Submitted:
10 February 2026
Posted:
12 February 2026
You are already at the latest version
Abstract
Keywords:
MSC: 92C05, 92C15, 92C40, 92C45, 80Axx, 82Cxx, 82B35, 82C26
1. Introduction
2. A Historical Perspective of Photochemical Dissipative Structuring
3. Non-Linear Classical Irreversible Thermodynamic Theory
- 1.
- The existence of at least one relatively constant applied external generalized thermodynamic potential defining the environment - the applied thermodynamic forces.
- 2.
- The spontaneous generation of internal generalized thermodynamic flows resulting from these applied external generalized forces and the possibility of new internal forces that these flows themselves generate.
- 3.
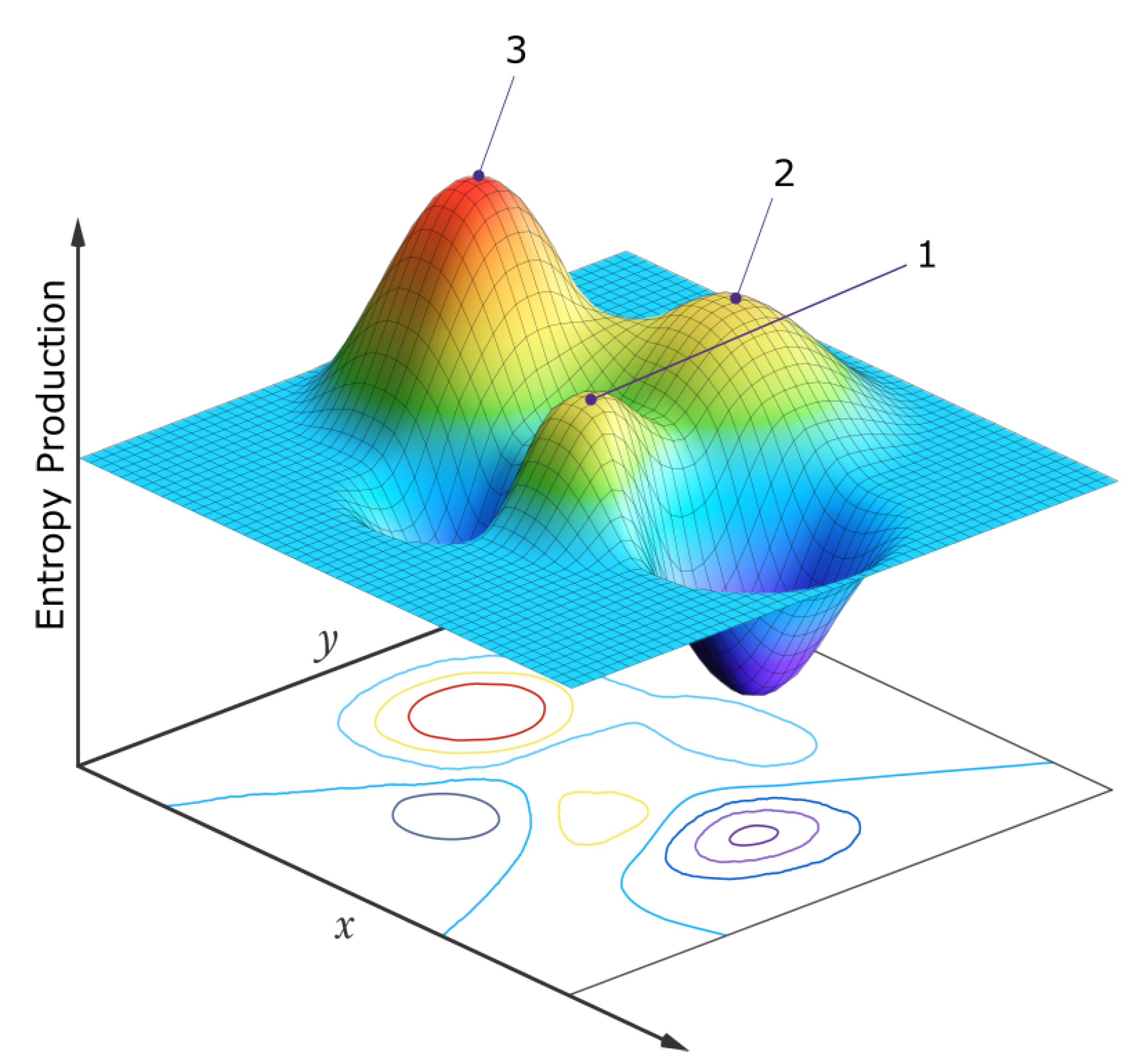
- The potentiality of various distinct sets of these internal forces and flows for non-linear systems for the same initial and boundary conditions, (i.e., multiple, locally stable, dissipative structures or processes, at stationary states) – each set of which can have a different rate of dissipation of the applied external potential (entropy production).
- 4.
- External or internal stochastic perturbations which, near a critical point, could cause the non-linear system to leave the local attractor basin in parameter space of one stationary state and evolve to that of another.
- 5.
- The non-deterministic (stochastic) tendency for evolution on perturbation to stationary states (dissipative structures) affording greater dissipation (entropy production), particularly through routes with autocatalytic and cross-catalytic steps, since these have a larger and thus more stable attractor basin in this generalized parameter space.
4. Molecular Dissipative Structuring
- 1.
- Sufficient energy per photon to overcome activation barriers, as well as sufficiently large photoreaction quantum efficiencies.
- 2.
- A general increase in photon extinction coefficients and wavelength bandwidth as the molecules evolve from simple precursors towards final pigments [55].
- 3.
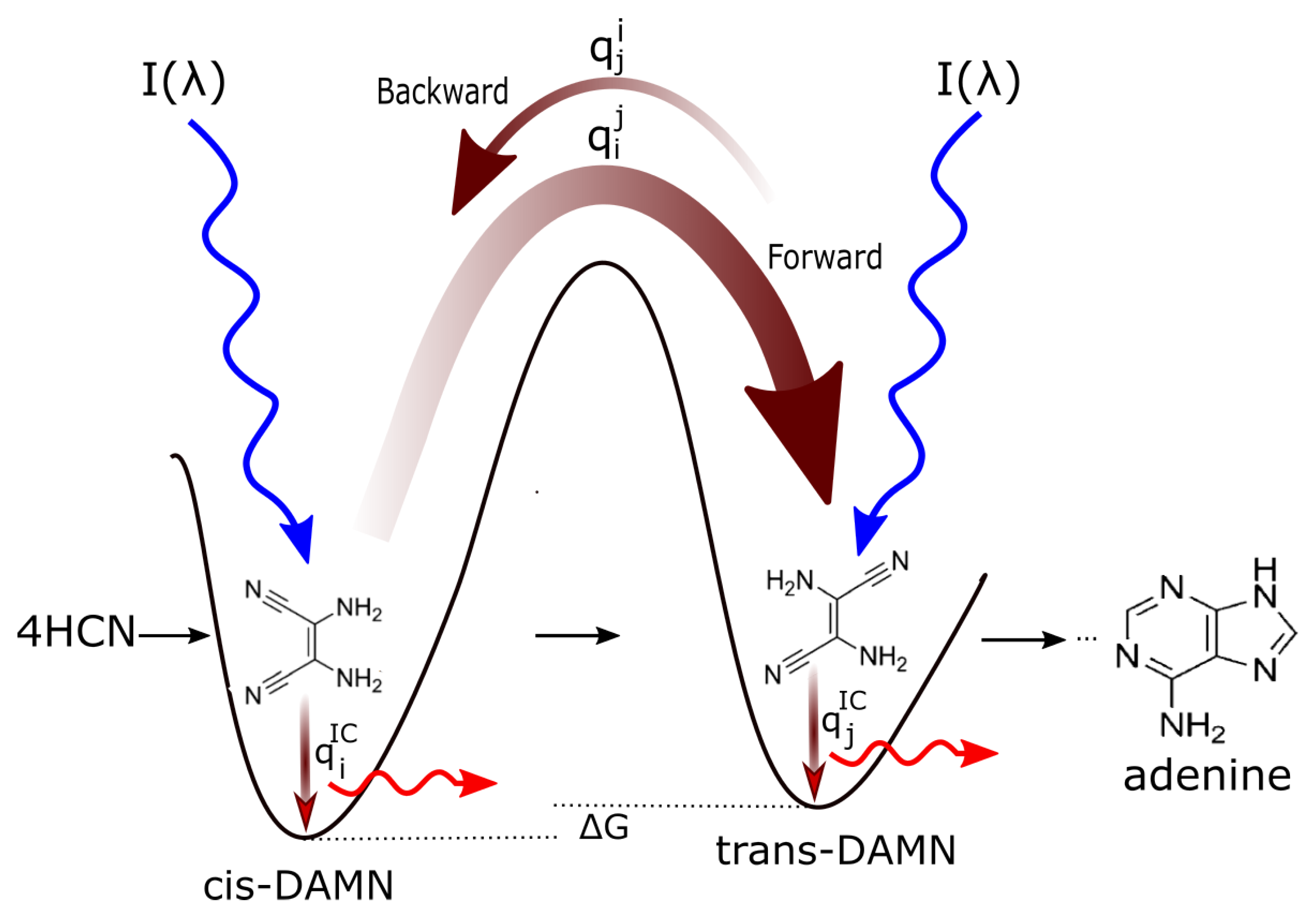
- The formation of conical intersections [15]connecting excited electronic states with the electronic ground state, allowing ultrafast (subpicosecond) radiationless dissipation (internal conversion).
- 4.
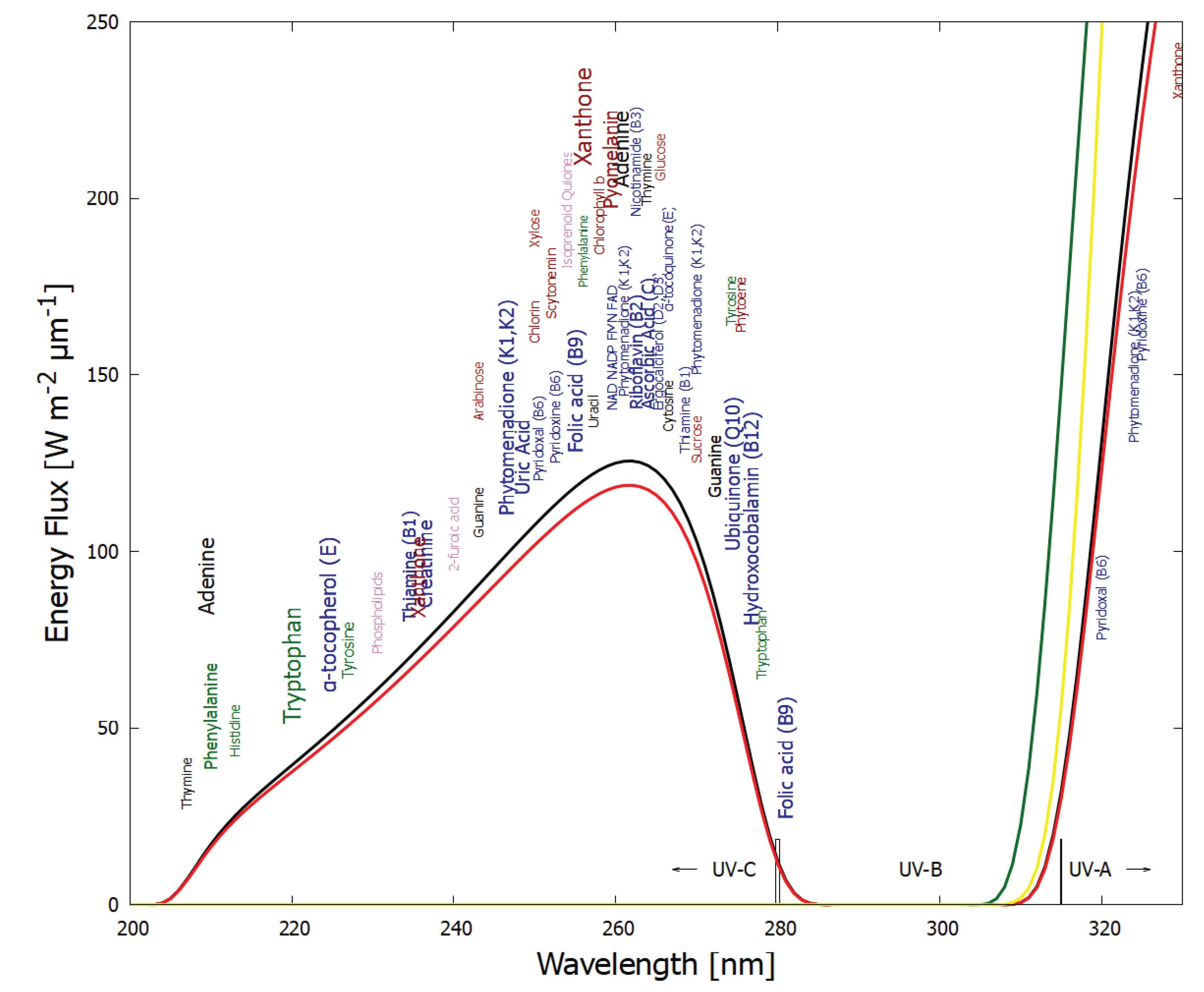
- A general trend towards dissipation of wavelengths of greater intensity of the prevailing surface solar spectrum.
- 5.
- Molecular ionization energies remaining greater than photon energies in the prevailing surface spectrum, inhibiting photon-induced degradation.
- 1.
- photon intensities at the different wavelengths, ,
- 2.
- the absorption cross section of the molecule as a function of wavelength ,
- 3.
- the widths of the phase-space paths leading to the particular conical intersection on the electronic excited state potential energy surface (i.e., the quantum efficiencies ) for particular molecular transformations or internal conversion. Reverse transformations , or transformations to other possible products (e.g., ), under the UV light are less probable if the quantum efficiencies are smaller (smaller phase-space path on the excited potential energy surface) as compared to the quantum efficiency for internal conversion to the ground state .
5. Examples of Molecular Dissipative Structuring
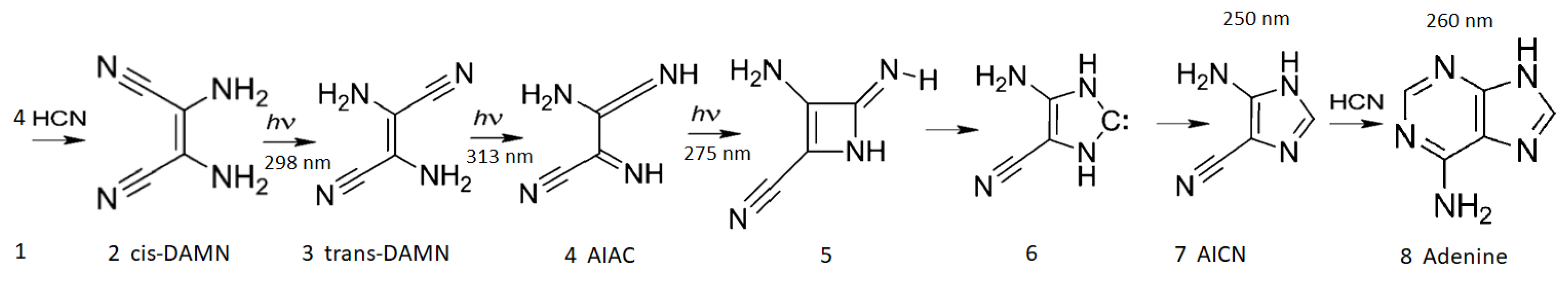
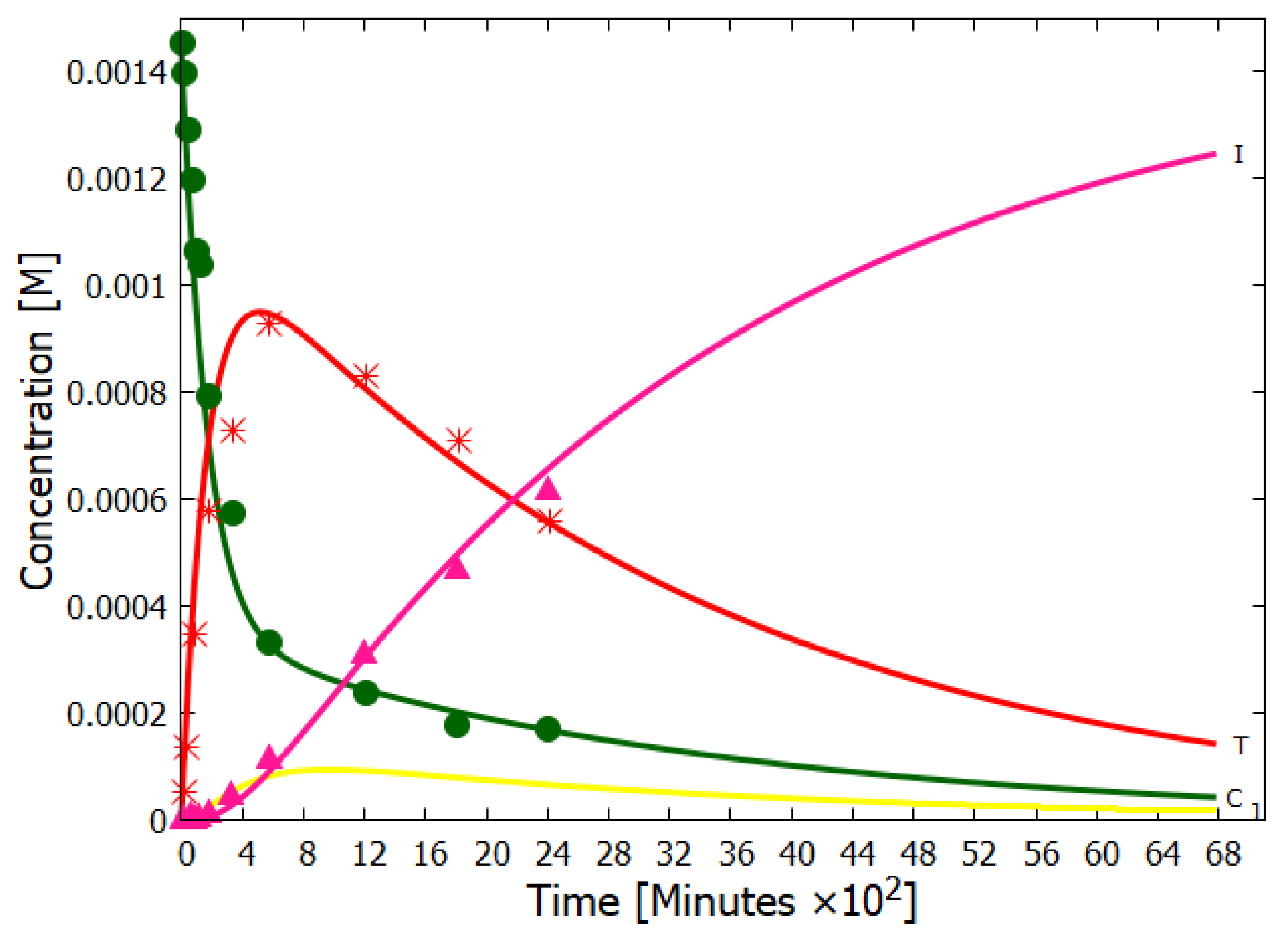
5.1. Nucleobases
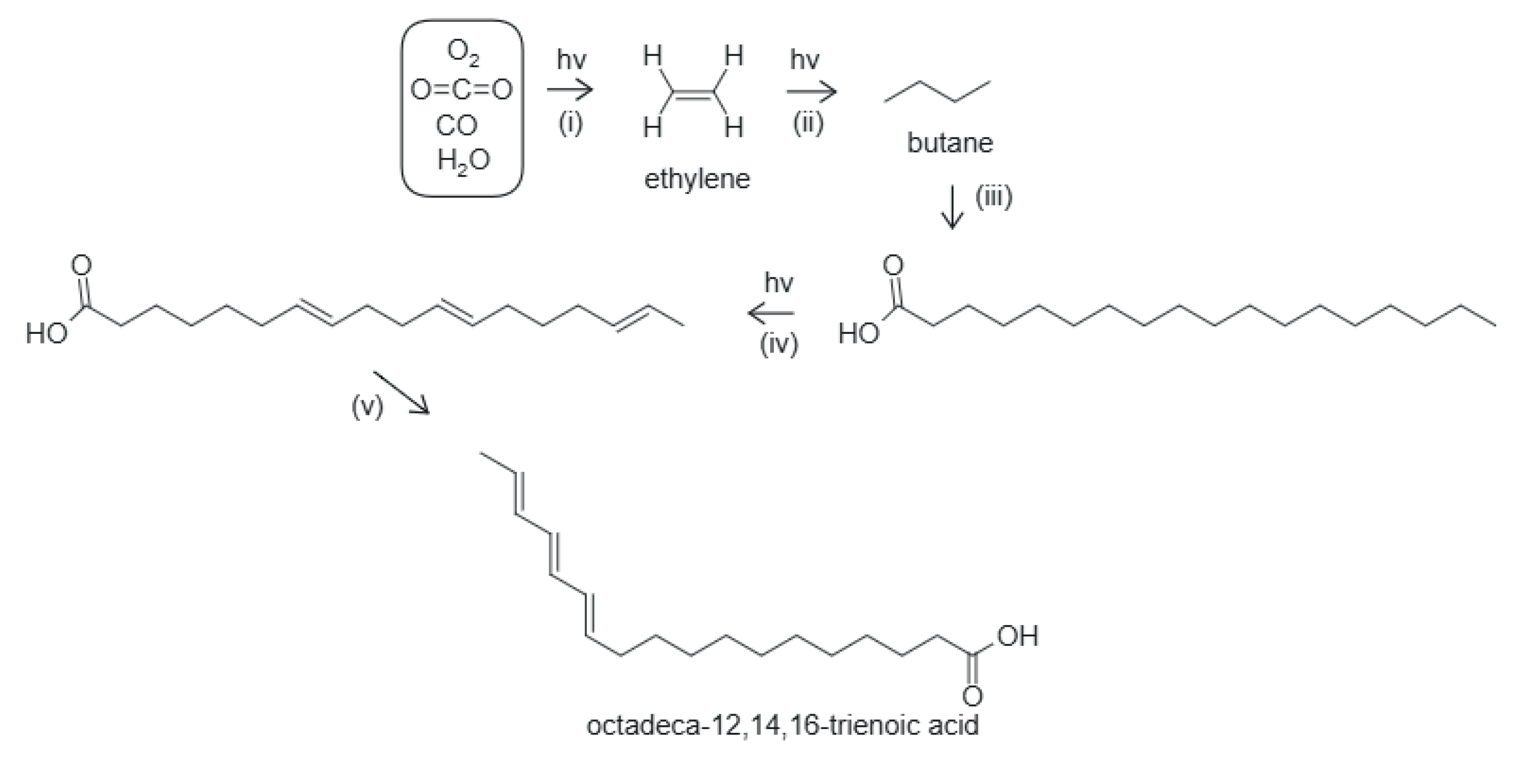
5.2. Fatty Acid Vesicles
- 1.
- UV-C-induced reduction of and CO in water saturated with these to form ethylene,
- 2.
- UV-C-induced polymerization of ethylene to form long hydrocarbon tails with an even number of carbon atoms,
- 3.
- oxidation and hydrolysis events to stop the growing of the chain and form the carboxyl group,
- 4.
- UV-C-induced excited-state dehydrogenation or hydrogen bond proton transfer of the tails to form a double bond,
- 5.
- double bond migration to give a conjugated diene or triene with a conical intersection and strong absorption within the Archean UV-C spectrum.
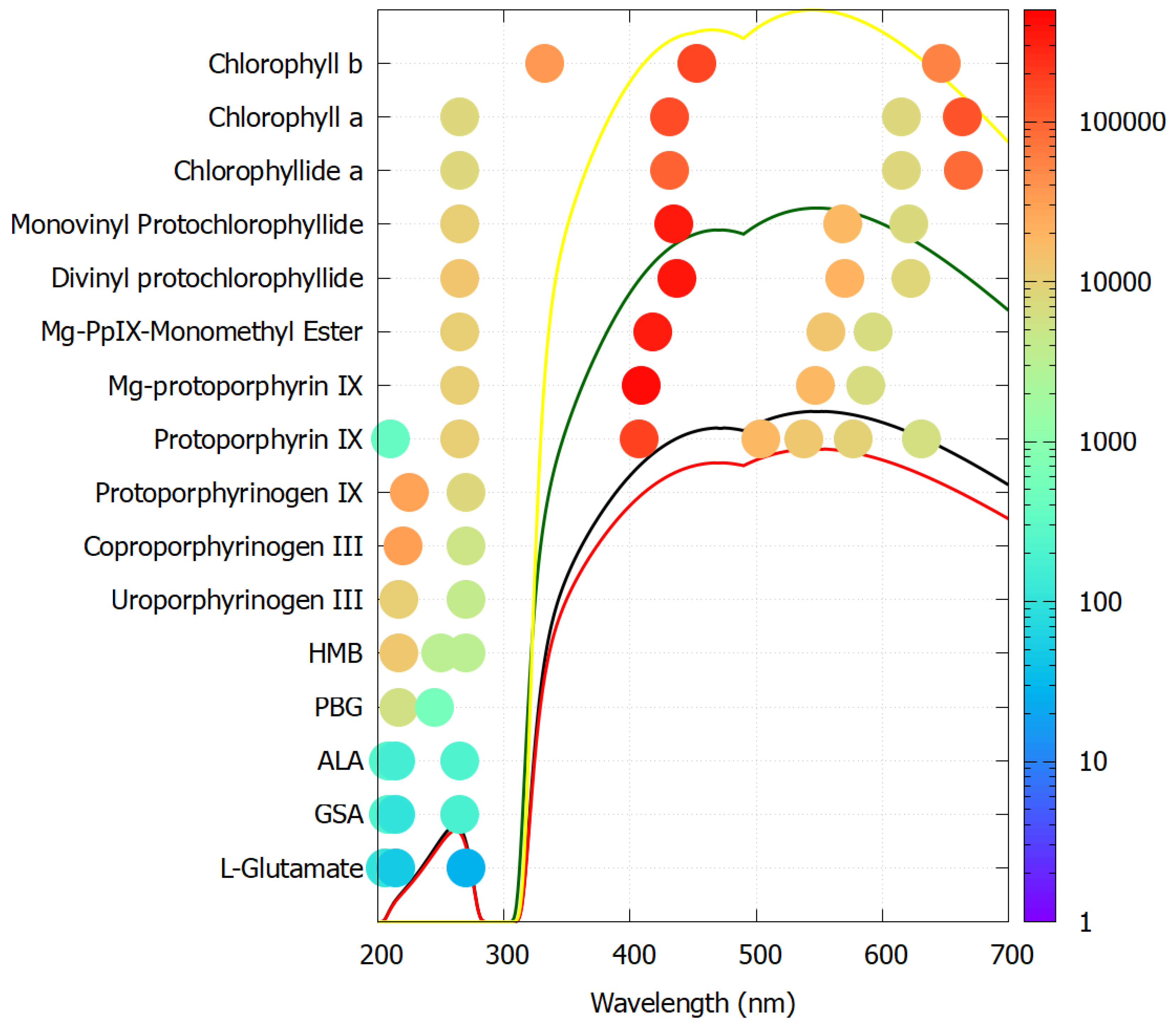
5.3. Pigments
6. The Fundamental Creative Force in Biology: Thermodynamic Selection of Dissipative Structuring
6.1. The Molecular Level
6.2. The Organism Level
6.3. The Ecosystem and Biosphere Levels
7. Conclusions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ALA | 5-Aminolevulinic Acid |
| ATP | Adenosine triphosphate |
| CIT | Classical Irreversible Thermodynamic theory |
| Carbon dioxide | |
| DNA | Deoxyribonucleic acid |
| GSA | Glutamate-1-Semialdehyde |
| S | Hydrogen sulfide |
| HCN | Hydrogen cyanide |
| HMB | Hydroxymethylbilane |
| LOV | Light-Oxygen-Voltage - organism blue-light-sensing protein modules |
| PBG | Porphobilinogen |
| RNA | Ribonucleic acid |
| Sulfur dioxide | |
| TDTOL | Thermodynamic Dissipation Theory of the Origin of Life |
| UV-A | Light within the region 315-400 nm |
| UV-B | Light within the region 280-315 nm |
| UV-C | Light within the region 100-280 nm |
| UV-C (hard) | Light in the region 100-205 nm |
| UV-C (soft) | Light within the region 205-285 nm |
References
- Prigogine, I. Introduction to Thermodynamics Of Irreversible Processes, third ed.; John Wiley & Sons, 1955. [Google Scholar]
- Prigogine, I. Introduction to Thermodynamics Of Irreversible Processes, third ed.; John Wiley & Sons: Hoboken, NJ, USA, 1967. [Google Scholar]
- Prigogine, I.; Nicolis, G. Biological order, structure and instabilities. Quarterly Reviews of Biophysics 1971, 4, 107–144. [Google Scholar] [CrossRef]
- Onsager, L. Reciprocal Relations in Irreversible Processes, I. Phys. Rev. 1931, 37, 405–426. [Google Scholar] [CrossRef]
- Onsager, L. Reciprocal Relations in Irreversible Processes, II. Phys. Rev. 1931, 38, 2265. [Google Scholar] [CrossRef]
- Onsager, L.; Machlup, S. Fluctuations and Irreversible Processes. Phys. Rev. 1953, 91, 1505–1512. [Google Scholar] [CrossRef]
- Vale, R.D.; Milligan, R.A. The way things move: Looking under the hood of molecular motor proteins. Science 2000, 288, 88–95. [Google Scholar] [CrossRef]
- Howard, J. Mechanics of Motor Proteins and the Cytoskeleton; Sinauer Associates: Sunderland, MA, 2001. [Google Scholar]
- Berg, H.C.; Brown, D.A. Chemotaxis in Escherichia coli analysed by three-dimensional tracking. Nature 1972, 239, 500–504. [Google Scholar] [CrossRef]
- Berg, H.C. E. coli in Motion; Springer: New York, 2003. [Google Scholar] [CrossRef]
- Zhang, B.; Cech, T.R. Peptide bond formation by in vitro selected ribozymes. Nature 1997, 390, 96–100. [Google Scholar] [CrossRef]
- Michaelian, K. Thermodynamic origin of life. ArXiv 2009, arXiv:physics.gen. [Google Scholar]
- Michaelian, K. Thermodynamic dissipation theory for the origin of life. Earth Syst. Dynam. 2011, 224, 37–51. Available online: https://esd.copernicus.org/articles/2/37/2011/esd-2-37-2011.html. [CrossRef]
- Michaelian, K. The Dissipative Photochemical Origin of Life: UVC Abiogenesis of Adenine. Entropy 2021, 23. Available online: https://www.mdpi.com/1099-4300/23/2/217. [CrossRef]
- Schuurman, M.S.; Stolow, A. Dynamics at Conical Intersections. Annu. Rev. Phys. Chem. 2018, 69, 427–450. [Google Scholar] [CrossRef]
- Meixnerová, J.; Blum, J.D.; Johnson, M.W.; Stüeken, E.E.; Kipp, M.A.; Anbar, A.D.; Buick, R. Mercury abundance and isotopic composition indicate subaerial volcanism prior to the end-Archean “whiff” of oxygen. Proceedings of the National Academy of Sciences 2021, 118, e2107511118. [Google Scholar] [CrossRef] [PubMed]
- Sagan, C. Ultraviolet Selection Pressure on the Earliest Organisms. J. Theor. Biol. 1973, 39, 195–200. [Google Scholar] [CrossRef]
- Meller, R.; Moortgat, G.K. Temperature dependence of the absorption cross sections of formaldehyde between 223 and 323 K in the wavelength range 225–375 nm. Journal of Geophysical Research: Atmospheres 2000, 105, 7089–7101. Available online: https://agupubs.onlinelibrary.wiley.com/doi/pdf/10.1029/1999JD901074. [CrossRef]
- Michaelian, K. The Non-Equilibrium Thermodynamics of Natural Selection: From Molecules to the Biosphere. Entropy 2023, 25. [Google Scholar] [CrossRef]
- Sagan, C. Radiation and the Origin of the Gene. Evolution 1957, 11, 40–55. [Google Scholar] [CrossRef]
- Mulkidjanian, A.Y.; Junge, W. Primordial UV-Protectors As Ancestors of the Photosynthetic Pigment-Proteins. In The Phototrophic Prokaryotes; Peschek, G.A., Löffelhardt, W., Schmetterer, G., Eds.; Springer: Boston, MA, 1999; pp. 669–682. [Google Scholar] [CrossRef]
- Mulkidjanian, A.Y.; Cherepanov, D.A.; Galperin, M.Y. Survival of the fittest before the beginning of life: selection of the first oligonucleotide-like polymers by UV light. BMC Evolutionary Biology 2003, 3, 12. [Google Scholar] [CrossRef] [PubMed]
- Solovchenko, A.; Merzlyak, M. Screening of visible and UV radiation as a photoprotective mechanism in plants. Russian Journal of Plant Physiology 2008, 55, 719–737. [Google Scholar] [CrossRef]
- Staleva, H.; et al. Mechanism of photoprotection in the cyanobacterial ancestor of plant antenna proteins. Nat. Chem. Biol. 2015, 11, 287–291. [Google Scholar] [CrossRef]
- Simkin, A.J.; Kapoor, L.; Doss, C.G.P.; Hofmann, T.A.; Lawson, T.; Ramamoorthy, S. The role of photosynthesis related pigments in light harvesting, photoprotection and enhancement of photosynthetic yield in plants. Photosynthesis Research 2022, 152, 23–42. [Google Scholar] [CrossRef]
- Zuo, G. Non-photochemical quenching (NPQ) in photoprotection: insights into NPQ levels required to avoid photoinactivation and photoinhibition. New Phytologist 2025. [Google Scholar] [CrossRef] [PubMed]
- Brøndsted Nielsen, S.; Sølling, T.I. Are Conical Intersections Responsible for the Ultrafast Processes of Adenine, Protonated Adenine, and the Corresponding Nucleosides? ChemPhysChem 2005, 6, 1276–1281. Available online: https://chemistry-europe.onlinelibrary.wiley.com/doi/pdf/10.1002/cphc.200400644. [CrossRef] [PubMed]
- Barbatti, M.; Aquino, A.; Szymczak, J.; Nachtigallová, D.; Hobza, P.; Lischka, H. Relaxation mechanisms of UV-photoexcited DNA and RNA nucleobases. Proc Natl Acad Sci U S A 2010, 107, 21453–21458. [Google Scholar] [CrossRef]
- Salmahaminati; Roca-Sanjuán, D. The Photophysics and Photochemistry of Phenylalanine, Tyrosine, and Tryptophan: A CASSCF/CASPT2 Study This study uses CASSCF/CASPT2 to explore conical intersections and non-radiative decay pathways in the three aromatic amino acids. In ACS Omega 2024; Salmahaminati, Roca-Sanjuán, Daniel, Eds.; first and corresponding author, Chemistry Department, Faculty of Mathematics and Natural Science, Islamic University of Indonesia Instituto de Ciencia Molecular: Universitat de València, 6 August 2024; Volume 9, pp. 35356–35363. [Google Scholar] [CrossRef]
- Lechuga, I.; Michaelian, K. Fatty Acid Vesicles as Hard UV-C Shields for Early Life. Foundations 2023, 3, 99–114. [Google Scholar] [CrossRef]
- Brasier, M.D.; Antcliffe, J.; Saunders, M.; Wacey, D. A Hydrothermal-Sedimentary Context for the Origin of Life. Astrobiology 2018, 18, 429–445. [Google Scholar] [CrossRef]
- Subbotin, V.; Fiksel, G. Aquatic Ferrous Solutions of Prebiotic Mineral Salts as Strong UV Protectants and Possible Loci of Life Origin. Astrobiology 2023, 23, 741–745. [Google Scholar] [CrossRef] [PubMed]
- Martin, W.; Baross, J.; Kelley, D. Hydrothermal vents and the origin of life. Nature Reviews Microbiol. 2008, 6, 805–814. [Google Scholar] [CrossRef]
- Mulkidjanian, A.Y.; Bychkov, A.Y.; Dibrova, D.V.; Galperin, M.Y.; Koonin, E.V. Origin of first cells at terrestrial, anoxic geothermal fields. Proceedings of the National Academy of Sciences 2012, 109, E821–E830. [Google Scholar] [CrossRef]
- Ferris, J.P.; Orgel, L.E. An Unusual Photochemical Rearrangement in the Synthesis of Adenine from Hydrogen Cyanide. J. Am. Chem. Soc. 1966, 88, 1074–1074. [Google Scholar] [CrossRef]
- Sanchez, R.A.; Ferris, J.P.; Orgel, L.E. Studies in Prebiodc Synthesis II: Synthesis of Purine Precursors and Amino Acids from Aqueous Hydrogen Cyanide. J. Mol. Biol. 1967, 80, 223–253. [Google Scholar]
- Sanchez, R.A.; Ferris, J.P.; Orgel, L.E. Studies in Prebiodc Synthesis IV: Conversion of 4-Aminoimidazole-5-carbonitrile Derivatives to Purines. J. Mol. Biol. 1968, 38, 121–128. [Google Scholar] [CrossRef]
- Ferris, J.; Joshi, P.; Edelson, E.; Lawless, J. HCN: a plausible source of purines, pyrimidines and amino acids on the primitive Earth. Journal of molecular evolution 1978, 11, 293–311. [Google Scholar] [CrossRef]
- Roy, D.; Najafian, K.; Schleyer, P.V.R. Chemical Evolution: the Mechanism Of The Formation Of Adenine Under Prebiotic Conditions. Proc. Natl. Acad. Sci. U.S.A. 2007, 104, 17272–17277. [Google Scholar] [CrossRef]
- Barks, H.L.; Buckley, R.; Grieves, G.A.; Di Mauro, E.; Hud, N.V.; Orlando, T.M. Guanine, Adenine, and Hypoxanthine Production in UV-Irradiated Formamide Solutions: Relaxation of the Requirements for Prebiotic Purine Nucleobase Formation. ChemBioChem 2010, 11, 1240–1243. Available online: https://chemistry-europe.onlinelibrary.wiley.com/doi/pdf/10.1002/cbic.201000074. [CrossRef] [PubMed]
- Saladino, R.; Crestini, C.; Pino, S.; Costanzo, G.; Di Mauro, E. Formamide and the origin of life. Physics of Life Reviews 2012, 9, 84–104. [Google Scholar] [CrossRef] [PubMed]
- Botta, L.; Bizzarri, B.M.; Piccinino, D.; Fornaro, T.; Brucato, J.R.; Saladino, R. Prebiotic synthesis of carboxylic acids, amino acids and nucleic acid bases from formamide under photochemical conditions. Eur. Phys. J. Plus 2017, 132, 317. [Google Scholar] [CrossRef]
- Sagan, C.; Khare, B.N. Long-Wavelength Ultraviolet Photoproduction of Amino Acids on the Primitive Earth. Science 1971, 173, 417–420. Available online: https://science.sciencemag.org/content/173/3995/417.full.pdf. [CrossRef]
- Ponnamperuma; Mariner, R. Formation of Ribose and Deoxyribose by Ultraviolet Irradiation of Formaldehyde in Water. Rad. Res. 1963, 19, 183. [Google Scholar]
- Ritson, D.; Sutherland, J. Prebiotic synthesis of simple sugars by photoredox systems chemistry. Nature Chem. 2012, 4, 895–899. [Google Scholar] [CrossRef]
- Ponnamperuma, C.; Sagan, C.; Mariner, R. Synthesis of adenosine triphosphate under possible primitive Earth conditions. Nature 1963, 199, 222–226. [Google Scholar] [CrossRef]
- Ponnamperuma; Mariner, R.; Sagan, C. Formation of Adenosine by Ultraviolet Irradiation of a Solution of Adenine and Ribose. Nature 1963, 198, 1199–1200. [Google Scholar] [CrossRef]
- Ponnamperuma, C.; Shimoyama, A.; Friebele, E. Possible Role of Ultraviolet Radiation in Prebiotic Synthesis. Origins of Life 1982, 12, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Kolesnikov, M.P.; Telegina, T.A.; Lyudnikova, T.A.; Kritsky, M.S. Abiogenic photophosphorylation of ADP to ATP sensitized by flavoproteinoid microspheres. Origins of Life and Evolution of Biospheres 2008, 38, 243–256. [Google Scholar] [CrossRef]
- Telegina, T.A.; Kolesnikov, M.P.; Buglak, A.A.; Vechtomova, Y.L.; Kritsky, M.S. Abiotic model of photophosphorylation of ADP to ATP: Characteristics of photoreceptor pigments and the role of organosilicate matrix. Paleontological Journal 2013, 47, 999–1005. [Google Scholar] [CrossRef]
- Powner, M.; Gerland, B.; Sutherland, J. Synthesis of activated pyrimidine ribonucleotides in prebiotically plausible conditions. Nature 2009, 459, 239–242. [Google Scholar] [CrossRef] [PubMed]
- Boulanger, E.; Anoop, A.; Nachtigallova, D.; Thiel, W.; Barbatti, M. Photochemical Steps in the Prebiotic Synthesis of Purine Precursors from HCN. Angew. Chem. Int. 2013, 52, 8000–8003. [Google Scholar] [CrossRef]
- Michaelian, K. The Pigment World: Life’s Origins as Photon-Dissipating Pigments. Life 2024, 14. [Google Scholar] [CrossRef] [PubMed]
- Michaelian, K. Non-Equilibrium Thermodynamic Foundations of the Origin of Life. Foundations 2022, 2, 308–337. [Google Scholar] [CrossRef]
- Michaelian, K. Microscopic Dissipative Structuring and Proliferation at the Origin of Life. Heliyon 2017, 3, e00424. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5647473/. [CrossRef]
- Michaelian, K.; Rodriguez, O. Prebiotic fatty acid vesicles through photochemical dissipative structuring. Revista Cubana de Química 2019, 31, 354–370. [Google Scholar]
- Michaelian, K.; Cano, R.E. A Photon Force and Flow for Dissipative Structuring: Application to Pigments, Plants and Ecosystems. Entropy 2022, 24, 76. [Google Scholar] [CrossRef]
- Hernández, C.; Michaelian, K. Dissipative Photochemical Abiogenesis of the Purines. Entropy 2022, 24, 1027. [Google Scholar] [CrossRef]
- Michaelian, K.; Santillán Padilla, N. DNA Denaturing through Photon Dissipation: A Possible Route to Archean Non-enzymatic Replication. bioRxiv. 2014. Available online: https://www.biorxiv.org/content/early/2014/11/24/009126.full.pdf. [CrossRef]
- Michaelian, K.; Santillan, N. UVC photon-induced denaturing of DNA: A possible dissipative route to Archean enzyme-less replication. Heliyon 2019, 5, e01902. Available online: https://www.heliyon.com/article/e01902. [CrossRef]
- Michaelian, K. Homochirality through Photon-Induced Denaturing of RNA/DNA at the Origin of Life. Life 2018, 8. Available online: http://www.mdpi.com/2075-1729/8/2/21. [CrossRef]
- Mejía Morales, J.; Michaelian, K. Photon Dissipation as the Origin of Information Encoding in RNA and DNA. Entropy 2020, 22. Available online: https://www.mdpi.com/1099-4300/22/9/940. [CrossRef]
- Michaelian, K.; Simeonov, A. Thermodynamic explanation of the cosmic ubiquity of organic pigments. Astrobiol. Outreach 2017, 5, 156. Available online: https://www.longdom.org/open-access/thermodynamic-explanation-for-the-cosmic-ubiquity-of-organicpigments-2332-2519-1000156.pdf. [CrossRef]
- Pollack, J.B.; Toon, O.B.; Whitten, R.C.; Boese, R.; Ragent, B.; Tomasko, M.; Esposito, L.; Travis, L.; Wiedman, D. Distribution and source of the UV absorption in Venus’ atmosphere. Journal of Geophysical Research 1980, 85, 8141–8150. [Google Scholar] [CrossRef]
- Eigenbrode, J.L.; Summons, R.E.; Steele, A.; Freissinet, C.; Millan, M.; Navarro-González, R.; Sutter, B.; Archer, P.D., Jr.; Franz, H.B.; Glavin, D.P.; et al. Organic matter preserved in 3-billion-year-old mudstones at Gale crater, Mars. Science 2018, 360, 1096–1101. [Google Scholar] [CrossRef] [PubMed]
- Feldman, P.D.; Weaver, H.A.; Festou, M.C. IUE observations of the UV spectrum of comet Bradfield. Nature 1980, 286, 132–135. [Google Scholar] [CrossRef]
- Pering, K.L.; Ponnamperuma, C. Aromatic hydrocarbons in the Murchison meteorite. Science 1971, 173, 237–239. [Google Scholar] [CrossRef] [PubMed]
- Botta, O.; Martins, Z.; Emmenegger, C.; Dworkin, J.; Glavin, D.; Harvey, R.; Zenobi, R.; Bada, J.; Ehrenfreund, P. Polycyclic aromatic hydrocarbons and amino acids in meteorites and ice samples from LaPaz Icefield, Antarctica. Meteoritics & Planetary Science 2008, 43, 1465–1480. Available online: https://onlinelibrary.wiley.com/doi/pdf/10.1111/j.1945-5100.2008.tb01021.x. [CrossRef]
- Buhl, D. Cosmochemical Evolution and the Origins of Life: Proceedings of the Fourth International Conference on the Origin of Life and the First Meeting of the International Society for the Study of the Origin of Life, June 25-28, 1973. Volume I: Invited Papers; Springer, 1973; chapter Galactic Clouds of Organic Molecules.
- Sagan, C.; Khare, B.N. Tholins: organic chemistry of interstellar grains and gas. Nature 1979, 277, 102–107. [Google Scholar] [CrossRef]
- Stecher, T.P. Interstellar Extinction in the Ultraviolet. The Astrophysical Journal 1965, 142, 1683–1684. [Google Scholar] [CrossRef]
- Glansdorff, P. Living Systems as Energy Converters. In chapter Energetic evolution of complex networks of reactions; North-Holland Publishing Company, 1977; pp. 41–54. [Google Scholar]
- Michaelian, K.; Simeonov, A. Fundamental molecules of life are pigments which arose and co-evolved as a response to the thermodynamic imperative of dissipating the prevailing solar spectrum. Biogeosciences 2015, 12, 4913–4937. Available online: https://bg.copernicus.org/articles/12/4913/2015/. [CrossRef]
- Michaelian, K. Thermodynamic Dissipation Theory of the Origin and Evolution of Life: Salient characteristics of RNA and DNA and other fundamental molecules suggest an origin of life driven by UV-C light; Printed by; CreateSpace: Mexico City, 2016; ISBN 9781541317482. Self-published. [Google Scholar]
- Prigogine, I.; Nicolis, G.; Babloyantz, A. Thermodynamics of evolution I. Physics Today 1972, 25, 23–28. [Google Scholar] [CrossRef]
- Babloyantz, A.; Hiernaux, J. Models for cell differentiation and generation of polarity in diffusion-governed morphogenetic fields. Bulletin of Mathematical Biology 1975, 37, 637–657. [Google Scholar] [CrossRef]
- Pascal, R.; Pross, A. On the Chemical Origin of Biological Cognition. Life 2022, 12. [Google Scholar] [CrossRef]
- Vitas, M. Towards a Possible Definition of Consciousness. BioSystems 2025, 254, 105526. [Google Scholar] [CrossRef]
- Michaelian, K. Thermodynamic stability of ecosystems. Journal of Theoretical Biology 2005, 237, 323–335. Available online: https://www.sciencedirect.com/science/article/pii/S0022519305001839?via%3Dihub. [CrossRef]
- Kleidon, A.; Malhi, Y.; Cox, P.M. Maximum entropy production in environmental and ecological systems. Philosophical Transactions of the Royal Society B: Biological Sciences 2010, 365, 1297–1302. Available online: https://royalsocietypublishing.org/doi/pdf/10.1098/rstb.2010.0018. [CrossRef]
- Kleidon, A. Maximum entropy production and general trends in biospheric evolution. Paleontological Journal 2009, 43, 980–985. [Google Scholar] [CrossRef]
- Michaelian, K. In The Biosphere; Ishwaran, N., Ed.; INTECH: London, 2012; chapter The biosphere: A thermodynamic imperative, pp. 51–60.
- Michaelian, K. Biological catalysis of the hydrological cycle: life’s thermodynamic function. Hydrol. Earth Syst. Sci. 2012, 16, 2629–2645. Available online: www.hydrol-earth-syst-sci.net/16/2629/2012/. [CrossRef]
- Prigogine, I.; Nicolis, G. On Symmetry-Breaking Instabilities in Dissipative Systems. J. Chem. Phys. 1967, 46, 3542. [Google Scholar] [CrossRef]
- Glansdorff, P.; Prigogine, I. Thermodynamic Theory of Structure, Stability and Fluctuations; Wiley - Interscience: Hoboken, NJ, USA, 1971. [Google Scholar]
- Hill, A. Entropy production as the selection rule between different growth morphologies. Nature 1990, 348, 426–428. [Google Scholar] [CrossRef]
- Atanasov, M.; Ganyushin, D.; Pantazis, D.A.; Sivalingam, K.; Neese, F. Vibronic Coupling in Inorganic Systems: Photochemistry, Conical Intersections, and the Jahn-Teller and Pseudo-Jahn-Teller Effects. Inorganic Chemistry 2011, 50, 7460–7477. [Google Scholar] [CrossRef]
- Minard, R.D.; Matthews, C.N. HCN World: Establishing Proteininucleic Acid Life via Hydrogen Cyanide Polymers. Abstr. Pap. Am. Chem. Soc. 2004, 228, U963–U963. [Google Scholar]
- Matthews, C.N. Origins: Genesis, Evolution and Diversity of Life. In Series: Cellular Origin and Life in Extreme Habitats and Astrobiology; Seckbach, J., Ed.; Kluwer: Dordrecht; chapter The HCN World, 2004; Vol. 6, pp. 121–135. [Google Scholar]
- Catling, D.C.; Zahnle, K.J. The Archean atmosphere. Science Advances 2020, 6, eaax1420. [Google Scholar] [CrossRef]
- Trainer, M.G.; Jimenez, J.L.; Yung, Y.L.; Toon, O.B.; Tolbert, M.A. Nitrogen Incorporation in CH4-N2 Photochemical Aerosol Produced by Far UV Irradiation. NASA archives. 2012. Available online: https://ntrs.nasa.gov/archive/nasa/casi.ntrs.nasa.gov/20120009529.pdf.
- Rimmer, P.B.; Rugheimer, S. Hydrogen cyanide in nitrogen-rich atmospheres of rocky exoplanets. Icarus 2019, 329, 124–137. [Google Scholar] [CrossRef]
- Koch, T.; Rodehorst, R. Quantitative investigation of the photochemical conversion of diaminomaleonitrile to diaminofumaronitrile and 4-amino-5-cyanoimidazole. J. Am. Chem. Soc. 1974, 96, 6707–6710. [Google Scholar] [CrossRef]
- Budin, I.; Prywes, N.; Zhang, N.; Szostak, J.W. Chain-Length Heterogeneity Allows for the Assembly of Fatty Acid Vesicles in Dilute Solutions. Biophysical Journal 2014, 107, 1582–1590. [Google Scholar] [CrossRef] [PubMed]
- Griffith, E.C.; Rapf, R.J.; Shoemaker, R.K.; Carpenter, B.K.; Vaida, V. Photoinitiated Synthesis of Self-Assembled Vesicles. Journal of the American Chemical Society 2014, 136, 3784–3787. [Google Scholar] [CrossRef] [PubMed]
- Martin, N.; Douliez, J.P. Fatty Acid Vesicles and Coacervates as Model Prebiotic Protocells. ChemSystemsChem 2021, 3, e2100024. Available online: https://chemistry-europe.onlinelibrary.wiley.com/doi/pdf/10.1002/syst.202100024. [CrossRef]
- Gözen, I.; Köksal, E.S.; Põldsalu, I.; Xue, L.; Spustova, K.; Pedrueza-Villalmanzo, E.; Ryskulov, R.; Meng, F.; Jesorka, A. Protocells: Milestones and Recent Advances. Small 2022, 18, 2106624. Available online: https://onlinelibrary.wiley.com/doi/pdf/10.1002/smll.202106624. [CrossRef]
- Schulz, H. Short history and present trends of Fischer-Tropsch synthesis. Applied Catalysis A: General 1999, 186, 3–12. [Google Scholar] [CrossRef]
- Getoff, N. Reduktion der Kohlensäure in wässeriger Lösung unter Einwirkung von UV-licht. Zeitschrift für Naturforschung B 1962, 17, 87–90. [Google Scholar] [CrossRef]
- Tan, S.S.; Zou, L.; Hu, E. Photocatalytic reduction of carbon dioxide into gaseous hydrocarbon using TiO2 pellets. Catalysis Today 2006, 115, 269–273. [Google Scholar] [CrossRef]
- Yu, L.; Razzaq, A.; Gooding, J.J.; Tilley, R.D. Plasmon-assisted photocatalytic CO2 reduction to multi-carbon products. Nanophotonics 2023. [Google Scholar] [CrossRef]
- Serrano-Perez, J.J.; de Vleeschouwer, F.; de Proft, F.; Mendive-Tapia, D.; Bearpark, M.J.; Robb, M.A. How the Conical Intersection Seam Controls Chemical Selectivity in the Photocycloaddition of Ethylene and Benzene. J. Org. Chem. 2013, 78, 1874–1886. [Google Scholar] [CrossRef] [PubMed]
- Vicente, A.; Antunes, R.; Almeida, D.; Franco, I.J.A.; Hoffmann, S.V.; Mason, N.J.; Eden, S.; Duflot, D.; Canneaux, S.; Delwiche, J.; et al. Photoabsorption measurements and theoretical calculations of the electronic state spectroscopy of propionic, butyric, and valeric acids. Phys. Chem. Chem. Phys. 2009, 11, 5729–5741. [Google Scholar] [CrossRef]
- Demchenko, A.P. Proton transfer reactions: From photochemistry to biochemistry and bioenergetics. BBA Advances 2023, 3, 100085. [Google Scholar] [CrossRef]
- Mandal, T.K.; Chatterjee, S.N. Ultraviolet- and Sunlight-Induced Lipid Peroxidation in Liposomal Membrane. Radiation Research 1980, 83, 290–302. [Google Scholar] [CrossRef]
- Lewis, G.N.; Calvin, M. The Color of Organic Substances. Chemical Reviews 1949, 25, 273–328. [Google Scholar] [CrossRef]
- Bowman, C.N.; Kloxin, C.J. Toward an enhanced understanding and implementation of photopolymerization reactions. AIChE J. 2008, 54, 2775–2795. [Google Scholar] [CrossRef]
- Mochida, M.; Kitamori, Y.; Kawamura, K.; Nojiri, Y.; Suzuki, K. Fatty acids in the marine atmosphere: Factors governing their concentrations and evaluation of organic films on sea-salt particles. Journal of Geophysical Research: Atmospheres 2002, 107, AAC 1–1–AAC 1–10. Available online: https://agupubs.onlinelibrary.wiley.com/doi/pdf/10.1029/2001JD001278. [CrossRef]
- Wellen, B.A.; Lach, E.A.; Allen, H.C. Surface pKa of octanoic, nonanoic, and decanoic fatty acids at the air-water interface: applications to atmospheric aerosol chemistry. Phys. Chem. Chem. Phys. 2017, 19, 26551–26558. [Google Scholar] [CrossRef]
- Celani, P.; Garavelli, M.; Ottani, S.; Bemardi, F.; Robb, M.A.; Olivucci, M. Molecular “Trigger” for Radiationless Deactivation of Photoexcited Conjugated Hydrocarbons. J. Am. Chem. Soc. 1995, 117, 11584–11585. [Google Scholar] [CrossRef]
- Fan, Y.; Fang, Y.; Ma, L. The self-crosslinked ufasome of conjugated linoleic acid: Investigation of morphology, bilayer membrane and stability. Colloids and Surfaces B: Biointerfaces 2014, 123, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Calvin, M. Occurrence of fatty acids and aliphatic hydrocarbons in a 3.4 billion-year-old sediment. Nature 1969, 224, 576–577. [Google Scholar] [CrossRef] [PubMed]
- Van Hoeven, W.; Maxwell, J.; Calvin, M. Fatty acids and hydrocarbons as evidence of life processes in ancient sediments and crude oils. Geochimica et Cosmochimica Acta 1969, 33, 877–881. [Google Scholar] [CrossRef]
- Kritsky, M.S.; Kolesnikov, M.P.; Telegina, T.A. Modeling of abiogenic synthesis of ATP. Doklady Biochemistry and Biophysics 2007, 417, 313–315. [Google Scholar] [CrossRef]
- Michaelian, K.; Simeonov, A. The Dissipative Photochemical Origin of Photosynthesis. in process 2026. [Google Scholar]
- Niyogi, K.K.; Wolosiuk, R.A.; Malkin, R. Photosynthesis. In Biochemistry and Molecular Biology of Plants, 2 ed.; Buchanan, B.B., Gruissem, W., Jones, R.L., Eds.; Wiley Blackwell: Chichester, West Sussex, 2015; pp. 515–519. [Google Scholar]
- Falahati, K.; Hamerla, C.; Huix-Rotllant, M.; Burghardt, I. Ultrafast photochemistry of free-base porphyrin: a theoretical investigation of B → Q internal conversion mediated by dark states. Physical Chemistry Chemical Physics 2018, 20, 12483–12492. [Google Scholar] [CrossRef]
- Vass, I. Molecular mechanisms of photodamage in the Photosystem II complex. Biochimica et Biophysica Acta (BBA) - Bioenergetics;Photosystem II 2012, 1817, 209–217. [Google Scholar] [CrossRef] [PubMed]
- Gould, S.J. The structure of evolutionary theory / Stephen Jay Gould; Belknap Press of Harvard University Press: Cambridge, MA, USA, 2002; pp. 1–1433. [Google Scholar]
- Chen, R.F. Fluorescence Quantum Yields of Tryptophan and Tyrosine. Analytical Letters 1967, 1, 35–42. [Google Scholar] [CrossRef]
- Yarus, M.; Widmann, J.; Knight, R. RNA-Amino Acid Binding: A Stereochemical Era for the Genetic Code. J Mol Evol 2009, 69, 406–429. [Google Scholar] [CrossRef] [PubMed]
- Nikogosyan, D.N.; Letokhov, V.S. Fluorescence of the purine and pyrimidine bases of the nucleic acids in neutral aqueous solution at 300 degrees K. Doklady Akademii Nauk SSSR 1970, 190, 146–149. [Google Scholar]
- Dimicoli, J.L.; Hélène, C. Specific Recognition of Single-Stranded Regions in Ultraviolet-Irradiated and Heat-Denatured DNA by Tryptophan-Containing Peptides. Proceedings of the National Academy of Sciences 1974, 71, 3185–3188. [Google Scholar] [CrossRef]
- Blicharska, Z.; Antosiewicz, J.; Dygut, J.M.; Słowiński, J. Fluorescence quenching studies of Trp repressor-operator interaction. Journal of Protein Chemistry 1999, 18, 823–830. [Google Scholar] [CrossRef]
- Gehlen, M.H. The centenary of the Stern-Volmer equation of fluorescence quenching: From the single line plot to the SV quenching map. Journal of Photochemistry and Photobiology C: Photochemistry Reviews 2020, 42, 100338. [Google Scholar] [CrossRef]
- Wehbi, S.; Wheeler, A.; Morel, B.; Manepalli, N.; Minh, B.Q.; Lauretta, D.S.; Masel, J. Order of amino acid recruitment into the genetic code resolved by last universal common ancestor’s protein domains. Proceedings of the National Academy of Sciences 2024, 121, e2410311121. [Google Scholar] [CrossRef]
- Ripple, W.J.; Beschta, R.L. Trophic cascades in Yellowstone: The first 15years after wolf reintroduction. Biological Conservation 2012, 145, 205–213. [Google Scholar] [CrossRef]
- Barrick, J.E.; Yu, D.S.; Yoon, S.H.; Jeong, H.; Oh, T.K.; Schneider, D.; Lenski, R.E.; Kim, J.F. Genome evolution and adaptation in a long-term experiment with Escherichia coli. Nature 2009, 461, 1243–1247. [Google Scholar] [CrossRef] [PubMed]
- Swenson, R. The Cybernetics of Complex Systems: Self-organization, Evolution, and Social Change chapter End-directed physics and evolutionary ordering: Obviating the problem of the population of one; Intersystems Publications: Cambridge, MA, USA, 1991; pp. 41–60. [Google Scholar]
- Lovelock, J.E. The Ages of Gaia; A Biography of Our Living Earth; W. W. Norton&Company: New York, NY, USA, 1988. [Google Scholar]
- Kleidon, A.; Fraedrich, K.; Heimann, M.A. Green Planet Versus a Desert World: Estimating the Maximum Effect of Vegetation on the Land Surface Climate. Climatic Change 2000, 44, 471–493. [Google Scholar] [CrossRef]
- Gnanadesikan, A.; Emanuel, K.; Vecchi, G.A.; Anderson, W.G.; Hallberg, R. How ocean color can steer Pacific tropical cyclones. Geophysical Research Letters 2010, 37, L18802. Available online: https://agupubs.onlinelibrary.wiley.com/doi/pdf/10.1029/2010GL044514. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



