Submitted:
02 January 2026
Posted:
04 January 2026
You are already at the latest version
Abstract
Keywords:
Introduction
| Year | Country (and affiliation) of authors [reference] | Oil |
| Oil-in-water emulsions | ||
| 1999-2018 | Japan [1,2,3,4,5,6] |
|
| 2003-2004 | University of California, USA [7,8] |
|
| 2004 | Bristol University, UK [9] |
|
| 2004, 2025 | Beattie and Djerdjev from Sydney University, Australia [10,11] |
|
| 2020 | China [17] |
|
| 2022 | Korea [12] |
|
| 2023 | China, Japan, Canada [16] |
|
| 2025 | USA and China [18] |
|
| 2018,2025 | France [20,21,22] |
|
| Water-in-oil emulsion | ||
| 2010-2018 | Japan [13,14,15] |
|
Materials
Sample Preparation and Measurement Protocol
Instruments
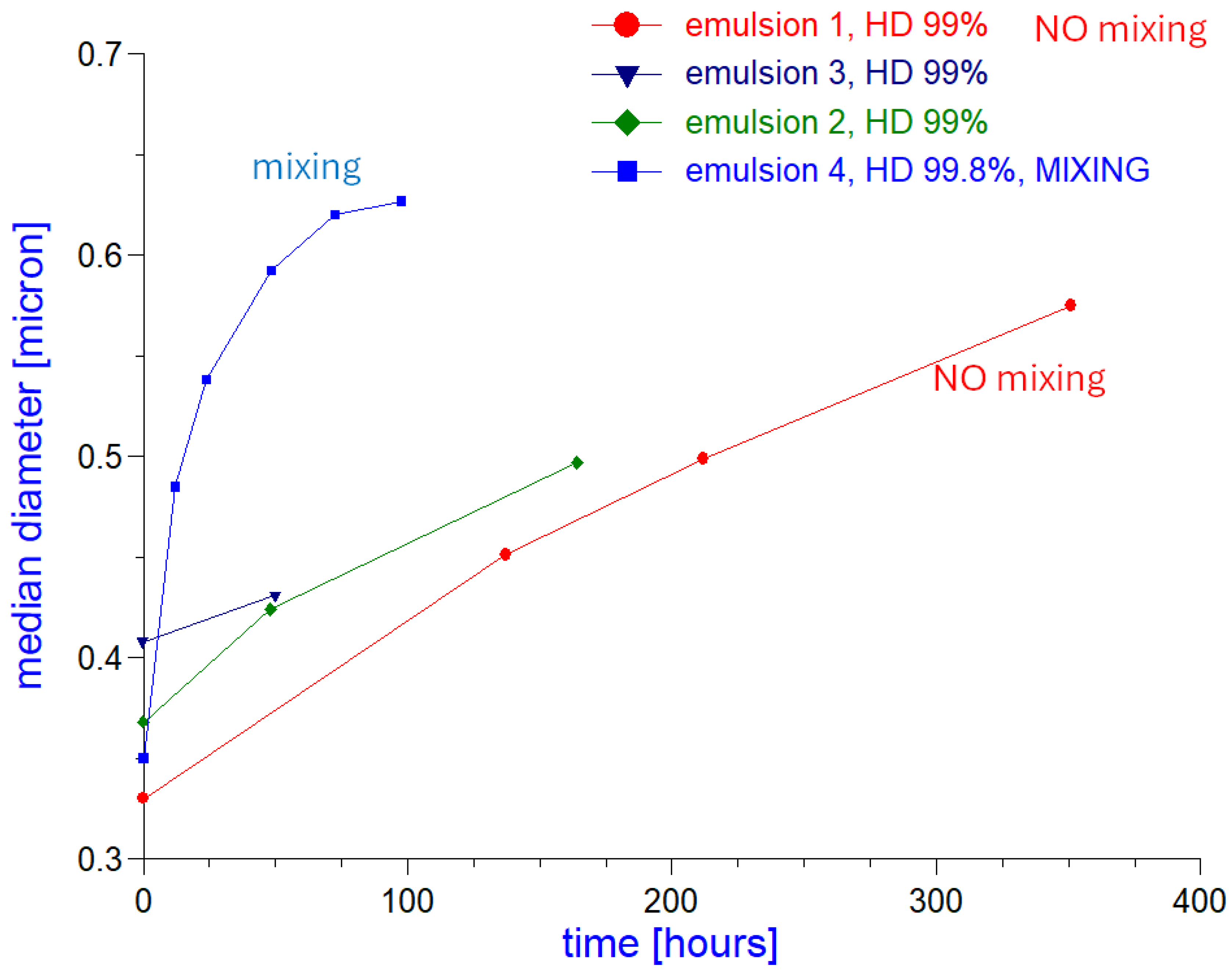
Experimental Results
Discussion
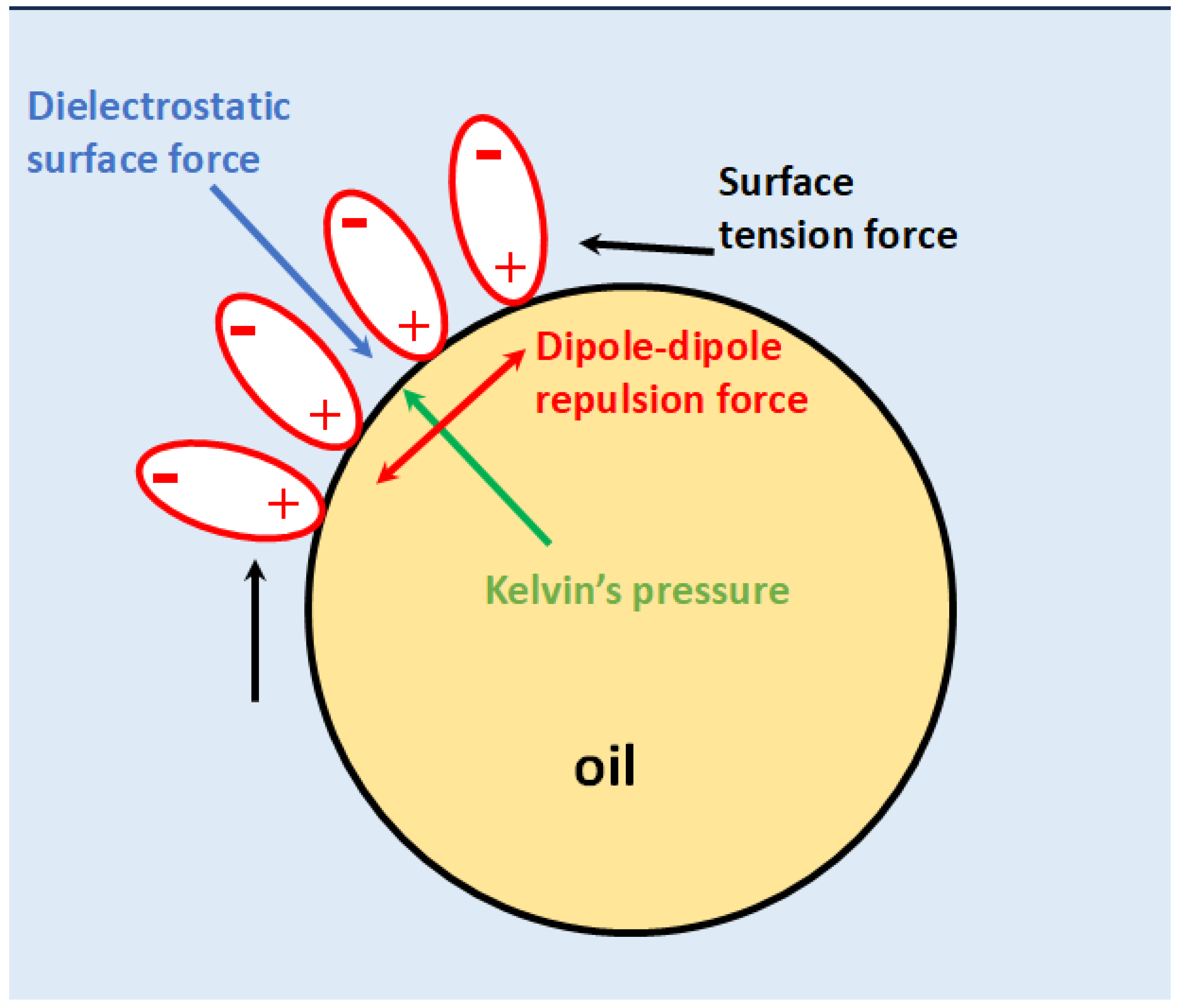
Dielectrostatic Force Determines Stable Size
Repulsion of Oriented Water Dipoles
Balance of Forces at Pristine Interfaces with Structured Water Layer
What Liquids Could Form “Pristine Emulsion”
Conclusions
Appendix 1. Arguments Against Hypothesis of Impurities as Origin of Observed Emulsification
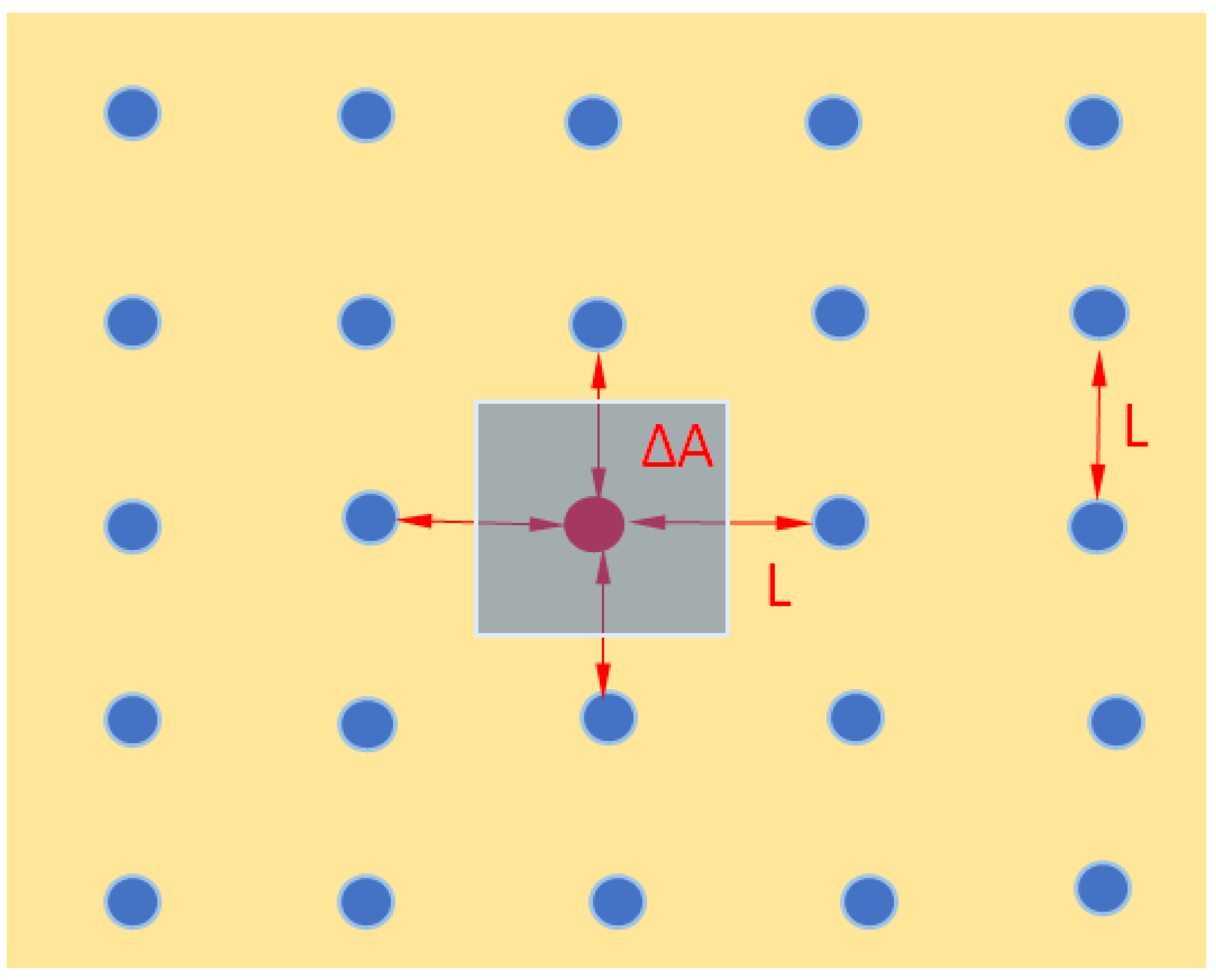
Appendix 2. Calculation of Lifshitz-Slyozov-Wagner (LSW) [28,29] Time for Kinetics of Ostwald Ripening [24].
- a is an average radius of the droplets in [m]
- t is time in [sec]
- D is diffusion coefficient of the emulsion droplet liquid in [m2/sec]
- Csol is solubility in [g/m3]
- γ is surface tension in [N/m]
- M is molecular weight in [g/mol]
- ρ is density in [g/m3]
- R is gas constant in [J/K0 mol]
- T is absolute temperature in K0.
References
- Kamogawa, K; Matsumoto, M; Kobayashi, T; Sakai, T; Sakai, H; Abe, M. Langmuir 1999, 15, 1913–1919. [CrossRef]
- Sakai, T; Kamogawa, K; Harusawas, F; Momozawa, N; Sakai, H; Abe, M. “Direct observation of flocculation/coalescence of metastable oil droplets in surfactant-free oil/water emulsion by freeze-fracture electron microscopy”. Langmuir 2001, 17(2), 255–259. [Google Scholar] [CrossRef]
- Kamogawa, K; Akatsuka, H; Matsumoto, M; Yokoyama, S; Sakai, T; Sakai, H; Abe, M. “Surfactant-free o/w emulsion formation of oleic acid and its esters with ultrasonic dispersion”. Colloids and Surfaces, A 2001, 180, 41–53. [Google Scholar] [CrossRef]
- Sakai, T; Kamogawa, K; Nishiama, K; Sakai, H; Abe, M. “Molecular diffusion of oil/water emulsions in surfactant-free conditions”. Langmuir 2002, 18(6), 1985–1990. [Google Scholar] [CrossRef]
- Sakai, T; Takeda, Y; Mafune, F; Abe, M; Kondow, T. “Monitoring growth of surfactant-free nanodroplets dispersed in water by single-droplet detection”. J. Phys. Chem. 2003, 107(13), 2921–2926. [Google Scholar] [CrossRef]
- Sakai, T. “Surfactant-free emulsions”. Current Opinion Colloid Interface Science 2008, 13(4), 228–235. [Google Scholar] [CrossRef]
- Pashley, RM. “Effect of degassing on the formation and stability of surfactant-free emulsions and fine Teflon dispersions”. J. Phys. Chem. 2003, B 107, 1714–1720. [Google Scholar] [CrossRef]
- Maeda, N; Rosenberg, KJ; Israelachvili, JN; Pashley, RM. “Further studies on the effect of degassing on the dispersion and stability of surfactant-free emulsions”. Langmuir 2004, 20, 3129–3137. [Google Scholar] [CrossRef] [PubMed]
- Burnett, GR; Atkin, R; Hicks, S; Eastoe, J. “Surfactant-free “emulsions” generated by freeze-thaw”. Langmuir 2004, 20, 5673–5678. [Google Scholar] [CrossRef] [PubMed]
- Beattie, JK; Djerdjev, AM. “The Pristine Oil/Water Interface: Surfactant-Free Hydroxide-Charged Emulsions”. Angew. Chem. Int. Ed. 2004, 43, 3568–3571. [Google Scholar] [CrossRef]
- Djerdjev, AM; Beattie, JK. “Hydroxide and hydrophobic terabutylammonium ions at the hydrophobe-water interface”. Molecules 2025, 30, 875. [Google Scholar] [CrossRef]
- Hwanbo, SAH; Lee, SY; Kim, BA; Moon, CK. “Preparation of surfactant-free nano oil particles in water using ultrasonic system and mechanism of emulsion stability”. Nanomaterials 2022, 12, 1547. [Google Scholar] [CrossRef]
- Horikoshi, S.; Yu, Akao; Ogura, T; Sakai, H; Abe, M; Serpone, N. “On the stability of surfactant-free water-in-oil emulsions and synthesis of hollow SiO2 nanospheres”. Coll. Surf. A 2010, 372, 55–60. [Google Scholar] [CrossRef]
- Sakai, T; Seo, K. “Colloidal stability of emulsifier-free water-in-oil emulsions: effect of oil property”. J. Japan Soc Colloid Mater 2014, 87(11), 1–6. [Google Scholar] [CrossRef]
- Sakai, T; Oishi, T. “Colloidal stabilization of surfactant-free emulsion by control of molecular diffusion among droplets”. J. Taiwan Inst. Chem. Engin. 2018, 92, 123–128. [Google Scholar] [CrossRef]
- Alizadeh, A; Huang, Y; Liu, F; Daiguji, H; Wang, M. “A streaming-potential-based microfluidic measurement of surface charge at immiscible liquid-liquid interface”. Int. J. Mech. Sci. 2023, 247, 108200. [Google Scholar] [CrossRef]
- Xiong, H; Lee, JK; Zare, RN; Min, W. “Strong electric field observed at the interface of aqueous microdroplets”. The Journal of Physical Chemistry Letters 2020, 11, 7423–7428. [Google Scholar] [CrossRef] [PubMed]
- Shi, L; LaCour, RA; Oian, N; Heindel, JP; Lang, X; Zhao, R; Head-Gordon, T; Min, W. “Water structure and electric fields at the interface of oil droplets”. Nature 2025, 640, 87–93. [Google Scholar] [CrossRef] [PubMed]
- LaCour, RA; Heindel, JP; Zhao, R; Head-Gordon, G.T. “The role of interfaces and charge for chemical reactivity in microdroplets”. Journal of American Chemical Society 2025, 147, 6299–6317. [Google Scholar] [CrossRef]
- Ganachaud, F. “An alternative hypothesis of enhanced deep supercooling of water: nucleator inhibition via bicarbonate adsorption”. The Journal of Physical Chemistry Letters 2025, 16, 261–264. [Google Scholar] [CrossRef]
- Yan, X; Delgado, M; Aubry, J; Gribelin, O; Stocco, A; Cruz, FBD; Bernard, J; Ganachaud, F. “Central role of bicarbonate anions in charging water/hydrophobic interfaces”. The Journal of Physical Chemistry Letters 2018, 9, 96–103. [Google Scholar] [CrossRef]
- Yan, X; Stocco, A; Bernard, J; Ganachaud, F. “Freeze/thaw-induced carbon dioxide trapping promotes emulsification of oil in water”. The Journal of Physical Chemistry Letters 2018, 9, 5998–6002. [Google Scholar] [CrossRef]
- Pickering, Spencer Umfreville. Emulsions. Journal of the Chemical Society, Transactions 91, 2001–2021, 1907. [CrossRef]
- Ostwald, W. Studien über die Bildung und Umwandlung fester Körper" [Studies on the formation and transformation of solid bodies] (PDF). Zeitschrift für Physikalische Chemise 22, 289–33 1897.
- Ninham, BW; Nostro, PL. Unexpected Properties of Degassed Solutions. J Phys Chem B 2020, 124, 7872–8. [Google Scholar] [CrossRef]
- Snoeyink, VL; Jenkins, D. Water Chemistry; John Wiley & Sons: NY, 1980. [Google Scholar]
- Kowacz, M; Pollack, GH. “Moving Water Droplets: “The Role of Atmospheric CO2 and Incident Radiant Energy in Charge Separation at the Air−Water Interface”. J. Phys. Chem. B 2019, 123, 11003−11013. [Google Scholar] [CrossRef] [PubMed]
- Lifshitz, IM; Slyozov, VV. The Kinetics of Precipitation from Supersaturated Solid Solutions. Journal of Physics and Chemistry of Solids 1961, 19(1–2), 35–50. [Google Scholar] [CrossRef]
- Wagner, C. Theory of the aging of precipitates by dissolution-reprecipitation (Ostwald ripening). Zeitschrift für Elektrochemie 1961, 65(7), 581–591. [Google Scholar]
- Meegoda, JN; Hewage, SA; Batagoda, JH. “Stability of Nanobubbles”. Environ Eng Sci 2018, 35(11), 1216–1227. [Google Scholar] [CrossRef]
- Dukhin, AS; Xu, R. “A new approach to explain nano-bubbles paradoxical longevity”. Colloids and Surfaces, A 2024, 700, 134805. [Google Scholar] [CrossRef]
- Perrin, L; Desobry-Banon, S; Gillet, G; Desobry, S. “Review of High-Frequency Ultrasounds Emulsification Methods and Oil/Water Interfacial Organization in Absence of any Kind of Stabilizer”. Foods 2022, 11, 2194. [Google Scholar] [CrossRef]
- Gaikward, SG; Pandit, AB. “Ultrasound emulsification: Effect of ultrasonic and physicochemical properties on dispersed phase volume and droplet size”. Ultrasonic sonochemistry 2008, 15, 554–563. [Google Scholar] [CrossRef]
- Dukhin, AS; Goetz, JP. Characterization of Liquids, Nano- and Microparticulates, and Porous Bodies using Ultrasound Ed 3; Elsevier: London, 2017. [Google Scholar]
- ISO 20998-1; Measurement and characterization of particles by acoustic methods. Part 1. Concepts and procedures in ultrasonic attenuation spectroscopy. ISO: Geneva, 2006.
- ISO 20998-2; Measurement and characterization of particles by acoustic methods. Part 2 Guidelines for linear theory. ISO: Geneva, 2012.
- ISO 13099-1; Colloidal systems - Methods for zeta-potential determination - Part 1: Electroacoustic and electrokinetic phenomena. ISO: Geneva, 2012.
- ISO 13099-3; Colloidal systems - Methods for zeta potential determination - Part 3: Acoustic methods. ISO: Geneva, 2014.
- Dukhin, A.S.; Riesel, A. “Overlapping double layers in electrokinetics of concentrated dispersion”. J. of Colloid and interface Science 2022, 609, 664–674. [Google Scholar] [CrossRef] [PubMed]
- Lyklema, J. Fundamentals of Interface and Colloid Science, Volumes 1; Academic Press, 1993. [Google Scholar]
- Hardy, WB. “An application of the principle of dynamical similitude to molecular physics”. P R Soc Lond A-Cont 1915, 92, 82–100. [Google Scholar]
- Frank, HS; Evans, MW. “Free volume and entropy in condensed systems. III. Entropy in binary liquid mixtures; Partial molal entropy in dilute solutions; structure and thermodynamics in aqueous electrolytes”. Journal of Chemical Physics 13(11), 507–532, 1945. [CrossRef]
- Bockris, JO’M; Devanathan, MAV; Müller, K. “On the Structure of Charged Interfaces”. Proc R Soc Lon Ser-A 1963, 274(1356), 55–79. [Google Scholar] [CrossRef]
- Pashley, RM; Israelachvili, JN. “Molecular layering of water in thin films between mica surfaces and its relation to hydration forces”. J Colloid Interf Sci 1984, 101(2), 500–514. [Google Scholar] [CrossRef]
- Craig, VSJ; Ninham, BW; Pashley, RM. “Direct measurement of hydrophobic forces: a study of dissolved gas, approach rate, and neutron irradiation”. Langmuir 1999, 15, 1913–1919. [Google Scholar] [CrossRef]
- Ninham, BW; Pashley, RM; Nostro, PL. Surface forces: Changing concepts and complexity with dissolved gas, bubbles, salt and heat. Curr Opin Colloid In 2017, 27, 25–32. [Google Scholar] [CrossRef]
- Derjaguin, BV; Dukhin, SS; Yaroschuk, AE. “On the Role of the Electrostatic Factor in Stabilization of Dispersions Protected by Adsorption Layers of Polymers”. J Colloid Interface Sci 1987, 115(1), 234–239. [Google Scholar] [CrossRef]
- Mancui, M; Ruckenstein, E. “The Polarization Model for Hydration/Double Layer Interactions: The Role of the Electrolyte Ions”. Adv Colloid Interface Science 2004, 112(1-3), 109–128. [Google Scholar]
- Hunter, RJ. Zeta Potential in Colloid Science; Academic Press; 1986. [Google Scholar]
- Hauner, IM; Deblais, A; Beattie, JK; Kellay, H; Bonn, D. “The dynamic surface tension of water”. J Phys Chem Lett 2017, B, 1599–1603. [Google Scholar] [CrossRef]
- Anim-Danso, E.; Zhang, Y.; Dhinojwala, A. “Surface Charge Affects the Structure of Interfacial Ice”. Journal of Physical Chemistry C 2016, 120, 3741−3748. [Google Scholar] [CrossRef]
- Arias, FJ. “Comment on the paper ‘A new approach to explaining nano-bubbles paradoxical longevity’ Dukhin A and Xu R [Colloids Surf A 700 (2024) 34805”. Colloids and Surfaces A 2025, 727(2), 137962. [Google Scholar] [CrossRef]
- Eastoe, J; Ellis, C. “De-gassed water and surfactant-free emulsion. History, controversy, and possible application”. Adv in Colloid and Interface Sci. 2007, 134-135, 89–95. [Google Scholar] [CrossRef]
- Craig, VSJ; Ninham, BW; Pashley, RM. The effect of electrolytes on bubble coalescence in water. J Phys Chem 1993, 97, 10192–7. [Google Scholar] [CrossRef]
- Onsager, L; Samaras, NNT. The Surface Tension of Debye-Hückel Electrolytes. J Chem Phys 1934, 2, 628. [Google Scholar] [CrossRef]
- Pohl, HA. Dielectrophoresis: The Behavior of Neutral Matter in Nonuniform Electric Fields; Cambridge University Press: Cambridge, 1978. [Google Scholar]
- Thomson, William. “On the equilibrium of vapor at a curved surface of liquid”. Philosophical Magazine 1871, 4(42 (282)), 448–452. [Google Scholar]
- Goebel, A; Lunkenheimer, K. Interfacial tension of the water/n-Alkane interface. Langmuir 1997, 13, 369–372. [Google Scholar] [CrossRef]
- CRC Handbook of Chemistry and Physics, 95th Edition; Haynes, W.M., Ed.; CRC Press LLC: Boca Raton: FL; p. p. 3-294, 2014-2015.
- Klochko, L.; Noel, J.; Sgreva, NR.; Leclerc, S.; Metivier, C.; Lacroix, D.; Isaev, M. Thermophysical properties of n-hexadecane: Combined molecular dynamics and experimental investigation”. International communications in heat and mass transfer 2022, 137, 106234. [Google Scholar] [CrossRef]
- ECHA. Hexadecane (CAS 544-76-3) Registered Substances Dossier. European Chemical Agency, 2015.
- Coates, M. Environ Sci Technol 1985, 19, 628–32. [CrossRef] [PubMed]
- Boron, WF; Boulpaer, EL. Medical Physiology: A Cellular and Molecular Approach; Elsevier, 2005. [Google Scholar]
- Smoluchowski, M.Z. Phys.Chem 1917, 92, 129.
| Emulsion 3 | Emulsion 4 | ||||
| Experiment Date | Measurement Date | Zeta [mV] | Experiment Date | Measurement Date | Zeta [mV] |
| 9/19/2024 14:36 | 9/19/2024 14:38 | -40.8 | 11/24/2024 13:14 | 11/24/2024 13:25 | -34.3 |
| 9/19/2024 14:41 | -40.4 | 11/24/2024 13:32 | -34.5 | ||
| 9/19/2024 14:43 | -40.8 | 11/24/2024 13:39 | -34.8 | ||
| 9/19/2024 15:22 | 9/19/2024 15:26 | -35.5 | 11/24/2024 13:48 | -34.2 | |
| 9/19/2024 15:28 | -34.2 | 11/24/2024 13:55 | -34.3 | ||
| 9/19/2024 15:31 | -35.2 | 11/25/2024 12:29 | 11/25/2024 12:39 | -36.3 | |
| 9/19/2024 15:33 | -34.6 | 11/25/2024 12:47 | -35.7 | ||
| 9/23/2024 9:43 | 9/23/2024 9:46 | -42.2 | 11/25/2024 12:55 | -35.4 | |
| 9/23/2024 9:48 | -40.8 | 11/25/2024 13:03 | -35.6 | ||
| 9/23/2024 9:50 | -40.4 | 11/25/2024 13:12 | -35.4 | ||
| 9/23/2024 9:53 | -40.6 | 11/26/2024 13:41 | 11/26/2024 13:52 | -35.1 | |
| 9/23/2024 9:55 | -43.3 | 11/26/2024 13:59 | -35.2 | ||
| 9/26/2024 10:31 | 9/26/2024 10:44 | -43.6 | 11/26/2024 14:06 | -35.4 | |
| 9/26/2024 10:54 | -38.4 | 11/26/2024 14:13 | -35.1 | ||
| 9/26/2024 11:04 | -43.9 | 11/26/2024 14:22 | -34.9 | ||
| 9/26/2024 11:13 | -43.2 | 11/27/2024 11:41 | 11/27/2024 11:52 | -36.2 | |
| 9/26/2024 11:22 | -38.3 | 11/27/2024 11:59 | -36.4 | ||
| 11/27/2024 12:07 | -35.9 | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




