Submitted:
12 January 2026
Posted:
13 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Clinical Data and Adverse Event Assessment
2.3. DNA Extraction and IL7 rs16906115 Genotyping
2.4. Statistical Analysis
2.5. Survival Analysis
2.6. Ethical Considerations
3. Results
3.1. Patient Characteristics
3.2. IL7 rs16906115 Genotype Distribution and Association with Adverse Events
3.3. Genetic Models and Multivariable Analysis
3.4. Association with Clinical Subgroups and Outcomes
| Clinical Feature | Non-Carriers (GG) | Risk Carriers (AG/AA) | P-value |
|---|---|---|---|
| Toxicity Severity, n (%) | n=106 | n=18 | |
| Low Grade (1-2) | 89 (84.0%) | 16 (88.9%) | 0.86 |
| High Grade (3-5) | 17 (16.0%) | 2 (11.1%) | |
| Survival Outcomes, median (months) | n=96 | n=19 | |
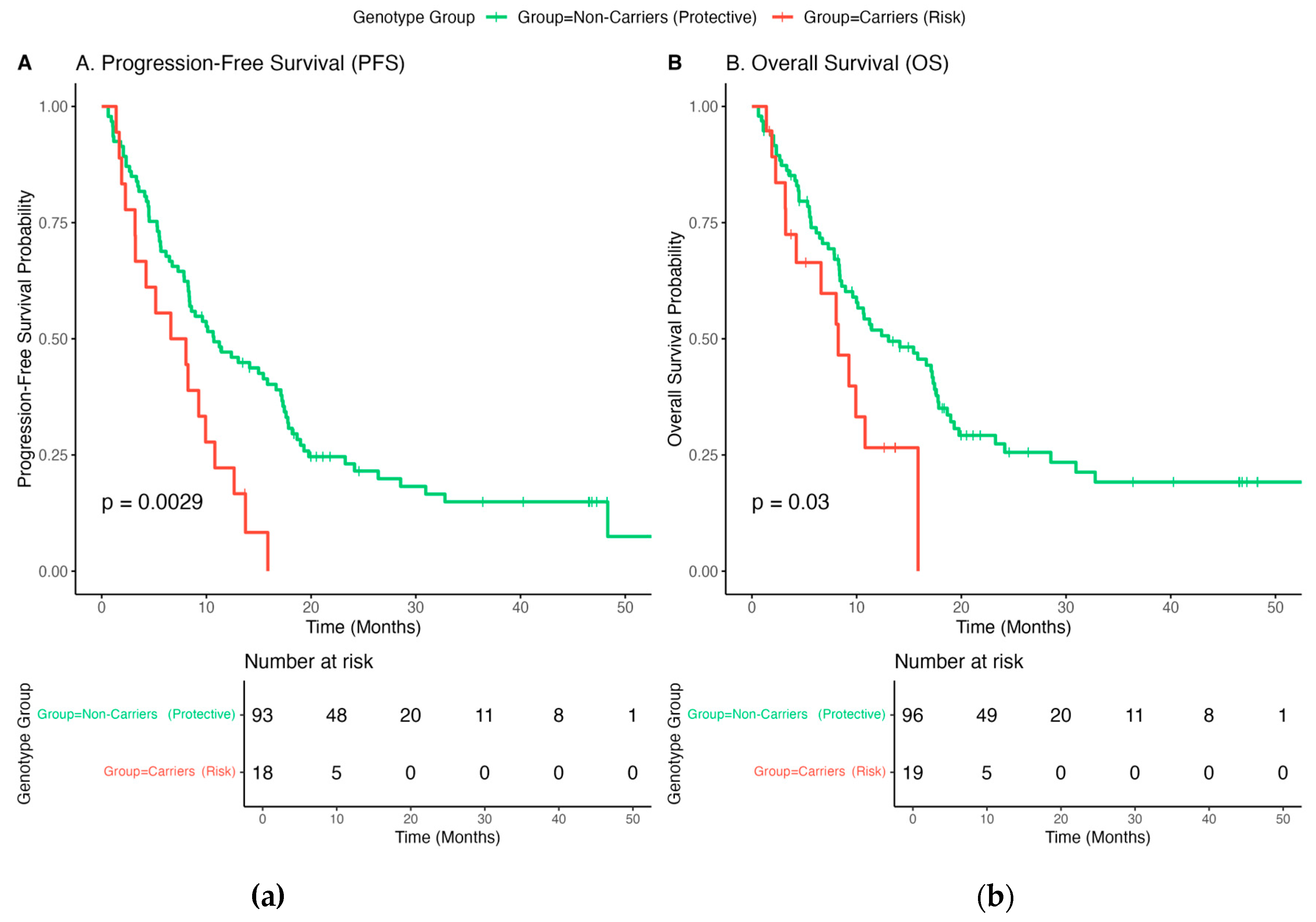
| Progression-Free Survival (PFS) | 10.0 (8.2 –11.9) | 6.6 (4.4 – 8.8) | 0.0029 |
| Overall Survival (OS) | 13.0 (9.6–17.6) | 8.3 (4.2–NR) | 0.03 |
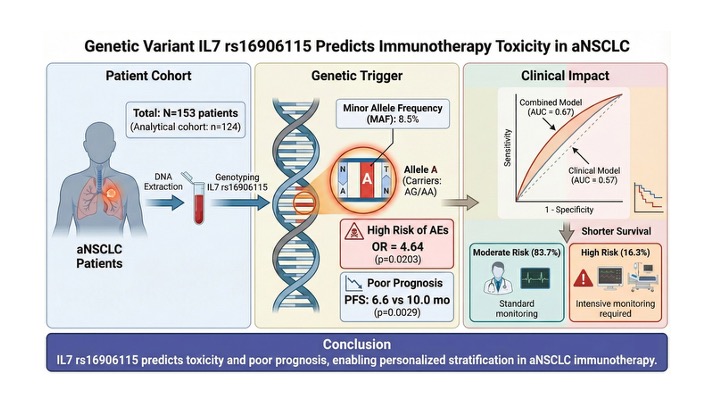
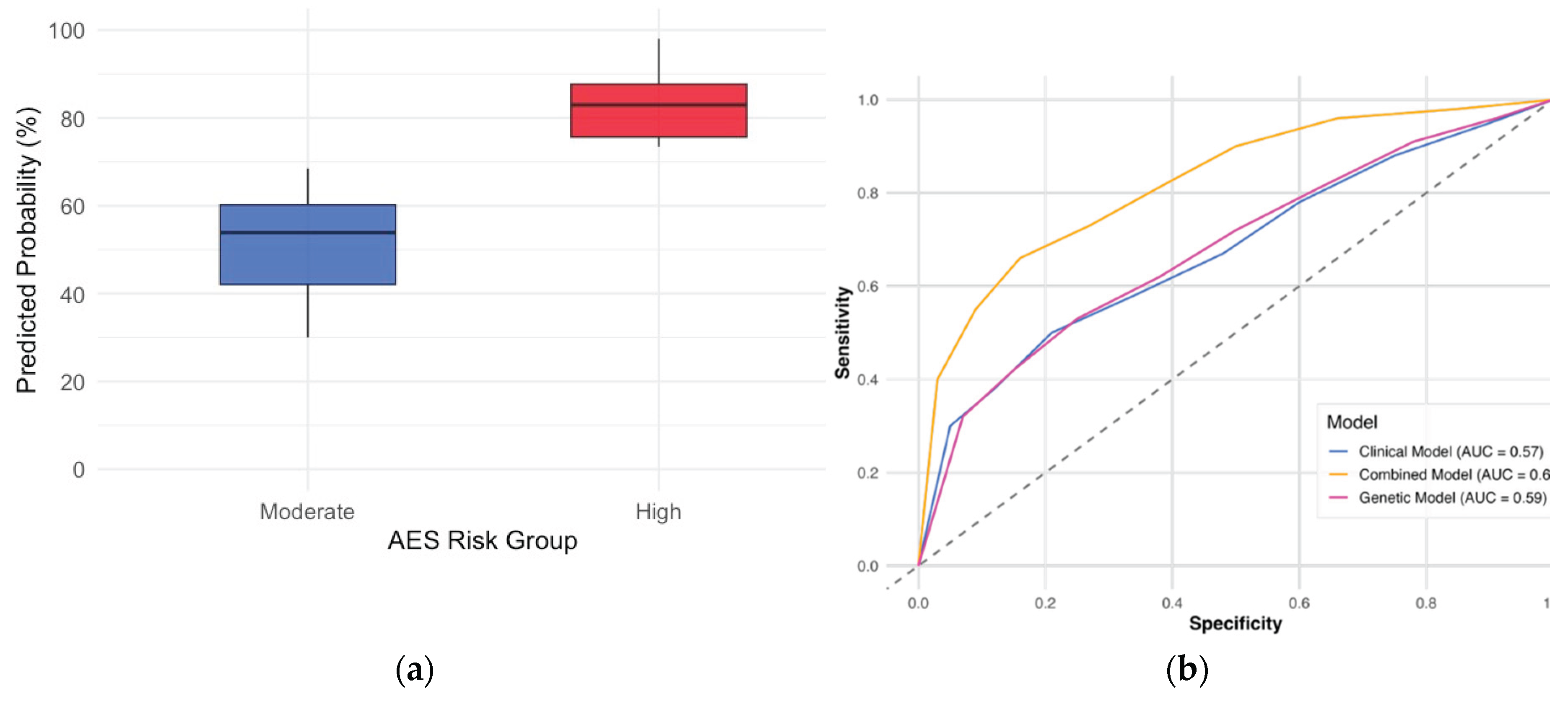
3.5. Predictive Scoring Model for AE Risk
3.6. Association of IL7 Genotype with Survival Outcomes
4. Discussion
4.1. Biological Plausibility and Mechanistic Insight
4.2. Comparison with Prior Studies and Population Specificity
4.3. Clinical Utility and Predictive Performance
4.4. Limitations and Future Directions
5. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Abbreviation | Full Term |
| AEs | Adverse Events |
| aNSCLC | Advanced Non-Small Cell Lung Cancer |
| AUC | Area Under the Curve |
| CI | Confidence Interval |
| CTCAE | Common Terminology Criteria for Adverse Events |
| ECOG | Eastern Cooperative Oncology Group |
| eQTL | Expression Quantitative Trait Locus |
| HR | Hazard Ratio |
| ICIs | Immune Checkpoint Inhibitors |
| IL-7 | Interleukin-7 |
| irAEs | Immune-related Adverse Events |
| MAF | Minor Allele Frequency |
| NSCLC | Non-Small Cell Lung Cancer |
| OR | Odds Ratio |
| OS | Overall Survival |
| PD-1 | Programmed Cell Death Protein 1 |
| PD-L1 | Programmed Death-Ligand 1 |
References
- Brahmer, J.; Reckamp, K.L.; Baas, P.; Crinò, L.; Eberhardt, W.E.E.; Poddubskaya, E.; Antonia, S.; Pluzanski, A.; Vokes, E.E.; Holgado, E.; et al. Nivolumab versus Docetaxel in Advanced Squamous-Cell Non–Small-Cell Lung Cancer. New England Journal of Medicine 2015, 373, 123–135. [CrossRef]
- Topalian, S.L.; Hodi, F.S.; Brahmer, J.R.; Gettinger, S.N.; Smith, D.C.; McDermott, D.F.; Powderly, J.D.; Carvajal, R.D.; Sosman, J.A.; Atkins, M.B.; et al. Safety, Activity, and Immune Correlates of Anti–PD-1 Antibody in Cancer. New England Journal of Medicine 2012, 366, 2443–2454. [CrossRef]
- Jayathilaka, B.; Mian, F.; Franchini, F.; Au-Yeung, G.; IJzerman, M. Cancer and Treatment Specific Incidence Rates of Immune-Related Adverse Events Induced by Immune Checkpoint Inhibitors: A Systematic Review. Br J Cancer 2025, 132, 51–57. [CrossRef]
- Ricciuti, B.; Genova, C.; De Giglio, A.; Bassanelli, M.; Dal Bello, M.G.; Metro, G.; Brambilla, M.; Baglivo, S.; Grossi, F.; Chiari, R. Impact of Immune-Related Adverse Events on Survival in Patients with Advanced Non-Small Cell Lung Cancer Treated with Nivolumab: Long-Term Outcomes from a Multi-Institutional Analysis. J Cancer Res Clin Oncol 2019, 145, 479–485. [CrossRef]
- Hellmann, M.D.; Ciuleanu, T.-E.; Pluzanski, A.; Lee, J.S.; Otterson, G.A.; Audigier-Valette, C.; Minenza, E.; Linardou, H.; Burgers, S.; Salman, P.; et al. Nivolumab plus Ipilimumab in Lung Cancer with a High Tumor Mutational Burden. N Engl J Med 2018, 378, 2093–2104. [CrossRef]
- Queirolo, P.; Dozin, B.; Morabito, A.; Banelli, B.; Piccioli, P.; Fava, C.; Leo, C.; Carosio, R.; Laurent, S.; Fontana, V.; et al. Association of CTLA-4 Gene Variants with Response to Therapy and Long-Term Survival in Metastatic Melanoma Patients Treated with Ipilimumab: An Italian Melanoma Intergroup Study. Front Immunol 2017, 8, 386. [CrossRef]
- Khan, Z.; Hammer, C.; Carroll, J.; Di Nucci, F.; Acosta, S.L.; Maiya, V.; Bhangale, T.; Hunkapiller, J.; Mellman, I.; Albert, M.L.; et al. Genetic Variation Associated with Thyroid Autoimmunity Shapes the Systemic Immune Response to PD-1 Checkpoint Blockade. Nat Commun 2021, 12, 3355. [CrossRef]
- Shahabi, V.; Berman, D.; Chasalow, S.D.; Wang, L.; Tsuchihashi, Z.; Hu, B.; Panting, L.; Jure-Kunkel, M.; Ji, R.-R. Gene Expression Profiling of Whole Blood in Ipilimumab-Treated Patients for Identification of Potential Biomarkers of Immune-Related Gastrointestinal Adverse Events. J Transl Med 2013, 11, 75. [CrossRef]
- Fry, T.J.; Mackall, C.L. Interleukin-7: From Bench to Clinic. Blood 2002, 99, 3892–3904. [CrossRef]
- Todd, J.A.; Walker, N.M.; Cooper, J.D.; Smyth, D.J.; Downes, K.; Plagnol, V.; Bailey, R.; Nejentsev, S.; Field, S.F.; Payne, F.; et al. Robust Associations of Four New Chromosome Regions from Genome-Wide Analyses of Type 1 Diabetes. Nat Genet 2007, 39, 857–864. [CrossRef]
- International Multiple Sclerosis Genetics Consortium; Hafler, D.A.; Compston, A.; Sawcer, S.; Lander, E.S.; Daly, M.J.; De Jager, P.L.; de Bakker, P.I.W.; Gabriel, S.B.; Mirel, D.B.; et al. Risk Alleles for Multiple Sclerosis Identified by a Genomewide Study. N Engl J Med 2007, 357, 851–862. [CrossRef]
- Mazzucchelli, R.; Durum, S.K. Interleukin-7 Receptor Expression: Intelligent Design. Nat Rev Immunol 2007, 7, 144–154. [CrossRef]
- Lundström, W.; Highfill, S.; Walsh, S.T.R.; Beq, S.; Morse, E.; Kockum, I.; Alfredsson, L.; Olsson, T.; Hillert, J.; Mackall, C.L. Soluble IL7Rα Potentiates IL-7 Bioactivity and Promotes Autoimmunity. Proc Natl Acad Sci U S A 2013, 110, E1761-1770. [CrossRef]
- Gregory, S.G.; Schmidt, S.; Seth, P.; Oksenberg, J.R.; Hart, J.; Prokop, A.; Caillier, S.J.; Ban, M.; Goris, A.; Barcellos, L.F.; et al. Interleukin 7 Receptor Alpha Chain (IL7R) Shows Allelic and Functional Association with Multiple Sclerosis. Nat Genet 2007, 39, 1083–1091. [CrossRef]
- Taylor, C.A.; Watson, R.A.; Tong, O.; Ye, W.; Nassiri, I.; Gilchrist, J.J.; de los Aires, A.V.; Sharma, P.K.; Koturan, S.; Cooper, R.A.; et al. IL7 Genetic Variation and Toxicity to Immune Checkpoint Blockade in Patients with Melanoma. Nat Med 2022, 28, 2592–2600. [CrossRef]
- Groha, S.; Alaiwi, S.A.; Xu, W.; Naranbhai, V.; Nassar, A.H.; Bakouny, Z.; El Zarif, T.; Saliby, R.M.; Wan, G.; Rajeh, A.; et al. Germline Variants Associated with Toxicity to Immune Checkpoint Blockade. Nat Med 2022, 28, 2584–2591. [CrossRef]
- Miyamoto, H.; Kondo, Y.; Itobayashi, E.; Uehara, M.; Hiraoka, A.; Kudo, M.; Kakizaki, S.; Kagawa, T.; Miuma, S.; Suzuki, T.; et al. Evaluation of the Associations of Interlukin-7 Genetic Variants with Toxicity and Efficacy of Immune Checkpoint Inhibitors: A Replication Study of a Japanese Population, Based on the Findings of a European Genome-Wide Association Study. Hepatology Research n/a. [CrossRef]
- Saad, E.; Mehrabad, E.M.; Labaki, C.; Saliby, R.M.; Semaan, K.; Eid, M.; Machaalani, M.; Chehade, R.E.H.; Nawfal, R.; Sun, M.; et al. 22 Association of a Germline Single Nucleotide Polymorphism (SNP) in the Interleukin-7 (IL7) Gene with Immune-Related Adverse Events (irAEs). Oncologist 2024, 29, S1–S2. [CrossRef]
- Takada, H.; Osawa, L.; Komiyama, Y.; Muraoka, M.; Suzuki, Y.; Sato, M.; Kobayashi, S.; Yoshida, T.; Takano, S.; Maekawa, S.; et al. Interleukin-7 Risk Allele, Lymphocyte Counts, and Autoantibodies for Prediction of Risk of Immune-Related Adverse Events in Patients Receiving Atezolizumab plus Bevacizumab Therapy for Unresectable Hepatocellular Carcinoma. Oncology 2024, 103, 37–47. [CrossRef]
- Haratani, K.; Hayashi, H.; Chiba, Y.; Kudo, K.; Yonesaka, K.; Kato, R.; Kaneda, H.; Hasegawa, Y.; Tanaka, K.; Takeda, M.; et al. Association of Immune-Related Adverse Events With Nivolumab Efficacy in Non-Small-Cell Lung Cancer. JAMA Oncol 2018, 4, 374–378. [CrossRef]
- Auton, A.; Abecasis, G.R.; Altshuler, D.M.; Durbin, R.M.; Abecasis, G.R.; Bentley, D.R.; Chakravarti, A.; Clark, A.G.; Donnelly, P.; Eichler, E.E.; et al. A Global Reference for Human Genetic Variation. Nature 2015, 526, 68–74. [CrossRef]
| Characteristic | Total Cohort (N=153) | AEs YES (n=71) | AEs NO (n=53) | P-value |
|---|---|---|---|---|
| Median Age (Range), years | 64 (42–84) | 66 (42–83) | 64 (50–84) | 0.42 |
| Sex, n (%) | ||||
| Male | 102 (66.7%) | 53 (74.6%) | 40 (75.5%) | 0.89 |
| Female | 51 (33.3%) | 18 (25.4%) | 13 (24.5%) | |
| Histology, n (%) | ||||
| Adenocarcinoma | 84 (54.9%) | 43 (60.6%) | 36 (67.9%) | 0.58 |
| Squamous | 39 (25.5%) | 21 (29.6%) | 14 (26.4%) | |
| Other | 30 (19.6%) | 7 (9.8%) | 3 (5.7%) | |
| PD-L1 Expression, n (%) | ||||
| <1% (Negative) | 25 (16.3%) | 16 (22.5%) | 9 (17.0%) | 0.61 |
| ≥1% (Positive) | 128 (83.7%) | 55 (77.5%) | 42 (79.2%) | |
| Metastatic Sites, n (%) | ||||
| Brain | 16 (9.9%) | 9 (12.9%) | 6 (11.1%) | 0.76 |
| Lung | 67 (41.4%) | 34 (48.6%) | 28 (51.9%) | 0.72 |
| Liver | 16 (9.9%) | 8 (11.4%) | 8 (14.8%) | 0.59 |
| Bone | 47 (29.0%) | 23 (32.9%) | 17 (31.5%) | 0.87 |
| Treatment Modality, n (%) | ||||
| ICI Monotherapy | 41 (26.8%) | 25 (35.2%) | 16 (30.2%) | 0.67 |
| Chemo-Immunotherapy | 112 (73.2%) | 46 (64.8%) | 37 (69.8%) | |
| Outcomes, n (%) | ||||
| Treatment interruption | 91 (76.8%) | 59 (84.2%) | 34 (61.8%) | 0.004 |
| Response at 1 year | 39 (31.7%) | 20 (29.9%) | 18 (38.3%) | 0.34 |
| Genotype / Allele | AEs YES (n=71) | AEs NO (n=53) | OR (95% CI) | P-value |
|---|---|---|---|---|
| Genotype, n (%) | ||||
| GG (Wild-type) | 56 (78.9%) | 50 (94.3%) | Reference | - |
| AG (Heterozygous) | 13 (18.3%) | 3 (5.7%) | 3.03 (0.88–10.3) | 0.037 |
| AA (Homozygous Risk) | 2 (2.8%) | 0 (0.0%) | 2.31 (0.10–49.2) | 0.21 |
| Allele Frequency, n (%) | ||||
| G Allele | 125 (88.0%) | 103 (97.2%) | Reference | - |
| A Allele (Risk) | 17 (12.0%) | 3 (2.8%) | 3.71 (1.14–12.0) | 0.0081 |
| Dominant Model, n (%) | ||||
| Non-Carriers (GG) | 56 (78.9%) | 50 (94.3%) | Reference | - |
| Carriers (AG + AA) | 15 (21.1%) | 3 (5.7%) | 4.64 (1.50–17.2)a | 0.0203 |
| Model Type | AUC | Sensitivity | Specificity | Risk Stratification Criteria |
|---|---|---|---|---|
| Clinical Only | 0.57 | 45.1% | 66.0% | Age, Sex, Histology, PD-L1 |
| Genetic Only (IL7) | 0.59 | 21.1% | 94.3% | rs16906115 Genotype |
| Combined Model | 0.67 | 52.3% | 88.6% | Clinical + IL7 Genotype |
| Model Type | AUC | Sensitivity | Specificity | Risk Stratification Criteria |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




