Submitted:
22 January 2026
Posted:
23 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
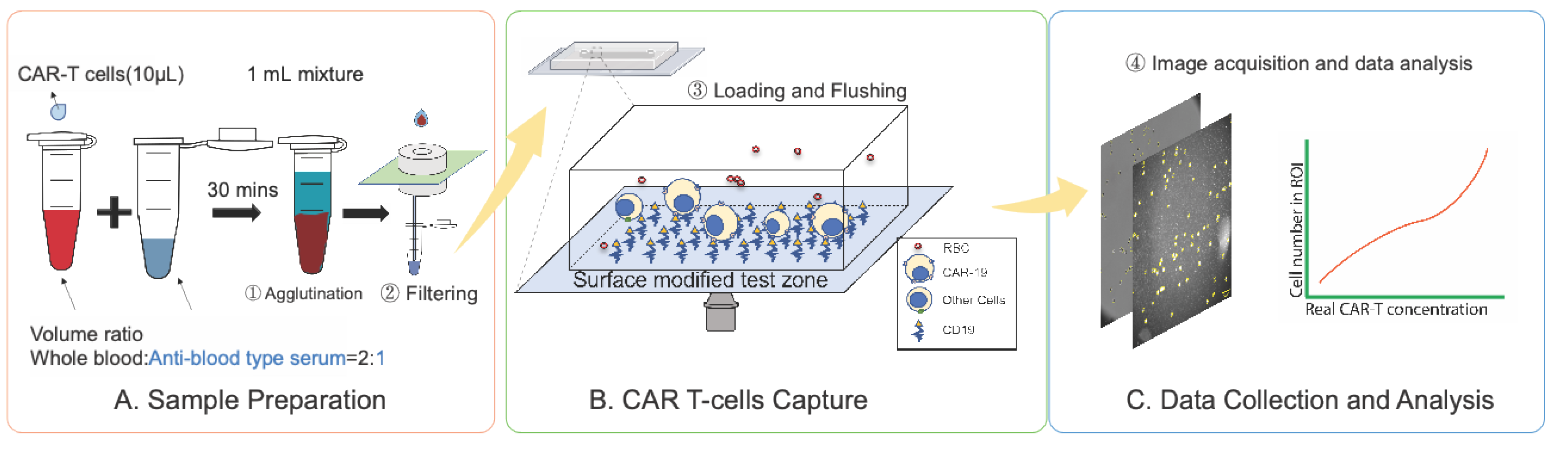
2.1. Overview of the CAR T-Cell Detection Workflow
2.2. Sample Delivery and Detection System
2.3. Microfluidic Chip Fabrication
2.4. Surface Chemistry
2.5. Sample Preparation
2.6. Sample Loading and Processing
2.7. Optical Image Acquisition
2.8. Cell Culture
2.9. Cell Counting with a Cell Counter
2.10. Immunostaining
2.11. Flow Cytometry
2.12. Image Processing
3. Results
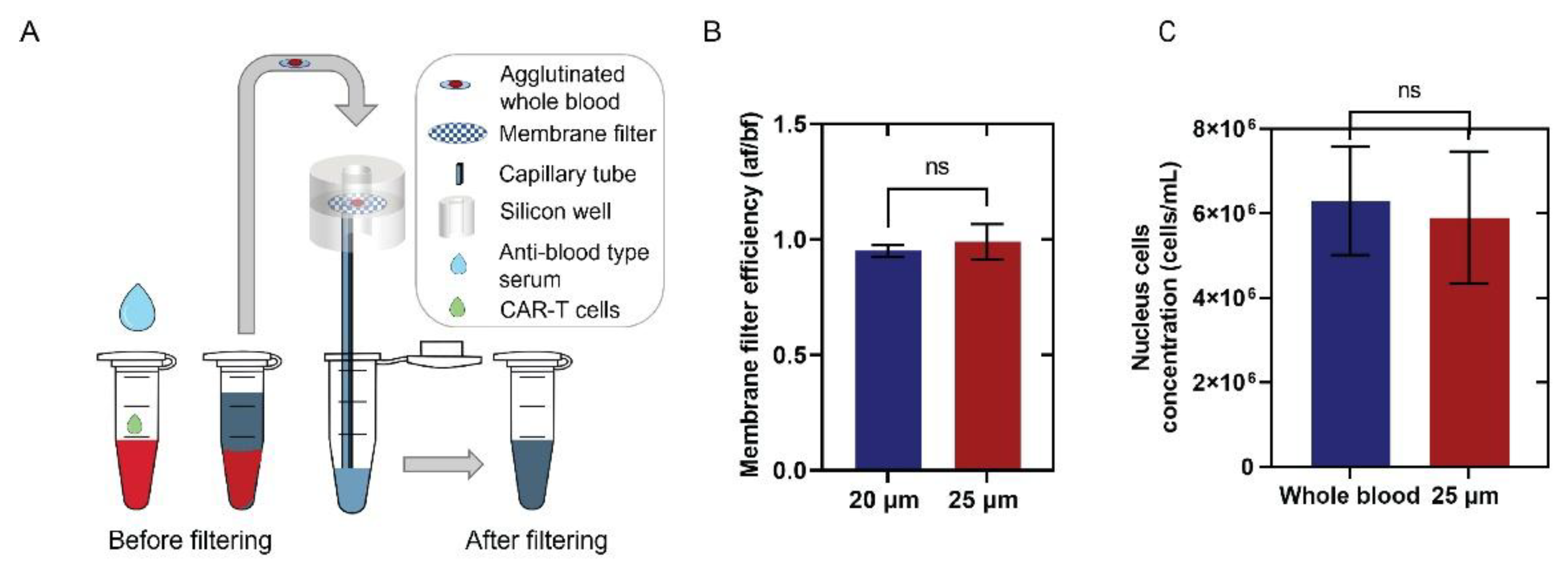
Centrifuge-Free, Agglutination-Based RBCs Removal
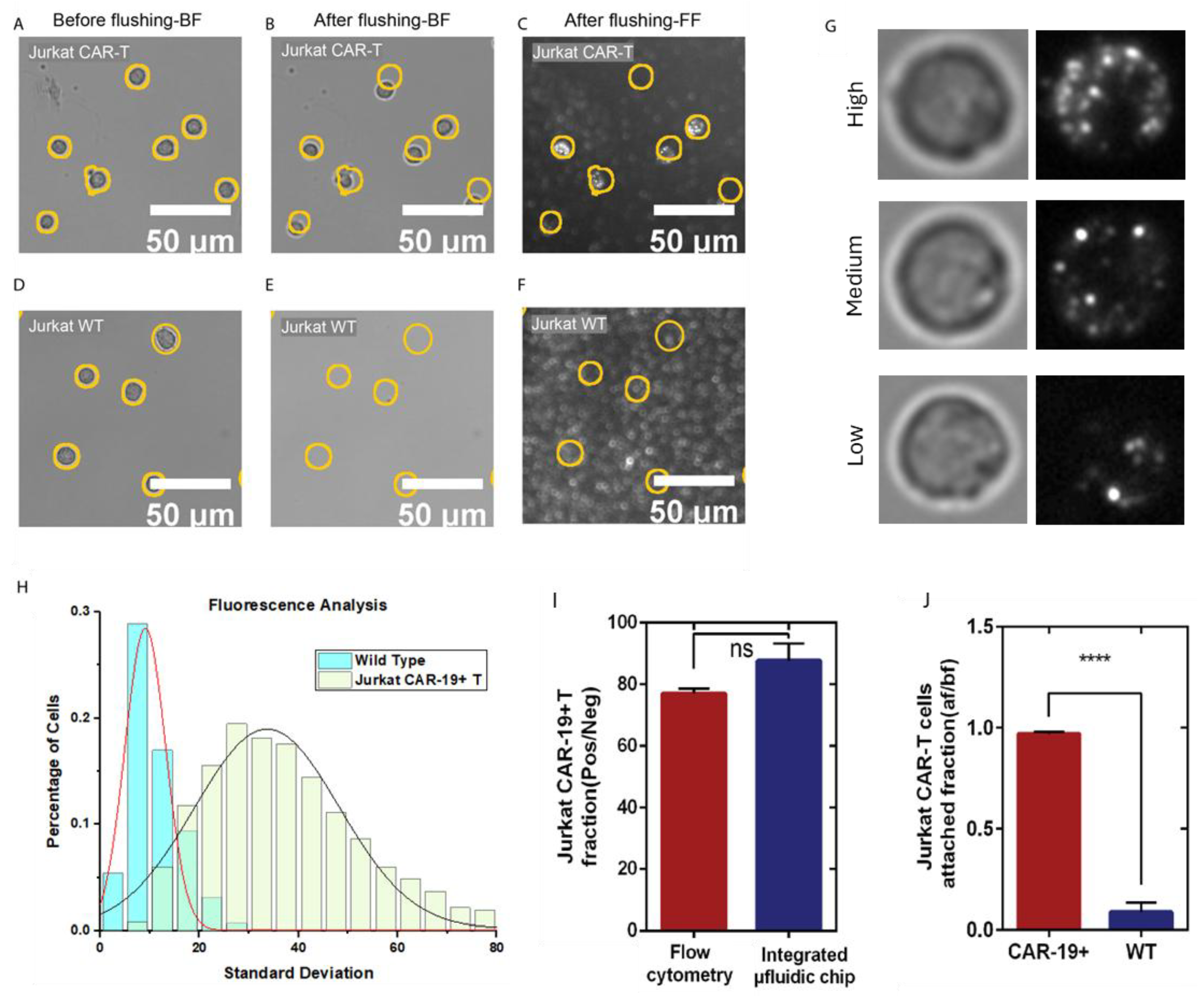
CAR T-Cell Capture, Detection, and Validation
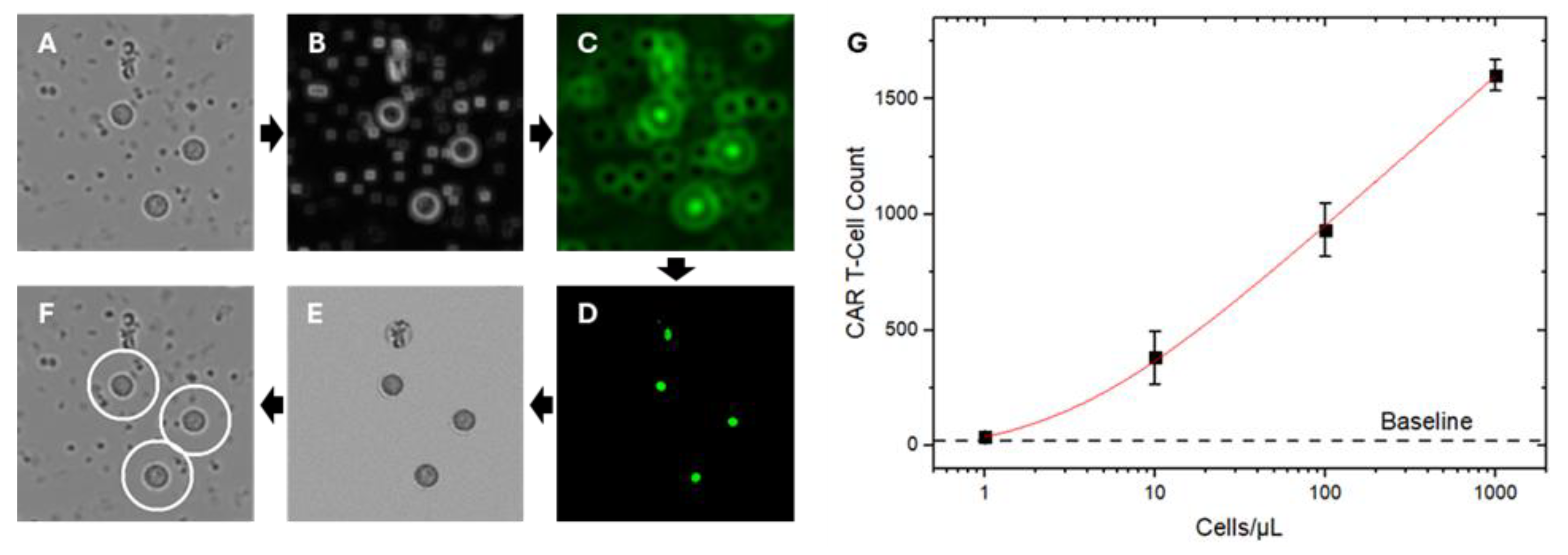
CAR T-Cell Quantification in Whole Blood
4. Discussion
6. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kochenderfer, J.N.; Somerville, R.P.T.; Lu, T.; Shi, V.; Bot, A.; Rossi, J.; Xue, A.; Goff, S.L.; Yang, J.C.; Sherry, R.M.; et al. Lymphoma Remissions Caused by Anti-CD19 Chimeric Antigen Receptor T Cells Are Associated With High Serum Interleukin-15 Levels. J Clin Oncol 2017, 35, 1803–1813. [Google Scholar] [CrossRef]
- Locke, F.L.; Neelapu, S.S.; Bartlett, N.L.; Siddiqi, T.; Chavez, J.C.; Hosing, C.M.; Ghobadi, A.; Budde, L.E.; Bot, A.; Rossi, J.M.; et al. Phase 1 Results of ZUMA-1: A Multicenter Study of KTE-C19 Anti-CD19 CAR T Cell Therapy in Refractory Aggressive Lymphoma. Mol Ther 2017, 25, 285–295. [Google Scholar] [CrossRef] [PubMed]
- Chong, E.A.; Ruella, M.; Schuster, S.J.; Lymphoma Program Investigators at the University of; P. Five-Year Outcomes for Refractory B-Cell Lymphomas with CAR T-Cell Therapy. N Engl J Med 2021, 384, 673–674. [Google Scholar] [CrossRef] [PubMed]
- Ghafouri, S.; Timmerman, J.; Larson, S.; Mead, M.D. Axicabtagene Ciloleucel CAR T-cell therapy for relapsed/refractory secondary CNS non-Hodgkin lymphoma: comparable outcomes and toxicities, but shorter remissions may warrant alternative consolidative strategies? Bone Marrow Transplant 2020. [Google Scholar] [CrossRef] [PubMed]
- Neelapu, S.S.; Tummala, S.; Kebriaei, P.; Wierda, W.; Gutierrez, C.; Locke, F.L.; Komanduri, K.V.; Lin, Y.; Jain, N.; Daver, N.; et al. Chimeric antigen receptor T-cell therapy — assessment and management of toxicities. Nature Reviews Clinical Oncology 2018, 15, 47–62. [Google Scholar] [CrossRef]
- Choi, M.Y.; Widhopf, G.F., 2nd; Ghia, E.M.; Kidwell, R.L.; Hasan, M.K.; Yu, J.; Rassenti, L.Z.; Chen, L.; Chen, Y.; Pittman, E.; et al. Phase I Trial: Cirmtuzumab Inhibits ROR1 Signaling and Stemness Signatures in Patients with Chronic Lymphocytic Leukemia. Cell Stem Cell 2018, 22, 951–959 e953. [Google Scholar] [CrossRef] [PubMed]
- Jena, B.; Maiti, S.; Huls, H.; Singh, H.; Lee, D.A.; Champlin, R.E.; Cooper, L.J. Chimeric antigen receptor (CAR)-specific monoclonal antibody to detect CD19-specific T cells in clinical trials. PLoS One 2013, 8, e57838. [Google Scholar] [CrossRef]
- Cavallo, M.C.; Cavazza, M.; Bonifazi, F.; Casadei, B.; Cutini, I.; Tonietti, B.; Saccardi, R.; Zinzani, P.; Jommi, C. Cost of implementing CAR-T activity and managing CAR-T patients: an exploratory study. BMC Health Serv Res 2024, 24, 121. [Google Scholar] [CrossRef]
- Kim, H.; Zhbanov, A.; Yang, S. Microfluidic Systems for Blood and Blood Cell Characterization. Biosensors (Basel) 2022, 13. [Google Scholar] [CrossRef] [PubMed]
- Lu, N.; Tay, H.M.; Petchakup, C.; He, L.; Gong, L.; Maw, K.K.; Leong, S.Y.; Lok, W.W.; Ong, H.B.; Guo, R.; et al. Label-free microfluidic cell sorting and detection for rapid blood analysis. Lab Chip 2023, 23, 1226–1257. [Google Scholar] [CrossRef]
- Yuan, H.; Miao, Z.; Wan, C.; Wang, J.; Liu, J.; Li, Y.; Xiao, Y.; Chen, P.; Liu, B.F. Recent advances in centrifugal microfluidics for point-of-care testing. Lab Chip 2025, 25, 1015–1046. [Google Scholar] [CrossRef]
- Kant, K.; Shahbazi, M.A.; Dave, V.P.; Ngo, T.A.; Chidambara, V.A.; Than, L.Q.; Bang, D.D.; Wolff, A. Microfluidic devices for sample preparation and rapid detection of foodborne pathogens. Biotechnol Adv 2018, 36, 1003–1024. [Google Scholar] [CrossRef]
- Aran, K.; Sasso, L.A.; Kamdar, N.; Zahn, J.D. Irreversible, direct bonding of nanoporous polymer membranes to PDMS or glass microdevices. Lab on a Chip 2010, 10, 548. [Google Scholar] [CrossRef]
- Wu, D.; Zhao, B.; Dai, Z.; Qin, J.; Lin, B. Grafting epoxy-modified hydrophilic polymers onto poly(dimethylsiloxane) microfluidic chip to resist nonspecific protein adsorption. Lab Chip 2006, 6, 942–947. [Google Scholar] [CrossRef]
- Friend, J.; Yeo, L. Fabrication of microfluidic devices using polydimethylsiloxane. Biomicrofluidics 2010, 4, 026502. [Google Scholar] [CrossRef]
- Ghanbari, A.; Attar, M.M. A study on the anticorrosion performance of epoxy nanocomposite coatings containing epoxy-silane treated nano-silica on mild steel substrate. Journal of Industrial and Engineering Chemistry 2015, 23, 145–153. [Google Scholar] [CrossRef]
- Mrad, M.; Montemor, M.F.; Dhouibi, L.; Triki, E. Deposition of hybrid 3-GPTMS's film on AA2024-T3: Dependence of film morphology and protectiveness performance on coating conditions. Progress in Organic Coatings 2012, 73, 264–271. [Google Scholar] [CrossRef]
- Ojea-Jiménez, I.; Urbán, P.; Barahona, F.; Pedroni, M.; Capomaccio, R.; Ceccone, G.; Kinsner-Ovaskainen, A.; Rossi, F.; Gilliland, D. Highly Flexible Platform for Tuning Surface Properties of Silica Nanoparticles and Monitoring Their Biological Interaction. ACS Applied Materials & Interfaces 2016, 8, 4838–4850. [Google Scholar] [CrossRef]
- Jing, W.; Wang, Y.; Chen, C.; Zhang, F.; Yang, Y.; Ma, G.; Yang, E.H.; Snozek, C.L.N.; Tao, N.; Wang, S. Gradient-Based Rapid Digital Immunoassay for High-Sensitivity Cardiac Troponin T (hs-cTnT) Detection in 1 muL Plasma. ACS Sens 2020. [Google Scholar] [CrossRef]
- Connell, L.S.; Romer, F.; Suarez, M.; Valliant, E.M.; Zhang, Z.; Lee, P.D.; Smith, M.E.; Hanna, J.V.; Jones, J.R. Chemical characterisation and fabrication of chitosan-silica hybrid scaffolds with 3-glycidoxypropyl trimethoxysilane. J Mater Chem B 2014, 2, 668–680. [Google Scholar] [CrossRef] [PubMed]
- Fan, H.; Lu, J.; Guo, Y.; Li, D.; Zhang, Z.M.; Tsai, Y.H.; Pi, W.C.; Ahn, J.H.; Gong, W.; Xiang, Y.; et al. BAHCC1 binds H3K27me3 via a conserved BAH module to mediate gene silencing and oncogenesis. Nat Genet 2020, 52, 1384–1396. [Google Scholar] [CrossRef]
- Cheng, R.; Zhang, F.; Li, M.; Wo, X.; Su, Y.-W.; Wang, W. Influence of Fixation and Permeabilization on the Mass Density of Single Cells: A Surface Plasmon Resonance Imaging Study. Frontiers in Chemistry 2019, 7. [Google Scholar] [CrossRef] [PubMed]
- Wainwright, M. Dyes, trypanosomiasis and DNA: a historical and critical review. Biotechnic & Histochemistry 2010, 85, 341–354. [Google Scholar] [CrossRef] [PubMed]
- Burry, R.W. Controls for immunocytochemistry: an update. [CrossRef] [PubMed]
- Finak, G.; Langweiler, M.; Jaimes, M.; Malek, M.; Taghiyar, J.; Korin, Y.; Raddassi, K.; Devine, L.; Obermoser, G.; Pekalski, M.L.; et al. Standardizing Flow Cytometry Immunophenotyping Analysis from the Human ImmunoPhenotyping Consortium. Sci Rep 2016, 6, 20686. [Google Scholar] [CrossRef]
- McKinnon, K.M. Flow Cytometry: An Overview. Curr Protoc Immunol 2018, 120, 5 1 1–5 1 11. [Google Scholar] [CrossRef] [PubMed]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: an open-source platform for biological-image analysis. Nat Methods 2012, 9, 676–682. [Google Scholar] [CrossRef]
- Berg, S.; Kutra, D.; Kroeger, T.; Straehle, C.N.; Kausler, B.X.; Haubold, C.; Schiegg, M.; Ales, J.; Beier, T.; Rudy, M.; et al. ilastik: interactive machine learning for (bio)image analysis. Nature Methods 2019, 16, 1226–1232. [Google Scholar] [CrossRef]
- Zhang, C.; Huber, F.; Knop, M.; Hamprecht, F.A. Yeast cell detection and segmentation in bright field microscopy. Proceedings of the 2014 IEEE 11th International Symposium on Biomedical Imaging (ISBI) 2014, 2014, 1267–1270. [Google Scholar]
- Laura, D. Blood and the cells it contains. In Blood Groups and Red Cell Antigens [Internet]; National Center for Biotechnology Information (US), 2005. [Google Scholar]
- Tatsumi, N.; Tsuda, I; Fau - Fukumori, T.; Fukumori, T; Fau - Hino, M.; Hino, M; Fau - Takubo, T.; Fau - Kondo, Takubo T; Kondo, H.H. A centrifuge-less plasma separation method from whole blood anticoagulated with EDTA-2K for the use of clinical laboratory tests.
- Böyum, A. Isolation of mononuclear cells and granulocytes from human blood. Isolation of monuclear cells by one centrifugation, and of granulocytes by combining centrifugation and sedimentation at 1 g. Scand J Clin Lab Invest Suppl 1968, 97, 77–89. [Google Scholar]
- Jia, Y.; Xu, H.; Li, Y.; Wei, C.; Guo, R.; Wang, F.; Wu, Y.; Liu, J.; Jia, J.; Yan, J.; et al. A Modified Ficoll-Paque Gradient Method for Isolating Mononuclear Cells from the Peripheral and Umbilical Cord Blood of Humans for Biobanks and Clinical Laboratories. Biopreservation and Biobanking 2017, 16, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Jaatinen, T.; Laine, J. Isolation of mononuclear cells from human cord blood by Ficoll-Paque density gradient. In Curr Protoc Stem Cell Biol 2007, Chapter 2; Unit 2A.1. [CrossRef]
- Bossuyt, X.; Marti, G.E.; Fleisher, T.A. Comparative analysis of whole blood lysis methods for flow cytometry. Cytometry 1997, 30, 124–133. [Google Scholar] [CrossRef]
- Horn, P.; Bork, S.; Wagner, W. Standardized isolation of human mesenchymal stromal cells with red blood cell lysis. Methods Mol Biol 2011, 698, 23–35. [Google Scholar] [CrossRef]
- Avci, F.Y.; Li, X.; Tsuji, M.; Kasper, D.L. Isolation of carbohydrate-specific CD4+ T cell clones from mice after stimulation by two model glycoconjugate vaccines. Nature Protocols 2012, 7, 2180–2192. [Google Scholar] [CrossRef]
- Brown, W.E.; Hu, J.C.; Athanasiou, K.A. Ammonium-Chloride-Potassium Lysing Buffer Treatment of Fully Differentiated Cells Increases Cell Purity and Resulting Neotissue Functional Properties. Tissue Eng Part C Methods 2016, 22, 895–903. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Chang, Y.; Deng, Q.; You, H. A simple and rapid method for blood plasma separation driven by capillary force with an application in protein detection. Anal Methods 2020, 12, 2560–2570. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Mauk, M.; Gross, R.; Bushman, F.D.; Edelstein, P.H.; Collman, R.G.; Bau, H.H. Membrane-based, sedimentation-assisted plasma separator for point-of-care applications. Anal Chem 2013, 85, 10463–10470. [Google Scholar] [CrossRef]
- Nabatiyan, A.; Parpia, Z.A.; Elghanian, R.; Kelso, D.M. Membrane-based plasma collection device for point-of-care diagnosis of HIV. J Virol Methods 2011, 173, 37–42. [Google Scholar] [CrossRef]
- Hou, H.W. Microfluidic Devices for Blood Fractionation. Micromachines 2011, 2. [Google Scholar] [CrossRef]
- Hauser, J.; Lenk, G.; Hansson, J.; Beck, O.; Stemme, G.; Roxhed, N. High-Yield Passive Plasma Filtration from Human Finger Prick Blood. Anal Chem 2018, 90, 13393–13399. [Google Scholar] [CrossRef] [PubMed]
- Ulum, M.F.; Maylina, L.; Noviana, D.; Wicaksono, D.H.B. EDTA-treated cotton-thread microfluidic device used for one-step whole blood plasma separation and assay. Lab on a Chip 16 2016, 1492–1504. [Google Scholar] [CrossRef]
- Gascoyne, P.R.; Noshari, J.; Anderson, T.J.; Becker, F.F. Isolation of rare cells from cell mixtures by dielectrophoresis. Electrophoresis 2009, 30, 1388–1398. [Google Scholar] [CrossRef]
- Kim, B.; Oh, S.; You, D.; Choi, S. Microfluidic Pipette Tip for High-Purity and High-Throughput Blood Plasma Separation from Whole Blood. Anal Chem 2017, 89, 1439–1444. [Google Scholar] [CrossRef] [PubMed]
- Kuan, D.-H.; Wu, C.-C.; Su, W.-Y.; Huang, N.-T. A Microfluidic Device for Simultaneous Extraction of Plasma, Red Blood Cells, and On-Chip White Blood Cell Trapping. Scientific Reports 2018, 8, 15345. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, Y.; Hata, S.; Yasuda, T. Blood plasma separation and extraction from a minute amount of blood using dielectrophoretic and capillary forces. Sensors and Actuators B: Chemical 2010, 145, 561–569. [Google Scholar] [CrossRef]
- Li, H.; Steckl, A.J. Paper Microfluidics for Point-of-Care Blood-Based Analysis and Diagnostics. Anal Chem 2019, 91, 352–371. [Google Scholar] [CrossRef] [PubMed]
- Carstensen, E.L.; Kelly, P.; Church, C.C.; Brayman, A.A.; Child, S.Z.; Raeman, C.H.; Schery, L. Lysis of erythrocytes by exposure to CW ultrasound. Ultrasound Med Biol 1993, 19, 147–165. [Google Scholar] [CrossRef]
- Davis, J.A. Deterministic hydrodynamics: Taking blood apart. Proceedings of the National Academy of Sciences 2006, 103. [Google Scholar] [CrossRef]
- Petchakup, C.; Tay, H.M.; Li, K.H.H.; Hou, H.W. integrated inertial-impedance cytometry for rapid label-free leukocyte isolation and profiling of neutrophil extracellular traps(NETs). Lab Chip 2019, 19, 1736–1746. [Google Scholar] [CrossRef] [PubMed]
- Xiang, N.; Wang, J.; Li, Q.; Han, Y.; Huang, D.; Ni, Z. Precise Size-Based Cell Separation via the Coupling of Inertial Microfluidics and Deterministic Lateral Displacement. Anal Chem 2019, 91, 10328–10334. [Google Scholar] [CrossRef]
- Wheater, P.R.; Burkitt, H.G.; Daniels, V.G.; Deakin, P.J. Functional Histology: A Text and Colour Atlas; Churchill Livingstone, 1979. [Google Scholar]
- Mayer, T.; Jagla B Fau - Wyler, M.R.; Wyler Mr Fau - Kelly, P.D.; Kelly Pd Fau - Aulner, N.; Aulner N Fau - Beard, M.; Beard M Fau - Barger, G.; Barger G Fau - Többen, U.; Többen U Fau - Smith, D.H.; Smith Dh Fau - Brandén, L.; Brandén L Fau - Rothman, J.E.; et al. Cell-based assays using primary endothelial cells to study multiple steps in inflammation.
- Ashcroft, R.G.; Lopez, P.A. Commercial high speed machines open new opportunities in high throughput flow cytometry (HTFC). [CrossRef]
- Maino, V.C.; Picker, L.J. Identification of functional subsets by flow cytometry: intracellular detection of cytokine expression. [CrossRef]
- Kummrow, A.; Frankowski, M; Fau - Bock, N.; Bock, N; Fau - Werner, C.; Werner C; Fau - Dziekan, T.; Dziekan, T; Fau - Neukammer, J.; Neukammer, J. Quantitative assessment of cell viability based on flow cytometry and microscopy. [CrossRef]
- Christiansen, E.M.; Yang, S.J.; Ando, D.M.; Javaherian, A.; Skibinski, G.; Lipnick, S.; Mount, E.; O’Neil, A.; Shah, K.; Lee, A.K.; Goyal, P.; Fedus, W.; Poplin, R.; Esteva, A.; Berndl, M.; Rubin, L.L.; Nelson, P.; Finkbeiner, S. In Silico Labeling: Predicting Fluorescent Labels in Unlabeled Images. Cell 2018, Cell, 792–803 e719. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Kim, S.; Lim, H.; Chung, A.J. Expanding CAR-T cell immunotherapy horizons through microfluidics. Lab on a Chip 2024, 24, 1088–1120. [Google Scholar] [CrossRef]
- Zeming, K.K.; Quek, K.Y.; Sin, W.-X.; Teo, D.B.L.; Cheung, K.-W.; Goh, C.R.; Kairi, F.; Lee, E.; Lim, F.L.W.I.; Seng, M.S.-F.; et al. Cell trajectory modulation: rapid microfluidic biophysical profiling of CAR T cell functional phenotypes. Nature Communications 2025, 16, 4775. [Google Scholar] [CrossRef]
- Ma, C.; Wang, H.; Liu, L.; Chen, R.; Mukherjee, N.; Tong, J.; Kazmi, S.; Fang, X.; Witkowski, M.T.; Aifantis, I.; et al. Bioengineered immunocompetent preclinical trial-on-chip tool enables screening of CAR T cell therapy for leukaemia. Nature Biomedical Engineering 2025. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro, L.B.; Coleman, V.A.; Hindson, C.M.; Herrmann, J.; Hindson, B.J.; Bhat, S.; Emslie, K.R. Evaluation of a Droplet Digital Polymerase Chain Reaction Format for DNA Copy Number Quantification. Analytical Chemistry 2012, 84, 1003–1011. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



