Submitted:
20 January 2026
Posted:
21 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Intrathecal Injection
2.3. Measurement of Hind Paw Thickness
2.4. Mechanical Pain Threshold Detection
2.5. Transcutaneous Electrical Nerve Stimulation Applications
2.6. Open Field Test
2.7. In Vivo Ca2+ Imaging of L4 DRG
2.8. Optogenetics
2.9. Immunofluorescence
2.10. Quantification of Calcium Imaging
2.11. Statistical Analysis
3. Results
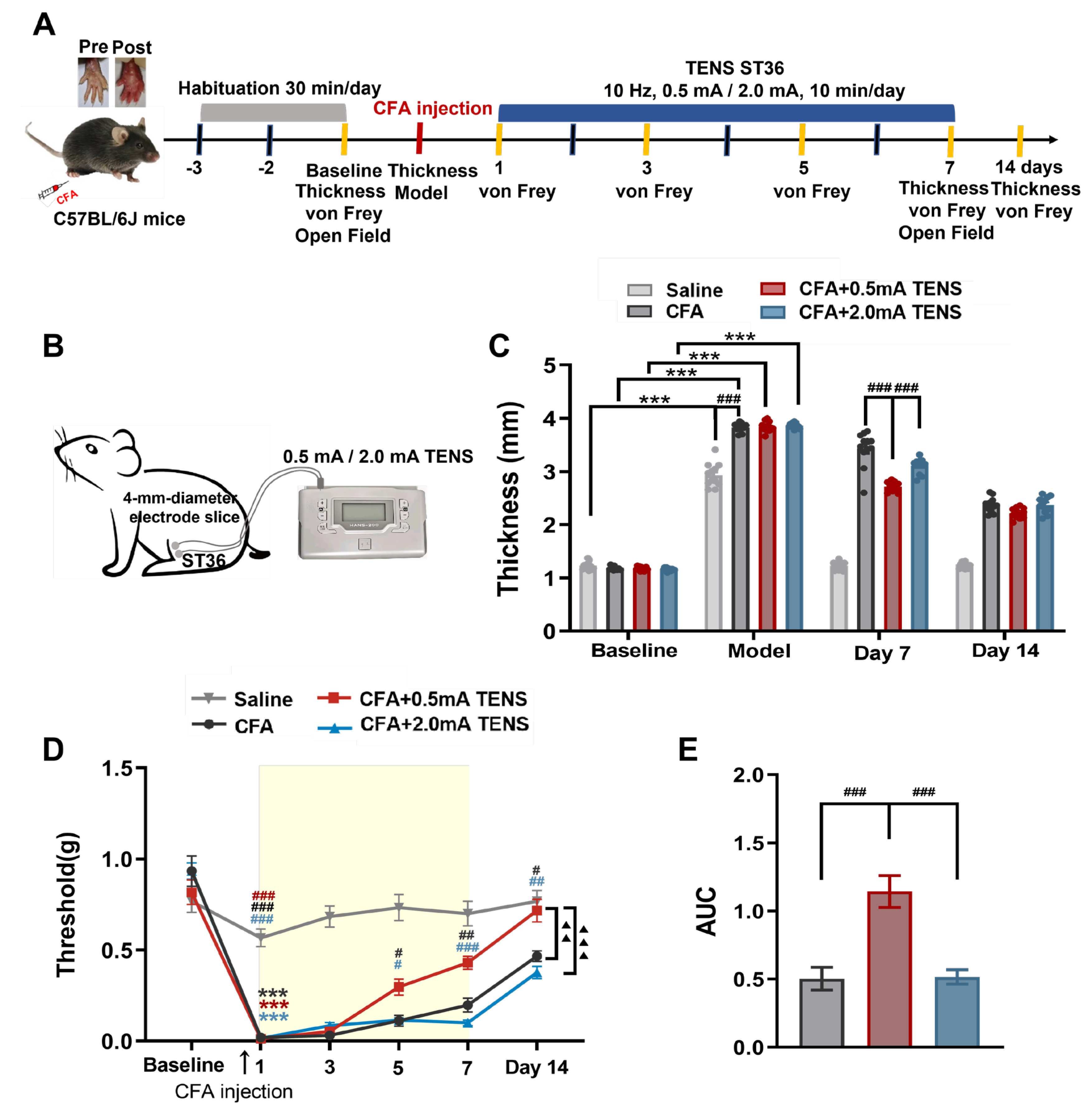
3.1. Effects of TENS ST36 on Hind Paw Thickness and Mechanical Pain Threshold in CFA Mice
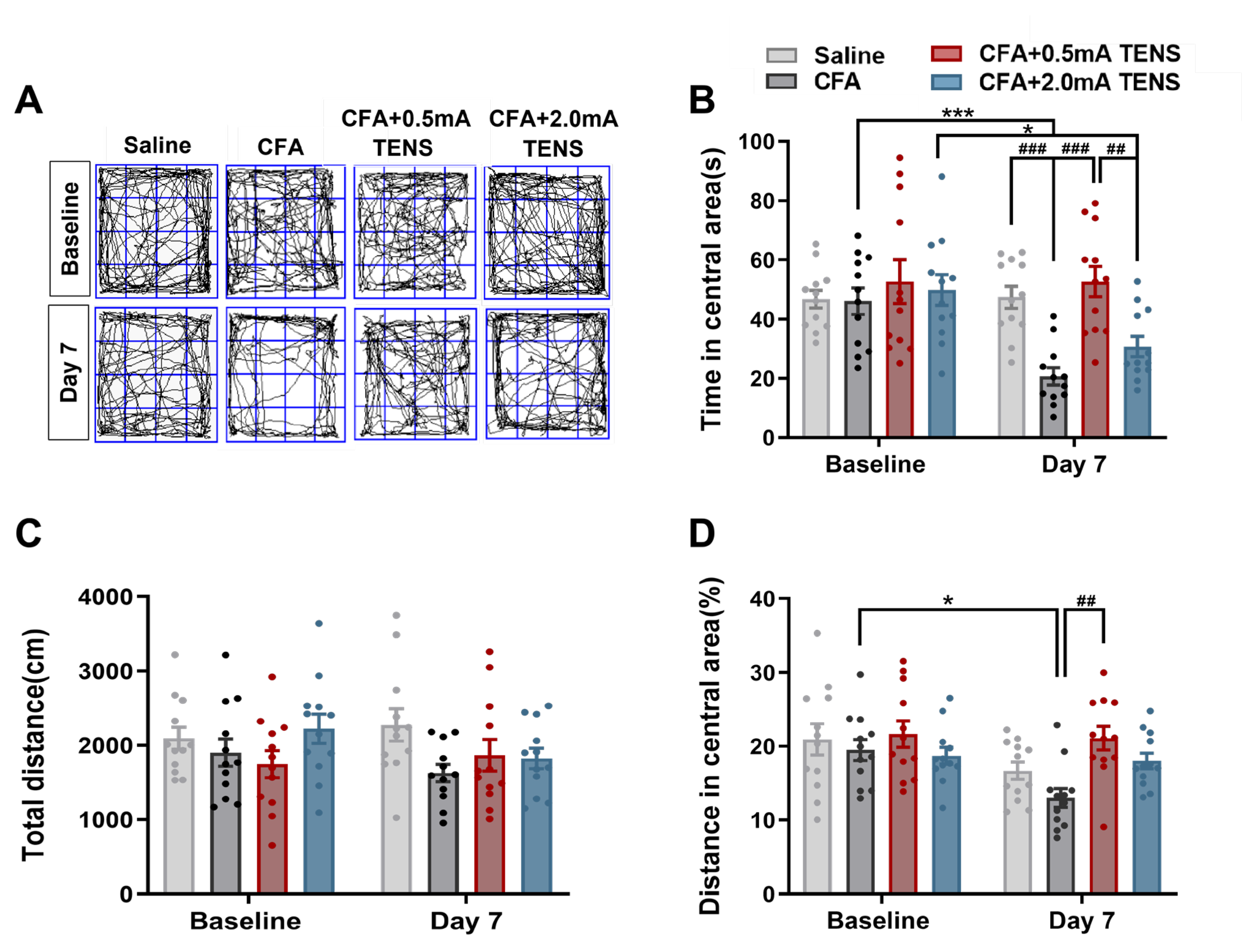
3.2. Effects of TENS ST36 on Anxiety-Like Behaviors in CFA Mice
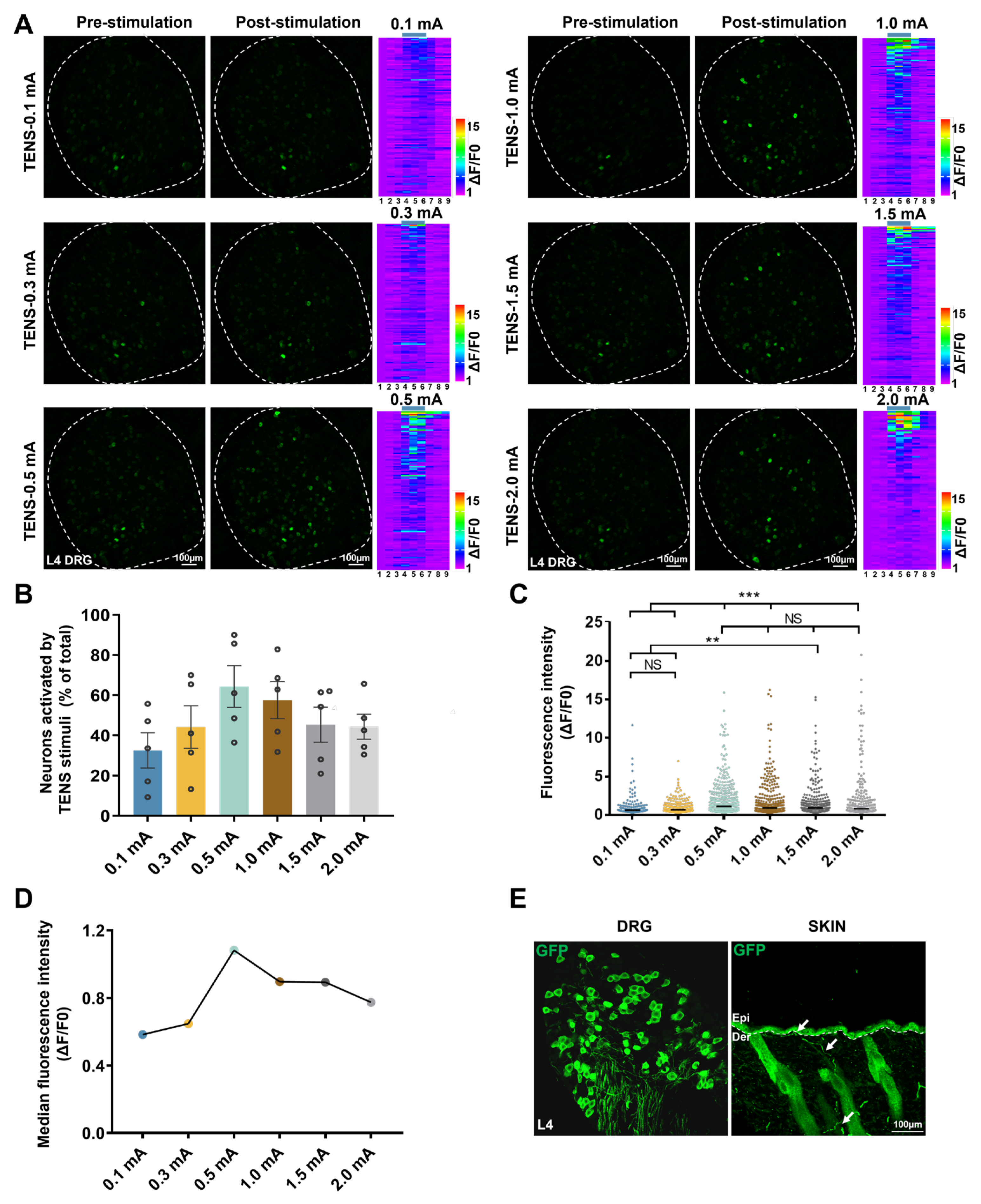
3.3. Responses of L4 DRG Mrgprb4-Lineage Neurons to Diverse TENS Stimulation In Vivo Ca2+ Imaging
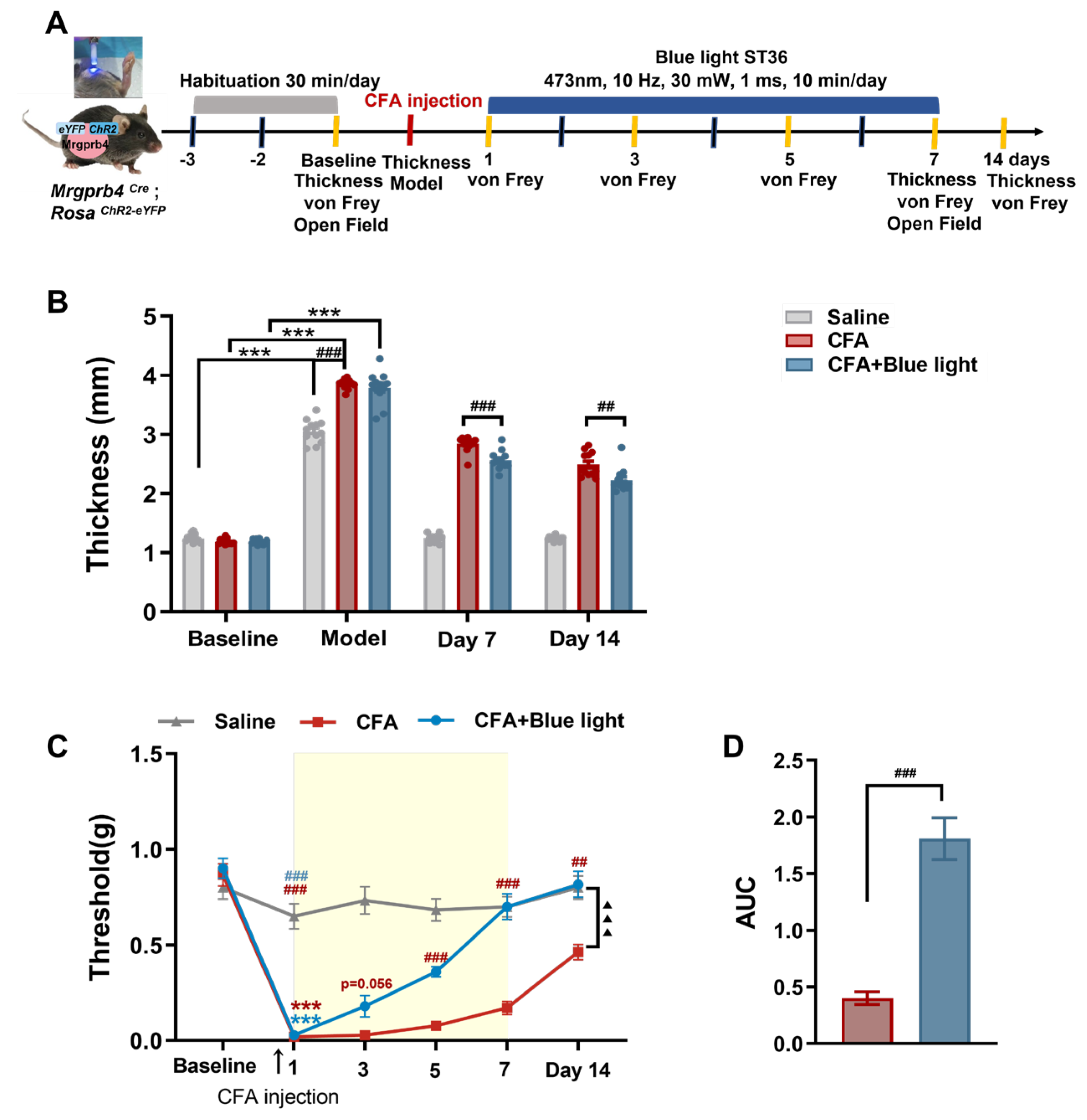
3.4. Effects of Mrgprb4-Lineage Neurons Activated by Blue Light on Hind Paw Thickness and Mechanical Pain Threshold of CFA Mice
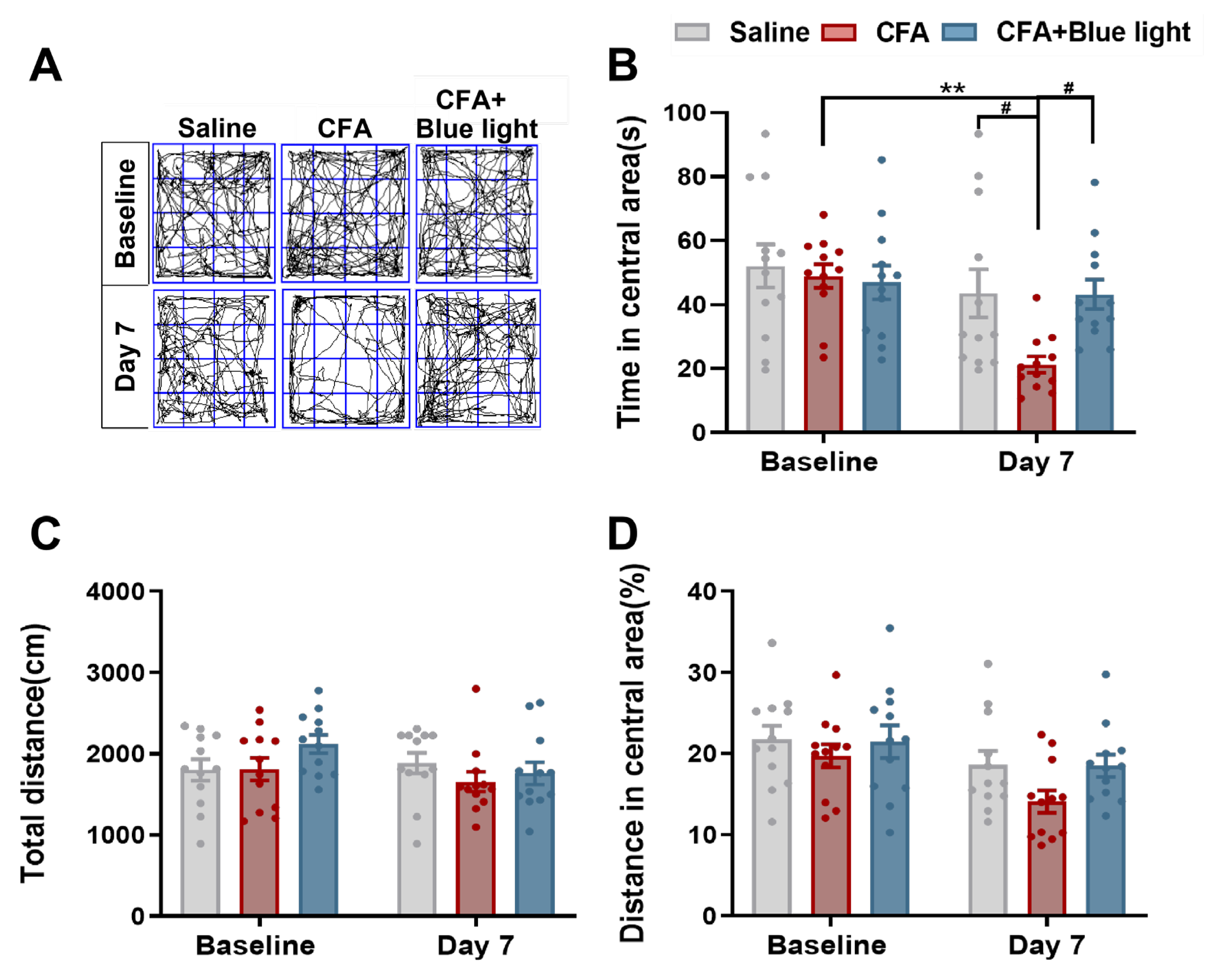
3.5. Effects of Mrgprb4-Lineage Neuron Activated by Blue Light on Anxiety-Like Behaviors of CFA Mice
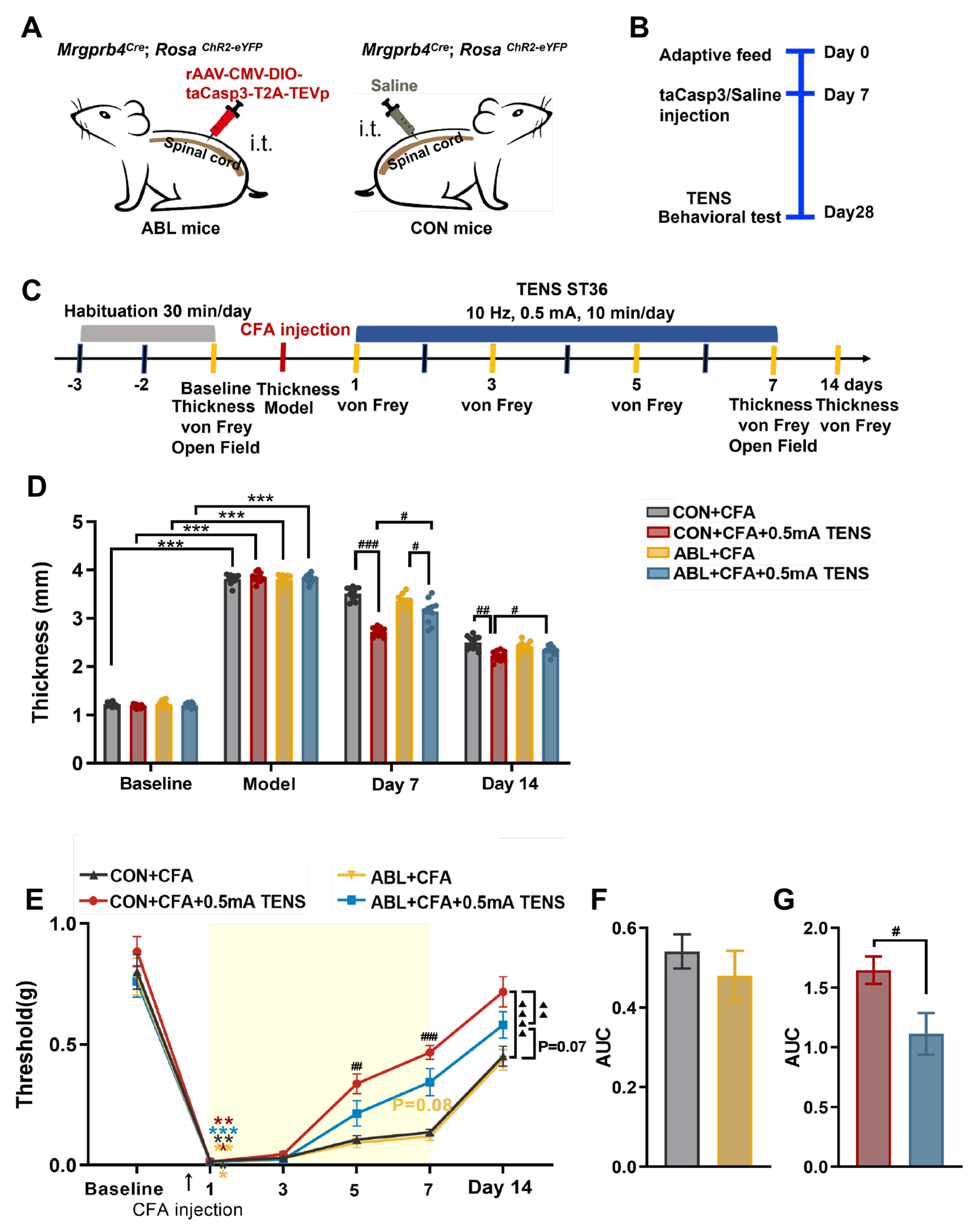
3.6. Effects of Virus Ablation of Mrgprb4-Lineage Neurons on Hind Paw Thickness and Mechanical Pain Threshold of CFA Mice
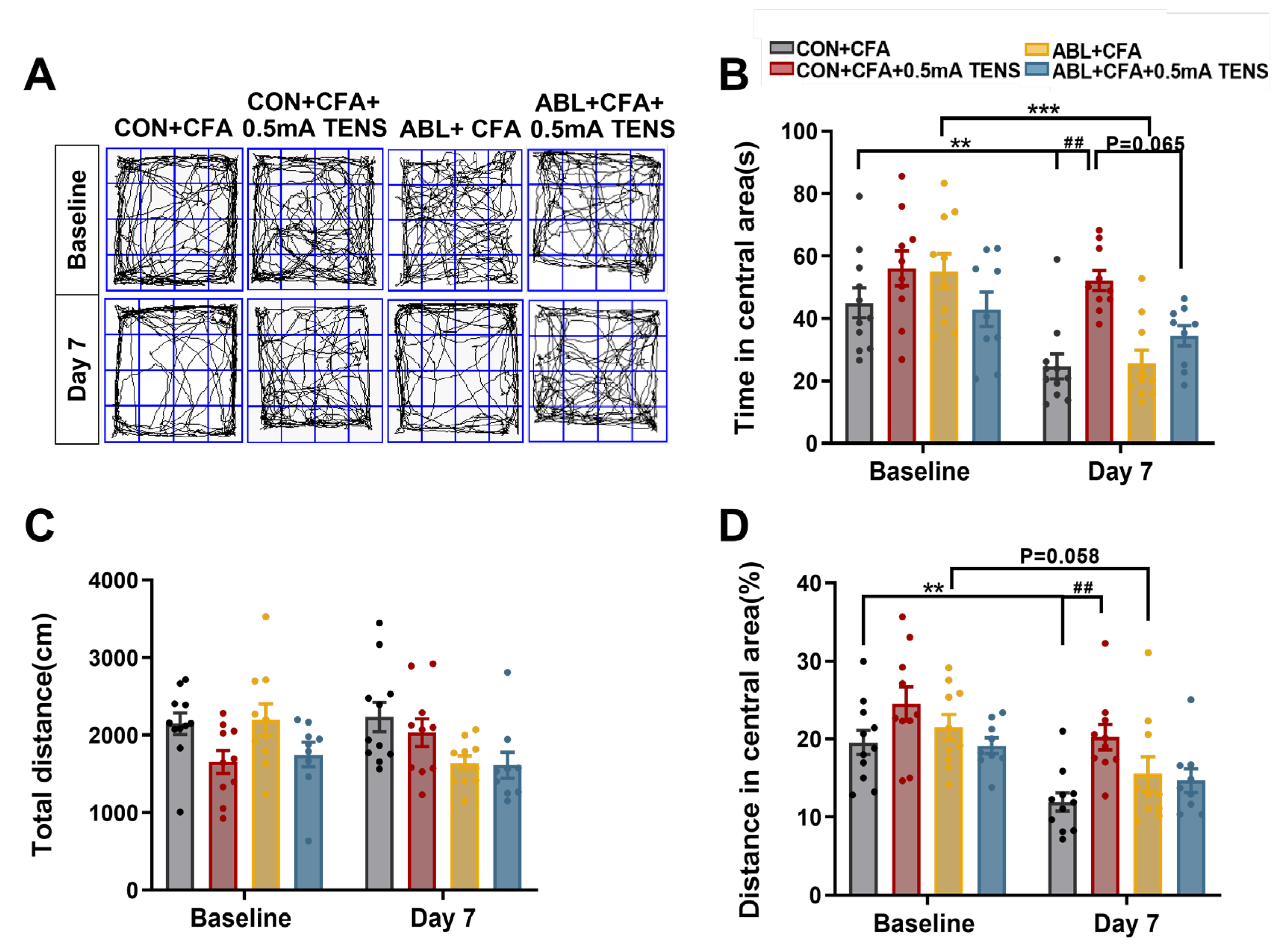
3.7. Effects of Virus Ablation of Mrgprb4-Lineage Neurons on Anxiety-Like Behaviors in CFA Mice
4. Discussion
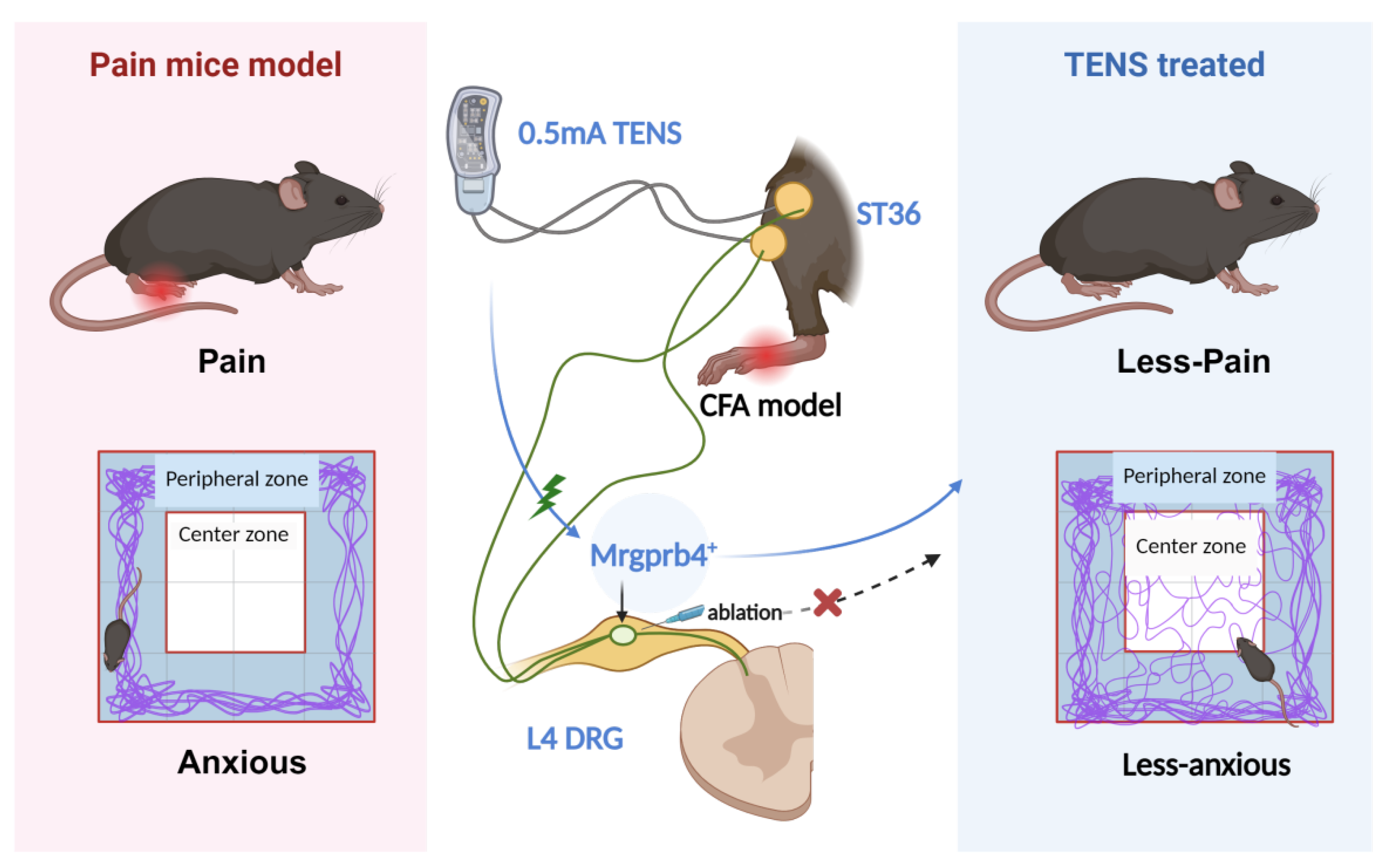
5. Conclusion
Limitations of the Study
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Consent for Publication
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| TRPV1 | Transient receptor potential vanilloid 1 |
| EA | Electroacupuncture |
| CFA | Complete Freund’s Adjuvant |
| Mrgprb4 | Mas-related G-protein-coupled receptor b4 |
| TENS | Transcutaneous electrical nerve stimulation |
| ST36 | Zusanli |
| DRG | Dorsal root ganglia |
| AUC | Area under the curve |
| 5-HT | 5-hydroxytryptamine |
| DA | Dopamine |
| SPB | Spinoparabrachial |
| Gpr83 | G protein-coupled receptor 83 |
References
- Price, D.D. Psychological and neural mechanisms of the affective dimension of pain. Science 2000, 288, 1769–1772. [Google Scholar] [CrossRef] [PubMed]
- Bouhassira, D. Neuropathic pain: Definition, assessment and epidemiology. Rev Neurol (Paris) 2019, 175, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Bandelow, B. Generalized Anxiety Disorder and Pain. Mod Trends Pharmacopsychiatry 2015, 30, 153–165. [Google Scholar] [CrossRef]
- Velly, A.M.; Mohit, S. Epidemiology of pain and relation to psychiatric disorders. Prog Neuropsychopharmacol Biol Psychiatry 2018, 87, 159–167. [Google Scholar] [CrossRef]
- Tsang, A.; Von Korff, M.; Lee, S.; Alonso, J.; Karam, E.; Angermeyer, M.C.; Borges, G.L.; Bromet, E.J.; Demytteneare, K.; de Girolamo, G.; et al. Common chronic pain conditions in developed and developing countries: gender and age differences and comorbidity with depression-anxiety disorders. J Pain 2008, 9, 883–891. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.; Correll, D.J.; Lechner, S.M.; Jazic, I.; Miao, X.; Shaw, D.; Simard, C.; Osteen, J.D.; Hare, B.; Beaton, A.; et al. Selective Inhibition of Na(V)1.8 with VX-548 for Acute Pain. N Engl J Med 2023, 389, 393–405. [Google Scholar] [CrossRef] [PubMed]
- Mecum, N.E.; Russell, R.; Lee, J.; Sullivan, C.; Meng, I.D. Optogenetic Inhibition of Nav1.8 Expressing Corneal Afferents Reduces Persistent Dry Eye Pain. Invest Ophthalmol Vis Sci 2021, 62, 15. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.Y.; Zhang, Y.L.; Sun, Q.; Zhang, P.A.; Wang, X.X.; Xu, G.Y.; Hu, J.; Zhang, H.H. Alpha-lipoic acid downregulates TRPV1 receptor via NF-κB and attenuates neuropathic pain in rats with diabetes. CNS Neurosci Ther 2020, 26, 762–772. [Google Scholar] [CrossRef]
- Barton, N.J.; McQueen, D.S.; Thomson, D.; Gauldie, S.D.; Wilson, A.W.; Salter, D.M.; Chessell, I.P. Attenuation of experimental arthritis in TRPV1R knockout mice. Exp Mol Pathol 2006, 81, 166–170. [Google Scholar] [CrossRef]
- Cavanaugh, D.J.; Lee, H.; Lo, L.; Shields, S.D.; Zylka, M.J.; Basbaum, A.I.; Anderson, D.J. Distinct subsets of unmyelinated primary sensory fibers mediate behavioral responses to noxious thermal and mechanical stimuli. Proc Natl Acad Sci U S A 2009, 106, 9075–9080. [Google Scholar] [CrossRef]
- Liao, H.Y.; Lin, Y.W. Electroacupuncture Attenuates Chronic Inflammatory Pain and Depression Comorbidity through Transient Receptor Potential V1 in the Brain. Am J Chin Med 2021, 49, 1417–1435. [Google Scholar] [CrossRef]
- Du, L.; Cheng, H.; Cui, X.; Cao, Q.; Li, X.; Wang, S.; Wang, X.; Liu, Y.; Zhu, B.; Gao, X.; et al. Mrgprb4-lineage neurons indispensable in pressure induced pleasant sensation are polymodal. iScience 2025, 28, 111940. [Google Scholar] [CrossRef]
- Kun Liu, B.Z. Significance of pleasant touch and state-of-the-art neuroscience technologies in acupuncture research. Acupuncture and Herbal Medicine 2023, 3, 55–58. [Google Scholar]
- Elias, L.J.; Succi, I.K.; Schaffler, M.D.; Foster, W.; Gradwell, M.A.; Bohic, M.; Fushiki, A.; Upadhyay, A.; Ejoh, L.L.; Schwark, R.; et al. Touch neurons underlying dopaminergic pleasurable touch and sexual receptivity. Cell 2023, 186, 577–590.e516. [Google Scholar] [CrossRef]
- Hylands-White, N.; Duarte, R.V.; Raphael, J.H. An overview of treatment approaches for chronic pain management. Rheumatol Int 2017, 37, 29–42. [Google Scholar] [CrossRef]
- Rocchio, R.J.; Ward, K.E. Intranasal Ketamine for Acute Pain. Clin J Pain 2021, 37, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Dailey, D.L.; Vance, C.G.T.; Rakel, B.A.; Zimmerman, M.B.; Embree, J.; Merriwether, E.N.; Geasland, K.M.; Chimenti, R.; Williams, J.M.; Golchha, M.; et al. Transcutaneous Electrical Nerve Stimulation Reduces Movement-Evoked Pain and Fatigue: A Randomized, Controlled Trial. Arthritis Rheumatol 2020, 72, 824–836. [Google Scholar] [CrossRef] [PubMed]
- Dailey, D.L.; Rakel, B.A.; Vance, C.G.T.; Liebano, R.E.; Amrit, A.S.; Bush, H.M.; Lee, K.S.; Lee, J.E.; Sluka, K.A. Transcutaneous electrical nerve stimulation reduces pain, fatigue and hyperalgesia while restoring central inhibition in primary fibromyalgia. Pain 2013, 154, 2554–2562. [Google Scholar] [CrossRef] [PubMed]
- Sabino, G.S.; Santos, C.M.; Francischi, J.N.; de Resende, M.A. Release of endogenous opioids following transcutaneous electric nerve stimulation in an experimental model of acute inflammatory pain. J Pain 2008, 9, 157–163. [Google Scholar] [CrossRef]
- DeSantana, J.M.; Santana-Filho, V.J.; Guerra, D.R.; Sluka, K.A.; Gurgel, R.Q.; da Silva, W.M., Jr. Hypoalgesic effect of the transcutaneous electrical nerve stimulation following inguinal herniorrhaphy: a randomized, controlled trial. J Pain 2008, 9, 623–629. [Google Scholar] [CrossRef]
- Osiri, M.; Welch, V.; Brosseau, L.; Shea, B.; McGowan, J.; Tugwell, P.; Wells, G. Transcutaneous electrical nerve stimulation for knee osteoarthritis. Cochrane Database Syst Rev 2000, Cd002823. [Google Scholar] [CrossRef]
- Schuster, G.D.; Infante, M.C. Pain relief after low back surgery: the efficacy of transcutaneous electrical nerve stimulation. Pain 1980, 8, 299–302. [Google Scholar] [CrossRef]
- Tanaka, M.; Vécsei, L. From Lab to Life: Exploring Cutting-Edge Models for Neurological and Psychiatric Disorders. Biomedicines 2024, 12. [Google Scholar] [CrossRef] [PubMed]
- Cebalo, N.; Negovetić Vranić, D.; Bašić Kes, V. The Effect of Transcutaneous Electric Nerve Stimulation (TENS) on Anxiety and Fear in Children Aged 9-14 Years. Acta Stomatol Croat 2020, 54, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, J.; Stamey, W.; Strutt, A.M.; Adam, O.R.; Jankovic, J. Transcutaneous electrical stimulation (TENS) for psychogenic movement disorders. J Neuropsychiatry Clin Neurosci 2011, 23, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Al-Zamil, M.; Minenko, I.A.; Kulikova, N.G.; Mansur, N.; Nuvakhova, M.B.; Khripunova, O.V.; Shurygina, I.P.; Topolyanskaya, S.V.; Trefilova, V.V.; Petrova, M.M.; et al. Efficiency of Direct Transcutaneous Electroneurostimulation of the Median Nerve in the Regression of Residual Neurological Symptoms after Carpal Tunnel Decompression Surgery. Biomedicines 2023, 11. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Tang, J.; White, P.F.; Sloninsky, A.; Wender, R.H.; Naruse, R.; Kariger, R. The effect of location of transcutaneous electrical nerve stimulation on postoperative opioid analgesic requirement: acupoint versus nonacupoint stimulation. Anesth Analg 1998, 87, 1129–1134. [Google Scholar]
- Liu, B.; Qiao, L.; Liu, K.; Liu, J.; Piccinni-Ash, T.J.; Chen, Z.F. Molecular and neural basis of pleasant touch sensation. Science 2022, 376, 483–491. [Google Scholar] [CrossRef]
- Zhongren, L. China Press of Traditional Chinese Medicine. In Experimental Acupuncturology; Beijing, China, 2003. [Google Scholar]
- Gao, X.; Han, S.; Huang, Q.; He, S.Q.; Ford, N.C.; Zheng, Q.; Chen, Z.; Yu, S.; Dong, X.; Guan, Y. Calcium imaging in population of dorsal root ganglion neurons unravels novel mechanisms of visceral pain sensitization and referred somatic hypersensitivity. Pain 2021, 162, 1068–1081. [Google Scholar] [CrossRef]
- Chen, Z.; Huang, Q.; Song, X.; Ford, N.C.; Zhang, C.; Xu, Q.; Lay, M.; He, S.Q.; Dong, X.; Hanani, M.; et al. Purinergic signaling between neurons and satellite glial cells of mouse dorsal root ganglia modulates neuronal excitability in vivo. Pain 2022, 163, 1636–1647. [Google Scholar] [CrossRef]
- Liu, K.; Liu, Y.; Li, X.; Wang, S.; Wang, X.; Zhang, Z.; Gao, X. In Vivo Calcium Imaging of Dorsal Root Ganglia Neurons’ Response to Somatic and Visceral Stimuli. J Vis Exp 2024. [Google Scholar] [CrossRef]
- Lampe, G.N. Introduction to the use of transcutaneous electrical nerve stimulation devices. Phys Ther 1978, 58, 1450–1454. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.J.; Wang, E.J.; Loprinzi, C.L. Cutaneous Electroanalgesia for Relief of Chronic and Neuropathic Pain. N Engl J Med 2023, 389, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Masson, E.A.; Veves, A.; Fernando, D.; Boulton, A.J. Current perception thresholds: a new, quick, and reproducible method for the assessment of peripheral neuropathy in diabetes mellitus. Diabetologia 1989, 32, 724–728. [Google Scholar] [CrossRef] [PubMed]
- Reeve, A.J.; Walker, K.; Urban, L.; Fox, A. Excitatory effects of galanin in the spinal cord of intact, anaesthetized rats. Neurosci Lett 2000, 295, 25–28. [Google Scholar] [CrossRef] [PubMed]
- Dufour, A.; Guergova, S.; Pebayle, T.; Touzalin-Chretien, P. On the selective activation of unmyelinated C-fibers using sinusoidal electrical stimulation: an ERP study. Clin Neurophysiol 2011, 122, 1042–1047. [Google Scholar] [CrossRef]
- Wright, A. Exploring the evidence for using TENS to relieve pain. Nurs Times 2012, 108, 20–23. [Google Scholar] [PubMed]
- Liu, S.; Long, S.S.; Li, F.; Yang, H.; Pu, S.; Du, D.; Luo, X.; Zhang, Y.Q.; Han, Q. Neural basis of transcutaneous electrical nerve stimulation for neuropathic pain relief. Neuron 2025, 113, 3616–3631.e3616. [Google Scholar] [CrossRef] [PubMed]
- Sjölund, BH; EM, L.J. Transcutaneous and implanted electric stimulation of peripheral nerves. In Management of Pain; Lea & Febiger: Philadelphia, 1990; pp. 1852–1861 p. [Google Scholar]
- Lee, J.H.; Choi, Y.H.; Choi, B.T. The anti-inflammatory effects of 2 Hz electroacupuncture with different intensities on acute carrageenan-induced inflammation in the rat paw. Int J Mol Med 2005, 16, 99–102. [Google Scholar] [CrossRef]
- Guo Zi, W.N.; Ru, Ye; Tiancheng, Sun; Shuang, Qiu; Xiaomei, Shao; Xiaochang, Ge; Lu, Guan; Junfang, Fang; Jianqiao, Fang; Junying, Du. Map activation of various brain regions using different frequencies of electroacupuncture ST36, utilizing the Fos-CreER strategy. Acupuncture and Herbal Medicine 2024, 4, 386–398. [Google Scholar]
- Li, Y.J.; Du, W.J.; Liu, R.; Zan, G.Y.; Ye, B.L.; Li, Q.; Sheng, Z.H.; Yuan, Y.W.; Song, Y.J.; Liu, J.G.; et al. Paraventricular nucleus-central amygdala oxytocinergic projection modulates pain-related anxiety-like behaviors in mice. CNS Neurosci Ther 2023, 29, 3493–3506. [Google Scholar] [CrossRef]
- Guo, H.; Hu, W.C.; Xian, H.; Shi, Y.X.; Liu, Y.Y.; Ma, S.B.; Pan, K.Q.; Wu, S.X.; Xu, L.Y.; Luo, C.; et al. CCL2 Potentiates Inflammation Pain and Related Anxiety-Like Behavior Through NMDA Signaling in Anterior Cingulate Cortex. Mol Neurobiol 2024, 61, 4976–4991. [Google Scholar] [CrossRef]
- McCarson, K.E.; Fehrenbacher, J.C. Models of Inflammation: Carrageenan- or Complete Freund’s Adjuvant (CFA)-Induced Edema and Hypersensitivity in the Rat. Curr Protoc 2021, 1, e202. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Song, Y.; Yang, J.; Zhang, Y.; Zhao, P.; Zhu, X.J.; Su, H.C. The contribution of TNF-α in the amygdala to anxiety in mice with persistent inflammatory pain. Neurosci Lett 2013, 541, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Melzack, R.; Wall, P.D. Pain mechanisms: a new theory. Science 1965, 150, 971–979. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Perl, E.R. A specific inhibitory pathway between substantia gelatinosa neurons receiving direct C-fiber input. J Neurosci 2003, 23, 8752–8758. [Google Scholar] [CrossRef]
- Han, J.S. Acupuncture: neuropeptide release produced by electrical stimulation of different frequencies. Trends Neurosci 2003, 26, 17–22. [Google Scholar] [CrossRef]
- Sluka, K.A.; Lisi, T.L.; Westlund, K.N. Increased release of serotonin in the spinal cord during low, but not high, frequency transcutaneous electric nerve stimulation in rats with joint inflammation. Arch Phys Med Rehabil 2006, 87, 1137–1140. [Google Scholar] [CrossRef]
- Rokugo, T.; Takeuchi, T.; Ito, H. A histochemical study of substance P in the rat spinal cord: effect of transcutaneous electrical nerve stimulation. J Nippon Med Sch 2002, 69, 428–433. [Google Scholar] [CrossRef]
- Sluka, K.A.; Vance, C.G.; Lisi, T.L. High-frequency, but not low-frequency, transcutaneous electrical nerve stimulation reduces aspartate and glutamate release in the spinal cord dorsal horn. J Neurochem 2005, 95, 1794–1801. [Google Scholar] [CrossRef]
- Chen, Y.W.; Tzeng, J.I.; Lin, M.F.; Hung, C.H.; Hsieh, P.L.; Wang, J.J. High-frequency transcutaneous electrical nerve stimulation attenuates postsurgical pain and inhibits excess substance P in rat dorsal root ganglion. Reg Anesth Pain Med 2014, 39, 322–328. [Google Scholar] [CrossRef]
- Chen, Y.W.; Tzeng, J.I.; Lin, M.F.; Hung, C.H.; Wang, J.J. Transcutaneous electrical nerve stimulation attenuates postsurgical allodynia and suppresses spinal substance P and proinflammatory cytokine release in rats. Phys Ther 2015, 95, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Sluka, K.A.; Bailey, K.; Bogush, J.; Olson, R.; Ricketts, A. Treatment with either high or low frequency TENS reduces the secondary hyperalgesia observed after injection of kaolin and carrageenan into the knee joint. Pain 1998, 77, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Price, D.D.; Hu, J.W.; Dubner, R.; Gracely, R.H. Peripheral suppression of first pain and central summation of second pain evoked by noxious heat pulses. Pain 1977, 3, 57–68. [Google Scholar] [CrossRef]
- Zarrindast, M.R.; Khakpai, F. The Modulatory Role of Dopamine in Anxiety-like Behavior. Arch Iran Med 2015, 18, 591–603. [Google Scholar]
- Choi, S.; Hachisuka, J.; Brett, M.A.; Magee, A.R.; Omori, Y.; Iqbal, N.U.; Zhang, D.; DeLisle, M.M.; Wolfson, R.L.; Bai, L.; et al. Parallel ascending spinal pathways for affective touch and pain. Nature 2020, 587, 258–263. [Google Scholar] [CrossRef] [PubMed]
- Tzschentke, T.M. Measuring reward with the conditioned place preference (CPP) paradigm: update of the last decade. Addict Biol 2007, 12, 227–462. [Google Scholar] [CrossRef] [PubMed]
- Vrontou, S.; Wong, A.M.; Rau, K.K.; Koerber, H.R.; Anderson, D.J. Genetic identification of C fibres that detect massage-like stroking of hairy skin in vivo. Nature 2013, 493, 669–673. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



