Submitted:
14 January 2026
Posted:
15 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
- The importance of the pharmacokinetic data of established drugs/compounds,
- Investigational compound/drug versus proven drug(s) for cancer treatment,
- Route of drug administration, dose conversion, and frequency of drug administration in preclinical studies,
- The age, gender, strain of mice, and the importance of the ARRIVE guideline in preclinical studies,
- Subcutaneous versus metastatic mouse models in preclinical studies,
- Comorbidities and related drug studies in preclinical models,
- The acidic tumor microenvironment (TME) and the efficacy of the drug candidate.
2. The Importance of the Pharmacokinetic Data of Established Drugs/Compounds
3. Investigational Compound/Drug Versus Proven Drug(s) for Cancer Treatment
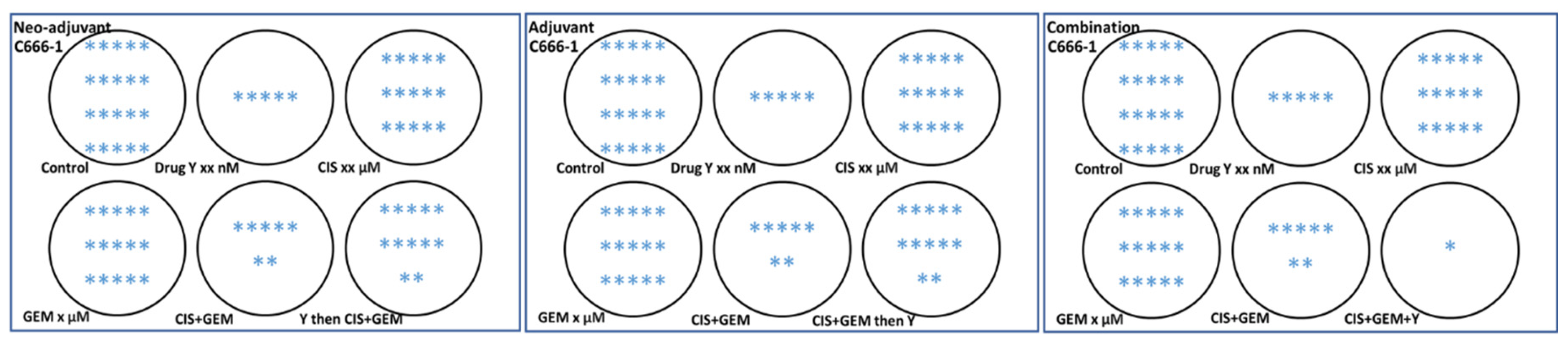
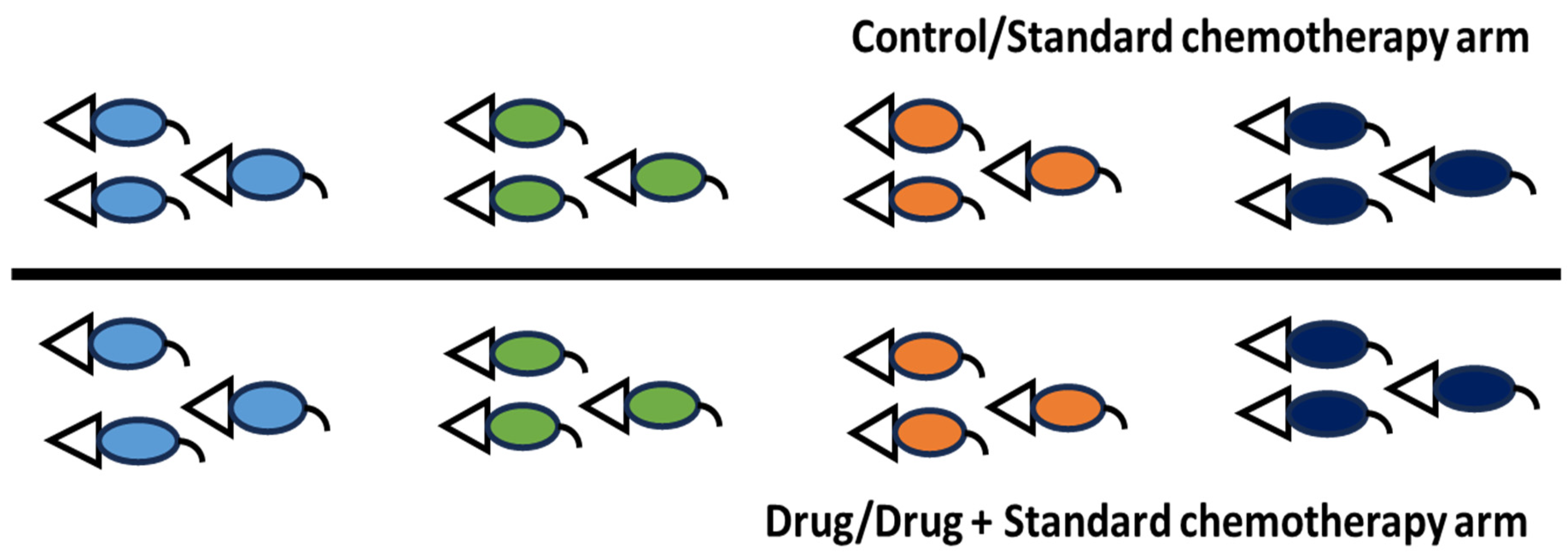
4. Route of Drug Administration, Dose Conversion, Frequency of Drug Administration and Study Design for Preclinical Research
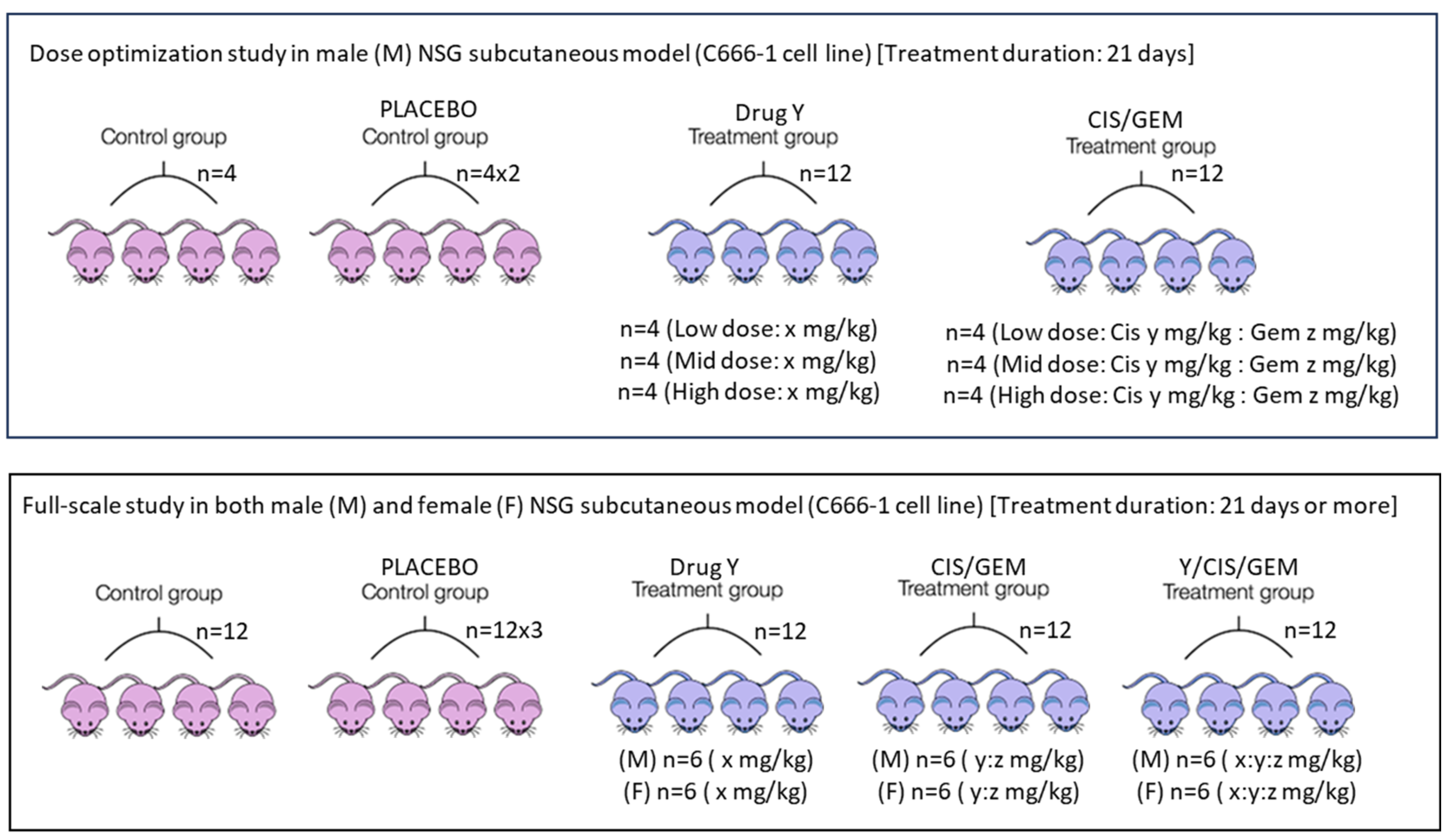
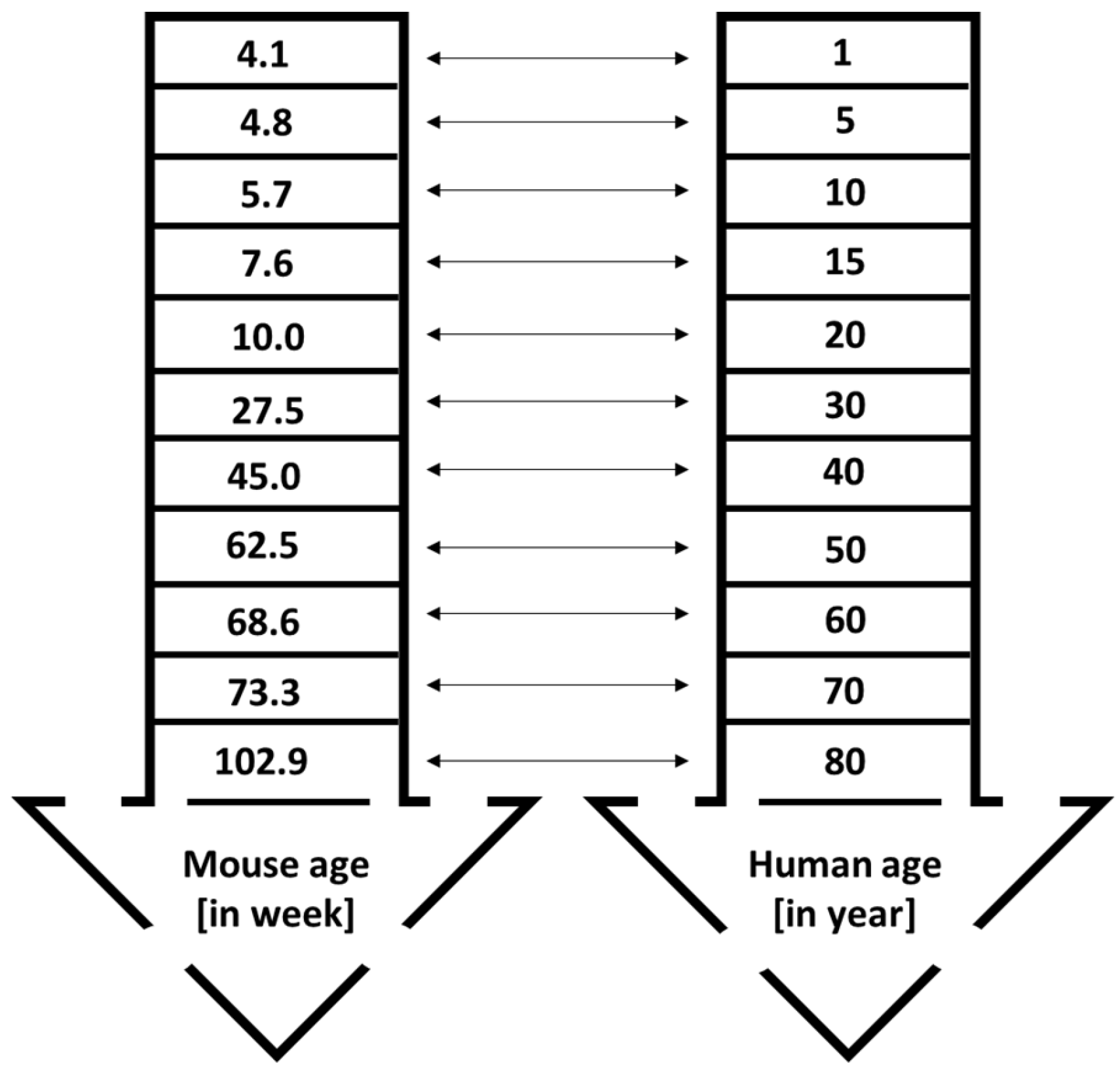
5. The Age, Gender, Strain of Mice, and the Importance of the ARRIVE Guideline in Preclinical Studies
6. Subcutaneous Versus Metastatic Mouse Models in Preclinical Studies
7. Comorbidities and Related Drug Studies in Preclinical Models
8. The Acidic Tumor Microenvironment (TME) and the Efficacy of the Drug Candidate
9. Discussion and Conclusions
10. Future Directions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Marshall, L.J.; Bailey, J.; Cassotta, M.; Herrmann, K.; Pistollato, F. Poor translatability of biomedical research using animals—A Narrative Review. Altern. Lab. Anim. 2023, 51, 102–135. [Google Scholar] [CrossRef]
- Seyhan, A.A. Lost in translation: The valley of death across preclinical and clinical divide -identification of problems and overcoming obstacles. Transl. Med. Comm. 2019, 4, 18. [Google Scholar] [CrossRef]
- Sun, D.; Gao, W.; Hu, H.; Zhou, S. Why 90% of clinical drug development fails and how to improve it? Acta Pharm. Sin. B 2022, 12, 3049–3062. [Google Scholar] [CrossRef] [PubMed]
- Mohi-Ud-Din, R.; Chawla, A.; Sharma, P.; Mir, P.A.; Potoo, F.H.; Reiner, Ž.; Reiner, I.; Ateşşahin, D.A.; Sharifi-Rad, J.; Mir, R.H.; et al. Repurposing approved non-oncology drugs for cancer therapy: A comprehensive review of mechanisms, efficacy, and clinical prospects. Eur. J. Med. Res. 2023, 28, 345. [Google Scholar] [CrossRef] [PubMed]
- Ramchandani, S.; Mohan, C.D.; Mistry, J.R.; Su, Q.; Naz, I.; Rangappa, K.S.; Ahn, K.S. The multifaceted antineoplastic role of pyrimethamine against human malignancies. IUBMB Life 2022, 74, 198–212. [Google Scholar] [CrossRef]
- Manoharan, S.; Ying, Y.L. Pyrimethamine reduced tumour growth in pre-clinical cancer models: A systematic review to identify potential pre-clinical studies for subsequent human clinical trials. Biol. Methods Protoc. 2024, 9, bpae021. [Google Scholar] [CrossRef]
- Jacobson, J.M.; Davidian, M.; Rainey, P.M.; Hafner, R.; Raasch, R.H.; Luft, B.J. Pyrimethamine pharmacokinetics in human immunodeficiency virus-positive patients seropositive for Toxoplasma gondii. Antimicrob. Agents Chemother. 1996, 40, 1360–1365. [Google Scholar] [CrossRef]
- Novus Biological, A Bio-Techne Brand. Molarity and Concentration Calculators. Available online: https://www.novusbio.com/resources/calculators (accessed on 24 June 2025).
- Zhou, X.; Zhang, J.; Hu, X.; He, P.; Guo, J.; Li, J.; Lan, T.; Liu, J.; Peng, L.; Li, H. Pyrimethamine elicits antitumor effects on prostate cancer by inhibiting the p38-NF-κB Pathway. Front. Pharmacol. 2020, 11, 758. [Google Scholar] [CrossRef]
- COLCRYS (Colchicine USP) Tablets for Oral Use Initial, U.S. Approval: 1961. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/022352s026lbl.pdf (accessed on 23 June 2025).
- Stamp, L.K.; Horsley, C.; Te Karu, L.; Dalbeth, N.; Barclay, M. Colchicine: The good, the bad, the ugly and how to minimize the risks. Rheumatology 2024, 63, 936–944. [Google Scholar] [CrossRef]
- Chang, L.S.; Oblinger, J.L.; Burns, S.S.; Huang, J.; Anderson, L.W.; Hollingshead, M.G.; Shen, R.; Pan, L.; Agarwal, G.; Ren, Y.; et al. Targeting protein translation by rocaglamide and didesmethylrocaglamide to treat mpnst and other sarcomas. Mol. Cancer Ther. 2020, 19, 731–741. [Google Scholar] [CrossRef]
- Iwasaki, S.; Floor, S.N.; Ingolia, N.T. Rocaglates convert DEAD-box protein eIF4A into a sequence-selective translational repressor. Nature 2016, 534, 558–561. [Google Scholar] [CrossRef] [PubMed]
- Markovic, M.; Ben-Shabat, S.; Dahan, A. Prodrugs for improved drug delivery: Lessons learned from recently developed and marketed products. Pharmaceutics 2020, 12, 1031. [Google Scholar] [CrossRef] [PubMed]
- Rautio, J.; Meanwell, N.A.; Di, L.; Hageman, M.J. The expanding role of prodrugs in contemporary drug design and development. Nat. Rev. Drug Discov. 2018, 17, 559–587. [Google Scholar] [CrossRef] [PubMed]
- Ge, C.; Huang, X.; Zhang, S.; Yuan, M.; Tan, Z.; Xu, C.; Jie, Q.; Zhang, J.; Zou, J.; Zhu, Y.; et al. In vitro co-culture systems of hepatic and intestinal cells for cellular pharmacokinetic and pharmacodynamic studies of capecitabine against colorectal cancer. Cancer Cell Int. 2023, 23, 14. [Google Scholar] [CrossRef]
- Reyes-Hernández, O.D.; Figueroa-González, G.; Quintas-Granados, L.I.; Gutiérrez-Ruíz, S.C.; Hernández-Parra, H.; Romero-Montero, A.; Del Prado-Audelo, M.L.; Bernal-Chavez, S.A.; Cortés, H.; Peña-Corona, S.I.; et al. 3,3′-Diindolylmethane and indole-3-carbinol: Potential therapeutic molecules for cancer chemoprevention and treatment via regulating cellular signaling pathways. Cancer Cell Int. 2023, 23, 180. [Google Scholar] [CrossRef]
- Bradlow, H.L.; Zeligs, M.A. Diindolylmethane (DIM) spontaneously forms from indole-3-carbinol (I3C) during cell culture experiments. In Vivo 2010, 24, 387–391. [Google Scholar]
- Aarón, R.H.; Sheila, C.M.; Julio Emmanuel, G.P.; Oscar, J.-G.; Aurelio, L.-M.; Ismael, M.-C.J. In Silico strategies for drug discovery: Optimizing natural compounds from foods for therapeutic applications. Discov. Chem. 2025, 2, 133. [Google Scholar] [CrossRef]
- Shaker, B.; Ahmad, S.; Lee, J.; Jung, C.; Na, D. In silico methods and tools for drug discovery. Comput. Biol. Med. 2021, 137, 104851. [Google Scholar] [CrossRef]
- Sabri, N.M.; Halim, S.N.A.; Zain, S.M.; Lee, V.S. Modification of Anti-cancer Co-crystal for Thymidylate Synthase Inhibition: Molecular Dynamics Study. Chiang Mai J. Sci. 2018, 45, 2361–2373. [Google Scholar]
- Kawasaki, Y.; Freire, E. Finding a better path to drug selectivity. Drug Discov. Today 2011, 16, 985–990. [Google Scholar] [CrossRef]
- Sadybekov, A.V.; Katritch, V. Computational approaches streamlining drug discovery. Nature 2023, 616, 673–685. [Google Scholar] [CrossRef]
- NHS-South West Strategic Clinical Network. Docetaxel and Prednisolone (Prostate). 2016. Available online: https://www.swagcanceralliance.nhs.uk/wp-content/uploads/2020/09/DocetaxelProstatev2.pdf (accessed on 5 June 2025).
- Park, H.S.; Kim, B.; Oh, J.H.; Kim, Y.C.; Lee, Y.J. First-pass metabolism of decursin, a bioactive compound of Angelica gigas, in rats. Planta Med. 2012, 78, 909–913. [Google Scholar] [CrossRef]
- Muralikrishnan, A.; Sekar, M.; Kumarasamy, V.; Gan, S.H.; Ravi, S.; Subramaniyan, V.; Wong, L.S.; Wong, L.S.; Wu, Y.S.; Khattulanuar, F.S.; Rani, N.N.I.M. Chemistry, Pharmacology and Therapeutic Potential of Decursin: A Promising Natural Lead for New Drug Discovery and Development. Drug Des. Dev. Ther. 2024, 18, 3741–3763. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Li, L.; Hale, T.W.; Chee, W.; Xing, C.; Jiang, C.; Lü, J. Single oral dose pharmacokinetics of decursin and decursinol angelate in healthy adult men and women. PLoS ONE 2015, 10, e0114992. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Tang, S.N.; Zhang, Y.; Puppala, M.; Cooper, T.K.; Xing, C.; Jiang, C.; Lü, J. Prostate Cancer Xenograft Inhibitory Activity and Pharmacokinetics of Decursinol, a Metabolite of Angelica gigas Pyranocoumarins, in Mouse Models. Am. J. Chin. Med. 2017, 45, 1773–1792. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). Clinical Care of Toxoplasmosis. 2024. Available online: https://www.cdc.gov/toxoplasmosis/hcp/clinical-care/index.html#:~:text=Pyrimethamine%20is%20a%20folic%20acid,the%20toxic%20effects%20of%20pyrimethamine (accessed on 25 June 2025).
- Ianevski, A.; Giri, A.K.; Aittokallio, T. SynergyFinder 2.0: Visual analytics of multi-drug combination synergies. Nucleic Acids Res. 2020, 48, W488–W493. [Google Scholar] [CrossRef]
- Tanahashi, M.; Niwa, H.; Yukiue, H.; Suzuki, E.; Haneda, H.; Yoshii, N. Adjuvant chemotherapy based on the in vitro histoculture drug response assay for non-small cell lung cancer improves survival. J. Thorac. Oncol. 2010, 5, 1376–1381. [Google Scholar] [CrossRef]
- Lau, G.I.; Loo, W.T.; Chow, L.W. Neoadjuvant chemotherapy for breast cancer determined by chemosensitivity assay achieves better tumor response. Biomed. Pharmacother. 2007, 61, 562–565. [Google Scholar] [CrossRef]
- Singer, C.F.; Klinglmüller, F.; Stratmann, R.; Staudigl, C.; Fink-Retter, A.; Gschwantler, D.; Helmy, S.; Pfeiler, G.; Dressler, A.C.; Sartori, C.; et al. Response prediction to neoadjuvant chemotherapy: Comparison between pre-therapeutic gene expression profiles and in vitro chemosensitivity assay. PLoS ONE 2013, 8, e66573. [Google Scholar] [CrossRef]
- Al-Zeheimi, N.; Adham, S.A. Modeling neoadjuvant chemotherapy resistance in vitro increased NRP-1 and HER2 expression and converted MCF7 breast cancer subtype. Br. J. Pharmacol. 2020, 177, 2024–2041. [Google Scholar] [CrossRef]
- Al Shoyaib, A.; Archie, S.R.; Karamyan, V.T. Intraperitoneal route of drug administration: Should it be used in experimental animal studies? Pharm. Res. 2019, 37, 12. [Google Scholar] [CrossRef] [PubMed]
- Ohshima, M.; Taguchi, A.; Tsuda, H.; Sato, Y.; Yamahara, K.; Harada-Shiba, M.; Miyazato, M.; Ikeda, T.; Iida, H.; Tsuji, M. Intraperitoneal and intravenous deliveries are not comparable in terms of drug efficacy and cell distribution in neonatal mice with hypoxia-ischemia. Brain Dev. 2015, 37, 376–386. [Google Scholar] [CrossRef] [PubMed]
- Perše, M. Cisplatin mouse models: Treatment, toxicity and translatability. Biomedicines 2021, 9, 1406. [Google Scholar] [CrossRef] [PubMed]
- Perše, M.; Večerić-Haler, Ž. Cisplatin-induced rodent model of kidney injury: Characteristics and challenges. Biomed. Res. Int. 2018, 2018, 1462802. [Google Scholar] [CrossRef]
- Dou, S.; Smith, M.; Wang, Y.; Rusckowski, M.; Liu, G. Intraperitoneal injection is not always a suitable alternative to intravenous injection for radiotherapy. Cancer Biother. Radiopharm. 2013, 28, 335–342. [Google Scholar] [CrossRef]
- Cai, Z.Y.; Galettis, P.; Lu, Y.; Morris, D.L.; Pourgholami, M.H. Pharmacokinetics of albendazole in New Zealand white rabbits: Oral versus intraperitoneal administration. Anticancer Res. 2007, 27, 417–422. [Google Scholar]
- Weng, J.H.; Koch, P.D.; Luan, H.H.; Tu, H.-C.; Shimada, K.; Ngan, I.; Ventura, R.; Jiang, R.; Mitchison, T.J. Colchicine acts selectively in the liver to induce hepatokines that inhibit myeloid cell activation. Nat. Metab. 2021, 3, 513–522. [Google Scholar] [CrossRef]
- NHS-Somerset; Wiltshire; Avon and Gloucestershire Cancer Alliance. Cisplatin and Gemcitabine (Nasopharynx). 2022. Available online: https://www.swagcanceralliance.nhs.uk/wp-content/uploads/2023/02/Cis-Gem-nasopharynx-v1.pdf (accessed on 26 June 2025).
- Thompson, B.R.; Shi, J.; Zhu, H.J.; Smith, D.E. Pharmacokinetics of gemcitabine and its amino acid ester prodrug following intravenous and oral administrations in mice. Biochem Pharmacol. 2020, 180, 114127. [Google Scholar] [CrossRef]
- Arita, M.; Watanabe, S.; Aoki, N.; Kuwahara, S.; Suzuki, R.; Goto, S.; Abe, Y.; Takahashi, M.; Sato, M.; Hokari, S.; et al. Combination therapy of cisplatin with cilastatin enables an increased dose of cisplatin, enhancing its antitumor effect by suppression of nephrotoxicity. Sci. Rep. 2021, 11, 750. [Google Scholar] [CrossRef]
- Nakano, K.; Ike, O.; Wada, H.; Hitomi, S.; Amano, Y.; Ogita, I.; Nakai, N.; Takada, K. Oral sustained-release cisplatin preparation for rats and mice. J. Pharm. Pharmacol. 1997, 49, 485–490. [Google Scholar] [CrossRef]
- White Paper: The Ultimate Guide to Mouse Clinical Trials. Crown Bioscience. Available online: https://www.crownbio.com/wp/mouse-clinical-trial-with-pdx-guide?utm_campaign=Integrated%20Solutions%20-%20MCT&utm_source=Scientist.com&utm_medium=blog&utm_content=Oct%202024 (accessed on 27 August 2025).
- Alrashed, S.H.; Alkatan, H.M.; Alarfaj, M.; Alnahdi, M.A.; Almeshari, N.Z.; Almutairi, F.J.; Maktabi, A.M.Y. Nasopharyngeal carcinoma (NPC) in the ophthalmic practice: A serious neoplasm presenting initially to ophthalmologists. Saudi J. Ophthalmol. 2022, 35, 225–229. [Google Scholar] [CrossRef] [PubMed]
- Harvard Chan Bioinformatics Core. Experimental Design Considerations. Available online: https://hbctraining.github.io/Intro-to-rnaseq-fasrc-salmon-flipped/lessons/02_experimental_planning_considerations.html (accessed on 30 June 2025).
- Manoharan, S. Mice age in cancer research: Overlooked implications for clinical translation. Int. Med. Res. J. 2024, 10, 67–71. [Google Scholar]
- Laconi, E.; Marongiu, F.; DeGregori, J. Cancer as a disease of old age: Changing mutational and microenvironmental landscapes. Br. J. Cancer 2020, 122, 943–952. [Google Scholar] [CrossRef] [PubMed]
- Downing, J.R.; Allison, J.P.; Honjo, T. NIH to require both sexes in preclinical studies. Cancer Discov. 2014, 4, 860. [Google Scholar] [CrossRef]
- Wang, S.; Lai, X.; Deng, Y.; Song, Y. Correlation between mouse age and human age in anti-tumor research: Significance and method establishment. Life Sci. 2020, 242, 117242. [Google Scholar] [CrossRef]
- Yanai, S.; Endo, S. Functional aging in male C57BL/6J mice across the life-span: A systematic behavioral analysis of motor, emotional, and memory function to define an aging phenotype. Front. Aging Neurosci. 2021, 13, 697621. [Google Scholar] [CrossRef]
- The Jackson Laboratory. Aged C57BL/6J Mice. Available online: https://www.jax.org/jax-mice-and-services/find-mice/most-popular-jax-mice-strains/aged-b6 (accessed on 3 July 2025).
- Charles River Laboratories. C57BL/6 Mice. Available online: https://www.criver.com/products-services/find-model/c57bl6-mouse?region=3616 (accessed on 29 July 2024).
- The Jackson Laboratory. Immunodeficient & Nude Mouse Models. Available online: https://www.jax.org/jax-mice-and-services/find-mice/most-popular-jax-mice-strains/immunodeficient-mouse-and-xenograft-host-comparisons (accessed on 29 July 2024).
- Hirenallur-Shanthappa, D.K.; Ramírez, J.A.; Iritani, B.M. Chapter 5—Immunodeficient mice: The backbone of patient-derived tumor xenograft models. In Patient Derived Tumor Xenograft Models; Uthamanthil, R., Tinkey, P., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 57–73. [Google Scholar]
- Muqbil, I.; Philip, P.A.; Mohammad, R.M. Chapter 9—A Guide to Tumor Assessment Methodologies in Cancer Drug Discovery. In Animal Models in Cancer Drug Discovery; Elsevier: Amsterdam, The Netherlands, 2019; pp. 233–248. [Google Scholar]
- Chen, J.; Liao, S.; Xiao, Z.; Pan, Q.; Wang, X.; Shen, K.; Wang, S.; Yang, L.; Guo, F.; Liu, H.-F.; et al. The development and improvement of immunodeficient mice and humanized immune system mouse models. Front. Immunol. 2022, 13, 1007579. [Google Scholar] [CrossRef]
- Begley, C.G.; Ellis, L.M. Drug development: Raise standards for preclinical cancer research. Nature 2012, 483, 531–533. [Google Scholar] [CrossRef]
- Percie du Sert, N.; Hurst, V.; Ahluwalia, A.; Alam, S.; Avey, M.T.; Baker, M.; Browne, W.J.; Clark, A.; Cuthill, I.C.; Dirnagl, U.; et al. The ARRIVE guidelines 2.0: Updated guidelines for reporting animal research. PLoS Biol. 2020, 18, e3000410. [Google Scholar]
- Fares, J.; Fares, M.Y.; Khachfe, H.H.; Salhab, H.A.; Fares, Y. Molecular principles of metastasis: A hallmark of cancer revisited. Signal Transduct. Target Ther. 2020, 5, 28. [Google Scholar] [CrossRef]
- Stribbling, S.M.; Beach, C.; Ryan, A.J. Orthotopic and metastatic tumour models in preclinical cancer research. Pharmacol. Ther. 2024, 257, 108631. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.A.; Merritt, D.; Barr, L.; Thorley-Lawson, D.A. An orthotopic model of metastatic nasopharyngeal carcinoma and its application in elucidating a therapeutic target that inhibits metastasis. Genes Cancer 2011, 2, 1023–1033. [Google Scholar] [CrossRef] [PubMed]
- Oncology. Orthotopic vs. Subcutaneous: Unpacking the Strengths and Limitations of Tumor Model Implantation Sites. 2023. Available online: https://blog.td2inc.com/orthotopic-vs.-subcutaneous (accessed on 5 July 2025).
- Fernandez, J.L.; Årbogen, S.; Sadeghinia, M.J.; Haram, M.; Snipstad, S.; Torp, S.H.; Einen, C.; Mühlenpfordt, M.; Maardalen, M.; Vikedal, K.; et al. A comparative analysis of orthotopic and subcutaneous pancreatic tumour models: Tumour microenvironment and drug delivery. Cancers 2023, 15, 5415. [Google Scholar] [CrossRef] [PubMed]
- Mizuta, K.; Gallagher, S.; Wang, A.; Chang, N.; Morinaga, S.; Sato, M.; Kang, B.M.; Hoffman, R.M. Head-to-head comparison of green fluorescent protein (GFP) imaging with luciferase-luciferin imaging in vivo using single-nanometer laser-excitation tuning and an ultra-low-light-detection camera and optics demonstrates the superiority of GFP. Anticancer Res. 2024, 44, 2823–2826. [Google Scholar] [CrossRef]
- Zeng, Q.; Liu, Y.M.; Liu, J.; Han, J.; Guo, J.-X.; Lu, S.; Huang, X.-M.; Yi, P.; Lang, J.-Y.; Zhang, P.; et al. Inhibition of ZIP4 reverses epithelial-to-mesenchymal transition and enhances the radiosensitivity in human nasopharyngeal carcinoma cells. Cell Death Dis. 2019, 10, 588. [Google Scholar] [CrossRef]
- Sato, S.; Bartolák-Suki, E.; Parameswaran, H.; Hamakawa, H.; Suki, B. Scale dependence of structure-function relationship in the emphysematous mouse lung. Front. Physiol. 2015, 6, 146. [Google Scholar] [CrossRef]
- O’Brien, M.; Ernst, M.; Poh, A.R. An intrasplenic injection model of pancreatic cancer metastasis to the liver in mice. STAR Protoc. 2023, 4, 102021. [Google Scholar] [CrossRef]
- Matsuda, Y.; Yoshimura, H.; Ueda, J.; Naito, Z.; Korc, M.; Ishiwata, T. Nestin delineates pancreatic cancer stem cells in metastatic foci of NOD/Shi-scid IL2Rγ(null) (NOG) mice. Am. J. Pathol. 2014, 184, 674–685. [Google Scholar] [CrossRef]
- Cheung, S.T.; Chan, S.L.; Lo, K.W. Contaminated and misidentified cell lines commonly use in cancer research. Mol. Carcinog. 2020, 59, 573–574. [Google Scholar] [CrossRef]
- Makowska, A.; Weiskirchen, R. Nasopharyngeal carcinoma cell lines: Reliable alternatives to primary nasopharyngeal cells? Cells 2024, 13, 559. [Google Scholar] [CrossRef]
- Panigrahi, G.; Ambs, S. How comorbidities shape cancer biology and survival. Trends Cancer 2021, 7, 488–495. [Google Scholar] [CrossRef]
- Sebastian, D.; Joseph, J.; Mathews, E. The prevalence and correlates of comorbidities among patients with cancer attending a tertiary care cancer center in South India: An analytical cross-sectional study. Cancer Res. Stat. Treat 2023, 6, 526–533. [Google Scholar] [CrossRef]
- Fowler, H.; Belot, A.; Ellis, L.; Maringe, C.; Luque-Fernandez, M.A.; Njagi, E.N.; Navani, N.; Sarfati, D.; Rachet, B. Comorbidity prevalence among cancer patients: A population-based cohort study of four cancers. BMC Cancer 2020, 20, 2. [Google Scholar] [CrossRef]
- Mao, X.; Wu, S.; Huang, D.; Li, C. Complications and comorbidities associated with antineoplastic chemotherapy: Rethinking drug design and delivery for anticancer therapy. Acta Pharm. Sin. B 2024, 14, 2901–2926. [Google Scholar] [CrossRef]
- Riechelmann, R.P.; Krzyzanowska, M.K. Drug interactions and oncological outcomes: A hidden adversary. Ecancermedicalscience 2019, 13, ed88. [Google Scholar] [CrossRef]
- Zayed, A.L.; Hroot, J.A.; Darweesh, R.S.; Qinna, N.A.; Alshekhhossin, A.M.; Al Kilani, O.M. Resveratrol alters erlotinib pharmacokinetics in rats: Application of a rapid HPLC-fluorescence method for quantifying erlotinib in plasma. Naunyn Schmiedebergs Arch. Pharmacol. 2025, in press. [Google Scholar] [CrossRef]
- Adamiszak, A.; Drobińska, J.; Niewiadomska-Wojnałowicz, I.; Derwich, K.; Grześkowiak, E.; Bienert, A. Potential drug-drug interactions analysis in Polish pediatric hemato-oncologic unit, including acute lymphoblastic leukemia patients. Pharmacol. Rep. 2025, 77, 751–760. [Google Scholar] [CrossRef] [PubMed]
- Talbot, S.R.; Heider, M.; Wirth, M.; Jörns, A.; Naujok, O. Exploring dose-response variability and relative severity assessment in STZ-induced diabetes male NSG mice. Sci. Rep. 2024, 14, 16559. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Hapach, L.A.; Griggs, L.; Smart, K.; Wu, Y.; Taufalele, P.V.; Rowe, M.M.; Young, K.M.; Bates, M.E.; Johnson, A.C.; et al. Diabetic hyperglycemia promotes primary tumor progression through glycation-induced tumor extracellular matrix stiffening. Sci. Adv. 2022, 8, eabo1673. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.; Dong, Z.; Tao, D.; Zhang, Y.; Liu, Z. The acidic tumor microenvironment: A target for smart cancer nano-theranostics. Natl. Sci. Rev. 2018, 5, 269–286. [Google Scholar] [CrossRef]
- Xu, J.; Li, Y.; Li, Z.; Shao, W.; Song, J.; Wei, J. Acidic tumor microenvironment promotes pancreatic cancer through miR-451a/MEF2D axis. J. Oncol. 2022, 2022, 3966386. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.A.; Yadab, M.K.; Ali, M.M. Emerging role of extracellular pH in tumor microenvironment as a therapeutic target for cancer immunotherapy. Cells 2024, 13, 1924. [Google Scholar] [CrossRef] [PubMed]
- Kato, Y.; Ozawa, S.; Miyamoto, C.; Maehata, Y.; Suzuki, A.; Maeda, T.; Baba, Y. Acidic extracellular microenvironment and cancer. Cancer Cell Int. 2013, 13, 89. [Google Scholar] [CrossRef] [PubMed]
- Worsley, C.M.; Veale, R.B.; Mayne, E.S. The acidic tumour microenvironment: Manipulating the immune response to elicit escape. Hum. Immunol. 2022, 83, 399–408. [Google Scholar] [CrossRef]
- Ward, C.; Meehan, J.; Gray, M.E.; Murray, A.F.; Argyle, D.J.; Kunkler, I.H.; Langdon, S.P. The impact of tumour pH on cancer progression: Strategies for clinical intervention. Explor. Target Antitumor. Ther. 2020, 1, 71–100. [Google Scholar] [CrossRef]
- Carvalho, T.M.A.; Di Molfetta, D.; Greco, M.R.; Koltai, T.; Alfarouk, K.O.; Reshkin, S.J.; Cardone, R.A. Tumor microenvironment features and chemoresistance in pancreatic ductal adenocarcinoma: Insights into targeting physicochemical barriers and metabolism as therapeutic approaches. Cancers 2021, 13, 6135. [Google Scholar] [CrossRef]
- Liang, C.; Shi, S.; Meng, Q.; Liang, D.; Ji, S.; Zhang, B.; Qin, Y.; Xu, J.; Ni, Q.; Yu, X. Complex roles of the stroma in the intrinsic resistance to gemcitabine in pancreatic cancer: Where we are and where we are going. Exp. Mol. Med. 2017, 49, e406. [Google Scholar] [CrossRef]
- Jin, M.Z.; Jin, W.L. The updated landscape of tumor microenvironment and drug repurposing. Sig. Transduct. Target Ther. 2020, 5, 166. [Google Scholar] [CrossRef]
- Estrella, V.; Chen, T.; Lloyd, M.; Wojtkowiak, J.; Cornnell, H.H.; Ibrahim-Hashim, A.; Bailey, K.; Balagurunathan, Y.; Rothberg, J.M.; Sloane, B.F.; et al. Acidity generated by the tumor microenvironment drives local invasion. Cancer Res. 2013, 73, 1524–1535. [Google Scholar] [CrossRef]
- Loftsson, T.; Moya-Ortega, M.D.; Alvarez-Lorenzo, C.; Concheiro, A. Pharmacokinetics of cyclodextrins and drugs after oral and parenteral administration of drug/cyclodextrin complexes. J. Pharm. Pharmacol. 2016, 68, 544–555. [Google Scholar] [CrossRef]
- Manoharan, S.; Ying, L.Y. Efficacy and safety of immune checkpoint inhibitors for locoregionally advanced, recurrent and metastatic nasopharyngeal carcinoma: A systematic review of phase III randomised controlled trials. Naunyn Schmiedebergs Arch. Pharmacol. 2025. [Google Scholar] [CrossRef]
- Lensen, S. When to pool data in a meta-analysis (and when not to)? Fertil. Steril. 2023, 119, 902–903. [Google Scholar] [CrossRef]
- Manoharan, S.; Ying, L.Y. Does baricitinib reduce mortality and disease progression in SARS-CoV-2 virus infected patients? A systematic review and meta-analysis. Respir. Med. 2022, 202, 106986. [Google Scholar] [CrossRef]
- Carlson, R.B.; Martin, J.R.; Beckett, R.D. Ten simple rules for interpreting and evaluating a meta-analysis. PLoS Comput. Biol. 2023, 19, e1011461. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



