Submitted:
04 January 2026
Posted:
07 January 2026
You are already at the latest version
Abstract
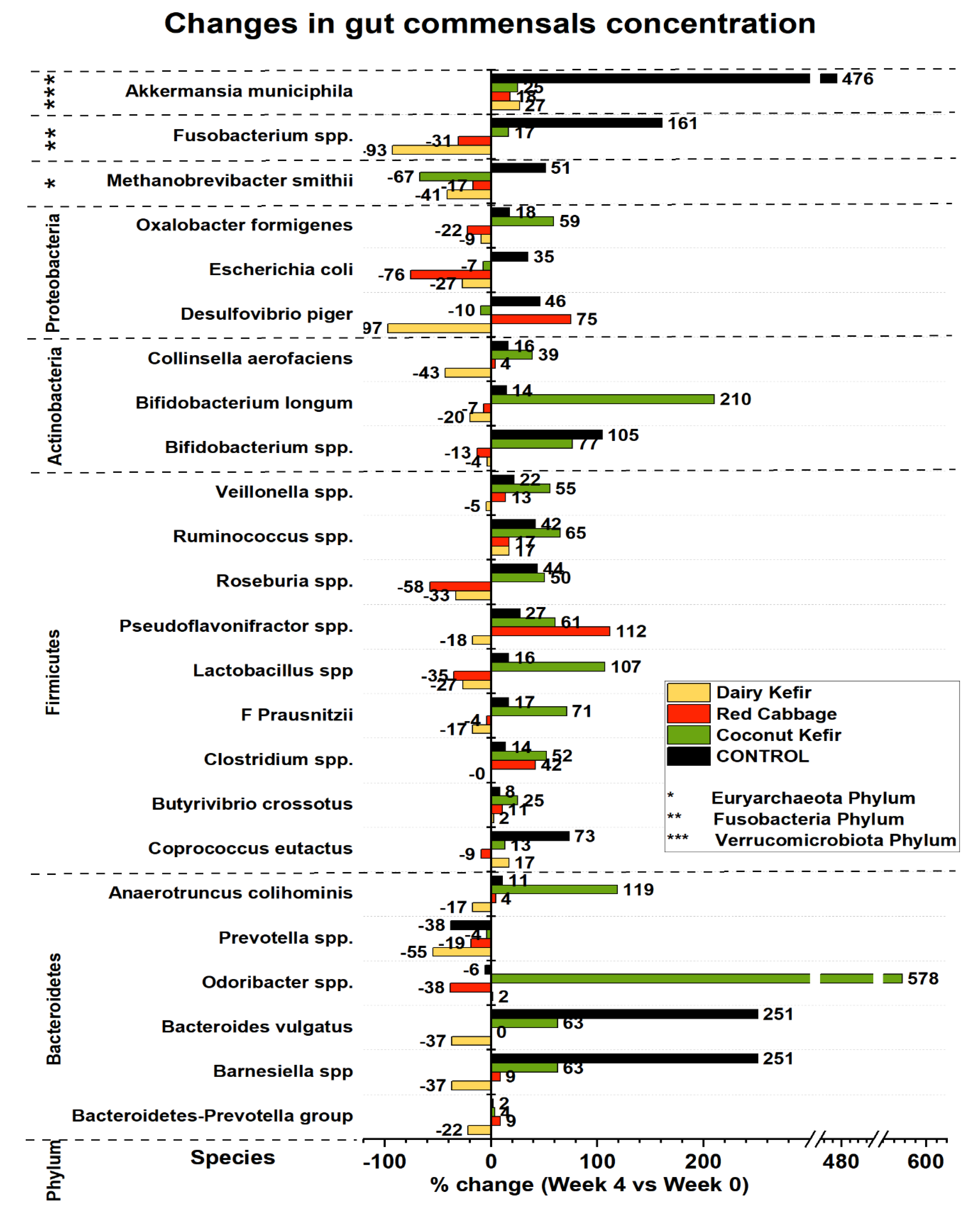
Fermented foods are increasingly recognized for their potential to support gut and brain health via microbiome modulation. However, most research focuses on isolated probiotics or lab-prepared products, leaving limited evidence for real-world fermented foods with live bacteria. This study evaluated the effects of three commercially available fermented foods—dairy kefir, coconut kefir, and fermented red cabbage and beetroot—on gastrointestinal, cognitive, and emotional outcomes in healthy adults. Over a 4-week randomized controlled intervention, cognitive function was assessed using the CANTAB, emotional health via validated self-report measures, and stool samples analysed using the Genova Diagnostics GI Effects test. Dairy kefir improved decision-making, sustained attention, working memory, reduced depression, anxiety and stress. The coconut kefir reduced waiting impulsivity, enhanced short-term memory, improved total mood, and increased butyrate-associated commensals, Faecalibacterium prausnitzii, Bifidobacterium spp., Lactobacillus spp., and Anaerotruncus colihominis, alongside elevated butyrate levels. The fermented red cabbage and beetroot improved sustained attention, working memory, reduced stress, improved total mood, and increased both butyrate and propionate. In contrast, the control group showed a rise in Fusobacterium spp. These findings support fermented foods as functional dietary interventions for gut–brain health.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Participants
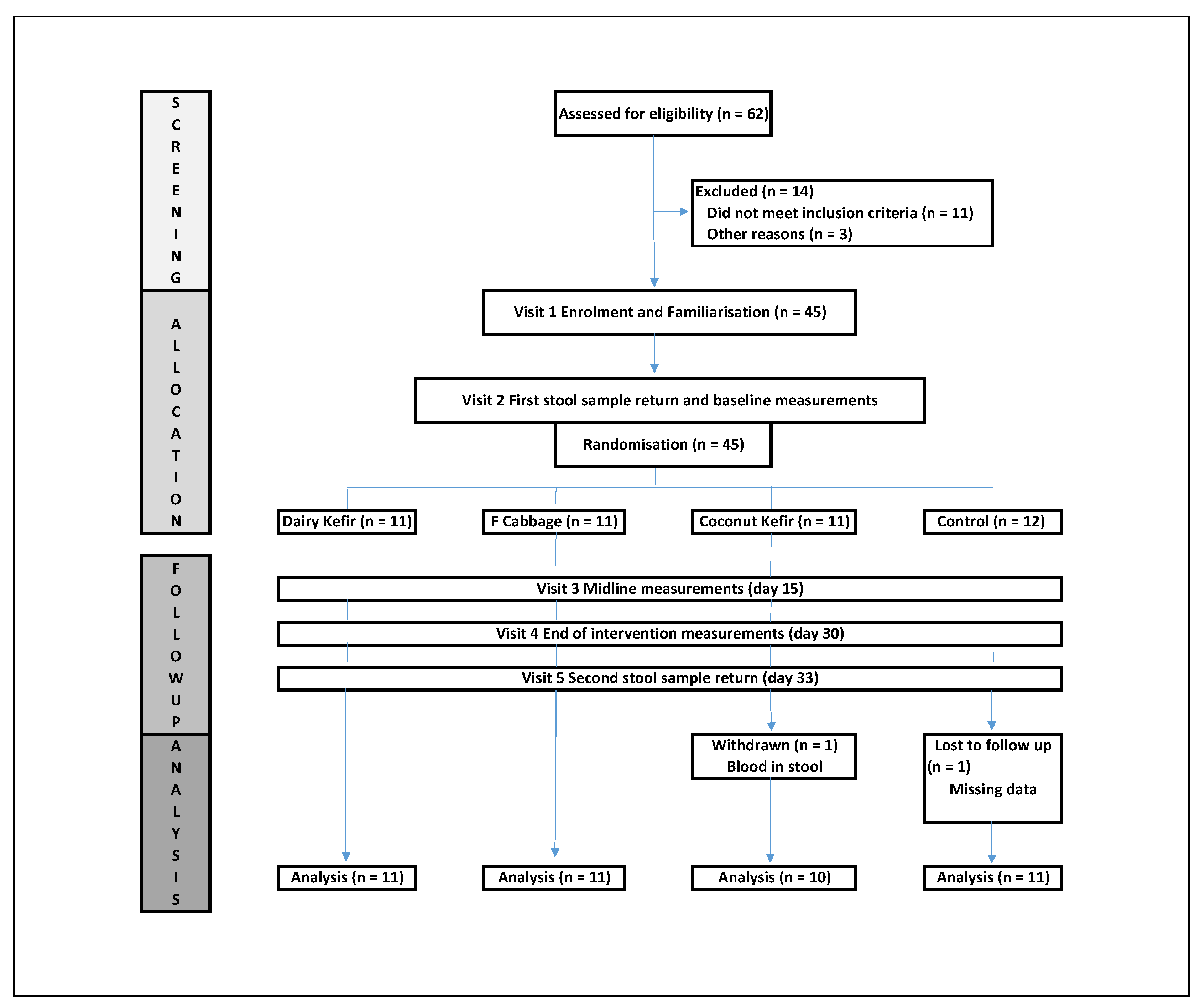
2.1.1. Recruitment Procedure
2.1.2. Randomisation and Group-Allocation Procedure
2.1.3. Intervention Products
2.1.4. Study Design and Procedure
2.1.5. Data Collection
2.1.5.1. MYMOP®
2.1.5.2. Cognitive Function
2.1.5.3. Emotional Health
2.1.5.4. Gastrointestinal Health
2.2. Data Analyses
2.2.1. MYMOP®
2.2.2. Cognitive Function
2.2.3. Emotional Health
2.2.4. GI Health
2.2.4.1. Inflammatory and Immune Biomarkers
2.2.4.2. Gastro-Intestinal Metabolites
2.2.4.3. Commensal Bacteria
2.2.4.4. Bristol Stool Scale
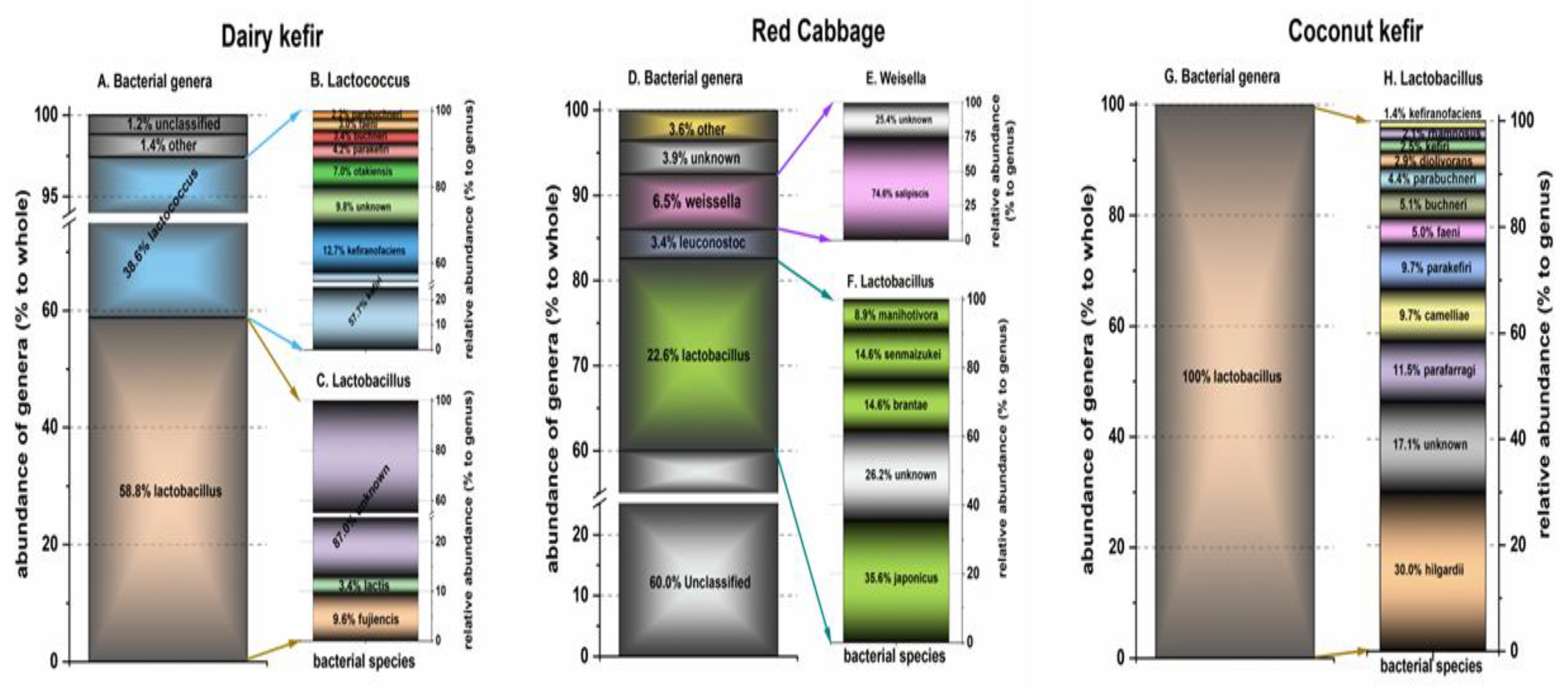
2.2.5. Intervention Products’ Metagenomics
2.3. Statistical Analyses
3. Results
3.1. Participant Group Comparisons
3.2. MYMOP ®
3.3. Cognitive Function
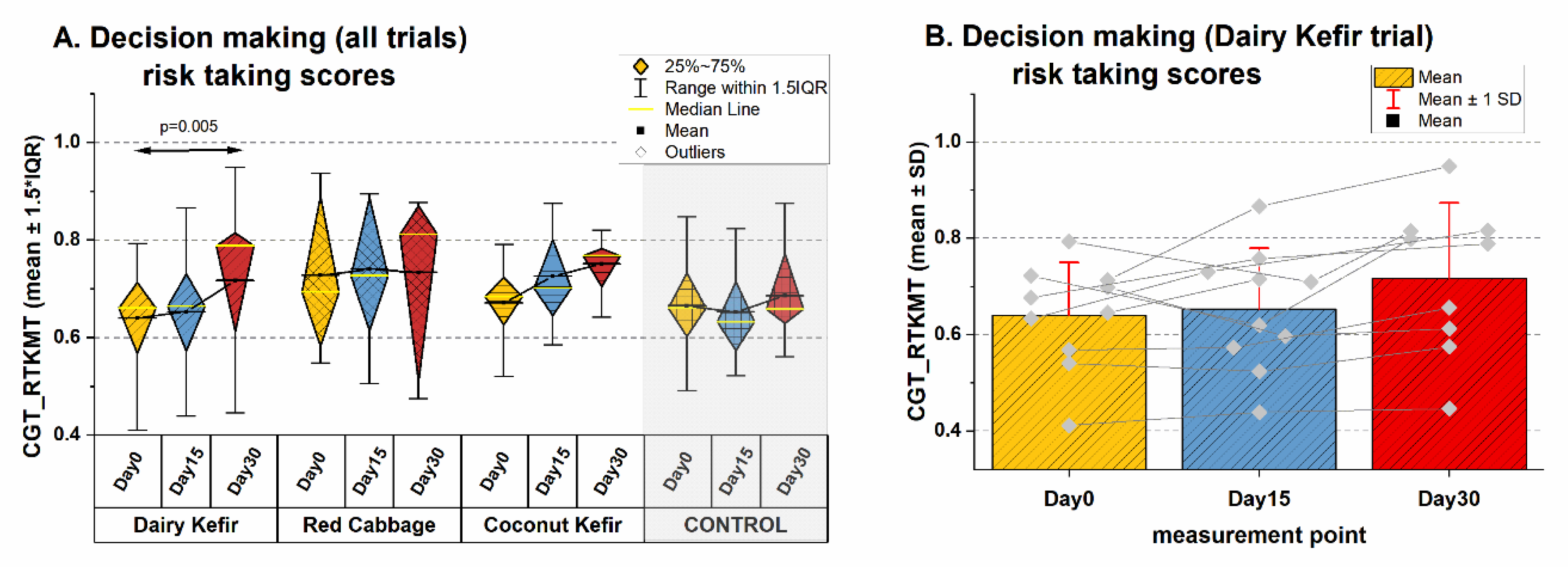
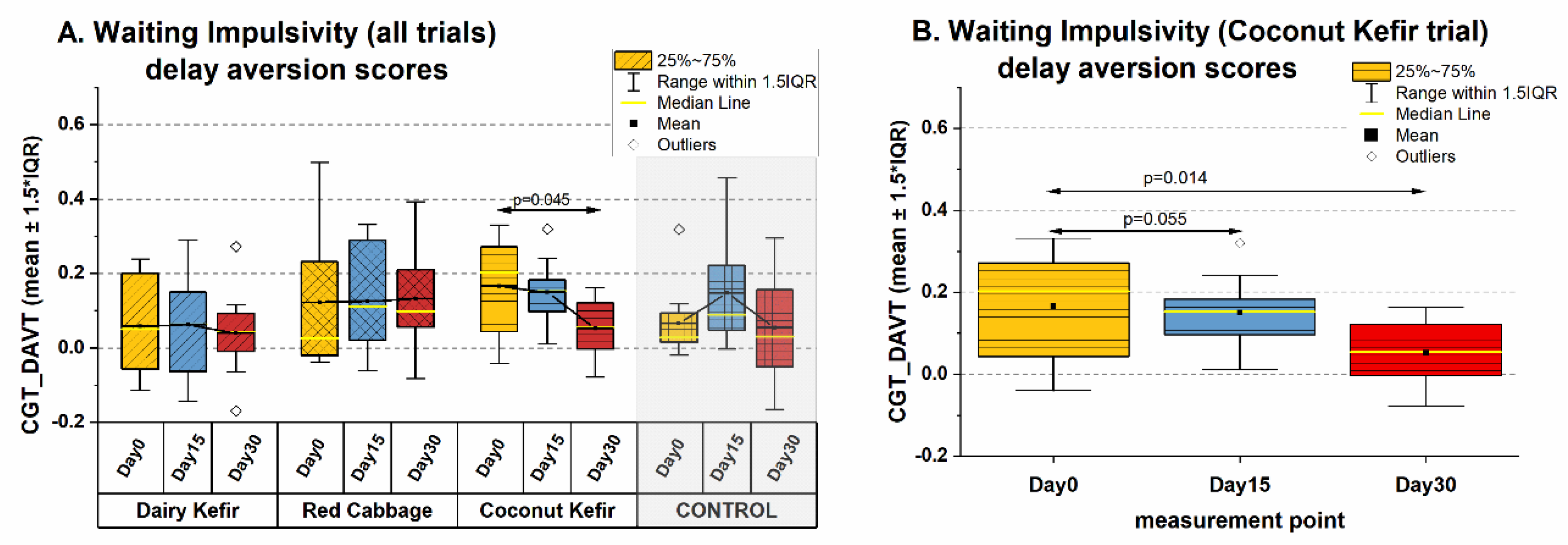
3.3.1. Effects on Decision Making and Impulsivity (Executive Function)
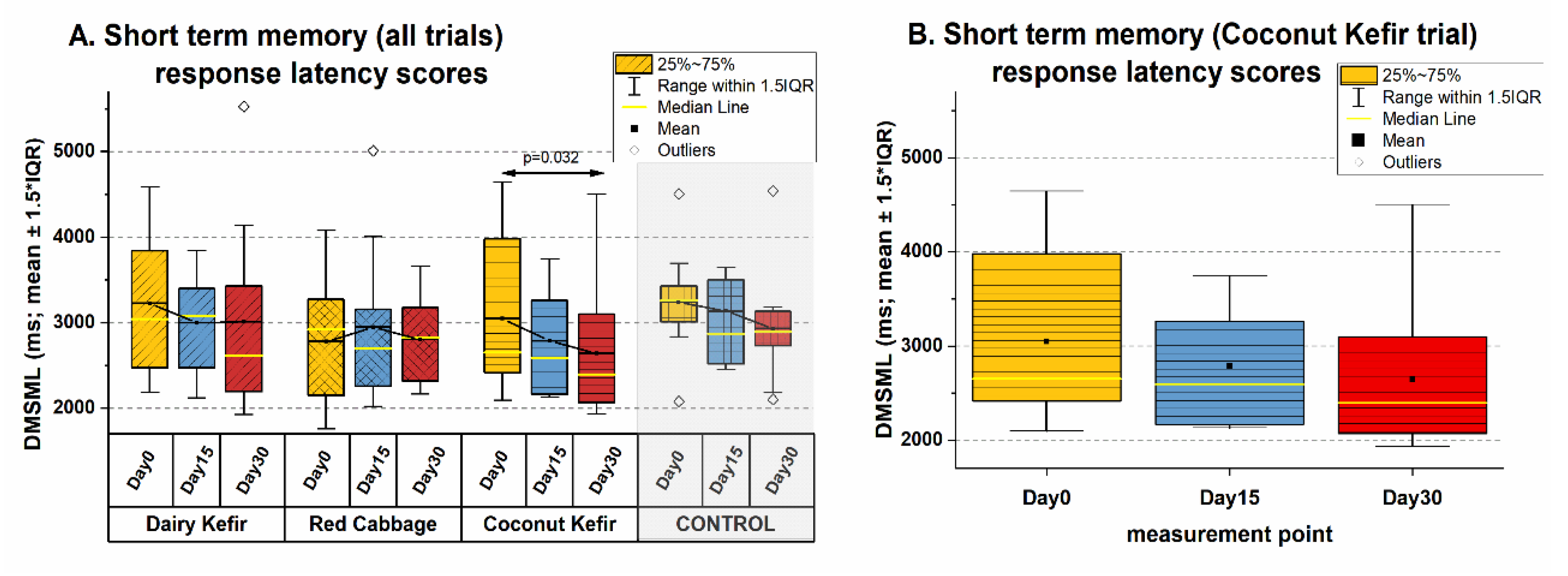
3.3.2. Effects on Short Term Memory
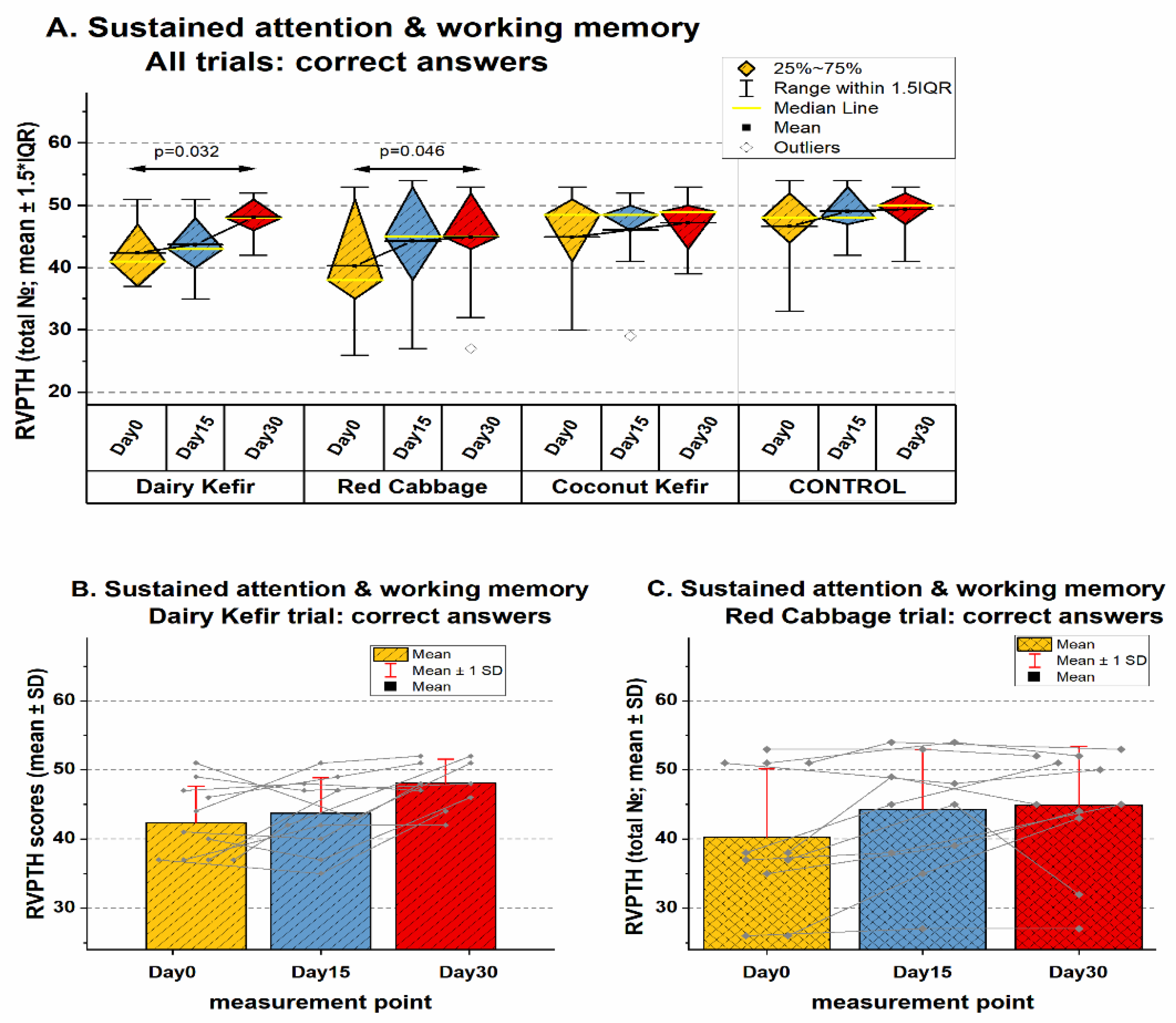
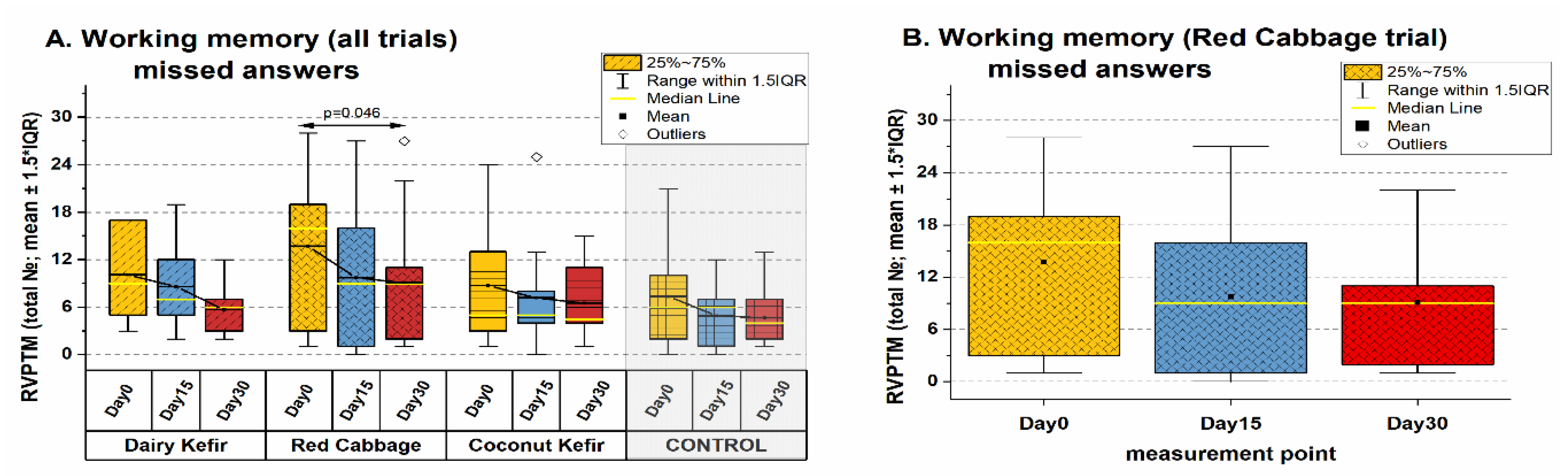
3.3.3. Effects on Sustained Attention and Working Memory
3.4. Emotional Health
3.4.1. POMS
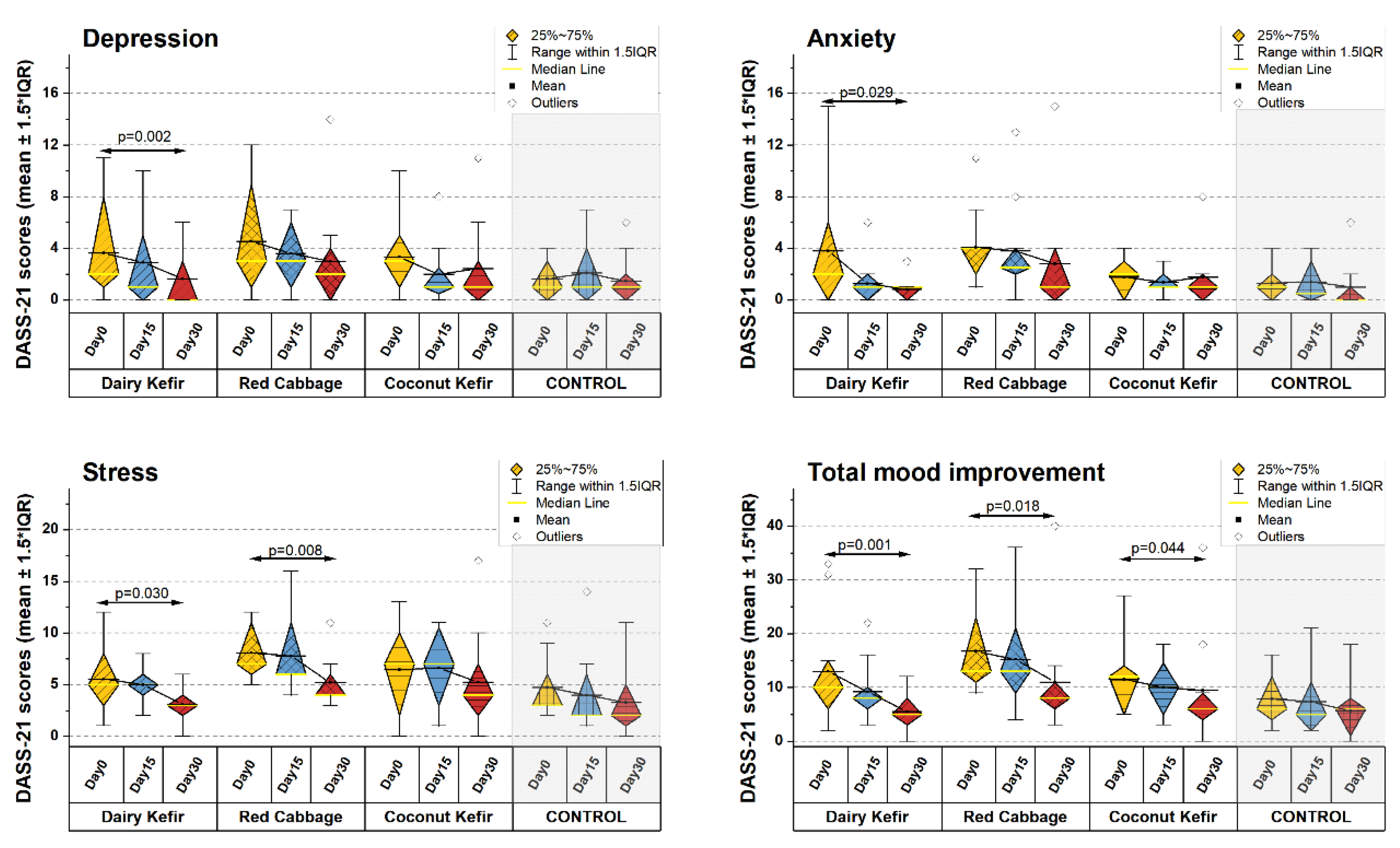
3.4.2. DASS21
3.5. Gastrointestinal Health
3.5.1. Bristol Stool Scale
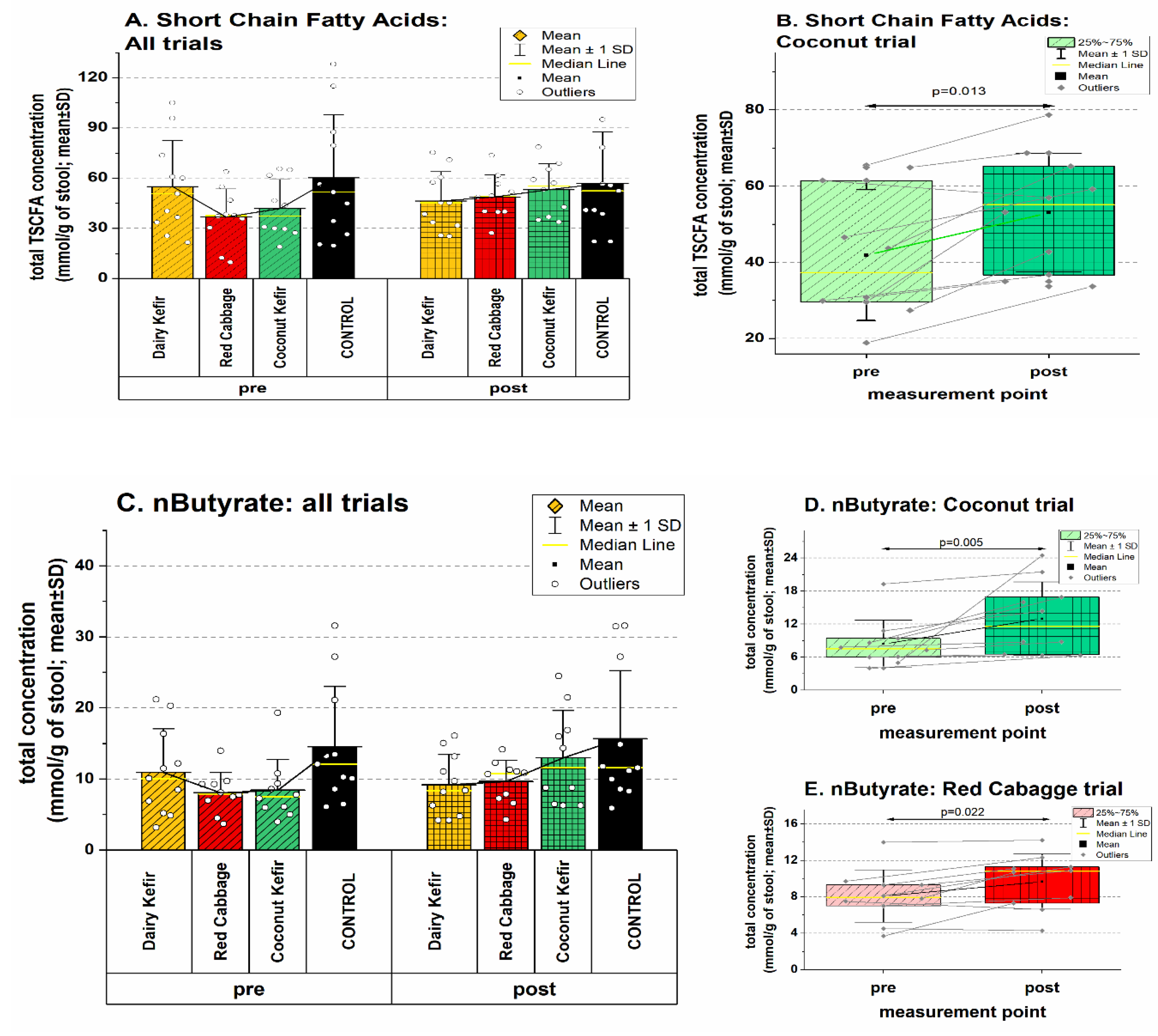
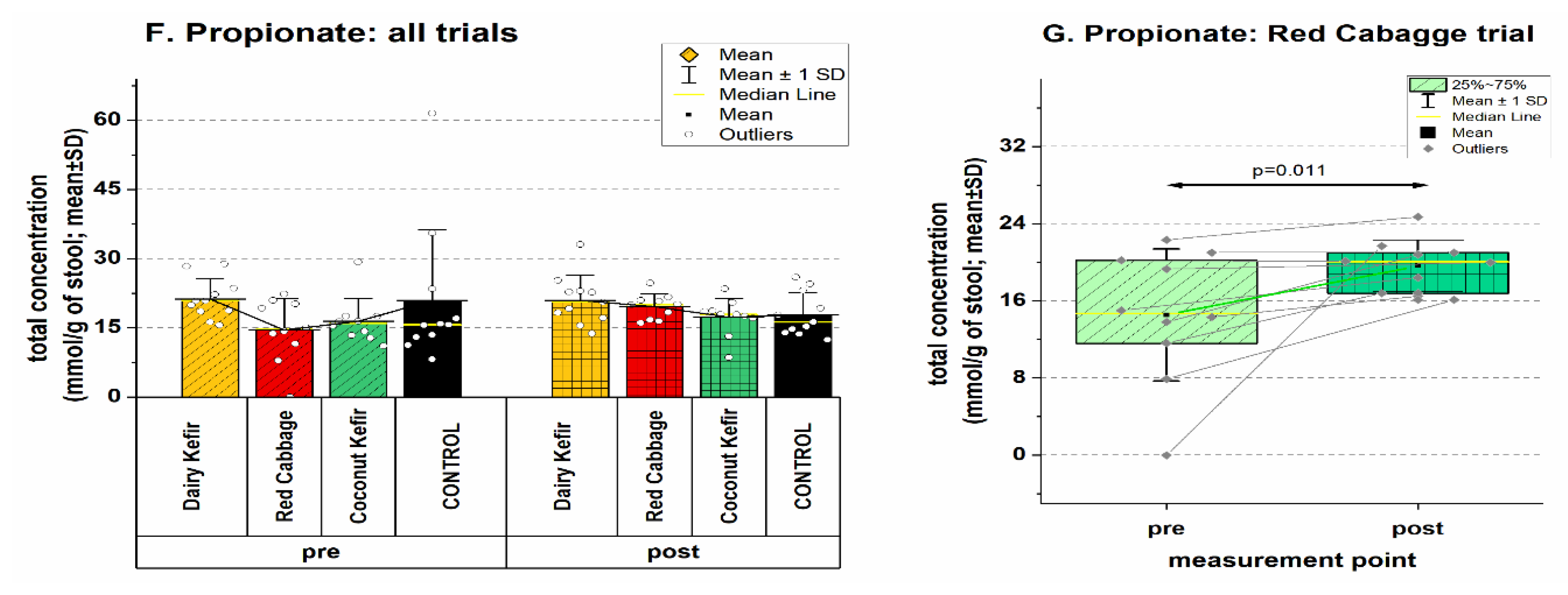
3.5.2. Effects on Short Chain Fatty Acids
3.5.3. Inflammation and Immunity
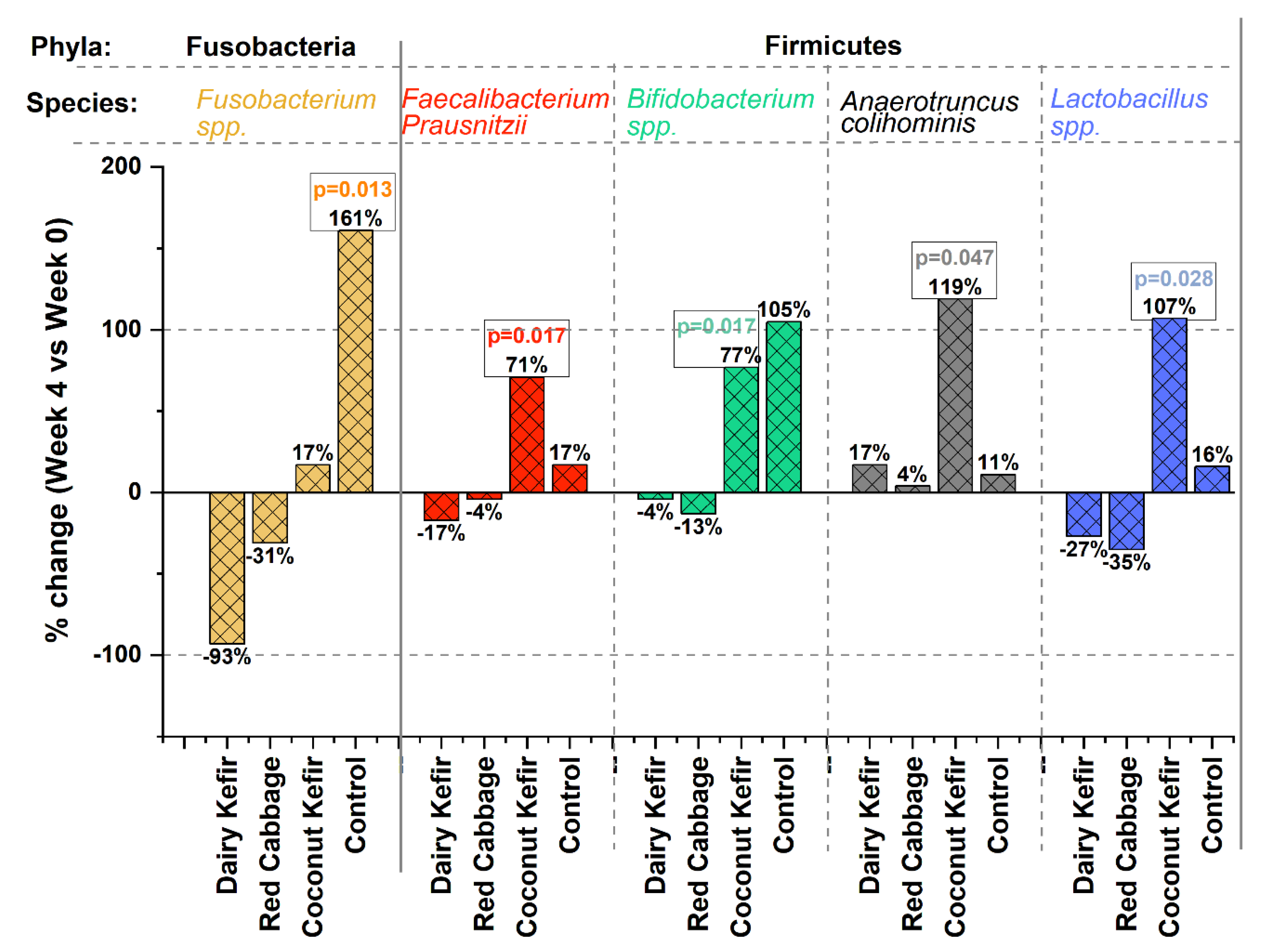
3.5.4. Effects on Gut Microbiota
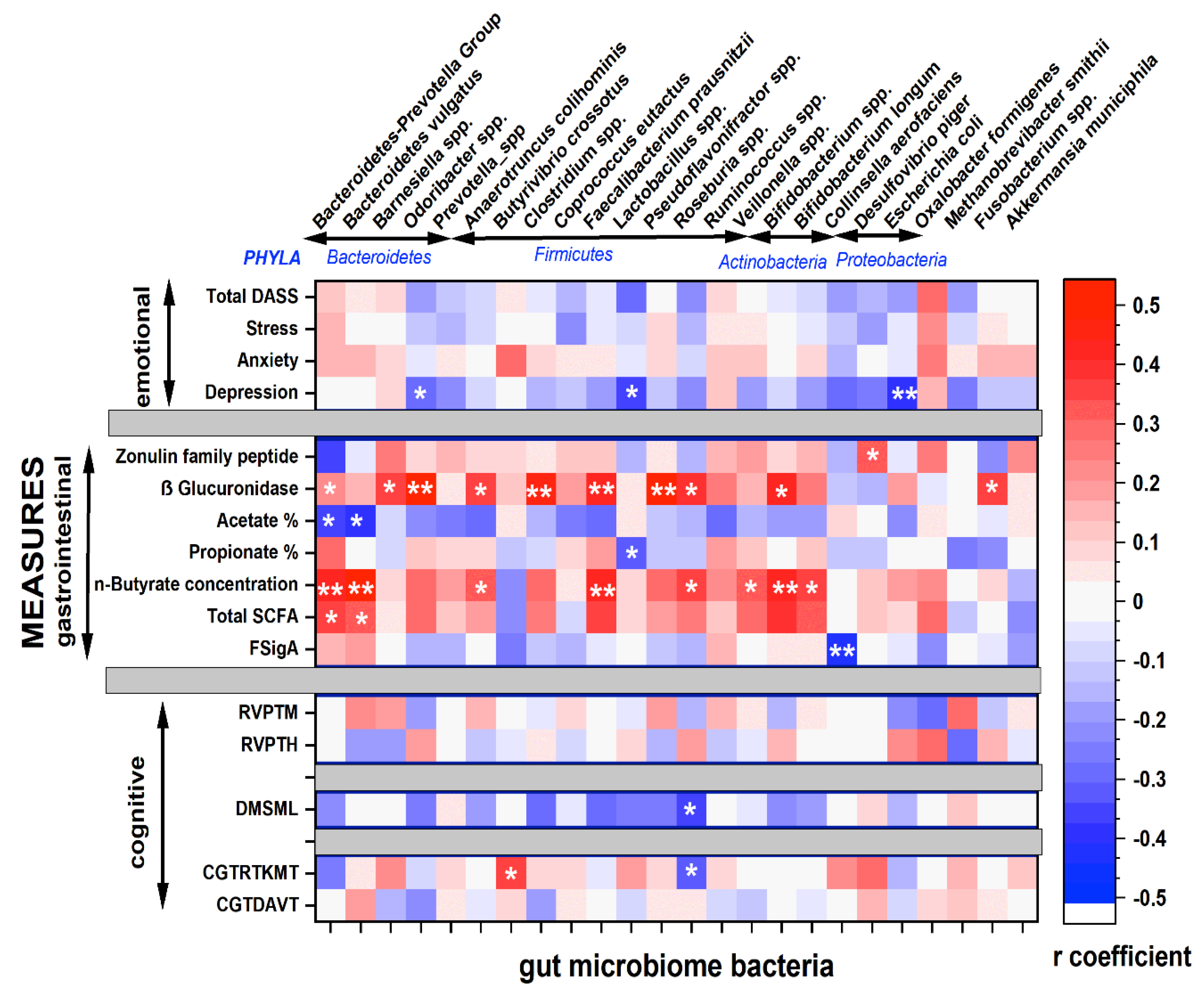
3.6. Correlation Analyses
3.6.1. Cognitive Function
3.6.2. Emotional Health
3.6.3. Gastro-Intestinal Health Biomarkers
3.7. Intervention Products
3.7.1. Dairy Kefir
3.7.2. Fermented Red Cabbage and Beetroot
3.7.3. Coconut Kefir
4. Discussion
4.1. Cognitive Function
4.2. Emotional Health
4.3. Gastrointestinal Health
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| 5HT | Serotonin |
| BBB | Blood–Brain Barrier |
| BDNF | Brain-Derived Neurotrophic Factor |
| CANTAB | Cambridge Neuropsychological Test Automated Battery |
| CAZymes | Carbohydrate-Active Enzymes |
| CG3 | Cyanidin-3-glucoside |
| CGT | Cambridge Gambling Task |
| CGTDAVT | Cambridge Gambling Task – Delay Aversion |
| CGTDMGMT | Cambridge Gambling Task – Decision-Making under Risk |
| CGTDMMT | Cambridge Gambling Task – Delay Match-to-Memory |
| CGTRTKMT | Cambridge Gambling Task – Risk-Taking Measure |
| CHO | Carbohydrate |
| CNS | Central Nervous System |
| DA | Dopamine |
| DASS | Depression Anxiety and Stress Scale |
| DMS | Delayed Matching to Sample |
| DMSPC | DMS – Percent Correct |
| DMSTC | DMS – Total Correct |
| EC | Enterochromaffin cells |
| EDN | Eosinophil-Derived Neurotoxin |
| EPX | Eosinophil Protein X |
| FFAR2 | Free Fatty Acid Receptor 2 |
| FFAR3 | Free Fatty Acid Receptor 3 |
| fSigA | Fecal Secretory Immunoglobulin A |
| GABA | Gamma-Aminobutyric Acid |
| GBA | Gut–Brain Axis |
| GI | Gastrointestinal |
| GPCR | G-Protein-Coupled Receptor |
| GPR41 | G-Protein-Coupled Receptor 41 |
| GPR43 | G-Protein-Coupled Receptor 43 |
| GUS | β-Glucuronidase |
| GUSome | Glucuronidase Enzyme System |
| HDAC | Histone Deacetylase |
| HPA | Hypothalamic–Pituitary–Adrenal Axis |
| ISAPP | International Scientific Association for Probiotics and Prebiotics |
| KYN | Kynurenine |
| KynA | Kynurenic Acid |
| LAB | Lactic Acid Bacteria |
| MCT | Medium chain triglycerides |
| MYMOP | Measure Yourself Medical Outcome Profile |
| PA | Proteolytic activity |
| PAZymes | Polyphenol Active Enzymes |
| POMS | Profile of Mood States |
| RVP | Rapid Visual Processing |
| RVPA | RVP – Omissions Accuracy |
| RVPML | RVP – Mean Latency |
| RVPTFA | RVP – Total False Alarms |
| RVPTH | RVP – Total Hits |
| SCFA | Short-Chain Fatty Acid |
| TDASS | Total Depression, Anxiety and Stress Score |
| TMD | Total Mood Disturbance |
| TRP | Tryptophan |
References
- Byun, M.; Yu, O.; Cha, Y.; Park, T. Korean traditional Chungkookjang improves body composition, lipid profiles and atherogenic indices in overweight/obese subjects: a double-blind, randomized, crossover, placebo-controlled clinical trial. European Journal of Clinical Nutrition 2016, 70(10), 1116–1122. [Google Scholar] [CrossRef]
- Lim, J; Jung, E; Choi, E; Jeong, D; Jo, S; Jin, J; et al. Supplementation with Aspergillus oryzae-fermented kochujang lowers serum cholesterol in subjects with hyperlipidemia. Clinical Nutrition 2015, 34(3), 383. [Google Scholar] [CrossRef] [PubMed]
- Tu, M; Chen, H; Tung, Y; Kao, C; Hu, F; Chen, C. Short-Term Effects of Kefir-Fermented Milk Consumption on Bone Mineral Density and Bone Metabolism in a Randomized Clinical Trial of Osteoporotic Patients. PLoS ONE 2015, 10(12). [Google Scholar] [CrossRef]
- Han, K; Bose, S; Wang, J; Kim, B; Kim, MJ; Kim, E; et al. Contrasting effects of fresh and fermented kimchi consumption on gut microbiota composition and gene expression related to metabolic syndrome in obese Korean women. Molecular Nutrition Food Res 2015, 59(5), 1004. [Google Scholar] [CrossRef] [PubMed]
- Özcan, H; Oskay, Ü; Bodur, AF. Effects of Kefir on Quality of Life and Sleep Disturbances in Postmenopausal Women. Holistic Nursing Practice 2019, 33(4), 207. [Google Scholar] [CrossRef]
- Pražnikar, ZJ; Kenig, S; Vardjan, T; Bizjak, MČ; Petelin, A. Effects of kefir or milk supplementation on zonulin in overweight subjects. Journal of Dairy Science 2020, 103(5), 3961. [Google Scholar] [CrossRef]
- Marsh, AJ; O’sullivan, O; Hill, C; Ross, RP; Cotter, PD. Sequencing-Based Analysis of the Bacterial and Fungal Composition of Kefir Grains and Milks from Multiple Sources. PLoS ONE 2013, 8(7). [Google Scholar] [CrossRef]
- Marco, ML; Heeney, D; Binda, S; Cifelli, CJ; Cotter, PD; Foligné, B; et al. Health benefits of fermented foods: microbiota and beyond. Current Opinion in Biotechnology 2017, 44, 94–102. [Google Scholar] [CrossRef]
- Peluzio, MDCG; Dias, MDME; Martinez, JA; Milagro, FI. Kefir and Intestinal Microbiota Modulation: Implications in Human Health. Front Nutr 2021, 8. [Google Scholar] [CrossRef]
- Melini, F; Melini, V; Luziatelli, F; Ficca, AG; Ruzzi, M. Health-Promoting Components in Fermented Foods: An Up-to-Date Systematic Review. Nutrients 2019, 11(5), 1189. [Google Scholar] [CrossRef] [PubMed]
- Stiemsma, LT; Nakamura, RE; Nguyen, JG; Michels, KB. Does Consumption of Fermented Foods Modify the Human Gut Microbiota? The Journal of Nutrition 2020, 150(7), 1680. [Google Scholar] [CrossRef]
- Dimidi, E; Cox, SR; Rossi, M; Whelan, K. Fermented Foods: Definitions and Characteristics, Impact on the Gut Microbiota and Effects on Gastrointestinal Health and Disease. Nutrients 2019, 11(8), 1806. [Google Scholar] [CrossRef]
- Leeuwendaal, NK; Stanton, C; O’toole, PW; Beresford, TP. Fermented Foods, Health and the Gut Microbiome. Nutrients 2022, 14(7). [Google Scholar] [CrossRef] [PubMed]
- Rul, F; Béra-Maillet, C; Champomier-Vergès, MC; El-Mecherfi, KE; Foligné, B; Michalski, MC; et al. Underlying evidence for the health benefits of fermented foods in humans. Food Funct 2022, 13(9), 4804. [Google Scholar] [CrossRef]
- Balasubramanian, R; Schneider, E; Gunnigle, E; Cotter, PD; Cryan, JF. Fermented foods: Harnessing their potential to modulate the microbiota-gut-brain axis for mental health. Neuroscience & Biobehavioral Reviews 2024, 158. [Google Scholar] [CrossRef]
- Bell, V; Ferrão, J; Pimentel, L; Pintado, M; Fernandes, T. One Health, Fermented Foods, and Gut Microbiota. Foods (Basel, Switzerland) 2018, 7(12), 195. [Google Scholar] [CrossRef] [PubMed]
- Marco, ML; Hill, C; Hutkins, R; Slavin, J; Tancredi, DJ; Merenstein, D; et al. Should There Be a Recommended Daily Intake of Microbes? The Journal of nutrition 2020, 150(12), 3061–3067. [Google Scholar] [CrossRef]
- Pedersen, MGB; Søndergaard, E; Nielsen, CB; Johannsen, M; Gormsen, LC; Møller, N; et al. Oral lactate slows gastric emptying and suppresses appetite in young males. Clinical Nutrition 2022, 41(2), 517. [Google Scholar] [CrossRef]
- Savaiano, DA; Hutkins, RW. Yogurt, cultured fermented milk, and health: a systematic review. Nutrition Reviews 2020, 79(5), 599. [Google Scholar] [CrossRef]
- Järbrink-Sehgal, E; Andreasson, A. The gut microbiota and mental health in adults. Current Opinion in Neurobiology 2020, 62, 102. [Google Scholar] [CrossRef]
- Casertano, M; Fogliano, V; Ercolini, D. Psychobiotics, gut microbiota and fermented foods can help preserving mental health. Food Research International 2021, 152. [Google Scholar] [CrossRef]
- Scott, SA; Fu, J; Chang, PV. Microbial tryptophan metabolites regulate gut barrier function via the aryl hydrocarbon receptor. Proc Natl Acad Sci USA 2020, 19376–19387. [Google Scholar] [CrossRef] [PubMed]
- Wei, L; Marco, ML. The fermented cabbage metabolome and its protection against cytokine-induced intestinal barrier disruption of Caco-2 monolayers. Appl Environ Microbiol 2025, 91(5). [Google Scholar] [CrossRef]
- Stachulski, AV; Knausenberger, NA; Shah, SN; Hoyles, L; Mcarthur, S. A host–gut microbial amino acid co-metabolite, p-cresol glucuronide, promotes blood–brain barrier integrity in vivo. Tissue Barriers 2022, 11(1). [Google Scholar] [CrossRef]
- Tan, Y; Zhong, H; Zhao, D; Du, H; Xu, Y. Succession rate of microbial community causes flavor difference in strong-aroma Baijiu making process. International Journal of Food Microbiology 2019, 311. [Google Scholar] [CrossRef] [PubMed]
- Leech, J; Cabrera-Rubio, R; Walsh, AM; Macori, G; Walsh, CJ; Barton, W; Finnegan, L; Crispie, F; O’Sullivan, O; Claesson, MJ; Cotter, PD. Fermented-Food Metagenomics Reveals Substrate-Associated Differences in Taxonomy and Health-Associated and Antibiotic Resistance Determinants. mSystems 2020, 5(6), e00522-20. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bell, V; Ferrão, J; Fernandes, T. Nutritional Guidelines and Fermented Food Frameworks. Foods (Basel, Switzerland) 2017, 6(8), 65. [Google Scholar] [CrossRef] [PubMed]
- Salminen, S; Van Loveren, H. Probiotics and prebiotics: health claim substantiation. Microbial Ecology in Health & Disease 2012, 23(0). [Google Scholar] [CrossRef]
- Campana, R; Van Hemert, S; Baffone, W. Strain-specific probiotic properties of lactic acid bacteria and their interference with human intestinal pathogens invasion. Gut Pathog 2017, 9(1). [Google Scholar] [CrossRef]
- Blasche, S; Kim, Y; Mars, RAT; Machado, D; Maansson, M; Kafkia, E; et al. Metabolic cooperation and spatiotemporal niche partitioning in a kefir microbial community. Nat Microbiol 2021, 6(2), 196. [Google Scholar] [CrossRef]
- Du, R.; Xiong, W.; Xu, L.; Xu, Y.; Wu, Q. Metagenomics reveals the habitat specificity of biosynthetic potential of secondary metabolites in global food fermentations. Microbiome 2023, 11, 115. [Google Scholar] [CrossRef] [PubMed]
- Lee, M; Choi, Y; Lee, H; Hwang, S; Lee, HJ; Park, SJ; et al. Influence of Salinity on the Microbial Community Composition and Metabolite Profile in Kimchi. Fermentation 2021, 7(4). [Google Scholar] [CrossRef]
- Yang, X; Hu, W; Xiu, Z; Jiang, A; Yang, X; Saren, G; et al. Microbial Community Dynamics and Metabolome Changes During Spontaneous Fermentation of Northeast Sauerkraut From Different Households. Front Microbiol 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Van Reckem, E; Geeraerts, W; Charmpi, C; Van Der Veken, D; De Vuyst, L; Leroy, F. Exploring the Link Between the Geographical Origin of European Fermented Foods and the Diversity of Their Bacterial Communities: The Case of Fermented Meats. Front Microbiol 2019, 10. [Google Scholar] [CrossRef]
- Genova Diagnostics. GI Effects® Stool Profiles. n.d. Available online: https://www.gdx.net/products/gi-effects.
- Sealed Envelope Ltd. Randomisation and online databases for clinical trials. [cited 2025 Oct 11]. n.d. Available online: https://www.sealedenvelope.com.
- Paterson, C. Measuring outcome in primary care: A patient-generated measure, MYMOP, compared to the SF-36 health survey. BMJ 1996, 312(7037), 1016–1020. [Google Scholar] [CrossRef]
- Cambridge Cognition. CANTAB® cognitive assessment software [Internet]. [cited 2025 Oct 11]. Available online: https://www.cambridgecognition.com.
- McNair, D. M.; Lorr, M.; Droppleman, L. F. Manual for the Profile of Mood States; Educational and Industrial Testing Service: San Diego, CA, 1971; Available online: https://api.semanticscholar.org/CorpusID:67926195.
- Lovibond, S. H.; Lovibond, P. F. Manual for the Depression Anxiety Stress Scales, 2nd ed.; Psychology Foundation of Australia: Sydney, 1995. [Google Scholar]
- Lewis, S. J.; Heaton, K. W. Stool form scale as a useful guide to intestinal transit time. Scandinavian Journal of Gastroenterology 1997, 32(9), 920–924. [Google Scholar] [CrossRef]
- Pathirana, WGW; Chubb, SP; Gillett, MJ; Vasikaran, SD. Faecal Calprotectin. Clin Biochem Rev. 2018, 39(3), 77–90. [Google Scholar] [PubMed] [PubMed Central]
- Siddiqui, I; Majid, H; Abid, S. Update on clinical and research application of fecal biomarkers for gastrointestinal diseases. WJGPT 2017, 8(1). [Google Scholar] [CrossRef]
- Yamamura, R; Nakamura, K; Kitada, N; Aizawa, T; Shimizu, Y; Nakamura, K; et al. Associations of gut microbiota, dietary intake, and serum short-chain fatty acids with fecal short-chain fatty acids. Biosci Microbiota Food Health 1 2019, 39(1), 11–17. [Google Scholar] [CrossRef]
- Banasiewicz, T; Domagalska, D; Borycka-Kiciak, K; Rydzewska, G. Determination of butyric acid dosage based on clinical and experimental studies - a literature review. Prz Gastroenterol 2020, 15(2), 119–125. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Farup, PG; Rudi, K; Hestad, K. Faecal short-chain fatty acids - a diagnostic biomarker for irritable bowel syndrome? BMC Gastroenterol 2016, 16(1), 51. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ching, Jian; Sonja, Kanerva; Sami, Qadri; Hannele, Yki-Järvinen; Anne, Salonen. In vitro Effects of Bacterial Exposure on Secretion of Zonulin Family Peptides and Their Detection in Human Tissue Samples. In Frontiers in Microbiology; 2022; Volume 13. [Google Scholar] [CrossRef]
- González-Orozco, Brianda D.; García-Cano, Israel; Jiménez-Flores, Rafael; Alvárez, Valente B. Milk kefir microbiota—Direct and indirect antimicrobial effects. Journal of Dairy Science 2022, 105(5), 3707–3715. [Google Scholar] [CrossRef]
- Mohajeri, MH; Wittwer, J; Vargas, K; Hogan, E; Holmes, A; Rogers, PJ; et al. Chronic treatment with a tryptophan-rich protein hydrolysate improves emotional processing, mental energy levels and reaction time in middle-aged women. Br J Nutr 2015, 113(2), 350. [Google Scholar] [CrossRef] [PubMed]
- Ton, AMM; Campagnaro, BP; Alves, GA; Aires, R; Côco, LZ; Arpini, CM; et al. Oxidative Stress and Dementia in Alzheimer’s Patients: Effects of Synbiotic Supplementation. Oxidative Medicine and Cellular Longevity 2020, 1. [Google Scholar] [CrossRef]
- Van De Wouw, M; Walsh, AM; Crispie, F; Van Leuven, L; Lyte, JM; Boehme, M; et al. Distinct actions of the fermented beverage kefir on host behaviour, immunity and microbiome gut-brain modules in the mouse. Microbiome 2020, 8(1). [Google Scholar] [CrossRef] [PubMed]
- Braga, JD; Thongngam, M; Kumrungsee, T. Gamma-aminobutyric acid as a potential postbiotic mediator in the gut–brain axis. npj Sci Food 2024, 8(1). [Google Scholar] [CrossRef]
- Boonstra, E; de Kleijn, R; Colzato, LS; Alkemade, A; Forstmann, BU; Nieuwenhuis, S. Neurotransmitters as food supplements: the effects of GABA on brain and behavior. Front Psychol 2015, 6, 1520. [Google Scholar] [CrossRef]
- Noori, N; Bangash, M; Motaghinejad, M; Hosseini, P; Noudoost, B. Kefir protective effects against nicotine cessation-induced anxiety and cognition impairments in rats. Adv Biomed Res 2014, 3(1). [Google Scholar] [CrossRef]
- Anwar, M; Ali, OS; Rahsed, LA; Badawi, A. The effect of using kefir grains and mesenchymal stem cells in LPS-induced Alzheimer’s disease neuroinflammatory model. Neurobiologia 2019, 10((25). [Google Scholar] [CrossRef]
- Farag, MA; Jomaa, SA; Abd El-Wahed, A.R.; El-Seedi, H. The Many Faces of Kefir Fermented Dairy Products: Quality Characteristics, Flavour Chemistry, Nutritional Value, Health Benefits, and Safety. Nutrients 2020, 12(2). [Google Scholar] [CrossRef]
- Yılmaz, C; Gökmen, V. Determination of tryptophan derivatives in kynurenine pathway in fermented foods using liquid chromatography tandem mass spectrometry. Food Chemistry 2017, 243, 420. [Google Scholar] [CrossRef]
- Liutkevičius, A; Šarkinas, A. Studies on the growth conditions and composition of kefir grains – as a food and forage biomass. Vet Zootec 2004, 25, 64–70. [Google Scholar]
- Jenkins, TA; Nguyen, JCD; Polglaze, KE; Bertrand, PP. Influence of Tryptophan and Serotonin on Mood and Cognition with a Possible Role of the Gut-Brain Axis. Nutrients 2016, 8(1). [Google Scholar] [CrossRef]
- Pratt, O.E. Kinetics of Tryptophan Transport Across the Blood-Brain Barrier. In Transport Mechanisms of Tryptophan in Blood Cells, Nerve Cells, and at the Blood-Brain Barrier. Journal of Neural Transmission; Baumann, P., Ed.; Springer: Vienna, 1979; Volume 15. [Google Scholar] [CrossRef]
- Mobini, S; Chiang, T; Ho, M; Bradshaw, CM; Szabadi, E. Effects of central 5-hydroxytryptamine depletion on sensitivity to delayed and probabilistic reinforcement. Psychopharmacology 2000, 152(4), 390. [Google Scholar] [CrossRef]
- Rogers, RD; Everitt, BJ; Baldacchino, A; Blackshaw, AJ; Swainson, R; Wynne, K; et al. Dissociable Deficits in the Decision-Making Cognition of Chronic Amphetamine Abusers, Opiate Abusers, Patients with Focal Damage to Prefrontal Cortex, and Tryptophan-Depleted Normal Volunteers: Evidence for Monoaminergic Mechanisms. Neuropsychopharmacology 1999. [Google Scholar] [CrossRef] [PubMed]
- Walderhaug, E; Magnusson, A; Neumeister, A; Lappalainen, J; Lunde, H; Refsum, H; et al. Interactive Effects of Sex and 5-HTTLPR on Mood and Impulsivity During Tryptophan Depletion in Healthy People. Biological Psychiatry 2007, 62(6), 593. [Google Scholar] [CrossRef] [PubMed]
- Sambeth, A; Riedel, WJ; Tillie, DE; Blokland, A; Postma, A; Schmitt, J. Memory impairments in humans after acute tryptophan depletion using a novel gelatin-based protein drink. J Psychopharmacol 2008, 23(1), 56. [Google Scholar] [CrossRef] [PubMed]
- Haider, S; Khaliq, S; Haleem, DJ. Enhanced serotonergic neurotransmission in the hippocampus following tryptophan administration improves learning acquisition and memory consolidation in rats. Pharmacological Report 2007, 59, 53–57. [Google Scholar]
- Suzuki, H; Yamashiro, D; Ogawa, S; Kobayashi, M; Cho, D; Iizuka, A; et al. Intake of Seven Essential Amino Acids Improves Cognitive Function and Psychological and Social Function in Middle-Aged and Older Adults: A Double-Blind, Randomized, Placebo-Controlled Trial. Front Nutr 2020, 7. [Google Scholar] [CrossRef]
- Juhasz, G; Downey, D; Hinvest, N; Thomas, E; Chase, D; Toth, ZG; et al. Risk-Taking Behavior in a Gambling Task Associated with Variations in the Tryptophan Hydroxylase 2 Gene: Relevance to Psychiatric Disorders. Neuropsychopharmacol 2009, 35(5), 1109. [Google Scholar] [CrossRef]
- Murphy, SE; Longhitano, CRE; Ayres, RE; Cowen, PJ; Harmer, CJ. Tryptophan supplementation induces a positive bias in the processing of emotional material in healthy female volunteers. Psychopharmacology 2006, 187(1), 121. [Google Scholar] [CrossRef] [PubMed]
- Ostapiuk, A; Urbanska, EM. Kynurenic acid in neurodegenerative disorders-unique neuroprotection or double-edged sword? CNS Neurosci Ther 2022, 28(1), 19–35. [Google Scholar] [CrossRef]
- Young, KD; Drevets, WC; Dantzer, R; Teague, TK; Bodurka, J; Savitz, J. Kynurenine pathway metabolites are associated with hippocampal activity during autobiographical memory recall in patients with depression. Brain, Behavior, and Immunity 2017, 56, 335. [Google Scholar] [CrossRef] [PubMed]
- Hartai, Z; Juhász, A; Rimanóczy, Á; Janáky, T; Donkó, T; Dux, L; et al. Decreased serum and red blood cell kynurenic acid levels in Alzheimer’s disease. Neurochemistry International 2006, 50(2), 308. [Google Scholar] [CrossRef] [PubMed]
- Martos, D; Tuka, B; Tanaka, M; Vécsei, L; Telegdy, G. Memory Enhancement with Kynurenic Acid and Its Mechanisms in Neurotransmission. Biomedicines 2022, 10(4). [Google Scholar] [CrossRef]
- Turski, M.P.; Turska, M.; Zgrajka, W.; et al. Presence of kynurenic acid in food and honeybee products. Amino Acids 2009, 36, 75–80. [Google Scholar] [CrossRef]
- Dalley, JW; Roiser, JP. Dopamine, serotonin and impulsivity. Neuroscience 2012, 215, 42. [Google Scholar] [CrossRef]
- Neufang, S; Akhrif, A; Herrmann, CG; Drepper, C; Homola, GA; Nowak, J; et al. Serotonergic modulation of ‘waiting impulsivity’ is mediated by the impulsivity phenotype in humans. Transl Psychiatry 2016, 6(11), e940. [Google Scholar] [CrossRef]
- Hayes, DJ; Jupp, B; Sawiak, SJ; Merlo, E; Caprioli, D; Dalley, JW. Brain γ-aminobutyric acid: a neglected role in impulsivity. Eur J of Neuroscience 2014, 39(11), 1921. [Google Scholar] [CrossRef]
- Prehn-Kristensen, A; Zimmermann, A; Tittmann, L; Lieb, W; Schreiber, S; Baving, L; et al. Reduced microbiome alpha diversity in young patients with ADHD. PLoS ONE 2018, 13(7). [Google Scholar] [CrossRef]
- Kayser, AS; Allen, DC; Navarro-Cebrian, A; Mitchell, JM; Fields, HL. Dopamine, Corticostriatal Connectivity, and Intertemporal Choice. Journal of Neuroscience 2012, 32(27), 9402. [Google Scholar] [CrossRef]
- Fujihara, K; Narita, K; Suzuki, Y; Takei, Y; Suda, M; Tagawa, M; et al. Relationship of γ-aminobutyric acid and glutamate + glutamine concentrations in the perigenual anterior cingulate cortex with performance of Cambridge Gambling Task. NeuroImage 2015, 109, 102. [Google Scholar] [CrossRef]
- Coray, R; Quednow, BB. The role of serotonin in declarative memory: A systematic review of animal and human research. Neuroscience & Biobehavioral Reviews 2022, 139. [Google Scholar] [CrossRef]
- Švob Štrac, D.; Pivac, N.; Mück-Šeler, D. The serotonergic system and cognitive function. Translational Neuroscience 2016, 7(1), 35–49. [Google Scholar] [CrossRef]
- Gasbarri, A.; Pompili, A. 3 - The Role of GABA in Memory Processes, in: Identification of Neural Markers Accompanying Memory; Elsevier Inc., 2014; pp. 47–62. [Google Scholar] [CrossRef]
- Shohamy, D; Adcock, RA. Dopamine and adaptive memory. Trends in Cognitive Sciences 2010, 14(10), 464. [Google Scholar] [CrossRef]
- Moigradean, Diana; Poiana, Mariana-Atena; Alda, Liana-Maria; Gogoasa, Ioan. Quantitative identification of fatty acids from walnut and coconut oils using GC-MS method. Journal of Agroalimentary Processes and Technologies 2013, 19, 459–463. [Google Scholar]
- Lal, S; Kirkup, AJ; Brunsden, AM; Thompson, DG; Grundy, D. Vagal afferent responses to fatty acids of different chain length in the rat. Am J Physiol Gastrointest Liver Physiol 2001, 281(4), G907–G915. [Google Scholar] [CrossRef] [PubMed]
- Han, W; Tellez, LA; Perkins, MH; Perez, IO; Qu, T; Ferreira, J; et al. A Neural Circuit for Gut-Induced Reward. Cell 2018, 175(3), 665. [Google Scholar] [CrossRef] [PubMed]
- O’mahony, SM; Clarke, G; Borre, YE; Dinan, TG; Cryan, JF. Serotonin, tryptophan metabolism and the brain-gut-microbiome axis. Behavioural Brain Research 2014, 277, 32. [Google Scholar] [CrossRef] [PubMed]
- Boonchooduang, N; Louthrenoo, O; Chattipakorn, N; Chattipakorn, SC. Possible links between gut–microbiota and attention-deficit/hyperactivity disorders in children and adolescents. Eur J Nutr 2020, 59(8), 3391. [Google Scholar] [CrossRef] [PubMed]
- Cryan, JF; Dinan, TG. Mind-altering microorganisms: the impact of the gut microbiota on brain and behaviour. Nat Rev Neurosci 2012, 13(10), 701. [Google Scholar] [CrossRef]
- Srivastav, S.; Neupane, S.; Bhurtel, S.; Katila, N.; Maharjan, S.; Choi, H.; Hong, J. T.; Choi, D. Y. Probiotics mixture increases butyrate, and subsequently rescues the nigral dopaminergic neurons from MPTP and rotenone-induced neurotoxicity. Journal of Nutritional Biochemistry 2019, 69, 73–86. [Google Scholar] [CrossRef]
- Hamamah, S; Aghazarian, A; Nazaryan, A; Hajnal, A; Covasa, M. Role of Microbiota-Gut-Brain Axis in Regulating Dopaminergic Signaling. Biomedicines 2022, 10(2). [Google Scholar] [CrossRef] [PubMed]
- Hata, T; Asano, Y; Yoshihara, K; Kimura-Todani, T; Miyata, N; Zhang, X; et al. Regulation of gut luminal serotonin by commensal microbiota in mice. PLoS ONE 2017, 12(7). [Google Scholar] [CrossRef]
- Chang, F; Siuti, P; Laurent, S; Williams, T; Glassey, E; Sailer, AW; et al. Gut-inhabiting Clostridia build human GPCR ligands by conjugating neurotransmitters with diet- and human-derived fatty acids. Nat Microbiol 2021, 6(6), 792. [Google Scholar] [CrossRef] [PubMed]
- Schlatterer, K; Peschel, A; Kretschmer, D. Short-Chain Fatty Acid and FFAR2 Activation – A New Option for Treating Infections? Front Cell Infect Microbiol 2021, 11. [Google Scholar] [CrossRef]
- Cui, Y; Miao, K; Niyaphorn, S; Qu, X. Production of Gamma-Aminobutyric Acid from Lactic Acid Bacteria: A Systematic Review. IJMS 2020, 21(3). [Google Scholar] [CrossRef] [PubMed]
- Kalkan, AE; Binmowyna, MN; Raposo, A; Ahmad, MF; Ahmed, F; Otayf, AY; et al. Beyond the Gut: Unveiling Butyrate’s Global Health Impact Through Gut Health and Dysbiosis-Related Conditions: A Narrative Review. Nutrients 2025, 17(8). [Google Scholar] [CrossRef]
- Asano, Y; Hiramoto, T; Nishino, R; Aiba, Y; Kimura, T; Yoshihara, K; Koga, Y; Sudo, N. Critical role of gut microbiota in the production of biologically active, free catecholamines in the gut lumen of mice. Am J Physiol Gastrointest Liver Physiol;Epub 2012, 303(11), G1288–G1295. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Li, E.; Sun, Z.; Fu, D.; Duan, Y.; Li, H. Altered gut microbiota and short chain fatty acids in Chinese children with autism spectrum disorder. Scientific Reports 2020, 10, 10156. [Google Scholar] [CrossRef]
- Roopashree, PG; Shetty, SS; Suchetha Kumari, N. Effect of medium chain fatty acid in human health and disease. Journal of Functional Foods 2021, 87. [Google Scholar] [CrossRef]
- Ota, M; Matsuo, J; Ishida, I; Hattori, K; Teraishi, T; Tonouchi, H; et al. Effect of a ketogenic meal on cognitive function in elderly adults: potential for cognitive enhancement. Psychopharmacology 2016, 233(21-22), 3797. [Google Scholar] [CrossRef]
- Courchesne-Loyer, Alexandre; Fortier, Mélanie; Tremblay-Mercier, Jennifer; Chouinard-Watkins, Raphaël; Roy, Maggie; Nugent, Scott; Castellano, Christian-Alexandre; Cunnane, Stephen C. Stimulation of mild, sustained ketonemia by medium-chain triacylglycerols in healthy humans: Estimated potential contribution to brain energy metabolism. Nutrition 2013, 29(4), 635–640. [Google Scholar] [CrossRef] [PubMed]
- Hughes, SD; Kanabus, M; Anderson, G; Hargreaves, IP; Rutherford, T; Donnell, MO; et al. The ketogenic diet component decanoic acid increases mitochondrial citrate synthase and complex I activity in neuronal cells. Journal of Neurochemistry 2014, 129(3), 426. [Google Scholar] [CrossRef]
- Ota, M; Matsuo, J; Ishida, I; Takano, H; Yokoi, Y; Hori, H; et al. Effects of a medium-chain triglyceride-based ketogenic formula on cognitive function in patients with mild-to-moderate Alzheimer’s disease. Neuroscience Letters 2018, 690, 232. [Google Scholar] [CrossRef] [PubMed]
- Ashton, Jake S.; Roberts, James W.; Wakefield, Caroline J.; Page, Richard M.; MacLaren, Don P.M.; Marwood, Simon; Malone, James J. The effects of medium chain triglyceride (MCT) supplementation using a C8:C10 ratio of 30:70 on cognitive performance in healthy young adults. Physiology & Behavior 229(2021), 113252. [CrossRef]
- Aziz, Abdul; Aziz, Ramlan; Sarmidi, Muhammad; Suean, Chua; Annuar, Nur; Noor, Norhayati; Nor, Rorfarahiyah. Fermented coconut milk and coconut oil. In Handbook of Plant-Based Fermented Food and Beverage Technology, 2nd ed.; Hui, Y.H., Evranuz, E.Ö., Eds.; CRC Press, 2012; pp. 665–675. [Google Scholar] [CrossRef]
- Gardener, SL; Rainey-Smith, SR; Weinborn, M; Bondonno, CP; Martins, RN. Intake of Products Containing Anthocyanins, Flavanols, and Flavanones, and Cognitive Function: A Narrative Review. Front Aging Neurosci 2021, 13. [Google Scholar] [CrossRef]
- Willis, LM; Shukitt-Hale, B; Joseph, JA. Recent advances in berry supplementation and age-related cognitive decline. Current Opinion in Clinical Nutrition & Metabolic Care 2009, 12(1), 91. [Google Scholar]
- Ockermann, P; Headley, L; Lizio, R; Hansmann, J. A Review of the Properties of Anthocyanins and Their Influence on Factors Affecting Cardiometabolic and Cognitive Health. Nutrients 2021, 13(8). [Google Scholar] [CrossRef] [PubMed]
- Krikorian, R; Nash, TA; Shidler, MD; Shukitt-Hale, B; Joseph, JA. Concord grape juice supplementation improves memory function in older adults with mild cognitive impairment. Br J Nutr 2009, 103(5), 730. [Google Scholar] [CrossRef]
- Lamport, DJ; Lawton, CL; Merat, N; Jamson, H; Myrissa, K; Hofman, D; et al. Concord grape juice, cognitive function, and driving performance: a 12-wk, placebo-controlled, randomized crossover trial in mothers of preteen children. The American Journal of Clinical Nutrition 2016, 103(3), 775. [Google Scholar] [CrossRef]
- Haskell-Ramsay, CF; Stuart, RC; Okello, EJ; Watson, AW. Cognitive and mood improvements following acute supplementation with purple grape juice in healthy young adults. Eur J Nutr 2017, 56(8), 2621. [Google Scholar] [CrossRef] [PubMed]
- Bowtell, JL; Aboo-Bakkar, Z; Conway, ME; Adlam, AR; Fulford, J. Enhanced task-related brain activation and resting perfusion in healthy older adults after chronic blueberry supplementation. Appl Physiol Nutr Metab 2017, 42(7), 773. [Google Scholar] [CrossRef] [PubMed]
- Andres-Lacueva, C; Shukitt-Hale, B; Galli, RL; Jauregui, O; Lamuela-Raventos, RM; Joseph, JA. Anthocyanins in aged blueberry-fed rats are found centrally and may enhance memory. Nutr Neurosci 2005, 8(2), 111–120. [Google Scholar] [CrossRef] [PubMed]
- Wiczkowski, W; Szawara-Nowak, D; Romaszko, J. The impact of red cabbage fermentation on bioavailability of anthocyanins and antioxidant capacity of human plasma. Food Chemistry 2015, 190, 730. [Google Scholar] [CrossRef]
- Shukitt-Hale, Barbara; Carey, Amanda; Simon, Laura; Mark, David A.; Joseph, James A. Effects of Concord grape juice on cognitive and motor deficits in aging. Nutrition 2006, 22, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Tayab, MA; Islam, MN; Chowdhury, KAA; Tasnim, FM. Targeting neuroinflammation by polyphenols: A promising therapeutic approach against inflammation-associated depression. Biomedicine & Pharmacotherapy 2022, 147. [Google Scholar] [CrossRef]
- Romo-Araiza, A; Gutiérrez-Salmeán, G; Galván, EJ; Hernández-Frausto, M; Herrera-López, G; Romo-Parra, H; et al. Probiotics and Prebiotics as a Therapeutic Strategy to Improve Memory in a Model of Middle-Aged Rats. Front Aging Neurosci 2018, 10. [Google Scholar] [CrossRef]
- Liu, P.; Zou, L.B.; Wang, L.H.; Wu, Y.F. Exercise improves memory impairment in a rat model of Alzheimer’s disease by increasing brain-derived neurotrophic factor expression. Neural Regeneration Research 2015, 10(10), 1615–1620. [Google Scholar] [CrossRef]
- Ryan, SM; Nolan, YM. Neuroinflammation negatively affects adult hippocampal neurogenesis and cognition: can exercise compensate? Neuroscience & Biobehavioral Reviews 2015, 61, 121. [Google Scholar] [CrossRef]
- Grande, I; Fries, GR; Kunz, M; Kapczinski, F. The Role of BDNF as a Mediator of Neuroplasticity in Bipolar Disorder. Psychiatry Investig 2010, 7(4). [Google Scholar] [CrossRef] [PubMed]
- Pang, PT; Lu, B. Regulation of late-phase LTP and long-term memory in normal and aging hippocampus: role of secreted proteins tPA and BDNF. Ageing Research Reviews 2004, 3(4), 407. [Google Scholar] [CrossRef]
- Skelly, DT; Griffin, ÉW; Murray, CL; Harney, S; O’boyle, C; Hennessy, E; et al. Acute transient cognitive dysfunction and acute brain injury induced by systemic inflammation occur by dissociable IL-1-dependent mechanisms. Mol Psychiatry 2018. [Google Scholar] [CrossRef]
- Canipe, LG; Sioda, M; Cheatham, CL. Diversity of the gut-microbiome related to cognitive behavioral outcomes in healthy older adults. Archives of Gerontology and Geriatrics 2021, 96. [Google Scholar] [CrossRef]
- Cunha, C; Brambilla, R; Thomas, KL; Kind, PC. A simple role for BDNF in learning and memory? Front Mol Neurosci 2010. [Google Scholar] [CrossRef] [PubMed]
- Heyck, M; Ibarra, A. Microbiota and memory: A symbiotic therapy to counter cognitive decline? Brain Circulation 2019, 5(3), 124–129. [Google Scholar] [CrossRef]
- Miranda, M; Morici, JF; Zanoni, MB; Bekinschtein, P. Brain-Derived Neurotrophic Factor: A Key Molecule for Memory in the Healthy and the Pathological Brain. Front Cell Neurosci 2019, 13. [Google Scholar] [CrossRef] [PubMed]
- Nieto, RR; Carrasco, A; Corral, S; Castillo, R; Gaspar, PA; Bustamante, ML; et al. BDNF as a Biomarker of Cognition in Schizophrenia/Psychosis: An Updated Review. Front Psychiatry 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Firuzi, O; Moosavi, F; Hosseini, R; Saso, L. Modulation of neurotrophic signaling pathways by polyphenols. DDDT 2015, 12. [Google Scholar] [CrossRef]
- Reyes-Izquierdo, T; Nemzer, B; Shu, C; Huynh, L; Argumedo, R; Keller, R; et al. Modulatory effect of coffee fruit extract on plasma levels of brain-derived neurotrophic factor in healthy subjects. Br J Nutr 2013, 110(3), 420. [Google Scholar] [CrossRef]
- Stilling, RM; Van De Wouw, M; Clarke, G; Stanton, C; Dinan, TG; Cryan, JF. The neuropharmacology of butyrate: The bread and butter of the microbiota-gut-brain axis? Neurochemistry International 2016, 99, 110. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Dong, J.; Cheng, Y.; Jiang, G. Propionate ameliorates diabetes-induced neurological dysfunction through regulating the PI3K/Akt/eNOS signaling pathway. bioRxiv 2021. [Google Scholar] [CrossRef]
- Pražnikar, ZJ; Kenig, S; Vardjan, T; Bizjak, MČ; Petelin, A. Effects of kefir or milk supplementation on zonulin in overweight subjects. Journal of Dairy Science 2020, 103(5), 3961. [Google Scholar] [CrossRef]
- Sun, Y; Geng, W; Pan, Y; Wang, J; Xiao, P; Wang, Y. Supplementation with Lactobacillus kefiranofaciens ZW3 from Tibetan Kefir improves depression-like behavior in stressed mice by modulating the gut microbiota. Food Funct 2019, 10(2), 925. [Google Scholar] [CrossRef]
- Sivamaruthi, BS; Kesika, P; Chaiyasut, C. Impact of Fermented Foods on Human Cognitive Function—A Review of Outcome of Clinical Trials. Sci Pharm 2018, 86(2). [Google Scholar] [CrossRef]
- Sahab, NRM; Subroto, E; Balia, RL; Utama, GL. γ-Aminobutyric acid found in fermented foods and beverages: current trends. Heliyon 2020, 6(11). [Google Scholar] [CrossRef] [PubMed]
- Xia, Y; Zha, M; Feng, C; Li, Y; Chen, Y; Shuang, Q. Effect of a co-fermentation system with high-GABA-yielding strains on soymilk properties: microbiological, physicochemical, and aromatic characterisations. Food Chemistry 2023, 423. [Google Scholar] [CrossRef]
- Hurtado-Romero, A; Del Toro-Barbosa, M; Gradilla-Hernández, MS; Garcia-Amezquita, LE; García-Cayuela, T. Probiotic Properties, Prebiotic Fermentability, and GABA-Producing Capacity of Microorganisms Isolated from Mexican Milk Kefir Grains: A Clustering Evaluation for Functional Dairy Food Applications. Foods 2021, 10(10). [Google Scholar] [CrossRef] [PubMed]
- Galli, V; Venturi, M; Mari, E; Guerrini, S; Granchi, L. Gamma-aminobutyric acid (GABA) production in fermented milk by lactic acid bacteria isolated from spontaneous raw milk fermentation. International Dairy Journal 2021, 127. [Google Scholar] [CrossRef]
- Hepsomali, P; Groeger, JA; Nishihira, J; Scholey, A. Effects of Oral Gamma-Aminobutyric Acid (GABA) Administration on Stress and Sleep in Humans: A Systematic Review. Front Neurosci 2020, 14. [Google Scholar] [CrossRef]
- Tillisch, K; Labus, J; Kilpatrick, L; Jiang, Z; Stains, J; Ebrat, B; et al. Consumption of Fermented Milk Product With Probiotic Modulates Brain Activity. Gastroenterology 2013, 144(7), 1394. [Google Scholar] [CrossRef] [PubMed]
- Moses, S.; Deeseenthum, S. Properties and benefits of kefir – A review. Songklanakarin Journal of Science and Technology 2015, 37(3), 275–282. [Google Scholar] [CrossRef]
- Diniz, RO; Garla, LK; Schneedorf, JM; Carvalho, JCT. Study of anti-inflammatory activity of Tibetan mushroom, a symbiotic culture of bacteria and fungi encapsulated into a polysaccharide matrix. Pharmacological Research 47(6), 563–568. [CrossRef] [PubMed]
- Rodrigues, KL; Caputo, LRG; Carvalho, JCT; Evangelista, J; Schneedorf, JM. Antimicrobial and healing activity of kefir and kefiran extract. International Journal of Antimicrobial Agents 2005, 25(5), 404. [Google Scholar] [CrossRef]
- Kim, D; Jeong, D; Kim, H; Kang, I; Chon, J; Song, K; et al. Antimicrobial Activity of Kefir against Various Food Pathogens and Spoilage Bacteria. Korean Journal for Food Science of Animal Resources 2016, 36(6), 787. [Google Scholar] [CrossRef]
- Gamba, RR; Yamamoto, S; Abdel-Hamid, M; Sasaki, T; Michihata, T; Koyanagi, T; et al. Chemical, Microbiological, and Functional Characterization of Kefir Produced from Cow’s Milk and Soy Milk. International Journal of Microbiology 2020, 1. [Google Scholar] [CrossRef]
- Demir, H. Comparison of traditional and commercial kefir microorganism compositions and inhibitory effects on certain pathogens. International Journal of Food Properties 2020, 23(1), 375. [Google Scholar] [CrossRef]
- Kim, D; Jeong, D; Kim, H; Seo, K. Modern perspectives on the health benefits of kefir in next generation sequencing era: Improvement of the host gut microbiota. Critical Reviews in Food Science and Nutrition 2018, 59(11), 1782. [Google Scholar] [CrossRef]
- Valles-Colomer, M.; Falony, G.; Darzi, Y.; Tigchelaar, E.F.; Wang, J.; Tito, R.Y.; Schiweck, C.; Kurilshikov, A.; Joossens, M.; Wijmenga, C.; Claes, S.; Van Oudenhove, L.; Zhernakova, A.; Vieira-Silva, S.; Raes, J. The neuroactive potential of the human gut microbiota in quality of life and depression. Nature Microbiology 2019, 4, 623–632. [Google Scholar] [CrossRef]
- Zheng, P; Zeng, B; Zhou, C; Liu, M; Fang, Z; Xu, X; et al. Gut microbiome remodeling induces depressive-like behaviors through a pathway mediated by the host’s metabolism. Mol Psychiatry 2016, 21(6), 786. [Google Scholar] [CrossRef]
- Westfall, S; Pasinetti, GM. The Gut Microbiota Links Dietary Polyphenols With Management of Psychiatric Mood Disorders. Front Neurosci 2019, 13. [Google Scholar] [CrossRef]
- Rong, H; Xie, X; Zhao, J; Lai, W; Wang, M; Xu, D; et al. Similarly in depression, nuances of gut microbiota: Evidences from a shotgun metagenomics sequencing study on major depressive disorder versus bipolar disorder with current major depressive episode patients. Journal of Psychiatric Research 2019, 113, 90. [Google Scholar] [CrossRef]
- Hopkins Medicine, J; Goes, FS. The Importance of Anxiety States in Bipolar Disorder. Curr Psychiatry Rep 2015, 17(2). [Google Scholar] [CrossRef]
- Li, W.; Ren, M.; Duo, L.; Li, J.; Wang, S.; Sun, Y.; Li, M.; Ren, W.; Hou, Q.; Yu, J.; Sun, Z.; Sun, T. Fermentation characteristics of Lactococcus lactis subsp. lactis isolated from naturally fermented dairy products and screening of potential starter isolates. Frontiers in Microbiology 2020, 11, 1794. [Google Scholar] [CrossRef] [PubMed]
- Santos, A; San, M; Sanchez, A; Torres, JM; Marquina, D. The Antimicrobial Properties of Different Strains of Lactobacillus spp. Isolated from Kefir. Systematic and Applied Microbiology 26(3), 434–437. [CrossRef] [PubMed]
- Bali, V; Panesar, PS; Bera, MB; Kennedy, JF. Bacteriocins: Recent Trends and Potential Applications. Critical Reviews in Food Science and Nutrition 2016, 56(5), 817. [Google Scholar] [CrossRef]
- Zhang, T; Zhang, Y; Li, L; Jiang, X; Chen, Z; Zhao, F; et al. Biosynthesis and Production of Class II Bacteriocins of Food-Associated Lactic Acid Bacteria. Fermentation 2022, 8(5). [Google Scholar] [CrossRef]
- Yüksekdağ, ZN; Beyatli, Y; Aslim, B. Determination of some characteristics coccoid forms of lactic acid bacteria isolated from Turkish kefirs with natural probiotic. LWT - Food Science and Technology 2004, 37(6), 663. [Google Scholar] [CrossRef]
- Bravo, JA; Forsythe, P; Chew, MV; Escaravage, E; Savignac, HM; Dinan, TG; et al. Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. Proc Natl Acad Sci U S A 2011, 108(38), 16050. [Google Scholar] [CrossRef]
- Janik, R; Thomason, LAM; Stanisz, AM; Forsythe, P; Bienenstock, J; Stanisz, GJ. Magnetic resonance spectroscopy reveals oral Lactobacillus promotion of increases in brain GABA, N-acetyl aspartate and glutamate. NeuroImage 2015, 125, 988. [Google Scholar] [CrossRef]
- Pollet, RM; D’Agostino, EH; Walton, WG; Xu, Y; Little, MS; Biernat, KA; et al. An Atlas of β-Glucuronidases in the Human Intestinal Microbiome. Structure 2018, 25(7), 967. [Google Scholar] [CrossRef]
- Edwinson, AL; Yang, L; Peters, S; Hanning, N; Jeraldo, P; Jagtap, P; et al. Gut microbial β-glucuronidases regulate host luminal proteases and are depleted in irritable bowel syndrome. Nat Microbiol 2022, 7(5), 680. [Google Scholar] [CrossRef]
- Hiippala, K; Barreto, G; Burrello, C; Diaz-Basabe, A; Suutarinen, M; Kainulainen, V; et al. Novel Odoribacter splanchnicus Strain and Its Outer Membrane Vesicles Exert Immunoregulatory Effects in vitro. Front Microbiol 2020, 11. [Google Scholar] [CrossRef]
- Gao, X; Tang, Y; Lei, N; Luo, Y; Chen, P; Liang, C; et al. Symptoms of anxiety/depression is associated with more aggressive inflammatory bowel disease. Sci Rep 2021, 11(1). [Google Scholar] [CrossRef]
- Rea, K; Dinan, TG; Cryan, JF. The microbiome: A key regulator of stress and neuroinflammation. Neurobiology of Stress 2016, 4, 23. [Google Scholar] [CrossRef]
- Selinger, CP; Bannaga, AS. Inflammatory bowel disease and anxiety: links, risks, and challenges faced. CEG 2015. [Google Scholar] [CrossRef] [PubMed]
- Yoon, H; Schaubeck, M; Lagkouvardos, I; Blesl, A; Heinzlmeir, S; Hahne, H; et al. Increased Pancreatic Protease Activity in Response to Antibiotics Impairs Gut Barrier and Triggers Colitis. Cellular and Molecular Gastroenterology and Hepatology 2018, 6(3), 370. [Google Scholar] [CrossRef]
- Carroll, IM; Maharshak, N; Im, C. Enteric bacterial proteases in inflammatory bowel disease- pathophysiology and clinical implications. WJG 2013, 19(43). [Google Scholar] [CrossRef] [PubMed]
- Edgington-Mitchell, LE. Pathophysiological roles of proteases in gastrointestinal disease. [CrossRef]
- Edogawa, S; Edwinson, AL; Peters, SA; Chikkamenahalli, LL; Sundt, W; Graves, S; et al. Serine proteases as luminal mediators of intestinal barrier dysfunction and symptom severity in IBS. Gut 2019, 69(1), 62. [Google Scholar] [CrossRef]
- Van De Wouw, M; Boehme, M; Lyte, JM; Wiley, N; Strain, C; O’Sullivan, O; et al. Short-chain fatty acids: microbial metabolites that alleviate stress-induced brain–gut axis alterations. The Journal of Physiology 2018, 596(20), 4923. [Google Scholar] [CrossRef]
- Kelly, CJ; Zheng, L; Campbell, EL; Saeedi, B; Scholz, CC; Bayless, AJ; et al. Crosstalk between Microbiota-Derived Short-Chain Fatty Acids and Intestinal Epithelial HIF Augments Tissue Barrier Function. Cell Host & Microbe 2016, 17(5), 662. [Google Scholar] [CrossRef]
- Tong, L; Wang, Y; Wang, Z; Liu, W; Sun, S; Li, L; et al. Propionate Ameliorates Dextran Sodium Sulfate-Induced Colitis by Improving Intestinal Barrier Function and Reducing Inflammation and Oxidative Stress. Front Pharmacol 2016, 7. [Google Scholar] [CrossRef]
- Simeoli, R.; Montague, K.; Jones, H.R.; Castaldi, L.; Chambers, D.; Kelleher, J.H.; Kostovcikova, K.; Themistocleous, A.C.; McMahon, S.B.; Holton, J.L.; Malcangio, M. Exogenous short-chain fatty acids improve neuropathic pain via modulation of microglia and T cells. Nature Communications 2017, 8, 1692. [Google Scholar] [CrossRef]
- Wei, L; Marco, ML. The fermented cabbage metabolome and its protection against cytokine-induced intestinal barrier disruption of Caco-2 monolayers. Appl Environ Microbiol 2025, 91(5). [Google Scholar] [CrossRef] [PubMed]
- Ozdal, T; Sela, DA; Xiao, J; Boyacioglu, D; Chen, F; Capanoglu, E. The Reciprocal Interactions between Polyphenols and Gut Microbiota and Effects on Bioaccessibility. Nutrients 2016, 8(2). [Google Scholar] [CrossRef]
- Herman, FJ; Pasinetti, GM. Principles of inflammasome priming and inhibition: Implications for psychiatric disorders. Brain, Behavior, and Immunity 2019, 73, 66. [Google Scholar] [CrossRef]
- Howland, R.H. Vagus nerve stimulation. Current Behavioral Neuroscience Reports 2014, 1, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Frank, M.G.; Miguel, Z.D.; Watkins, L.R.; Maier, S.F. Prior exposure to glucocorticoids sensitizes the neuroinflammatory and peripheral inflammatory responses to E. coli in rats. Brain, Behavior, and Immunity 2012, 26(3), 373–381. [Google Scholar] [CrossRef]
- Lee, J.C.; Simonyi, A.; Sun, A.Y.; Sun, G.Y. Magnolia polyphenols attenuate oxidative and inflammatory responses in neurons and microglial cells. Journal of Neuroinflammation 2013, 10, 15. [Google Scholar] [CrossRef]
- Lenzi, M.; Fimognari, C.; Hrelia, P. Sulforaphane: A promising molecule for fighting chronic disease. In Oxidative Medicine and Cellular Longevity; 2015; Volume 2015, p. 269378. [Google Scholar] [CrossRef]
- Cheruku, S.P.; Ramalingayya, G.V.; Chamallamudi, M.R.; Saxena, V. Catechin ameliorates doxorubicin-induced neuronal cytotoxicity in vitro and episodic memory deficit in vivo in Wistar rats. Neurological Sciences 2018, 39(7), 1243–1251. [Google Scholar] [CrossRef]
- Jiang, T.; Sun, Q.; Chen, S. Oxidative stress: A major pathogenesis and potential therapeutic target of antioxidative agents in Parkinson’s disease and Alzheimer’s disease. Progress in Neurobiology 2017, 147, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Rojanathammanee, L.; Murphy, E.J.; Combs, C.K. Expression of fractalkine receptor CX3CR1 on microglia correlates with neuronal loss in a mouse model of Alzheimer’s disease. Journal of Neuroimmune Pharmacology 2013, 8(4), 807–818. [Google Scholar] [CrossRef]
- Jeong, J; Lee, WS; Shin, SC; Kim, G; Choi, B; Choi, YH. Anthocyanins Downregulate Lipopolysaccharide-Induced Inflammatory Responses in BV2 Microglial Cells by Suppressing the NF-κB and Akt/MAPKs Signaling Pathways. IJMS 2013, 14(1), 1502. [Google Scholar] [CrossRef]
- Fernando Valenzuela, C.; Kazlauskas, A.; Weiner, J.L. Roles of platelet-derived growth factor in the developing and mature nervous systems. Brain Research Reviews 1997, 23(1–2), 77–89. [Google Scholar] [CrossRef] [PubMed]
- Kettenmann, H; Kirchhoff, F; Verkhratsky, A. Microglia: New Roles for the Synaptic Stripper. Neuron 2013, 77(1), 10. [Google Scholar] [CrossRef] [PubMed]
- Wolf, Y; Yona, S; Kim, K; Jung, S. Microglia, seen from the CX3CR1 angle. Front Cell Neurosci 2013, 7. [Google Scholar] [CrossRef]
- Meireles, M; Marques, C; Norberto, S; Fernandes, I; Mateus, N; Rendeiro, C; et al. The impact of chronic blackberry intake on the neuroinflammatory status of rats fed a standard or high-fat diet. The Journal of Nutritional Biochemistry 2015, 26(11), 1166. [Google Scholar] [CrossRef]
- Marques, C; Fernandes, I; Meireles, M; Faria, A; Spencer, JPE; Mateus, N; et al. Gut microbiota modulation accounts for the neuroprotective properties of anthocyanins. Sci Rep 2018, 8(1). [Google Scholar] [CrossRef]
- Clarke, G; Sandhu, KV; Griffin, BT; Dinan, TG; Cryan, JF; Hyland, NP. Gut Reactions: Breaking Down Xenobiotic–Microbiome Interactions. Pharmacological Reviews 2019, 71(2), 198. [Google Scholar] [CrossRef]
- Kearns, R. Gut–Brain Axis and Neuroinflammation: The Role of Gut Permeability and the Kynurenine Pathway in Neurological Disorders. Cell Mol Neurobiol 2024, 44(1). [Google Scholar] [CrossRef]
- Ahles, S.; Joris, P.J.; Plat, J. Effects of Berry Anthocyanins on Cognitive Performance, Vascular Function and Cardiometabolic Risk Markers: A Systematic Review of Randomized Placebo-Controlled Intervention Studies in Humans. International Journal of Molecular Sciences 2021, 22(12), 6482. [Google Scholar] [CrossRef]
- Wasiak, J; Gawlik-Kotelnicka, O. Intestinal permeability and its significance in psychiatric disorders – A narrative review and future perspectives. Behavioural Brain Research 2023, 448. [Google Scholar] [CrossRef]
- Vital, M.; Howe, A.C.; Tiedje, J.M. Revealing the Bacterial Butyrate Synthesis Pathways by Analyzing (Meta)genomic Data. mBio 2014, 5(2), e00889-14. [Google Scholar] [CrossRef]
- Duncan, SH; Belenguer, A; Holtrop, G; Johnstone, AM; Flint, HJ; Lobley, GE. Reduced Dietary Intake of Carbohydrates by Obese Subjects Results in Decreased Concentrations of Butyrate and Butyrate-Producing Bacteria in Feces. Appl Environ Microbiol 2006, 73(4), 1073. [Google Scholar] [CrossRef]
- Parada Venegas, D; De La Fuente, MK; Landskron, G; González, MJ; Quera, R; Dijkstra, G; et al. Short Chain Fatty Acids (SCFAs)-Mediated Gut Epithelial and Immune Regulation and Its Relevance for Inflammatory Bowel Diseases. Front Immunol 2019, 10. [Google Scholar] [CrossRef]
- Rivière, A; Selak, M; Lantin, D; Leroy, F; De Vuyst, L. Bifidobacteria and Butyrate-Producing Colon Bacteria: Importance and Strategies for Their Stimulation in the Human Gut. Front Microbiol 2016, 7. [Google Scholar] [CrossRef]
- De Vuyst, L; Leroy, F. Cross-feeding between bifidobacteria and butyrate-producing colon bacteria explains bifdobacterial competitiveness, butyrate production, and gas production. International Journal of Food Microbiology 2011, 149(1), 73. [Google Scholar] [CrossRef] [PubMed]
- Esquivel-Elizondo, S.; Ilhan, Z.E.; Garcia-Peña, E.I.; Krajmalnik-Brown, R. Insights into Butyrate Production in a Controlled Fermentation System via Gene Predictions. mSystems 2017, 2(4), e00051-17. [Google Scholar] [CrossRef]
- Gotoh, A; Ojima, MN; Katayama, T. Minority species influences microbiota formation: the role of Bifidobacterium with extracellular glycosidases in bifidus flora formation in breastfed infant guts. Microbial Biotechnology 2019, 12(2), 259. [Google Scholar] [CrossRef] [PubMed]
- Kumar, H; Collado, MC; Wopereis, H; Salminen, S; Knol, J; Roeselers, G. The Bifidogenic Effect Revisited—Ecology and Health Perspectives of Bifidobacterial Colonization in Early Life. Microorganisms 2020, 8(12). [Google Scholar] [CrossRef]
- O’Callaghan, A; Van Sinderen, D. Bifidobacteria and Their Role as Members of the Human Gut Microbiota. Front Microbiol 2016, 7. [Google Scholar] [CrossRef]
- Wong, CB; Odamaki, T; Xiao, J. Insights into the reason of Human-Residential Bifidobacteria (HRB) being the natural inhabitants of the human gut and their potential health-promoting benefits. FEMS Microbiology Reviews 2020, 44(3), 369. [Google Scholar] [CrossRef]
- Malard, F; Dore, J; Gaugler, B; Mohty, M. Introduction to host microbiome symbiosis in health and disease. Mucosal Immunology 2020, 14(3), 547. [Google Scholar] [CrossRef]
- Santos-Pujol, E.; Noguera-Castells, A.; Casado-Pelaez, M.; García-Prieto, C.A.; Vasallo, C.; Campillo-Marcos, I.; et al. The Multiomics Blueprint of Extreme Human Lifespan. bioRxiv 2025. [Google Scholar] [CrossRef]
- Roedlger, EW. Role of anaerobic bacteria in the metabolic welfare of the colonic mucosa in man. Gut 1980, 21(9), 793. [Google Scholar] [CrossRef] [PubMed]
- Tabat, MW; Marques, TM; Markgren, M; Löfvendahl, L; Brummer, RJ; Wall, R. Acute Effects of Butyrate on Induced Hyperpermeability and Tight Junction Protein Expression in Human Colonic Tissues. Biomolecules 2020, 10(5). [Google Scholar] [CrossRef] [PubMed]
- Vancamelbeke, M; Vermeire, S. The intestinal barrier: a fundamental role in health and disease. Expert Review of Gastroenterology & Hepatology 2018, 11(9), 821. [Google Scholar] [CrossRef]
- Hoel, H; Heggelund, L; Reikvam, DH; Stiksrud, B; Ueland, T; Michelsen, AE; et al. Elevated markers of gut leakage and inflammasome activation in COVID-19 patients with cardiac involvement. J Intern Med 2020, 289(4), 523. [Google Scholar] [CrossRef]
- Clark, RL; Connors, BM; Stevenson, DM; Hromada, SE; Hamilton, JJ; Amador-Noguez, D; et al. Design of synthetic human gut microbiome assembly and butyrate production. Nat Commun 2021, 12(1). [Google Scholar] [CrossRef] [PubMed]
- Stoeva, MK; Garcia-So, J; Justice, N; Myers, J; Tyagi, S; Nemchek, M; et al. Butyrate-producing human gut symbiont, Clostridium butyricum, and its role in health and disease. Gut Microbes 2021, 13(1). [Google Scholar] [CrossRef]
- Fung, KYC; Cosgrove, L; Lockett, T; Head, R; Topping, DL. A review of the potential mechanisms for the lowering of colorectal oncogenesis by butyrate. Br J Nutr 2012, 108(5), 820. [Google Scholar] [CrossRef]
- Rosser, EC; Piper, CJM; Matei, DE; Blair, PA; Rendeiro, AF; Orford, M; et al. Microbiota-Derived Metabolites Suppress Arthritis by Amplifying Aryl-Hydrocarbon Receptor Activation in Regulatory B Cells. Cell Metabolism 2020, 31(4), 837. [Google Scholar] [CrossRef] [PubMed]
- Kimura, I; Ozawa, K; Inoue, D; Imamura, T; Kimura, K; Maeda, T; et al. The gut microbiota suppresses insulin-mediated fat accumulation via the short-chain fatty acid receptor GPR43. Nat Commun 2013, 4(1). [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Yi, C.-X.; Katiraei, S.; Kooijman, S.; Zhou, E.; Chung, C.K.; Gao, Y.; van den Heuvel, J.K.; Meijer, O.C.; Berbée, J.F.P.; Heijink, M.; Giera, M.; Willems van Dijk, K.; Groen, A.K.; Rensen, P.C.N.; Wang, Y. Butyrate reduces appetite and activates brown adipose tissue via the gut-brain neural circuit. Gut 2017, 67(7), 1269–1279. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.V.; Frassetto, A.; Kowalik, E.J., Jr.; Nawrocki, A.R.; Lu, M.M.; Kosinski, J.R.; Hubert, J.A.; Szeto, D.; Yao, X.; Forrest, G.; Marsh, D.J. Butyrate and Propionate Protect against Diet-Induced Obesity and Regulate Gut Hormones via Free Fatty Acid Receptor 3-Independent Mechanisms. PLoS ONE 2012, 7(4), e35240. [Google Scholar] [CrossRef]
- Zhang, L; Du, J; Yano, N; Wang, H; Zhao, YT; Dubielecka, PM; et al. Sodium Butyrate Protects Against High Fat Diet-Induced Cardiac Dysfunction and Metabolic Disorders in Type II Diabetic Mice. J Cell Biochem 2018, 118(8), 2395. [Google Scholar] [CrossRef] [PubMed]
- Silva, YP; Bernardi, A; Frozza, RL. The Role of Short-Chain Fatty Acids From Gut Microbiota in Gut-Brain Communication. Front Endocrinol 2020, 11. [Google Scholar] [CrossRef]
- Siddiqui, MT; Cresci, GA. The Immunomodulatory Functions of Butyrate. JIR 2021, 14, 6025. [Google Scholar] [CrossRef]
- Matt, SM; Allen, JM; Lawson, MA; Mailing, LJ; Woods, JA; Johnson, RW. Butyrate and Dietary Soluble Fiber Improve Neuroinflammation Associated With Aging in Mice. Front Immunol 2018, 9. [Google Scholar] [CrossRef]
- De Vos, WM; Tilg, H; Van Hul, M; Cani, PD. Gut microbiome and health: mechanistic insights. Gut 2022, 71(5), 1020. [Google Scholar] [CrossRef]
- Sun, Y; O’riordan, MXD. Regulation of Bacterial Pathogenesis by Intestinal Short-Chain Fatty Acids. Advances in Applied Microbiology 2014, 93. [Google Scholar] [CrossRef]
- Zhao, C; Dong, H; Zhang, Y; Li, Y. Discovery of potential genes contributing to the biosynthesis of short-chain fatty acids and lactate in gut microbiota from systematic investigation in E. coli. npj Biofilms Microbiomes 2019, 5(1). [Google Scholar] [CrossRef] [PubMed]
- Fernández-Veledo, S; Vendrell, J. Gut microbiota-derived succinate: Friend or foe in human metabolic diseases? Rev Endocr Metab Disord 2019, 20(4), 439. [Google Scholar] [CrossRef]
- Daniel, SL; Moradi, L; Paiste, H; Wood, KD; Assimos, DG; Holmes, RP; et al. Forty Years of Oxalobacter formigenes, a Gutsy Oxalate-Degrading Specialist. Appl Environ Microbiol 2021, 87(18). [Google Scholar] [CrossRef] [PubMed]
- Ticinesi, A; Nouvenne, A; Chiussi, G; Castaldo, G; Guerra, A; Meschi, T. Calcium Oxalate Nephrolithiasis and Gut Microbiota: Not just a Gut-Kidney Axis. A Nutritional Perspective. Nutrients 2020, 12(2). [Google Scholar] [CrossRef]
- Liu, M.; Devlin, J.C.; Hu, J.; Volkova, A.; Battaglia, T.W.; Byrd, A.; Loke, P.; Li, H.; Ruggles, K.V.; Tsirigos, A.; Blaser, M.J.; Nazzal, L. Microbial genetic and transcriptional contributions to oxalate degradation by the gut microbiota in health and disease. eLife 2021, 10, e63642. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.-M.; Hung, J.-H.; Huang, S.-L. Nitrate promotes the growth and the production of short-chain fatty acids and tryptophan from commensal anaerobe Veillonella dispar in the lactate-deficient environment by facilitating the catabolism of glutamate and aspartate. NPJ Biofilms and Microbiomes 2024, 10(1). [Google Scholar] [CrossRef]
- Detman, A.; Mielecki, D.; Chojnacka, A.; Salamon, A.; Błaszczyk, M.K.; Sikora, A. Cell factories converting lactate and acetate to butyrate: Clostridium butyricum and microbial communities from dark fermentation bioreactors. Microbial Cell Factories 2019, 18(1), 36. [Google Scholar] [CrossRef]
- Wiczkowski, W; Szawara-Nowak, D; Topolska, J. Red cabbage anthocyanins: Profile, isolation, identification, and antioxidant activity. Food Research International 2013, 51(1), 303. [Google Scholar] [CrossRef]
- Podsędek, A; Redzynia, M; Klewicka, E; Koziołkiewicz, M. Matrix Effects on the Stability and Antioxidant Activity of Red Cabbage Anthocyanins under Simulated Gastrointestinal Digestion. BioMed Research International 2014, 2014, 1. [Google Scholar] [CrossRef]
- Chen, L; Zhu, Y; Hu, Z; Wu, S; Jin, C. Beetroot as a functional food with huge health benefits: Antioxidant, antitumor, physical function, and chronic metabolomics activity. Food Science & Nutrition 2021, 9(11), 6406. [Google Scholar] [CrossRef]
- Miller, T.L.; Wolin, M.J. Pathways of acetate, propionate, and butyrate formation by the human fecal microbial flora. Appl. Environ. Microbiol. 1996, 62, 1589–1592. [Google Scholar] [CrossRef]
- Colucci Cante, R; Nigro, F; Passannanti, F; Lentini, G; Gallo, M; Nigro, R; et al. Gut health benefits and associated systemic effects provided by functional components from the fermentation of natural matrices. Comp Rev Food Sci Food Safe 2024, 23(3). [Google Scholar] [CrossRef]
- Eker, ME; Aaby, K; Budic-Leto, I; Rimac Brnčić, S; El, SN; Karakaya, S; et al. A Review of Factors Affecting Anthocyanin Bioavailability: Possible Implications for the Inter-Individual Variability. Foods 2019, 9(1). [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Díaz, I.; Fernández-Navarro, T.; Salazar, N.; Bartolomé, B.; Moreno-Arribas, M.V.; López, P.; Suárez, A.; González de los Reyes-Gavilán, C.; Gueimonde, M.; González, S. Could fecal phenylacetic and phenylpropionic acids be used as indicators of health status? J. Agric. Food Chem. 2018, 66, 10438–10446. [Google Scholar] [CrossRef]
- Aura, A; Martin-Lopez, P; O’leary, KA; Williamson, G; Oksman-Caldentey, K; Poutanen, K; et al. In vitro metabolism of anthocyanins by human gut microflora. Eur J Nutr 2004, 44(3), 133. [Google Scholar] [CrossRef]
- El Mohsen, MA; Marks, J; Kuhnle, G; Moore, K; Debnam, E; Srai, SK; et al. Absorption, tissue distribution and excretion of pelargonidin and its metabolites following oral administration to rats. Br J Nutr 2006, 95(1), 51. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.; Veličković, D.; Garcia, A.; Temperton, N.; Saha, S.; Warden, C.; Kroon, P.A. Anthocyanins are not absorbed intact but reach the colon where they may exert health effects via microbial metabolism. Food & Function 2014, 5(6), 1132–1137. [Google Scholar] [CrossRef]
- Van Rymenant, E; Abrankó, L; Tumova, S; Grootaert, C; Van Camp, J; Williamson, G; et al. Chronic exposure to short-chain fatty acids modulates transport and metabolism of microbiome-derived phenolics in human intestinal cells. The Journal of Nutritional Biochemistry 2017, 39, 156. [Google Scholar] [CrossRef] [PubMed]
- Berkhout, MD; Plugge, CM; Belzer, C. How microbial glycosyl hydrolase activity in the gut mucosa initiates microbial cross-feeding. Glycobiology 2021, 32(3), 182. [Google Scholar] [CrossRef]
- Rodriguez-Gaza, C.; Henrissat, B.; Terrapon, N.; Lombard, V.; Crouch, L.I.; Gilbert, H.J. The CAZyome of human gut bacteria: Carbohydrate-active enzymes and their role in the microbiome. Microbiome 2021, 9, 61. [Google Scholar] [CrossRef]
- Ćurko, N.; Tomašević, M.; Cvjetko Bubalo, M.; Gracin, L.; Radojčić Redovniković, I.; Kovačević Ganić, K. Extraction of Proanthocyanidins and Anthocyanins from Grape Skin by Using Ionic Liquids. Food Technol. Biotechnol. 2017, 55(3), 429–437. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y; Morozova, K; Scampicchio, M; Ferrentino, G. Non-Extractable Polyphenols from Food By-Products: Current Knowledge on Recovery, Characterisation, and Potential Applications. Processes 2020, 8(8). [Google Scholar] [CrossRef]
- Gloux, K; Berteau, O; El Oumami, H; Béguet, F; Leclerc, M; Doré, J. A metagenomic β-glucuronidase uncovers a core adaptive function of the human intestinal microbiome. Proc Natl Acad Sci U S A 2010, 108 (supplement_1), 4539. [Google Scholar] [CrossRef]
- Ting, NL; Lau, HC; Yu, J. Cancer pharmacomicrobiomics: targeting microbiota to optimise cancer therapy outcomes. Gut 2022, 71(7), 1412. [Google Scholar] [CrossRef]
- Dabek, M; Mccrae, SI; Stevens, VJ; Duncan, SH; Louis, P. Distribution of β-glucosidase and β-glucuronidase activity and of β-glucuronidase gene gus in human colonic bacteria. FEMS Microbiology Ecology 2008, 66(3), 487. [Google Scholar] [CrossRef]
- Drula, E; Garron, M; Dogan, S; Lombard, V; Henrissat, B; Terrapon, N. The carbohydrate-active enzyme database: functions and literature. Nucleic Acids Research 2021, 50(D1), D571. [Google Scholar] [CrossRef] [PubMed]
- Schropp, N; Bauer, A; Stanislas, V; Huang, KD; Lesker, T; Bielecka, AA; et al. The impact of regular sauerkraut consumption on the human gut microbiota: a crossover intervention trial. Microbiome 2025, 13(1). [Google Scholar] [CrossRef]
- Karačić, A; Zonjić, J; Stefanov, E; Radolović, K; Starčević, A; Renko, I; et al. Short-Term Supplementation of Sauerkraut Induces Favorable Changes in the Gut Microbiota of Active Athletes: A Proof-of-Concept Study. Nutrients 2024, 16(24). [Google Scholar] [CrossRef]
- Peng, L.; Zhang, Y.; Li, W.; Dai, T.; Nie, L.; Xie, J. Polyphenol Extract of Moringa oleifera Leaves Alleviates Colonic Inflammation in Dextran Sulfate Sodium-Treated Mice. Evid.-Based Complement. Altern. Med. 2020, 2020, 6295402. [Google Scholar] [CrossRef]
- Gu, Y.; Yang, X.; Yu, Y.; Li, H.; Shen, T.; Tang, Y. Dietary polyphenols promote growth of Barnesiella and Clostridium in the gut microbiota of rodents, contributing to improved metabolic outcomes. Journal of Functional Foods 2019, 57, 364–375. [Google Scholar] [CrossRef]
- Anhê, F.F.; Roy, D.; Pilon, G.; Dudonné, S.; Matamoros, S.; Varin, T.V.; Garofalo, C.; Moine, Q.; Desjardins, Y.; Levy, E.; Marette, A. A polyphenol-rich cranberry extract protects from diet-induced obesity, insulin resistance and intestinal inflammation in association with increased Akkermansia spp. population in the gut microbiota of mice. Gut 2015, 64(6), 872–883. [Google Scholar] [CrossRef]
- Fernandez, J.; Redondo-Blanco, S.; Gutiérrez-del-Río, I.; Miguélez, E.M.; Villar, C.J.; Lombó, F. Colon microbiota fermentation of dietary polyphenols: A review on their effects on human health. Food & Function 2018, 9(10), 5617–5631. [Google Scholar] [CrossRef]
- Jennings, A.; Koch, M.; Jensen, M.K.; Bang, C.; Kassubek, J.; Müller, H.P.; Nöthlings, U.; Franke, A.; Lieb, W.; Cassidy, A. The role of the gut microbiome in the association between habitual anthocyanin intake and visceral abdominal fat in population-level analysis. The American Journal of Clinical Nutrition 2023, 117(3), 492–502. [Google Scholar] [CrossRef] [PubMed]
- Baxter, N.T.; Schmidt, A.W.; Venkataraman, A.; Kim, K.S.; Waldron, C.; Schmidt, T.M. Dynamics of Human Gut Microbiota and Short-Chain Fatty Acids in Response to Dietary Interventions with Three Fermentable Fibers. mBio 2019, 10(1), e02566-18. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Castaño, GP; Rey, FE; Caro-Quintero, A; Acosta-González, A. Gut-derived Flavonifractor species variants are differentially enriched during in vitro incubation with quercetin. PLoS ONE 2020, 15(12). [Google Scholar] [CrossRef]
- Goris, T; Cuadrat, RRC; Braune, A. Flavonoid-Modifying Capabilities of the Human Gut Microbiome—An In Silico Study. Nutrients 2021, 13(8). [Google Scholar] [CrossRef] [PubMed]
- Pudlo, N.A.; Urs, K.; Crawford, R.; Pirani, A.; Atherly, T.; Jimenez, R. Phenotypic and Genomic Diversification in Complex Carbohydrate-Degrading Human Gut Bacteria. mSystems 2022, 7(1), e01250-21. [Google Scholar] [CrossRef]
- Annunziata, G.; Arnone, A.; Ciampaglia, R.; Tenore, G.C.; Novellino, E. Short-Time Lactic-Acid Fermentation Improves the Nutraceutical Value of Black Tea Beverage. ResearchGate 2018. [Google Scholar] [CrossRef]
- Annunziata, G; Arnone, A; Ciampaglia, R; Tenore, GC; Novellino, E. Fermentation of Foods and Beverages as a Tool for Increasing Availability of Bioactive Compounds. Focus on Short-Chain Fatty Acids. Foods 2020, 9(8). [Google Scholar] [CrossRef]
- Uțoiu, E; Matei, F; Toma, A; Diguță, C; Ștefan, L; Mănoiu, S; et al. Bee Collected Pollen with Enhanced Health Benefits, Produced by Fermentation with a Kombucha Consortium. Nutrients 2018, 10(10). [Google Scholar] [CrossRef]
- Bhat, R; Suryanarayana, LC; Chandrashekara, KA; Krishnan, P; Kush, A; Ravikumar, P. Lactobacillus plantarum mediated fermentation of Psidium guajava L. fruit extract. Journal of Bioscience and Bioengineering 2014, 119(4), 430. [Google Scholar] [CrossRef]
- Lampe, JW. Interindividual differences in response to plant-based diets: implications for cancer risk. The American Journal of Clinical Nutrition 2009, 89(5), 1553S. [Google Scholar] [CrossRef]
- Padayachee, A; Netzel, G; Netzel, M; Day, L; Zabaras, D; Mikkelsen, D; et al. Binding of polyphenols to plant cell wall analogues – Part 2: Phenolic acids. Food Chemistry 2012, 135(4), 2287. [Google Scholar] [CrossRef]
- Hur, SJ; Lee, SY; Kim, Y; Choi, I; Kim, G. Effect of fermentation on the antioxidant activity in plant-based foods. Food Chemistry 2014, 160, 346. [Google Scholar] [CrossRef] [PubMed]
- Pellock, SJ; Walton, WG; Biernat, KA; Torres-Rivera, D; Creekmore, BC; Xu, Y; et al. Three structurally and functionally distinct β-glucuronidases from the human gut microbe Bacteroides uniformis. Journal of Biological Chemistry 2018, 293(48), 18559. [Google Scholar] [CrossRef] [PubMed]
- Dashnyam, P; Mudududdla, R; Hsieh, T; Lin, T; Lin, H; Chen, P; et al. β-Glucuronidases of opportunistic bacteria are the major contributors to xenobiotic-induced toxicity in the gut. Sci Rep 2018, 8(1). [Google Scholar] [CrossRef] [PubMed]
- Gaur, G; Oh, J; Filannino, P; Gobbetti, M; Van Pijkeren, J; Gänzle, MG. Genetic Determinants of Hydroxycinnamic Acid Metabolism in Heterofermentative Lactobacilli. Appl Environ Microbiol 2020, 86(5). [Google Scholar] [CrossRef]
- Gutiérrez-Díaz, I.; Fernández-Navarro, T.; Salazar, N.; Bartolomé, B.; Moreno-Arribas, M.V.; López, P.; Suárez, A.; de Los Reyes-Gavilán, C.G.; Gueimonde, M.; González, S. Could Fecal Phenylacetic and Phenylpropionic Acids Be Used as Indicators of Health Status? Journal of Agricultural and Food Chemistry 2018, 66(40), 10438–10446. [Google Scholar] [CrossRef]
- Deleu, S; Machiels, K; Raes, J; Verbeke, K; Vermeire, S. Short chain fatty acids and its producing organisms: An overlooked therapy for IBD? eBioMedicine 2021, 66. [Google Scholar] [CrossRef]
- Fang, W; Xue, H; Chen, X; Chen, K; Ling, W. Supplementation with Sodium Butyrate Modulates the Composition of the Gut Microbiota and Ameliorates High-Fat Diet-Induced Obesity in Mice. The Journal of Nutrition 2019, 149(5), 747. [Google Scholar] [CrossRef]
- Smith, PM; Howitt, MR; Panikov, N; Michaud, M; Gallini, CA; Bohlooly-Y, M; et al. The Microbial Metabolites, Short-Chain Fatty Acids, Regulate Colonic T reg Cell Homeostasis. Science 2013, 341(6145), 569. [Google Scholar] [CrossRef]
- Pouteau, E.; Nguyen, P.; Ballèvre, O.; Krempf, M. Production rates and metabolism of short-chain fatty acids in the colon and whole body using stable isotopes. Proceedings of the Nutrition Society 2003, 62(1), 87–93. [Google Scholar] [CrossRef] [PubMed]
- Van Der Beek, CM; Bloemen, JG; Van Den Broek, MA; Lenaerts, K; Venema, K; Buurman, WA; et al. Hepatic Uptake of Rectally Administered Butyrate Prevents an Increase in Systemic Butyrate Concentrations in Humans1–3. The Journal of Nutrition 2015, 145(9), 2019. [Google Scholar] [CrossRef]
- Shimizu, H; Masujima, Y; Ushiroda, C; Mizushima, R; Taira, S; Ohue-Kitano, R; et al. Dietary short-chain fatty acid intake improves the hepatic metabolic condition via FFAR3. Sci Rep 2019, 9(1). [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Fang, J.-Y. Fusobacterium nucleatum, a key pathogenic factor and microbial biomarker for colorectal cancer. Trends in Microbiology 2023, 31(1), 10–19. [Google Scholar] [CrossRef]
- Zhu, Q.; Hou, Q.; Huang, S.; Ou, Q.; Huo, D.; Peng, Y. Eubacterium rectale contributes to colorectal cancer initiation via promoting colitis. Gut Pathogens 2021, 13, 2. [Google Scholar] [CrossRef]
- Louis, P; Flint, HJ. Formation of propionate and butyrate by the human colonic microbiota. Environmental microbiology 2017, 19(1), 29–41. [Google Scholar] [CrossRef]
- Zonjić, J; Karačić, A; Brodić, I; Starčević, A; Renko, I; Krznarić, Ž; et al. The Short- and Long-Term Effects of a Short Course of Sauerkraut Supplementation on the Gut Microbiota of Active Athletes: A Pilot Follow-Up Study. Nutrients 2025, 17(5). [Google Scholar] [CrossRef] [PubMed]
- Taylor, B.C.; Lejzerowicz, F.; Poirel, M.; Shaffer, J.P.; Jiang, L.; Aksenov, A.; Litwin, N.; Humphrey, G.; Martino, C.; Miller-Montgomery, S. Consumption of fermented foods is associated with systematic differences in the gut microbiome and metabolome. mSystems 2020, 5(2), e00901-19. [Google Scholar] [CrossRef] [PubMed]
- Candeliere, F.; Raimondi, S.; Ranieri, R.; Musmeci, E.; Zambon, A.; Amaretti, A.; Rossi, M. β-Glucuronidase Pattern Predicted From Gut Metagenomes Indicates Potentially Diversified Pharmacomicrobiomics. Frontiers in Microbiology 2022, 13, 826994. [Google Scholar] [CrossRef] [PubMed]
| Dairy Kefir Group A |
Fermented Red Cabbage Group B |
Fermented Coconut Milk Group C | |
|---|---|---|---|
| portion | 247ml | ~50g | 100ml |
| Lactic Acid Bacteria (LAB), cfu/ml / cfu/g | 105 | 106 | 1011 |
| Energy | 66.9kJ/16kCal | 125kJ/30kCal | 141kJ/34kCal |
| Total Fat | 0.2g | 0.9g | 2.0g |
| Of which saturates | 1.9g | 0.4g | 1.9g |
| CHO (carbohydrate) | 4.2g | 2.1g | 2.8G |
| Of which sugars | 3.8g | No detectible amount | 2.0g |
| Fibre | No detectible amount | 2.8g | 1.7g |
| Protein | 3.25g | 2.0g | 0.8g |
| Salt | No detectible amount | 1.56g | No detectible amount |
| Study Activities | Screening | Visit1 familiarisation | Visit 2 baseline | Visit 3 (day 15) mid-line |
Visit 4 (day 30) end | Visit 5 post testing |
|---|---|---|---|---|---|---|
| Informed Consent | x | |||||
| Health Questionnaire | x | |||||
| Randomization | x | |||||
| Plant Food Diary | x | |||||
| Diet and Lifestyle Questionnaire | x | |||||
| Brain Health | ||||||
| Cognitive Function | x | x | x | x | ||
| Emotional Health | ||||||
| POMS | x | x | x | |||
| DASS-21 | x | x | x | |||
| Gastrointestinal Health | ||||||
| Bristol Stool Scale | x receive | x return/receive | x return | |||
| Stool Test Kit receive | x | x | ||||
| Stool Test Kit return | x | x | ||||
| Quality of Life | ||||||
| MYMOP® | x | x follow-up | x follow-up | x follow-up | ||
| Product receive | x | x | ||||
| Adverse Symptom Assessment | x | x | ||||
| Phylum | Genus | Reference range (CFU/g stool) |
|---|---|---|
| Bacteroidetes | Bacteroidetes-Prevotella group | 3.4E6 – 1.5E9 |
| Bacteroidetes vulgatus | <=2.2E9 | |
| Barnsiella spp. | <=1.6E8 | |
| Odoribacter spp. | <=8.0E7 | |
| Prevotella spp. | 1.4E5 – 1.6E7 | |
| Firmicutes | Anaerotruncus colihominus | <=3.2E7 |
| Butyrivibrio cossotus | 5.5E3 – 5.9E5 | |
| Clostridium spp. | 1.7E8 – 1.5 E10 | |
| Coprococcus eutactus | <=1.2E8 | |
| Faecalibacterium prausnitzii | 5.8E7 – 4.7E9 | |
| Lactobacillus spp. | 8.3E6 – 5.2E9 | |
| Pseudoflavonifractor spp. | 4.2E5 – 1.3E8 | |
| Roseburia spp. | 1.3E8 – 1.2E10 | |
| Ruminococcus spp. | 9.5E7 – 1.6E9 | |
| Veillonella spp. | 1.2E5 – 5.5E7 | |
| Actinobacteria | Bifidobacterium spp. | <=6.4E9 |
| Bifidobacterium longum | <=7.2E8 | |
| Collinsella formigenes | 1.4E7 – 1.9E9 | |
| Proteobacteria | Desolfovibrio piger | <=1.8E7 |
| Escherichia coli | 9.0E4 – 4.6E7 | |
| Oxalobacter formigenes | <=1.5E7 | |
| Euryarchaeota | Methanobrevibacter smithii | <=8.6E7 |
| Fusobacteria | Fusobacterium spp. | <=2.4E5 |
| Verrucomicrobia | Akkermansia municiphila | >=1.2E6 |
| Groups | Dairy Kefir (A) | Fermented Cabbage (B) | Coconut Kefir (C) | Control (D) | Group difference p value |
|---|---|---|---|---|---|
| Anthropometric characteristics: | |||||
| Age (years) | 39.09(8.53) | 38.64(8.56) | 38.82(7.18) | 38.55(8.68) | .623 |
| Age range (years) | 20-49 | 27-52 | 25-48 | 25-51 | |
| Female/Male | 6/5 | 7/4 | 7/4 | 7/4 | .962 |
| Body weight (kg) | 65.1 (10.2) | 70.6 (13) | 66.2 (11.1) | 61.5 (5.2) | .595 |
| Habitual dietary intake (portions): | |||||
| Daily Vegetable | 3.9(2.3) | 4.7(2.3) | 5.5(2.1) | 6.7(3.0) | .127 |
| Daily Fruit | 2.4(2.3) | 1.4(3.0) | 1.9(1.4) | 1.8(1.0) | .849 |
| Weekly Fermented Foods | 1.9(2.2) | 1.9(3.1) | 4.2(3.0) | 2.8(2.4) | .117 |
| Education Levels | |||||
| Left formal education <age 16 | 0 | 0 | 0 | 0 | .215 |
| Left Formal education at age 16 | 0 | 2 | 0 | 0 | |
| Left Formal education at age 17-18 | 1 | 0 | 3 | 2 | |
| Undergraduate degree or equivalent | 8 | 8 | 7 | 6 | |
| Postgraduate degree or equivalent | 1 | 0 | 1 | 0 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.





