Submitted:
19 January 2026
Posted:
21 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cell and Viruses
2.2. Immunocytochemistry
2.3. RNA Fluorescence In Situ Hybridization (FISH)
2.4. Transmission Electron Microscopy
3. Results
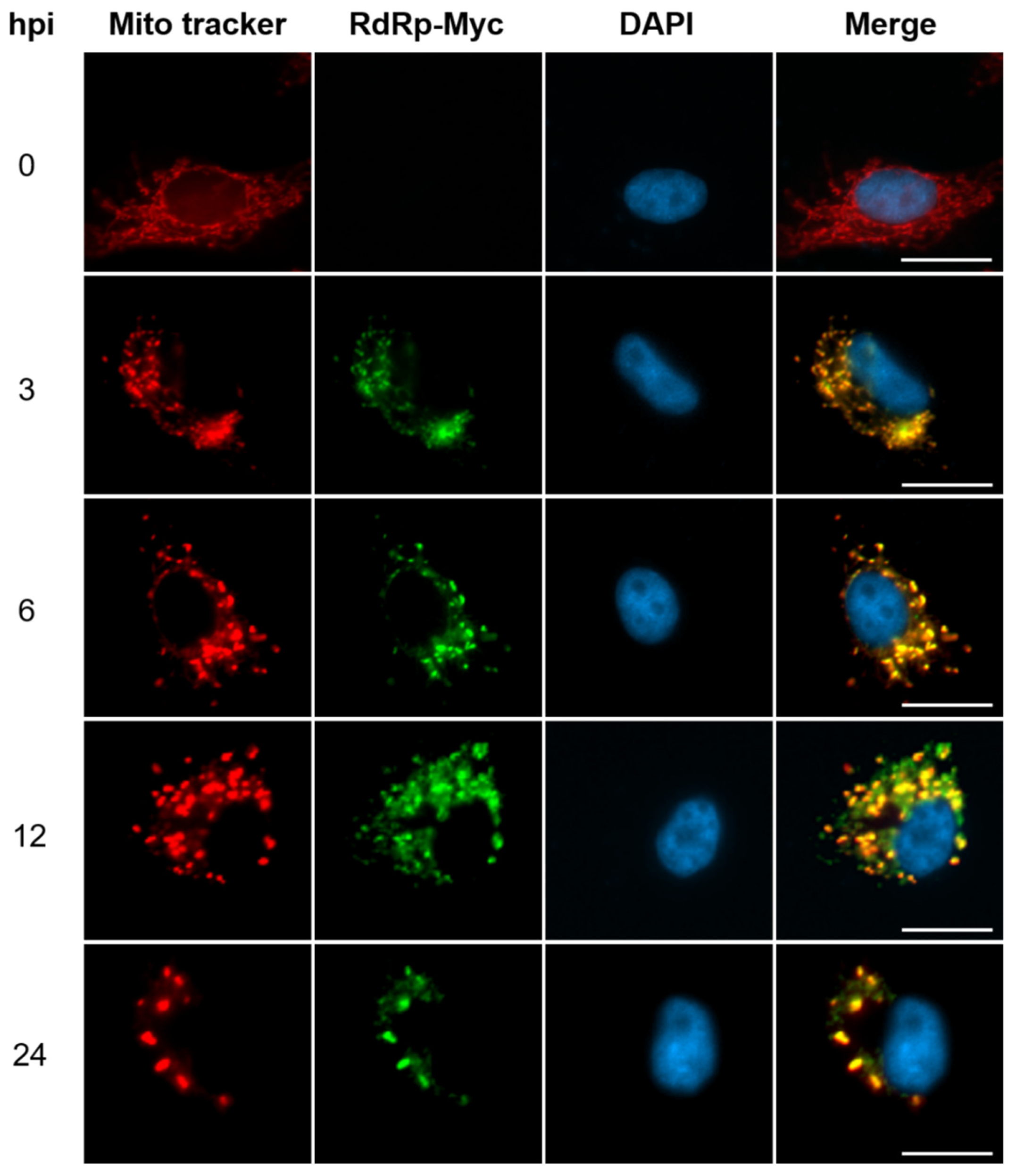
3.1. Translocation of RdRp into Mitochondria for NNV RNAs Synthesis
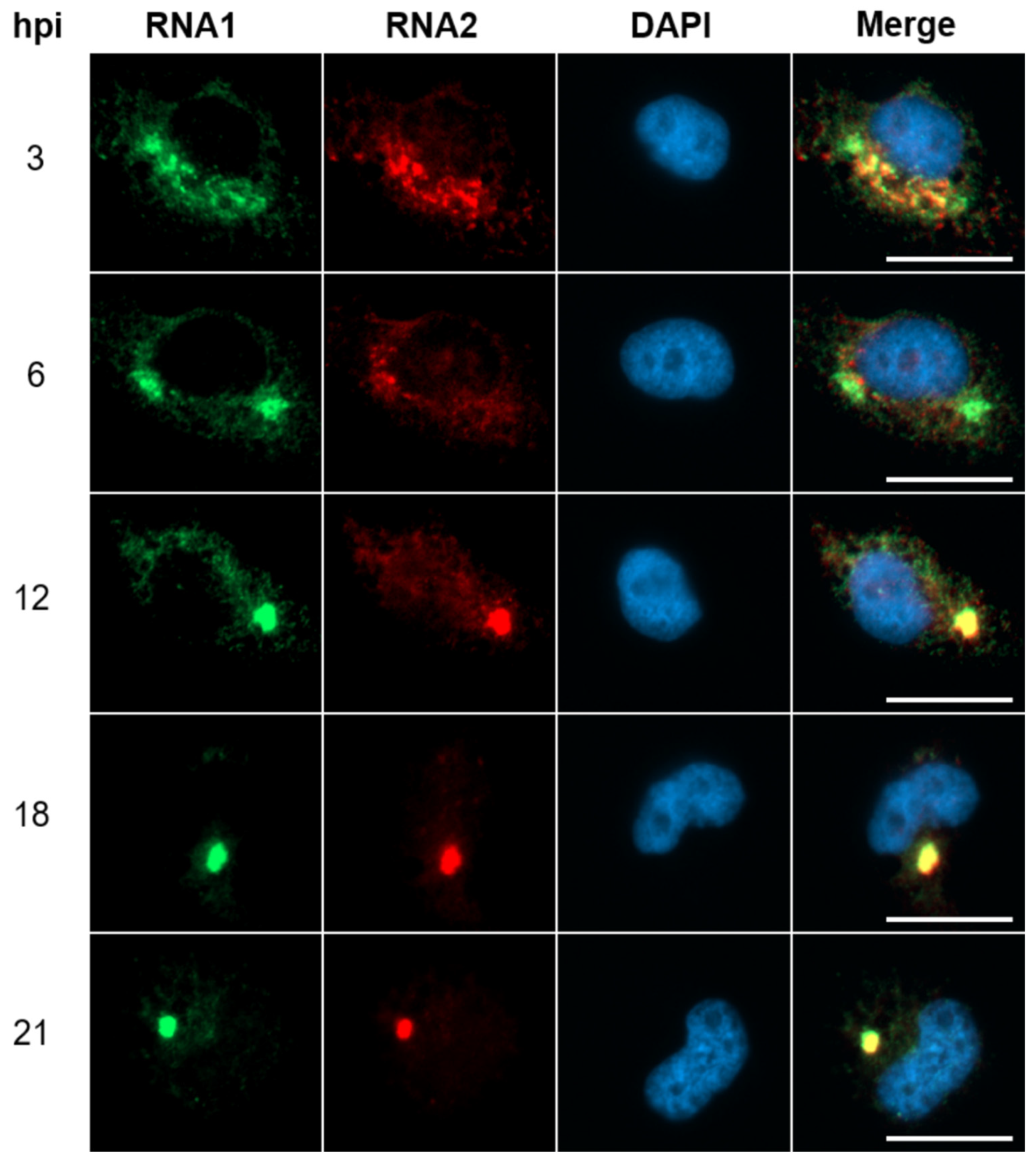
3.2. Neosynthesized NNV RNAs Move Toward Perinuclear Compartment
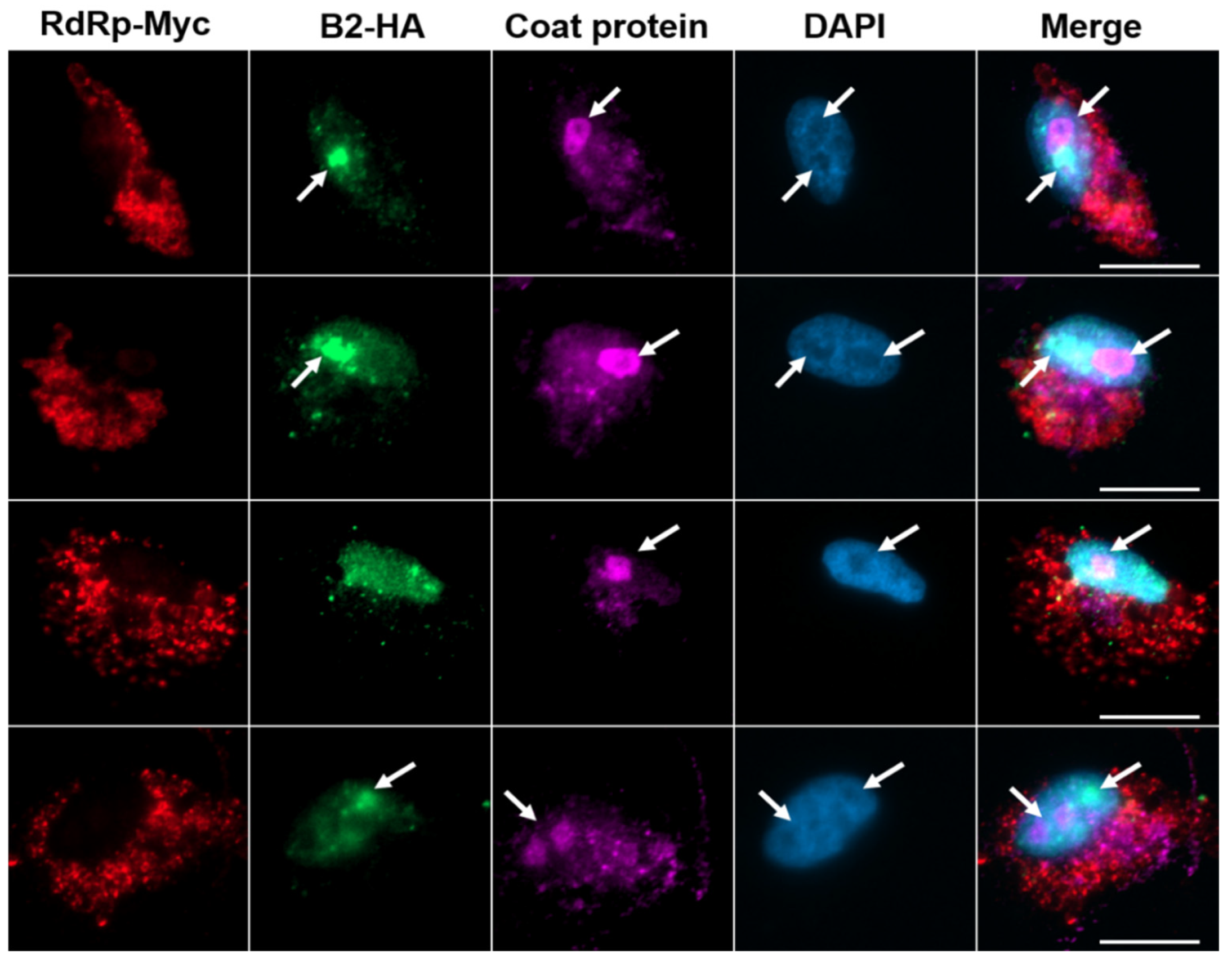
3.3. Transportation of NNV Viral Proteins into Nucleolus
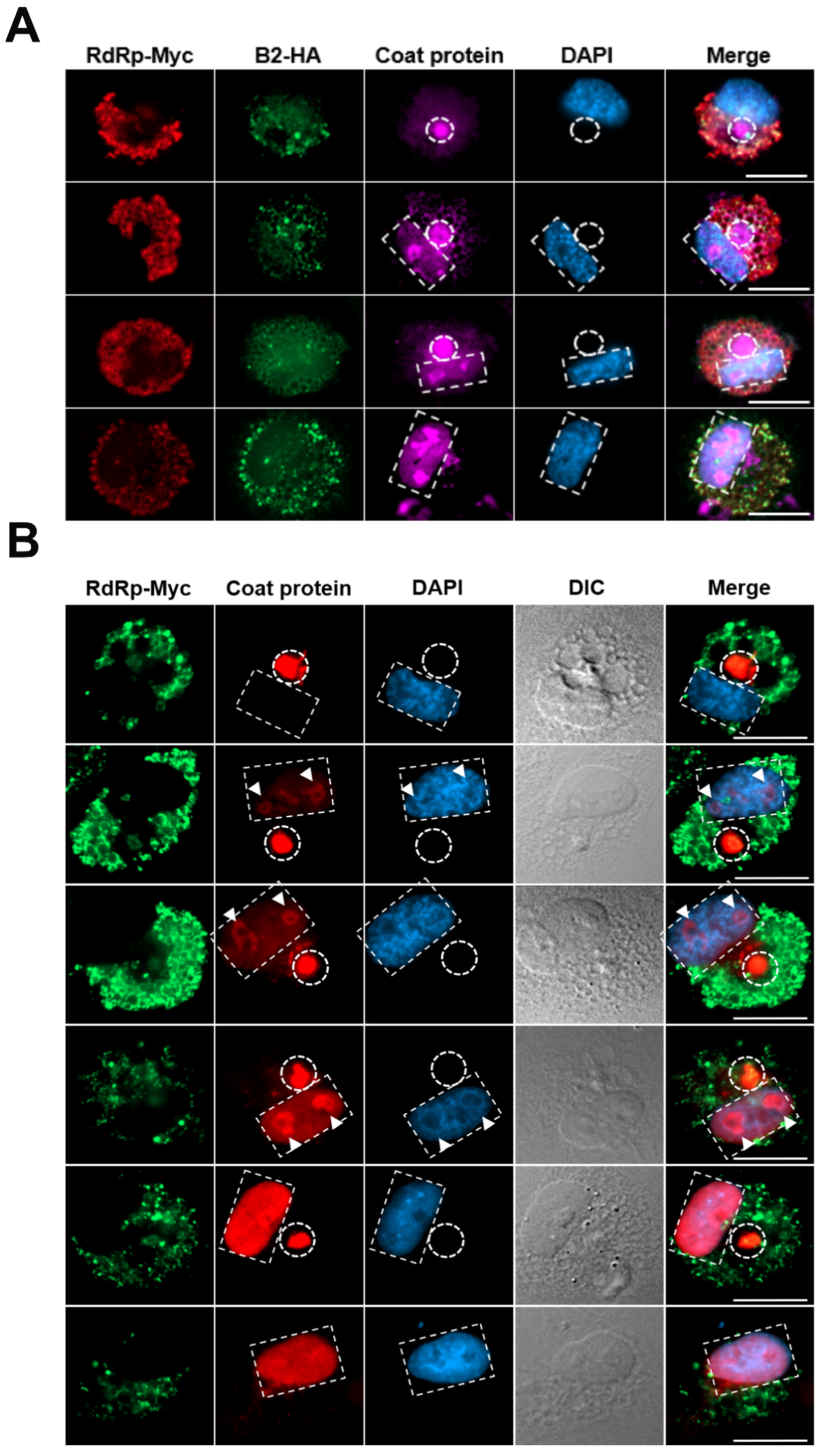
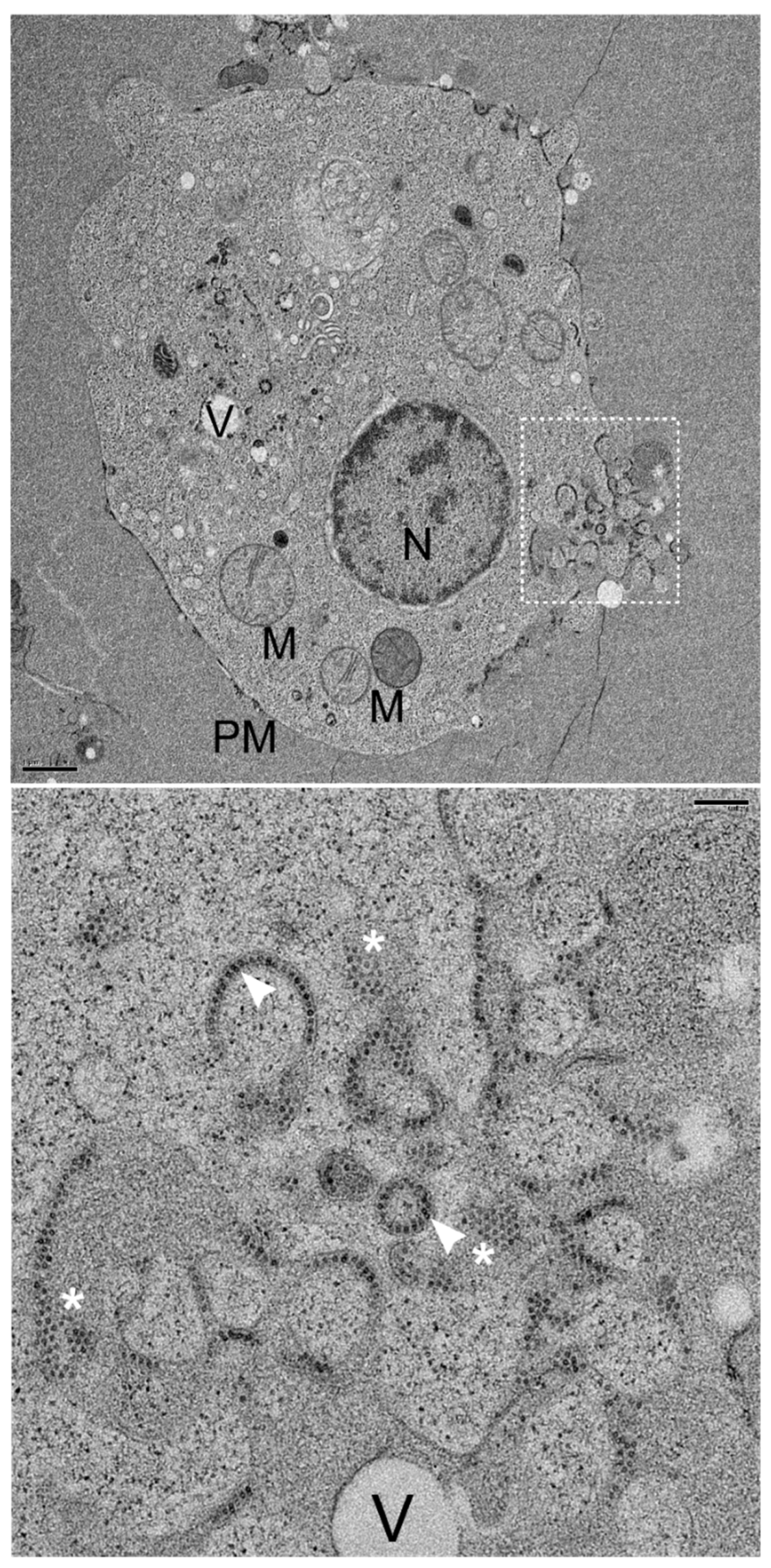
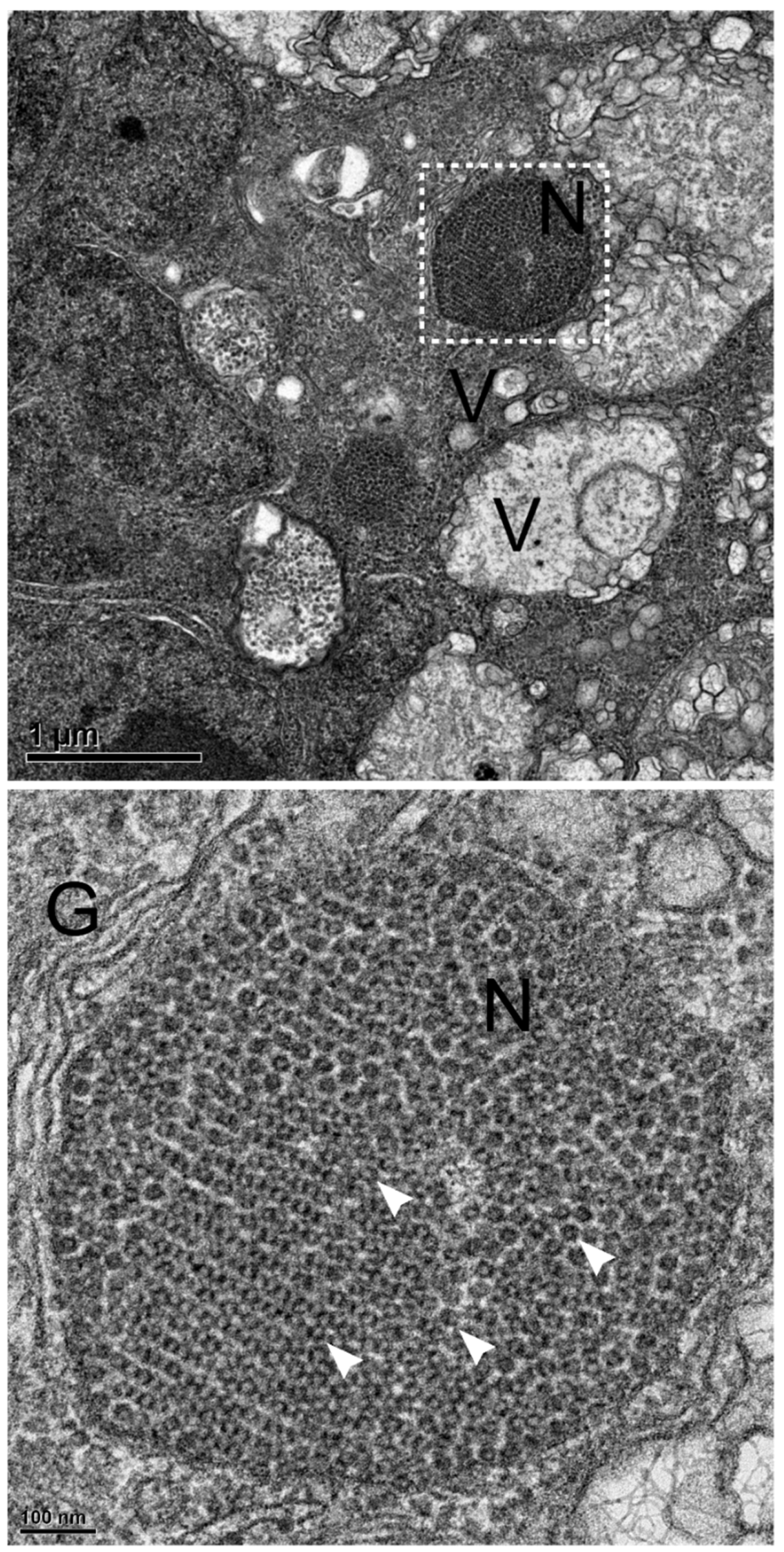
3.4. Convergence of NNV Coat Protein into Remodeled MTOC
3.5. NNV Particles Assembly in Remodeled MTOC
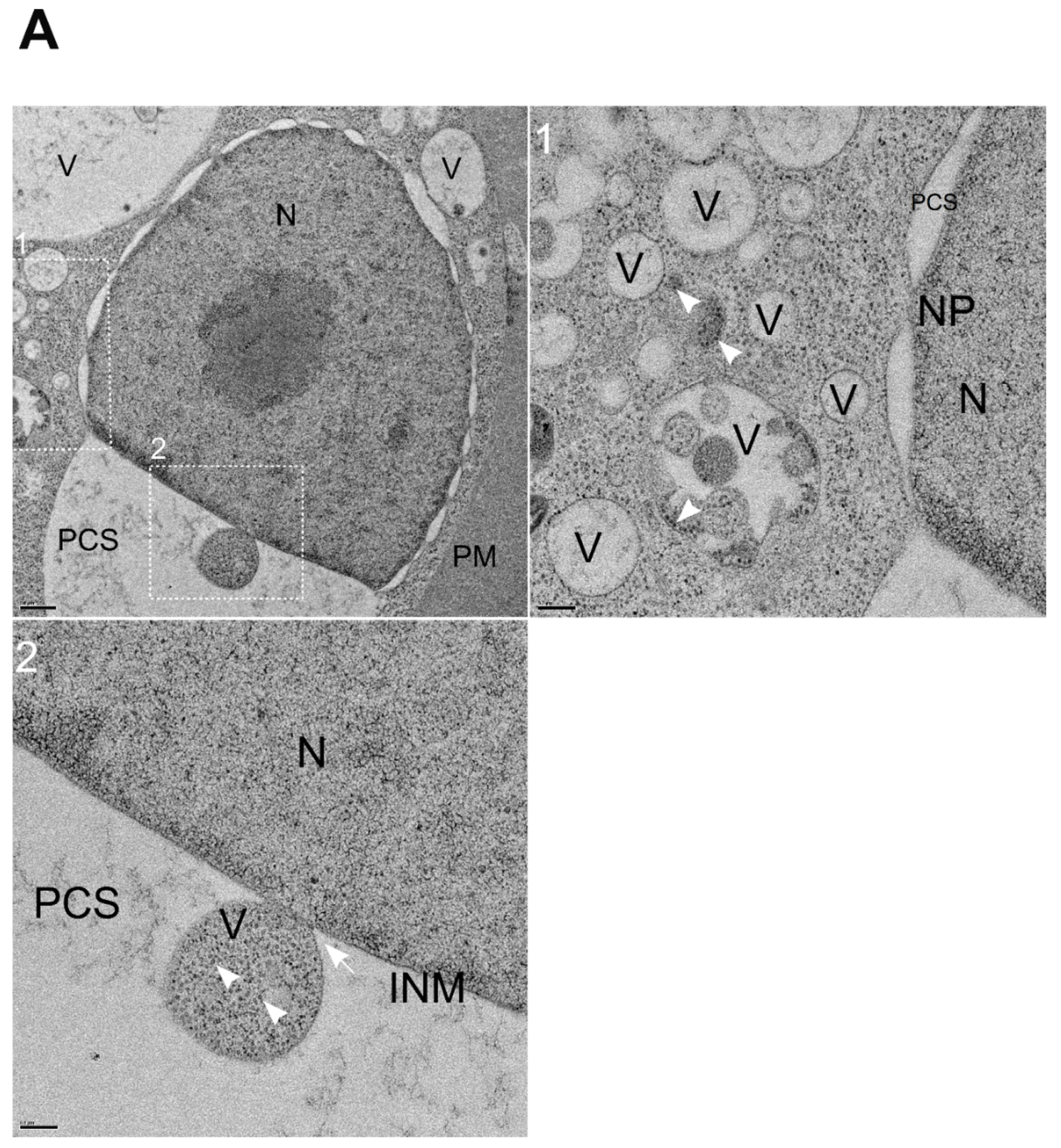
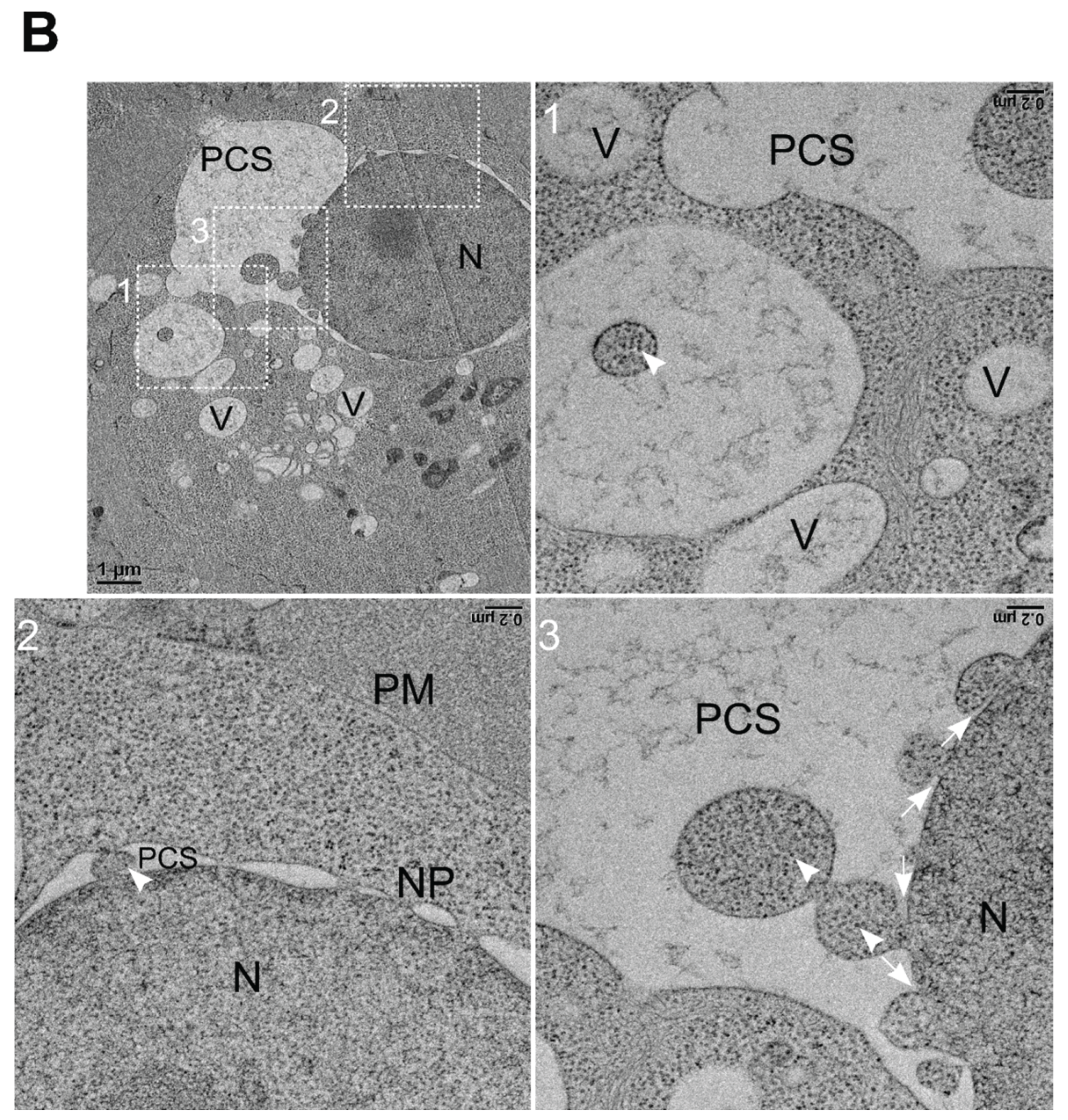
3.6. NNV Particles Containing Vesicles Merging into Nucleus
3.7. Storage of NNV Particles Inside Nucleus
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hetzer, M.W. The nuclear envelope. Cold Spring Harb Perspect Biol 2010, 2, a000539. [Google Scholar] [CrossRef] [PubMed]
- Dubois, M.-L.; Boisvert, F.-M. The Nucleolus: Structure and Function. In The Functional Nucleus; Bazett-Jones, D.P., Dellaire, G., Eds.; Springer International Publishing: Cham, 2016; pp. 29–49. [Google Scholar]
- Amineva, S.P.; Aminev, A.G.; Palmenberg, A.C.; Gern, J.E. Rhinovirus 3C protease precursors 3CD and 3CD’ localize to the nuclei of infected cells. J Gen Virol 2004, 85, 2969–2979. [Google Scholar] [CrossRef]
- Cheng, C.A.; Luo, J.M.; Chiang, M.H.; Fang, K.Y.; Li, C.H.; Chen, C.W.; Wang, Y.S.; Chang, C.Y. Nervous Necrosis Virus Coat Protein Mediates Host Translation Shutoff through Nuclear Translocalization and Degradation of Polyadenylate Binding Protein. J Virol 2021, 95, e0236420. [Google Scholar] [CrossRef]
- Aminev, A.G.; Amineva, S.P.; Palmenberg, A.C. Encephalomyocarditis viral protein 2A localizes to nucleoli and inhibits cap-dependent mRNA translation. Virus Res 2003, 95, 45–57. [Google Scholar] [CrossRef]
- Wang, X.; Zhu, J.; Zhang, D.; Liu, G. Ribosomal control in RNA virus-infected cells. Front Microbiol 2022, 13, 1026887. [Google Scholar] [CrossRef]
- Lu, J.; Wu, T.; Zhang, B.; Liu, S.; Song, W.; Qiao, J.; Ruan, H. Types of nuclear localization signals and mechanisms of protein import into the nucleus. Cell Commun Signal 2021, 19, 60. [Google Scholar] [CrossRef]
- Hiscox, J.A. RNA viruses: hijacking the dynamic nucleolus. Nat Rev Microbiol 2007, 5, 119–127. [Google Scholar] [CrossRef]
- Rowland, R.R.; Schneider, P.; Fang, Y.; Wootton, S.; Yoo, D.; Benfield, D.A. Peptide domains involved in the localization of the porcine reproductive and respiratory syndrome virus nucleocapsid protein to the nucleolus. Virology 2003, 316, 135–145. [Google Scholar] [CrossRef]
- Rowland, R.R.; Chauhan, V.; Fang, Y.; Pekosz, A.; Kerrigan, M.; Burton, M.D. Intracellular localization of the severe acute respiratory syndrome coronavirus nucleocapsid protein: absence of nucleolar accumulation during infection and after expression as a recombinant protein in vero cells. J Virol 2005, 79, 11507–11512. [Google Scholar] [CrossRef]
- Peränen, J.; Rikkonen, M.; Liljeström, P.; Kääriäinen, L. Nuclear localization of Semliki Forest virus-specific nonstructural protein nsP2. J Virol 1990, 64, 1888–1896. [Google Scholar] [CrossRef]
- Mori, Y.; Okabayashi, T.; Yamashita, T.; Zhao, Z.; Wakita, T.; Yasui, K.; Hasebe, F.; Tadano, M.; Konishi, E.; Moriishi, K.; et al. Nuclear localization of Japanese encephalitis virus core protein enhances viral replication. J Virol 2005, 79, 3448–3458. [Google Scholar] [CrossRef]
- Dearing, S.C.; Scotti, P.D.; Wigley, P.J.; Dhana, S.D. A small RNA virus isolated from the grass grub, Costelytra zealandica (Coleoptera: Scarabaeidae). New Zealand Journal of Zoology 1980, 7, 267–269. [Google Scholar] [CrossRef]
- YOSHIKOSHI, K.; INOUE, K. Viral nervous necrosis in hatchery-reared larvae and juveniles of Japanese parrotfish, Oplegnathus fasciatus (Temminck & Schlegel). Journal of Fish Diseases 1990, 13, 69–77. [Google Scholar] [CrossRef]
- Arcier, J.; Herman, F.; Lightner, D.V.; Redman, R.M.; Mari, J.; Bonami, J. A viral disease associated with mortalities in hatchery-reared postlarvae of the giant freshwater prawn Macrobrachium rosenbergii. Diseases of Aquatic Organisms 1999, 38, 177–181. [Google Scholar] [CrossRef]
- Munday, B.L.; Langdon, J.S.; Hyatt, A.; Humphrey, J.D. Mass mortality associated with a viral-induced vacuolating encephalopathy and retinopathy of larval and juvenile barramundi, Lates calcarifer Bloch. Aquaculture 1992, 103, 197–211. [Google Scholar] [CrossRef]
- Iwamoto, T.; Mise, K.; Takeda, A.; Okinaka, Y.; Mori, K.I.; Arimoto, M.; Okuno, T.; Nakai, T. Characterization of Striped jack nervous necrosis virus subgenomic RNA3 and biological activities of its encoded protein B2. J Gen Virol 2005, 86, 2807–2816. [Google Scholar] [CrossRef]
- Ertel, K.J.; Benefield, D.; Castaño-Diez, D.; Pennington, J.G.; Horswill, M.; den Boon, J.A.; Otegui, M.S.; Ahlquist, P. Cryo-electron tomography reveals novel features of a viral RNA replication compartment. Elife 2017, 6. [Google Scholar] [CrossRef]
- Bajpai, V.; Li, C.H.; Lai, C.S.; Fang, K.Y.; Chiang, M.H.; Chen, W.D.; Chen, W.Y.; Chen, C.W.; Wang, Y.S.; Chen, S.C.; et al. Translation of nervous necrosis virus involves eIF4E but not RPS6 phosphorylation and viral particle assembly in remodeled microtubule-organizing center. Virol J 2025, 22, 341. [Google Scholar] [CrossRef]
- Lin, C.H.; Christopher John, J.A.; Lin, C.H.; Chang, C.Y. Inhibition of nervous necrosis virus propagation by fish Mx proteins. Biochem Biophys Res Commun 2006, 351, 534–539. [Google Scholar] [CrossRef]
- Chen, C.W.; Wu, M.S.; Huang, Y.J.; Cheng, C.A.; Chang, C.Y. Recognition of Linear B-Cell Epitope of Betanodavirus Coat Protein by RG-M18 Neutralizing mAB Inhibits Giant Grouper Nervous Necrosis Virus (GGNNV) Infection. PLoS One 2015, 10, e0126121. [Google Scholar] [CrossRef]
- Lai, Y.-S.; Murali, S.; Chiu, H.-C.; Ju, H.-Y.; Lin, Y.-S.; Chen, S.-C.; Guo, I.-C.; Fang, K.; Chang, C.-Y. Propagation of yellow grouper nervous necrosis virus (YGNNV) in a new nodavirus-susceptible cell line from yellow grouper, Epinephelus awoara (Temminck & Schlegel), brain tissue. Journal of Fish Diseases 2001, 24, 299–309. [Google Scholar] [CrossRef]
- Cheng, C.A.; Lai, C.S.; Liao, F.E.; Yeh, S.L.; Chiang, M.H.; Li, C.H.; Fang, K.Y.; Liao, Y.C.; Hsu, H.C.; Bajpai, V.; et al. Giant grouper nervous necrosis virus subgenomic RNA3 is transcribed by a premature termination mechanism via intragenomic RNA-RNA pairing. Virol J 2025, 22, 340. [Google Scholar] [CrossRef]
- Lai, Y.-S.; Chiu, H.-C.; Murali, S.; Guo, I.-C.; Chen, S.-C.; Fang, K.; Chang, C.-Y. In vitro neutralization by monoclonal antibodies against yellow grouper nervous necrosis virus (YGNNV) and immunolocalization of virus infection in yellow grouper, Epinephelus awoara (Temminck & Schlegel). Journal of Fish Diseases 2001, 24, 237–244. [Google Scholar] [CrossRef]
- Guo, Y.X.; Chan, S.W.; Kwang, J. Membrane association of greasy grouper nervous necrosis virus protein A and characterization of its mitochondrial localization targeting signal. J Virol 2004, 78, 6498–6508. [Google Scholar] [CrossRef]
- Guo, Y.X.; Dallmann, K.; Kwang, J. Identification of nucleolus localization signal of betanodavirus GGNNV protein alpha. Virology 2003, 306, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.M.; Su, Y.L.; Shie, P.S.; Huang, S.L.; Yang, H.L.; Chen, T.Y. Grouper Mx confers resistance to nodavirus and interacts with coat protein. Dev Comp Immunol 2008, 32, 825–836. [Google Scholar] [CrossRef] [PubMed]
- Eichwald, C.; Arnoldi, F.; Laimbacher, A.S.; Schraner, E.M.; Fraefel, C.; Wild, P.; Burrone, O.R.; Ackermann, M. Rotavirus viroplasm fusion and perinuclear localization are dynamic processes requiring stabilized microtubules. PLoS One 2012, 7, e47947. [Google Scholar] [CrossRef]
- Smith, R.W.; Gray, N.K. Poly(A)-binding protein (PABP): a common viral target. Biochem J 2010, 426, 1–12. [Google Scholar] [CrossRef]
- Harb, M.; Becker, M.M.; Vitour, D.; Baron, C.H.; Vende, P.; Brown, S.C.; Bolte, S.; Arold, S.T.; Poncet, D. Nuclear localization of cytoplasmic poly(A)-binding protein upon rotavirus infection involves the interaction of NSP3 with eIF4G and RoXaN. J Virol 2008, 82, 11283–11293. [Google Scholar] [CrossRef]
- Blakqori, G.; van Knippenberg, I.; Elliott, R.M. Bunyamwera orthobunyavirus S-segment untranslated regions mediate poly(A) tail-independent translation. J Virol 2009, 83, 3637–3646. [Google Scholar] [CrossRef] [PubMed]
- Mai, W.; Huang, F.; Chen, H.; Zhou, Y.; Chen, Y. Nervous necrosis virus capsid protein exploits nucleolar phosphoprotein Nucleophosmin (B23) function for viral replication. Virus Res 2017, 230, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, Y.; Mori, Y.; Abe, T.; Yamashita, T.; Okamoto, T.; Ichimura, T.; Moriishi, K.; Matsuura, Y. Nucleolar protein B23 interacts with Japanese encephalitis virus core protein and participates in viral replication. Microbiol Immunol 2006, 50, 225–234. [Google Scholar] [CrossRef]
- Yoo, D.; Wootton, S.K.; Li, G.; Song, C.; Rowland, R.R. Colocalization and interaction of the porcine arterivirus nucleocapsid protein with the small nucleolar RNA-associated protein fibrillarin. J Virol 2003, 77, 12173–12183. [Google Scholar] [CrossRef]
- Slack, J.; Arif, B.M. The baculoviruses occlusion-derived virus: virion structure and function. Adv Virus Res 2007, 69, 99–165. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



