Submitted:
15 January 2026
Posted:
15 January 2026
You are already at the latest version
Abstract
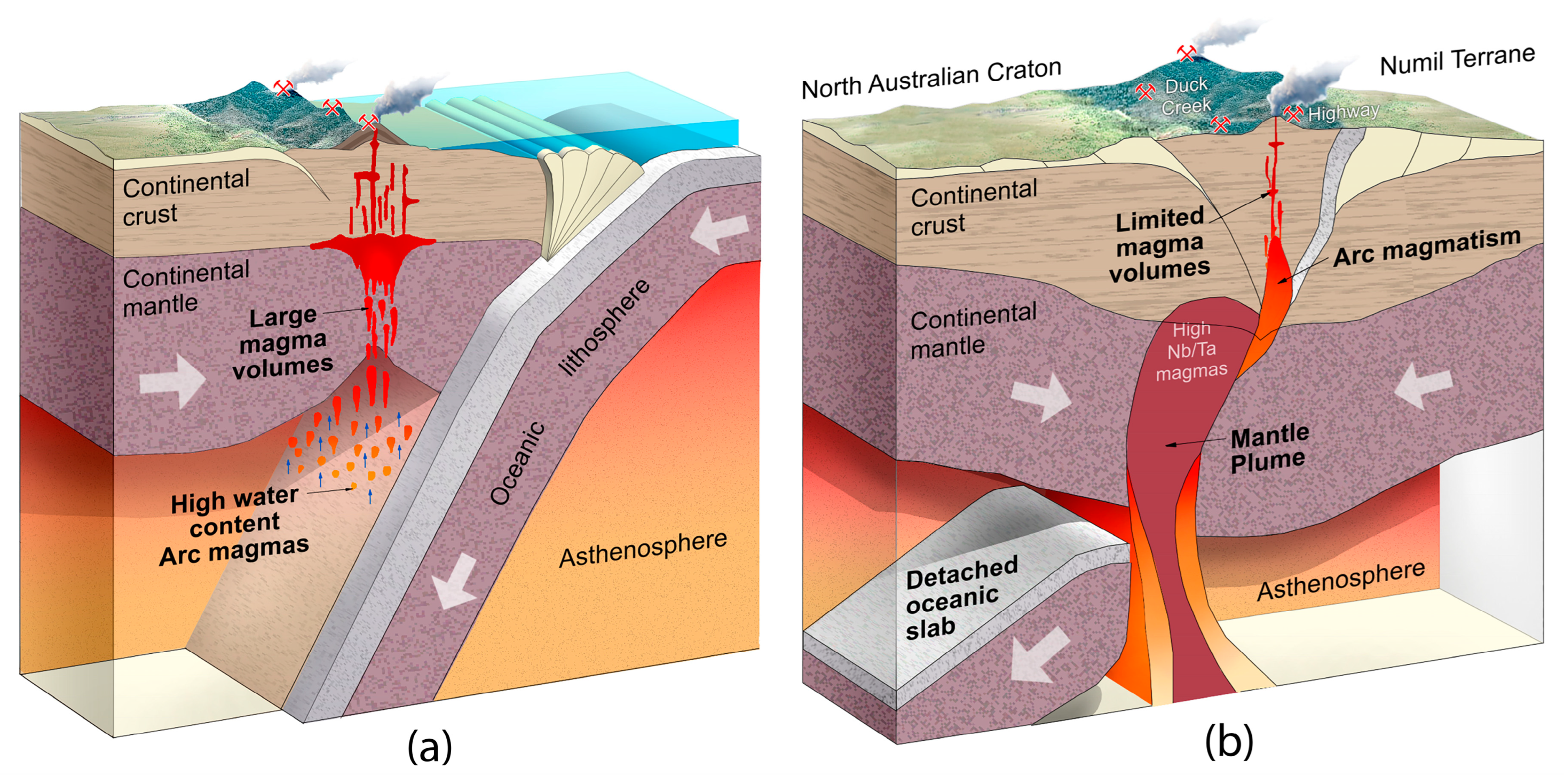
Post-collisional Cu-Au-Ni-Co-Pt-Pd-Sc porphyry, [Duck Creek porphyry system (DCPS)], with overlying Au-Te-Bi-W-HRE epithermal mineralisation, [Highway epithermal system (HES)] has been discovered in the core of the Mitakoodi anticline, southwest of Cloncurry. Xenotime and monazite geochronology indicates mineralisation occurred between ~1490 and 1530 Ma. Host rock lithologies show widespread potassic and/or propylitic to phyllic alteration. Paragenesis of porphyry sulphides indicate early crystallisation of pyrite, followed by chalcopyrite, with bornite forming by hydrothermal alteration chalcopyrite. Cu sulphides also show the effect of supergene oxidation alteration with rims of covellite, digenite and chalcocite. Redox conditions deduced from V/Sc systematics indicate that the DCPS contains both highly oxidized (typical of porphyries) and reduced lithologies, typical of plume generated tholeiitic and alkaline suites. Ni/Te and Cu/Te systematics plot within the fields defined by epithermal and porphyry deposits. Duck Creek chalcophile and highly siderophile element (Cu, MgO and Pd) systematics resemble data from porphyry mineral systems, at Cadia, Bingham Canyon, Grasberg, Skouries, Kalmakyr, Elaisite, Assarel and Medet. SAM geophysical inversion models suggest the presence of an extensive porphyry system below the HES. A progressive increase in molar Cu/Au ratios with depth from the HES to the DCPS, supports this conclusion. Three metal sources contributed to the DCPS-HES viz., tholeiitic ferrogabbro, potassic ultramafic to mafic system and a Fe and Ca-rich alkaline system. The latter two imparted non-crustal superchondritic Nb/Ta ratios that are characteristic of many deposits in the eastern Mount Isa Block. The associated tholeiite and alkaline magmatism reflect mantle plume upwelling through a palaeo-slab window that had accreted below the eastern flank of the North Australian craton following west verging collision by the Numil Terrane. Discovery of this linked mineral system provides a new paradigm for mineral exploration in the region.
Keywords:
1. Introduction
2. Geological Background
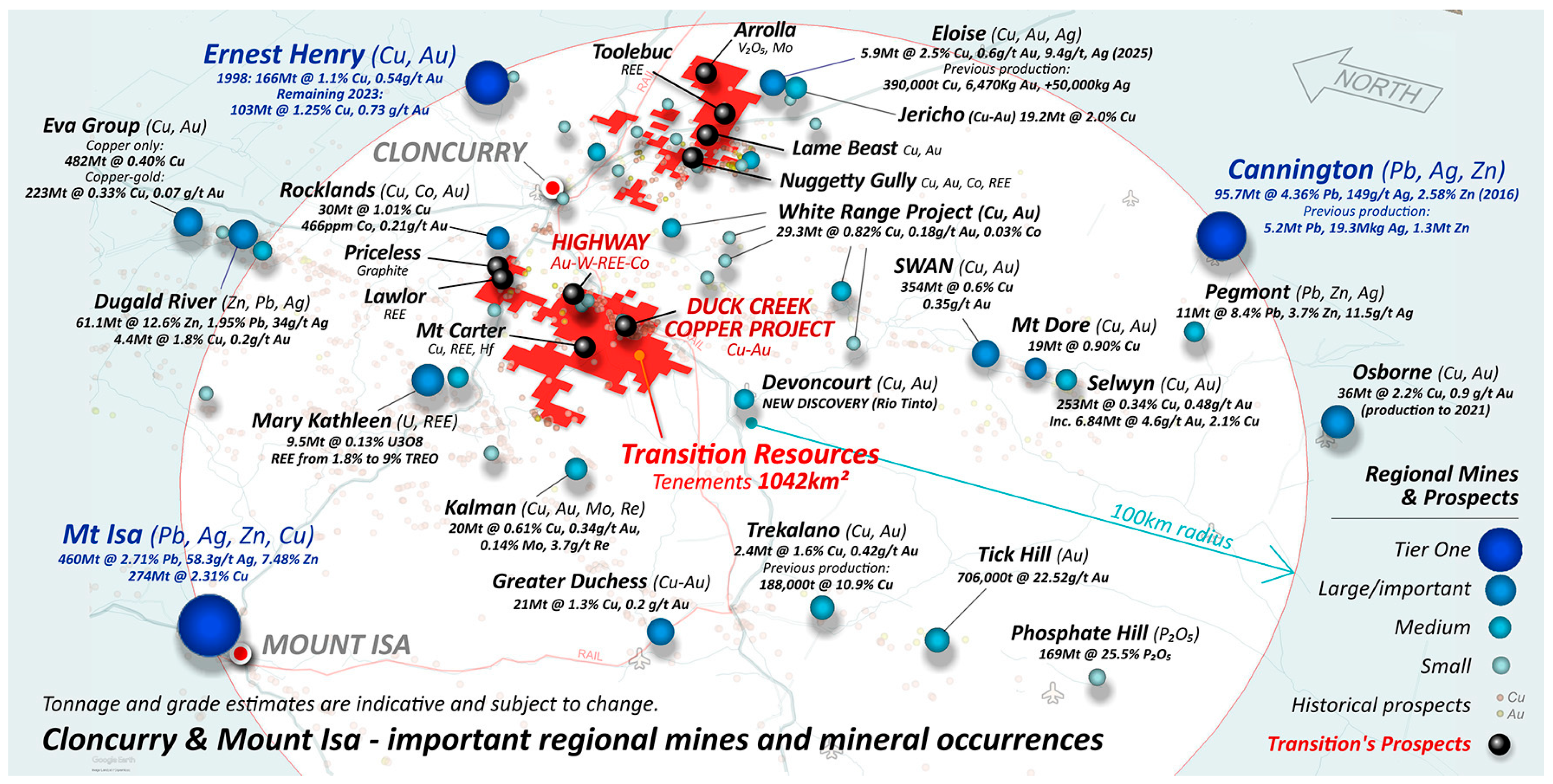
2.1. The Highway - Duck Creek Mineral System
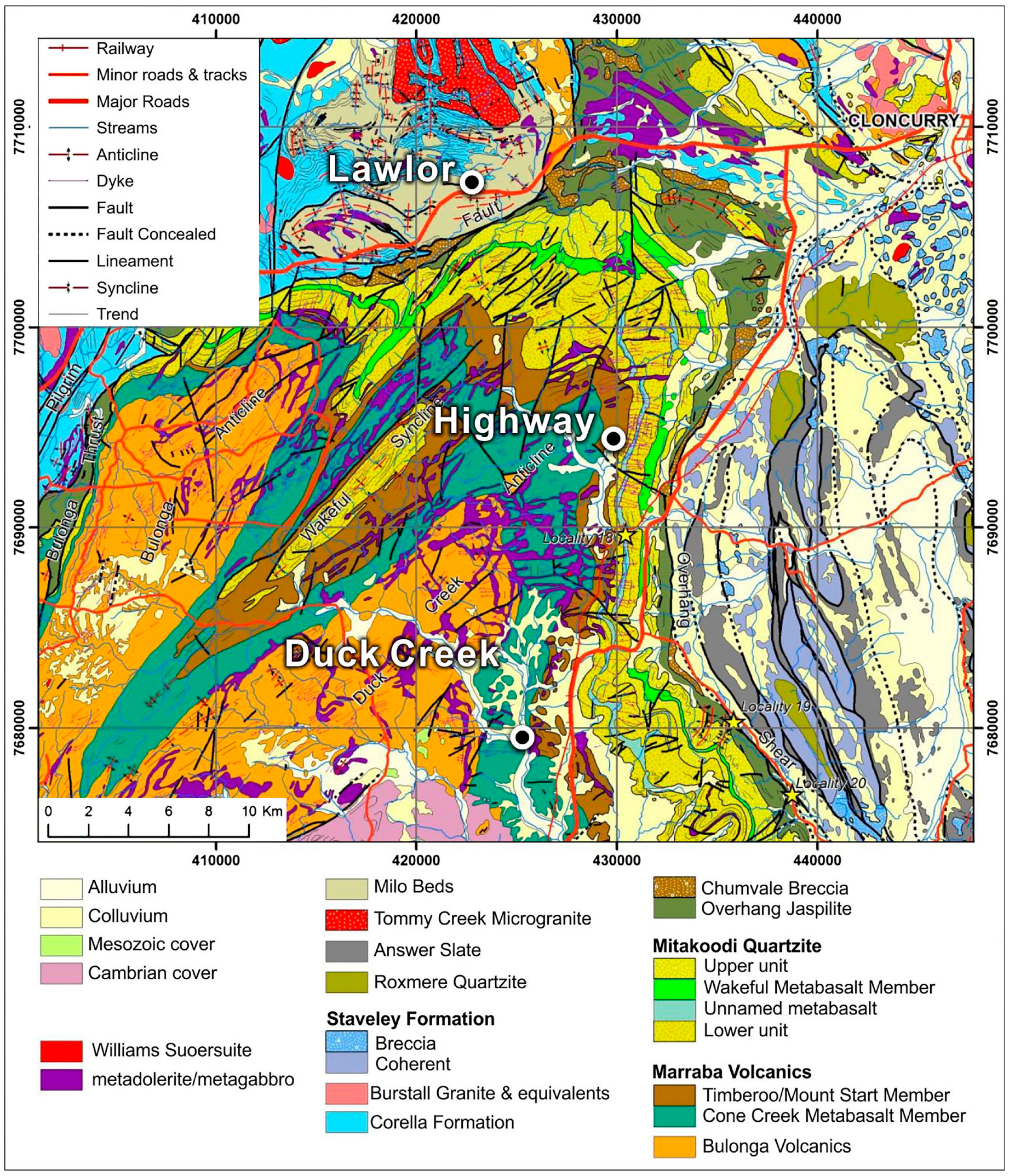
2.2. Geological Setting of the Mitakoodi Domain
2.3. Field Relationships and Petrology



3.4. Duck Creek Mafic Intrusive Complex

2.5. Alteration in the Highway - Duck Creek Mineral System

3. Highway - Duck Creek Mineralisation
4. Materials and Methods
4.1. Materials
4.2. Methods
| #1 | #2 | #3 | #4 | #5 | #6 | #7 | #8 | #9 | #10 | #11 | #12 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Number of Analyses | 154 | 783 | 426 | 888 | 12 | 64 | 1045 | 722 | 47 | 435 | 15 | 163 |
| SiO2 | 51.71 | 52.86 | 53.49 | 50.51 | 59.84 | 58.37 | 60.69 | 61.91 | 57.35 | 58.02 | 73.87 | 70.66 |
| TiO2 | 0.40 | 0.81 | 0.64 | 1.55 | 0.47 | 0.67 | 0.59 | 0.49 | 0.44 | 0.23 | 0.30 | 0.26 |
| Al2O3 | 12.74 | 11.89 | 12.39 | 11.63 | 13.49 | 12.19 | 13.72 | 13.20 | 9.46 | 5.28 | 4.75 | 7.44 |
| Fe2O3 | 16.99 | 13.91 | 10.74 | 16.42 | 14.48 | 9.64 | 9.21 | 7.58 | 8.87 | 7.77 | 12.21 | 12.32 |
| MnO | 0.13 | 0.17 | 0.13 | 0.17 | 0.18 | 0.13 | 0.08 | 0.12 | 0.26 | 0.38 | 0.69 | 0.22 |
| MgO | 4.40 | 4.73 | 4.00 | 4.55 | 2.96 | 4.76 | 3.81 | 3.58 | 3.32 | 2.94 | 1.52 | 1.77 |
| CaO | 2.12 | 6.50 | 4.65 | 6.02 | 1.25 | 6.55 | 3.48 | 5.71 | 13.97 | 20.46 | 1.33 | 2.11 |
| Na2O | 1.01 | 1.62 | 1.40 | 2.01 | 0.84 | 1.28 | 2.24 | 2.10 | 0.94 | 0.40 | 0.65 | 0.33 |
| K2O | 2.23 | 2.27 | 2.37 | 1.38 | 3.18 | 3.25 | 3.01 | 2.15 | 2.24 | 1.37 | 1.56 | 1.73 |
| P2O5 | 0.27 | 0.24 | 0.19 | 0.26 | 0.31 | 0.16 | 0.17 | 0.16 | 0.15 | 0.15 | 0.12 | 0.16 |
| LOI | 8.00 | 5.00 | 10.00 | 5.50 | 3.00 | 3.00 | 3.00 | 3.00 | 3.00 | 3.00 | 3.00 | 3.00 |
| Total | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
| Li | 18.3 | 13.7 | 15.1 | 13.2 | 10.8 | 5.9 | 16.0 | 13.0 | 10.5 | 7.0 | 14.2 | 8.6 |
| Sc | 35 | 33 | 24 | 38 | 36 | 24 | 18 | 17 | 19 | 14 | 23 | 25 |
| V | 359 | 322 | 208 | 356 | 378 | 193 | 142 | 127 | 172 | 108 | 151 | 203 |
| Cr | 74 | 64 | 66 | 49 | 78 | 86 | 67 | 57 | 51 | 28 | 48 | 55 |
| Co | 126 | 73 | 57 | 78 | 75 | 40 | 34 | 28 | 60 | 237 | 229 | 170 |
| Ni | 55 | 47 | 42 | 41 | 50 | 48 | 42 | 37 | 37 | 38 | 44 | 43 |
| Cu | 53 | 94 | 73 | 413 | 30 | 39 | 35 | 29 | 34 | 86 | 38 | 22 |
| Zn | 26 | 21 | 28 | 37 | 22 | 26 | 25 | 12 | 31 | 12 | 10 | 21 |
| Ga | 22 | 21 | 19 | 22 | 26 | 19 | 20 | 20 | 16 | 11 | 9 | 14 |
| Ge | 1.33 | 1.27 | 1.44 | 0.73 | 1.14 | 3.88 | 1.74 | 1.28 | 1.63 | 0.85 | 1.30 | 1.17 |
| As | 4.8 | 5.6 | 3.8 | 7.0 | 2.9 | 4.0 | 2.7 | 2.1 | 2.9 | 6.5 | 7.3 | 4.7 |
| Se | 1.97 | 1.98 | 2.37 | 1.53 | 0.94 | 2.06 | 1.57 | 1.85 | 1.58 | 7.33 | 9.05 | 2.94 |
| Rb | 100 | 101 | 101 | 72 | 133 | 143 | 122 | 93 | 94 | 57 | 81 | 76 |
| Sr | 34 | 28 | 25 | 62 | 21 | 43 | 31 | 28 | 37 | 33 | 15 | 19 |
| Y | 47 | 38 | 35 | 50 | 46 | 32 | 31 | 29 | 35 | 72 | 244 | 112 |
| Zr | 168 | 170 | 201 | 161 | 185 | 174 | 214 | 224 | 160 | 109 | 65 | 112 |
| Nb | 13.3 | 15.0 | 12.9 | 11.7 | 13.5 | 11.6 | 13.9 | 13.9 | 12.6 | 12.1 | 5.8 | 12.7 |
| Mo | 2.25 | 4.19 | 3.01 | 2.73 | 3.83 | 3.26 | 2.69 | 3.13 | 3.09 | 13.07 | 5.31 | 4.63 |
| Ag | 0.22 | 0.17 | 0.29 | 0.32 | 0.07 | 0.45 | 0.35 | 0.15 | 0.37 | 0.45 | 0.34 | 0.28 |
| Sn | 3.91 | 4.13 | 3.92 | 2.87 | 6.57 | 3.13 | 4.00 | 4.34 | 3.92 | 4.35 | 2.13 | 4.59 |
| Sb | 0.96 | 0.53 | 1.66 | 0.73 | 0.76 | 5.32 | 1.50 | 0.70 | 3.40 | 0.83 | 0.53 | 0.58 |
| Te | 19.20 | 3.06 | 4.79 | 5.04 | 1.03 | 8.41 | 5.94 | 2.03 | 4.81 | 13.28 | 25.15 | 28.14 |
| Ba | 300 | 361 | 362 | 201 | 503 | 337 | 547 | 301 | 348 | 208 | 160 | 294 |
| La | 38.9 | 36.5 | 43.1 | 28.5 | 40.5 | 32.4 | 47.5 | 51.7 | 47.7 | 178.1 | 43.8 | 57.7 |
| Ce | 78.2 | 76.7 | 94.6 | 65.3 | 81.8 | 69.3 | 97.1 | 102.3 | 94.3 | 309.7 | 92.6 | 112.3 |
| Pr | 9.6 | 9.2 | 11.1 | 8.1 | 10.0 | 8.3 | 11.1 | 11.8 | 11.0 | 38.6 | 11.6 | 13.3 |
| Nd | 38.6 | 36.6 | 41.7 | 33.1 | 40.0 | 31.8 | 41.3 | 44.1 | 41.6 | 146.0 | 50.9 | 54.1 |
| Sm | 8.76 | 8.16 | 8.96 | 8.36 | 8.86 | 6.60 | 7.98 | 8.59 | 9.39 | 32.26 | 18.39 | 13.72 |
| Eu | 2.63 | 2.28 | 2.34 | 2.47 | 2.55 | 1.56 | 1.71 | 1.95 | 2.48 | 9.10 | 8.38 | 4.77 |
| Gd | 9.96 | 8.61 | 8.47 | 10.10 | 9.69 | 6.33 | 7.10 | 7.71 | 8.54 | 27.95 | 37.66 | 19.57 |
| Tb | 1.62 | 1.38 | 1.33 | 1.77 | 1.55 | 0.99 | 1.05 | 1.14 | 1.28 | 3.74 | 8.32 | 3.72 |
| Dy | 10.09 | 8.45 | 7.99 | 11.80 | 9.54 | 5.94 | 6.20 | 6.58 | 7.38 | 19.69 | 57.51 | 24.91 |
| Ho | 1.98 | 1.68 | 1.52 | 2.39 | 1.86 | 1.19 | 1.21 | 1.26 | 1.40 | 3.45 | 11.73 | 4.97 |
| Er | 5.50 | 4.64 | 4.24 | 6.50 | 5.35 | 3.40 | 3.44 | 3.48 | 3.83 | 8.92 | 30.50 | 13.29 |
| Tm | 0.78 | 0.65 | 0.59 | 0.90 | 0.79 | 0.49 | 0.49 | 0.50 | 0.53 | 1.22 | 4.13 | 1.87 |
| Yb | 4.87 | 3.98 | 3.76 | 5.50 | 4.97 | 3.11 | 3.14 | 3.12 | 3.26 | 7.42 | 23.81 | 11.04 |
| Lu | 0.69 | 0.59 | 0.54 | 0.77 | 0.74 | 0.46 | 0.48 | 0.48 | 0.48 | 0.93 | 2.83 | 1.40 |
| Hf | 4.57 | 4.48 | 5.43 | 4.46 | 5.14 | 4.72 | 5.79 | 5.94 | 4.29 | 2.83 | 1.81 | 3.02 |
| Ta | 0.85 | 0.86 | 0.97 | 0.70 | 0.97 | 0.95 | 1.10 | 1.08 | 0.90 | 0.82 | 0.33 | 0.70 |
| W | 135 | 169 | 211 | 25 | 172 | 25 | 92 | 147 | 70 | 227 | 715 | 615 |
| Au g/t | 1.03 | 0.37 | 0.19 | 0.23 | 0.12 | 0.03 | 0.16 | 0.25 | 0.22 | 3.46 | 6.14 | 3.28 |
| Tl | 0.69 | 0.24 | 2.45 | 0.24 | 0.31 | 9.78 | 2.59 | 0.77 | 5.10 | 1.31 | 0.18 | 0.20 |
| Pb | 2.27 | 2.28 | 2.40 | 3.03 | 1.23 | 2.11 | 2.12 | 1.87 | 2.23 | 5.34 | 4.52 | 3.21 |
| Bi | 11.71 | 1.60 | 1.11 | 1.23 | 0.32 | 2.66 | 1.34 | 0.72 | 2.01 | 11.55 | 21.88 | 24.08 |
| Th | 6.79 | 6.33 | 12.35 | 5.37 | 7.78 | 17.26 | 15.99 | 14.99 | 11.68 | 5.99 | 4.72 | 5.24 |
| U | 4.46 | 3.94 | 4.74 | 2.12 | 5.90 | 8.31 | 6.07 | 5.81 | 6.42 | 10.37 | 7.06 | 5.17 |
| S wt% | 0.15 | 0.37 | 0.22 | 0.29 | N.D. | 0.06 | 0.13 | 0.24 | 0.22 | 1.35 | 1.01 | 0.47 |
| Y/Ho | 23.88 | 22.91 | 22.82 | 20.78 | 24.96 | 26.69 | 25.30 | 22.75 | 24.98 | 20.89 | 20.79 | 22.50 |
| Zr/Hf | 36.84 | 38.08 | 36.98 | 36.08 | 35.92 | 36.93 | 36.90 | 37.79 | 37.20 | 38.59 | 35.83 | 37.06 |
| Nb/Ta | 15.69 | 17.38 | 13.30 | 16.79 | 13.93 | 12.22 | 12.57 | 12.93 | 13.99 | 14.63 | 17.44 | 18.18 |
| Nb/Y | 0.28 | 0.39 | 0.37 | 0.24 | 0.29 | 0.37 | 0.45 | 0.49 | 0.36 | 0.17 | 0.02 | 0.11 |
| La/Yb | 7.99 | 9.17 | 11.45 | 5.18 | 8.15 | 10.41 | 15.11 | 16.56 | 14.63 | 24.00 | 1.84 | 5.22 |
| Molar Cu/Au | 159 | 786 | 1198 | 5571 | 784 | 4051 | 680 | 357 | 475 | 77 | 19 | 21 |
| Columbiad | Comet | Dulce | Easter Gift. | Eva | Forget-Me-Not | Hideaway | Horseshoe | |
|---|---|---|---|---|---|---|---|---|
| No. Analyses | n=130 | n=44 | n=61 | n=4 | n=13 | n=157 | n=66 | n=481 |
| SIO2 | 62.96 | 64.16 | 59.16 | 69.09 | 63.87 | 62.90 | 63.20 | 62.27 |
| TiO2 | 0.90 | 1.48 | 0.74 | 0.68 | 0.43 | 0.64 | 1.24 | 0.93 |
| Al2O3 | 10.33 | 10.30 | 12.55 | 10.94 | 6.96 | 10.84 | 9.42 | 10.56 |
| Fe2O3 | 16.39 | 15.58 | 15.60 | 11.51 | 20.51 | 15.05 | 18.65 | 15.34 |
| MnO | 0.09 | 0.32 | 0.13 | 0.09 | 0.11 | 0.08 | 0.12 | 0.12 |
| MgO | 4.76 | 2.30 | 5.83 | 3.78 | 3.43 | 6.11 | 2.32 | 5.45 |
| CaO | 1.46 | 3.28 | 2.86 | 0.43 | 3.31 | 1.49 | 2.75 | 2.60 |
| Na2O | 2.14 | 1.28 | 2.03 | 2.52 | 0.74 | 1.74 | 1.07 | 1.83 |
| K2O | 0.79 | 1.16 | 1.00 | 0.84 | 0.57 | 1.05 | 1.13 | 0.78 |
| P2O5 | 0.18 | 0.14 | 0.10 | 0.12 | 0.08 | 0.11 | 0.11 | 0.13 |
| SUM | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| Fe/(Fe+Mg) | 0.63 | 0.77 | 0.57 | 0.61 | 0.75 | 0.55 | 0.80 | 0.59 |
| Na2O+K2O | 2.93 | 2.44 | 3.03 | 3.36 | 1.30 | 2.79 | 2.20 | 2.61 |
| Trace Elements ppm | ||||||||
| Li | 10.8 | 10.6 | 17.6 | 13.1 | 10.1 | 21.0 | 10.1 | 16.3 |
| Be | 1.65 | 1.26 | 0.94 | 0.85 | 0.66 | 1.10 | 1.00 | 1.05 |
| Sc | 21 | 30 | 25 | 19 | 18 | 24 | 24 | 26 |
| V | 224 | 329 | 216 | 213 | 274 | 215 | 352 | 244 |
| Cr | 52 | 54 | 92 | 43 | 70 | 81 | 68 | 55 |
| Co | 208 | 97 | 159 | 100 | 204 | 114 | 59 | 164 |
| Ni | 64 | 46 | 93 | 45 | 108 | 85 | 102 | 67 |
| Cu | 704 | 2544 | 5789 | 11596 | 19414 | 4645 | 1727 | 6382 |
| Cu wt.% | 0.07 | 0.25 | 0.58 | 1.16 | 1.94 | 0.46 | 0.17 | 0.64 |
| Zn | 28 | 27 | 37 | 22 | 29 | 36 | 41 | 34 |
| Ga | 20 | 15 | 22 | 14 | 14 | 21 | 15 | 19 |
| Rb | 37 | 50 | 47 | 31 | 26 | 74 | 54 | 30 |
| Sr | 30 | 58 | 50 | 16 | 24 | 36 | 60 | 40 |
| Y | 23 | 29 | 19 | 36 | 14 | 15 | 23 | 20 |
| Zr | 103 | 120 | 103 | 126 | 35 | 68 | 102 | 85 |
| Nb | 7.0 | 8.2 | 6.5 | 4.4 | 1.8 | 4.4 | 8.1 | 5.5 |
| Mo | 1.99 | 0.64 | 2.96 | 0.45 | 1.38 | 1.56 | 1.11 | 2.89 |
| Pd | 2.92 | 3.24 | 7.05 | 2.83 | 20.93 | 6.42 | 7.42 | 8.89 |
| Ag | 0.15 | 0.34 | 0.35 | 0.33 | 0.98 | 0.44 | 0.90 | 0.40 |
| Sn | 1.81 | 1.84 | 1.43 | 1.80 | 2.46 | 1.71 | 1.71 | 1.74 |
| Sb | 26.3 | 0.63 | 0.37 | 0.40 | 0.50 | 0.43 | 2.39 | 0.38 |
| Cs | 1.45 | 1.52 | 1.46 | 0.79 | 1.05 | 2.76 | 1.28 | 1.23 |
| Ba | 176 | 283 | 239 | 118 | 179 | 217 | 191 | 177 |
| La | 19.5 | 20.2 | 19.9 | 13.2 | 6.2 | 10.7 | 16.6 | 14.2 |
| Ce | 39.2 | 46.2 | 41.7 | 27.3 | 13.0 | 22.1 | 36.0 | 30.1 |
| Pr | 4.6 | 5.2 | 5.0 | 3.6 | 1.8 | 2.7 | 4.3 | 3.7 |
| Nd | 18.3 | 21.5 | 19.6 | 15.6 | 7.7 | 11.1 | 17.4 | 15.3 |
| Sm | 4.02 | 5.00 | 4.05 | 4.41 | 1.95 | 2.56 | 4.08 | 3.50 |
| Eu | 1.03 | 1.46 | 0.90 | 1.15 | 0.48 | 0.62 | 1.08 | 0.83 |
| Gd | 4.26 | 5.55 | 3.82 | 5.50 | 2.20 | 2.71 | 4.31 | 3.65 |
| Tb | 0.70 | 0.85 | 0.57 | 0.91 | 0.36 | 0.44 | 0.68 | 0.59 |
| Dy | 4.30 | 5.43 | 3.62 | 6.11 | 2.40 | 2.90 | 4.33 | 3.82 |
| Ho | 0.87 | 1.11 | 0.72 | 1.27 | 0.51 | 0.60 | 0.88 | 0.78 |
| Er | 2.55 | 3.57 | 2.13 | 4.15 | 1.64 | 1.78 | 2.66 | 2.29 |
| Tm | 0.36 | 0.52 | 0.31 | 0.63 | 0.24 | 0.27 | 0.38 | 0.34 |
| Yb | 2.30 | 3.47 | 2.04 | 4.25 | 1.59 | 1.65 | 2.45 | 2.09 |
| Lu | 0.34 | 0.56 | 0.31 | 0.66 | 0.25 | 0.25 | 0.37 | 0.33 |
| Hf | 3.03 | 3.18 | 2.83 | 3.42 | 0.95 | 1.98 | 2.77 | 2.55 |
| Ta | 0.53 | 0.60 | 0.48 | 0.34 | 0.12 | 0.32 | 0.59 | 0.42 |
| W | 3.31 | 12.78 | 1.91 | 4.38 | 5.30 | 3.52 | 1.69 | 3.83 |
| Pt | 3.66 | 3.44 | 6.14 | 1.40 | 12.68 | 6.46 | 3.94 | 7.41 |
| Au ppm | 0.02 | 0.12 | 0.04 | 0.04 | 2.36 | 0.07 | 0.07 | 0.06 |
| Tl | 0.16 | 0.20 | 0.20 | 0.10 | 0.12 | 0.29 | 0.24 | 0.13 |
| Pb | 4.12 | 4.24 | 2.16 | 19.33 | 8.24 | 1.85 | 40.20 | 2.02 |
| Bi | 0.85 | 1.00 | 1.19 | 0.41 | 3.85 | 1.33 | 4.19 | 0.68 |
| Th | 5.49 | 6.14 | 6.66 | 5.89 | 1.10 | 3.39 | 5.53 | 3.62 |
| U | 2.27 | 1.97 | 2.13 | 4.56 | 3.44 | 2.37 | 1.55 | 1.99 |
| TREE ppm | 102 | 121 | 105 | 89 | 40 | 60 | 96 | 81 |
| logAg/Au | 0.91 | 0.46 | 0.98 | 0.90 | -0.38 | 0.78 | 1.12 | 0.82 |
| Molar Cu/Au | 122967 | 66077 | 495563 | 866068 | 25471 | 195163 | 78261 | 326323 |
| Y/Ho | 26.29 | 26.48 | 26.65 | 28.42 | 27.87 | 25.52 | 26.09 | 25.35 |
| Zr/Hf | 33.90 | 37.63 | 36.46 | 36.87 | 36.44 | 34.48 | 36.73 | 33.53 |
| Nb/Ta | 13.41 | 13.84 | 13.50 | 13.02 | 14.52 | 13.84 | 13.62 | 13.12 |
| Nb/Y | 0.31 | 0.28 | 0.34 | 0.12 | 0.13 | 0.29 | 0.35 | 0.28 |
| Nb+Y | 29.95 | 37.70 | 25.59 | 40.46 | 16.09 | 19.84 | 31.12 | 25.26 |
| Junction | Micawber | Mountain Maid | New Dollar | Ready Rhino | Success | Mount Cobalt |
Mary Kathleen* |
|||
|---|---|---|---|---|---|---|---|---|---|---|
| Elements wt% | n=52 | n=67 | n=216 | n=168 | n=145 | n=196 | n=3 | n=640 | ||
| SIO2 | 55.62 | 60.80 | 64.04 | 61.79 | 61.31 | 65.01 | 46.33 | 46.74 | ||
| TiO2 | 0.94 | 0.64 | 1.06 | 0.86 | 0.86 | 1.18 | 1.51 | 4.78 | ||
| Al2O3 | 14.01 | 12.44 | 9.94 | 10.80 | 11.61 | 10.08 | 13.45 | 0.18 | ||
| Fe2O3 | 15.37 | 13.91 | 15.53 | 15.70 | 14.38 | 16.98 | 17.74 | 19.02 | ||
| MnO | 0.13 | 0.11 | 0.10 | 0.11 | 0.11 | 0.16 | 0.16 | 0.32 | ||
| MgO | 6.50 | 5.66 | 4.28 | 6.08 | 6.69 | 3.21 | 9.20 | 3.99 | ||
| CaO | 3.07 | 2.65 | 1.85 | 1.69 | 2.04 | 1.34 | 5.05 | 23.65 | ||
| Na2O | 3.00 | 2.30 | 1.71 | 1.43 | 1.53 | 0.98 | 1.88 | 0.65 | ||
| K2O | 1.20 | 1.38 | 1.29 | 1.43 | 1.33 | 0.93 | 4.55 | 0.26 | ||
| P2O5 | 0.17 | 0.11 | 0.20 | 0.12 | 0.15 | 0.11 | 0.12 | 0.42 | ||
| SUM | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | ||
| Fe/(Fe+Mg) | 0.54 | 0.55 | 0.65 | 0.57 | 0.52 | 0.73 | 0.49 | 0.85 | ||
| Na2O+K2O | 4.21 | 3.68 | 3.00 | 2.86 | 2.86 | 1.92 | 4.68 | 0.90 | ||
| Li | 19.7 | 17.2 | 14.3 | 25.9 | 29.1 | 15.7 | 26.7 | 6.89 | ||
| Be | 1.48 | 1.10 | 1.63 | 1.18 | 1.25 | 1.31 | 1.71 | |||
| Sc | 32 | 26 | 23 | 29 | 31 | 27 | 52 | 3.34 | ||
| V | 250 | 201 | 246 | 263 | 265 | 329 | 573 | 79.71 | ||
| Cr | 75 | 63 | 54 | 74 | 66 | 66 | 113 | 9.18 | ||
| Co | 60 | 124 | 156 | 80 | 76 | 208 | 148 | 104 | ||
| Ni | 67 | 101 | 62 | 70 | 80 | 67 | 147 | 203 | ||
| Cu | 187 | 2197 | 2932 | 4796 | 2774 | 4820 | 184 | 2039 | ||
| Cu wt.% | 0.02 | 0.22 | 0.29 | 0.48 | 0.28 | 0.48 | 0.02 | 0.20 | ||
| Zn | 42 | 38 | 33 | 32 | 38 | 25 | 34 | 23.44 | ||
| Ga | 22 | 21 | 19 | 19 | 19 | 17 | 36 | 15.45 | ||
| Rb | 55 | 79 | 73 | 65 | 61 | 43 | 423 | 11.16 | ||
| Sr | 69 | 51 | 39 | 29 | 48 | 33 | 59 | 18.90 | ||
| Y | 25 | 18 | 25 | 18 | 22 | 23 | 32 | 38.78 | ||
| Zr | 125 | 97 | 117 | 75 | 85 | 118 | 84 | 46.29 | ||
| Nb | 8.2 | 6.0 | 8.1 | 4.4 | 5.2 | 7.2 | 7.7 | 9.70 | ||
| Mo | 1.51 | 1.45 | 2.07 | 1.29 | 1.33 | 0.92 | 1.50 | 1.83 | ||
| Pd | 9.13 | 7.24 | 2.79 | 7.07 | 8.22 | 4.44 | 1.80 | |||
| Ag | 0.18 | 0.16 | 0.32 | 0.29 | 0.28 | 0.42 | 1.67 | 0.11 | ||
| Sn | 2.12 | 1.40 | 1.82 | 1.33 | 1.43 | 2.39 | 0.08 | 45.72 | ||
| Sb | 0.57 | 0.33 | 0.33 | 0.40 | 0.48 | 0.53 | 0.43 | 0.33 | ||
| Cs | 2.89 | 4.06 | 2.66 | 3.39 | 2.68 | 1.18 | 13.78 | 0.36 | ||
| Ba | 293 | 292 | 217 | 189 | 234 | 200 | 461 | 52.6 | ||
| La | 34.1 | 17.5 | 20.4 | 13.6 | 14.2 | 22.9 | 152.6 | 159.5 | ||
| Ce | 66.6 | 36.4 | 43.1 | 27.4 | 29.4 | 46.0 | 270.4 | 303.3 | ||
| Pr | 7.7 | 4.4 | 5.2 | 3.3 | 3.7 | 5.5 | 30.9 | 29.5 | ||
| Nd | 29.7 | 17.4 | 20.8 | 13.7 | 15.4 | 21.9 | 119.4 | 101.24 | ||
| Sm | 5.85 | 3.71 | 4.58 | 3.18 | 3.67 | 4.83 | 25.89 | 16.39 | ||
| Eu | 1.63 | 0.82 | 1.16 | 0.82 | 0.94 | 1.35 | 2.65 | 3.49 | ||
| Gd | 5.38 | 3.68 | 4.80 | 3.44 | 3.97 | 4.78 | 19.38 | 11.69 | ||
| Tb | 0.79 | 0.55 | 0.79 | 0.55 | 0.63 | 0.73 | 2.22 | 1.48 | ||
| Dy | 4.83 | 3.47 | 4.79 | 3.50 | 4.07 | 4.51 | 9.58 | 7.28 | ||
| Ho | 0.95 | 0.68 | 0.97 | 0.71 | 0.82 | 0.89 | 1.44 | 1.28 | ||
| Er | 2.75 | 2.04 | 2.84 | 2.10 | 2.43 | 2.62 | 3.35 | 3.22 | ||
| Tm | 0.40 | 0.29 | 0.41 | 0.30 | 0.34 | 0.37 | 0.43 | 0.43 | ||
| Yb | 2.54 | 1.95 | 2.63 | 1.93 | 2.24 | 2.46 | 2.39 | 2.59 | ||
| Lu | 0.38 | 0.31 | 0.39 | 0.29 | 0.34 | 0.37 | 0.31 | 0.39 | ||
| Hf | 3.47 | 2.68 | 3.25 | 2.13 | 2.37 | 3.20 | 2.30 | 1.09 | ||
| Ta | 0.61 | 0.45 | 0.58 | 0.31 | 0.36 | 0.54 | 0.23 | 0.26 | ||
| W | 3.34 | 2.04 | 3.27 | 3.58 | 4.26 | 3.10 | 1.00 | 13.76 | ||
| Pt | 8.43 | 5.52 | 2.76 | 6.66 | 7.52 | 3.97 | 1.50 | |||
| Au ppm | 0.00 | 0.03 | 0.03 | 0.03 | 0.02 | 0.15 | 0.01 | 0.04 | ||
| Tl | 0.23 | 0.28 | 0.25 | 0.19 | 0.23 | 0.18 | 0.92 | 0.05 | ||
| Pb | 1.92 | 1.96 | 2.19 | 2.42 | 2.13 | 4.26 | 9.80 | |||
| Bi | 0.25 | 0.58 | 3.94 | 1.26 | 0.93 | 2.16 | 0.06 | 2.81 | ||
| Th | 8.03 | 6.52 | 5.79 | 2.37 | 3.75 | 6.02 | 1.22 | 55.81 | ||
| U | 2.43 | 2.23 | 2.57 | 2.06 | 1.93 | 2.17 | 0.86 | 17.95 | ||
| TREE ppm | 164 | 93 | 113 | 75 | 82 | 119 | 641 | 642 | ||
| logAg/Au | 1.62 | 0.74 | 1.06 | 0.94 | 1.06 | 0.45 | 2.44 | 0.41 | ||
| Molar Cu/Au | 133087 | 230183 | 333822 | 438564 | 345827 | 99015 | 94882 | 143049 | ||
| Y/Ho | 26.19 | 26.69 | 26.36 | 25.83 | 26.45 | 25.89 | 22.03 | 30.39 | ||
| Zr/Hf | 36.14 | 36.17 | 36.06 | 35.38 | 36.09 | 36.87 | 36.52 | 42.38 | ||
| Nb/Ta | 13.30 | 13.52 | 14.01 | 14.08 | 14.38 | 13.38 | 32.86 | 37.20 | ||
| Nb/Y | 0.33 | 0.33 | 0.32 | 0.24 | 0.24 | 0.31 | 0.24 | 0.25 | ||
| Nb+Y | 33.09 | 24.30 | 33.57 | 22.69 | 26.90 | 30.21 | 39.47 | 48.48 | ||
5. Results
5.1. Metallogenic Affinity
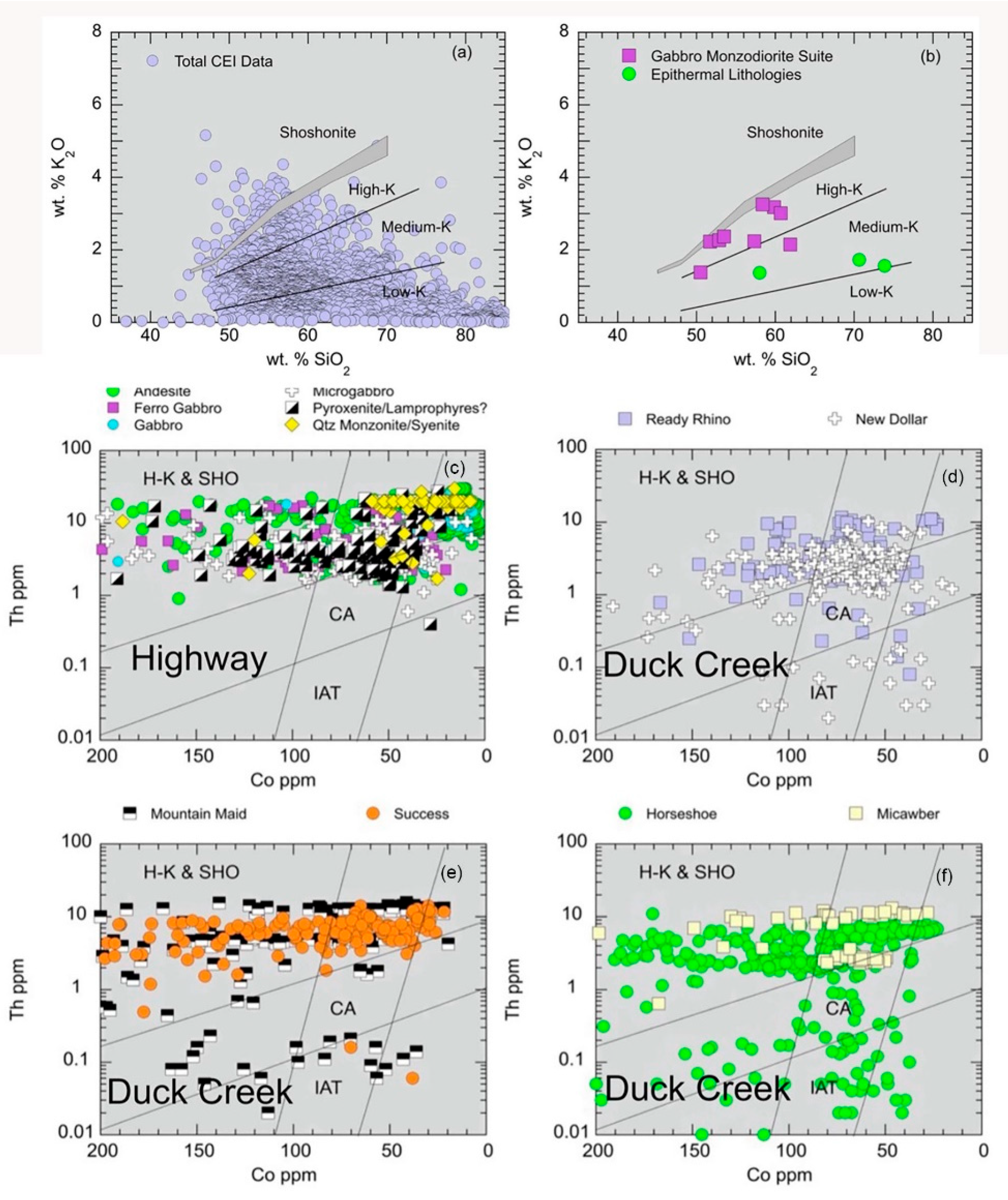
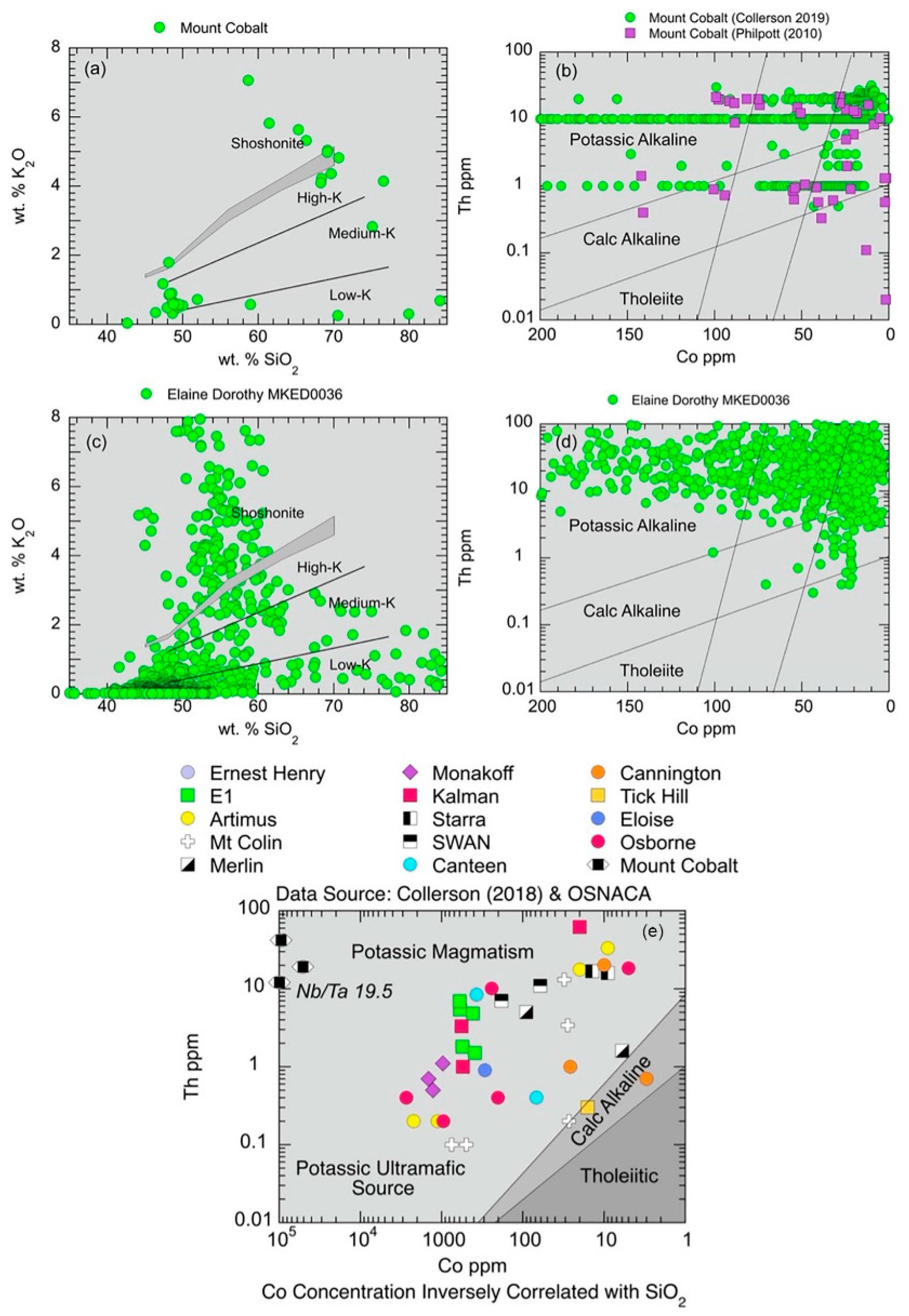
5.1.1. K2O-SiO2 and Th-Co Discrimination Plots
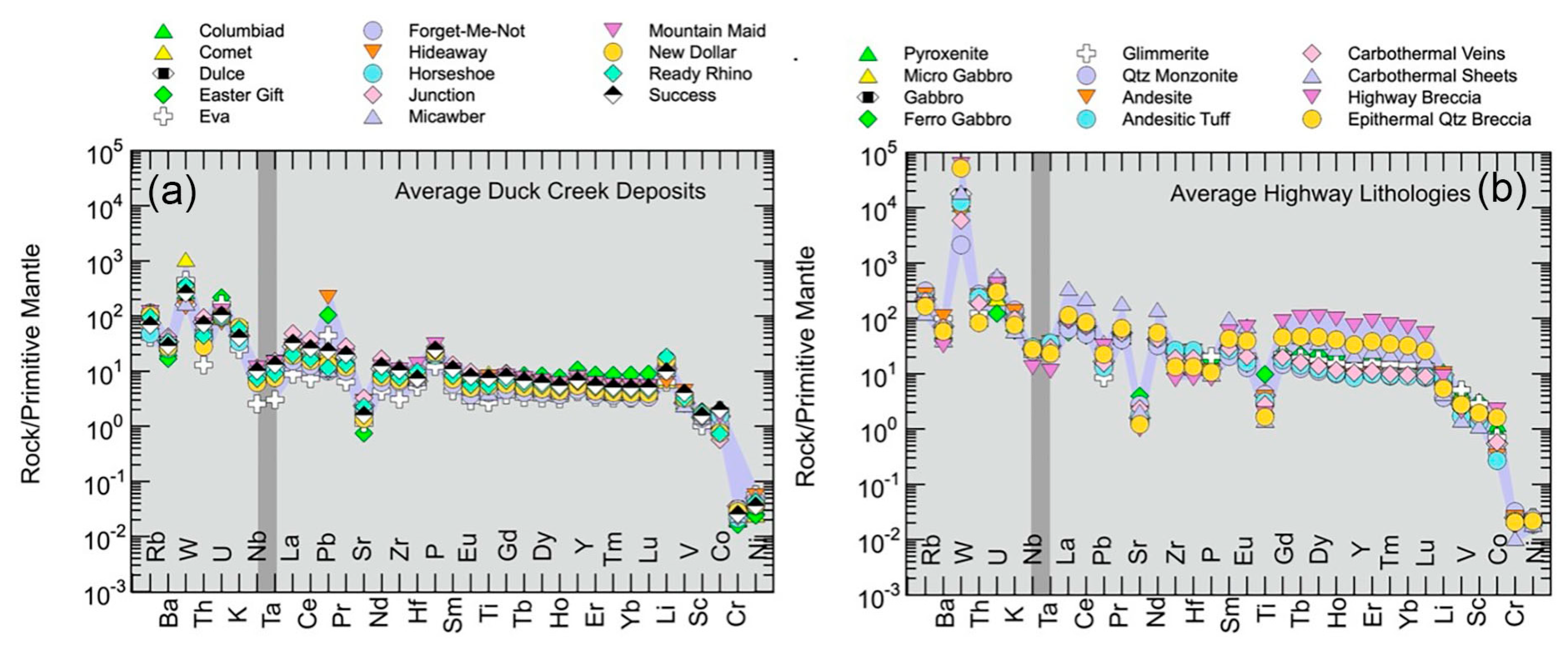
5.1.2. Multi-element Geochemical Variation
5.1.3. Multi-element Geochemical Variation
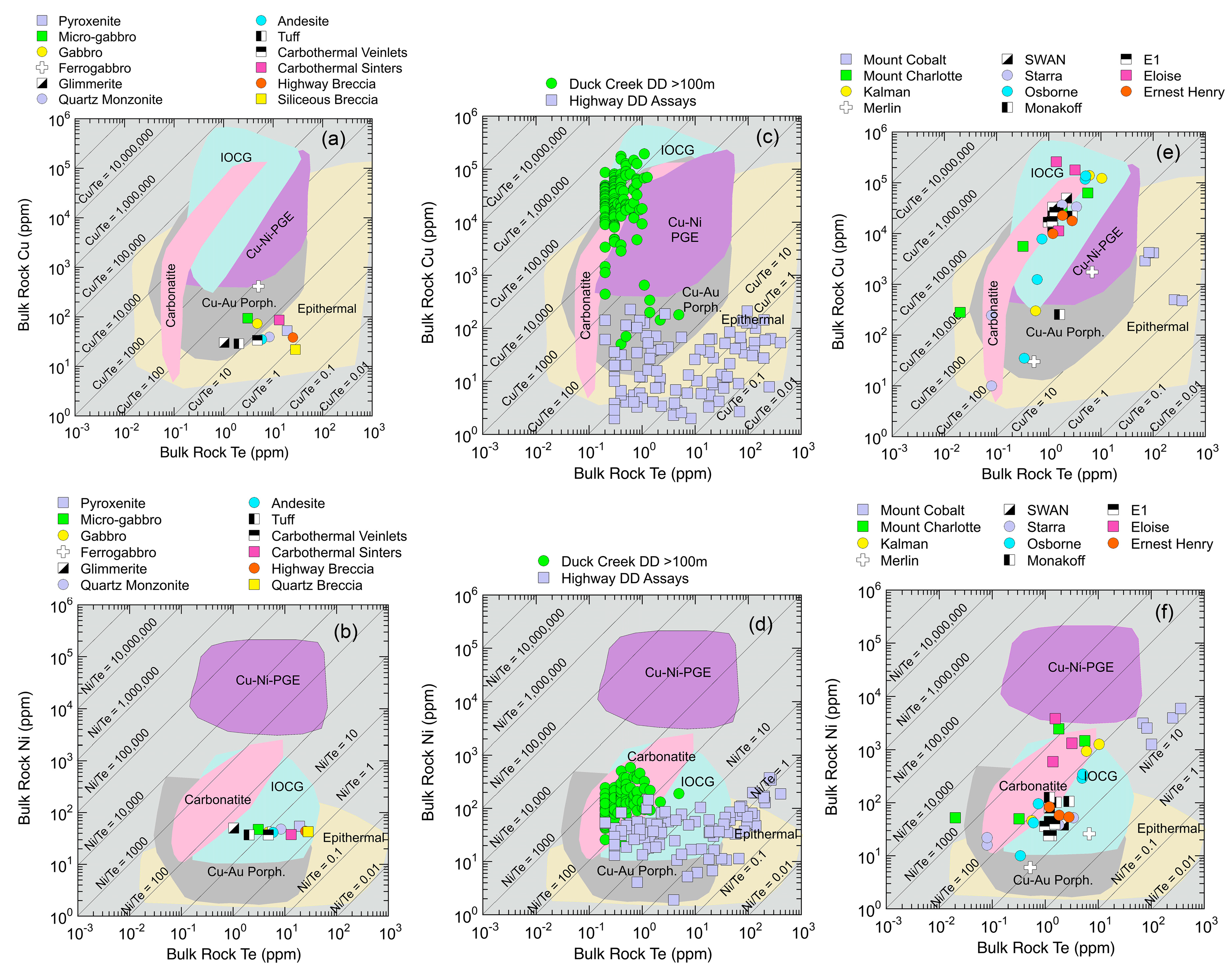
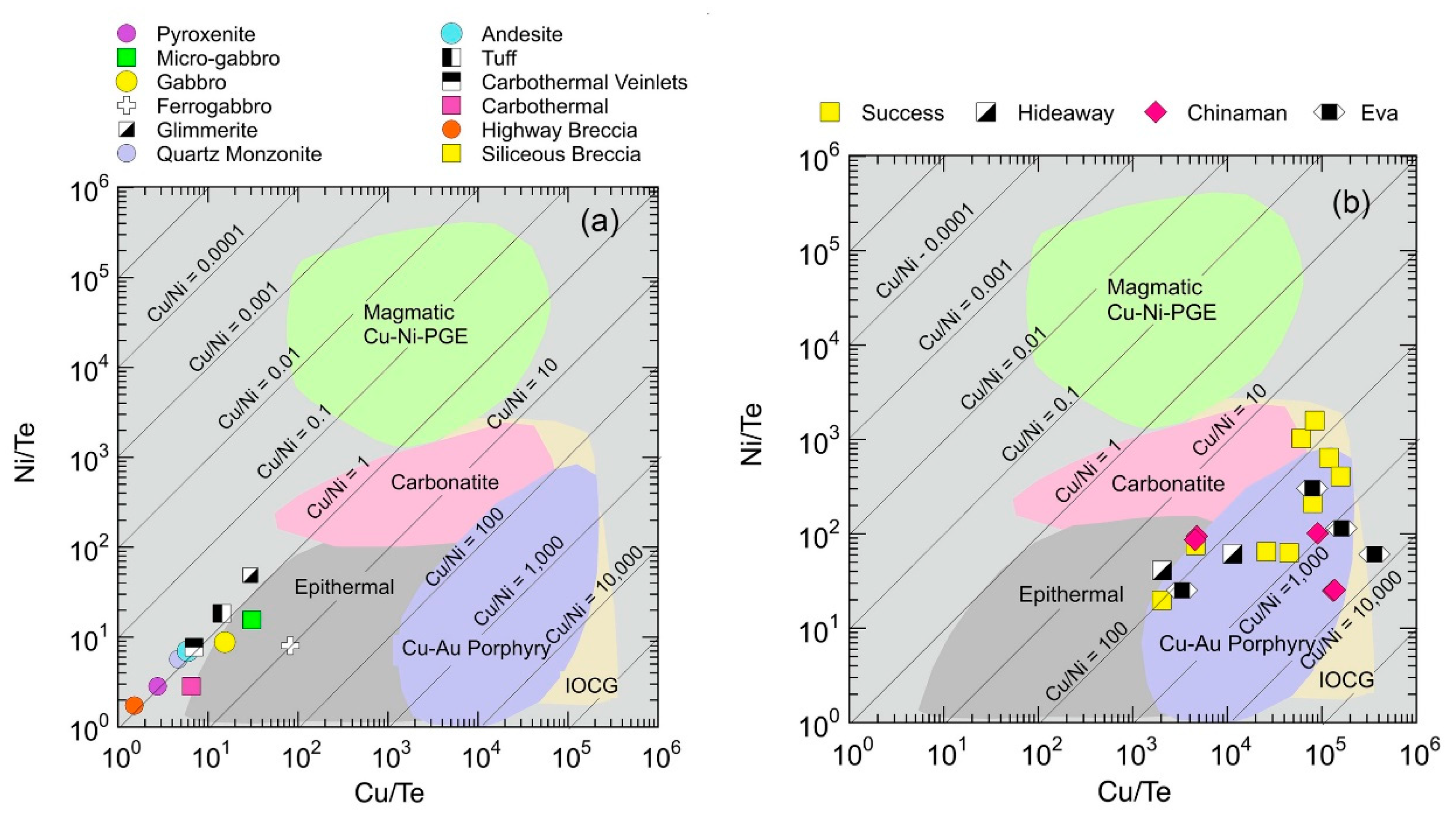
5.1.4. Metal Source Constraints from Te Cu Ni Systematics
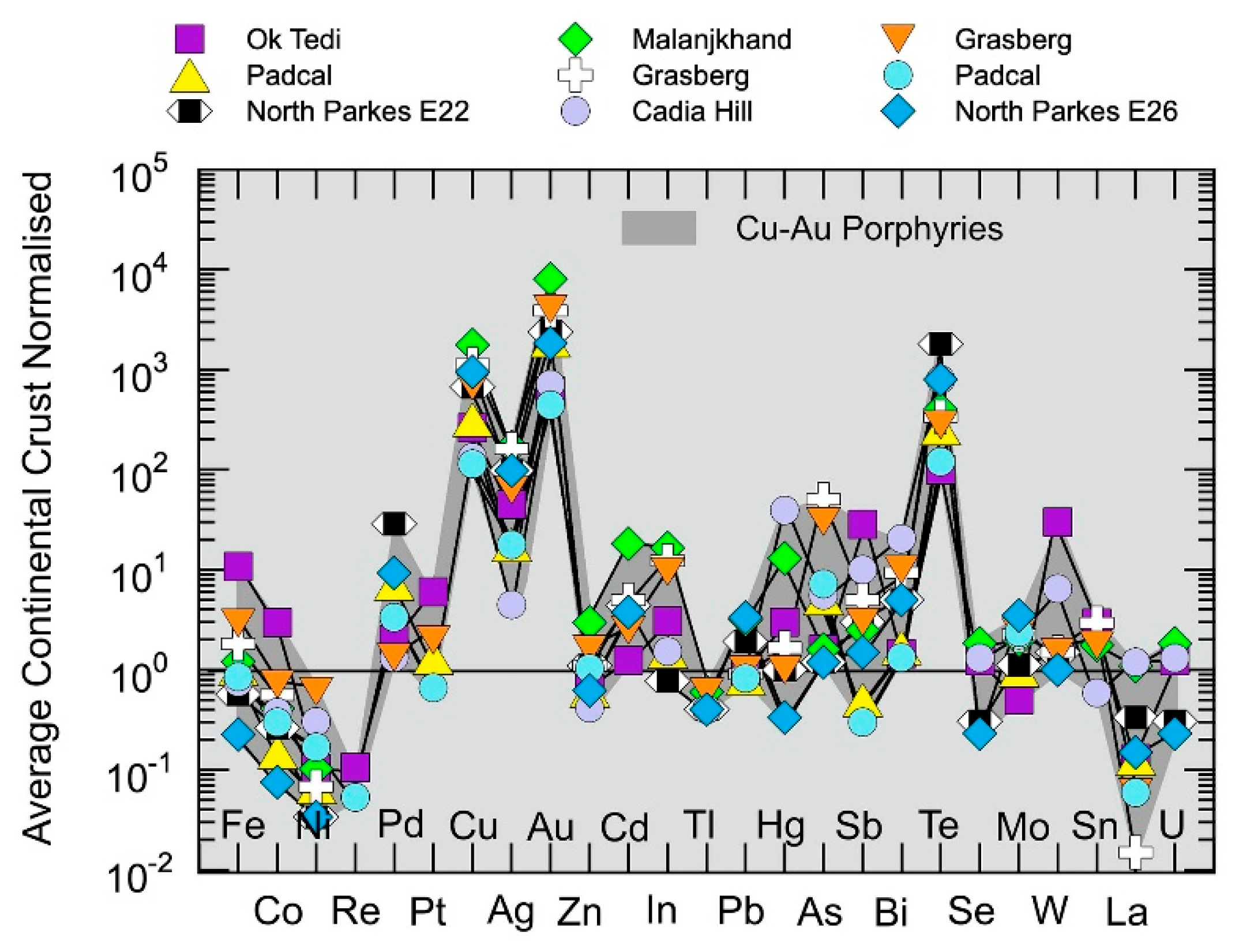
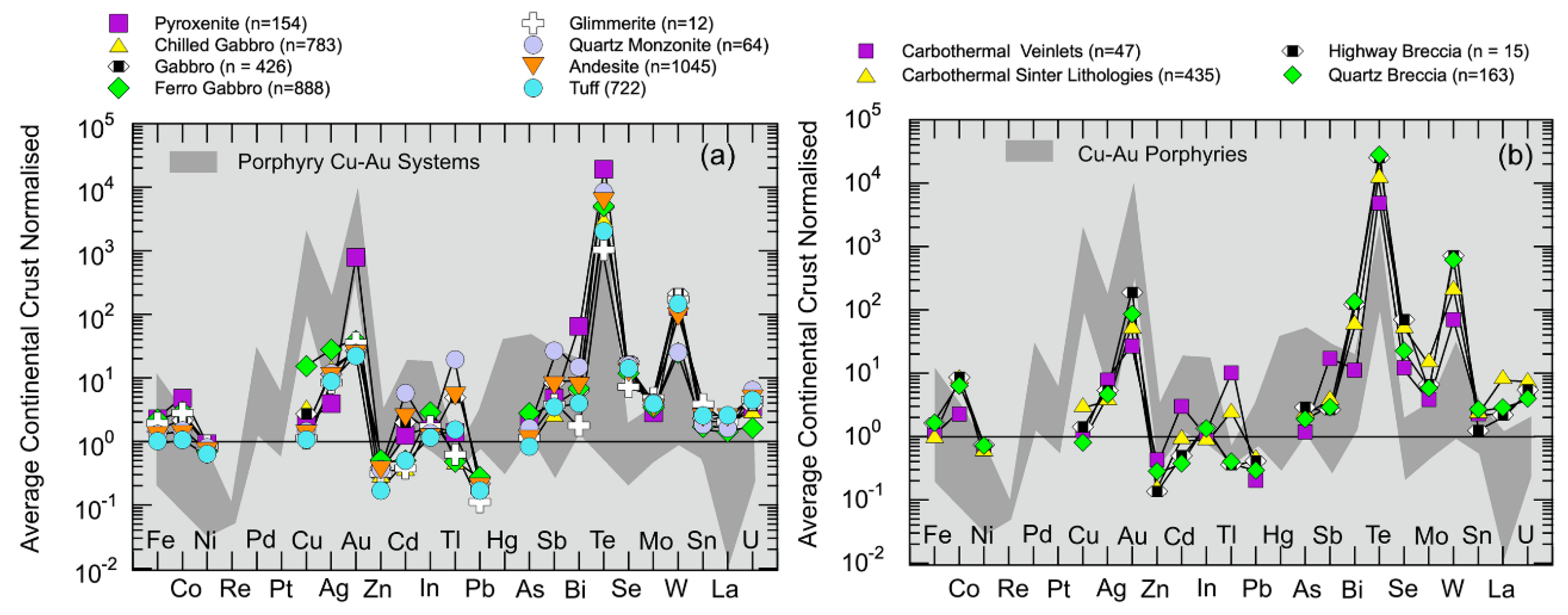
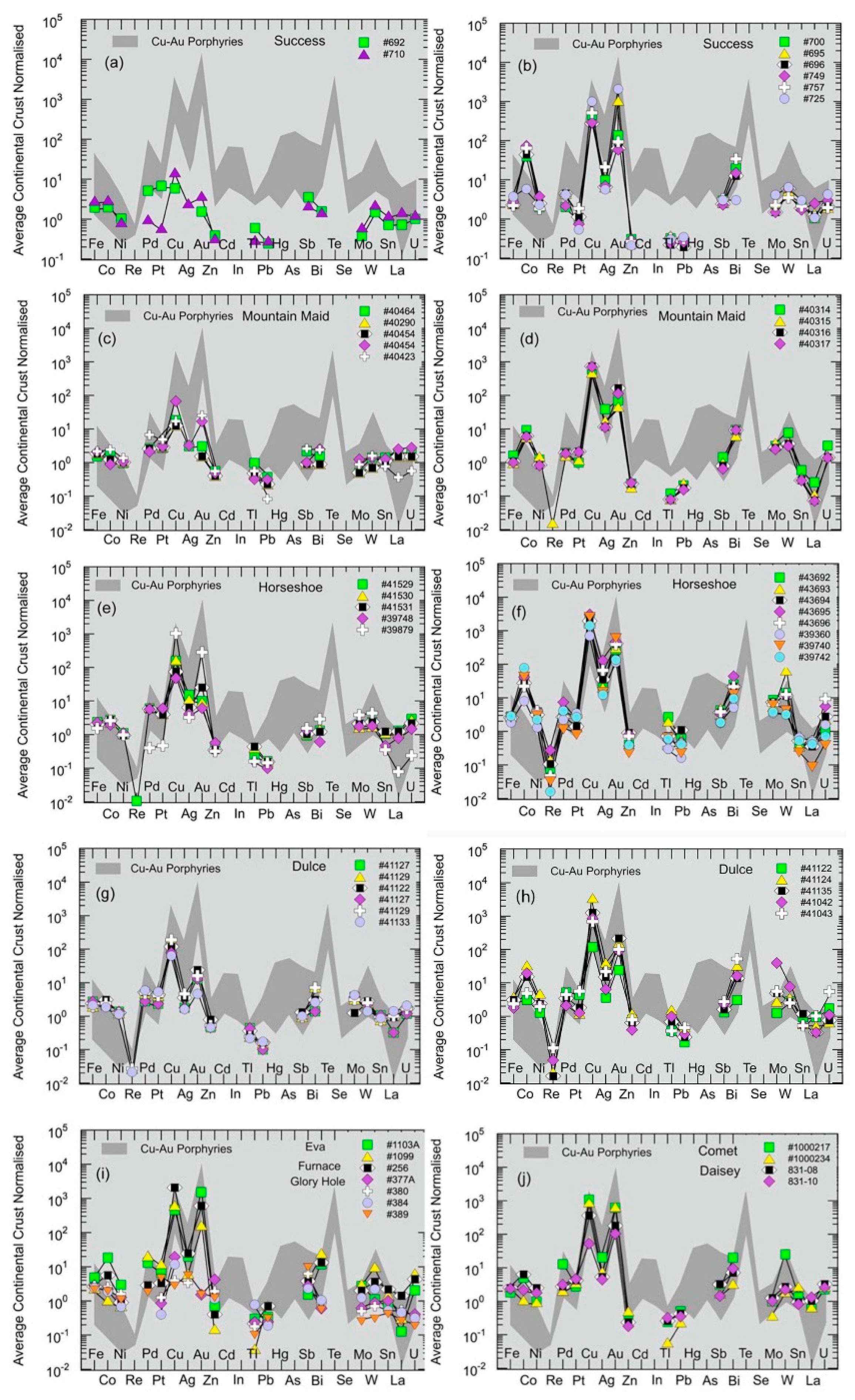
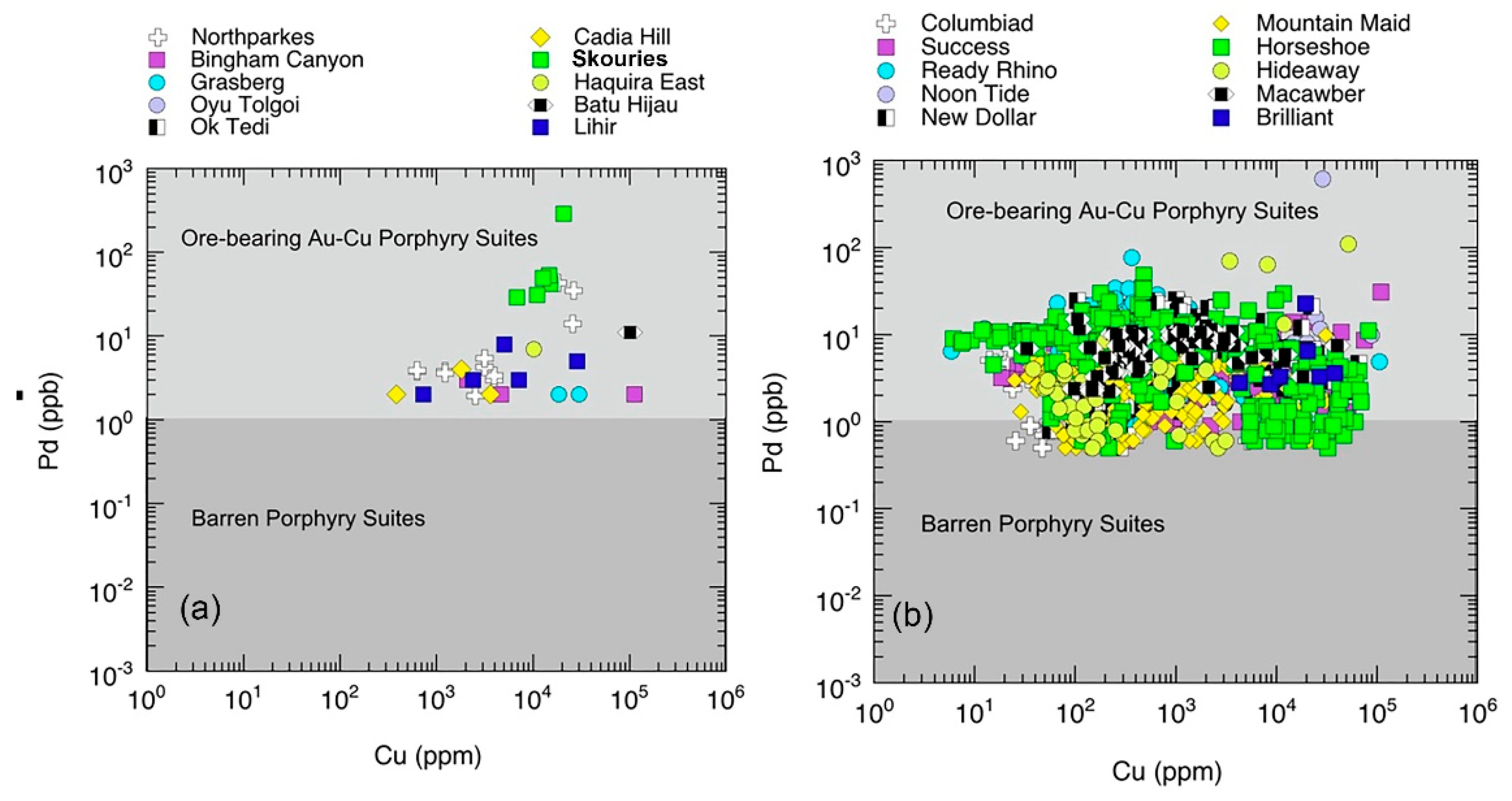
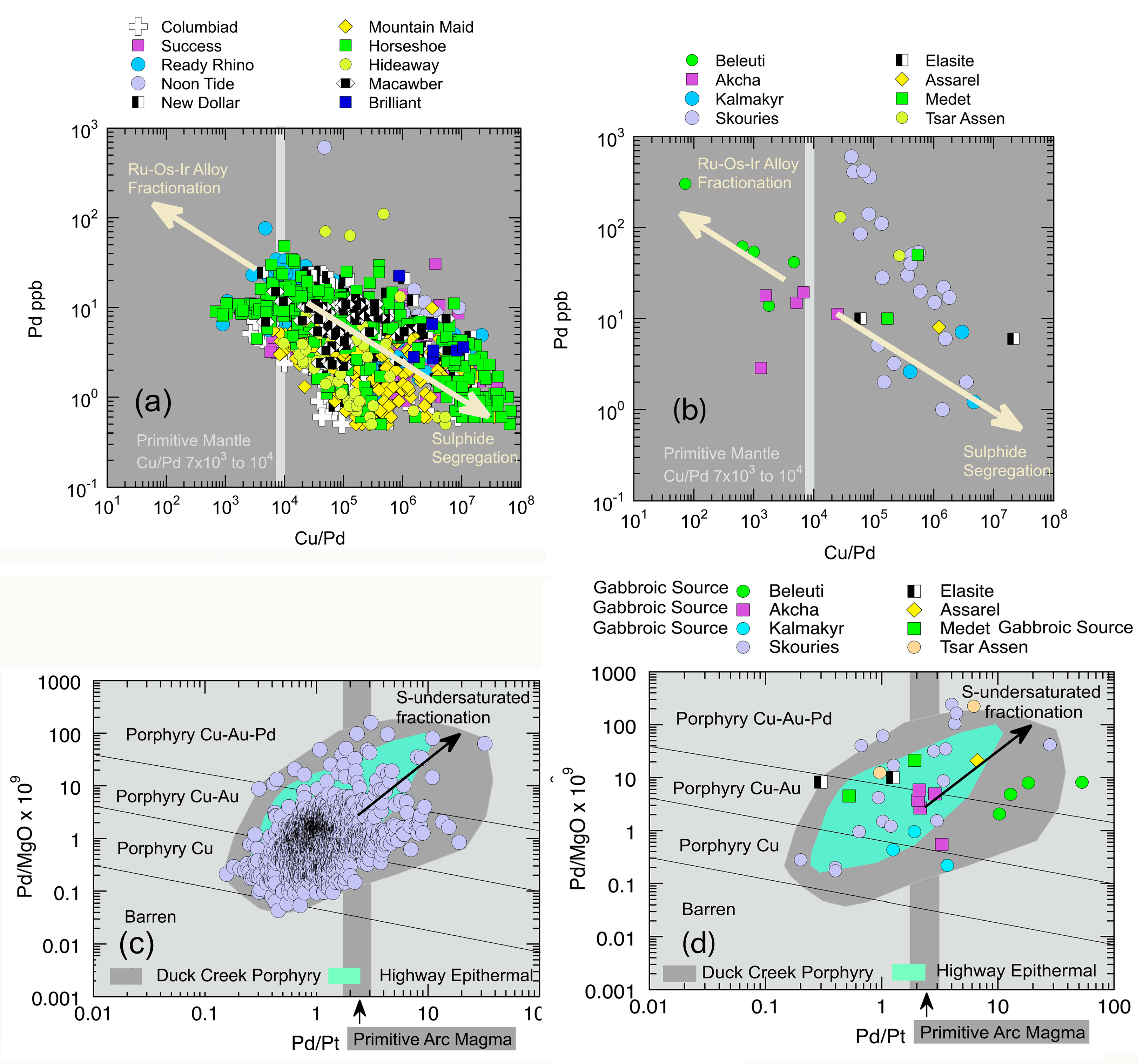
5.1.5. Chalcophile Element and Highly Siderophile Element Systematics
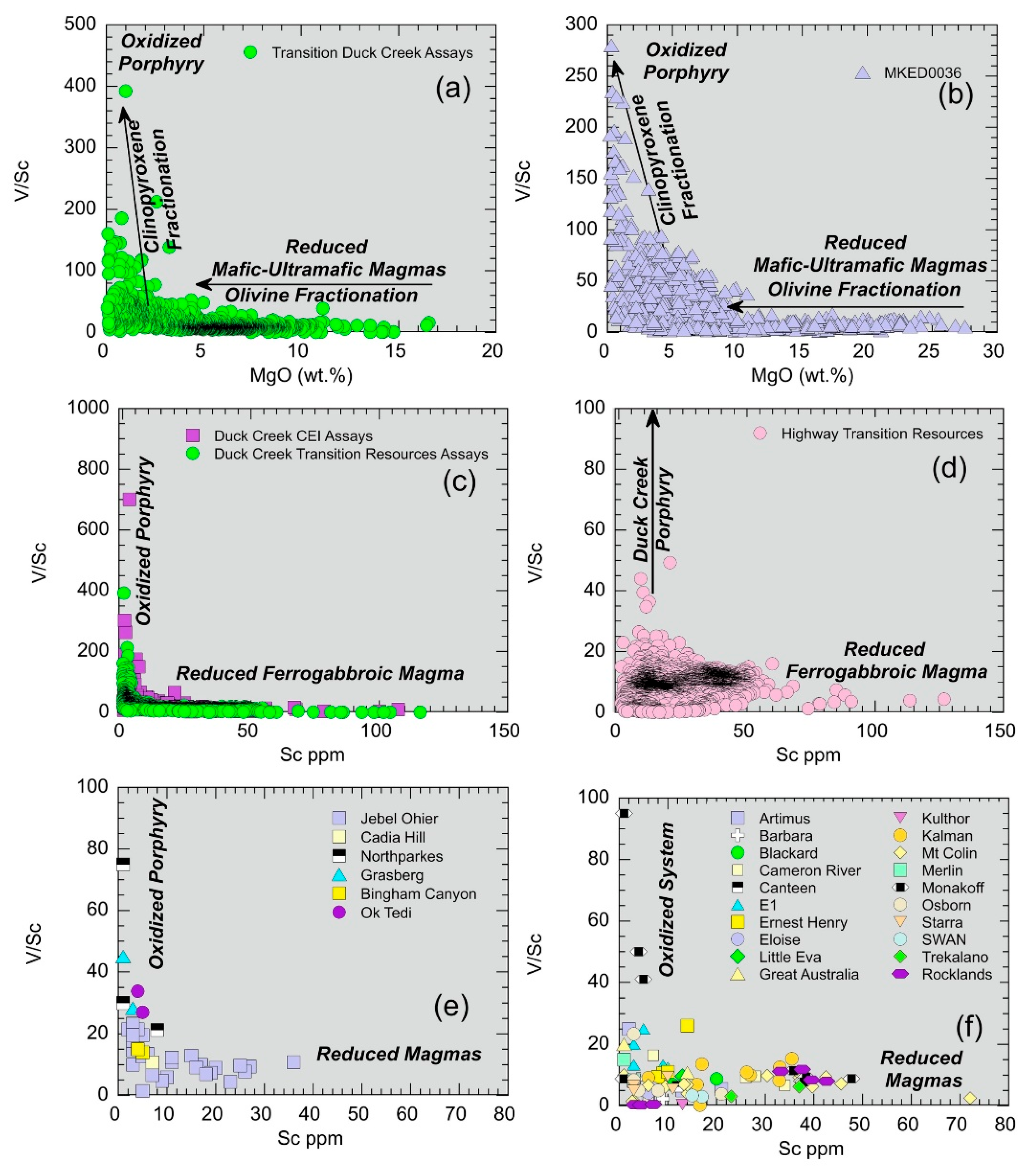
5.1.6. Deducing Redox Conditions using Scandium and Vanadium Systematics
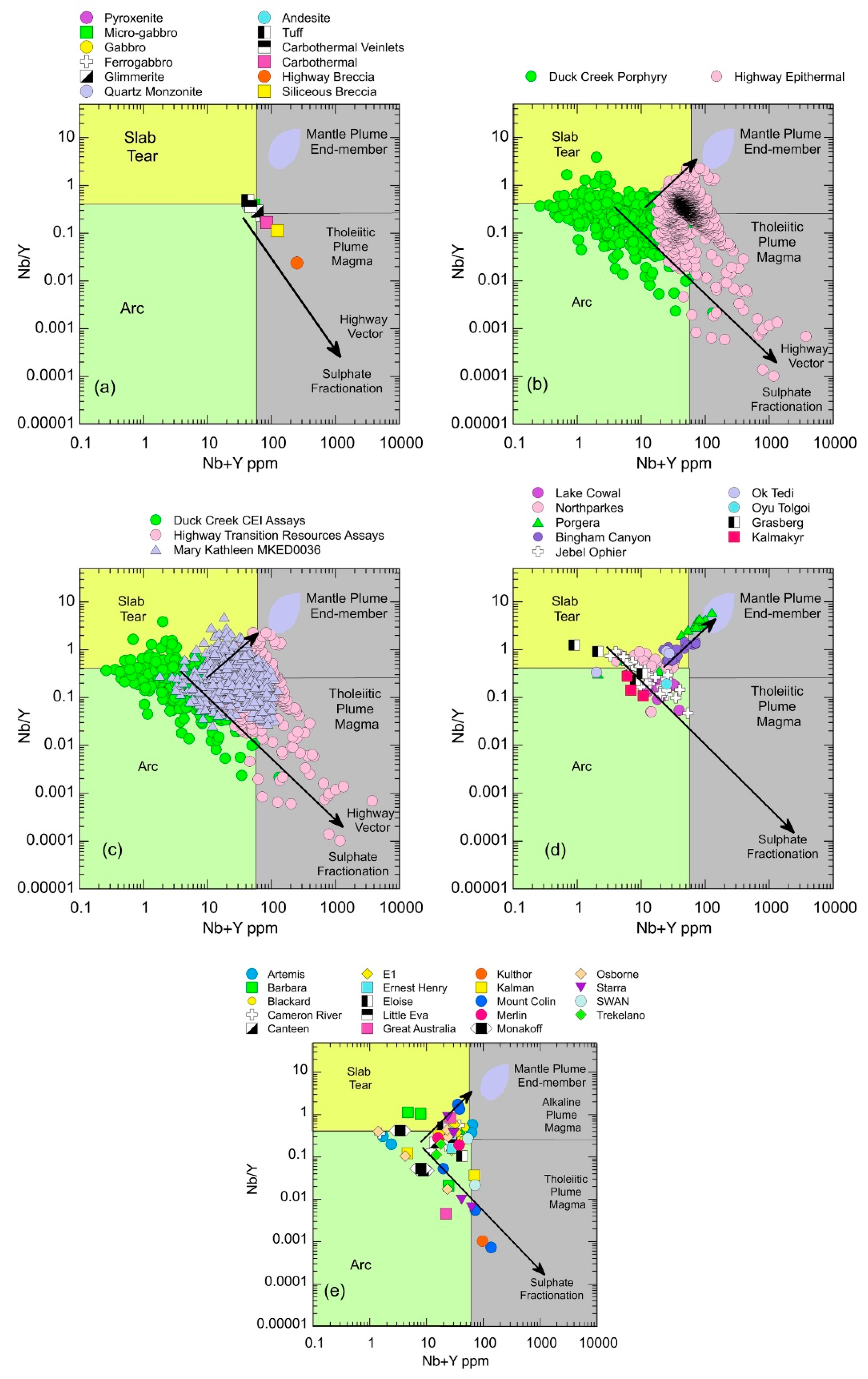
5.1.7. Trace Element Vectors Indicating Tectonic Setting and Role of Sulphate Fractionation
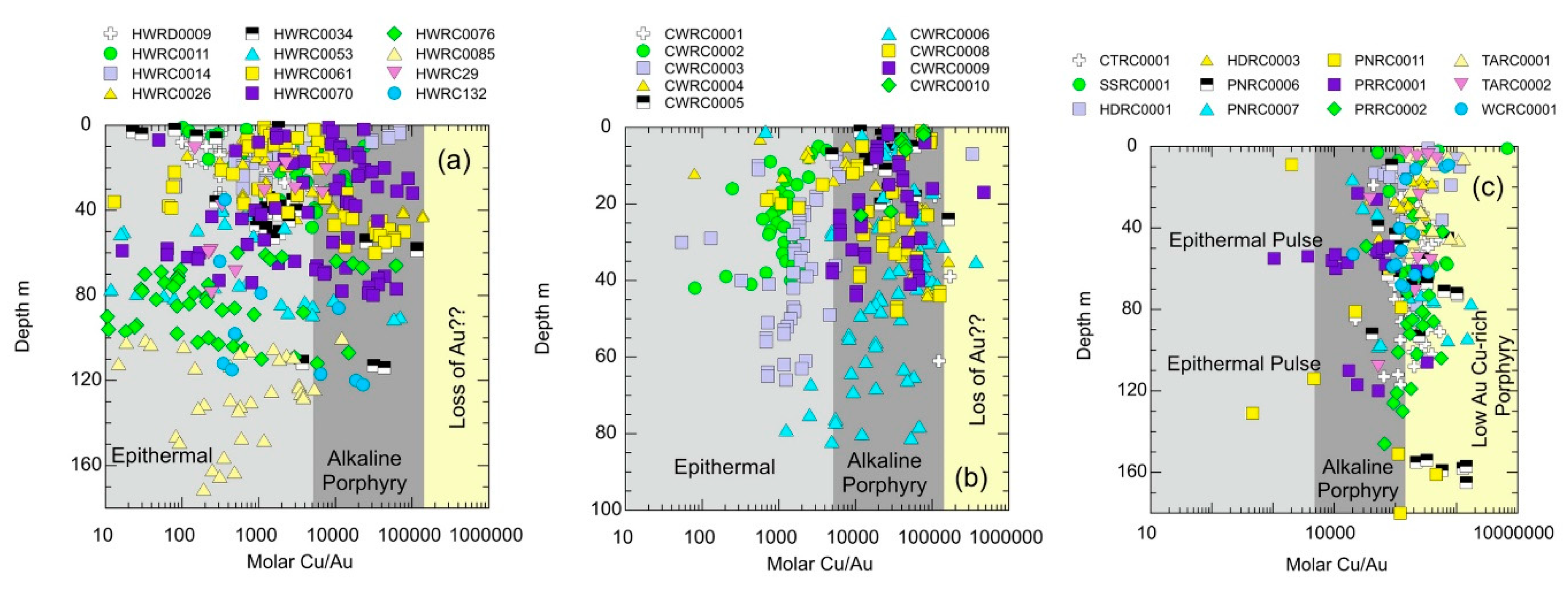
5.1.8. Variation in Molar Cu/Au with Depth
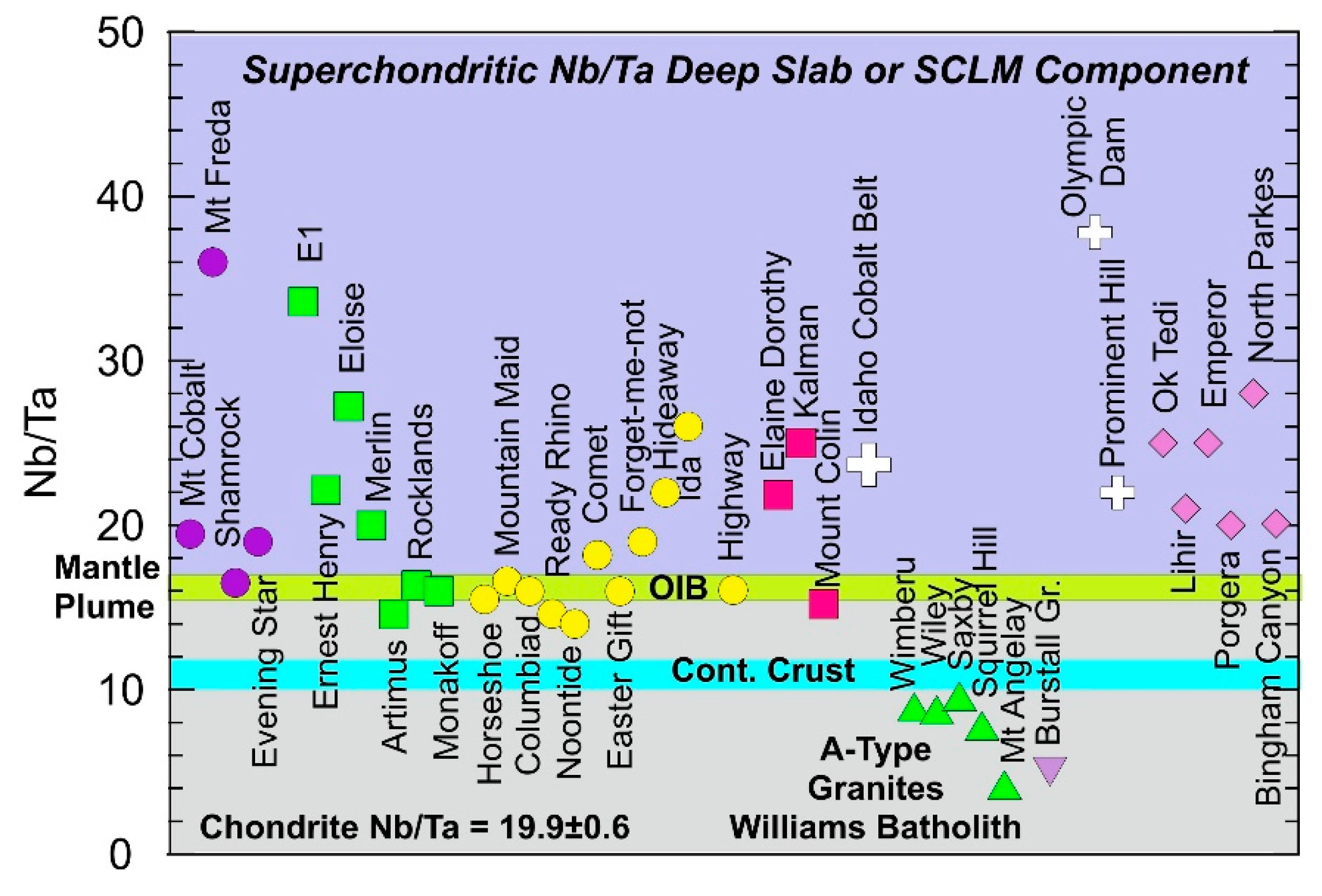
5.1.9. Nb/Ta Ratios and the Role of a Mantle Component in NWMP Mineralisation
6. Summary and Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sillitoe, R.H. Porphyry copper systems. Econ. Geol. 2020, 105, 3–41. [Google Scholar] [CrossRef]
- Richards, J.P. Porphyry copper deposit formation in arcs: What are the odds? Geosphere 2021, 18, 130–155. [Google Scholar] [CrossRef]
- Sillitoe, R.H. A plate tectonic model for the origin of porphyry copper deposits. Econ. Geol. 1972, 67, 184–197. [Google Scholar] [CrossRef]
- Wilkinson, J.J. Triggers for the formation of porphyry ore deposits in magmatic arcs. Nature Geosci. 2013, 6, 917–925. [Google Scholar] [CrossRef]
- Lamont, T.N.; Loader, M.A.; Roberts, N.M.; Cooper, F.J.; Wilkinson, J.J.; Bevan, D.; Gorecki, A.; Kemp, A.; Elliott, T.; Gardiner, N.J.; et al. Porphyry copper formation driven by water-fluxed crustal melting during flat-slab subduction. Nature Geosci. 2024, 17, 1306–1315. [Google Scholar] [CrossRef]
- Hou, Z.; Yang, Z.; Lu, Y.; Kemp, A.; Zheng, Y.; Li, Q.; Tang, J.; Yang, Z.; Duan, L. A genetic linkage between subduction-and collision-related porphyry Cu deposits in continental collision zones. Geology 2015, 43, 247–250. [Google Scholar] [CrossRef]
- Wang, Z.; Zheng, Y.; Chiaradia, M.; Xu, B.; Hao, H.; Hou, Z. Divergent paradigms of porphyry copper deposits in subduction and collision zones. Earth ArXiv (under review). 2025. [Google Scholar]
- Collerson, K.; Lal, S.; Williams, Q.; Rost, S. Cracking the metallogenic code for Fijian epithermal gold mineralisation. Pacrim 2015 Congress Extended Abstracts, Hong Kong, China, 2015; pp. 219–226. [Google Scholar]
- Holm, R.J.; Tapster, S.; Jelsma, H.A.; Rosenbaum, G.; Mark, D.F. Tectonic evolution and copper-gold metallogenesis of the Papua New Guinea and Solomon Islands region. Ore Geol. Rev. 2019, 104, 208–226. [Google Scholar] [CrossRef]
- Ward, J. F.; Rosenbaum, G.; Ubide, T.; Sandiford, M. Slab segmentation, anomalous arc volcanism, and giant porphyry copper deposits in Indonesia. Earth Planet. Sci. Lett. 2024, 626, 118532. [Google Scholar] [CrossRef]
- Collerson, K.D; Williams, Q.; Ewart, A.E.; Murphy, D.T. Origin of HIMU and EM-1domains sampled by ocean island basalts, kimberlites and carbonatites: The role of CO2-fluxed lower mantle melting in thermochemical upwellings. Phys. Earth. Planet. Int. 2010, 181, 112–131. [Google Scholar] [CrossRef]
- Tarkian, M.; Hünken, U.; Tokmakchieva, M.; Bogdanov, K. Precious-metal distribution and fluid-inclusion petrography of the Elatsite porphyry copper deposit, Bulgaria. Miner. Depos. 2003, 38, 261–281. [Google Scholar] [CrossRef]
- Economou-Eliopoulos, M; Eliopoulos, DG. Palladium, platinum and gold concentration in porphyry copper systems of Greece and their genetic significance. Ore Geol. Rev. 2000, 16, 59–70. [Google Scholar] [CrossRef]
- Economou-Eliopoulos, M. Exploration for Deposits of Platinum-Group Elements. Mineral. Assoc. Canada Short Course 2005, 35, 203–246. [Google Scholar]
- Economou-Eliopoulos, M. Platinum-group elements (PGE) in various geotectonic settings: opportunities and risks. Hell. J. Geosci. 2010, 45, 65–82. [Google Scholar]
- Economou-Eliopoulos, M.; Zaccarini, F.; Garuti, G. Fertility indicators for porphyry-Cu- Au+Pd±Pt deposits: Evidence from Skouries, Chalkidiki Peninsula, Greece, and comparison with worldwide mineralizations. Minerals 2023, 13, 1413. [Google Scholar] [CrossRef]
- Sun, W.; Huang, R-f.; Li, H.; Hu, Y-b.; Zhang, C-c.; Sun, S-J.; Zhang, L-P.; Ding, X.; Li, C-Y.; Zartman, R.E.; Ling, M-x. Porphyry deposits and oxidized magmas. Ore Geol. Rev. 2015, 65, 97–131. [Google Scholar] [CrossRef]
- Yuan, F.; Zhou, T.; Zhang, D.; Jowitt, S.M.; Keays, R.; Liu, S.; Fan, Y. Siderophile and chalcophile metal variations in basalts: Implications for the sulphide saturation history and Ni–Cu–PGE mineralization potential of the Tarim continental flood basalt province, Xinjiang Province, China. Ore Geol. Rev. 2012, 45, 5–15. [Google Scholar] [CrossRef]
- Li, J.; Liu, W.; Su, L.; Li, D.; Zhang, S.; Chen, H. Porphyry copper mineralization triggered by sulfate reduction and alkali metasomatism: Constraints from an experimental investigation. GSA Bull. 2023, 135, 2407–2427. [Google Scholar] [CrossRef]
- Simmons, S.F.; White, N.C.; John, D.A. Geological characteristics of epithermal precious and base metal deposits. Econ. Geol. 2005, 100, 485–522. [Google Scholar]
- Sillitoe, R.H. Epithermal paleosurfaces. Miner Deposita 2015, 50, 767–973. [Google Scholar] [CrossRef]
- Dong, G.; Morrison, G.; Jaireth, S. Quartz textures in epithermal veins, Queensland: Classification, origin and implication. Econ. Geol. 1995, 90, 1841–1856. [Google Scholar] [CrossRef]
- Kesler, S.E.; Wilkinson, B.H. The role of exhumation in the temporal distribution of ore deposits. Econ. Geol. 2006, 101, 919–922. [Google Scholar] [CrossRef]
- Groves, D.I.; Bierlein, F.P.; Meinert, L.D.; Hitzman, M.W. Iron oxide-copper-gold (IOCG) deposits through Earth History: Implications for origin, lithospheric setting and distinction from other epigenetic iron oxide deposits. Econ. Geol. 2010, 105, 641–654. [Google Scholar] [CrossRef]
- Kalinin, A.A.; Kudryashov, N.M. Porphyry-related metamorphosed Au-Ag and Cu-Mo Deposits in the Precambrian of the Fennoscandian Shield. Minerals 2021, 11, 139. [Google Scholar] [CrossRef]
- Marshall, A. E. Low temperature-low pressure (“epithermal”) siliceous vein deposits of the North Pilbara granite-greenstone terrane, Western Australia. Australian Geological Survey Organisation Record 2000, 2000/1. [Google Scholar]
- McCuaig, T. C.; Behn, M.; Stein, H.; Hagemann, S. G.; McNaughton, N. J.; Cassidy, K. F.; Champion, D. C.; Wyborn, L. The Boddington gold mine: a new style of Archaean Au-Cu deposit. Geosci. Australia Record 2001, 2001/37, 453–455. [Google Scholar]
- Lopes, A.A.C.; Moura, M.A. Gold Deposit, Tapajós Mineral Province (Brazil): Geology, Petrology and Fluid Inclusion Evidence for Ore-Forming Processes. Minerals 2019, 9, 29. [Google Scholar] [CrossRef]
- Mumin, A.H.; Somarin, A.K.; Jones, B.; Corriveau, L.; Ootes, L.; Camier, J. The IOCG- porphyry-epithermal continuum of deposit types in the Great Bear magmatic zone, North- west Territories, Canada. In Exploring for Iron Oxide Copper-Gold Deposits: Geol. Assoc. Canada, Short Course Notes; Corriveau, L., Mumin, H., Eds.; 2020; Volume 20, pp. 59–78. [Google Scholar]
- Richards, J.P.; Mumin, A.H. Magmatic-hydrothermal processes within an evolving Earth: Iron oxide-copper-gold and porphyry Cu ± Mo ± Au deposits. Geology 2013, 41, 767–770. [Google Scholar] [CrossRef]
- Tiddy, C.J; Giles, D. Suprasubduction model for metal endowment at 1.60-1.57 Ga in eastern Australia. Ore Geol. Rev. 2020, 122, 103483. [Google Scholar] [CrossRef]
- Tornos, F.; Velasco, F.; Barra, F.; Morata, D. The Tropezón Cu–Mo–(Au) deposit, Northern Chile: the missing link between IOCG and porphyry copper systems? Miner Deposita 2010, 45, 313–321. [Google Scholar] [CrossRef]
- Sillitoe, RH. Iron oxide–copper–gold deposits: an Andean view. Miner Deposita 2003, 38, 787–812. [Google Scholar] [CrossRef]
- Brauhart, C.W.; Groves, D.I. Australian intracratonic copper-gold and allied deposits: A clan of mineral systems. Ore Geol. Rev. 2025, 149, 158–182. [Google Scholar] [CrossRef]
- Williams, P.; Barton, M.D.; Johnson, D.A.; Fontboté, L.; Ad, H.; Mark, G.; Oliver, N.H.S.; Marschik, R. Iron oxide copper–gold deposits: geology, space–time distribution, and possible modes of origin. Econ. Geol. 2005, 100, 371–406. [Google Scholar]
- Gibson, G.M.; Meixner, A.J.; Withnall, I.W.; Korsch, R.J; Hutton, L.J.; Jones, L.E.A; Holzschuh, J.; Costelloe, R.D.; Henson, P.A.; Saygin, E. Basin architecture and evolution in the Mount Isa mineral province, northern Australia: Constraints from deep seismic reflection profiling and implications for ore genesis. Ore Geol. Rev. 2016, 76, 414–441. [Google Scholar] [CrossRef]
- Pollard, P.J. An intrusion-related origin for Cu- Au mineralization in iron oxide-copper-gold (IOCG) provinces: Mineral. Deposita 2006, 4, 179–187. [Google Scholar] [CrossRef]
- Baker, T. Alteration, mineralisation and fluid evolution at the Eloise Cu – Au deposit, Cloncurry district, NW Queensland. Econ. Geol. 1998, 93, 1213–1236. [Google Scholar] [CrossRef]
- Mark, G.; Oliver, N.H.S.; Carew, M.J. Insights into the genesis and diversity of epigenetic Cu – Au mineralisation in the Cloncurry district. Mt Isa Inlier, northwest Queensland. Aust. J. Earth Sci. 2006, 53, 109–124. [Google Scholar] [CrossRef]
- Williams, P. J.; Dong, G.; Ryan, C. G.; Pollard, P. J.; Rotherham, J.; Mernagh, T. P.; Chapman, I. H. Geochemistry of hypersaline fluid inclusions from the Starra (Fe oxide) – Cu – Au deposit, Cloncurry district, Queensland. Econ. Geol. 2001, 96, 875–884. [Google Scholar]
- Marshall, L.J.; Oliver, N.H.S. Constraints on hydrothermal fluid pathways within Mary Kathleen Group stratigraphy of the Cloncurry iron-oxide–copper–gold District, Australia. Precambrian Res. 2008, 163, 151–158. [Google Scholar] [CrossRef]
- Collerson, K. D. Cobalt and HREE Mineral Systems in the Mount Isa Block. Report for Geological Survey of Queensland, 2019. Available online: https://qdexguest.dnrm.qld.gov.au/portal/site/qdex/search?REPORT_ID=111110&COLLECTION_ID=999.
- Weidendorfer, D.; Schmidt, M.X.; Mattsson, H.B. Fractional crystallization of Si-undersaturated alkaline magmas leading to unmixing of carbonatites on Brava Island (Cape Verde) and a general model of carbonatite genesis in alkaline magma suites. Contrib. Mineral. Petrol. 2016, 171, 43. [Google Scholar] [CrossRef]
- Pfänder, J.A.; Jung, S.; Münker, C.; Stracke, A.; Mezger, K. A possible high Nb/Ta reservoir in the continental lithospheric mantle and consequences on the global Nb budget-Evidence from continental basalts from Central Germany. Geochim. Cosmochim. Acta 2012, 77, 232–251. [Google Scholar] [CrossRef]
- Tang, M.; Lee, C-T. A.; Chen, K.; Erdman, M.; Costin, G.; Jiang, H. Nb/Ta systematics in arc magma differentiation and the role of arclogites in continent formation. Nature Commun. 2019, 10, 235. [Google Scholar] [CrossRef] [PubMed]
- Collerson, K.D. Core lithologies from Elaine Dorothy and Blue Caesar Prospects: A geochemical interpretation based on assays from Elaine Dorothy; Mary Kathleen Belt, North Queensland. Report for China Yunnan Copper Resources Ltd. In HDR Salva; 2013; p. 95 pp. [Google Scholar]
- Wilson, I.H. Volcanism on a Proterozoic continental margin in northwestern Queensland. Precambrian Res. 1978, 7, 205–235. [Google Scholar] [CrossRef]
- Korsch, R.J.; Huston, D.L.; Henderson, R.A.; Blewett, R.S.; Withnall, I.W.; Fergusson, C.L.; Collins, W.J.; Saygin, E.; Kositcin, N.; Meixner, A.J.; Chopping, R.; Henson, P.A.; Champion, D.C.; Hutton, L.J.; Wormald, R.; Holzschuh, J.; Costelloe, R.D. Crustal architecture and geodynamics of North Queensland, Australia: Insights from deep seismic reflection profiling. Tectonophysics 2012, 572, 76–99. [Google Scholar] [CrossRef]
- Korsch, R.J.; Doublier, M.P.; Brown, D.D.; Simpson, J.M.; Cross, A.J.; Ross, D.; Costelloe, R.D.; Jiang, W. Crustal architecture and tectonic development of western Queensland, Australia, based on deep seismic reflection profiling: Implications for Proterozoic continental assembly and dispersal. Tectonophysics 2024, 878, 230302. [Google Scholar] [CrossRef]
- Betts, P.G.; Giles, D.; Schaefer, B.F.; Mark, G. 1600–1500 Ma hotspot track in eastern Australia: implications for Mesoproterozoic continental reconstructions. Terra Nova 2007, 19, 496–501. [Google Scholar] [CrossRef]
- Betts, P.G.; Giles, D.; Foden, F.; Schaefer, B.F.; Mark, G; Pankhurst, M.J.; Caroline Forbes, C.J.; Williams, H.A.; Chalmers, N.C.; Hills, Q. Mesoproterozoic plume-modified orogenesis in eastern Precambrian Australia. Tectonophysics 2009, 28, TC3006. [Google Scholar] [CrossRef]
- Gibson, G.M.; Champion, D.C.; Huston, D.L.; Withnall, I.W. Orogenesis in Paleo-Mesoproterozoic eastern Australia: a response to Arc–Continent and Continent–Continent Collision During Assembly of the Nuna Supercontinent. Tectonics 2020, 39, E2019TC005717. [Google Scholar] [CrossRef]
- Li, J.; Song, X. Tearing of Indian mantle lithosphere from high-resolution seismic images and its implications for lithosphere coupling in southern Tibet. Proc. Natl. Acad. Sci. USA 2018, 115, 8296–8300. [Google Scholar] [CrossRef] [PubMed]
- Rosenbaum, G.; Gasparon, M.; Lucente, F. P.; Peccerillo, A.; Miller, M. S. Kinematics of slab tear faults during subduction segmentation and implications for Italian magmatism. Tectonics 2008, 27. [Google Scholar] [CrossRef]
- Momme, P.; Oskarsson, N.; Keays, R.R.; Horan, M.F.; Brandon, A.D.; Neal, C.R. Platinum- group elements in the Icelandic rift system; melting processes and mantle sources beneath Iceland. Chem. Geol. 2003, 196, 209–234. [Google Scholar] [CrossRef]
- Webber, A.P.; Roberts, S.; Taylor, R.N.; Pitcairn, I.K. Golden plumes: Substantial gold enrichment of oceanic crust during ridge-plume interaction. Geology 2013, 41, 187–190. [Google Scholar] [CrossRef]
- Williams, P. R. Nature and timing of early extensional structures in the Mitakoodi Quartzite, Mount Isa Inlier, north- west Queensland. Aust. J. Earth Sci. 1989, 36, 283–296. [Google Scholar] [CrossRef]
- Potma, W.A.; Betts, P.G. Extension-related structures in the Mitakoodi Culmination: Implications for the nature and timing of extension, and effect on later shortening in the eastern Mt Isa Inlier. Aust. J. Earth Sci. 2006, 53(1), 55–67. [Google Scholar] [CrossRef]
- Skirrow, R.G. Iron oxide copper-gold (IOCG) deposits A review (Part 1): Settings, mineralogy, ore geochemistry and classification.ore geochemistry and classification. Ore Geol. Rev. 2022, 140, 104569. [Google Scholar] [CrossRef]
- MacCready, T.; Goleby, B.R.; Goncharov, A.; Drummond, B.J.; Lister, G.S. A framework of overprinting orogens based on interpretation of the Mount Isa deep seismic transect. Econ. Geol. 1989, 93, 1422–1434. [Google Scholar] [CrossRef]
- O’Dea, M.G.; Betts, P.G.; MacCready, T.; Aillères, L. Sequential development of a mid-crustal fold-thrust complex: evidence from the Mitakoodi Culmination in the eastern Mt. Isa Inlier, Australia. Aust. J. Earth Sci. 2006, 53, 69–90. [Google Scholar] [CrossRef]
- Neumann, N.L.; Gibson, G. M.; Southgate, P. N. New SHRIMP age constraints on the timing and duration of magmatism and sedimentation in the Mary Kathleen Fold Belt, Mt Isa Inlier, Australia. Aust. J. Earth Sci. 2009, 56, 965–983. [Google Scholar] [CrossRef]
- Hackenberg, O. The Wimberu Granite: Granitic phases, geochemistry, geochronology and thermobarometry. Unpublished BSc. Hons Thesis, University of Queensland, 2018; 143p. [Google Scholar]
- Clark, R.; Smith, D.J.; Naden, J.; Holwell, D.; Mann, S. Mineralogical constraints on the genesis of an alkalic-type epithermal Au-Te deposit: Tuvatu, Fiji. Ore Geol. Rev. 2023, 154, 105279. [Google Scholar] [CrossRef]
- Ryan, B. The Nain-Churchill Boundary and the Nain Plutonic Suite: A Regional Perspective on the Geologic Setting of the Voisey’s Bay Ni-Cu-Co Deposit. Econ. Geol. 2000, 95, 703–724. [Google Scholar]
- Baksheev, I. A.; Prokofev, V.Y.; Zaraisky, G.P.; Chitalin, A.F.; Yapaskurt, V.O. Tourmaline as a prospecting guide for the porphyry-style deposits. Eur. J. Mineral. 2012, 24, 957–979. [Google Scholar] [CrossRef]
- Wallace, C.J.; Maher, K.C. Phyllic alteration and the implications of fluid composition at the Copper Flat hydrothermal System, New Mexico, USA. Ore Geol. Rev. 2019, 104, 273–293. [Google Scholar] [CrossRef]
- Reich, M.; Deditius, A.; Chryssoulis, S.; Li, J-W.; Ma, C-Q.; Parada, M.A.; Barra, F.; Mittermayr, F. Pyrite as a record of hydrothermal fluid evolution in a porphyry copper system: A SIMS/EMPA trace element study. Geochim. Cosmochim. Acta 2013, 104, 42–62. [Google Scholar] [CrossRef]
- Philpott, R.C. Geology and geochemistry of the Mount Cobalt Co-Cu-Au-REE IOCG deposit, Cloncurry District, Queensland. Unpublished B.Env.Sc Hons Thesis, Monash University, 2010; p. 138pp. [Google Scholar]
- Peccerillo, A; Taylor, SR. Geochemistry of Eocene calk-alkaline volcanic rocks from the Kastamonu area, northern Turkey. Contrib. Mineral. Petrol. 1976, 58, 63–81. [Google Scholar] [CrossRef]
- Mathieu, L. Quantifying Hydrothermal Alteration: A Review of Methods. Geosciences 2019, 8, 245. [Google Scholar] [CrossRef]
- Hastie, A.R. Classification of Altered Volcanic Island Arc Rocks using Immobile Trace Elements: Development of the Th-Co Discrimination Diagram. J. Petrol. 2007, 48, 2341–2357. [Google Scholar] [CrossRef]
- Muller, D.; Groves, D.I. Potassic Igneous Rocks and Associated Gold-Copper Mineralization, 5th Ed. ed; Springer, 2019; p. 398pp. [Google Scholar]
- Soder, C.G.; Dunga, J.; Romer, R.L. Continental subduction controls regional magma heterogeneity and distribution of porphyry deposits in post-collisional settings. Geochim. Cosmochim. Acta 2024, 375, 217–228. [Google Scholar]
- McDonough, W. F.; Sun, S.-S. The composition of the Earth. Chem. Geol. 1995, 120, 223–253. [Google Scholar] [CrossRef]
- Lyubetskaya, T.; Korenaga, J. Chemical composition of the Earth’s primitive mantle and its variance. J. Geophys. Res. 2007, 112, B03211. [Google Scholar]
- Hikov, A. Geochemistry of strontium in advanced argillic alteration systems – Possible guide to exploration. In Bulgarian Geol. Soc., Abtr. An. Sci, Confr; 2004; pp. 29–31. [Google Scholar]
- Hikov, A.; Lerouge, C.; Velinova, N. Geochemistry of alunite group minerals in advanced argillic altered rocks from the Asarel porphyry copper deposit, Central Srednogorie. Rev. Bulg. Geol. Soc. 2010, 71, 133–148. [Google Scholar]
- Mavrogonatos, C.; Voudouris, P.; Spry, P.G.; Melfos, V.; Klemme, S.; Jasper Berndt, J.; Tim Baker, T.; Moritx, R.; Bissig, T.; Monecke, T.; Zaccarini, T. Mineralogical study of the advanced argillic alteration zone at the Konos Hill Mo–Cu–Re–Au Porphyry Prospect, NE Greece. Minerals 2018, 8, 479. [Google Scholar]
- Rudnick, R.L.; Gao, S. Composition of the continental crust. In The Crust; Holland, H.D., Turekian, K.K., Eds.; Elsevier-Pergamon: Oxford, 2003; pp. 1–64. [Google Scholar]
- Brauhart, C.W.; Grunsky, E.C.; Hagemann, S.G. Magmato-hydrothermal space: A new metric for geochemical characterisation of metallic ore deposits. Ore Geol. Rev. 2017, 86, 867–895. [Google Scholar] [CrossRef]
- Holwell, D.A.; Fiorentini, M.; McDonald; Lu, Y.; Giuliani, A.; Smith, D.J.; Keith, M.; Locmelis, M. A metasomatized lithospheric mantle control on the metallogenic signature of post-subduction magmatism. Nature Commun. 2019, 10, 3511. [Google Scholar] [CrossRef] [PubMed]
- Holwell, D. A.; McDonald, I. A review of the behaviour of platinum group elements within natural magmatic sulfide ore systems the importance of semimetals in governing partitioning behaviour. Platin. Met. Rev. 2010, 54, 26–36. [Google Scholar] [CrossRef]
- Grundler, P. V.; et al. Speciation of aqueous tellurium(IV) in hydrothermal solutions and vapors, and the role of oxidized tellurium species in Te transport and gold deposition. Geochim. Cosmochim. Acta 2013, 120, 298–325. [Google Scholar] [CrossRef]
- Brugger, J.; et al. XAS evidence for the stability of polytellurides in hydrothermal fluids up to 599 °C, 800 bar. Am. Mineral. 2012, 97, 1519–1522. [Google Scholar]
- McFall, K.A.; Naden, J.; Roberts, S.; Baker, T.; Spratt, J.; McDonald, I. Platinum-group minerals in the Skouries Cu-Au (Pd, Pt, Te) porphyry Deposit. Ore Geol. Rev. 2018, 99, 344–364. [Google Scholar] [CrossRef]
- Park, J-W; Campbell, I.H. Chalcophile element fertility and the formation of porphyry Cu ± Au Deposits. Mineral. Deposita 2019, 54, 657–670. [Google Scholar] [CrossRef]
- Cocker, H.A.; Valente, D.L.; Park, J-W; Campbell, I.H. Using platinum group elements to identify sulfide saturation in a porphyry Cu system: the El Abra porphyry Cu deposit, northern Chile. J. Petrol. 2015, 56, 2491–2514. [Google Scholar] [CrossRef]
- Hao, H.; Campbell, I.H.; Park, J.-W.; Cooke, D.R. Platinum-group element geochemistry used to determine Cu and Au fertility in the Northparkes igneous suites, New South Wales, Australia. Geochim. Cosmochim. Acta 2017, 216, 372–392. [Google Scholar] [CrossRef]
- Lowczak, J.N.; Campbell, I.H.; Cocker, H.; Park, J.-W.; Cooke, D.R. Platinum-group element geochemistry of the Forest Reef Volcanics, southeastern Australia: Implications for porphyry Au-Cu mineralisation. Geochim. Cosmochim. Acta 2018, 220, 385–406. [Google Scholar] [CrossRef]
- Mungall, J.E; Brenan, J.M. Partitioning of platinum-group elements and Au between sulfide liquid and basalt and the origins of mantle- crust fractionation of the chalcophile elements. Geochim. Cosmochim. Acta 2014, 125, 265–289. [Google Scholar] [CrossRef]
- Crocket, J.H. PGE in fresh basalt, hydrothermal alteration products, and volcanic incrustations of Kilauea volcano, Hawaii. Geochim Cosmochim. Acta 2000, 64, 1791–1807. [Google Scholar] [CrossRef]
- Park, J.-W.; Campbell, I.H.; Kim, J. Abundances of platinum group elements in native sulfur condensates from the Niuatahi-Motutahi submarine volcano, Tonga rear arc: implications for PGE mineralization in porphyry deposits. Geochim. Cosmochim. Acta 2016, 174, 236–246. [Google Scholar] [CrossRef]
- Lui, B.; Zhang, Z.; Cheng, Z.; Xie, Q.; Turesebekov, A.; Nurtaev, B.; Santosh, M. Platinum group elements in gabbroic intrusions from the Valerianov-Beltau-Kurama arc: Implications for genesis of the Kalmakyr porphyry Cu–Au deposit. Geol. J. 2020, 56, 46–59. [Google Scholar]
- Li, Z.X.A.; Lee, C.T.A. The constancy of upper mantle fO2 through time inferred from V/Sc ratios in basalts: Earth Planet. Sci. Lett. 2004, 228, 483–493. [Google Scholar]
- Toplis, M.J.; Corgne, A. An experimental study of element partitioning between magnetite, clinopyroxene and iron-bearing silicate liquids with particular emphasis on vanadium. Mineral. Petrol. 2002, 144, 22–37. [Google Scholar] [CrossRef]
- Loucks, R.R. Distinctive composition of copper-ore-forming arc magmas: Aust. Earth Sci. 2014, 61, 5–16. [Google Scholar]
- Williams-Jones, A.E.; Vasyukova, O.V. The economic geology of scandium, the runt of the rare earth element litter. Econ. Geol. 2018, 113, 973–988. [Google Scholar] [CrossRef]
- Whalen, J.B.; Hildebrand, R.S. Trace element discrimination of arc, slab failure, and A-type granitic rocks. Lithos 2019, 348–349, 105179. [Google Scholar]
- Smith, M.; Kynicky, j.; Xu, C.; Song, W.; Spratt, J.; Jeffries, T.; Brtnicky, M.; Kopriva, A.; Cangelosi, D. The origin of secondary heavy rare earth element enrichment in carbonatites: Constraints from the evolution of the Huanglongpu district, China. Lithos 2018, 308–309. [Google Scholar] [CrossRef]
- Cangelosi, D.; Smith, M.; David Banks, D.; Yardley, B. The role of sulfate-rich fluids in heavy rare earth enrichment at the Dashigou carbonatite deposit, Huanglongpu, China. Mineral. Mag. 2020, 84, 65–80. [Google Scholar] [CrossRef]
- Simon, A.C.; Pettke, T.; Candela, P.A.; Piccoli, P.M.; Heinrich, CA. Gold partitioning in melt - vapour - brine systems. Geochim. Cosmochim. Acta 2005, 69, 3321–3335. [Google Scholar] [CrossRef]
- Halter, W.E.; Pettke, T.; Heinrich, C.A. The origin of Cu/Au ratios in porphyry-type ore deposits. Science, 296: 1844 -1846.Hill KC, Kendrick RD, Crowhurst PV, Gow PA Copper-gold mineralisation in New Guinea: tectonics, lineaments, thermochronology and structure. Aust. J. Earth Sci. 2002, 49, 737-752. [Google Scholar]
- Heinrich, C.A.; Halter, W.E.; Landtwing, M.R.; Pettke, T. The formation of economic porphyry copper (- gold) deposits: constraints from microanalysis of fluid and melt inclusions. Geol. Soc. Lond. Spec. Publ. 2005, 248, 247–263. [Google Scholar]
- Heinrich, C.A.; Günther, D.; Audétat, A.; Ulrich, T.; Frischknecht, R. Metal fractionation between magmatic brine and vapor, determined by microanalysis of fluid inclusions. Geology 1999, 27, 755–758. [Google Scholar] [CrossRef]
- Pokrovski, G.S.; Anastassia, Yu.; Borisova, A.Y.; Harrichoury, J-C. The effect of sulfur on vapour - liquid fractionation of metals in hydrothermal systems. Earth Planet. Sci. Lett. 2008, 266, 345–362. [Google Scholar] [CrossRef]
- Ulrich, T.; Günther, D.; Heinrich, C.A. The evolution of a porphyry Cu–Au deposit, based on LA-ICP-MS analysis of fluid inclusions: Bajo de la Alumbrera, Argentina. Econ. Geol. 2001, 96, 1743–1774. [Google Scholar]
- Sutherland Brown, A. Porphyry deposits of the Canadian Cordillera of North America; Canadian Institute of Mining, Metallurgy and Petroleum: Montreal, 1976; Volume 15, p. 510. [Google Scholar]
- Cox, D.P.; Singer, D.A. Distribution of gold in porphyry copper deposits. U.S. Geological Survey Open File Report 1988, 88–46. [Google Scholar]
- Sillitoe, R.H. Characteristics and controls of the largest porphyry copper–gold and epithermal gold deposits in the circum-Pacific region. Aust. J. Earth Sci. 1997, 44, 373–388. [Google Scholar]
- Murakami, H.; Seo, J.H.; Heinrich, C.A. The relation between Cu/Au ratio and formation depth of porphyry-style Cu–Au ± Mo deposits. Miner. Deposita 2010, 45, 11–21. [Google Scholar]
- Barth, M.; McDonough, W. F.; Rudnick, R. L. Tracking the budget of Nb and Ta in the continental crust. Chem. Geol. 2000, 165, 197–213. [Google Scholar] [CrossRef]
- Kamber, B.S.; Greig, A.; Collerson, K.D. A new estimate for the composition of weathered young upper continental crust from alluvial sediments, Queensland, Australia. Geochim. Cosmochim. Acta 2005, 69, 1041–1058. [Google Scholar] [CrossRef]
- Kamber, B.S.; Collerson, K.D. Role of ‘hidden’ deeply subducted slabs in mantle depletion. Chem. Geol. 2000, 166, 241–254. [Google Scholar] [CrossRef]
- Münker, C.; Pfänder, J.A.; Weyer, S.; Büchl, A.; Kleine, T.; Mezger, K. Evolution of planetary cores and the Earth–Moon system from Nb/Ta systematics. Science 2003, 301, 84–87. [Google Scholar] [CrossRef] [PubMed]
- Doucelance, R; Hammouda, T; Moreira, M; Martins, JC. Geo- chemical constraints on depth of origin of oceanic carbonatites: the Cape Verde case. Geochim. Cosmochim. Acta 2010, 74, 7261–7282. [Google Scholar]
- Hoernle, K.; Tilton, G.; LeBas, M.J.; Duggen, S.; Garbe-Schönberg, D. Geochemistry of oceanic carbonatites compared with continental carbonatites: mantle recycling of oceanic crustal carbonate. Contrib. Mineral. Petrol. 2002, 142, 520–542. [Google Scholar]
- Yang, Y.; Neubauer, F.; Liu, Z.; Wang, G-C.; Tan, S-C; He, X-H.; Liang, C-Y. Enriched lithospheric mantle storage of the Earth’s missing niobium: New insights from alkaline rocks with superchondritic Nb/Ta ratios. Geosys. Envir. 2024, 3, 100289. [Google Scholar]
- Wade, J.; Wood, B.J. The Earth’s ‘missing’ Nb may be in the core? Nature 2001, 409, 75–78. [Google Scholar] [CrossRef]
- Hoss, A.; Haase, K.M.; Keith, M.; Klemd, R.; Melfos, V.; Gerlach, L.; Pelloth, F.; Falkenberg, J.J.; Voudouris, P.; Strauss, H.; Baker, T; Tarantola, A. Magmatic and hydrothermal evolution of the Skouries Au-Cu porphyry deposit, northern Greece. Ore Geol. Rev. 2024, 173, 106233. [Google Scholar] [CrossRef]
- Perring, C.; Pollard, P.; Nunn, A. Petrogenesis of the Squirrel Hills granite and associated magnetite-rich sill and vein complex: Lightning Creek prospect, Cloncurry district, northwest Queensland. Precambrian Res. 2001, 106, 213–238. [Google Scholar] [CrossRef]
- Mark, G. Petrogenesis of Mesoproterozoic K-rich granitoids, southern Mt Angelay igneous complex, Cloncurry district, northwest Queensland. Aust. J. Earth Sci. 1999, 46, 933–949. [Google Scholar]
- Pollard, P.J.; Mark, G.; Mitchell, L.C. Geochemistry of Post-1540 Ma Granites in the, Cloncurry District, Northwest Queensland. Econ. Geol. 1998, 93, 1330–1344. [Google Scholar] [CrossRef]
- Slack, J.F. Strata-bound Fe-Co-Cu-Au-Bi-Y-REE deposits of the Idaho Cobalt Belt: Multistage hydrothermal mineralisation in a magmatic-related iron oxide copper-gold system. Econ.Geol. 2012, 107, 1089–1113. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




