Submitted:
11 January 2026
Posted:
13 January 2026
You are already at the latest version
Abstract
Keywords:
Introduction
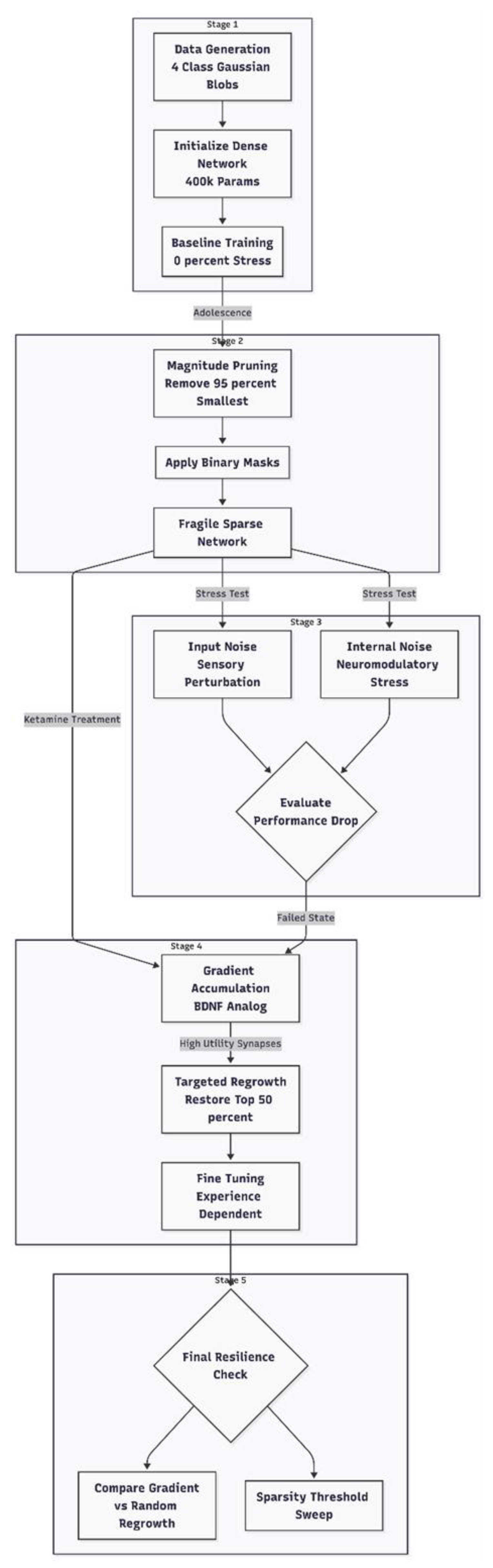
Methods
Computational Environment and Reproducibility
Task and Dataset
Network Architecture
Baseline Training
Pruning Procedure
Plasticity Restoration
Stress Tests and Evaluation
Results
Baseline Performance
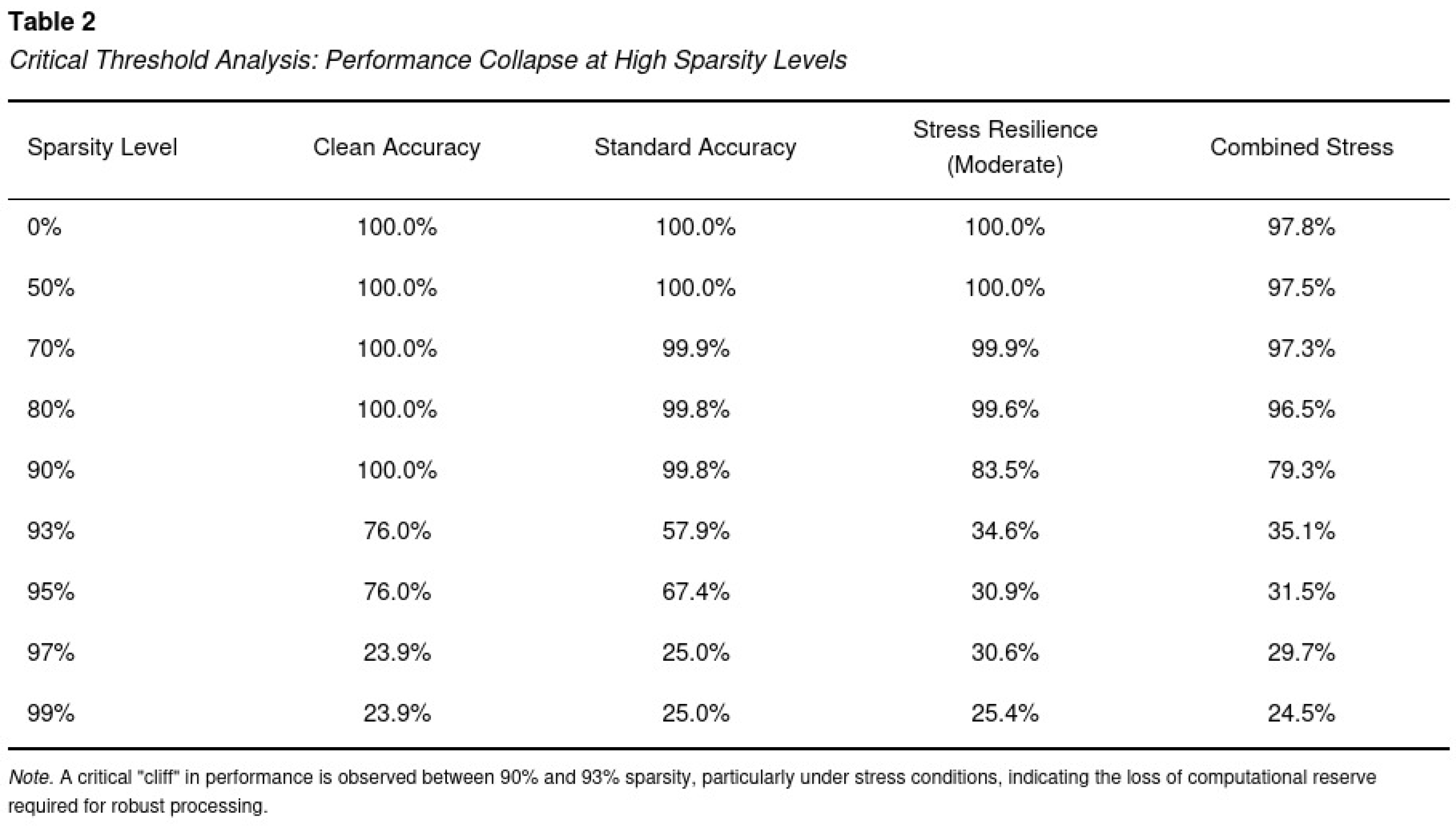
Consequences of 95 Percent Pruning
Recovery Through Gradient-Guided Regrowth
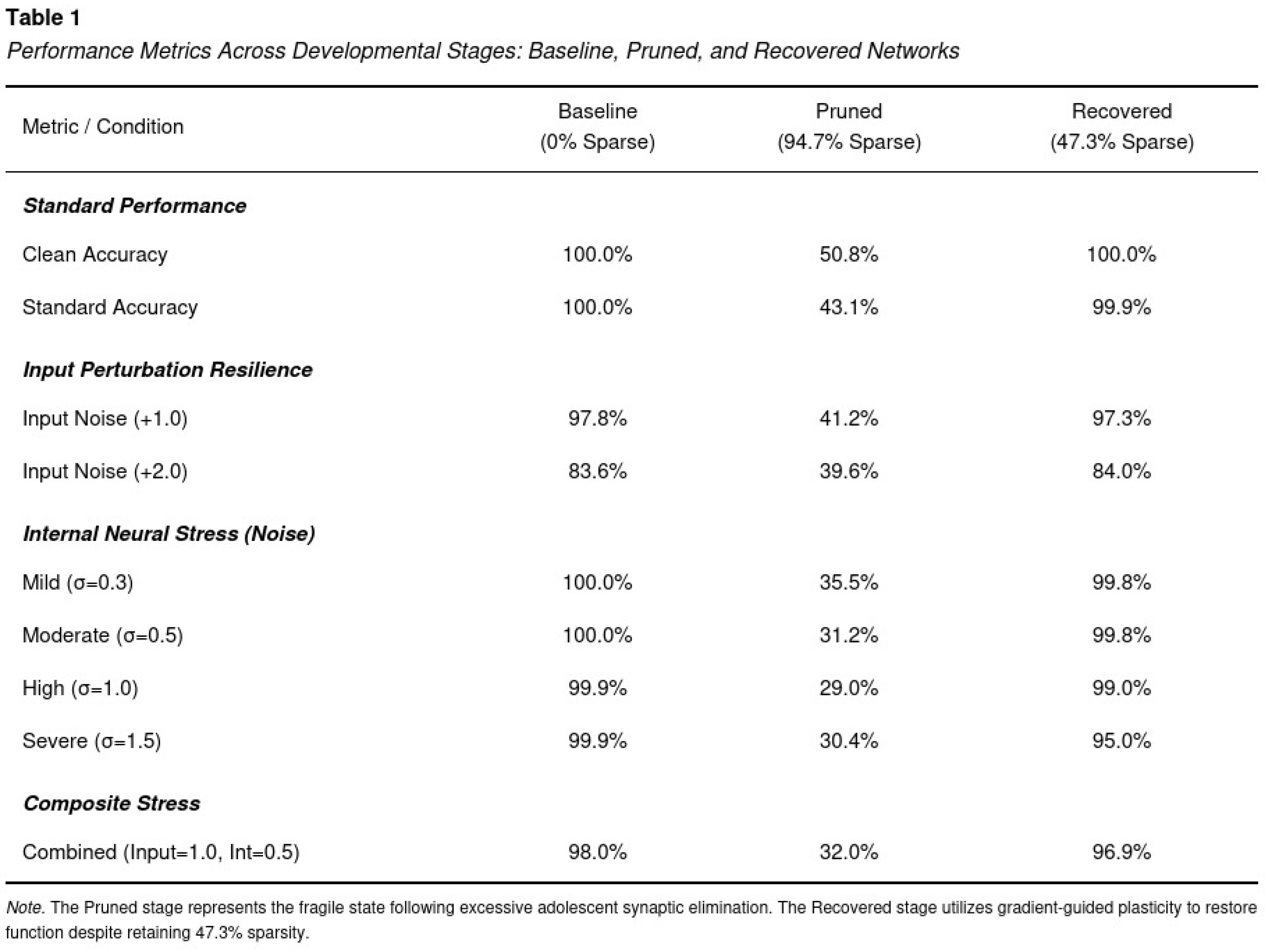
Random Versus Targeted Regrowth
Sparsity Threshold
Discussion
Interpretation of Results
New Insights from the Improved Model
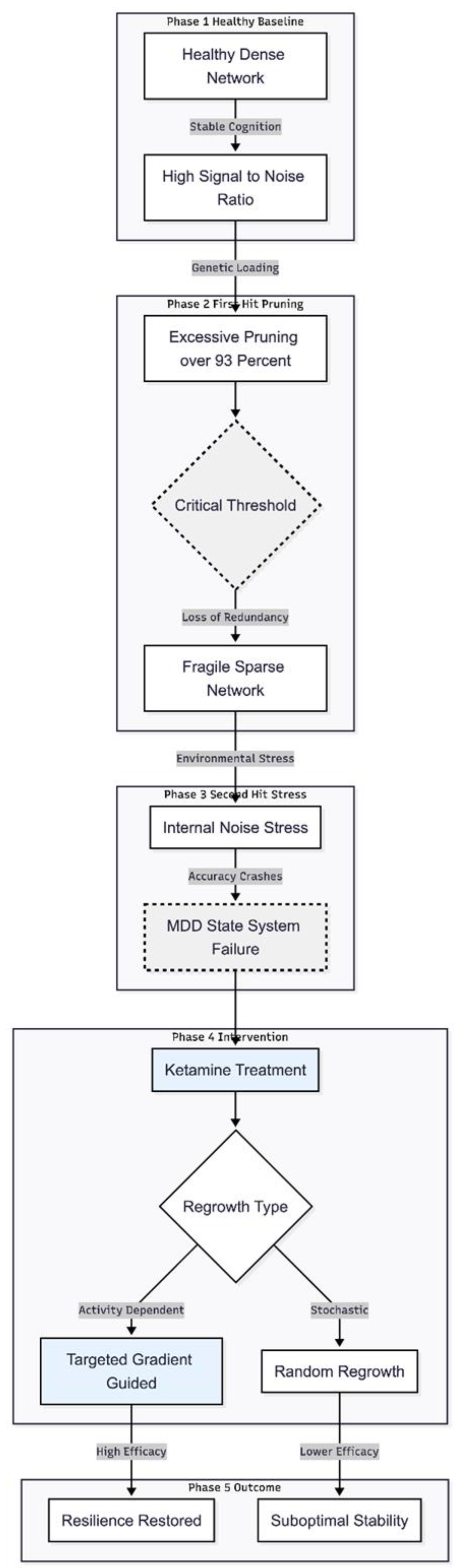
Implications for the Pruning-Mediated Plasticity Deficit Hypothesis
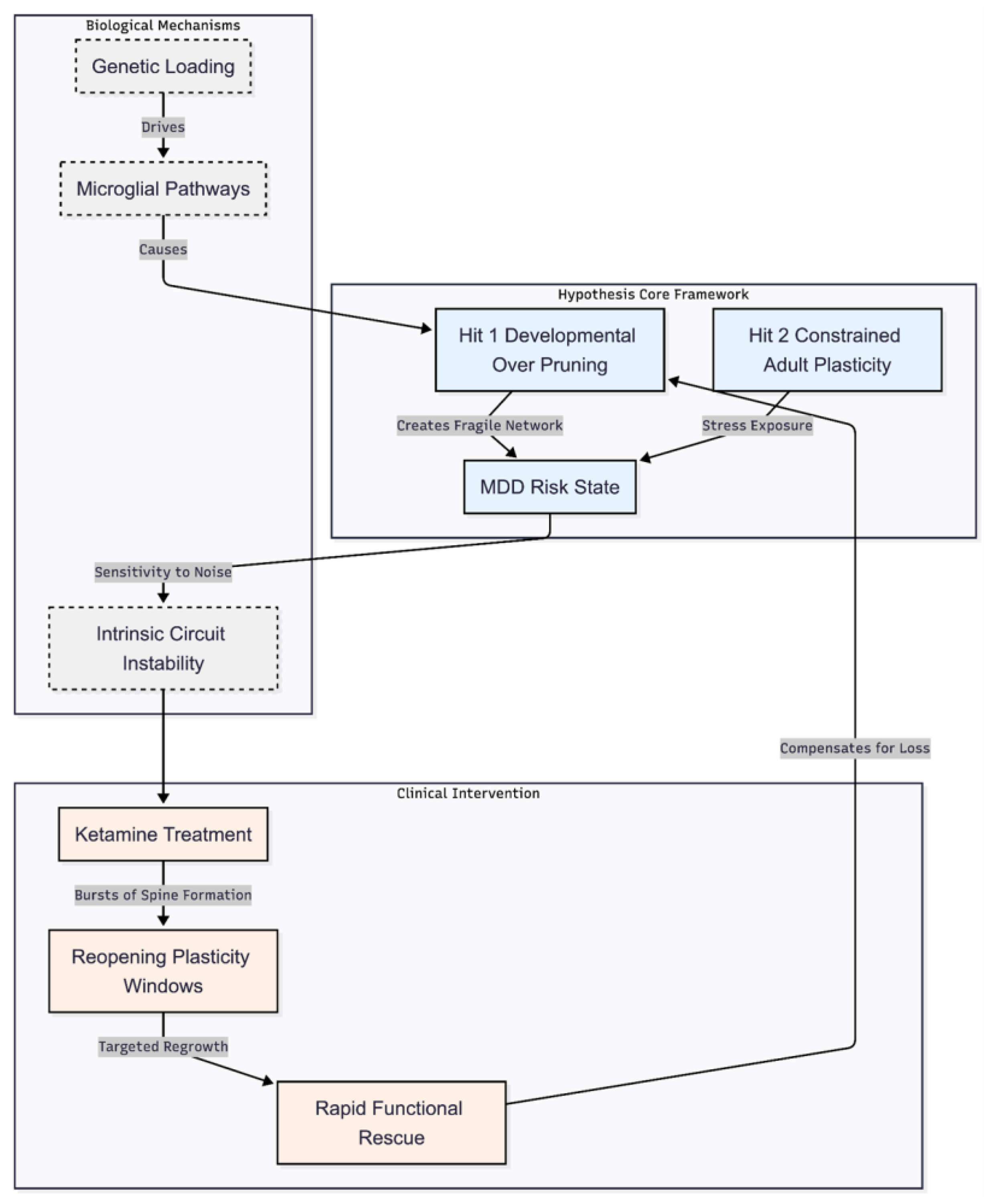
Novelty of the Present Model
Potential Impact and Translational Implications
Limitations
Conclusions
Funding Declaration
Ethics Declaration
Conflicts of Interest
References
- Sekar A, Bialas AR, de Rivera H, et al. Schizophrenia risk from complex variation of complement component 4. Nature. 2016;530(7589):177–183. [CrossRef]
- Duman RS, Aghajanian GK. Synaptic dysfunction in depression: Potential therapeutic targets. Science. 2012;338(6103):68–72. [CrossRef]
- Hoffman RE, Dobscha SK. Cortical pruning and the development of schizophrenia: A computer model. Schizophrenia Bulletin. 1989;15(3):477–490. [CrossRef]
- Averbeck BB. Pruning recurrent neural networks replicates adolescent changes in working memory and reinforcement learning. Proceedings of the National Academy of Sciences. 2022;119(22):e2121331119. [CrossRef]
- Scholl C, Rule ME, Hennig MH. The information theory of developmental pruning: Optimizing global network architectures using local synaptic rules. PLoS Computational Biology. 2021;17(10):e1009458. [CrossRef]
- Liu S, Chen T, Chen X, et al. Sparse training via boosting pruning plasticity with neuroregeneration. In Advances in Neural Information Processing Systems (Vol. 34, pp. 9908–9922). [CrossRef]
- Cheung N. From Pruning to Plasticity: Refining the Etiological Architecture of Major Depressive Disorder Through Causal and Polygenic Inference. Preprints. [CrossRef]
- Monteggia LM, Kavalali ET. Synaptic basis of rapid antidepressant action. European Archives of Psychiatry and Clinical Neuroscience. 2024;275:1539–1546. [CrossRef]
- Moda-Sava RN, Murdock MH, Parekh PK, et al. Sustained rescue of prefrontal circuit dysfunction by antidepressant-induced spine formation. Science. 2019;364(6436):eaat8078. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



