Submitted:
29 December 2025
Posted:
30 December 2025
You are already at the latest version
Abstract
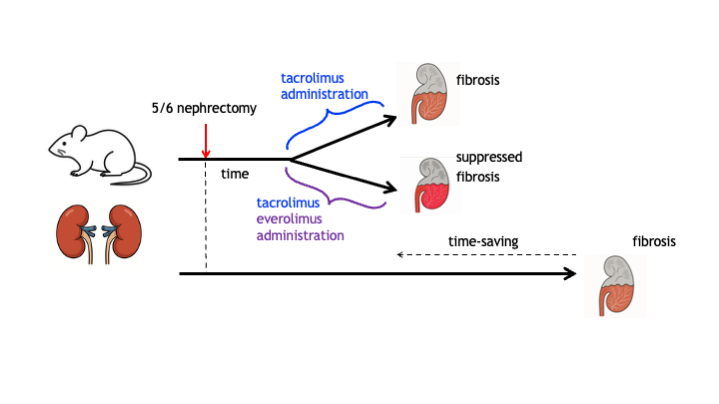
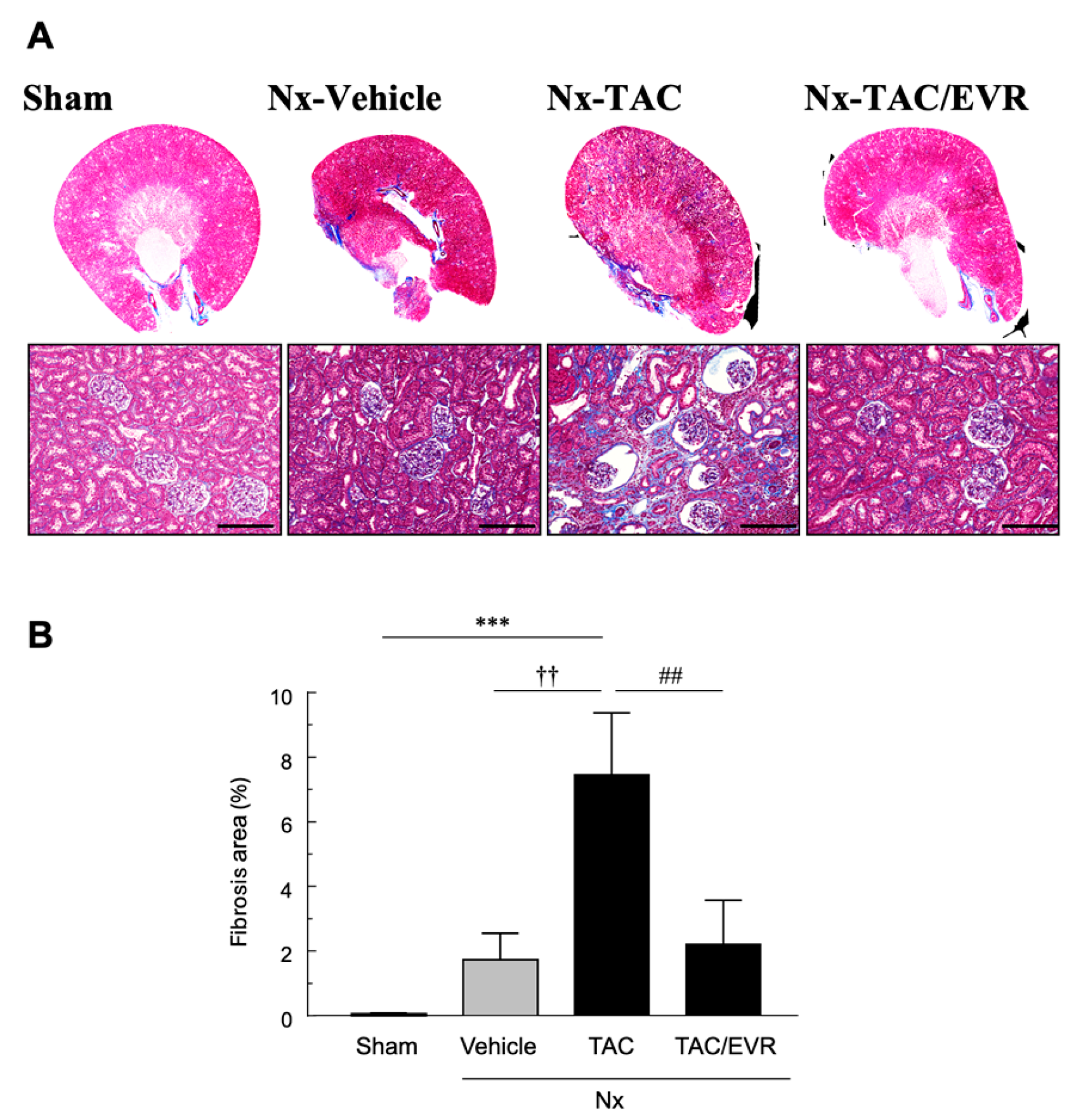
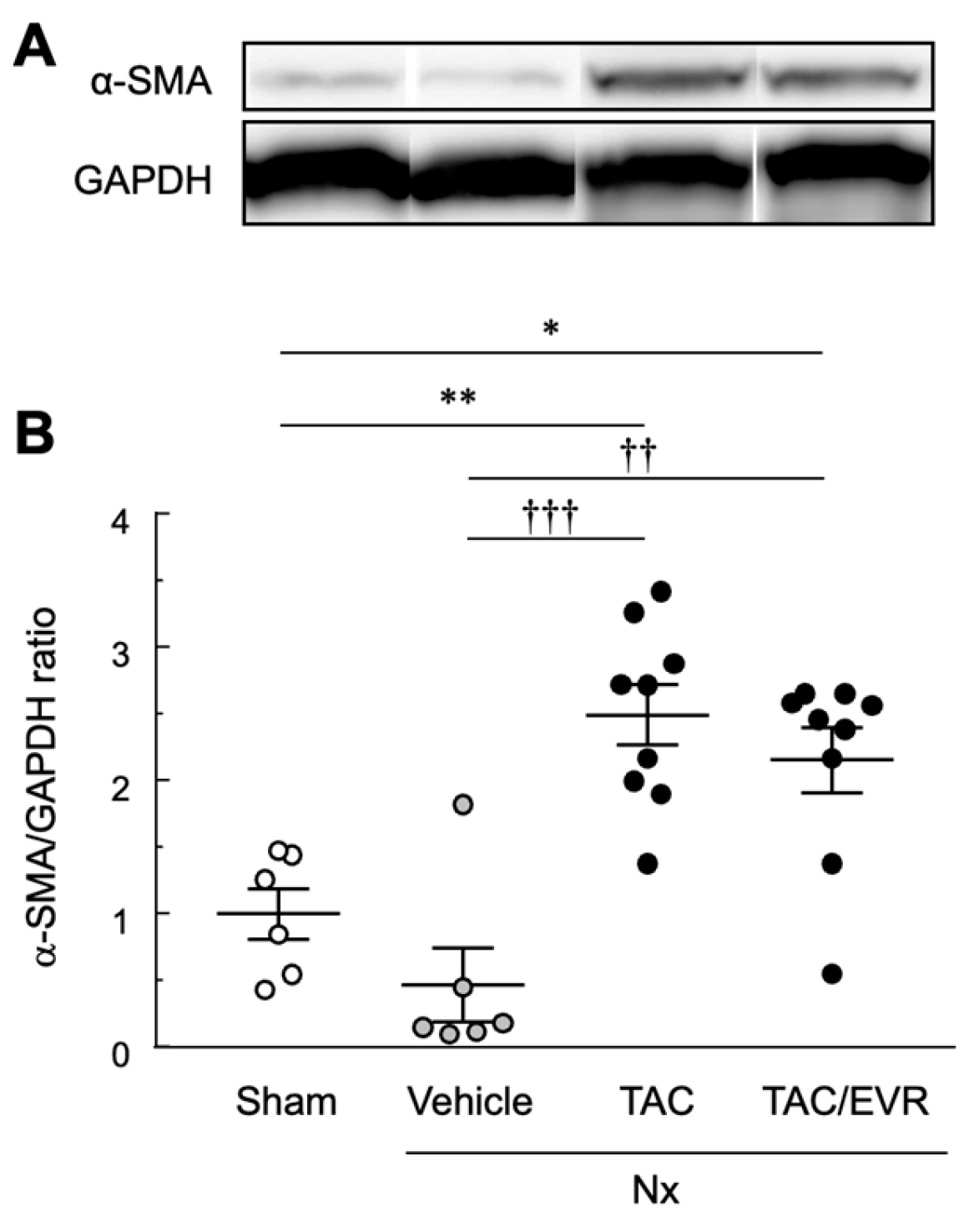
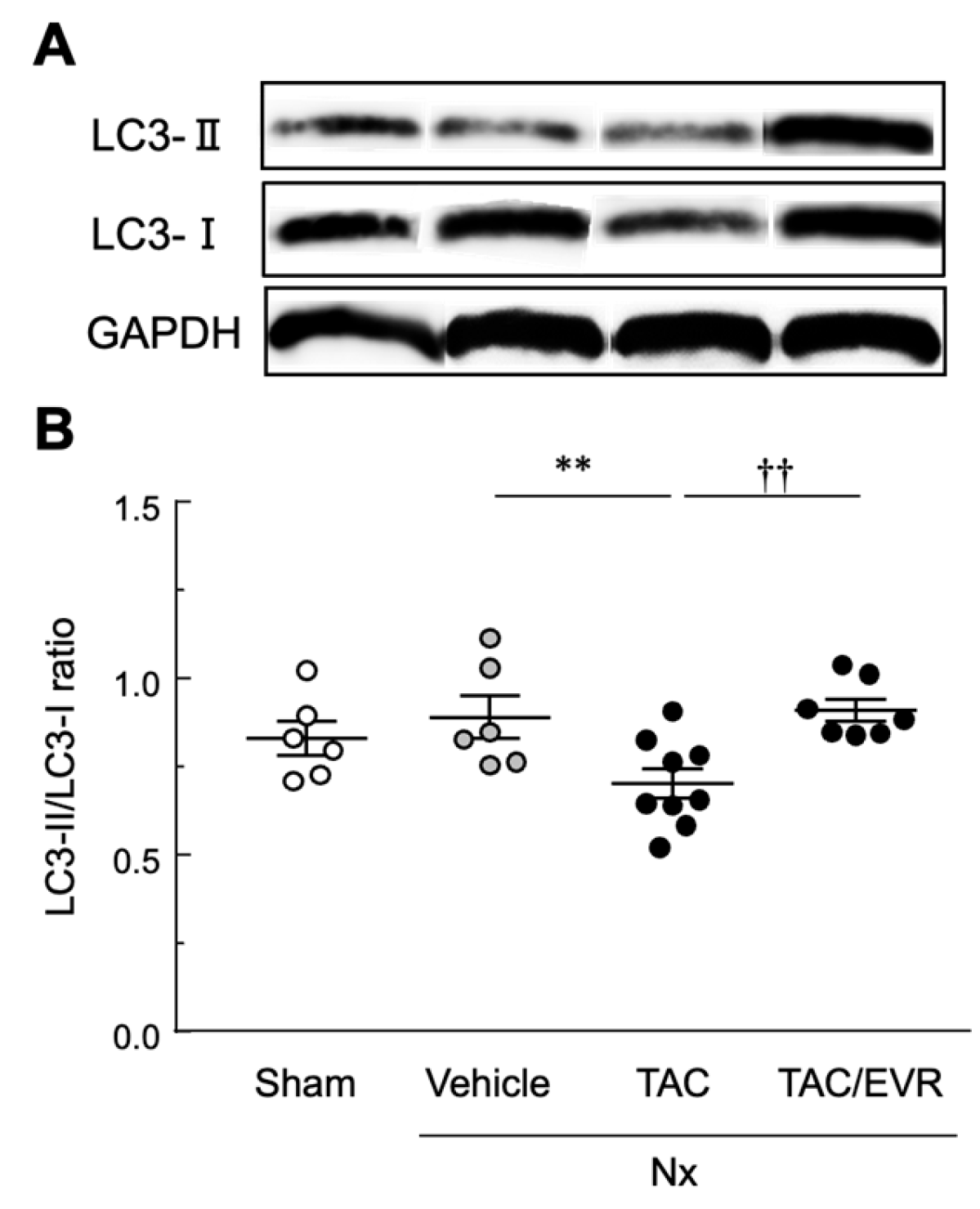
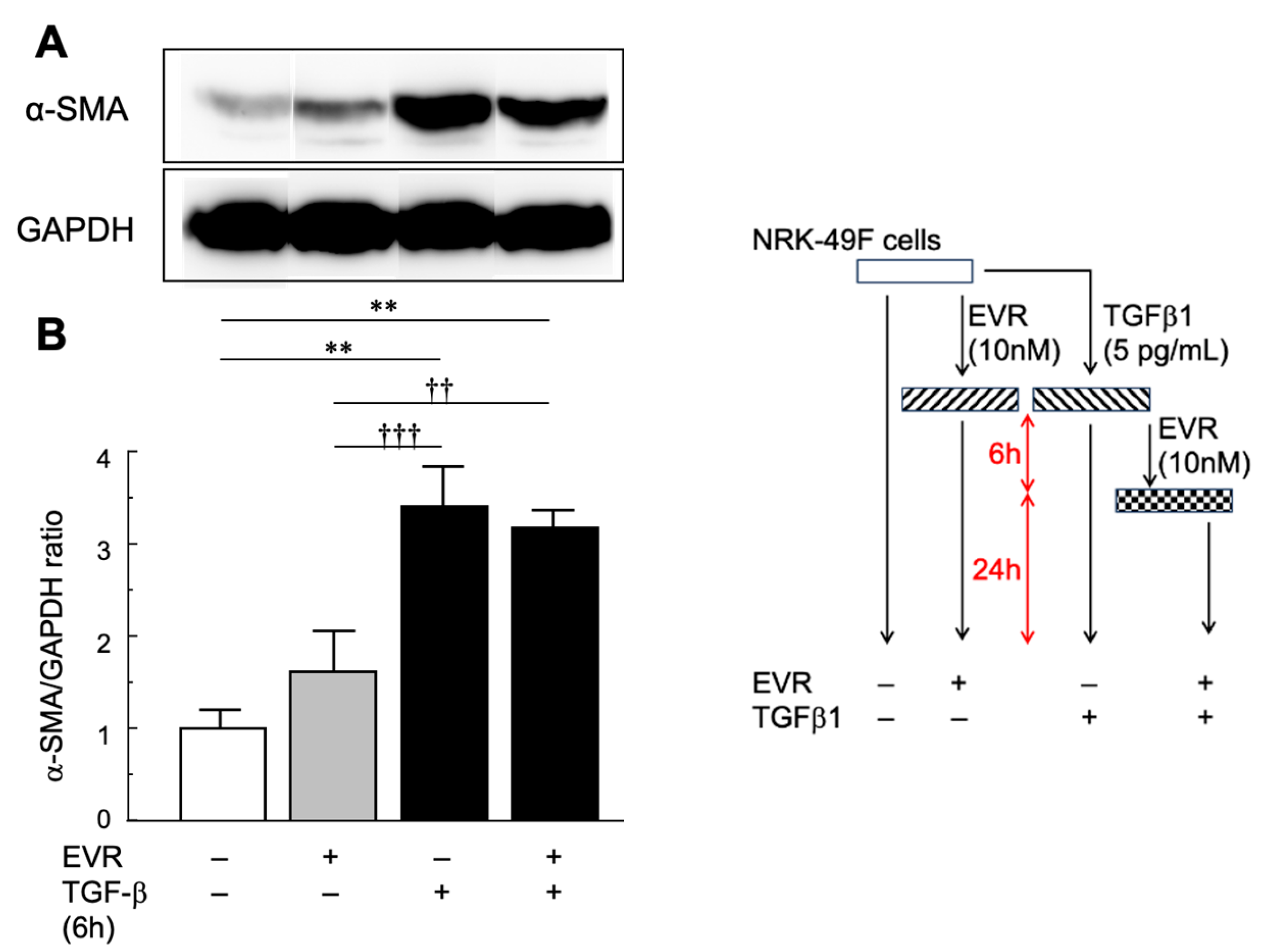
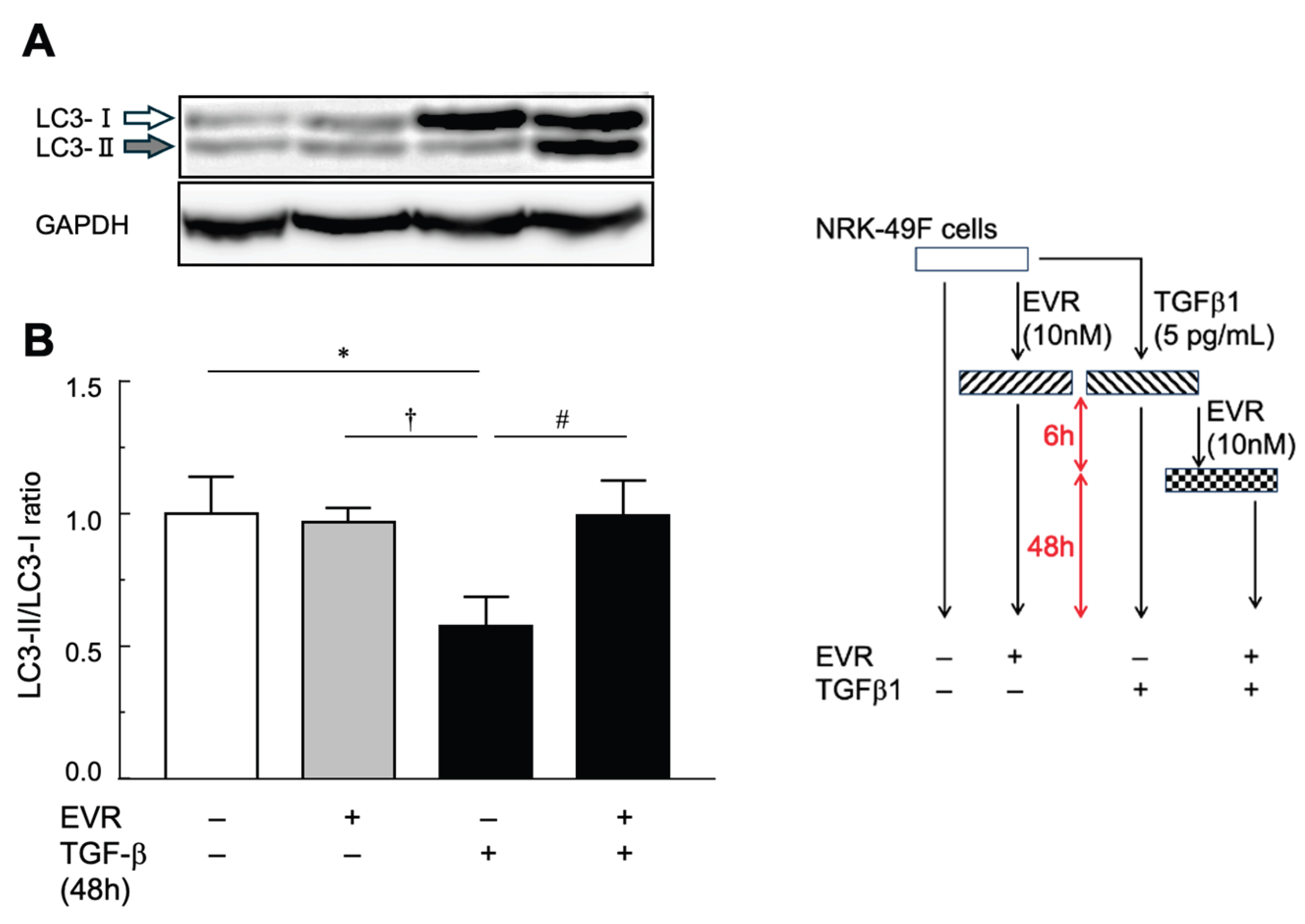
Chronic kidney disease (CKD) is characterized by progressive glomerulosclerosis and interstitial fibrosis, ultimately leading to irreversible renal dysfunction. Tacrolimus, a calcineurin inhibitor widely used after kidney transplantation, is known to accelerate chronic renal injury, whereas everolimus has been introduced as a calcineurin inhibitor–sparing agent. However, their direct effects on renal fibrogenesis remain incompletely defined. In this study, we established a chronic renal failure model using 5/6 nephrectomized rats to evaluate tacrolimus-accelerated renal interstitial fibrosis and the impact of concomitant everolimus treatment. Low-dose tacrolimus (1 mg/kg every other day) administered for two weeks starting four weeks after nephrectomy markedly accelerated interstitial fibrotic progression, accompanied by histological deterioration and increased albuminuria. Co-administration of low-dose everolimus (1 mg/kg every other day) significantly attenuated tacrolimus-accelerated fibrosis and improved functional and structural renal parameters. Histological analyses demonstrated reduced interstitial fibrosis and glomerular alterations in the everolimus-treated group. Although α-smooth muscle actin expression was not significantly suppressed, everolimus restored autophagic activity, as indicated by normalization of the LC3-II/LC3-I ratio in both in vivo and in vitro experiments. These findings indicate that tacrolimus accelerates renal fibrogenesis in a remnant kidney model and that concomitant low-dose everolimus effectively mitigates this progression, providing a time- and cost-efficient experimental platform for evaluating antifibrotic strategies in CKD.
Keywords:
1. Introduction
2. Results
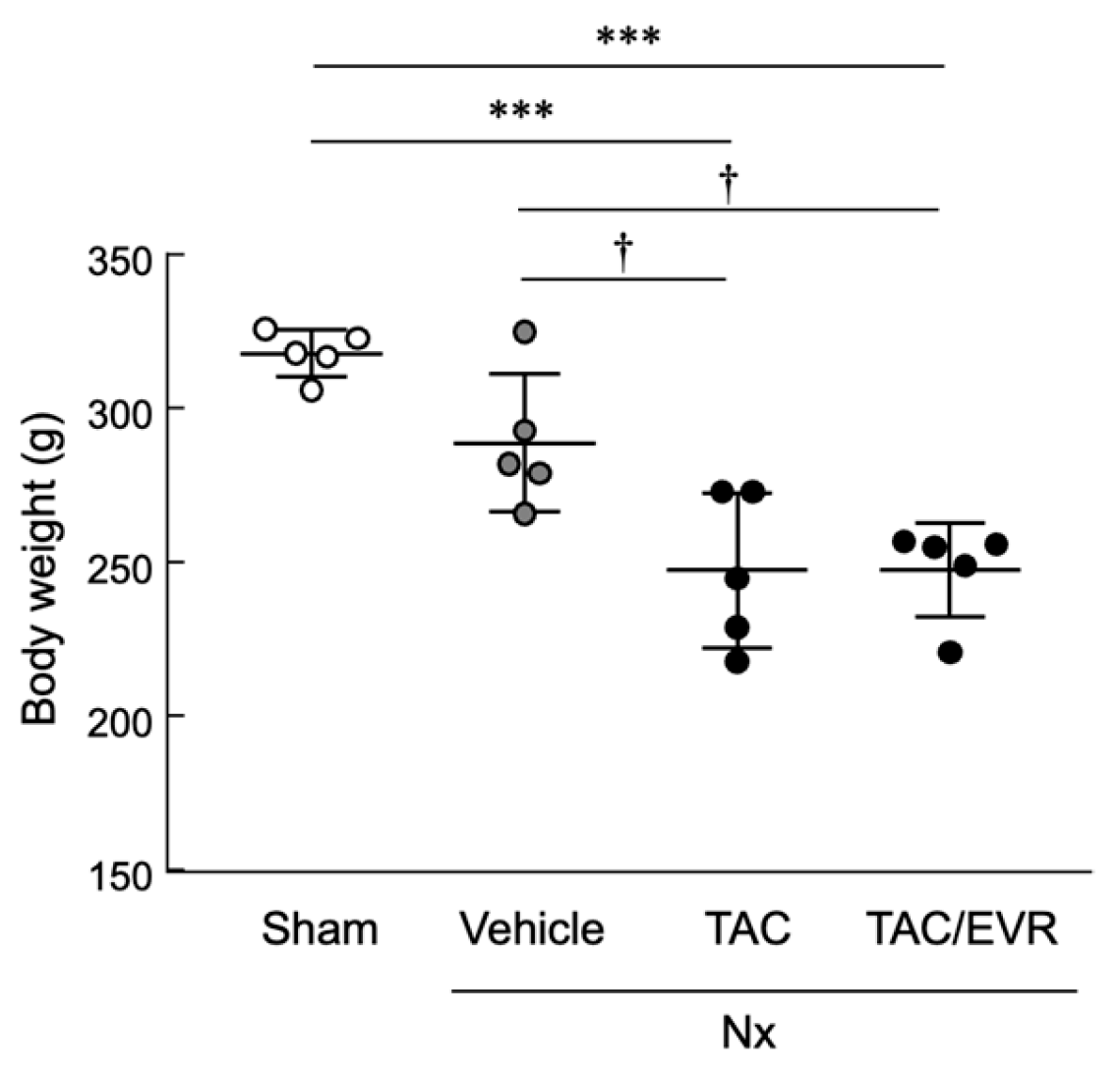
2.1. Evaluation of Renal Functions Following Drug Administration in Nx Rats
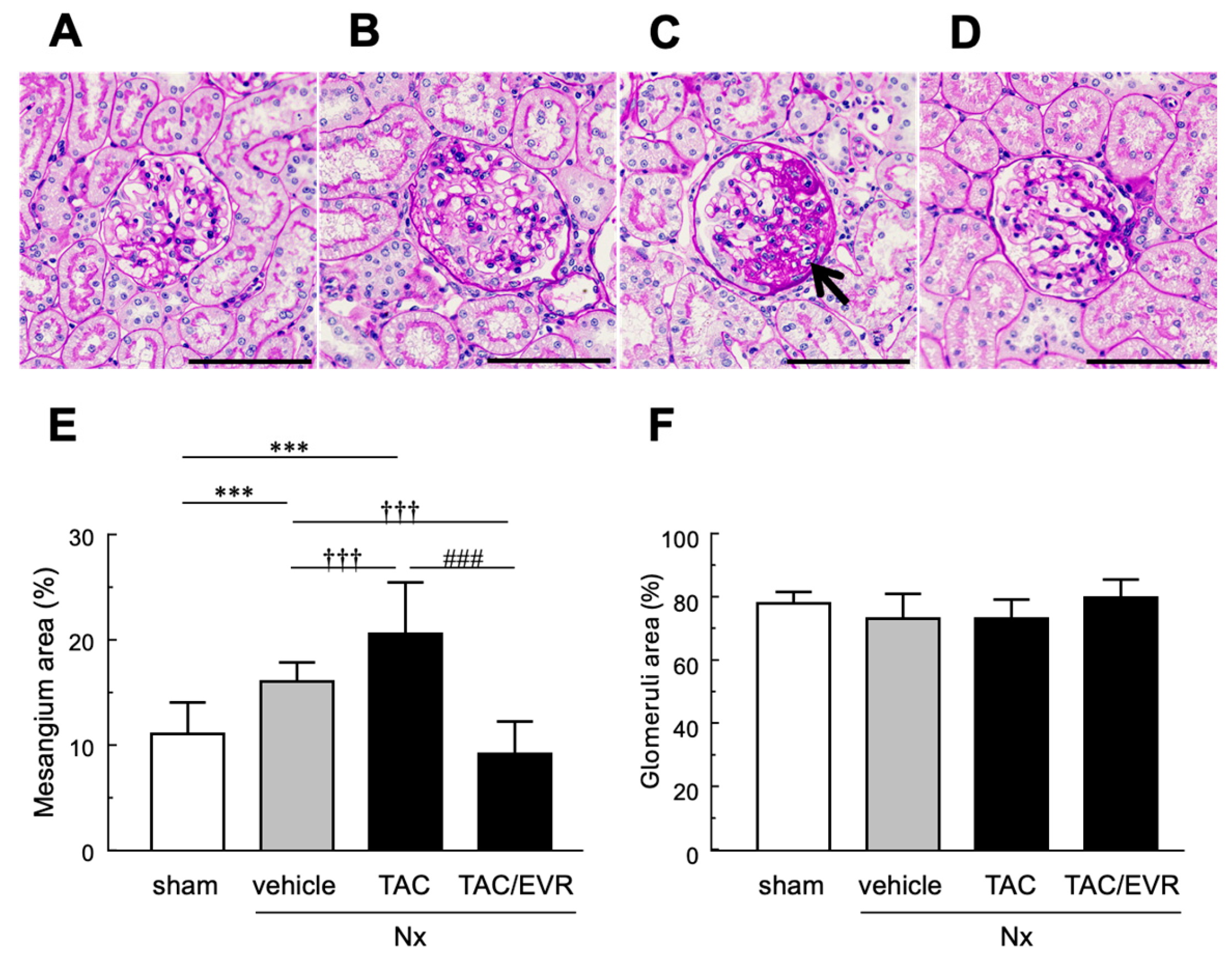
2.2. Histological Evaluation of Renal Tissue Following Drug Treatment
2.3. In Vitro Experiments Using NRK-49F Cells
3. Discussion
4. Materials and Methods
4.1. Test Substances and Reagents
4.2. In Vivo Experiments Using Rats
4.2.1. Drug Administration in the CRF Model
4.2.2. Evaluation of Renal Function and Injury
4.2.3. Morphological Evaluation of Renal Lesions
4.2.4. Western Blot Analysis
4.3. In Vitro Experiments Using Cell Lines
4.3.1. Cell Culture and Drug Treatment
4.3.2. Western Blot Analysis
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CRF | chronic renal failure |
| CKD | chronic kidney disease |
| ESRD | end-stage renal disease |
| GFR | glomerular filtration rate |
| TAC | tacrolimus |
| EVR | everolimus |
| Nx | 5/6 nephrectomy |
| PAS | periodic acid–Schiff |
| MT | Masson’s trichrome |
| Pcr | plasma creatinine |
| Ucr | urinary creatinine |
| Ualb | urinary albumin |
| UACR | urine albumin-creatinine ratio |
| GD | glomerulus diameter |
| BD | Bowman’s diameter |
| ANOVA | 1-way analysis of variance |
| α-SMA | alpha-smooth muscle actin |
| GAPDH | glyceraldehyde-3-phosphate dehydrogenase |
| LC3 | microtubule-associated protein 1 light chain 3 |
| TGF-β | transforming growth factor-beta |
| SEM | standard error of the mean |
| mTORC1 | mammalian target of rapamycin complex 1 |
| ECM | extracellular matrix |
| SDS-PAGE | Sodium Dodecyl Sulfate–Polyacrylamide Gel Electrophoresis |
| PVDF | polyvinylidene difluoride |
| DMEM | Dulbecco’s modified Eagle’s medium |
| PBS | phosphate-buffered saline |
| DTT | dithiothreitol |
| HRP | horseradish peroxidase |
References
- Chen, T. K.; Knicely, D. H.; Grams, M. E. Chronic Kidney Disease Diagnosis and Management: A Review. JAMA 2019, 322(13), 1294–1304. [Google Scholar] [CrossRef]
- Huang, R.; Fu, P.; Ma, L. Kidney fibrosis: from mechanisms to therapeutic medicines. Signal Transduct Target Ther 2023, 8(1), 129. [Google Scholar] [CrossRef] [PubMed]
- Jiang, M.; Chen, H.; Luo, J.; Chen, J.; Gao, L.; Zhu, Q. Characterization of diabetic kidney disease in 235 patients: clinical and pathological insights with or without concurrent non-diabetic kidney disease. BMC Nephrology 2025, 26, 29. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, T.; Isaka, Y. Pathological mechanisms of kidney disease in ageing. Nat Rev Nephrol 2024, 20(9), 603–615. [Google Scholar] [CrossRef]
- Imai, E.; Horio, M.; Watanabe, T.; Iseki, K.; Yamagata, K.; Hara, S.; Ura, N.; Kiyohara, Y.; Moriyama, T.; Ando, Y.; Fujimoto, S.; Konta, T.; Yokoyama, H.; Makino, H.; Hishida, A.; Matsuo, S. Prevalence of chronic kidney disease in the Japanese general population. Clin Exp Nephrol Correction in Clin Exp Nephrol 2009, 13, 631–632. https://doi.org/10.1007/s10157-009-0238-7.. 2009, 13(6), 621–30. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, A.; Hirano, K.; Okuda, T.; Ikenoue, T.; Yokoo, T.; Fukuma, S. Estimating the prevalence of chronic kidney disease in the older population using health screening data in Japan. Clin Exp Nephrol 2025, 29(3), 276–282. [Google Scholar] [CrossRef]
- Japanese Society of, N., CKD Clinical Guide 2024; Tokyo Igakusha: Tokyo, 2024.
- Japanese Society for Clinical Renal, T.; Japanese Society for, T. Renal transplant registry report in Japan: Annual statistics and follow-up results of cases performed in 2023. Japanese Journal of Transplantation 2024, 59(3), 217–236. [Google Scholar]
- Goto, S.; Fujii, H.; Mieno, M.; Yagisawa, T.; Abe, M.; Nitta, K.; Nishi, S. Survival benefit of living donor kidney transplantation in patients on hemodialysis. Clin Exp Nephrol 2024, 28(2), 165–174. [Google Scholar] [CrossRef]
- Hamahata, Y.; Tanabe, K.; Takagi, T. Outcomes of kidney transplants from elderly living donors: A retrospective cohort study in Japan. Tokyo Women’s Medical University Journal 2024, Advance Publication.
- Henkel, L.; Jehn, U.; Tholking, G.; Reuter, S. Tacrolimus-why pharmacokinetics matter in the clinic. Front Transplant 2023, 2, 1160752. [Google Scholar] [CrossRef]
- Kim, H.; Han, A.; Ahn, S.; Min, S. K.; Ha, J.; Min, S. Association of high intra-patient variability in tacrolimus exposure with calcineurin inhibitor nephrotoxicity in kidney transplantation. Sci Rep 2023, 13(1), 16502. [Google Scholar] [CrossRef]
- Okumi, M.; Unagami, K.; Kakuta, Y.; Ochi, A.; Takagi, T.; Ishida, H.; Tanabe, K.; Japan Academic Consortium of Kidney, T. Elderly living donor kidney transplantation allows worthwhile outcomes: The Japan Academic Consortium of Kidney Transplantation study. Int J Urol 2017, 24(12), 833–840. [Google Scholar] [CrossRef] [PubMed]
- Schagen, M. R.; Volarevic, H.; Francke, M. I.; Sassen, S. D. T.; Reinders, M. E. J.; Hesselink, D. A.; de Winter, B. C. M. Individualized dosing algorithms for tacrolimus in kidney transplant recipients: current status and unmet needs. Expert Opin Drug Metab Toxicol Correction in Expert Opin Drug Metab Toxicol 2023, 19(9), i. https://doi.org/10.1080/17425255.2023.2264099. 2023, 19(7), 429–445. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Liu, Z.; Chen, J.; Chai, Y.; Shao, X.; Xie, W.; Zheng, K.; You, J.; Wang, Z.; Feng, M. Impact of intra-patient variability of tacrolimus on allograft function and CD4 + /CD8 + ratio in kidney transplant recipients: a retrospective single-center study. Int J Clin Pharm 2024, 46(4), 918–925. [Google Scholar] [CrossRef] [PubMed]
- Liang, K.; Garvi, E. S.; Slaats, G.; Verhaar, M.; Masereeuw, R.; van Genderen, A. M. #2617 Advanced kidney models to better understand tacrolimus-induced nephrotoxicity. Nephrology Dialysis Transplantation 2025, 40 (Supplement_3), gfaf116.1098. [Google Scholar] [CrossRef]
- Maslauskiene, R.; Vaiciuniene, R.; Radzeviciene, A.; Tretjakovs, P.; Gersone, G.; Stankevicius, E.; Bumblyte, I. A. The Influence of Tacrolimus Exposure and Metabolism on the Outcomes of Kidney Transplants. Biomedicines 2024, 12(5), 1125. [Google Scholar] [CrossRef]
- van Schaik, M.; Bredewold, O. W.; Priester, M.; Michels, W. M.; Rabelink, T. J.; Rotmans, J. I.; Teng, Y. K. O. Long-term renal and cardiovascular risks of tacrolimus in patients with lupus nephritis. Nephrol Dial Transplant 2024, 39(12), 2048–2057. [Google Scholar] [CrossRef]
- Arns, W.; Philippe, A.; Ditt, V.; Hauser, I. A.; Thaiss, F.; Sommerer, C.; Suwelack, B.; Dragun, D.; Hillen, J.; Schiedel, C.; Elsasser, A.; Nashan, B. Everolimus plus reduced calcineurin inhibitor prevents de novo anti-HLA antibodies and humoral rejection in kidney transplant recipients: 12-month results from the ATHENA study. Front Transplant 2023, 2, 1264903. [Google Scholar] [CrossRef]
- Budde, K.; Chadban, S.; Ciechanowska, D.; et al. Two-year outcomes in de novo renal transplant recipients receiving everolimus-facilitated calcineurin inhibitor reduction regimen: results from the randomized TRANSFORM study. Transplantation 2021, 105(1), 101–110. [Google Scholar]
- Mulgaonkar, S.; Pascual, J.; Chadban, S.; et al. In Everolimus with reduced calcineurin inhibitor exposure in de novo kidney transplant recipients: efficacy and safety outcomes from the TRANSFORM study, American Transplant Congress 2018, Seattle, WA, 2018; Seattle, WA, 2018.
- Philippe, A.; Arns, W.; Ditt, V.; Hauser, I. A.; Thaiss, F.; Sommerer, C.; Suwelack, B.; Dragun, D.; Hillen, J.; Schiedel, C.; Elsasser, A.; Nashan, B. Impact of everolimus plus calcineurin inhibitor on formation of non-HLA antibodies and graft outcomes in kidney transplant recipients: 12-month results from the ATHENA substudy. Front Transplant 2023, 2, 1273890. [Google Scholar] [CrossRef]
- Su, L.; Tam, N.; Deng, R.; Chen, P.; Li, H.; Wu, L. Everolimus-based calcineurin-inhibitor sparing regimens for kidney transplant recipients: a systematic review and meta-analysis. Int Urol Nephrol 2014, 46(10), 2035–44. [Google Scholar] [CrossRef]
- Kajiwara, M.; Masuda, S. Role of mTOR Inhibitors in Kidney Disease. International Journal of Molecular Sciences 2016, 17(6), E975. [Google Scholar] [CrossRef]
- Masuda, S.; Lemaitre, F.; Barten, M. J.; Bergan, S.; Shipkova, M.; van Gelder, T.; Vinks, S.; Wieland, E.; Bornemann-Kolatzki, K.; Brunet, M.; de Winter, B.; Dieterlen, M.-T.; Elens, L.; Ito, T.; Johnson-Davis, K.; Kunicki, P. K.; Lawson, R.; Lloberas, N.; Marquet, P.; Millan, O.; Mizuno, T.; Moes, D. J. A. R.; Noceti, O.; Oellerich, M.; Pattanaik, S.; Pawinski, T.; Seger, C.; van Schaik, R.; Venkataramanan, R.; Walson, P.; Woillard, J.-B.; Langman, L. J. Everolimus Personalized Therapy: Second Consensus Report by the International Association of Therapeutic Drug Monitoring and Clinical Toxicology. Therapeutic Drug Monitoring 2025, 47(1), 4–31. [Google Scholar] [PubMed]
- Fu, R.; Tajima, S.; Shigematsu, T.; Zhang, M.; Tsuchimoto, A.; Egashira, N.; Ieiri, I.; Masuda, S. Establishment of an experimental rat model of tacrolimus-induced kidney injury accompanied by interstitial fibrosis. Toxicol Lett 2021, 341, 43–50. [Google Scholar] [PubMed]
- Yang, B.; Jain, S.; Pawluczyk, I. Z.; Imtiaz, S.; Bowley, L.; Ashra, S. Y.; Nicholson, M. L. Inflammation and caspase activation in long-term renal ischemia/reperfusion injury and immunosuppression in rats. Kidney Int 2005, 68(5), 2050–67. [Google Scholar] [PubMed]
- Brenner, B. M.; Lawler, E. V.; Mackenzie, H. S. The hyperfiltration theory: A paradigm shift in nephrology. Kidney Int 1996, 49(6), 1774–1777. [Google Scholar]
- Horiba, N.; Masuda, S.; Takeuchi, A.; Saito, H.; Okuda, M.; Inui, K. Gene expression variance based on random sequencing in rat remnant kidney. Kidney Int 2004, 66(1), 29–45. [Google Scholar]
- Nakamura, N.; Masuda, S.; Takahashi, K.; Saito, H.; Okuda, M.; Inui, K. Decreased expression of glucose and peptide transporters in rat remnant kidney. Drug Metab Pharmacokinet 2004, 19(1), 41–7. [Google Scholar] [CrossRef]
- Nakagawa, S.; Masuda, S.; Nishihara, K.; Inui, K. mTOR inhibitor everolimus ameliorates progressive tubular dysfunction in chronic renal failure rats. Biochem Pharmacol 2010, 79(1), 67–76. [Google Scholar] [CrossRef]
- Nishihara, K.; Masuda, S.; Ji, L.; Katsura, T.; Inui, K. Pharmacokinetic significance of luminal multidrug and toxin extrusion 1 in chronic renal failure rats. Biochem Pharmacol 2007, 73(9), 1482–90. [Google Scholar]
- Nakagawa, S.; Nishihara, K.; Inui, K.; Masuda, S. Involvement of autophagy in the pharmacological effects of the mTOR inhibitor everolimus in acute kidney injury. Eur J Pharmacol 2012, 696(1-3), 143–54. [Google Scholar]
- Ji, L.; Masuda, S.; Saito, H.; Inui, K. Down-regulation of rat organic cation transporter rOCT2 by 5/6 nephrectomy. Kidney Int 2002, 62(2), 514–24. [Google Scholar] [CrossRef]
- Takeuchi, A.; Masuda, S.; Saito, H.; Doi, T.; Inui, K. Role of kidney-specific organic anion transporters in the urinary excretion of methotrexate. Kidney Int 2001, 60(3), 1058–68. [Google Scholar] [CrossRef]
- Bradford, M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 1976, 72, 248–54. [Google Scholar] [CrossRef] [PubMed]
| treatment | Pcr | Ucr | Ualb | UACR |
|---|---|---|---|---|
| (unit) | (mg/dL) | (mg/dL) | (mg/dL) | |
| Sham | 0.57±0.04 | 28.3±6.7 | 30.5±2.0 | 1.5±0.4 |
| Nx | ||||
| Vehicle | 0.82±0.14 | 11.7±0.9** | 80.0±30.1 | 7.4±3.4* |
| TAC | 1.74±0.20**†† | 26.3±5.2†† | 155.1±57.5**† | 6.4±2.3* |
| TAC/EVR | 1.40±0.19**† | 14.9±1.5** | 84.8±18.9 | 5.7±1.2 |
| Indicator | BD | ratio to Sham | GD | ratio to Sham | GD/BD ratio |
|---|---|---|---|---|---|
| (unit) | (µm) | (µm) | |||
| Sham | 110±9 | – | 100±9 | — | 0.91 |
| Nx | |||||
| Vehicle | 106±10 | 0.96 | 95±9 | 0.95 | 0.90 |
| TAC | 128±11**†† | 1.16 | 106±10†† | 1.06 | 0.83 |
| TAC/EVR | 114±11†† | 1.04 | 103±9† | 1.03 | 0.90 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




