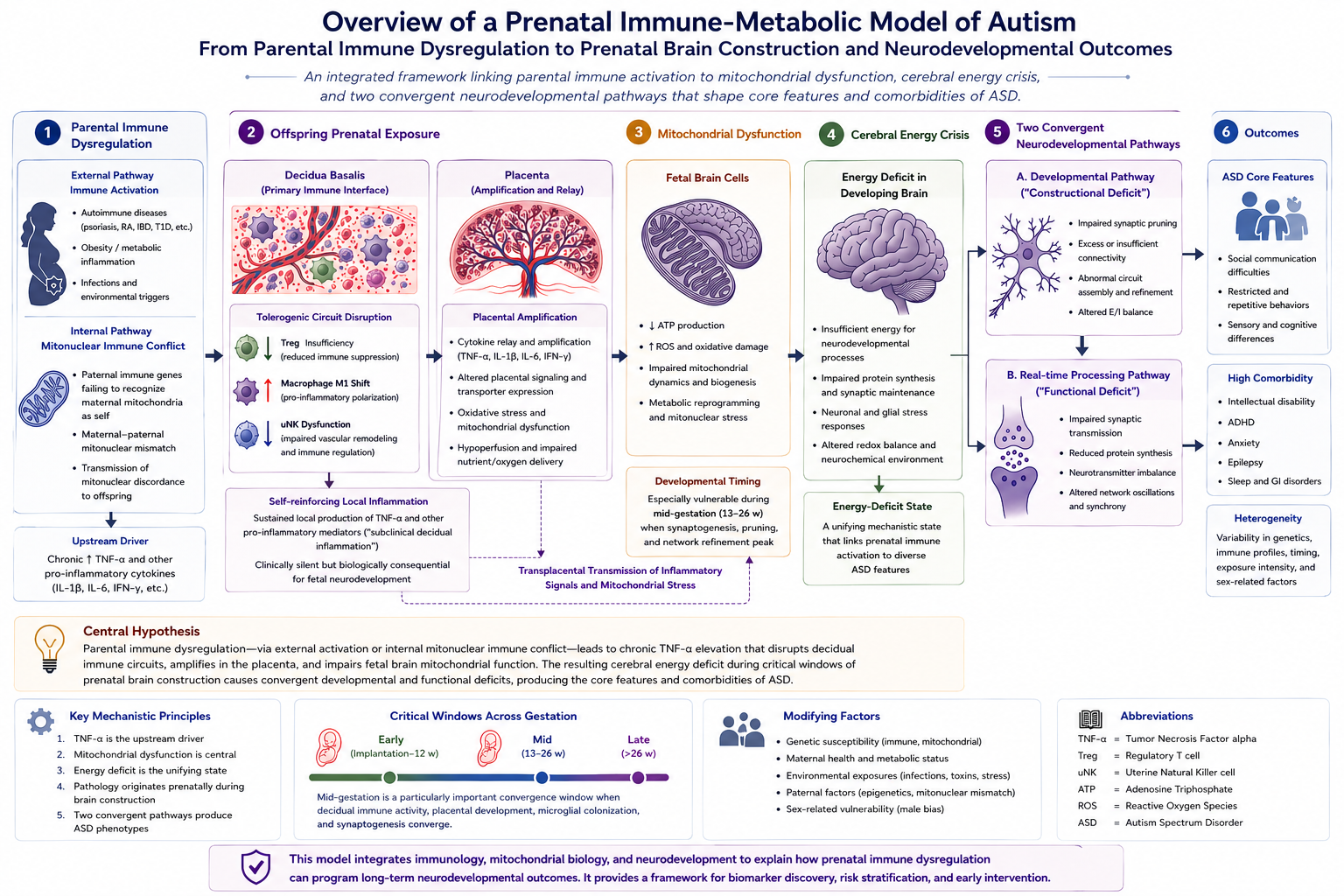
Background: Autism spectrum disorder (ASD) affects approximately 1–2% of children worldwide, yet its etiology remains incompletely understood. Emerging evidence suggests that offspring of parents with autoimmune diseases show elevated autism prevalence. Notably, children of parents with psoriasis (OR 1.59), type 1 diabetes (OR 1.49–2.36), and rheumatoid arthritis (OR 1.51) demonstrate particularly strong associations. Hypothesis: I propose that autism may be conceptualized as an immune-metabolic disorder in which TNF-α-mediated mitochondrial dysfunction contributes to cerebral energy deficiency. This energy deficit may impair three critical processes: (1) synaptic pruning during neurodevelopment, (2) real-time social cognition including gaze processing and emotion recognition, and (3) protein synthesis of critical synaptic scaffolding molecules. The proposed mechanism is TNF-α pathway dysregulation arising from inherited inflammatory susceptibility and/or direct fetal exposure to elevated maternal TNF-α during pregnancy. I further propose that the well-documented “firstborn effect” in autism reflects maternal immune maladaptation during primigravid pregnancies. Additionally, for cases without parental autoimmune history, a speculative secondary mechanism is proposed: mitonuclear immune conflict, in which paternal immune genes may partially recognize maternal mitochondria as non-self, generating endogenous TNF-α. A newly expanded component of the model is the decidua basalis–placenta–fetal brain transmission pathway, in which disruption of the decidual tolerogenic circuit, placental amplification of inflammatory signals, and fetal microglial reprogramming may together contribute to altered neurodevelopment during sensitive gestational windows. The model further proposes that this process may occur even in the setting of a clinically silent, chronic low-to-intermediate pro-inflammatory cytokine state that is insufficient to endanger maternal or fetal survival, yet sufficient to disrupt fetal brain development. Implications: This framework may provide an integrative account of disparate observations about autism pathophysiology and suggests that TNF-α-related pathways merit further investigation for potential risk modification, particularly in pregnancies identified as high-risk through parental autoimmune or inflammatory disease.