Submitted:
31 December 2025
Posted:
02 January 2026
You are already at the latest version
Abstract
Keywords:
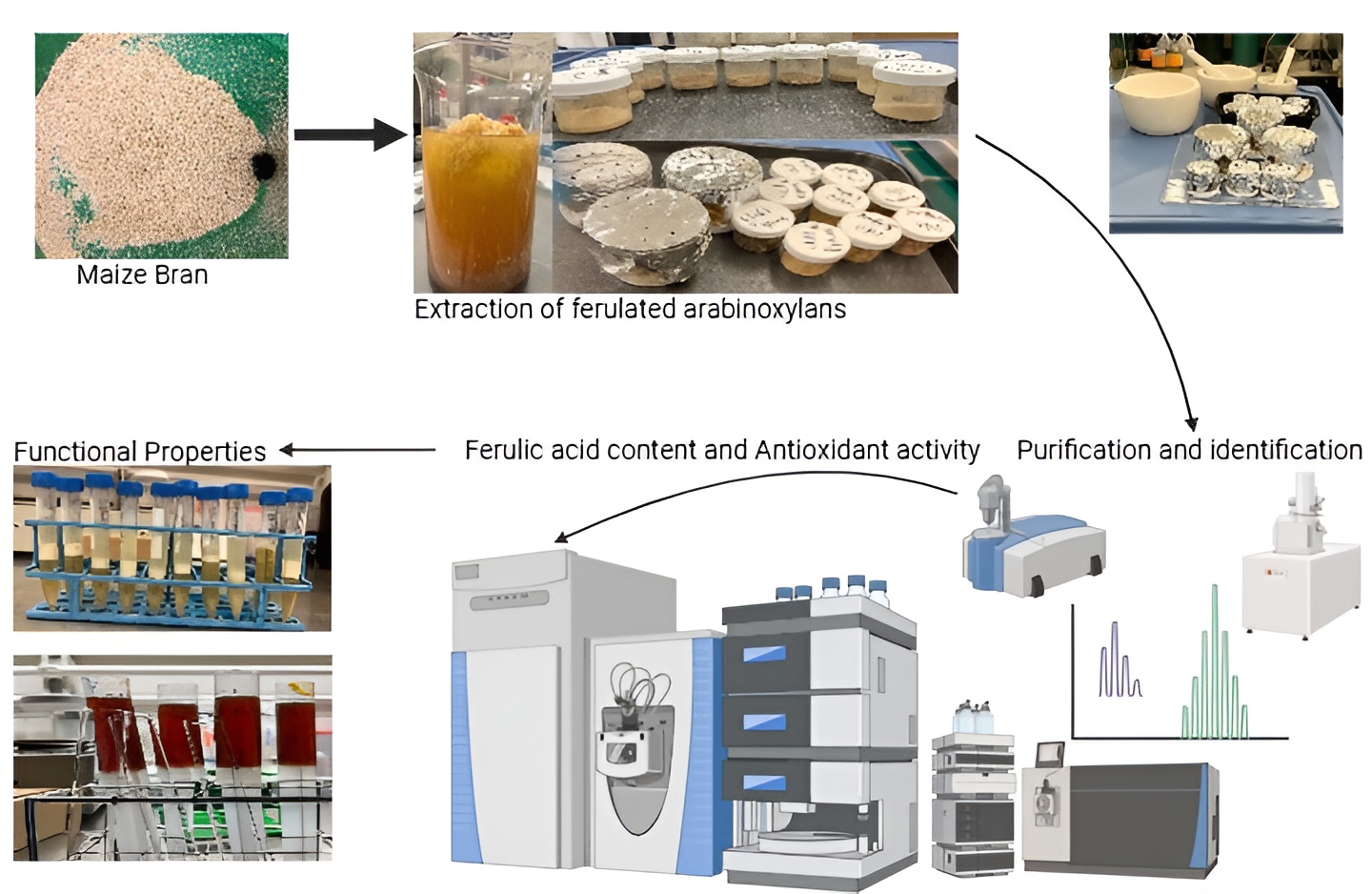
1. Introduction
2. Materials and Methods
2.1. Chemical Analysis
2.2. Extraction of Ferulated Arabinoxylans
1.2. Yield
1.3. Monosaccharaides Analysis
1.4. Arabinose to Xylose Ratio
1.5. Protein Content
1.6. Total Phenolic Content
1.7. Quantification of Ferulic Acid Content
1.8. Antioxidant Activity
1.9. Fourier Transform Infrared Spectroscopy
1.10. Scanning Electron Microscopy
1.11. Functional Properties of Ferulated arabinoxylans
1.12. Statistical Analysis
3. Results and Discussion
3.1. Chemical Composition of Maize Bran
3.2. Extraction Yield of FAX from Maize Bran
3.3. Protein Content
3.4. Monosaccharaides
3.5. Total Phenolic Content
3.6. Ferulic Acid Content
3.7. Antioxidant Activity
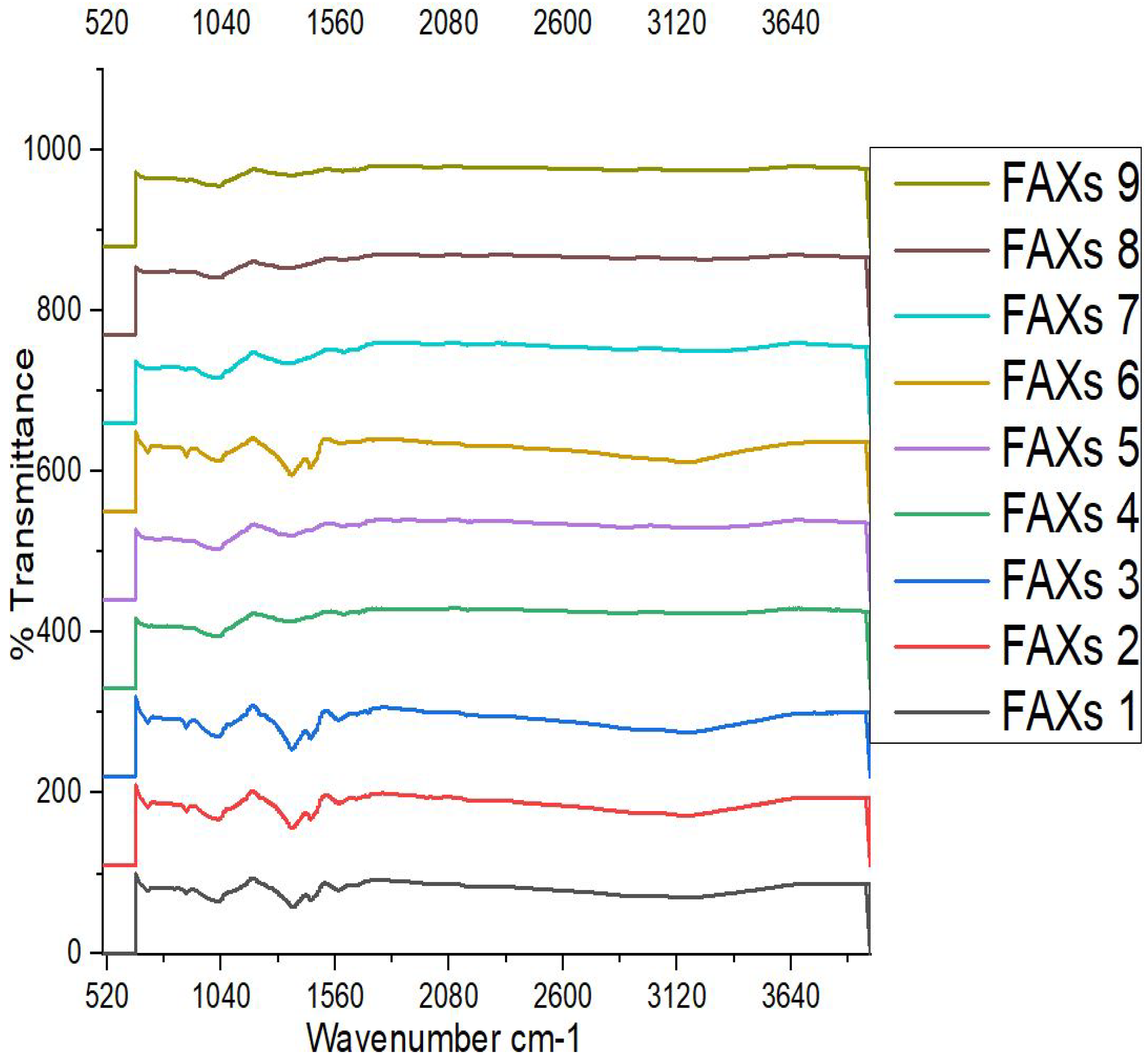
3.8. Fourier Transform Infrared (FT-IR) Spectroscopy
| Wave Number (cm-1) | Functional groups | Current Results | Reference |
|---|---|---|---|
| 520-760 | Polymer backbone | Peaks confirmed | (Sohni, et al. 2024; Kaur et al. 2021) |
| 900-1300 | Polysaccharides and nucleic acids (С-О-Н bendings) | Peaks confirmed | Rehman et al. 2021; Brian-Jaisson |
| 1500-1700 | Proteins (amide I and amide II) | Peaks confirmed | Jiao et al. 2010; Tian et al. 2012) |
| 2100-2200 | alkynes or nitriles bond in polymer structure (Triple bond of C≡C and N=N=N in azides) | Peaks confirmed | Nandiyanto et al. 2023; Shurvell, 2006 |
| 2880, 2900-3100 | СН2 groups in glycerol; C–H stretching bond (aliphatic H) | Peaks confirmed | Peng et al. 2009; Coelho et al. 2009; Bokovets |
| 3100-3300 | Broad absorbance bands for –OH group stretching | Peaks confirmed | Bokovets et al. 2023; Ludwig, B. 2022 |
3.9. Scanning Electron Microscopy
3.10. Functional Properties
4. Conclusion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mujtaba, M.; Fraceto, L. F.; Fazeli, M.; Mukherjee, S.; Savassa, S. M.; de Medeiros, G. A.; Vilaplana, F. Lignocellulosic biomass from agricultural waste to the circular economy: a review with focus on biofuels, biocomposites and bioplastics. Journal of Cleaner Production 2023, 402, 136815. [Google Scholar] [CrossRef]
- Skendi, A.; Zinoviadou, K. G.; Papageorgiou, M.; Rocha, J. M. Advances on the valorisation and functionalization of by-products and wastes from cereal-based processing industry. Foods 2020, 9(9), 1243. [Google Scholar] [CrossRef] [PubMed]
- Akin, M.; Jukic, M.; Lukinac, J.; Yilmaz, B.; Özogul, F.; Rocha, J. M. Valorization and Functionalization of Cereal-Based Industry By-Products for Nutraceuticals. Nutraceutics from Agri-Food By-Products 2025, 173–222. [Google Scholar]
- Guo, Y.; Liu, G.; Ning, Y.; Li, X.; Hu, S.; Zhao, J.; Qu, Y. Production of cellulosic ethanol and value-added products from corn fiber. Bioresources and Bioprocessing 2022, 9(1), 81. [Google Scholar] [CrossRef]
- Saeed, F.; Hussain, M.; Arshad, M. S.; Afzaal, M.; Munir, H.; Imran, M.; Anjum, F. M. Functional and nutraceutical properties of maize bran cell wall non-starch polysaccharides. International Journal of Food Properties 2021, 24(1), 233–248. [Google Scholar] [CrossRef]
- Lugo-Arias, J.; Vargas, S. B.; Maturana, A.; González-Álvarez, J.; Lugo-Arias, E.; Rico, H. Nutrient Removal from Aqueous Solutions Using Biosorbents Derived from Rice and Corn Husk Residues: A Systematic Review from the Environmental Management Perspective. Water 2024, 16(11), 1543. [Google Scholar] [CrossRef]
- Liu, S.; Ding, W.; Yang, Q.; Rose, D. J. Alkali treatment of maize bran affects utilization of arabinoxylan and other non-digestible carbohydrates by the human gut microbiota in vitro in a dose-dependent manner. Food Hydrocolloids 2024, 151, 109764. [Google Scholar] [CrossRef]
- Mule, T. A.; Sawant, S. S.; Odaneth, A. A. Maize bran as a potential substrate for production of β-glucosidase. Biomass Conversion and Biorefinery 2024, 14(3), 4029–4039. [Google Scholar] [CrossRef]
- Raza, M. A.; Saeed, F.; Afzaal, M.; Imran, A.; Niaz, B.; Hussain, M.; Al Jbawi, E. Comparative study of cross-and uncross-linked arabinoxylans extracted from maize bran with special reference to their structural and antioxidant potential. International Journal of Food Properties 2022, 25(1), 2495–2504. [Google Scholar] [CrossRef]
- Zannini, E.; Bravo Núñez, Á.; Sahin, A. W.; Arendt, E. K. Arabinoxylans as functional food ingredients: A review. Foods 2022, 11(7), 1026. [Google Scholar] [CrossRef]
- Weng, V.; Cardeira, M.; Bento-Silva, A.; Serra, A. T.; Brazinha, C.; Bronze, M. R. Arabinoxylan from corn fiber obtained through alkaline extraction and membrane purification: Relating bioactivities with the phenolic compounds. Molecules 2023, 28(15), 5621. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Guo, C.; Wang, C. Biochemical characterization of an organic solvent-and salt-tolerant xylanase and its application of arabinoxylan-oligosaccharides production from corn fiber gum. International Journal of Biological Macromolecules 2024, 136146. [Google Scholar] [CrossRef] [PubMed]
- Chateigner-Boutin, A. L.; Saulnier, L. Ferulic and coumaric acids in the cereal grain: Occurrence, biosynthesis, biological and technological functions. In Advances in Botanical Research; Academic Press, 2022; Vol. 104, pp. 169–213. [Google Scholar]
- Zhang, Z.; Yang, P.; Zhao, J. Ferulic acid mediates prebiotic responses of cereal-derived arabinoxylans on host health. Animal Nutrition 2022, 9, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Valério, R.; Crespo, J. G.; Galinha, C. F.; Brazinha, C. Effect of ultrafiltration operating conditions for separation of ferulic acid from arabinoxylans in corn fibre alkaline extract. Sustainability 2021, 13(9), 4682. [Google Scholar] [CrossRef]
- Marquez-Escalante, J. A.; Carvajal-Millan, E.; Martínez-López, A. L.; Martínez-Robinson, K. G.; Campa-Mada, A. C.; Rascon-Chu, A. Fine structural features and antioxidant capacity of ferulated arabinoxylans extracted from nixtamalized maize bran. Journal of the Science of Food and Agriculture 2023, 103(9), 4584–4591. [Google Scholar] [CrossRef]
- AlYammahi, J.; Rambabu, K.; Thanigaivelan, A.; Bharath, G.; Hasan, S. W.; Show, P. L.; Banat, F. Advances of non-conventional green technologies for phyto-saccharides extraction: current status and future perspectives. Phytochemistry Reviews 2023, 22(4), 1067–1088. [Google Scholar] [CrossRef]
- Jiang, Y.; Bai, X.; Lang, S.; Zhao, Y.; Liu, C.; Yu, L. Optimization of ultrasonic-microwave assisted alkali extraction of arabinoxylan from the corn bran using response surface methodology. International journal of biological macromolecules 2019, 128, 452–458. [Google Scholar] [CrossRef]
- Munk, L.; Muschiol, J.; Li, K.; Liu, M.; Perzon, A.; Meier, S.; Meyer, A. S. Selective enzymatic release and gel formation by cross-linking of feruloylated glucurono-arabinoxylan from corn bran. ACS sustainable chemistry & engineering 2020, 8(22), 8164–8174. [Google Scholar]
- Wang, Y. L.; Wang, W. K.; Wu, Q. C.; Yang, H. J. The release and catabolism of ferulic acid in plant cell wall by rumen microbes: A review. Animal Nutrition 2022, 9, 335–344. [Google Scholar] [CrossRef]
- Xiang, Z.; Runge, T. Emulsifying properties of succinylated arabinoxylan-protein gum produced from corn ethanol residuals. Food Hydrocolloids 2016, 52, 423–430. [Google Scholar] [CrossRef]
- Robertson, J. A.; de Monredon, F. D.; Dysseler, P.; Guillon, F.; Amado, R.; Thibault, J. F. Hydration properties of dietary fibre and resistant starch: a European collaborative study. LWT-Food Science and Technology 2000, 33(2), 72–79. [Google Scholar] [CrossRef]
- Kaur, A.; Singh, B.; Yadav, M. P.; Bhinder, S.; Singh, N. Isolation of arabinoxylan and cellulose-rich arabinoxylan from wheat bran of different varieties and their functionalities. Food Hydrocolloids 2021, 112, 106287. [Google Scholar] [CrossRef]
- Chemists, A. A. C. C. Approved methods of the AACC; The Association, 2000; p. 595. [Google Scholar]
- Gannasin, S. P.; Ramakrishnan, Y.; Adzahan, N. M.; Muhammad, K. Functional and preliminary characterisation of hydrocolloid from tamarillo (Solanum betaceum Cav.) puree. Molecules 2012, 17(6), 6869–6885. [Google Scholar] [CrossRef] [PubMed]
- AACC. Approved Methods of American Association of Cereal Chemists, 10th Ed. ed; American Association CerealChemists, Inc.; St. Paul, MN, 2000. [Google Scholar]
- Herrera-Balandrano, D. D.; Báez-González, J. G.; Carvajal-Millán, E.; Muy-Rangel, D.; Urías-Orona, V.; Martínez-López, A. L.; Niño-Medina, G. Alkali-extracted feruloylated arabinoxylans from nixtamalized maize bran byproduct: A synonymous with soluble antioxidant dietary fiber. Waste and Biomass Valorization 2020, 11, 403–409. [Google Scholar] [CrossRef]
- Sohni, S.; Begum, S.; Hashim, R.; Khan, S. B.; Mazhar, F.; Syed, F.; Khan, S. A. Physicochemical characterization of microcrystalline cellulose derived from underutilized orange peel waste as a sustainable resource under biorefinery concept. Bioresource Technology Reports 2024, 25, 101731. [Google Scholar] [CrossRef]
- Rehman, Z. U.; Vrouwenvelder, J. S.; Saikaly, P. E. Physicochemical properties of extracellular polymeric substances produced by three bacterial isolates from biofouled reverse osmosis membranes. Frontiers in microbiology 2021, 12, 668761. [Google Scholar] [CrossRef]
- Brian-Jaisson, F.; Molmeret, M.; Fahs, A.; Guentas-Dombrowsky, L.; Culioli, G.; Blache, Y.; Ortalo-Magné, A. Characterization and anti-biofilm activity of extracellular polymeric substances produced by the marine biofilm-forming bacterium Pseudoalteromonas ulvae strain TC14. Biofouling 2016, 32(5), 547–560. [Google Scholar] [CrossRef]
- Jiao, Y.; Cody, G. D.; Harding, A. K.; Wilmes, P.; Schrenk, M.; Wheeler, K. E.; Thelen, M. P. Characterization of extracellular polymeric substances from acidophilic microbial biofilms. Applied and environmental microbiology 2010, 76(9), 2916–2922. [Google Scholar] [CrossRef]
- Tian, K.; Shao, Z.; Chen, X. Investigation on thermally-induced conformation transition of soy protein film with variable-temperature FTIR spectroscopy. Journal of applied polymer science 2012, 124(4), 2838–2845. [Google Scholar] [CrossRef]
- Nandiyanto, A. B. D.; Ragadhita, R.; Fiandini, M. Interpretation of Fourier transform infrared spectra (FTIR): A practical approach in the polymer/plastic thermal decomposition. Indonesian Journal of Science and Technology 2023, 8(1), 113–126. [Google Scholar] [CrossRef]
- Shurvell, H. F. Spectra–structure correlations in the mid-and far-infrared; Handbook of vibrational spectroscopy, 2006. [Google Scholar]
- Brand-Williams, W.; Cuvelier, M.-E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT—Food Science and Technology 1995, 28(1), 25–30. [Google Scholar] [CrossRef]
- Peng, F.; Ren, J. L.; Xu, F.; Bian, J.; Peng, P.; Sun, R. C. Comparative study of hemicelluloses obtained by graded ethanol precipitation from sugarcane bagasse. Journal of agricultural and food chemistry 2009, 57(14), 6305–6317. [Google Scholar] [CrossRef] [PubMed]
- Coelho, R. R.; Hovell, I.; Moreno, E. L.; de Souza, A. L.; Rajagopal, K. Characterization of functional groups of asphaltenes in vacuum residues using molecular modelling and FTIR techniques. Petroleum science and technology 2007, 25(1-2), 41–54. [Google Scholar] [CrossRef]
- Bokovets, S. P.; Pertsevoi, F. V.; Murlykina, N. V.; Smetanska, I. M.; Borankulova, A. S.; Ianchyk, M. V.; Yarmosh, T. A. Investigation of infrared spectra of agar-based gel systems for the production of jelly bars. Journal of Chemistry and Technologies 2023, 31(1), 92–103. [Google Scholar]
- Ludwig, B. Infrared spectroscopy studies of aluminum oxide and metallic aluminum powders, part II: adsorption reactions of organofunctional silanes. Powders 2022, 1(2), 75–87. [Google Scholar] [CrossRef]
- Barberousse, Hélène; et al. Optimization of enzymatic extraction of ferulic acid from wheat bran, using response surface methodology, and characterization of the resulting fractions. Journal of the Science of Food and Agriculture 2009, 89.10, 1634–1641. [Google Scholar] [CrossRef]
- Kulathunga, J.; Simsek, S. Stone milling conditions and starter culture source influence phytic acid content and antioxidant activity in whole-grain sourdough bread. Cereal Chemistry 2024, 101(2), 313–322. [Google Scholar] [CrossRef]
- Dai, J.; Mumper, R. J. Plant phenolics: extraction, analysis and their antioxidant and anticancer properties. Molecules 2010, 15(10), 7313–7352. [Google Scholar] [CrossRef]
- Shi, L.; Zhao, W.; Yang, Z.; Subbiah, V.; Suleria, H. A. R. Extraction and characterization of phenolic compounds and their potential antioxidant activities. Environmental Science and Pollution Research 2022, 29(54), 81112–81129. [Google Scholar] [CrossRef]
- Sombutsuwan, P.; Durand, E.; Aryusuk, K. Effect of acidity/alkalinity of deep eutectic solvents on the extraction profiles of phenolics and biomolecules in defatted rice bran extract. PeerJ Analytical Chemistry 2024, 6, e29. [Google Scholar] [CrossRef]
- Huang, W.; Tian, F.; Wang, H.; Wu, S.; Jin, W.; Shen, W.; Liu, G. Comparative assessment of extraction, composition, and in vitro antioxidative properties of wheat bran polyphenols. LWT 2023, 180, 114706. [Google Scholar] [CrossRef]
- Tang, Q.; Huang, G. Improving method, properties and application of polysaccharide as emulsifier. Food chemistry 2022, 376, 131937. [Google Scholar] [CrossRef] [PubMed]
- Bai, L.; Huan, S.; Li, Z.; McClements, D. J. Comparison of emulsifying properties of food-grade polysaccharides in oil-in-water emulsions: Gum arabic, beet pectin, and corn fiber gum. Food Hydrocolloids 2017, 66, 144–153. [Google Scholar] [CrossRef]
- Schendel, R. R.; Meyer, M. R.; Bunzel, M. Quantitative profiling of feruloylated arabinoxylan side-chains from graminaceous cell walls. Frontiers in Plant Science 2016, 6, 1249. [Google Scholar] [CrossRef]
- Bauer, J. L.; Harbaum-Piayda, B.; Schwarz, K. Phenolic compounds from hydrolyzed and extracted fiber-rich by-products. LWT 2012, 47(2), 246–254. [Google Scholar] [CrossRef]
- Martínez-López, A. L.; Carvajal-Millan, E.; Lizardi-Mendoza, J.; Rascón-Chu, A.; López-Franco, Y. L.; Salas-Muñoz, E.; Ramírez-Wong, B. Ferulated arabinoxylans as by-product from maize wet-milling process: Characterization and gelling capability. Maize: Cultivation, uses and health benefits 2012, 65–74. [Google Scholar]
- Chanliaud, E.; Saulnier, L.; Thibault, J. F. Alkaline extraction and characterisation of heteroxylans from maize bran. Journal of Cereal Science 1995, 21(2), 195–203. [Google Scholar] [CrossRef]
- Mendez-Encinas, M. A.; Carvajal-Millan, E.; Rascon-Chu, A.; Astiazaran-Garcia, H. F.; Valencia-Rivera, D. E. Ferulated arabinoxylans and their gels: Functional properties and potential application as antioxidant and anticancer agent. Oxidative Medicine and Cellular Longevity 2018, 2018(1), 2314759. [Google Scholar] [CrossRef]
- Méndez, D. A.; Fabra, M. J.; Odriozola-Serrano, I.; Martín-Belloso, O.; Salvia-Trujillo, L.; López-Rubio, A.; Martínez-Abad, A. Influence of the extraction conditions on the carbohydrate and phenolic composition of functional pectin from persimmon waste streams. Food Hydrocolloids 2022, 123, 107066. [Google Scholar] [CrossRef]
- Ji, Y.; Yang, X.; Ji, Z.; Zhu, L.; Ma, N.; Chen, D.; Cao, Y. DFT-calculated IR spectrum amide I, II, and III band contributions of N-methylacetamide fine components. ACS omega 2020, 5(15), 8572–8578. [Google Scholar] [CrossRef]
- Hussain, M.; Saeed, F.; Niaz, B.; Imran, A.; Tufail, T. Biochemical and structural characterization of ferulated arabinoxylans extracted from nixtamalized and non-nixtamalized maize bran. Foods 2022, 11(21), 3374. [Google Scholar] [CrossRef]
- Pettolino, F. A.; Walsh, C.; Fincher, G. B.; Bacic, A. Determining the polysaccharide composition of plant cell walls. Nature protocols 2012, 7(9), 1590–1607. [Google Scholar] [CrossRef]
- Mendez-Encinas, M. A.; Carvajal-Millan, E.; Rascon-Chu, A.; Astiazaran-Garcia, H. F.; Valencia-Rivera, D. E. Ferulated arabinoxylans and their gels: Functional properties and potential application as antioxidant and anticancer agent. Oxidative Medicine and Cellular Longevity 2018, 2018(1), 2314759. [Google Scholar] [CrossRef]
- Rocchetti, G.; Gregorio, R. P.; Lorenzo, J. M.; Barba, F. J.; Oliveira, P. G.; Prieto, M. A.; Lucini, L. Functional implications of bound phenolic compounds and phenolics–food interaction: A review. Comprehensive Reviews in Food Science and Food Safety 2022, 21(2), 811–842. [Google Scholar] [CrossRef]
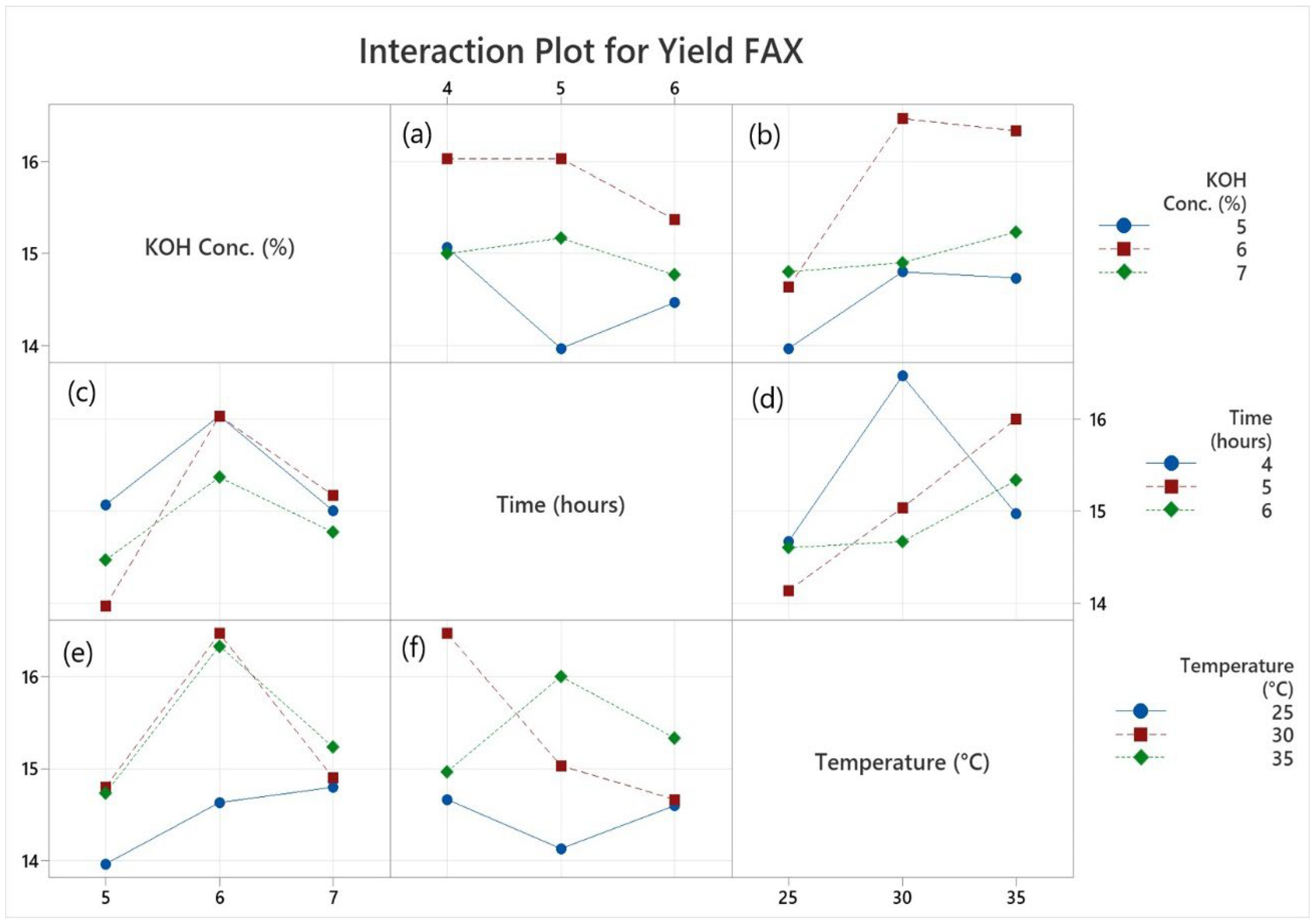

| Samples Run | Factors | Purified Yield of FAX (%) |
Coding of Selected Samples | ||
|---|---|---|---|---|---|
| KOH Conc. (%) | Time (hours) | Temperature (°C) | |||
| 1 | 5 | 4 | 25 | 14.50±0.04g | FAX1 |
| 2 | 5 | 4 | 30 | 14.50±0.04g | - |
| 3 | 5 | 4 | 35 | 14.55±0.05g | - |
| 4 | 5 | 5 | 25 | 18.90±0.05a | FAX2 |
| 5 | 5 | 5 | 30 | 18.90±0.04a | - |
| 6 | 5 | 5 | 35 | 18.90±0.04a | - |
| 7 | 5 | 6 | 25 | 16.20±0.04c | FAX3 |
| 8 | 5 | 6 | 30 | 16.20±0.04c | - |
| 9 | 5 | 6 | 35 | 16.20±0.04c | - |
| 10 | 6 | 4 | 25 | 15.20±0.04e | FAX4 |
| 11 | 6 | 4 | 30 | 15.20±0.04e | - |
| 12 | 6 | 4 | 35 | 15.20±0.04e | - |
| 13 | 6 | 5 | 25 | 16.10±0.02d | FAX5 |
| 14 | 6 | 5 | 30 | 16.10±0.03d | - |
| 15 | 6 | 5 | 35 | 16.10±0.04d | - |
| 16 | 6 | 6 | 25 | 14.70±0.02f | FAX6 |
| 17 | 6 | 6 | 30 | 14.70±0.04f | - |
| 18 | 6 | 6 | 35 | 14.70±0.04f | - |
| 19 | 7 | 4 | 25 | 15.80±0.05e | FAX7 |
| 20 | 7 | 4 | 30 | 15.80±0.04e | - |
| 21 | 7 | 4 | 35 | 15.80±0.04e | - |
| 22 | 7 | 5 | 25 | 16.30±0.04b | FAX8 |
| 23 | 7 | 5 | 30 | 16.30±0.04b | - |
| 24 | 7 | 5 | 35 | 16.30±0.04b | - |
| 25 | 7 | 6 | 25 | 16.10±0.04d | FAX9 |
| 26 | 7 | 6 | 30 | 16.10±0.04d | - |
| 27 | 7 | 6 | 35 | 16.10±0.04d | - |
| Chemical components | FAX1 | FAX2 | FAX3 | FAX4 | FAX5 | FAX6 | FAX7 | FAX8 | FAX9 |
|---|---|---|---|---|---|---|---|---|---|
| Arabinose (%) | 16.0±1.29e | 12.7±1.74f | 17.2±4.12e | 30.4±0.86a | 25.5±6.10c | 15.7±1.43e | 27.4±4.54b | 23.9±2.20d | 26.2±1.33b |
| Xylose (%) | 22.4±2.82e | 18.3±2.63f | 23.3±6.52e | 44.7±2.49a | 36.0±9.85c | 21.8±1.92e | 38.1±6.28b | 32.1±2.82d | 33.5±1.26d |
| Galactose (%) | 3.1±0.09e | 2.7±0.40f | 3.9±0.66d | 7.4±0.39a | 5.8±1.52c | 3.5±0.20d | 6.6±1.26b | 5.6±0.53c | 6.1±0.30c |
| Glucose (%) | 6.1±0.37c | 4.8±0.54e | 7.3±0.85b | 9.2±0.39a | 7.3±1.61b | 4.6±0.36e | 6.5±1.17c | 5.9±0.59d | 6.0±0.23c |
| Ara/Xyl Ratio | 0.71±0.02d | 0.69±0.01e | 0.73±0.03b | 0.68±0.01f | 0.68±0.03f | 0.72±0.03c | 0.71±0.02d | 0.74±0.03a | 0.73±0.02b |
| Protein (%) | 3.58±0.05a | 2.86±0.06c | 3.37±0.05b | 0.96±0.04g | 1.23±0.09f | 1.51±0.02e | 2.09±0.04d | 2.01±0.08d | 1.8±0.06e |
| Total Phenolic content | FAX1 | FAX2 | FAX3 | FAX4 | FAX5 | FAX6 | FAX7 | FAX8 | FAX9 |
|---|---|---|---|---|---|---|---|---|---|
| Free form of TPC (ug/g FAE) | 75.7±0.83bE | 39.4±0.08dD | 114.4±1.76aD | 23.5±0.44eE | 17.7±1.10fE | 48.1±0.08cE | 9.0±0.13gE | 18.3±0.13fE | 9.8±0.08gE |
| Alkaline extracted TPC (ug/g FAE) | 909.7±1.32bA | 592.8±0.44dA | 978.8±2.7aA | 155.0±5.3iA | 408.8±4.4gA | 634.4±3.5cA | 566.9±5.3eA | 536.3±0.9fA | 303.4±2.2hB |
| Acid extracted TPC (ug/g FAE) | 291.1±1.0cB | 264.3±0.7eB | 347.6±0.22aB | 105.4±0.22iB | 184.1±0.57hC | 281.0±0.75dB | 225.3±0.97fC | 188.2±0.13gC | 308.8±0.04bA |
| Fraction I, bound TPC (ug/g FAE) | 107.2±0.48aD | 27.1±0.13iE | 90.2±0.39dE | 100.8±0.26bC | 55.0±0.53hD | 88.5±0.44eD | 79.8±0.13fD | 67.5±0.30gD | 99.4±0.44cC |
| Fraction II, bound TPC (ug/g FAE) | 253.8±10.6bC | 255.9±10.1bC | 196.9±1.76eC | 58.1±1.76gD | 223.8±4.41dB | 186.3±5.30fC | 292.8±3.09aB | 230.9±3.09cB | 48.8±0.00hD |
| Ferulic acid | FAX1 | FAX2 | FAX3 | FAX4 | FAX5 | FAX6 | FAX7 | FAX8 | FAX9 |
|---|---|---|---|---|---|---|---|---|---|
| FFFA (ug/g) | 16.33 | 5.05 | 44.71 | 3.56 | 0.34 | 4.25 | 0.44 | 0.53 | 2.73 |
| Alkaline extracted FA (ug/g) | 252.21 | 165.56 | 267.88 | 63.49 | 140.59 | 262.5 | 204.36 | 181.56 | 115.93 |
| Acid extracted FA (ug/g) | 8.68 | 7.18 | 11.62 | 1.78 | 2.45 | 2.85 | 6.43 | 4.62 | 7.49 |
| Fraction I BFFA (ug/g) | 3.35 | 1.25 | 2.09 | 2.96 | 2.78 | 1.58 | 2.31 | 2.01 | 4.11 |
| Fraction II BFFA (ug/g) | 75.88 | 78.77 | 70.29 | 28.56 | 92.3 | 66.48 | 127.32 | 83.85 | 48.54 |
| Treatments | FAX1 | FAX2 | FAX3 | FAX4 | FAX5 | FAX6 | FAX7 | FAX8 | FAX9 |
|---|---|---|---|---|---|---|---|---|---|
| Ferric Reducing Antioxidant Power (FRAP) | |||||||||
| Free form of bioactive in FAXs extract ug/g TE | 56.5±0.19b | 49.5±0.12c | 122.6±0.24a | 29.4±0.19d | 11.6±0.05i | 19.5±0.01f | 16.5±0.19g | 25.7±0.01e | 13.2±0.01h |
| Alkaline extracted (Bound form extract) ug/g TE | 894.7±1.99b | 616.1±0.34e | 905.0±3.55a | 214.5±0.04i | 384.2±0.87g | 727.5±1.55c | 624.3±0.92d | 594.7±1.60f | 374.7±0.48h |
| Acid extracted (Bound form extract) ug/g TE | 246.7±3.39c | 223.6±2.36d | 260.0±0.75a | 119.8±0.83h | 177.1±0.54g | 245.2±0.47c | 217.8±0.40e | 205.9±2.28f | 256.1±1.41b |
| Fraction I (Bound form extract) ug/g TE | 108.6±0.20a | 25.3±0.06h | 94.3±0.12b | 81.3±0.20d | 46.7±0.01g | 83.6±0.36c | 63.6±0.13e | 48.0±0f | 80.5±0.20e |
| Fraction II (Bound form extract) ug/g TE | 298.6±1.07d | 310.5±1.07c | 286.5±0.43e | 118.7±0.43i | 283.7±0.53f | 242.3±1.16g | 367.9±1.36a | 318.8±0.48b | 188.3±0.24h |
| 2,2′-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) | |||||||||
| Free form of bioactive in FAXs extract ug/g TE | 75.1±0.01c | 74.7±0.03c | 428.7±4.19b | 74.0±0.11b | 29.3±0.31d | 73.7±0.12c | 34.0±0.25e | 40.5±0.30d | 74.2±0.16c |
| Alkaline extracted (Bound form extract) ug/g TE | 596.0±10.6c | 376.7±2.53a | 578.1±2.37a | 413.6±1.97a | 627.5±4.19a | 705.7±2.05a | 549.3±2.05a | 534.2±3.87a | 491.5±2.45a |
| Acid extracted (Bound form extract) ug/g TE | 71.7±0.18d | 68.0±0.29d | 74.5±0.03d | 51.5±0.37d | 60.8±0.27c | 67.6±0.23d | 65.7±0.30c | 61.8±0.34c | 70.3±0.19d |
| Fraction I (Bound form extract) ug/g TE | 39.3±1.03e | 11.0±0.63e | 35.1±0.55e | 37.0±0.45e | 14.8±0.25e | 46.9±0.42e | 45.5±0.33d | 32.0±0.30e | 40.7±0.49e |
| Fraction II (Bound form extract) ug/g TE | 250.2±3.00b | 264.2±3.24b | 266.6±2.45c | 67.9±2.21c | 200.7±3.16b | 146.4±3.32b | 220.3±3.32b | 221.6±4.03b | 136.2±4.11b |
| 2,2-diphenyl-1-picrylhydrazyl (DPPH) | |||||||||
| Free form of bioactive in FAXs extract ug/g TE | 20.6±0.23e | 21.1±0.58e | 64.4±3.74e | 13.4±0.49e | 13.7±0.03e | 4.8±0.35e | 12.3±0.04e | 9.3±0.02e | 2.9±0.21e |
| Alkaline extracted (Bound form extract) ug/g TE | 511.5±1.14a | 292.6±1.66a | 560.1±2.59a | 140.1±2.07b | 251.7±0.72a | 435.9±2.28a | 397.8±2.28a | 373.0±1.14a | 236.1±0.31a |
| Acid extracted (Bound form extract) ug/g TE | 73.6±0.29d | 67.6±0.34d | 80.7±0.18c | 43.3±0.17d | 55.5±0.31d | 68.2±0.41c | 60.1±0.31c | 60.8±0.38c | 71.5±0.36c |
| Fraction I (Bound form extract) ug/g TE | 78.6±0.27c | 74.5±0.32c | 79.6±0.23d | 69.3±0.32c | 59.5±0.39c | 34.8±0.20d | 34.2±0.13d | 32.6±0.21d | 43.1±0.31d |
| Fraction II (Bound form extract) ug/g TE | 200.8±2.59b | 212.4±2.59b | 197.2±2.91b | 180.7±1.66a | 246.0±3.63b | 289.0±3.95b | 241.5±2.80b | 223.1±4.78b | 196.8±2.28b |
| Treatments | Water holding capacity (g/g) | Oil holding capacity (g/g) | Emulsion activity (%) |
Emulsion stability (%) |
|---|---|---|---|---|
| FAX 1 | 9.2±0.05a | 2.96±0.01f | 51±0.08a | 89.7±2.4a |
| FAX 2 | 8.2±0.04b | 3.62±0.02b | 45±0.06b | 82.6±2.3b |
| FAX 3 | 8.9±0.06a | 3.42±0.02c | 49±0.07a | 88.7±2.9a |
| FAX 4 | 9.7±0.03a | 3.28±0.03d | 41±0.04c | 72.5±2.9d |
| FAX 5 | 8.6±0.05b | 3.64±0.05b | 43±0.05c | 77.2±2.2c |
| FAX 6 | 8.5±0.04b | 3.78±0.03a | 45±0.09b | 79.6±2.1c |
| FAX 7 | 8.3±0.06b | 3.44±0.04c | 46±0.04b | 82.5±1.8b |
| FAX 8 | 8.8±0.07a | 3.54±0.05c | 45+0.08b | 80.2±2.4b |
| FAX 9 | 9.4±0.03a | 3.12±0.03e | 42±0.07c | 81.6±3.2b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




