Submitted:
05 January 2026
Posted:
07 January 2026
You are already at the latest version
Abstract

Keywords:
1. Introduction
2. Materials and Methods
2. 1. Chemicals and Reagents
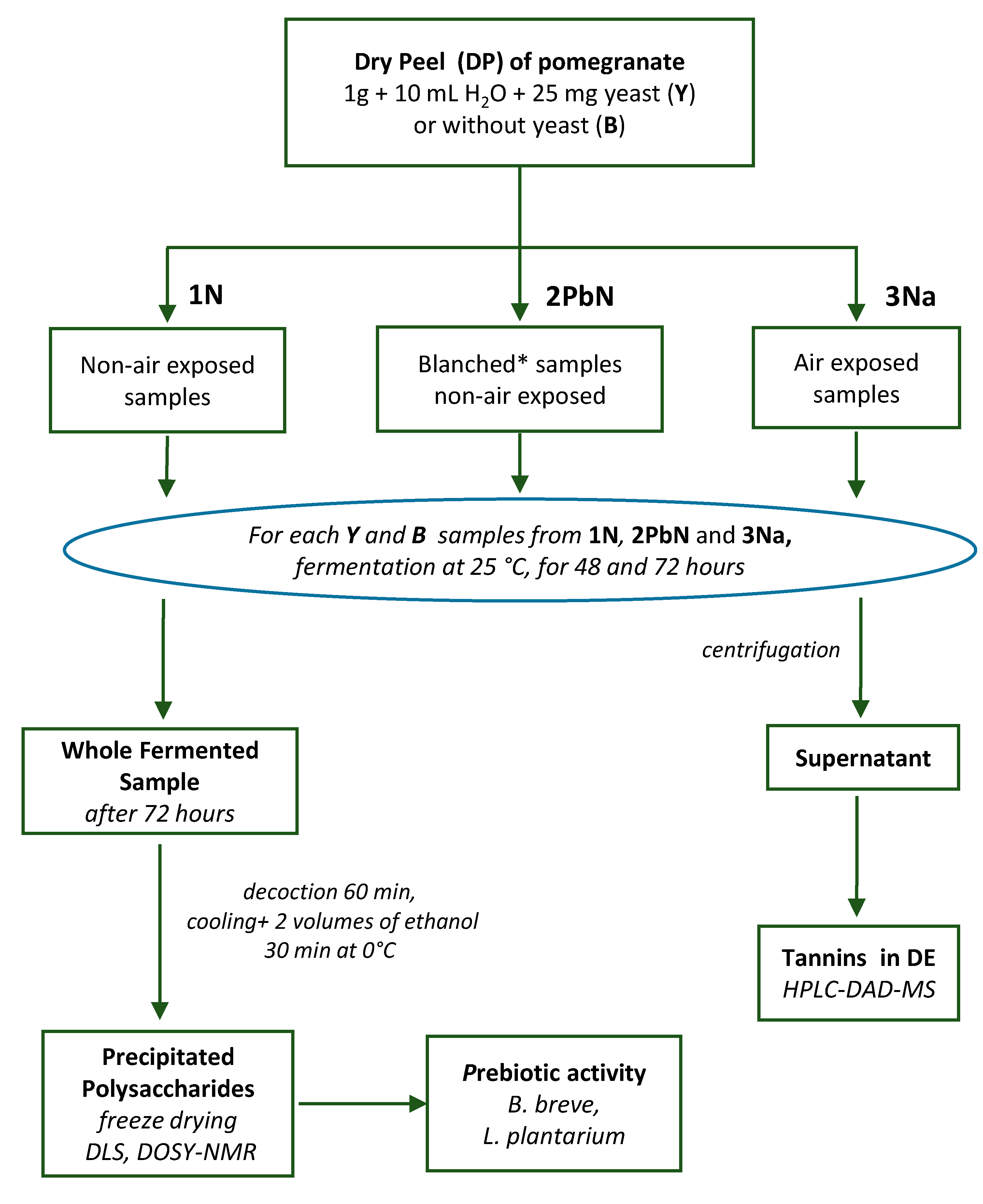
2. 2. Fermentation of Pomegranate Samples
2. 3. Extraction of Tannins and Polysaccharides
2. 4. Analysis of Tannin Content
2. 5. Analyses of Polysaccharides by DLS
2. 6. Analyses of Ethanol and Polysaccharides by 1H-NMR and DOSY
2. 7. In Vitro Evaluation of Prebiotic Properties
2. 8. Statistical Evaluation
3. Results and Discussion
3.1. Fermentation Trials on Pomegranate Peels with and Without Addition of S. cerevisiae
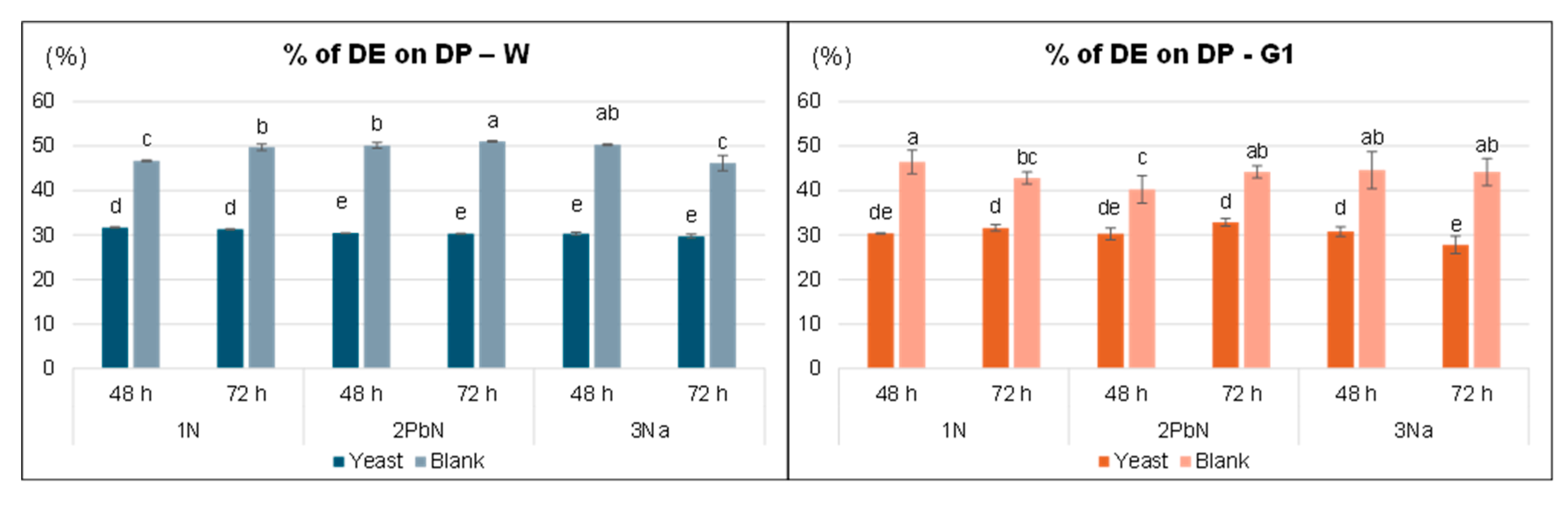
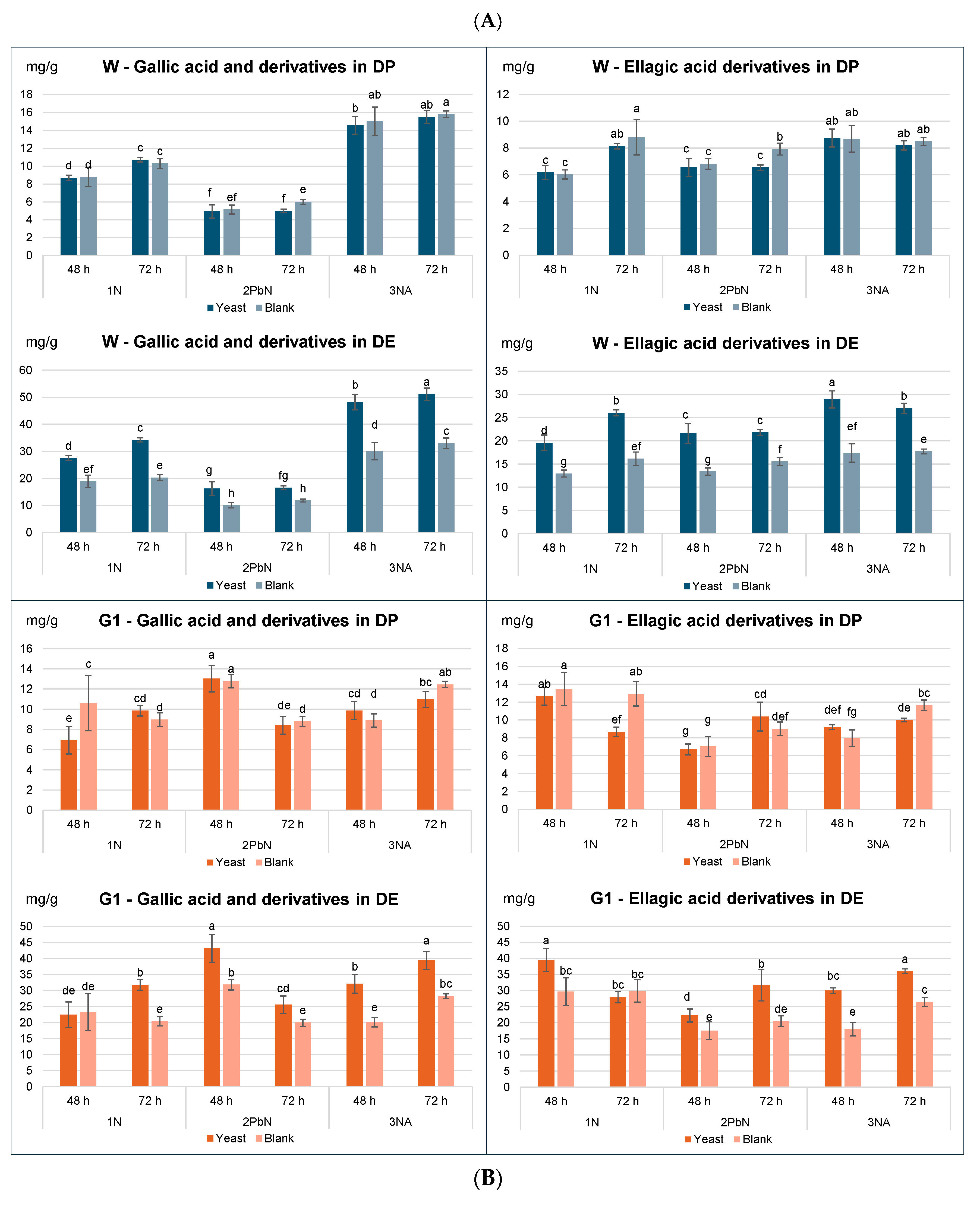
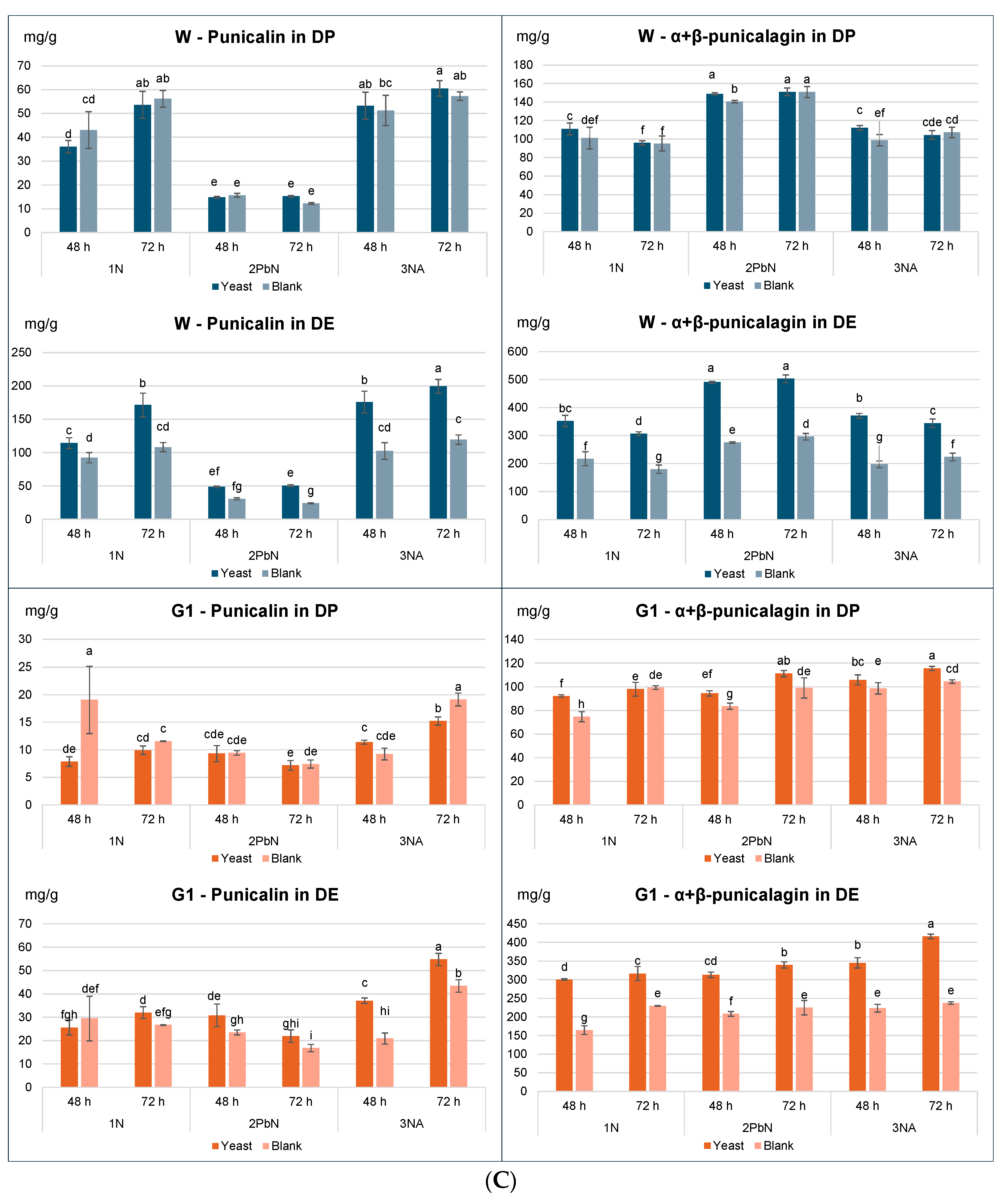
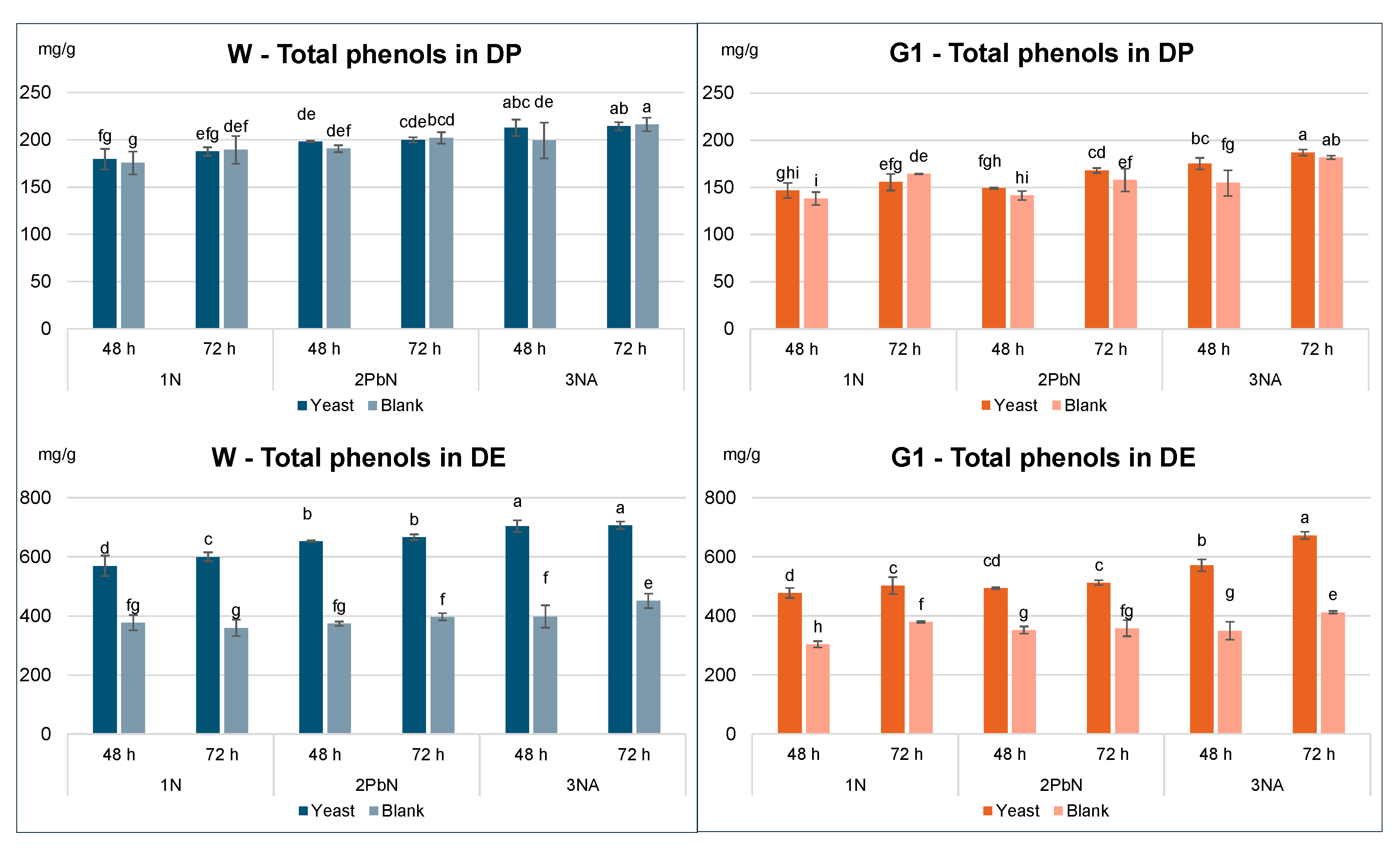
3. 2. Tannin Content in Dry Peel (DP) and Dry Extracts (DE)
3. 3. Chemical and Biological Properties of Polysaccharides
3.1.1. Extraction Yields
3.1.2. DLS Analysis
3.1.4. DOSY Experiments on Polysaccharides
3.1.5. In Vitro Prebiotic Activity on B. breve and L. plantarum
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| UAE | ultrasound-assisted extraction |
| MAE | microwave-assisted extraction |
| SFE | supercritical fluid extraction. |
| DE | dry extract. |
| DP | dry peel |
| HPLC-DAD | high-performance liquid chromatography-diode array detector. |
| DOSY-1H-NMR | Diffusion-Ordered Spectroscopy-proton Nuclear Magnetic Resonance. |
| DLS | dynamic light scattering. |
References
- Balli, D.; Cecchi, L.; Khatib, M.; Bellumori, M.; Cairone, F.; Carradori, S.; Zengin, G.; Cesa, S.; Innocenti, M.; Mulinacci, N. Characterization of Arils Juice and Peel Decoction of Fifteen Varieties of Punica Granatum L.: A Focus on Anthocyanins, Ellagitannins and Polysaccharides. Antioxidants 2020, 9. [Google Scholar] [CrossRef]
- Buenrostro-Figueroa, J.J.; Nevárez-Moorillón, G.V.; Chávez-González, M.L.; Sepúlveda, L.; Ascacio-Valdés, J.A.; Aguilar, C.N.; Pedroza-Islas, R.; Huerta-Ochoa, S.; Arely Prado-Barragán, L. Improved Extraction of High Value-Added Polyphenols from Pomegranate Peel by Solid-State Fermentation. Fermentation 2023, 9. [Google Scholar] [CrossRef]
- Grillo, G.; Capaldi, G.; Radošević, K.; Jakopović, Ž.; Markov, K.; Brncic, M.; Gallina, L.; Calcio Gaudino, E.; Cravotto, G. Unlocking the Bioactive Potential of Pomegranate Peels: A Green Extraction Approach. Antioxidants 2023, 12. [Google Scholar] [CrossRef] [PubMed]
- Hegazi, N.M.; El-Shamy, S.; Fahmy, H.; Farag, M.A. Pomegranate Juice as a Super-Food: A Comprehensive Review of Its Extraction, Analysis, and Quality Assessment Approaches. Journal of Food Composition and Analysis 2021, 97. [Google Scholar] [CrossRef]
- Lampakis, D.; Skenderidis, P.; Leontopoulos, S. Technologies and Extraction Methods of Polyphenolic Compounds Derived from Pomegranate (Punica Granatum) Peels. a Mini Review. Processes 2021, 9, 1–14. [Google Scholar] [CrossRef]
- Mulinacci, N.; Khatib, M.; Innocenti, M.; Giuliani, C.; Al- Tamimi, A.; Romani, A. Mesocarp and Exocarp of Laffan and Wonderful Pomegranate Varieties: By-Products as a Source of Ellagitannins. International Journal of Food and Nutritional Science 2017, 4, 60–66. [Google Scholar] [CrossRef]
- Abid, M.; Cheikhrouhou, S.; Renard, C.M.G.C.; Bureau, S.; Cuvelier, G.; Attia, H.; Ayadi, M.A. Characterization of Pectins Extracted from Pomegranate Peel and Their Gelling Properties. Food Chem 2017, 215, 318–325. [Google Scholar] [CrossRef]
- Khatib, M.; Giuliani, C.; Rossi, F.; Adessi, A.; Al-Tamimi, A.; Mazzola, G.; Di Gioia, D.; Innocenti, M.; Mulinacci, N. Polysaccharides from By-Products of the Wonderful and Laffan Pomegranate Varieties: New Insight into Extraction and Characterization. Food Chem 2017, 235, 58–66. [Google Scholar] [CrossRef]
- Balli, D.; Khatib, M.; Cecchi, L.; Adessi, A.; Melgarejo, P.; Nunes, C.; Coimbra, M.A.; Mulinacci, N. Pomegranate Peel as a Promising Source of Pectic Polysaccharides: A Multi-Methodological Analytical Investigation. Food Chem 2022, 397. [Google Scholar] [CrossRef]
- D’Agostino, S.; Cecchi, L.; Khatib, M.; Domizio, P.; Zoccatelli, G.; Civa, V.; Mainente, F.; Breschi, C.; Ugolini, T.; Meneguzzo, F.; et al. 1H-NMR, HPSEC-RID, and HPAEC-PAD Characterization of Polysaccharides Extracted by Hydrodynamic Cavitation from Apple and Pomegranate By-Products for Their Valorization: A Focus on Pectin. Processes 2024, 12. [Google Scholar] [CrossRef]
- Parisio, C.; Lucarini, E.; Micheli, L.; Toti, A.; Khatib, M.; Mulinacci, N.; Calosi, L.; Bani, D.; Mannelli, L.D.C.; Ghelardini, C. Pomegranate Mesocarp against Colitis-Induced Visceral Pain in Rats: Effects of a Decoction and Its Fractions. Int J Mol Sci 2020, 21, 1–16. [Google Scholar] [CrossRef]
- Tortora, K.; Femia, A. Pietro; Romagnoli, A.; Sineo, I.; Khatib, M.; Mulinacci, N.; Giovannelli, L.; Caderni, G. Pomegranate By-Products in Colorectal Cancer Chemoprevention: Effects in Apc-Mutated Pirc Rats and Mechanistic Studies In Vitro and Ex Vivo. Mol Nutr Food Res 2018, 62. [Google Scholar] [CrossRef]
- Faria, G.M.L.; Silva, E.K. Pulsed Electric Field, Ultrasound and Microwave Heating Based Extraction Techniques for Valorization of Pomegranate Peel by-Products: A Review. J Environ Chem Eng 2024, 12. [Google Scholar] [CrossRef]
- Xu, R.; Chen, J.; Yan, N.; Xu, B.; Lou, Z.; Xu, L. High-Value Utilization of Agricultural Residues Based on Component Characteristics: Potentiality and Challenges. Journal of Bioresources and Bioproducts 2025, 10, 271–294. [Google Scholar] [CrossRef]
- Demiray, E.; Karatay, S.E.; Dönmez, G. Evaluation of Pomegranate Peel in Ethanol Production by Saccharomyces Cerevisiae and Pichia Stipitis. Energy 2018, 159, 988–994. [Google Scholar] [CrossRef]
- Liu, H.M.; Xu, P.F.; Cheng, M.Y.; Lei, S.N.; Liu, Q.L.; Wang, W. Optimization of Fermentation Process of Pomegranate (Punica Granatum l.) Peel and Schisandra Chinensis (Turcz.) Baill and the Biological Activities of Fermentation Broth: Antioxidant Activity and Protective Effect against H2o2-Induced Oxidative Damage in Hacat Cells. Molecules 2021, 26. [Google Scholar] [CrossRef]
- Siddiqui, S.A.; Erol, Z.; Rugji, J.; Taşçı, F.; Kahraman, H.A.; Toppi, V.; Musa, L.; Di Giacinto, G.; Bahmid, N.A.; Mehdizadeh, M.; et al. An Overview of Fermentation in the Food Industry—Looking Back from a New Perspective. Bioresour Bioprocess 2023, 10. [Google Scholar] [CrossRef]
- Ezeora, K.C.; Setati, M.E.; Fawole, O.A.; Opara, U.L. Pomegranate Wine Production and Quality: A Comprehensive Review. Fermentation 2024, 10. [Google Scholar] [CrossRef]
- Berenguer, M.; Vegara, S.; Barrajón, E.; Saura, D.; Valero, M.; Martí, N. Physicochemical Characterization of Pomegranate Wines Fermented with Three Different Saccharomyces Cerevisiae Yeast Strains. Food Chem 2016, 190, 848–855. [Google Scholar] [CrossRef] [PubMed]
- Gumienna, M.; Szwengiel, A.; Górna, B. Bioactive Components of Pomegranate Fruit and Their Transformation by Fermentation Processes. European Food Research and Technology 2016, 242, 631–640. [Google Scholar] [CrossRef]
- Valero-Cases, E.; Nuncio-Jáuregui, N.; Frutos, M.J. Influence of Fermentation with Different Lactic Acid Bacteria and in Vitro Digestion on the Biotransformation of Phenolic Compounds in Fermented Pomegranate Juices. J Agric Food Chem 2017, 65, 6488–6496. [Google Scholar] [CrossRef]
- Liu, S.; Hu, J.; Zhong, Y.; Hu, X.; Yin, J.; Xiong, T.; Nie, S.; Xie, M. A Review: Effects of Microbial Fermentation on the Structure and Bioactivity of Polysaccharides in Plant-Based Foods. Food Chem 2024, 440. [Google Scholar] [CrossRef]
- Mall, U.P.; Patel, V.H. Evaluation of Pomegranate (Punica Granatum) Peel for Bioaccessibility of Polyphenols and Prebiotic Potential Using in Vitro Model. Food Chemistry Advances 2023, 2. [Google Scholar] [CrossRef]
- de Oliveira, D.P.; Todorov, S.D.; Fabi, J.P. Exploring the Prebiotic Potentials of Hydrolyzed Pectins: Mechanisms of Action and Gut Microbiota Modulation. Nutrients 2024, 16. [Google Scholar] [CrossRef]
- Cecchi, L.; Khatib, M.; Bellumori, M.; Civa, V.; Domizio, P.; Innocenti, M.; Balli, D.; Mulinacci, N. Industrial Drying for Agrifood By-Products Re-Use: Cases Studies on Pomegranate Peel (Punica Granatum L.) and Stoned Olive Pomace (Pâtè, Olea Europaea L.). Food Chem 2023, 403. [Google Scholar] [CrossRef] [PubMed]
- Jakati, S.V.; Gogineni, R.; Aley, M.S.; Chachada, A.; Atram, H.K.; Mankar, M. Glass Bead Sterilizer Comprehensively Defeats Hot Air Oven in Orthodontic Clinic. Journal of Indian Orthodontic Society 2015, 49, 134–138. [Google Scholar] [CrossRef]
- Khatib, M.; Al-Tamimi, A.; Cecchi, L.; Adessi, A.; Innocenti, M.; Balli, D.; Mulinacci, N. Phenolic Compounds and Polysaccharides in the Date Fruit (Phoenix Dactylifera L.): Comparative Study on Five Widely Consumed Arabian Varieties. Food Chem 2022, 395. [Google Scholar] [CrossRef]
- Ammon, J.; Andlauer, C.; Annweiler, E.; Bauer-Aymanns, H.; Bunzel, M.; Burgmaier-Thielert, E.; Brzezina, T.; Christoph, N.; Dietrich, H.; Dohr, A.; et al. Guide to NMR Method Development and Validation-Part II: Multivariate Data Analysis.
- Müller-Maatsch, J.; Caligiani, A.; Tedeschi, T.; Elst, K.; Sforza, S. Simple and Validated Quantitative 1H NMR Method for the Determination of Methylation, Acetylation, and Feruloylation Degree of Pectin. J Agric Food Chem 2014, 62, 9081–9087. [Google Scholar] [CrossRef]
- Zonfrillo, B.; Bellumori, M.; Truzzi, E.; Khatib, M.; Faraoni, P.; Bertelli, D.; Ranaldi, F.; Mulinacci, N. Ulvan as a Source of Oligosaccharides for Biological Applications: Enzymatic Hydrolysis in a Biocompatible Medium. Food Chem 2025, 492. [Google Scholar] [CrossRef]
- Mazaheri, D.; Orooji, Y.; Mazaheri, M.; Moghaddam, M.S.; Karimi-Maleh, H. Bioethanol Production from Pomegranate Peel by Simultaneous Saccharification and Fermentation Process. Biomass Convers Biorefin 2021. [Google Scholar] [CrossRef]
- Caballero, V.; Estévez, M.; Tomás-Barberán, F.A.; Morcuende, D.; Martín, I.; Delgado, J. Biodegradation of Punicalagin into Ellagic Acid by Selected Probiotic Bacteria: A Study of the Underlying Mechanisms by MS-Based Proteomics. J Agric Food Chem 2022, 70, 16273–16285. [Google Scholar] [CrossRef]
- Chaudhary, A.; Hussain, Z.; Aihetasham, A.; El-Sharnouby, M.; Abdul Rehman, R.; Azmat Ullah Khan, M.; Zahra, S.; Saleem, A.; Azhar, S.; Alhazmi, A.; et al. Pomegranate Peels Waste Hydrolyzate Optimization by Response Surface Methodology for Bioethanol Production. Saudi J Biol Sci 2021, 28, 4867–4875. [Google Scholar] [CrossRef]
- Mensah, J.K.M.; Twumasi, P. Use of Pineapple Waste for Single Cell Protein (SCP) Production and the Effect of Substrate Concentration on the Yield. J Food Process Eng 2017, 40. [Google Scholar] [CrossRef]
- Perez-Samper, G.; Cerulus, B.; Jariani, A.; Vermeersch, L.; Simancas, N.B.; Bisschops, M.M.M.; van den Brink, J.; Solis-Escalante, D.; Gallone, B.; De Maeyer, D.; et al. The Crabtree Effect Shapes the Saccharomyces Cerevisiae Lag Phase during the Switch between Different Carbon Sources. mBio 2018, 9, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Devecioglu, D.; Kara, D.; Tapan, R.; Karbancioglu-Guler, F.; Kahveci, D. Enhanced Production of Gamma-Aminobutyric Acid in Fermented Carrot Juice by Utilizing Pectin Hydrolysate Derived from Pomegranate Waste. Food Sci Nutr 2024, 12, 6534–6547. [Google Scholar] [CrossRef]
- Maina, N.H.; Pitkänen, L.; Heikkinen, S.; Tuomainen, P.; Virkki, L.; Tenkanen, M. Challenges in Analysis of High-Molar Mass Dextrans: Comparison of HPSEC, AsFlFFF and DOSY NMR Spectroscopy. Carbohydr Polym 2014, 99, 199–207. [Google Scholar] [CrossRef]
- Suárez, E.R.; Syvitski, R.; Kralovec, J.A.; Noseda, M.D.; Barrow, C.J.; Ewart, H.S.; Lumsden, M.D.; Grindley, T.B. Immunostimulatory Polysaccharides from Chlorella Pyrenoidosa. A New Galactofuranan. Measurement of Molecular Weight and Molecular Weight Dispersion by DOSY NMR. Biomacromolecules 2006, 7, 2368–2376. [Google Scholar] [CrossRef]
- Bachari, S.; Ghaderi-Ghahfarokhi, M.; Gavlighi, H.A.; Zarei, M. Ultrasonic Depolymerization of Pomegranate Peel Pectin: Effect of Sonication Time on Antioxidant, α-Amylase Inhibitory, and Prebiotic Properties. Food Chem X 2024, 24. [Google Scholar] [CrossRef]
- Grzesiuk, E.; Jankowska, A.; Laubitz, D.; Antushevich, H.; Zabielski, R. Competition of Lactobacillus Paracasei with Salmonella Enterica for Adhesion to Caco-2 Cells. J Biomed Biotechnol 2008, 2008. [Google Scholar] [CrossRef]
- Symonds, E.L.; O’Mahony, C.; Lapthorne, S.; O’Mahony, D.; Sharry, J. Mac; O’Mahony, L.; Shanahan, F. Bifidobacterium Infantis 35624 Protects against Salmonella-Induced Reductions in Digestive Enzyme Activity in Mice by Attenuation of the Host Inflammatory Response. Clin Transl Gastroenterol 2012, 3. [Google Scholar] [CrossRef] [PubMed]
- Aloisio, I.; Prodam, F.; Giglione, E.; Bozzi Cionci, N.; Solito, A.; Bellone, S.; Baffoni, L.; Mogna, L.; Pane, M.; Bona, G.; et al. Three-Month Feeding Integration With Bifidobacterium Strains Prevents Gastrointestinal Symptoms in Healthy Newborns. Front Nutr 2018, 5. [Google Scholar] [CrossRef]
- Koropatkin, N.M.; Cameron, E.A.; Martens, E.C. How Glycan Metabolism Shapes the Human Gut Microbiota. Nat Rev Microbiol 2012, 10, 323–335. [Google Scholar] [CrossRef] [PubMed]
| Samples | Polysaccharides yield (% on DP) | |
|---|---|---|
| G1 | Wonderful | |
| B-1N | 6.9 | 8.4 |
| Y-1N | 7.2 | 6.2 |
| B-2PbN | 10.5 | 8.3 |
| Y-2PbN | 8.3 | 6.3 |
| B-3Na | 8.3 | 10 |
| Y-3Na | 6.7 | 7.1 |
| Samples | PdI | Z-Pk1 | Pk1 | Pk 2 | Pk1 | Pk 2 |
|---|---|---|---|---|---|---|
| D (nm) | D (nm) | D (nm) | Area % | Area % | ||
| W-B-1N | 0.24 ± 0.02 | 342 ± 42 | 441 ± 58 | nd | 100 | nd |
| W-Y-1N | 0.24 ± 0.01 | 361 ± 19 | 463 ± 31 | 91 ± 1 | 99 ± 1 | 0.7 ± 0.2 |
| W-B-2PbN | 0.22 ± 0.02 | 749 ± 24 | 1017 ± 53 | 215 ± 14 | 90 ± 4 | 10.0 ± 3.5 |
| W-Y-2PbN | 0.26 ± 0.01 | 491 ± 14 | 563 ± 66 | 110 ± 11 | 89 ± 1 | 10.9 ± 1.2 |
| W-B-3Na | 0.23 ± 0.01 | 314 ± 10 | 390 ± 13 | nd | 99 ± 1 | 0.4 ± 0.5 |
| W-Y-3Na | 0.22 ± 0.01 | 282 ± 10 | 343 ± 13 | nd | 99 ± 1 | 0.8 ± 0.9 |
| W-Dec | 0.27 ± 0.03 | 716 ± 151 | 1059 ± 186 | 186 ± 12 | 88 ± 12 | 12.2 ± 12.3 |
| G1-B-1N | 0.26 ± 0.08 | 329 ± 8 | 390 ± 25 | 798 ± 25 | 99 ± 1 | 0.3 ± 0.5 |
| G1-Y-1N | 0.27 ± 0.02 | 395 ± 5 | 529 ± 37 | nd | 100 | nd |
| G1-B-2PbN | 0.27 ± 0.02 | 660 ± 29 | 943 ± 87 | 115 ± 11 | 96 ± 6 | 4.5 ± 5.7 |
| G1-Y-2PbN | 0.26 ± 0.01 | 408 ± 10 | 498 ± 24 | nd | 100 | 7.8 ± 1.1 |
| G1-B-3Na | 0.29 ± 0.02 | 461 ± 71 | 526 ± 126 | nd | 100 | nd |
| G1-Y-3Na | 0.33 ± 0.03 | 369 ± 7 | 525 ± 35 | nd | 100 | nd |
| G1-Dec | 0.27 ± 0.04 | 652 ± 1 | 908 ± 7 | 81 ± 15 | 97 ± 4 | 2.8 ± 3.9 |
| Samples | CH3OH | CH3COOH | Gal. Ac. | DM | DA |
|---|---|---|---|---|---|
| W-B-1N | 5.89 ± 0.06 | 2.54 ± 0.11 | 39.65 ± 1.20 | 77.96 | 38.91 |
| W-Y-1N | 5.64 ± 0.37 | 2.45 ± 0.24 | 50.71 ± 1.10 | 67.48 | 29.34 |
| W-B-2PbN | 6.05 ± 0.26 | 2.72 ± 0.02 | 46.57 ± 0.92 | 78.78 | 35.47 |
| W-Y-2PbN | 6.22 ± 0.28 | 2.86 ± 0.17 | 50.27 ± 0.95 | 75.08 | 34.56 |
| W-B-3Na | 6.14 ± 0.26 | 2.95 ± 0.07 | 44.77 ± 0.88 | 69.05 | 39.98 |
| W-Y-3Na | 5.83 ± 0.62 | 2.63 ± 0.16 | 52.10 ± 0.78 | 67.91 | 30.82 |
| Dec-W | 5.45 ± 0.15 | 2.61 ± 0.03 | 39.77 ± 1.25 | 74.29 | 33.72 |
| G1-B-1N | 5.66 ± 0.02 | 2.10 ± 0.25 | 45.29 ± 1.40 | 86.66 | 38.11 |
| G1-Y-1N | 6.01 ± 0.60 | 2.88 ± 0.09 | 46.73 ± 0.45 | 75.34 | 35.86 |
| G1-B-2PbN | 6.38 ± 0.08 | 2.37 ± 0.42 | 55.36 ± 0.89 | 69.90 | 37.55 |
| G1-Y-2PbN | 5.77 ± 0.21 | 2.46 ± 0.16 | 55.89 ± 0.98 | 64.52 | 37.21 |
| G1-B-3Na | 6.46 ± 0.06 | 1.97 ± 0.27 | 50.97 ± 1.20 | 64.78 | 35.95 |
| G1-Y-3Na | 5.44 ± 0.38 | 2.65 ± 0.11 | 44.83 ± 0.87 | 73.66 | 37.31 |
| Dec-G1 | 5.92 ± 0.15 | 2.49 ± 0.21 | 44.95 ± 0.72 | 79.88 | 37.76 |
| Populations KDa | |||||||
|---|---|---|---|---|---|---|---|
| Samples | 705-690 | 517-429 | 385-197 | 159-91 | 72-50 | 38-26 | < 20 |
| W-B-1N | 690 (10%) * | 516 (37%) | 357 (37%) | 109 (13%) | - | - | - |
| W-Y-1N | 700 (25%) * | 508 (36%) | 264 (17%) | 98 (19%) | 51 (3%) | - | - |
| W-B-2PbN | 701 (18%) * | - | 351 (42%) | 94 (7%) | 51 (5%) | - | < 20 (28%) |
| W-Y-2PbN | 693 (33%) * | 501 (67%) | - | - | - | - | - |
| W-B-3Na | - | - | 367 (21%) | - | 72 (59%) | - | < 20 (20%) |
| W-Y-3Na | - | - | 385 (31%) | 159 (40%) 108 (25%) |
69 (4%) | - | |
| W-Dec | 705 (96%) * | - | - | - | - | - | - |
| G1-B-1N | - | 509 (11%) | 266 (13%) | - | 70 (13%) | - | < 20 (63%)- |
| G1-Y-1N | - | 429 (4%) | 265 (59%) | 155 (16%) | 68 (10%) | - | < 20 (11%) |
| G1-B-2PbN | - | - | 278 (9%) 197 (10%) |
96 (14%) | - | 38 (10%) 26 (10%) |
< 20 (47%) |
| G1-Y-2PbN | - | 499 (69%) | 357 (26%) | - | 67 (5%) | - | - |
| G1-B-3Na | - | 500 (20%) | 356 (27%) | 138 (24%) | 69 (11%) | 38 (18%) | - |
| G1-Y-3Na | - | 495 (17%) | 367 (36%) | 91 (17%) | - | 34 (4%) | < 20 (26%) |
| G1-Dec | - | 517 (39%) | 367 (42%) | - | 71 (7%) | - | < 20 (12%) |
| B. breve | 0h | 12h | 24h | 30h | 48h |
|---|---|---|---|---|---|
| B632 | CFU/mL | CFU/mL | CFU/mL | CFU/mL | CFU/mL |
| W-Y-3Na | (2.1 ± 0.2) × 105 | (3.5 ± 0.2) × 107 b | (9.8 ± 0.2) × 108 a | (2.1 ± 0.3) × 109 a | (8.4 ± 0.3) × 108 a |
| W-Y-1N | (2.2 ± 0.2) × 105 | (2.7 ± 0.2) × 107 b | (6.6 ± 0.2) × 108 a | (8.7 ± 0.2) × 108 c | (2.1 ± 0.3) × 108 e |
| W-Y-2PbN | (2.1 ± 0.2) × 105 | (3.1 ± 0.2) × 107 b | (8.3 ± 0.3) × 108 b | (9.3 ± 0.4) × 108 c | (5.4 ± 0.3) × 108 c |
| W-B-3Na | (2.4 ± 0.2) × 105 | (3.3 ± 0.2) × 107 b | (4.4 ± 0.4) × 107 g | (3.4 ± 0.3) × 108 de | (3.6 ± 0.2) × 107 g |
| W-B-1N | (2.2 ± 0.2) × 105 | (2.5 ± 0.2) × 107 b | (1.8 ± 0.1) × 107 g | (8.6 ± 0.2) × 107 ef | (1.2 ± 0.3) × 107 g |
| W-B-2PbN | (2.4 ± 0.1) × 105 | (2.8 ± 0.1) × 107 b | (2,9 ± 0.2) × 107 g | (1.3 ± 0.3) × 108 ef | (2.5 ± 0.2) × 107 g |
| G1-Y-3Na | (2.3 ± 0.2) × 105 | (3.4 ± 0.2) × 107 b | (7.1 ± 0.3) × 108 c | (1.7 ± 0.2) × 109 b | (6.1 ± 0.3) × 108 b |
| G1-Y-1N | (2.1 ± 0.1) × 105 | (2.9 ± 0.2) × 107 b | (3.4 ± 0.2) × 108 f | (4.9 ± 0.3) × 108 d | (1.3 ± 0.1) × 108 f |
| G1-Y-2PbN | (2.2 ± 0.2) × 105 | (3.0 ± 0.3) × 107 b | (6.6 ± 0.3) × 108 d | (8.8 ± 0.2) × 108 c | (3.2 ± 0.2) × 108 d |
| G1-B-3Na | (2.6 ± 0.2) × 105 | (3.0 ± 0.3) × 107 b | (4.7 ± 0.3) × 107 g | (5.4 ± 0.2) × 108 d | (3.0 ± 0.3) × 107 g |
| G1-B-1N | (2.4 ± 0.2) × 105 | (2.6 ± 0.3) × 107 b | (1.4 ± 0.3) × 107 g | (8.8 ± 0.3) × 107 ef | (1.0 ± 0.5) × 107 g |
| G1-B-2PbN | (2.4 ± 0.1) × 105 | (2.7 ± 0.4) × 107 b | (3.2 ± 0.4) × 107 g | (1.2 ± 0.2) × 108 ef | (1.7 ± 0.3) × 107 g |
| Dec-W | (2.5 ± 0.2) × 105 | (8.4 ± 0.3) × 105 c | (8.4 ± 0.2) × 106 g | (8.2 ± 0.3) × 106 f | (6.7 ± 0.6) × 106 g |
| Dec-G1 | (2.3 ± 0.2) × 105 | (7.7 ± 0.2) × 105 c | (6.8 ± 0.1) × 106 g | (7.6 ± 0.2) × 106 f | (4.5 ± 0.3) × 106 g |
| No Carbon source | (2.1 ± 0.3) × 105 | (2.6 ± 0.2) × 105 c | (3.8 ± 0.2) × 106 g | (4.7 ± 0.3) × 106 f | (1.3 ± 0.2) × 106 g |
| Glucose | (2.2 ± 0.1) × 105 | (3.5 ± 0.2) × 108 a | (4.3 ± 0.2) × 108 e | (3.8 ± 0.3) × 108 de | (2.5 ± 0.2) × 108 e |
| S | ns | *** | *** | ** | *** |
| L. plantarum L12 | 0h | 12h | 24h | 30h | 48h |
|---|---|---|---|---|---|
| CFU/mL | CFU/mL | CFU/mL | CFU/mL | CFU/mL | |
| W-Y-3Na | (1.8 ± 0.1) × 105 a | (3.4 ± 0.2) × 108 c | (4.7 ± 0.3) × 109 d | (4.2 ± 0.3) × 109 c | (2.3 ± 0.2) × 109 c |
| W-Y-1-N | (1.5 ± 0.2) × 105 a | (1.5 ± 0.2) × 108 e | (3.8 ± 0.2) × 109 e | (3.5 ± 0.2) × 109 d | (1.5 ± 0.4) × 109 b |
| W-Y-2PbN | (1.6 ± 0.2) × 105 a | (2.4 ± 0.2) × 108 d | (3.4 ± 0.4) × 109 e | (3.4 ± 0.2) × 109 d | (1.9 ± 0.2) × 109 cd |
| W-B-3Na | (1.7 ± 0.2) × 105 a | (8.4 ± 0.2) × 107 f | (5.4 ± 0.3) × 108 fg | (3.7 ± 0.2) × 108 ef | (2.8 ± 0.3) × 108 e |
| W-B-1N | (1.4 ± 0.2) × 105 a | (7.5 ± 0.2) × 107 f | (3.5 ± 0.3) × 108 fgh | (1.4 ± 0.2) × 108 ef | (1.8 ± 0.2) × 108 e |
| W-B-2PbN | (1.7 ± 0.2) × 105 a | (7.8 ± 0.6) × 107 f | (4.6 ± 0.2) × 108 fgh | (1.9 ± 0.2) × 108 ef | (2.1 ± 0.4) × 108 e |
| G1-Y-3Na | (1.6 ± 0.2) × 105 a | (4.3 ± 0.2) × 108 b | (7.7 ± 0.4) × 109 b | (6.4 ± 0.3) × 109 b | (4.2 ± 0.2) × 109 a |
| G1-Y-1N | (1.3 ± 0.2) × 105 a | (3.6 ± 0.2) × 108 c | (6.5 ± 0.3) × 109 h | (6.3 ± 0.6) × 109 b | (3.3 ± 0.1) × 109 b |
| G1-Y-2PbN | (1.7 ± 0.2) × 105 a | (2.3 ± 0.4) × 108 d | (7.1 ± 0.3) × 109 b | (6.8 ± 0.2) × 109 b | (4.4 ± 0.4) × 109 a |
| G1-B-3Na | (1.5 ± 0.2) × 105 a | (7.8 ± 0.2) × 107 f | (5.8 ± 0.2) × 108 f | (5.1 ± 0.3) × 108 e | (4.6 ± 0.3) × 108 e |
| G1-B-1N | (1.8 ± 0.2) × 105 a | (4.8 ± 0.2) × 107 fg | (2.6 ± 0.3) × 108 fgh | (2.1 ± 0.3) × 108 ef | (1.8 ± 0.3) × 108 e |
| G1-B-2PbN | (1.9 ± 0.2) × 105 a | (6.6 ± 0.4) × 107 f | (5.9 ± 0.3) × 108 f | (5.7 ± 0.3) × 108 e | (2.7 ± 0.3) × 108 e |
| Dec-W | (1.7 ± 0.2) × 105 a | (8.4 ± 0.3) × 105 g | (8.4 ± 0.4) × 106 gh | (8.2 ± 0.2) × 106 f | (6.7 ± 0.1) × 106 e |
| Dec-G1 | (1.6 ± 0.2) × 105 a | (8.9 ± 0.2) × 105 g | (8.9 ± 0.2) × 106 gh | (1.5 ± 0.2) × 107 f | (2.4 ± 0.2) × 107 e |
| No Carbon source | (1.8 ± 0.2) × 105 a | (6.5 ± 0.1) × 105 g | (5.7 ± 0.2) × 106 b | (8.6 ± 0.3) × 106 f | (1.4 ± 0.3) × 107 e |
| Glucose | (1.4 ± 0.2) × 105 a | (8.5 ± 0.2) × 108 a | (8.3 ± 0.2) × 109 a | (7.8 ± 0.2) × 109 a | (4.5 ± 0.3) × 108 e |
| S | ns | *** | ** | *** | *** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




