Submitted:
05 January 2026
Posted:
06 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials
2.1. Instruments Used and Optical Fibers Tested
- Two Echolaser X4 systems were employed: one unit was used for tests involving Elesta–Oberon optical fibers equipped with an SMA905 connector featuring a 0.1-mm setback, while the second unit was used for tests performed with Oberon, Thorlabs or Asclepion optical fibers with a standard SMA905 connector;
- Ultrasound imaging was carried out using an Esaote MyLabOmega system equipped with a linear probe;
- Temperature monitoring was performed using an Agilent Data Acquisition/Switch Unit (SN US37034947) in combination with two type-K thermocouples: one RS thermocouple (SN 6212170) with a 0.3-mm probe diameter, 2-m length, and +600 °C range mounted on a 14G needle, and a second RS thermocouple (SN 8047990) with a 0.076-mm probe diameter, 2-m length, and +260 °C range mounted on a 21G needle;
- A Tsunami Medical balloon catheter (PBKK 11/20-20; outer diameter 11G, length 20 cm, balloon volume 5.5 ml) and a Tsunami Medical inflation kit (RK07) were used to perform balloon-assisted procedures;
- Optical power measurements were obtained using an Ophir power meter (7Z01565);
- Optical fibers were prepared using a Jonard Tools adjustable wire stripper (20–30 AWG, ST-500).
2.2. Thermocouples for Temperature Measurements
- 10 mm from the fiber tip in single-fiber tests;
- at the midpoint between the fibers in dual-fiber configurations.
2.3. Laser System
2.4. Angular Optical Power Distribution Measurement System (Goniophotometer)
- a dark chamber;
- a rotation stage controlled by a stepper motor;
- a photodiode sensor mounted on a rotating arm.
3. Methods
3.1. Optical Fiber Emission Theory – Numerical Aperture (NA)
3.2. Laser Ablation Phenomenon, Radiation-Matter Interaction and Thermal Effects
3.3. Sample Selection, Fiber and Thermocouples Insertion Techniques
- calf liver: commonly used substitute for prostate tissue due to comparable optical/thermal properties;
- porcine muscle: for preliminary verification tests.
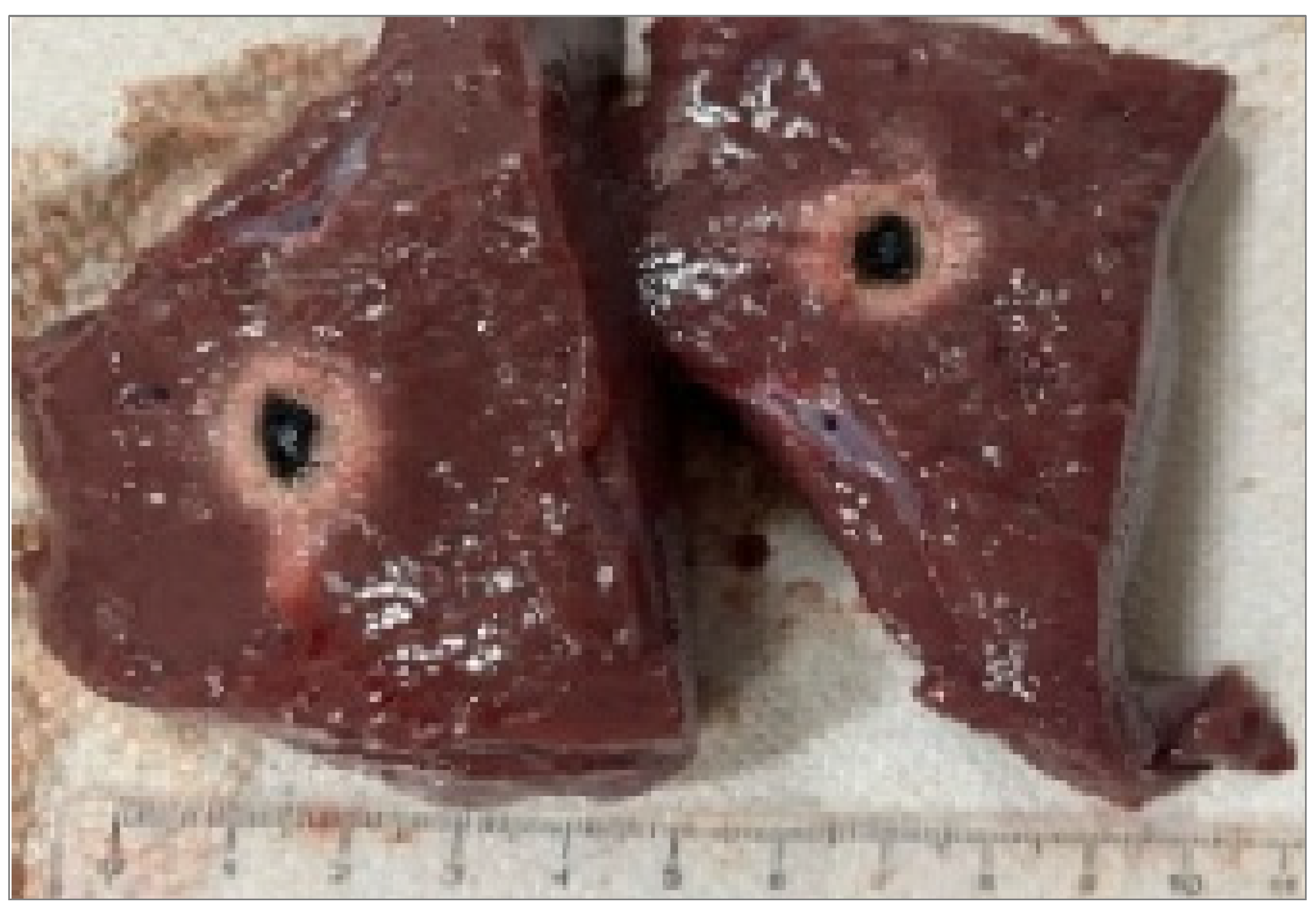
3.4. Recognition of Ablation Size
3.5. Tissues Irradiation Techniques
- Single fiber;
- Dual fiber with 10 mm spacing;
- Pull-back technique (fiber retraction after initial energy delivery);
- Balloon catheter with scattering medium (hydroxyapatite suspension) in selected trials.
- Single-fiber and multi-fiber configurations, to adapt ablation volume and shape to tissue geometry;
- Pull-back technique, in which fibers are retracted after initial energy delivery to extend the ablation region longitudinally;
3.6. Outcome Parameters
4. Results
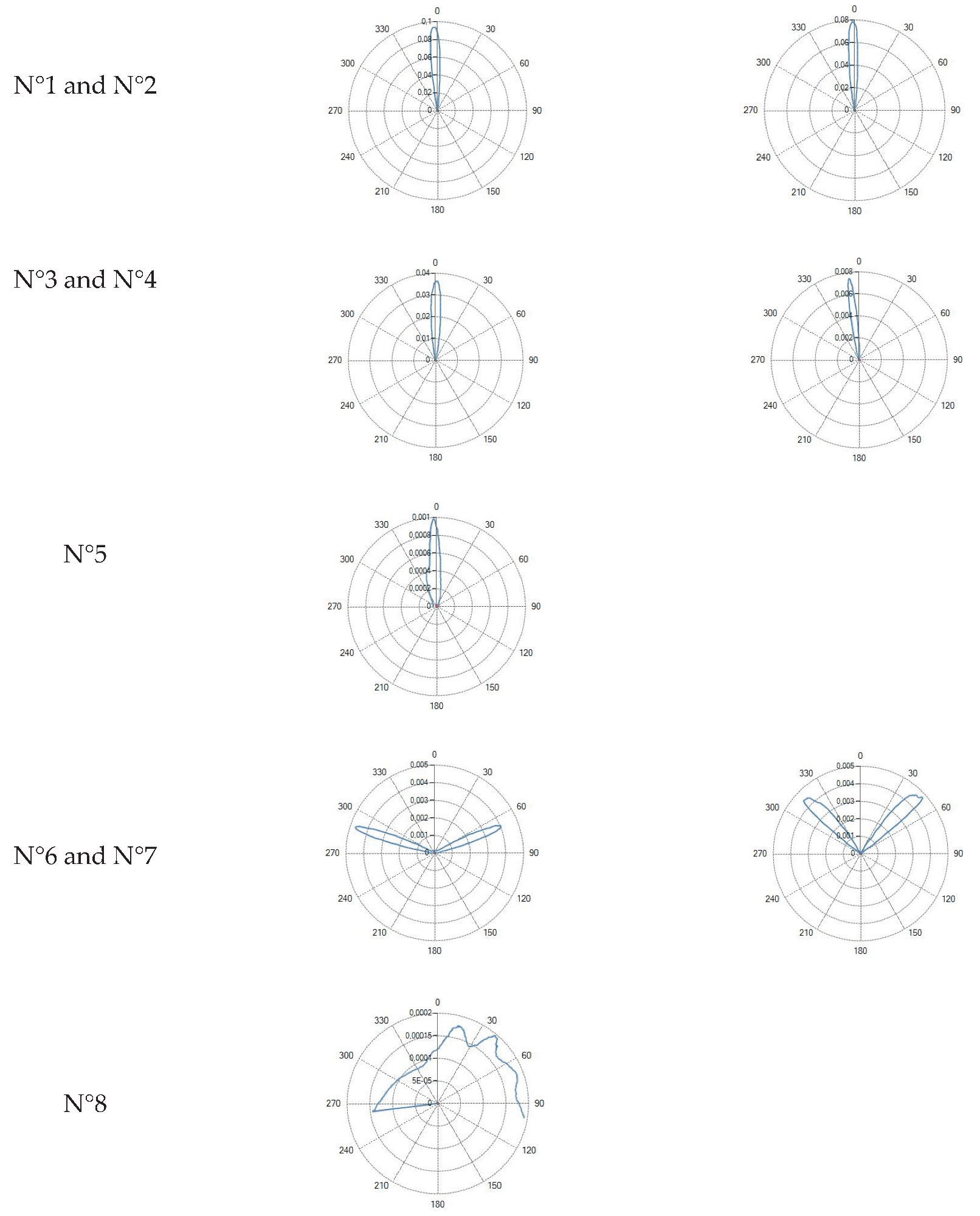
4.1. Radiation Profiles of the Eight Types of Optical Fibers Analyzed
| Type of optical fiber | Radiation profiles |
 | |
4.2. Optical Fibers with Flat Tip Geometry
- Core = 272 µm (Fiber N°1) –300 µm (Fiber N°2), NA = 0.22. Produced the smallest lesions, with longitudinal dimensions (L) typically exceeding transverse width (W), yielding an elongated, ellipsoidal profile.
- Core = 300 µm (Fiber N°3) – 400 µm (Fiber N°4), NA = 0.37. Resulted in a marked increase in W and a more spherical ablation shape. The transition from NA 0.22 to 0.37 produced the largest relative increase in W among all pairwise comparisons.
- Core = 600 µm (Fiber N°5), NA = 0.48. Generated the largest ablations overall. At 7 W, these fibers produced broad coagulation zones but also showed the highest degree of thermal spread, consistent with the combination of high NA and large mode volume.
4.3. Radial Emission Conical Fibers
- Ablation shapes were more spherical and homogeneous.
- L decreased relative to flat tip fibers, confirming the lateral redistribution of energy.
4.4. Spherical Curved Ball Tip Fiber
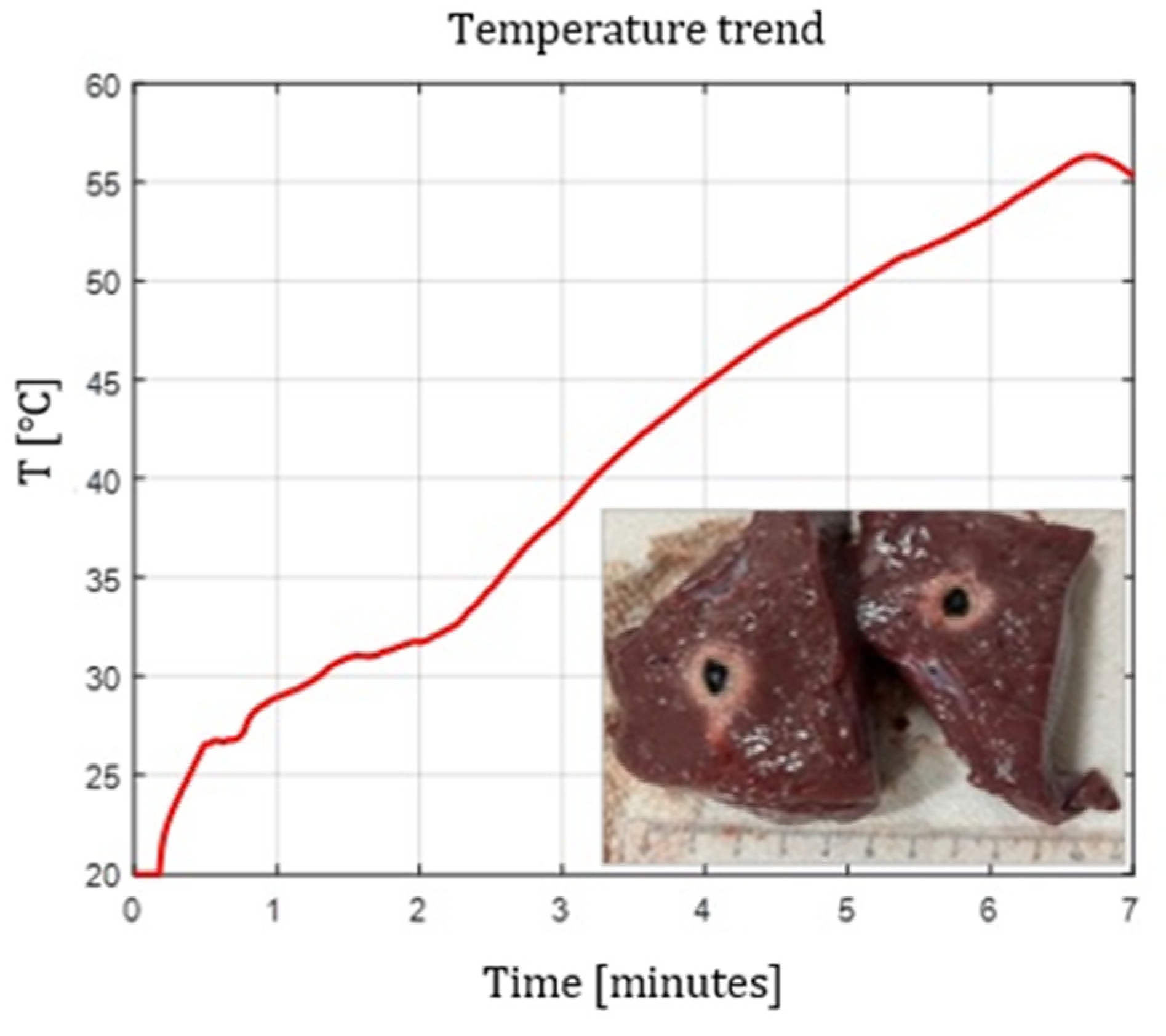
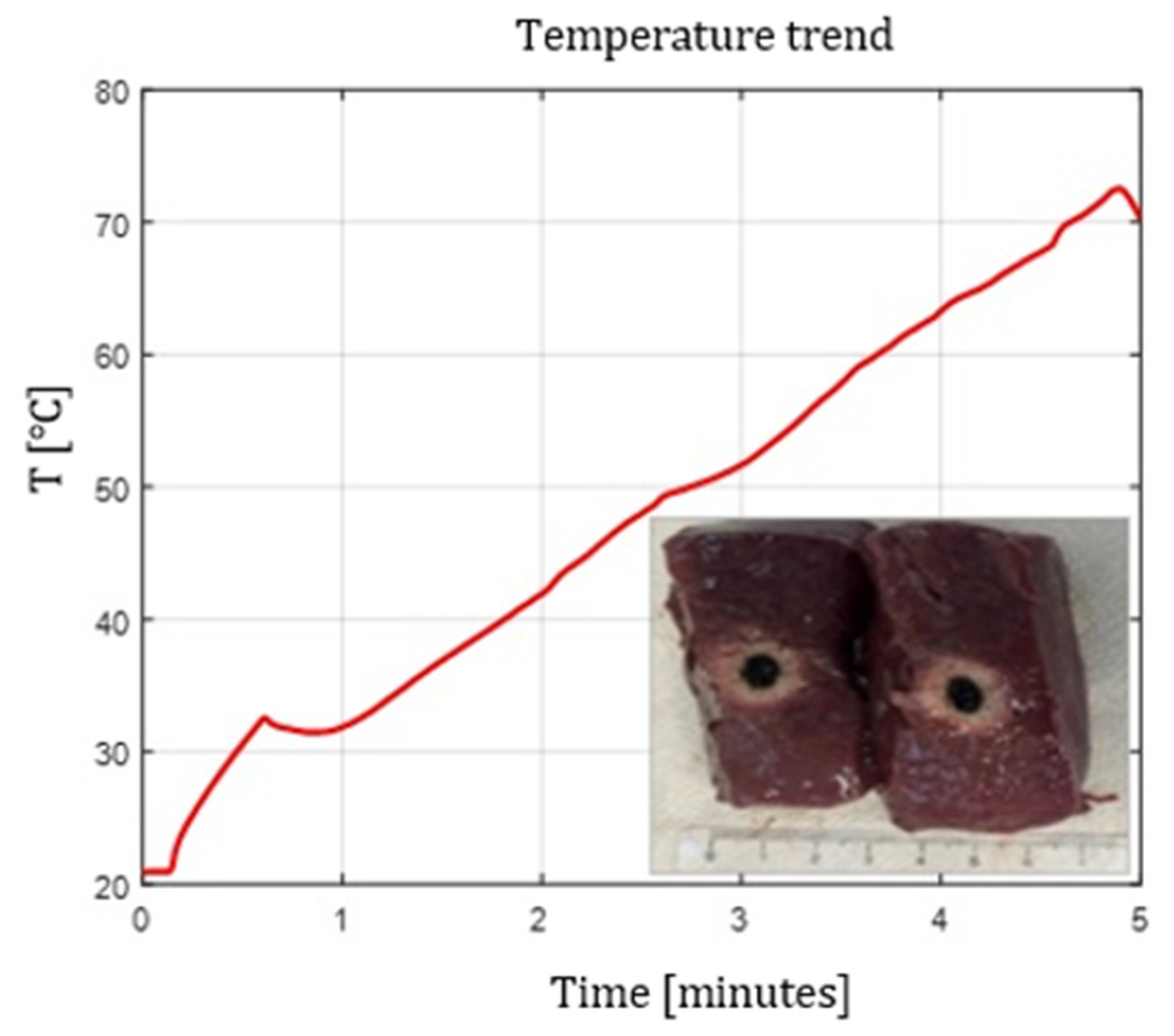
4.5. Temperature Profiles
- Flat tip fibers with higher NA and higher core diameter reached peak temperatures faster.
- Radial and ball-tip fibers exhibited lower initial temperature slopes, indicating slower local heating due to redistributed emission.
4.6. Multi-Fiber and Pull-Back Configurations
- substantial enlargement of the transverse ablation zone;
- partial merging of the two thermal fields;
- higher measured temperatures at the midpoint compared to single-fiber trials.
4.7. Balloon Catheter with Scattering Medium
5. Discussion
5.1. Influence of Core Diameter and Numerical Aperture
5.2. Radial and Spherical Emission for Uniform Ablation
5.3. Multi-Fiber and Pull-Back Strategies
5.4. Temperature Evolution and Thermal Spread
5.5. Balloon Catheter Feasibility
5.6. Limitations
6. Conclusion
References
- Gibson KF, Kernohan WG. Lasers in medicine—A review. Journal of Medical Engineering & Technology. 1993;17(5):197–203. https://pubmed.ncbi.nlm.nih.gov/8366508/. [CrossRef]
- Walser EM, Zimmerer R, Nance A, Masood I, Saleem A. Anatomic and Clinical Effects of Focal Laser Ablation of the Prostate on Symptomatic Benign Prostatic Hyperplasia. Cancers. 2025;17(3):475. [CrossRef]
- Tafuri A, Panunzio A, De Carlo F, Luperto E, Di Cosmo F, Cavaliere A, et al. Transperineal Laser Ablation for Benign Prostatic Enlargement: A Systematic Review and Pooled Analysis of Pilot Studies. J Clin Med. 2023;12(5):1860. [CrossRef]
- Fainberg JS, Al Hussein Al Awamlh B, DeRosa AP, Chesnut GT, Coleman JA, Lee T, et al. A Systematic Review of Outcomes after Thermal and Nonthermal Partial Prostate Ablation. Prostate Int. 2021;9(4):169–175. [CrossRef]
- Brace CL. Microwave tissue ablation: biophysics, technology, and applications. Crit Rev Biomed Eng. 2010;38:1. [CrossRef]
- Yang C-H, Barbulescu D-V, Marian L, Tung M-C, Ou Y-C, Wu C-H. High-Intensity Focused Ultrasound Ablation in Prostate Cancer: A Systematic Review. J Pers Med. 2024;14(12):1163. [CrossRef]
- Chin YF, Lynn N. Systematic Review of Focal and Salvage Cryotherapy for Prostate Cancer. Cureus. 2022;14(6):e26400. [CrossRef]
- Elesta S.p.A. (n.d.). EchoLaser: First, Unique, Micro-invasive, Integrated System Offering Diagnostic Ultrasound and Laser Ablation Treatments. https://www.elesta-echolaser.com.
- Papadopoulou-Schultheiss A, Schulze-Osthoff K, Schultheiss G. Thermal cell injury: the significance of temperature and exposure time. Radiother Oncol. 1990;19(2):129–145. PMID:2343609.
- McIntosh R, Anderson V. A Comprehensive Tissue Properties Database Provided for the Thermal Assessment of a Human at Rest. Biophys Rev Lett. 2010;5(3):129−151. [CrossRef]
- Giering K, Lamprecht I, Minet O, Handke A. Determination of the Specific Heat Capacity of Healthy and Tumorous Human Tissue. Thermochim Acta. 1995;251:199–205. [CrossRef]
- Data on thermal conductivity of tissues including statistical information on the standard deviation and the spread in the values: https://itis.swiss/virtual-population/tissue-properties/database/thermal-conductivity/.
- Data on density of tissues including statistical information on the standard deviation and the spread in the values: https://itis.swiss/virtual-population/tissue-properties/database/density/.
- Data on heat capacity of tissues including statistical information on the standard deviation and the spread in the values: https://itis.swiss/virtual-population/tissue-properties/database/heat-capacity/.
- Germer CT, Roggan A, Ritz JP, Isbert C, Albrecht D, Müller G, Buhr HJ. Optical Properties of Native and Coagulated Human Liver Tissue and Liver Metastases in the Near Infrared Range. Lasers Surg Med. 1998;23(4):194–203. PMID: 9829430. [CrossRef]
- Adamczak L, Chmiel M, Florowski T, Pietrzak D. Estimation of Chemical Composition of Pork Trimmings by Use of Density Measurement-Hydrostatic Method. Molecules. 2020;25(7):1736. [CrossRef]
- Zhang L, Zhang Z, Zhang Y, Zhang X. Effects of Different Cooking Methods on the Physicochemical Properties and Sensory Quality of Pork Muscle. Food Sci Nutr. 2018;6(5):1185–1193. [CrossRef]
- Engineering Toolbox. Specific Heat Capacity of Foods. https://www.engineeringtoolbox.com/specific-heat-capacity-food-d_295.html.
- Leitman, S. (1967). Experimental determination of thermal conductivity of various types of meat, including pork. Georgia Institute of Technology. https://repository.gatech.edu/bitstreams/304e0095-b44b-4265-8289-0ad293dd3a70/download.
- Saccomandi, P., Vogel, V., Bazrafshan, B., Maurer, J., Schena, E., Vogl, T. J., Silvestri, S., & Mäntele, W. (2015). Estimation of anisotropy coefficient of swine pancreas, liver and muscle at 1064 nm based on goniometric technique. Journal of Biophotonics, 8(5), 422–428. https://onlinelibrary.wiley.com/doi/10.1002/jbio.201400057.
- Thomas, S. (2016). Property Tables and Charts (SI Units), Appendix 1. Wright State University, College of Engineering and Computer Science. https://cecs.wright.edu/people/faculty/sthomas/htappendix01.pdf.
- Bergmann, F., Foschum, F., Marzel, L., & Kienle, A. (2021). Ex Vivo Determination of Broadband Absorption and Effective Scattering Coefficients of Porcine Tissue. Photonics, 8(9), 365. [CrossRef]
- Mosca, S., Lanka, P., Stone, N., Konugolu Venkata Sekar, S., Matousek, P., Valentini, G., & Pifferi, A. (2020). Optical Characterization of Porcine Tissues from Various Organs in the 650–1100 nm Range Using Time-Domain Diffuse Spectroscopy. Biomedical Optics Express, 11(3), 1697–1706. (PubMed Central: PMC7075607). [CrossRef]

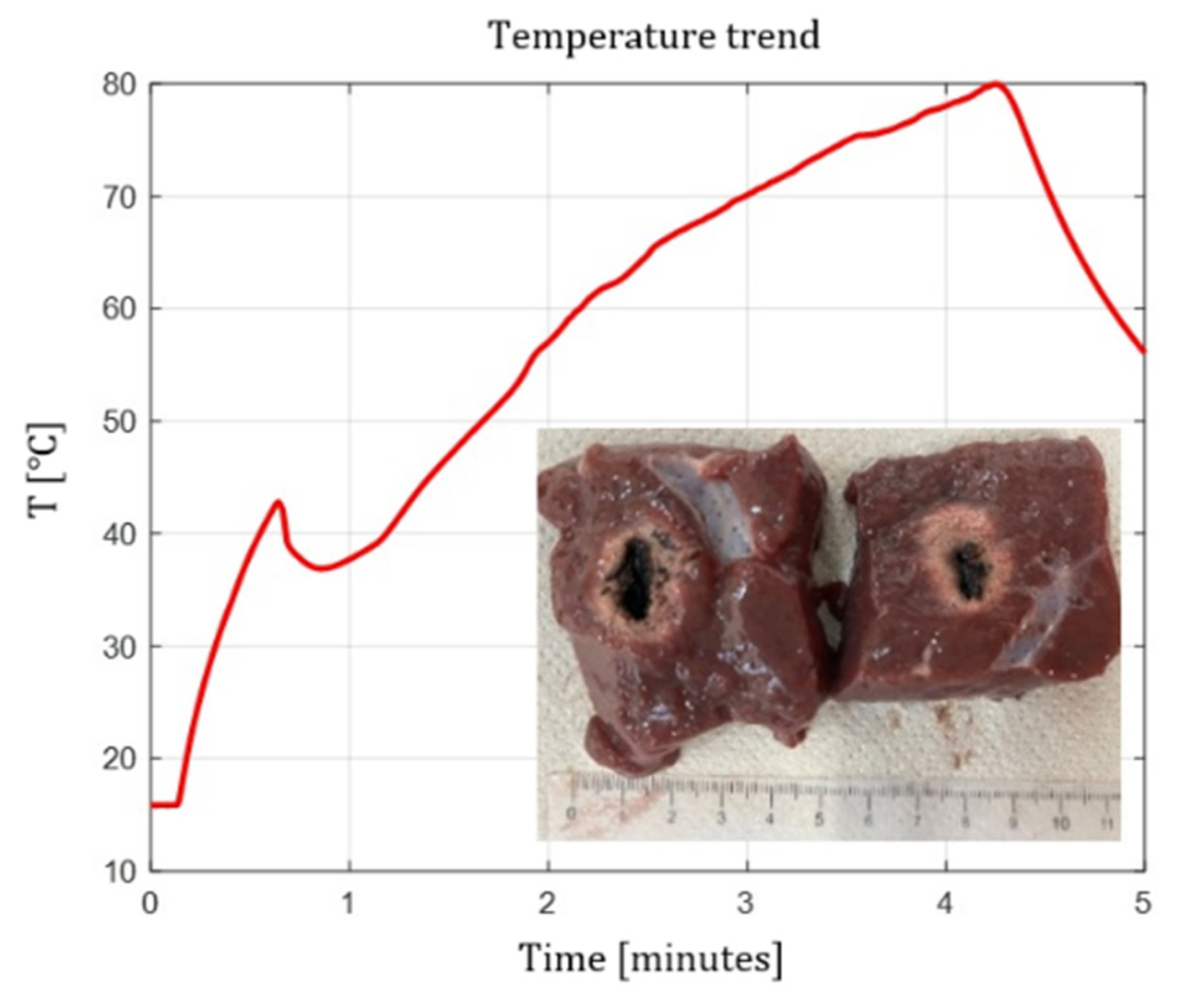
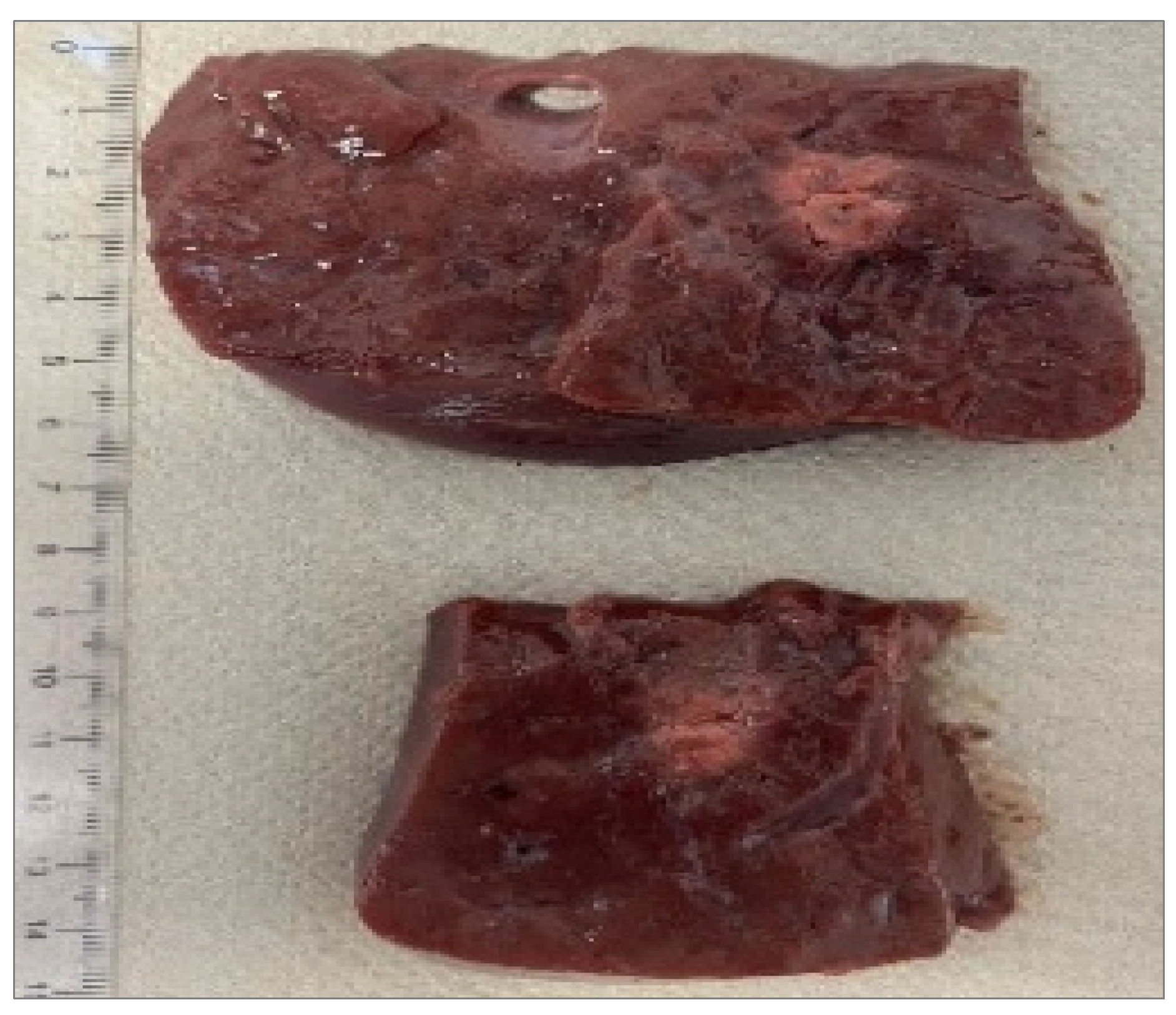
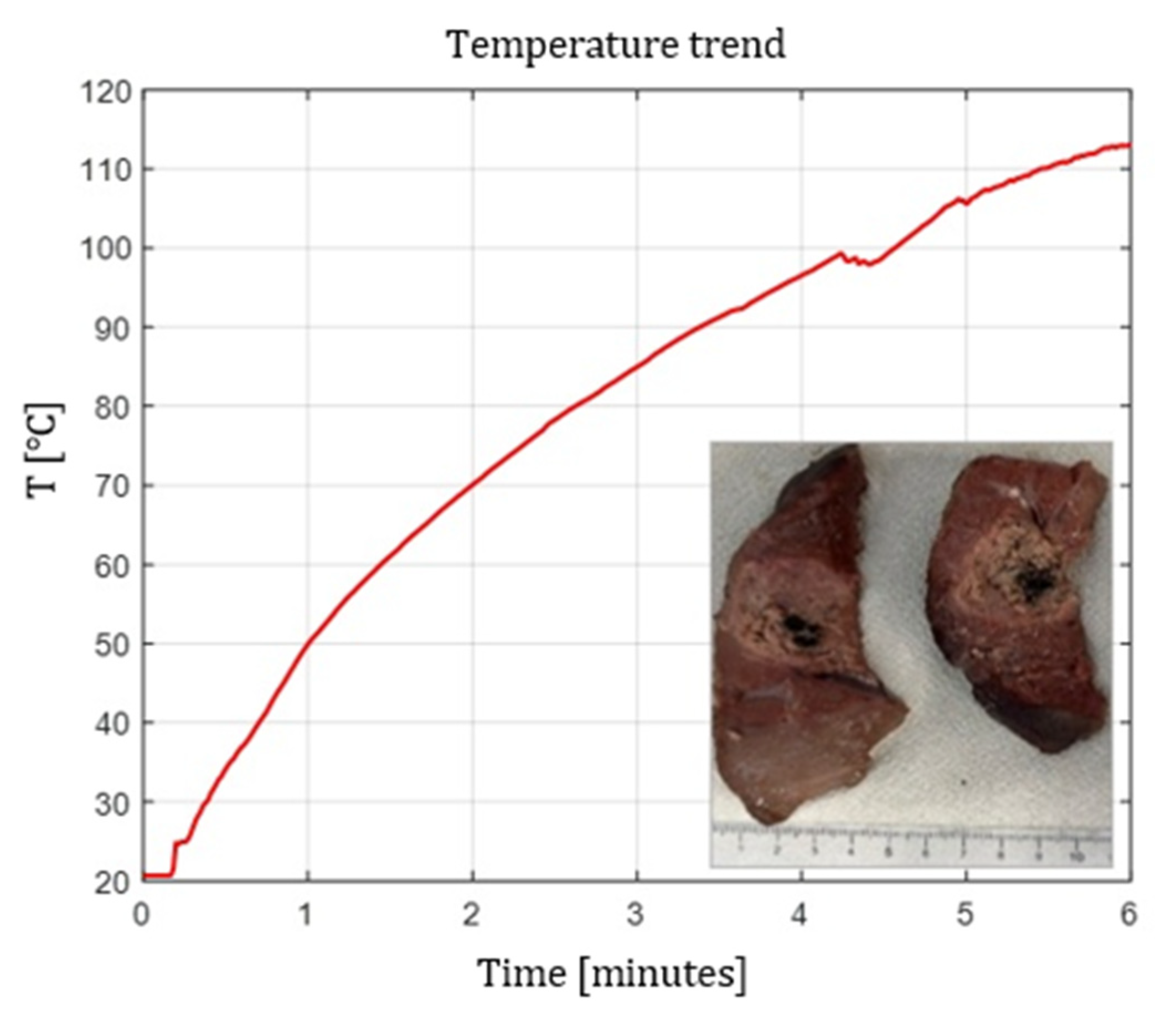
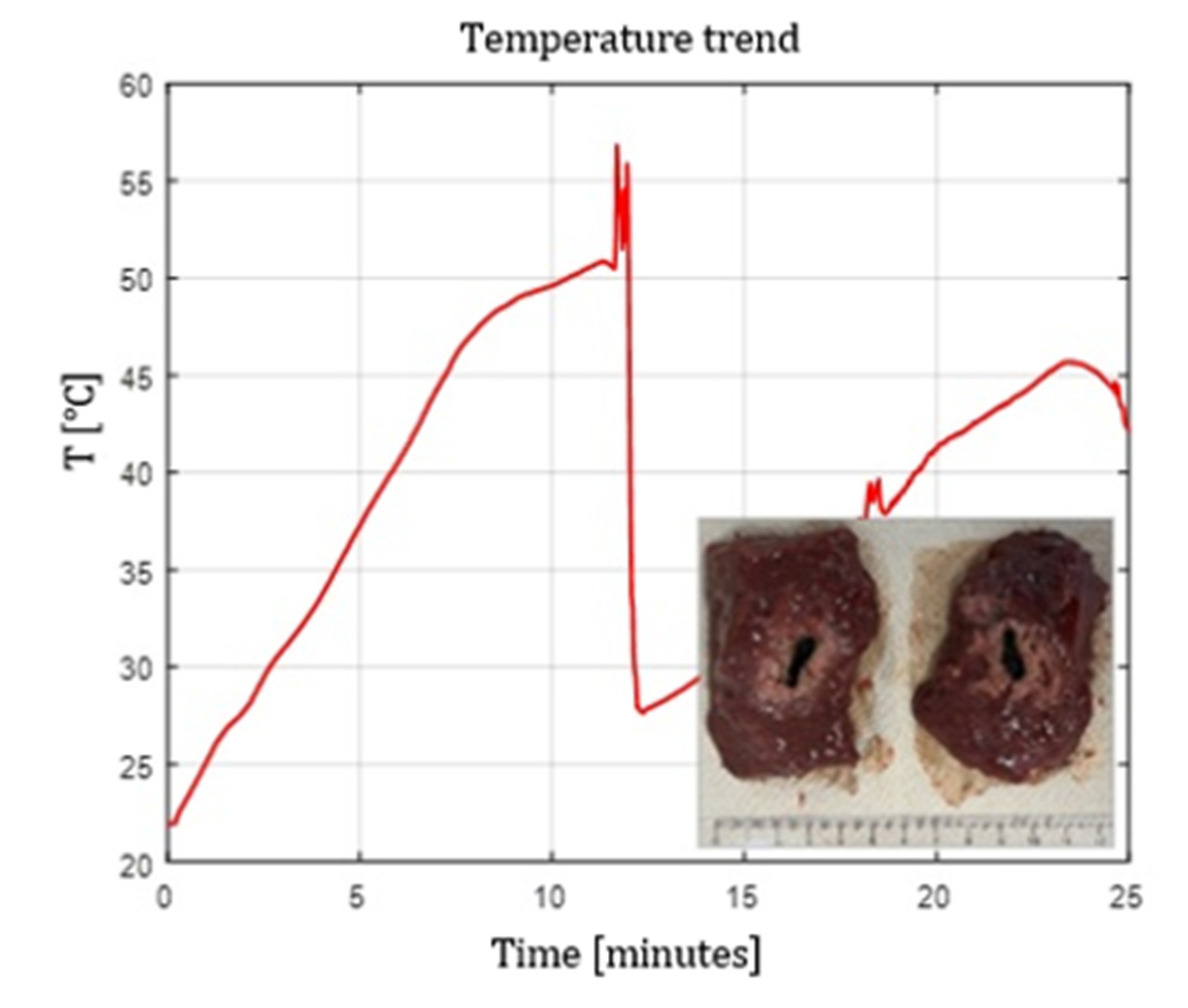
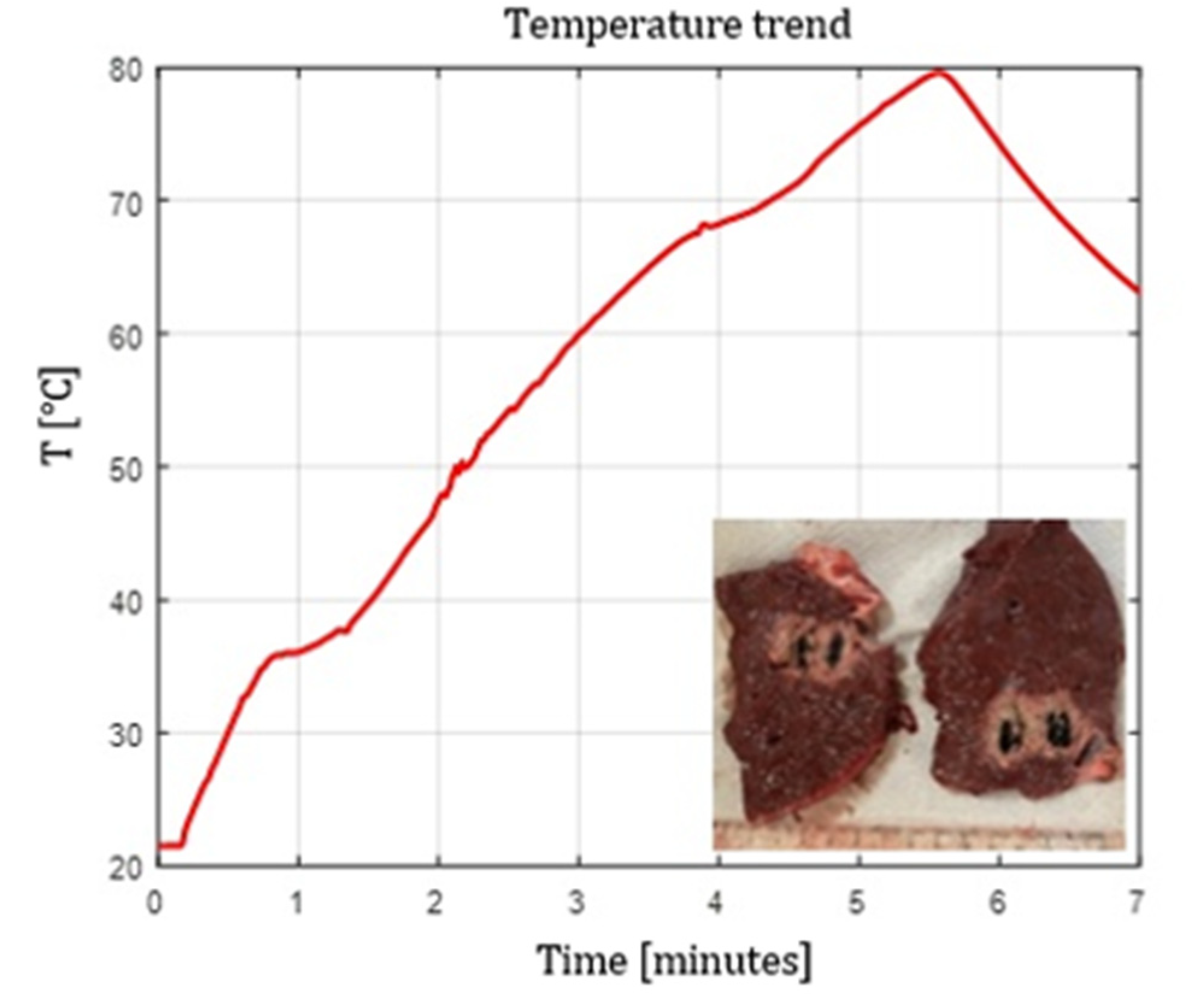
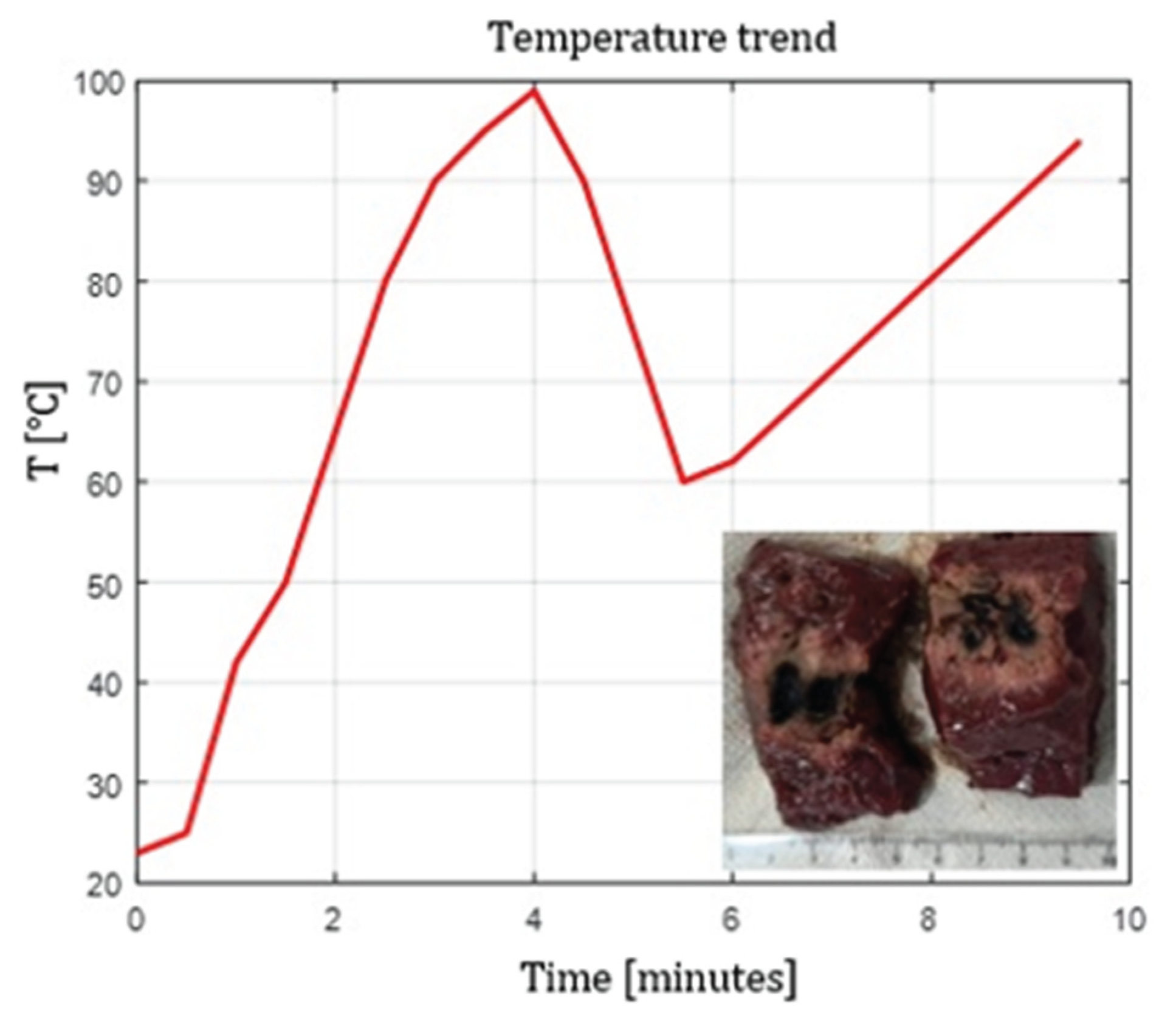
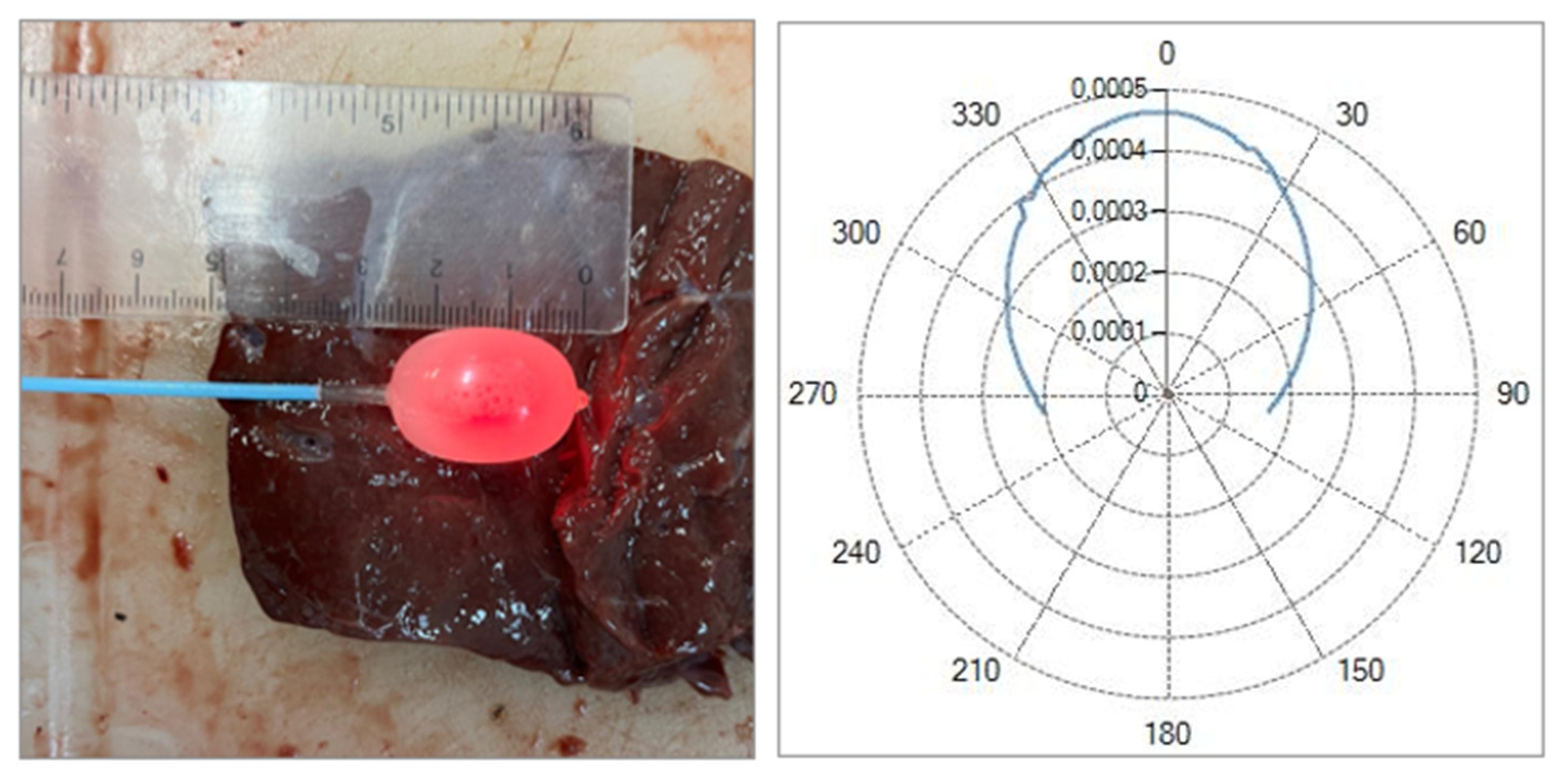
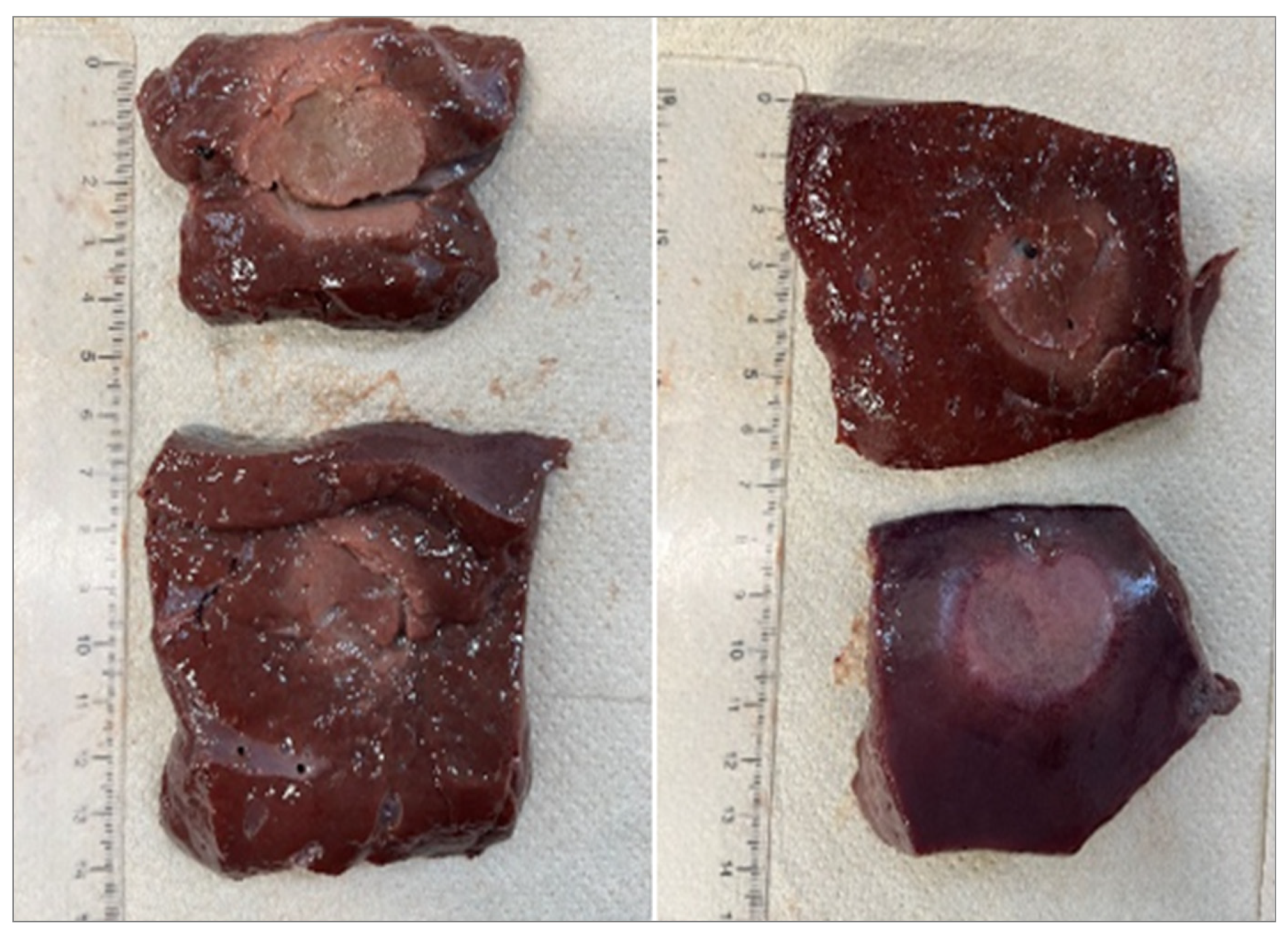
| Fiber ID | Core [µm] | OD [µm] | NA | Tip geometry | Manufacturer |
| N°1 | 272 | 420 | 0.22 | Flat tip | Oberon-Elesta custom |
| N°2 | 300 | 350 | 0.22 | Flat tip | Asclepion |
| N°3 | 300 | 650 | 0.37 | Flat tip | Thorlabs |
| N°4 | 400 | 730 | 0.37 | Flat tip | Oberon |
| N°5 | 600 | 1040 | 0.48 | Flat tip | Thorlabs |
| N°6 | 365 | 800 | 0.22 | Conical tip | Oberon-Elesta custom |
| N°7 | 400 | 950 | 0.22 | Conical tip | Oberon |
| N°8 | 600 | 890 | 0.22 | Curved ball tip | Oberon |
| Thermal properties |
% Water |
Optical properties (=1064 nm) | ||||
| Tissue density [kg m-3] | Specific heat[J kg-1 °C-1] | Thermal conductivity [W m-1 °C-1] | Absorption coefficient [cm-1] | Diffusion coefficient [cm-1] | ||
| Prostate | 1045 | 3715 | 0.51 | 82 | 0.4 | 110 |
| Calf liver | 1079 | 3540 | 0.52 | 75 – 81.9 | 0.3 – 0.5 | 150 – 169 |
| Porcine muscle | 1082 - 1100 | 3490 | 0.44 – 0.49 | 73 - 75 | 0.12 – 0.22 | 2.5 – 5.0 |
| Fiber | 3W@1800J | 5W@1800J | 7W@1800J |
| N°1 | L = [18.0 ± 0.9] mm W = [12.2 ± 1.0] mm ΔT = [30.9 ± 4.3] °C |
L = [19.3 ± 1.0] mm W = [12.5 ± 0.6] mm ΔT = [34.1 ± 3.2] °C |
L = [23.0 ± 1.2] mm W = [14.8 ± 1.0] mm ΔT = [48.0 ± 9.8] °C |
| N°2 | L = [20.0 ± 0.0] mm W = [13.0 ± 0.0] mm ΔT = [40.1 ± 0.0] °C |
L = [22.0 ± 0.0] mm W = [14.0 ± 0.0] mm ΔT = [45.4 ± 0.0] °C |
L = [23.5 ± 0.7] mm W = [14.0 ± 0.0] mm ΔT = [66.4 ± 0.0] °C |
| N°3 | L = [20.5 ± 0.7] mm W = [15.5 ± 0.7] mm ΔT = [34.2 ± 4.7] °C |
L = [22.5 ± 0.7] mm W = [15.5 ± 0.7] mm ΔT = [41.6 ± 0.0] °C |
L = [24.5 ± 0.7] mm W = [17.0 ± 1.4] mm ΔT = [56.6 ± 0.0] °C |
| N°4 | L = [20.0 ± 0.0] mm W = [13.5 ± 0.7] mm ΔT = [38.3 ± 5.6] °C |
L = [23.0 ± 0.0] mm W = [15.0 ± 0.0] mm ΔT = [40.7 ± 4.5] °C |
L = [28.0 ± 0.0] mm W = [17.0 ± 0.0] mm ΔT = [54.7 ± 0.0] °C |
| N°5 | L = [21.5 ± 0.7] mm W = [15.5 ± 0.7] mm ΔT = [45.3 ± 1.6] °C |
L = [23.0 ± 1.0] mm W = [16.0 ± 1.0] mm ΔT = [55.3 ± 0.0] °C |
L = [24.5 ± 0.7] mm W = [16.0 ± 1.4] mm ΔT = [64.2 ± 0.0] °C |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




