is expressed as:
with initial conditions
5.1. Objective Functional
We aim to minimize drug usage while promoting immune health and suppressing infection. The cost functional is
where
are weight constants on the benefit of the cost, and
penalty multipliers. The admissible control set is
The problem here is to find
such that
subject to the state system in (
18).
5.2. Existence of the Optimal Control Pair
The existence of an optimal pair is ensure under the following conditions:
- (i)
the control set U is nonempty, closed, convex, and bounded;
- (ii)
the right-hand side of (
18) is jointly continuous in
and locally Lipschitz in
for each pair
;
- (iii)
the integrand in (
20) is convex in
u and bounded below by an
function in
t.
Here, the dynamics are affine in
u, smooth in
x, and the integrand is strictly convex in
u (due to the presence of
) and are bounded. Therefore, an optimal control pair
exists for the system (
18).
5.3. The Hamiltonian
We formulate the Hamiltonian as
where
are adjoint variables.
According to maximum principle [
33], the necessary conditions for the optimality of the system are:
- (i)
The objective functional (
20) is minimized subject to the state system (
18) with given initial data in (
2),
- (ii)
Adjoint (costate) equations satisfy: with terminal conditions , ,
- (iii)
Optimal control parameters, () minimize the Hamiltonian i.e. a.e. on .
5.4. Adjoint system
Using the second condition mentioned above, we computing the partial derivatives of (
21) to get the adjoint system. The adjoint system is obtained as
with transversality
for
. Solving this system as a boundary value problem , we get the adjoint variables,
.
5.5. Characterization of the Optimal Control Pair
Using the third condition mentioned above we get the optimal control pair. Thus, we set the gradient of the Hamiltonian with respect to the controls to zero:
The corresponding unconstrained optimal controls are
Projecting onto the admissible set
yields the bounded optimal controls:
From the above analysis, we have the following Theorem:
Theorem 5.
The objective cost function over U attains its minimum for the optimal control corresponding to the interior equilibrium . Moreover, there exist adjoint variables satisfying the following system of equations:
together with the transversality condition for . Furthermore, the optimal controls are given by
Remark 2.
The optimality system comprises the coupled state equations (18), adjoint equations (22), and control laws (24), with initial conditions for and terminal conditions for . This defines a two-point boundary value problem.
6. Numerical Results
In this section, we present computer simulation results for model system (
1) by using MatLab (R2016a) algorithms. We choose the set of values of parameters from
Table 1. Firstly, we simulate the results from the system without optimal control, then we present the simulated results from the optimal control problem.
6.1. Dynamics of the system without control
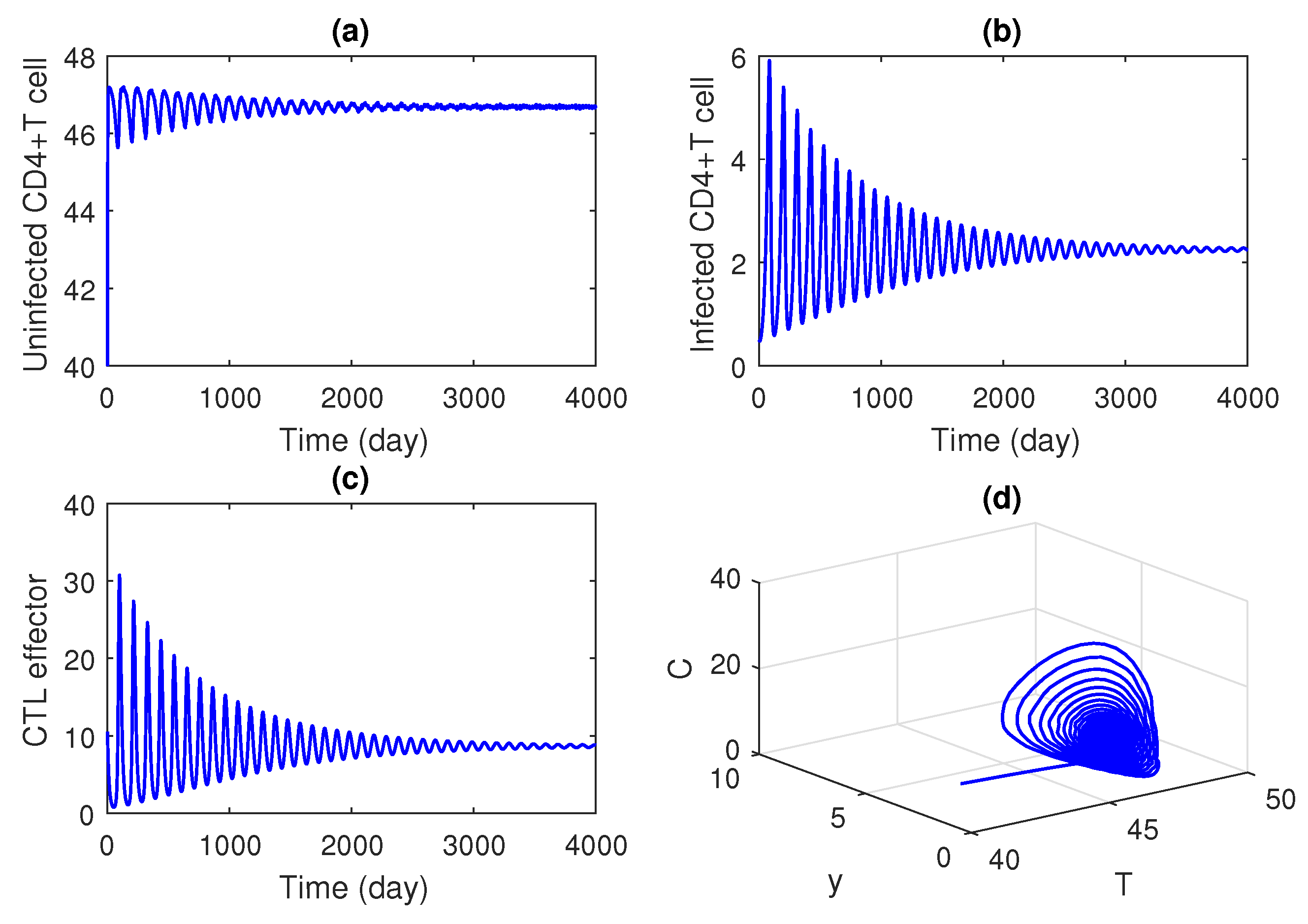
Time series solution of the system is plotted in
Figure 2, taking the values of the model parameters from
Table 1. It is observed that the coexistence equilibrium is asymptotically stable. It is seen that the system moves towards the endemic equilibrium. That means the system is stable in nature.
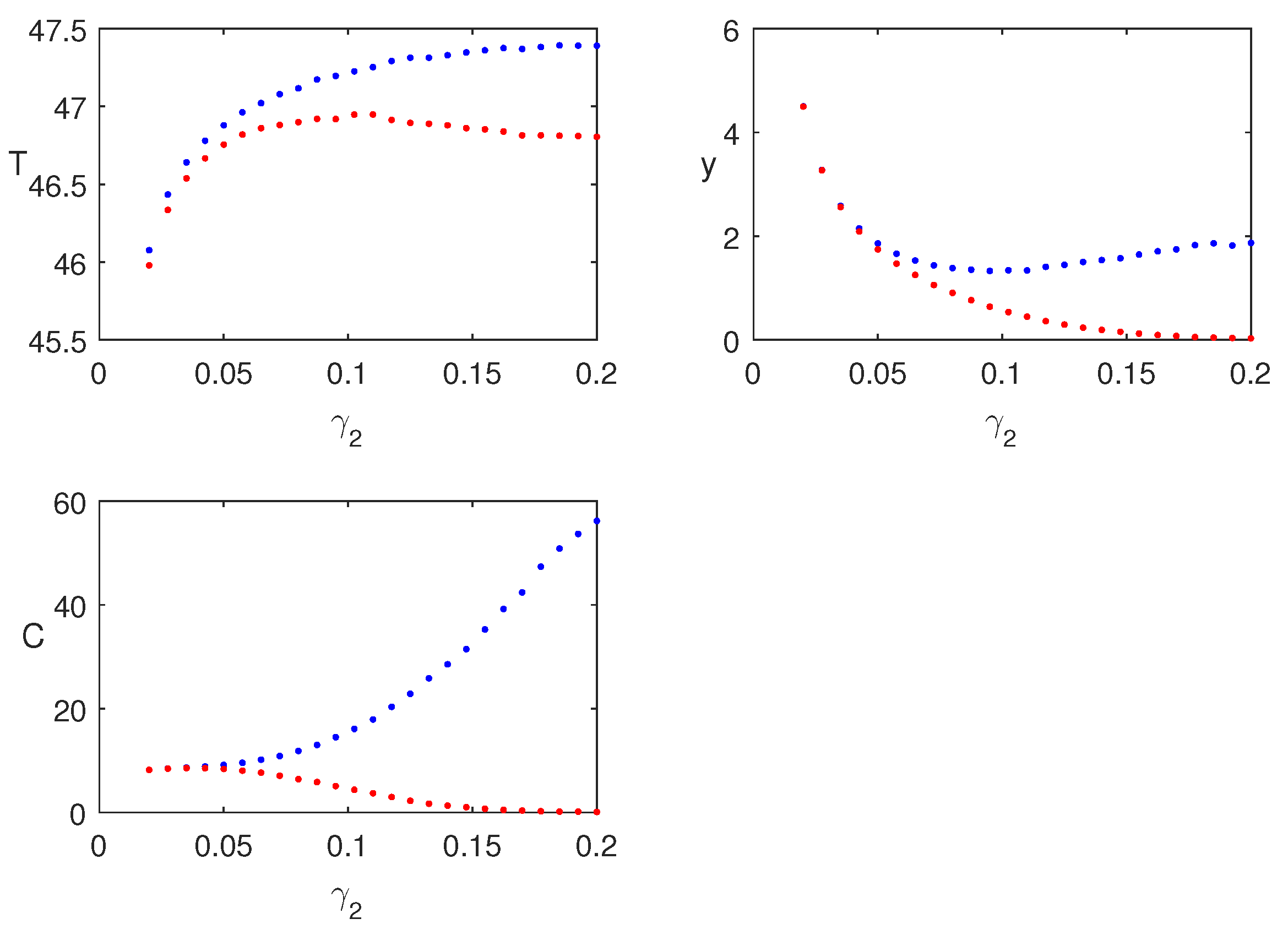
Bifurcation diagram of the system is shown in
Figure 3 by varying
. The system bifurcates into periodic solution when gamma passes the critical value,
(approx.).
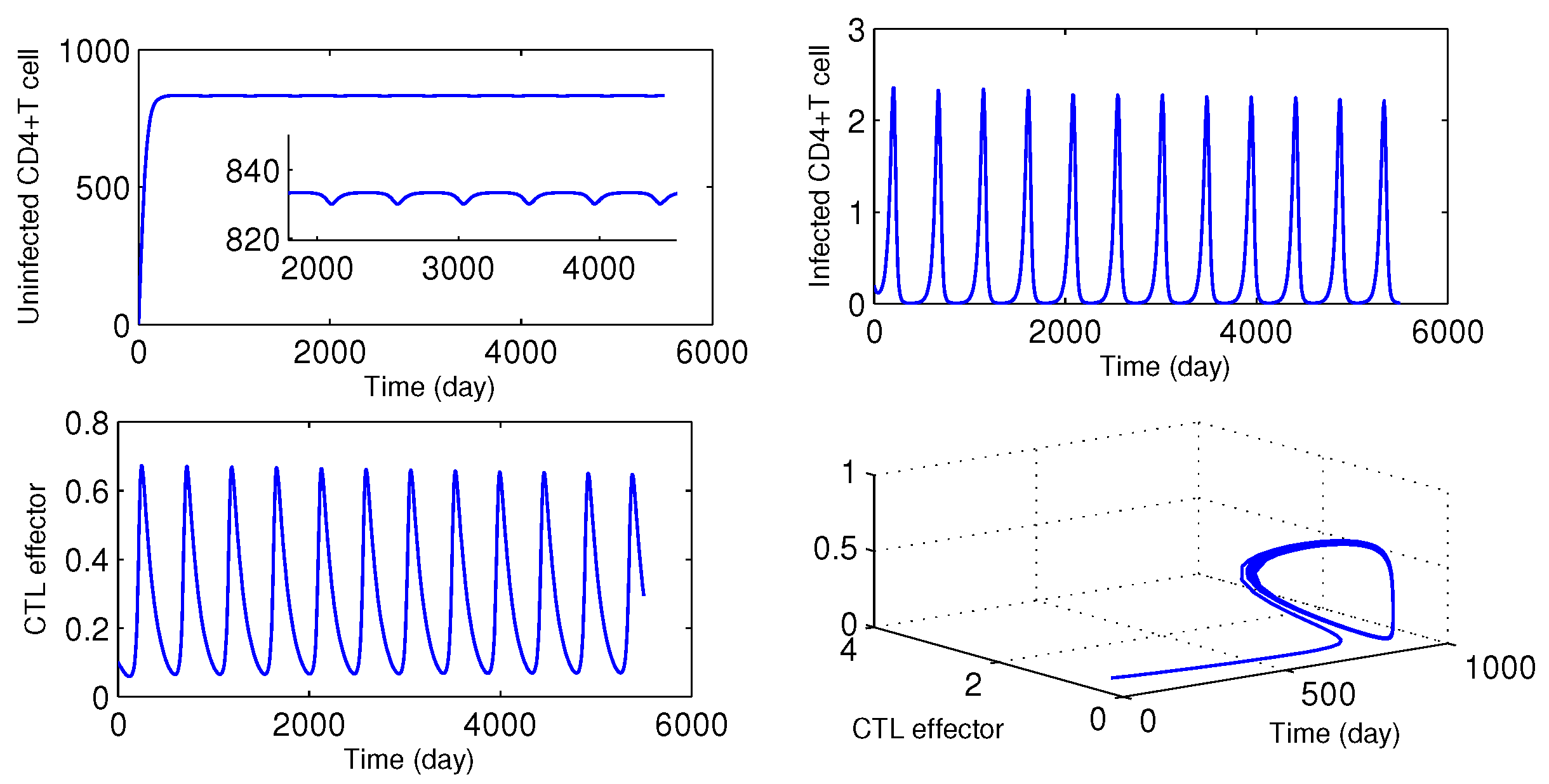
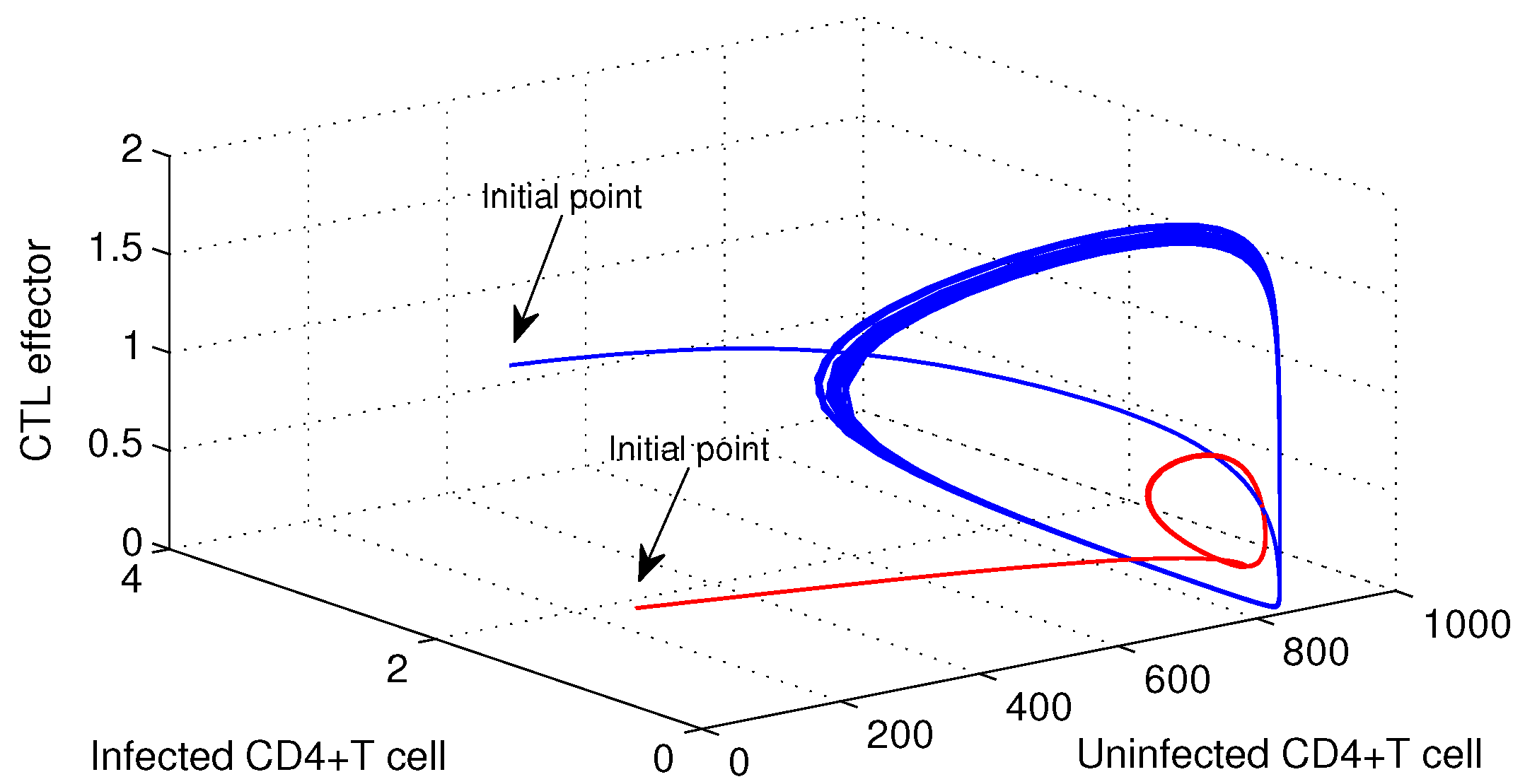
Time series solution is plotted for
, and the bifurcating periodic periodic solution is unstable (see
Figure 4). Also, for
, periodic solution is observed in
Figure 5.
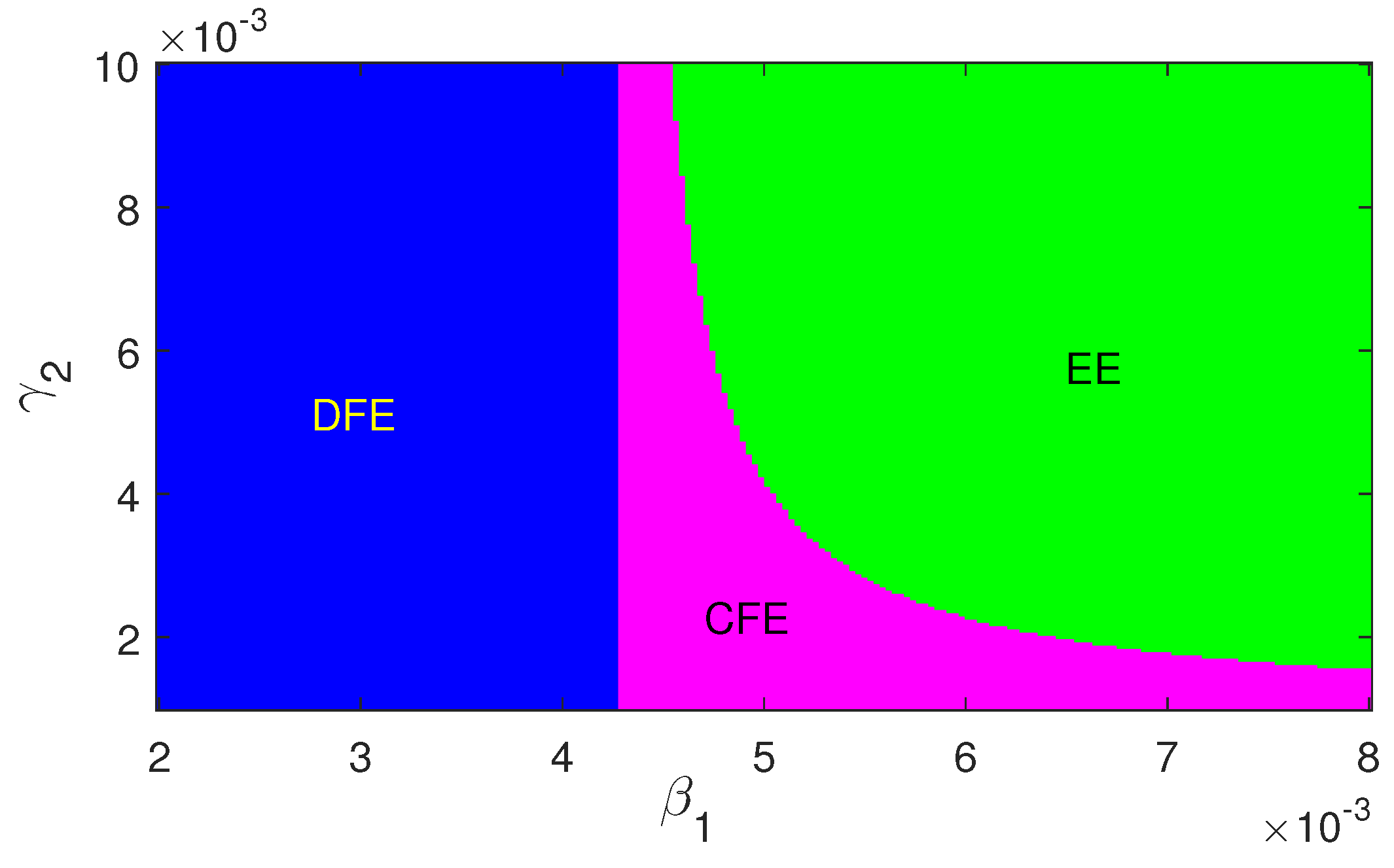
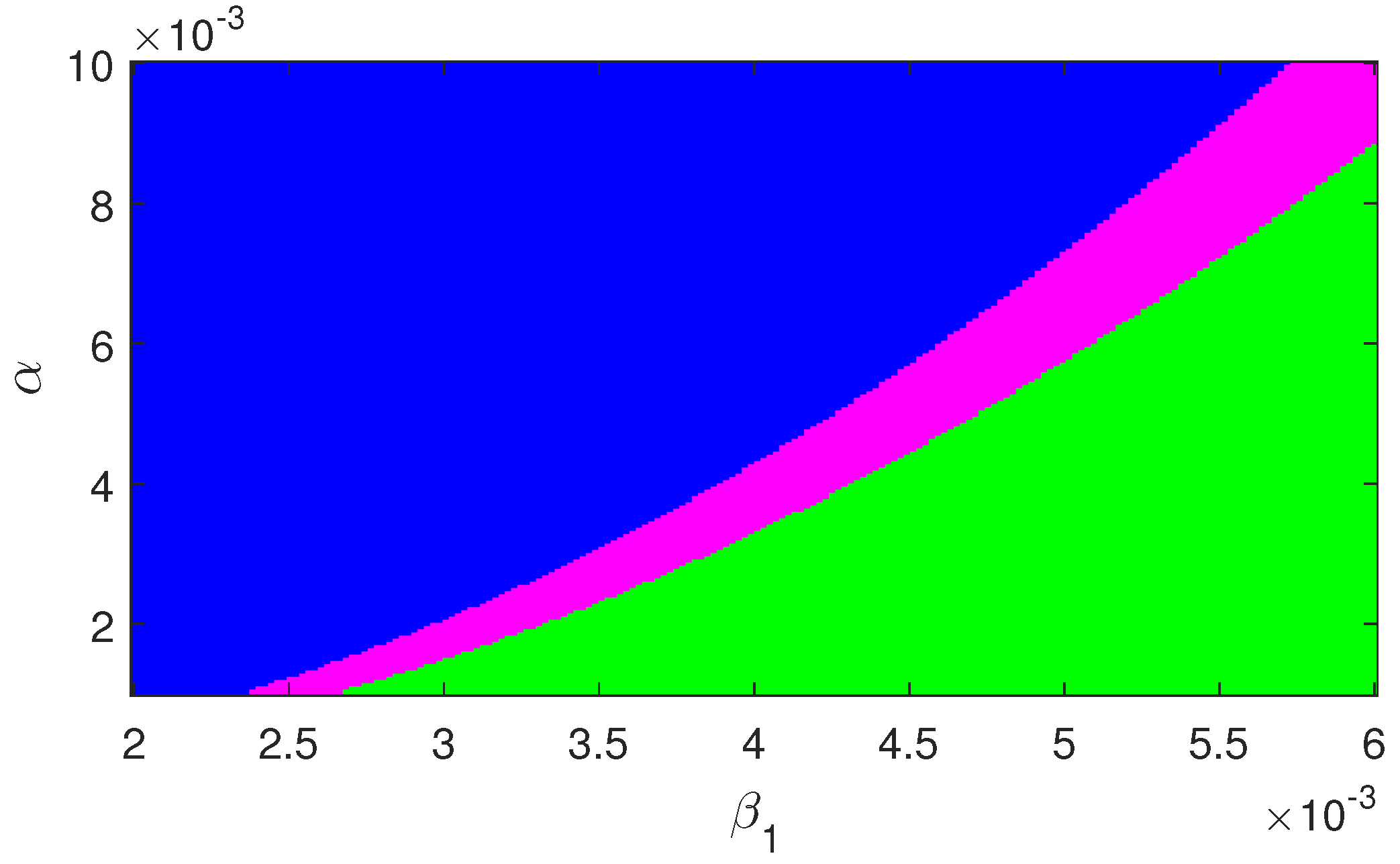
Regions of stability of the equilibrium points are plotted in
Figure 6 and
Figure 7. The figures show that for higher infection, the endemic equilibrium is feasible. Also it is stable in nature for the set of parameters used for these figures. Also,
has a stabilizing role in the dynamics.
6.2. Dynamics of the System with Optimal Control
The forward–backward sweep method is a widely used numerical technique for solving optimal control problems. It works by iteratively integrating the state equations forward in time with an initial guess for the controls, and then integrating the adjoint (costate) equations backward in time using terminal conditions. At each iteration, the controls are updated pointwise according to the optimality conditions derived from the Hamiltonian, typically with bounds enforced. This forward–backward cycle is repeated until the state, adjoint, and control trajectories converge, yielding an approximate solution to the optimal control problem.
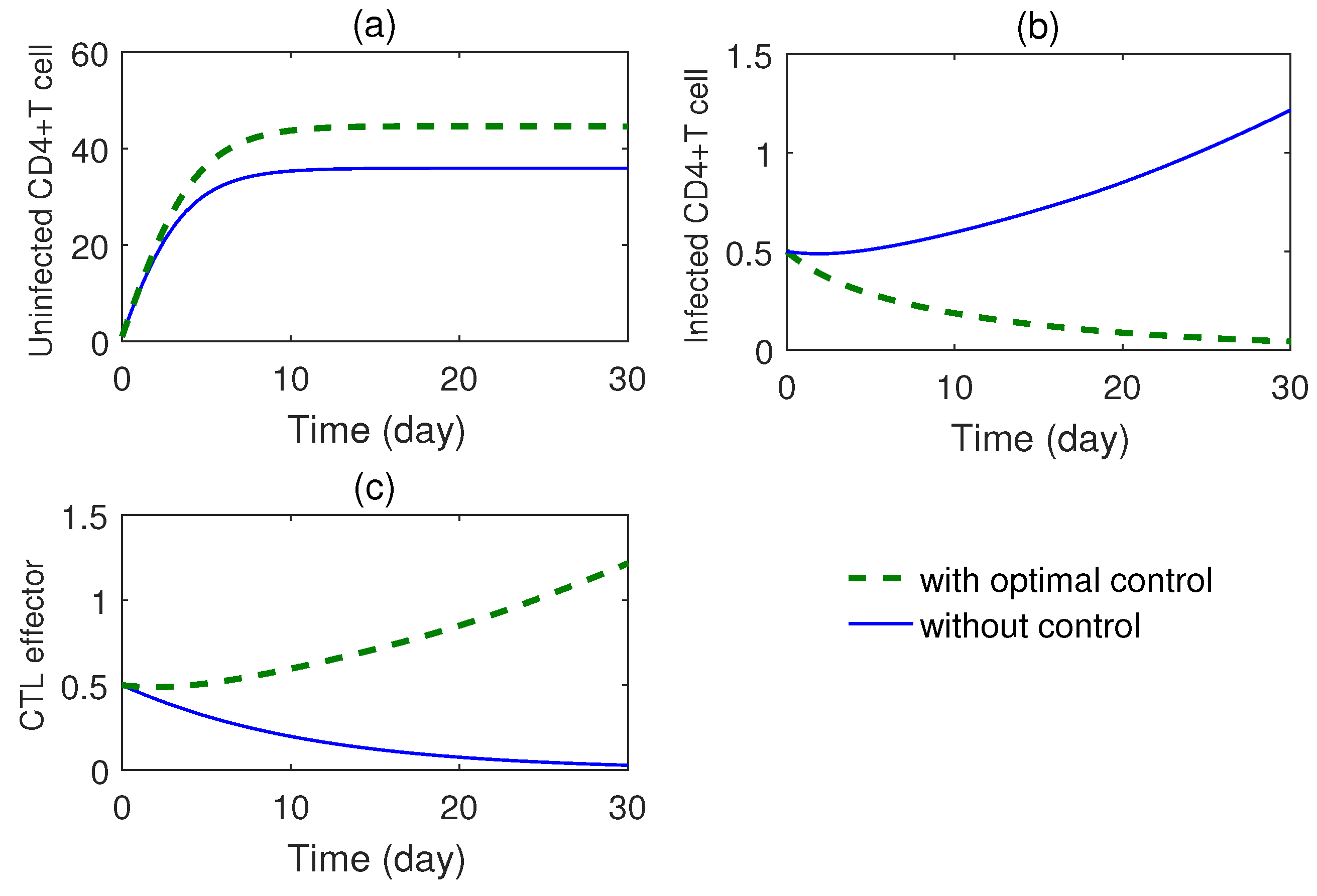
From optimal control studies (
Figure 8 and
Figure 9), several interesting results have been obtained. The CTL effector population is found to decrease in all the cases. Thus, optimal control approach will help in designing an innovative cost-effective safe therapeutic regimen of HAART and IL-2 where the uninfected cell population will be enhanced with simultaneous decrease in the infected cell population. Moreover, successful immune reconstitution can also be achieved with increase in precursor CTL population. Mathematical modeling of viral dynamics thus enables maximization of therapeutic outcome even in case of multiple therapies with specific goal of reversal of immunity impairment.
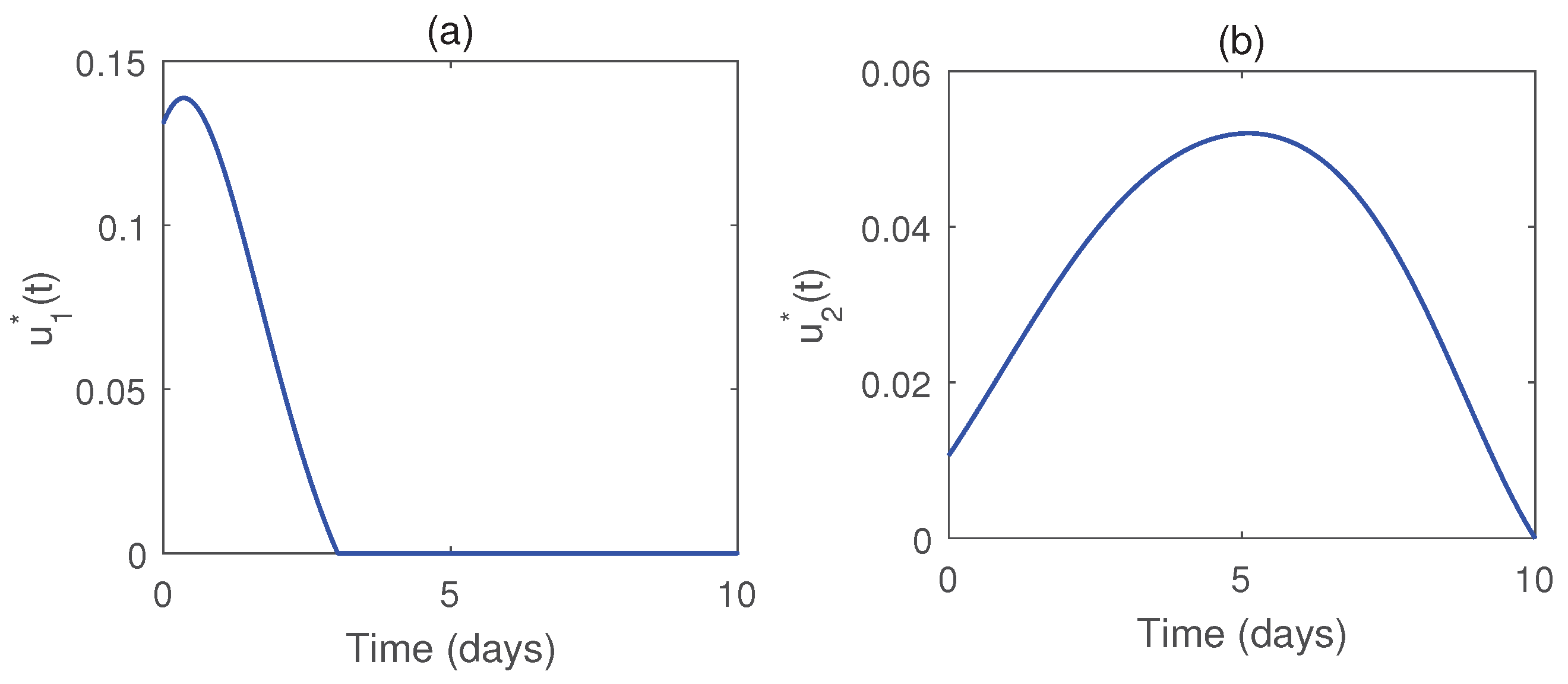
Figure 8, represents the control pair
for RTI drug for the parameter set as given in
Table 1. The RTI drug is administered at nearly full level for 20 days approximately and after that it is reduced to zero at 30 days. In
Figure 9, we see that during the treatment period, the infected T cells increase almost linearly, and the CTL responses also increases, whereas the virus producing cell linearly decreases.
7. Discussion and Conclusion
In this study, we derived a mathematical framework to elucidate the dynamics of the human immune system in response to HIV infection under the combined influence of Highly Active Antiretroviral Therapy (HAART) and Interleukin-2 (IL-2). Biologically, HIV infection is characterized by the progressive depletion of T lymphocytes, which compromises immune surveillance and predisposes patients to opportunistic infections. Our model captures these processes by incorporating viral replication, immune activation, and therapeutic intervention, thereby providing a quantitative lens through which the interplay of infection and treatment can be examined.
Analysis of the model system revealed that stability and oscillatory behavior depend critically on the infection rate parameter and immune response rate, . For certain values of these parameters, the immune system either stabilizes or destabilizes, reflecting biological scenarios of viral suppression or resurgence. Notably, bifurcating periodic solutions observed at correspond to oscillatory viral loads and T cell counts, a phenomenon consistent with clinical observations of viral blips during therapy. Using normal form theory we see that the bifurcating periodic solution is subcritical type.
From a control-theoretic perspective, we established the existence of optimal therapeutic strategies by applying the Pontryagin Minimum Principle. The derived adjoint equations and control laws formalize the biological trade-off between drug-induced viral suppression and immune stimulation. The resulting two-point boundary value problem (TPBVP) encapsulates the coupled dynamics of viral replication, immune recovery, and drug administration. Computationally, the forward–backward sweep method yielded dosing policies that balance pharmacological costs against immunological benefits, thereby reflecting the clinical objective of maximizing patient health while minimizing toxicity and expense.
Biologically, IL-2 serves as an immune activator that enhances the proliferation and survival of T cells, while HAART suppresses viral replication by targeting reverse transcriptase and other viral enzymes. Our findings demonstrate that the synergistic application of these therapies not only improves the uninfected T cell population but also reduces the reservoir of infected cells. This dual effect translates into prolonged immune competence and extended patient survival. Importantly, the optimal control strategy was shown to reduce side effects and improve cost-effectiveness, aligning with clinical goals of sustainable long-term therapy.
In conclusion, the integration of mathematical modeling with biological interpretation underscores the potential of optimal control theory in designing effective HIV treatment regimens. The combination of HAART and IL-2, when administered under an optimized dosing policy, enhances immune recovery, suppresses viral persistence, and improves life expectancy in HIV-infected individuals. This framework provides a rigorous foundation for future quantitative investigations into combination therapies and highlights the importance of mathematical biology in guiding clinical decision-making.
Data availability statement
The data sets used for this study are mentioned within the article.
Conflict of interests
The authors declare no conflict of interest.
References
- Organization, W.H. HIV and AIDS: Advances in Treatment and Research AIDS (Acquired Immunodeficiency Syndrome) can develop from HIV (Human Immunodeficiency Virus), a chronic infection that targets the immune system, if treatment is not received. Effective treatment enables people with HIV to live long, healthy lives even though there is no cure. Significant advancements in long-acting treatments and cure-focused research are highlighted by recent HIV research. Global Health Review 2025, 12, 233–240. [Google Scholar]
- Sciences, G. Breakthroughs in Long-Acting HIV Therapies Phase 2 trial results. Clinical Infectious Diseases 2025. [Google Scholar]
- Smith, J.; Patel, R. Twice-Yearly Lenacapavir Regimen with Broadly Neutralizing Antibodies. Lancet HIV 2025, 12, 101–110. [Google Scholar]
- Ndung’u, T.; Dong, K.; SenGupta, D. Ground-Breaking South African HIV Cure Trial Shows Promising Results. UKZN News / Africa Health Research Institute Results presented at the 2025 Conference on Retroviruses and Opportunistic Infections (CROI), San Francisco, 2025. [Google Scholar]
- Perelson, A.S.; Nelson, P.W. Mathematical analysis of HIV-1 dynamics in vivo. SIAM Review 1996, 39, 3–44. [Google Scholar] [CrossRef]
- Nowak, M.A.; May, R.M. Virus Dynamics: Mathematical Principles of Immunology and Virology; Oxford University Press, 2000. [Google Scholar]
- Abrams, D. Interleukin-2 therapy in patients with HIV infection. New England Journal of Medicine 2009, 361, 1548–1559. [Google Scholar] [CrossRef]
- Levy, J. Interleukin-2 and HIV therapy: the end of the story? Nature Reviews Immunology 2009, 9, 480–481. [Google Scholar]
- AlShamrani, N.H. A diffusion-based HIV model with inflammatory cytokines and adaptive immune impairment. Frontiers in Applied Mathematics and Statistics 2025, 11. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, L.; Zhang, J.; Liu, S.; Peng, Z. Dynamical modeling and data analysis of HIV infection with infection-age, CTLs immune response and delayed antibody immune response. Journal of Mathematical Biology 2025, 91. [Google Scholar] [CrossRef]
- de Carvalho, J.P.S.M. Qualitative analysis of HAART effects on HIV and SARS-CoV-2 coinfection model. Applications of Mathematics 2025, 70, 495–516. [Google Scholar] [CrossRef]
- Alsulami, I.M. On the stability, chaos and bifurcation analysis of a discrete-time chemostat model using the piecewise constant argument method. AIMS MATHEMATICS 2024, 9, 33861–33878. [Google Scholar] [CrossRef]
- Rathnayaka, N.S.; Wijerathna, J.K.; Pradeep, B.G.S.A. Stability Properties and Hopf Bifurcation of a Delayed HIV Dynamics Model with Saturation Functional Response, Absorption Effect and Cure Rate. International Journal of Analysis and Applications 2025, 23. [Google Scholar] [CrossRef]
- Hmarrass, H.; Qesmi, R. Global Stability and Hopf Bifurcation of a Delayed HIV Model with Macrophages, CD4+ T Cells with Latent Reservoirs and Immune Response. European Physical Journal Plus 2025, 140, 335. [Google Scholar] [CrossRef]
- IEEE. Mathematical Modeling and Optimal Control Strategies of HIV/AIDS with HAART. In Proceedings of the IEEE Xplore, 2023. [Google Scholar]
- Wiley. A New Mathematical Model for Assessing Therapeutic Strategies for HIV. In Wiley Online Library; 2002. [Google Scholar]
- Pontryagin, L.S.; Boltyanskii, V.G.; Gamkrelidze, R.V.; Mishchenko, E.F. The Mathematical Theory of Optimal Processes, 1962. Classic reference introducing the Pontryagin Maximum Principle.
- Campos, C.; Silva, C.J.; Torres, D.F.M. Numerical Optimal Control of HIV Transmission in Octave/MATLAB. Mathematics 2019, 7, 1–20. [Google Scholar] [CrossRef]
- Ouedraogo, B.; Zorom, M.; Gouba, E. Mathematical Modeling and Optimal Control of an HIV/AIDS Transmission Model. International Journal of Analysis and Applications 2024, 22. [Google Scholar] [CrossRef]
- Lemmon, M. Optimal Control Theory - Module 3: Maximum Principle, 2015.
- Al Basir, F.; Nisar, K.S.; Alsulami, I.M.; Chatterjee, A.N. Dynamics and optimal control of an extended SIQR model with protected human class and public awareness. The European Physical Journal Plus 2025, 140, 152. [Google Scholar] [CrossRef]
- Wikipedia contributors. Hamiltonian (control theory). Accessed. 2025. (accessed on November 2025).
- Musa, S.; John, S.; Salvation, H. Mathematical Modeling of HIV Transmission Dynamics: Incorporating Treatment and Removal as Control Strategies. International Journal of Scientific Research Accepted. 2025. [Google Scholar]
- Boukary, O.; Malicki, Z.; Elisée, G. Mathematical Modeling and Optimal Control of an HIV/AIDS Transmission Model. International Journal of Analysis and Applications 2024, 22. [Google Scholar] [CrossRef]
- Rana, P.S.; Sharma, N.; Priyadarshi, A. Mathematical Modeling and Optimal Control of a Deterministic SHATR Model of HIV/AIDS with Possibility of Rehabilitation: A Dynamic Analysis. Tamkang Journal of Mathematics 2024, 55, 267–285. [Google Scholar] [CrossRef]
- Kumar, N.; Kashif, M.; Chauhan, T.S.; Chauhan, I.S. Modeling and Control of HIV/AIDS Epidemics: A Stochastic and Optimal Control Perspective. Journal of Nonlinear Mathematical Physics 2025, 32. [Google Scholar] [CrossRef]
- Culshaw, R.; Ruan, S. A delay-differential equation model of HIV-1 infection of CD4+ T-cells. Mathematical Biosciences 2000, 165, 425–444. [Google Scholar] [CrossRef]
- Wodarz, D.; Nowak, M. Specific therapy regimes could lead to long term immunological control to HIV. Proceedings of the National Academy of Sciences USA 1999, 96, 14464–14469. [Google Scholar] [CrossRef]
- Brikhoff, G.; Rota, G. Ordinary Differential Equations; Ginn: Boston, 1982. [Google Scholar]
- Diekmann, O.; Heesterbeek, J.A.P.; Metz, J.A. On the definition and the computation of the basic reproduction ratio R0 in models for infectious diseases in heterogeneous populations. Journal of Mathematical Biology 1990, 28, 365–382. [Google Scholar] [CrossRef]
- Van den Driessche, P.; Watmough, J. Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Mathematical Biosciences 2002, 180, 29–48. [Google Scholar] [CrossRef]
- Hassard, B.D.; Kazarinoff, N.D.; Wan, Y.H. Theory and Applications of Hopf Bifurcation; Cambridge University Press: Cambridge, 1981. [Google Scholar]
- Fleming, W.H.; Rishel, R.W. Deterministic and Stochastic Optimal Control. In Applications of Mathematics; Springer: New York, 1975; Vol. 1. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).