Submitted:
02 January 2026
Posted:
04 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
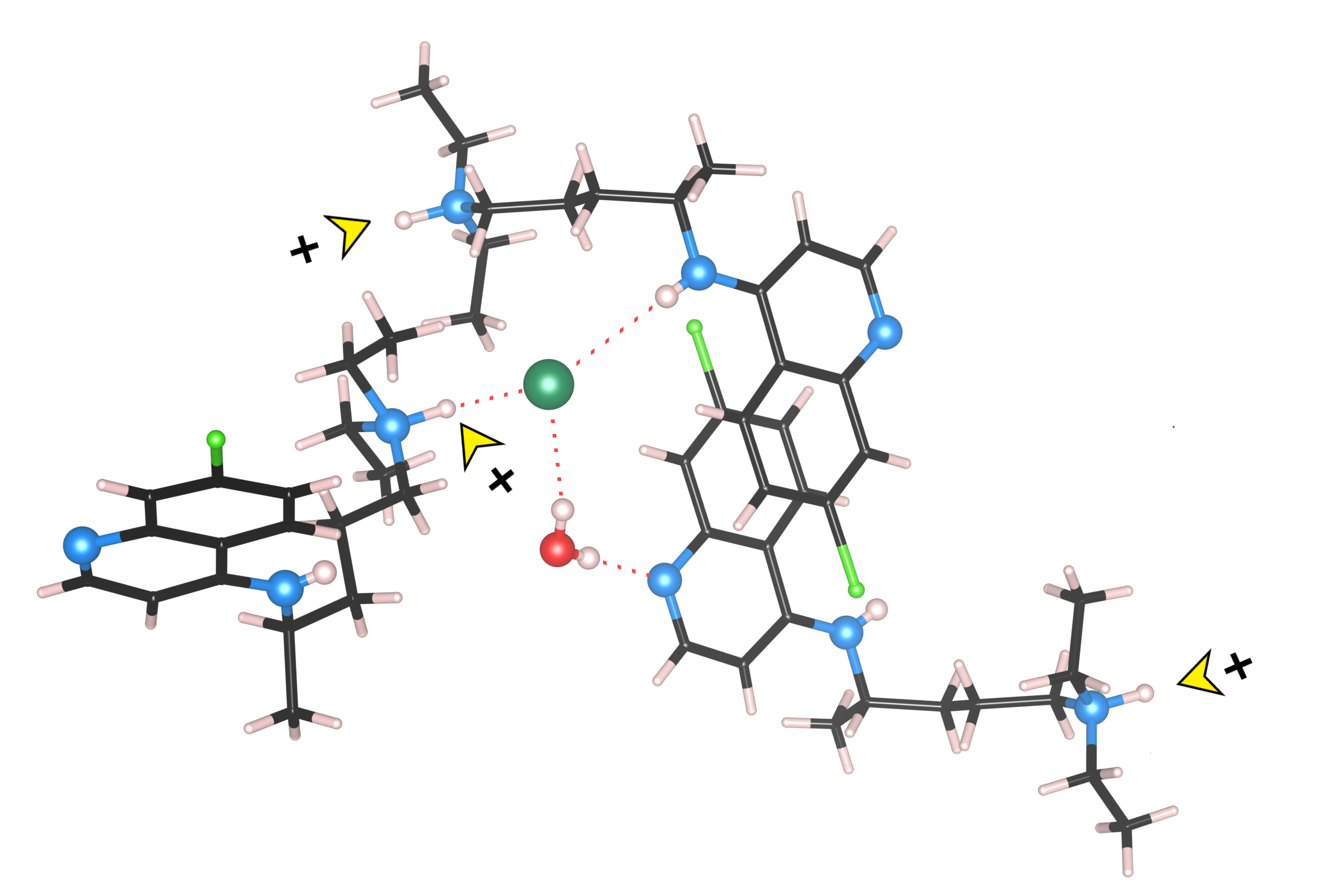
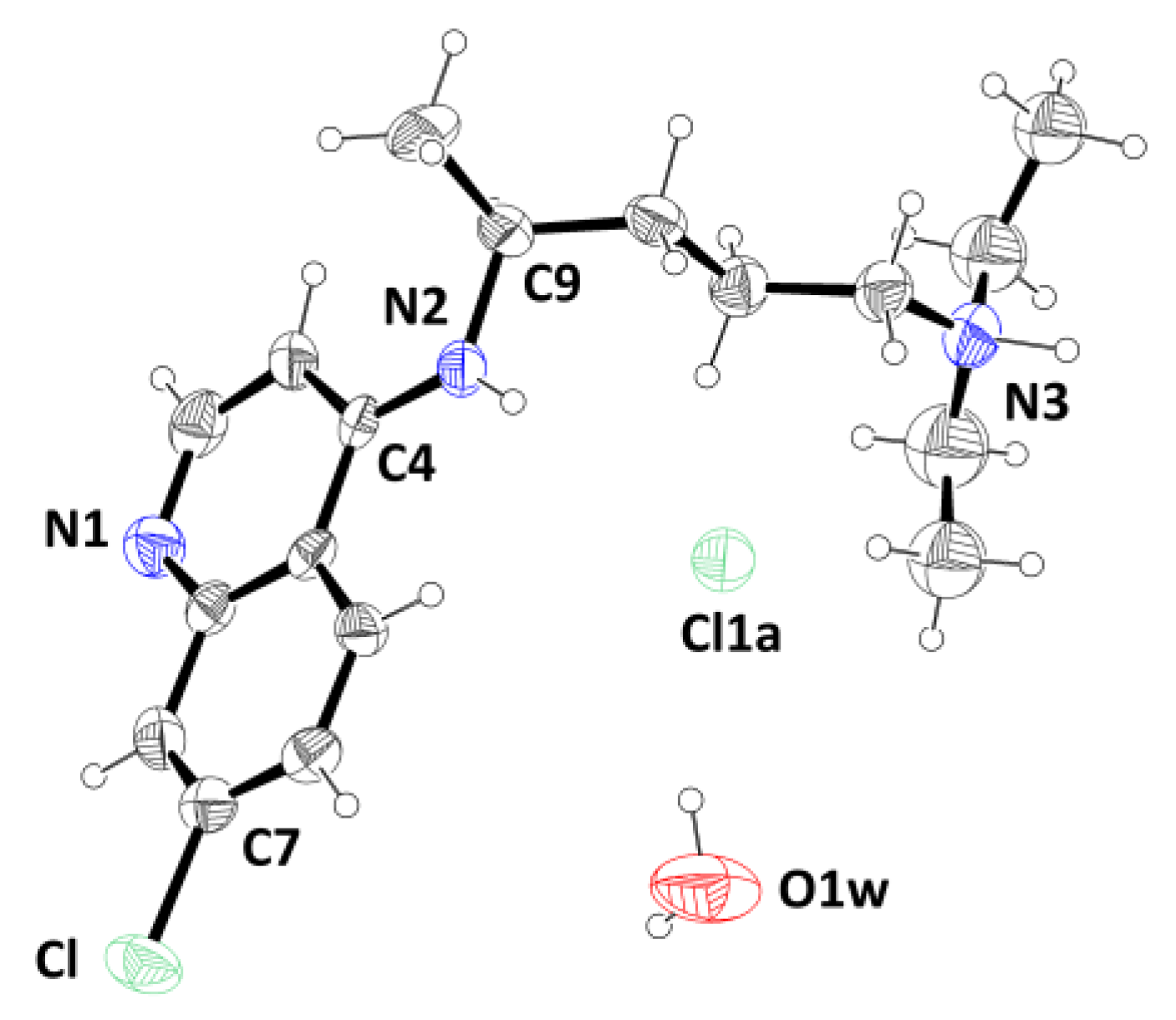
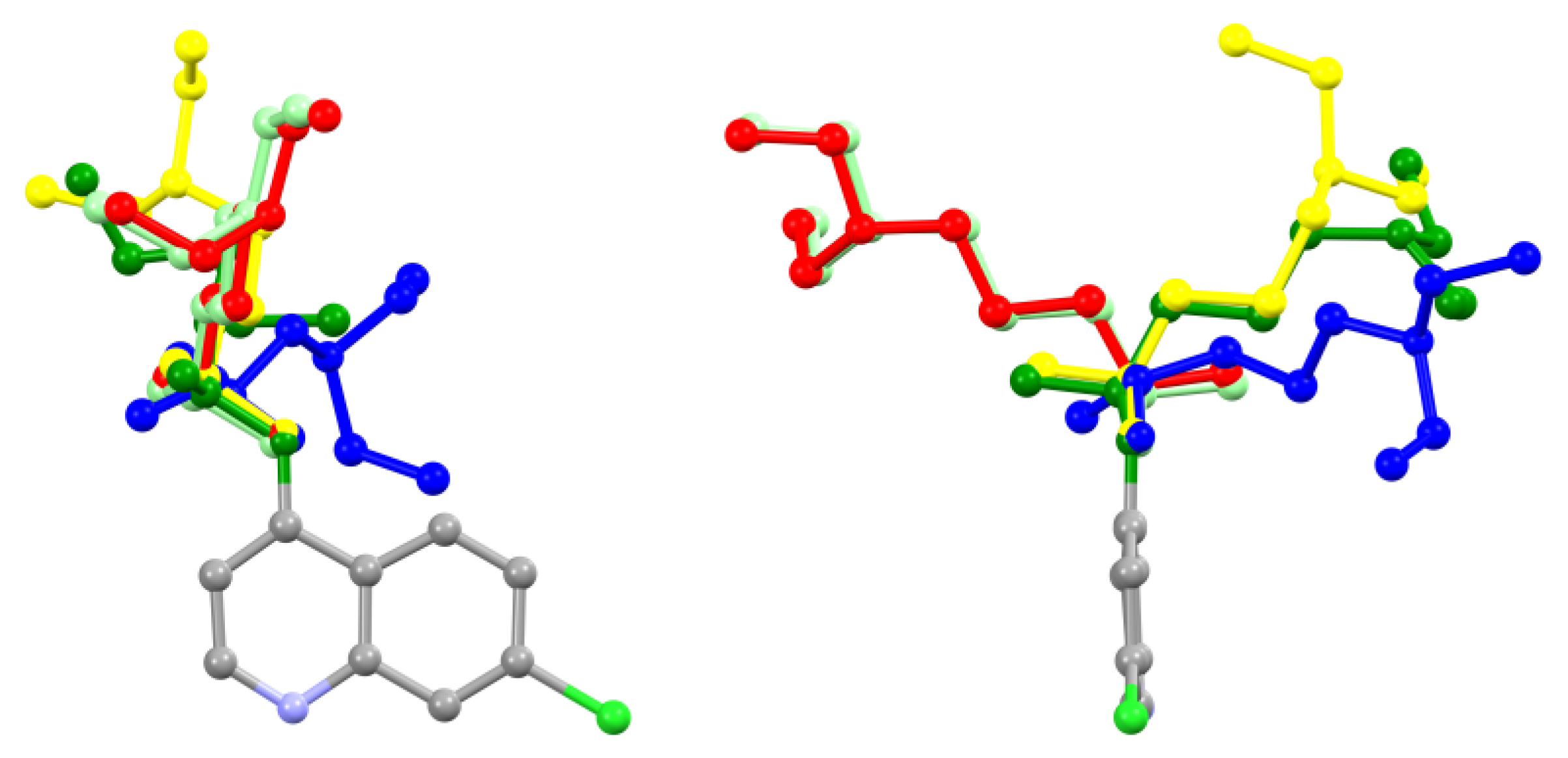
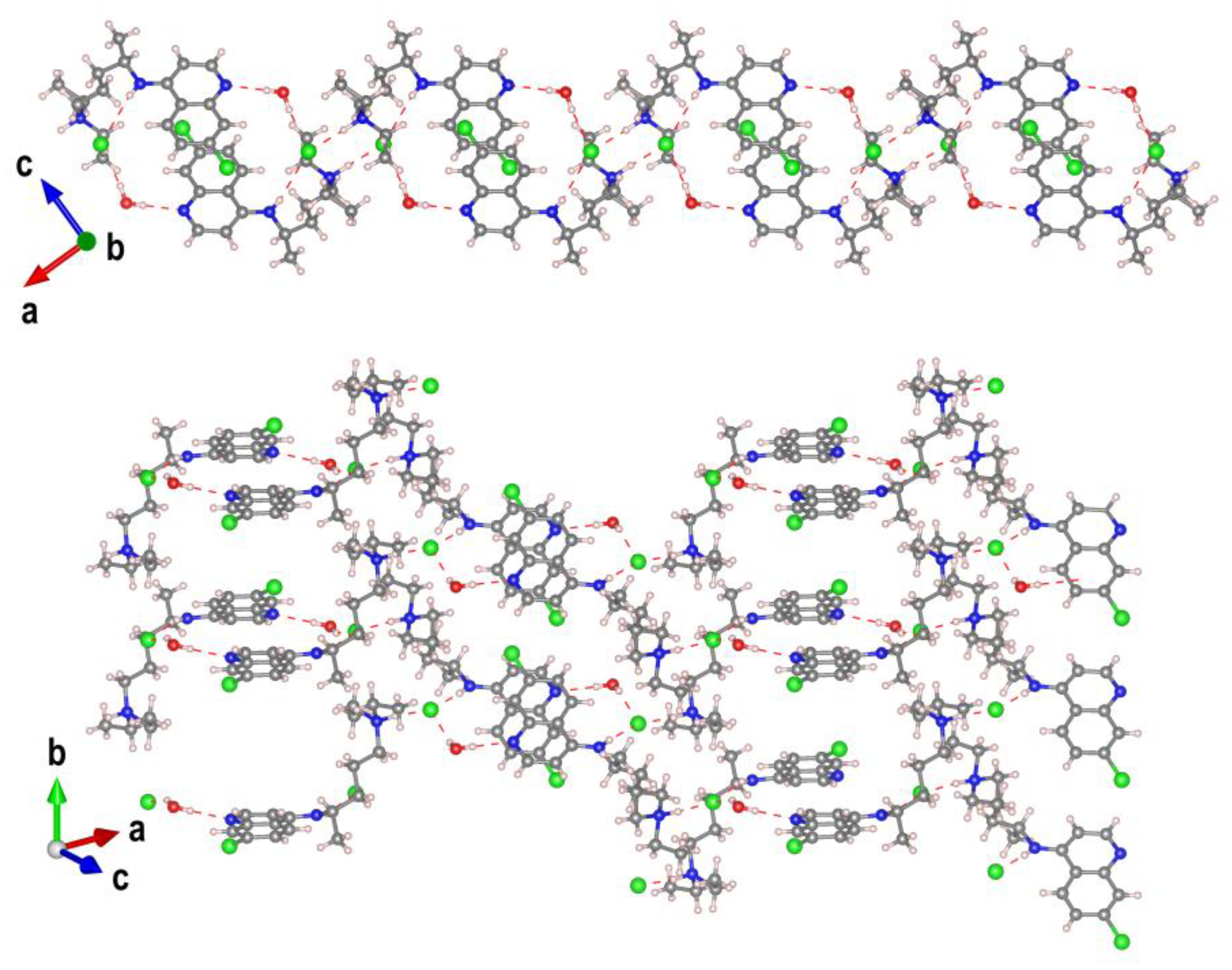
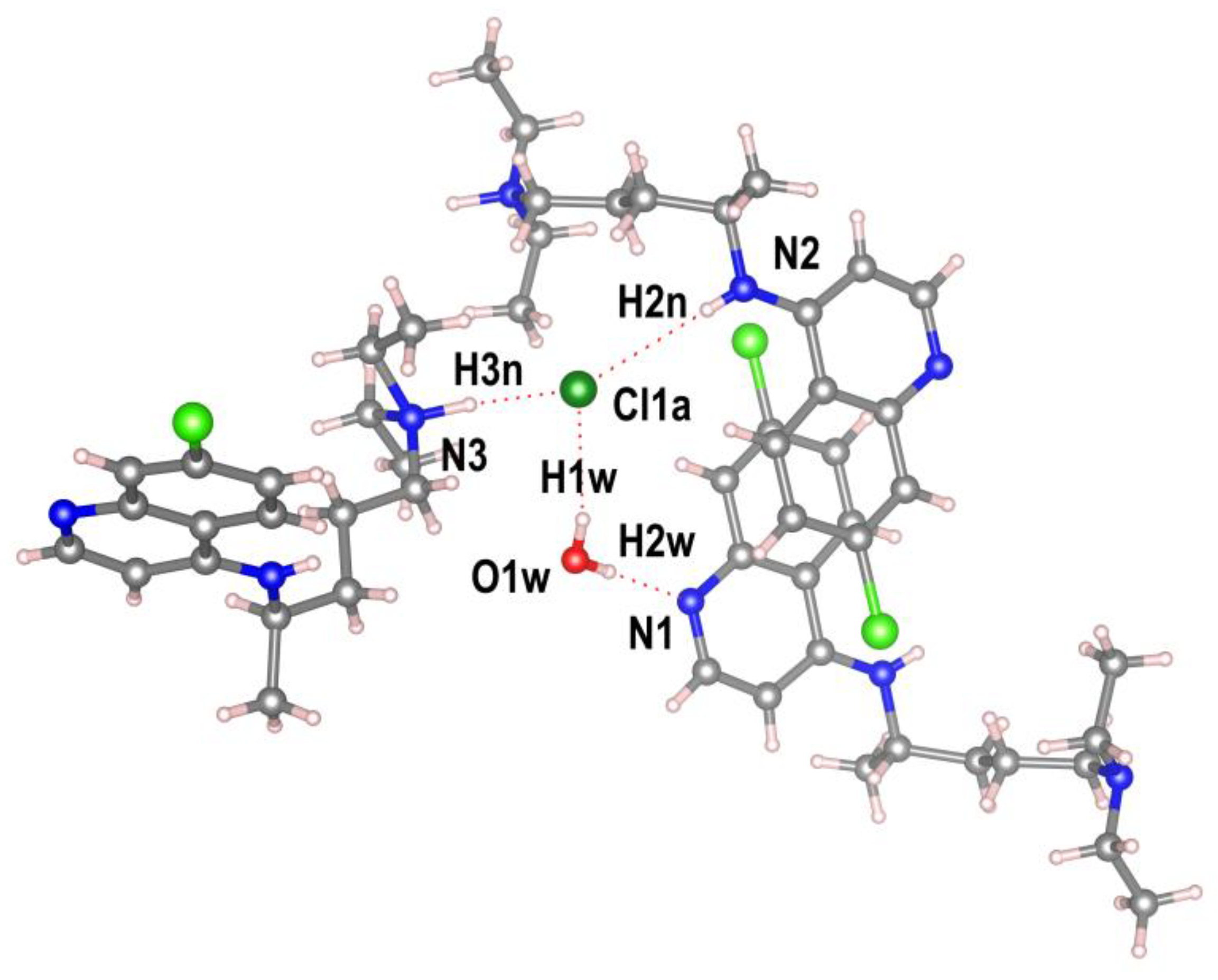
2.1. Solid-State Structure
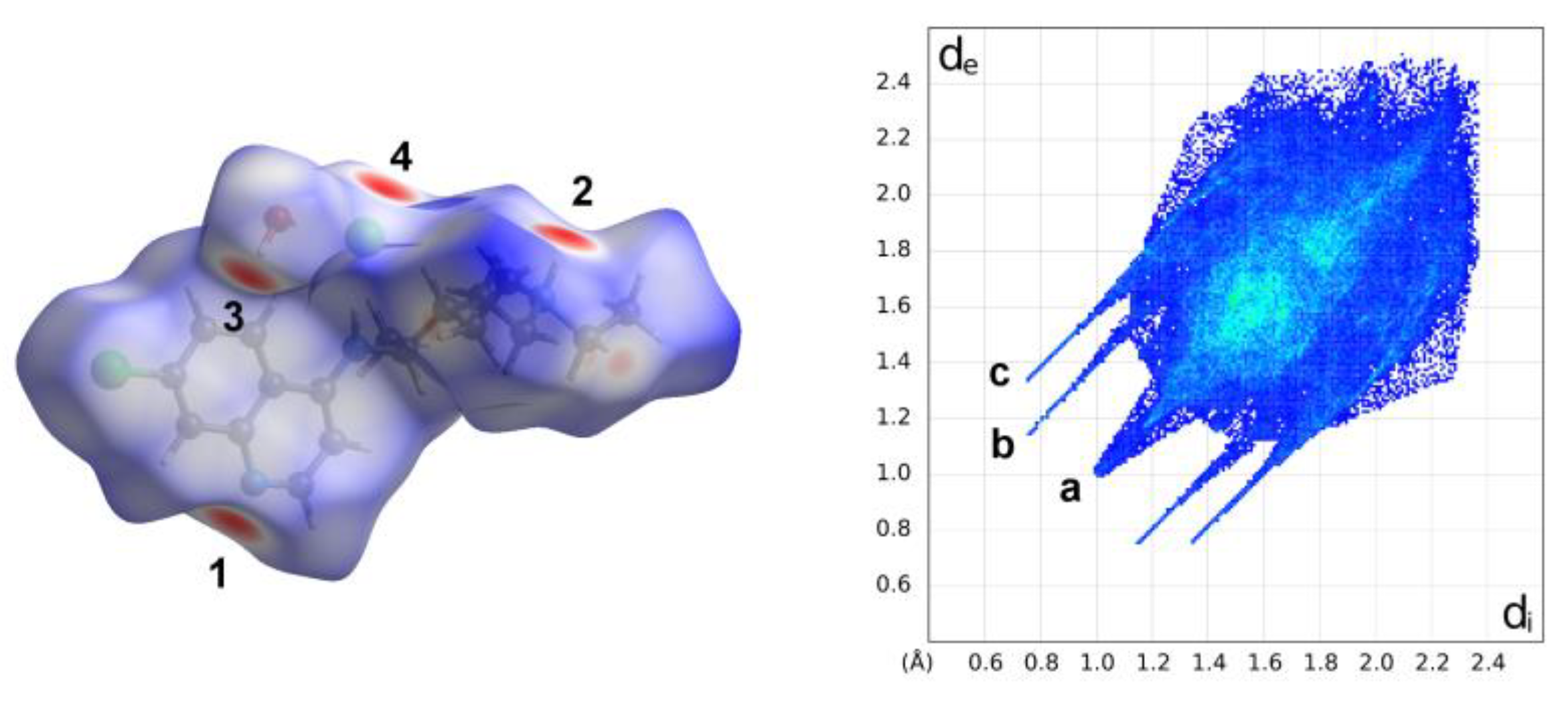
2.2. Intermolecular Interaction Analysis
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Synthesis
4.2.1. Free Base CQ
4.2.2. Salt Crystallization
4.3. Single-Crystal X-Ray Diffraction
4.4. X-Ray Powder Diffraction
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CQ | Chloroquine |
| CQCl·H2O | Chloroquine hydrochloride monohydrate |
| CQDP | Chloroquine diphosphate |
| AQ | Amodiaquine |
| PIP | Piperaquine |
| HCQ | Hydroxychloroquine |
| PXRD | X-ray Powder Diffraction |
| HS | Hirshfeld Surface |
| FP | 2D Fingerprints Plot |
References
- Zhou, W.; Wang, H.; Yang, Y.; Chen, Z.-S.; Zou, C.; Zhang, J. Chloroquine against Malaria, Cancers and Viral Diseases. Drug Discov. Today 2020, 25, 2012–2022. [Google Scholar] [CrossRef]
- Al-Bari, M.A.A. Chloroquine Analogues in Drug Discovery: New Directions of Uses, Mechanisms of Actions and Toxic Manifestations from Malaria to Multifarious Diseases. J. Antimicrob. Chemother. 2015, 70, 1608–1621. [Google Scholar] [CrossRef]
- Shah, S.M.A.; Rasheed, T.; Rizwan, K.; Bilal, M.; Iqbal, H.M.N.; Rasool, N.; Toma, S.; Marceanu, L.G.; Bobescu, E. Risk. Management Strategies and Therapeutic Modalities to Tackle COVID-19/SARS-CoV-2. J. Infect. Public Health 2021, 14, 331–346. [Google Scholar] [CrossRef] [PubMed]
- Verbeeck, R.K.; Junginger, H.E.; Midha, K.K.; Shah, V.P.; Barends, D.M. Biowaiver Monographs for Immediate Release Solid Oral Dosage Forms Based on Biopharmaceutics Classification System (BCS) Literature Data: Chloroquine Phosphate, Chloroquine Sulfate, and Chloroquine hydrochloride. J. Pharm. Sci. 2005, 94, 1389–1395. [Google Scholar] [CrossRef] [PubMed]
- Browning, D.J. Pharmacology of Chloroquine and Hydroxychloroquine. In Hydroxychloroquine and Chloroquine Retinopathy; Springer New York: New York, NY, 2014; pp. 35–63. ISBN 978-1-4939-0597-3. [Google Scholar]
- De Souza Santos, M.; De Morais Del Lama, M.P.F.; Siuiti Ito, A.; Zumstein Georgetto Naal, R.M. Binding of Chloroquine to Ionic Micelles: Effect of pH and Micellar Surface Charge. J. Lumin. 2014, 147, 49–58. [Google Scholar] [CrossRef]
- Espeau, P. Special Issue Pharmaceutical Solid Forms: From Crystal Structure to Formulation. Pharmaceutics 2025, 17, 312. [Google Scholar] [CrossRef] [PubMed]
- Iredale, J.; Fieger, H.; Wainer, I.W. Determination of the Stereoisomers of Hydroxychloroquine and Its Major Metabolites in Plasma and Urine Following a Single Oral Administration of Racemic Hydroxychloroquine. Semin. Arthritis Rheum. 1993, 23, 74–81. [Google Scholar] [CrossRef]
- Kalia, S.; Dutz, J.P. New Concepts in Antimalarial Use and Mode of Action in Dermatology. Dermatol. Ther. 2007, 20, 160–174. [Google Scholar] [CrossRef]
- Albesa-Jové, D.; Pan, Z.; Harris, K.D.M.; Uekusa, H. A Solid-State Dehydration Process Associated with a Significant Change in the Topology of Dihydrogen Phosphate Chains, Established from Powder X-Ray Diffraction. Cryst. Growth Des. 2008, 8, 3641–3645. [Google Scholar] [CrossRef]
- Furuseth, S.; Karlsen, J.; Mostad, A.; Romming, C.A.; Salmén, R.; Tønnesen, H.H.; Tokii, T. N4-(7-Chloro-4-Quinolinyl)-N1,N1-Diethyl-1,4-Pentanediamine. An X-Ray Diffraction Study of Chloroquine Diphosphate Hydrate. Acta Chem. Scand. 1990, 44, 741–745. [Google Scholar] [CrossRef]
- Macetti, G.; Loconte, L.; Rizzato, S.; Gatti, C.; Presti, L.L. Intermolecular Recognition of the Antimalarial Drug Chloroquine: A Quantum Theory of Atoms in Molecules-Density Functional Theory Investigation of the Hydrated Dihydrogen Phosphate Salt from the 103 K X-Ray Structure. Cryst. Growth Des. 2016, 16, 6043–6054. [Google Scholar] [CrossRef]
- Courseille, C.; Busetta, B.; Hospital, M. 7-Chloro-4(4-Diethylamino-1-Methylbutyl-Amino) Quinoline. Cryst.Struct.Commun. 1973, 2, 283. [Google Scholar]
- Macrae, C.F.; Sovago, I.; Cottrell, S.J.; Galek, P.T.A.; McCabe, P.; Pidcock, E.; Platings, M.; Shields, G.P.; Stevens, J.S.; Towler, M.; et al. Mercury 4.0: From Visualization to Analysis, Design and Prediction. J. Appl. Crystallogr. 2020, 53, 226–235. [Google Scholar] [CrossRef]
- Spackman, P.R.; Turner, M.J.; McKinnon, J.J.; Wolff, S.K.; Grimwood, D.J.; Jayatilaka, D.; Spackman, M.A. CrystalExplorer: A Program for Hirshfeld Surface Analysis, Visualization and Quantitative Analysis of Molecular Crystals. J Appl Crystallogr 2021, 54, 1006–1011. [Google Scholar] [CrossRef] [PubMed]
- Soave, Raffaella. CCDC 2130150: Experimental Crystal Structure Determination.
- Paulikat, M.; Vitone, D.; Schackert, F.K.; Schuth, N.; Barbanente, A.; Piccini, G.; Ippoliti, E.; Rossetti, G.; Clark, A.H.; Nachtegaal, M.; et al. Molecular Dynamics and Structural Studies of Zinc Chloroquine Complexes. J. Chem. Inf. Model. 2023, 63, 161–172. [Google Scholar] [CrossRef]
- Squarcina, A.; Franke, A.; Senft, L.; Onderka, C.; Langer, J.; Vignane, T.; Filipovic, M.R.; Grill, P.; Michalke, B.; Ivanović-Burmazović, I. Zinc Complexes of Chloroquine and Hydroxychloroquine versus the Mixtures of Their Components: Structures, Solution Equilibria/Speciation and Cellular Zinc Uptake. J. Inorg. Biochem. 2024, 252, 112478. [Google Scholar] [CrossRef]
- Rizzato, S. Unpublished results (Manuscript in Preparation).
- Cotting, G.; Urquidi, O.; Besnard, C.; Brazard, J.; Adachi, T.B.M. The Effect of Salt Additives on the Glycine Crystallization Pathway Revealed by Studying One Crystal Nucleation at a Time. Proc. Natl. Acad. Sci. 2025, 122, e2419638122. [Google Scholar] [CrossRef]
- Putra, O.D.; Pettersen, A.; Nilsson Lill, S.O.; Umeda, D.; Yonemochi, E.; Nugraha, Y.P.; Uekusa, H. Capturing a New Hydrate Polymorph of Amodiaquine Dihydrochloride Dihydrate via Heterogeneous Crystallisation. CrystEngComm 2019, 21, 2053–2057. [Google Scholar] [CrossRef]
- Takenaka, H.; Kawashima, Y.; Lin, S.Y. Polymorphism of Spray-Dried Microencapsulated Sulfamethoxazole with Cellulose Acetate Phthalate and Colloidal Silica, Montmorillonite, or Talc. J. Pharm. Sci. 1981, 70, 1256–1260. [Google Scholar] [CrossRef]
- Tipduangta, P.; Takieddin, K.; Fábián, L.; Belton, P.; Qi, S. A New Low Melting-Point Polymorph of Fenofibrate Prepared via Talc Induced Heterogeneous Nucleation. Cryst. Growth Des. 2015, 15, 5011–5020. [Google Scholar] [CrossRef]
- Braga, D.; Grepioni, F.; Chelazzi, L.; Nanna, S.; Rubini, K.; Curzi, M.; Giaffreda, S.L.; Saxell, H.E.; Bratz, M.; Chiodo, T. Bentazon: Effect of Additives on the Crystallization of Pure and Mixed Polymorphic Forms of a Commercial Herbicide. Cryst. Growth Des. 2014, 14, 5729–5736. [Google Scholar] [CrossRef]
- Bruker APEX2 V2014.1-1. Bruker AXS Inc.: Madison, Wisconsin, USA, 2015.
- Bruker SAINT v8.34A, XPREP V2013/3, SADABS. Bruker AXS Inc.: Madison, Wisconsin, USA, 2013.
- Sheldrick, G.M. Crystal Structure Refinement with SHELXL. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Farrugia, L.J. WinGX and ORTEP for Windows: An Update. J. Appl. Crystallogr. 2012, 45, 849–854. [Google Scholar] [CrossRef]
- Momma, K.; Izumi, F. VESTA 3 for Three-Dimensional Visualization of Crystal, Volumetric and Morphology Data. J. Appl. Crystallogr. 2011, 44, 1272–1276. [Google Scholar] [CrossRef]
- Rodriguez-Carvajal, J. FULLPROF: A Program for Rietveld Refinement and Pattern-Matching Analysis. In Proceedings of the Abstracts of the meeting Powder Diffraction, Toulouse, France, 1990; pp. 127–128. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



