Submitted:
29 December 2025
Posted:
30 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
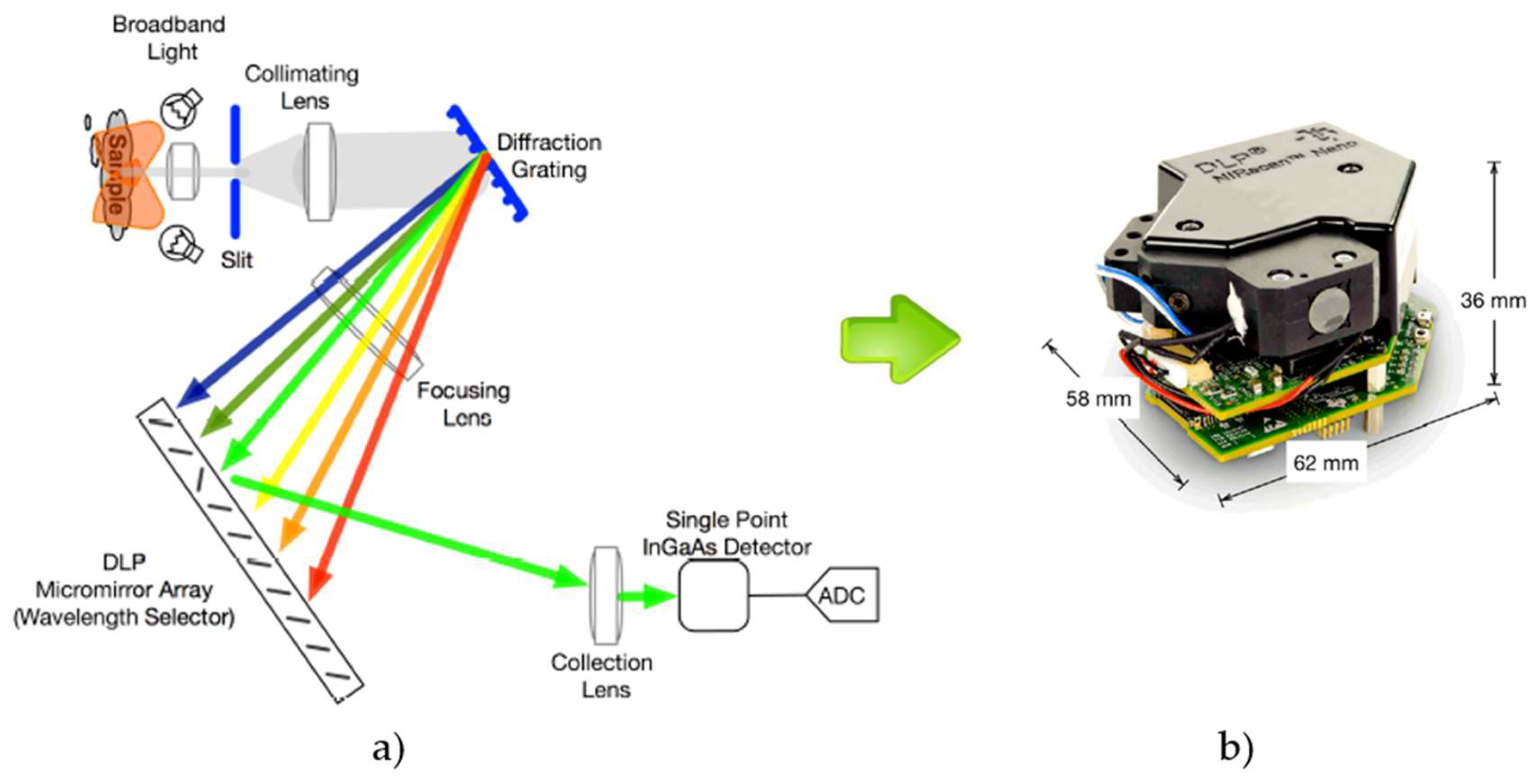
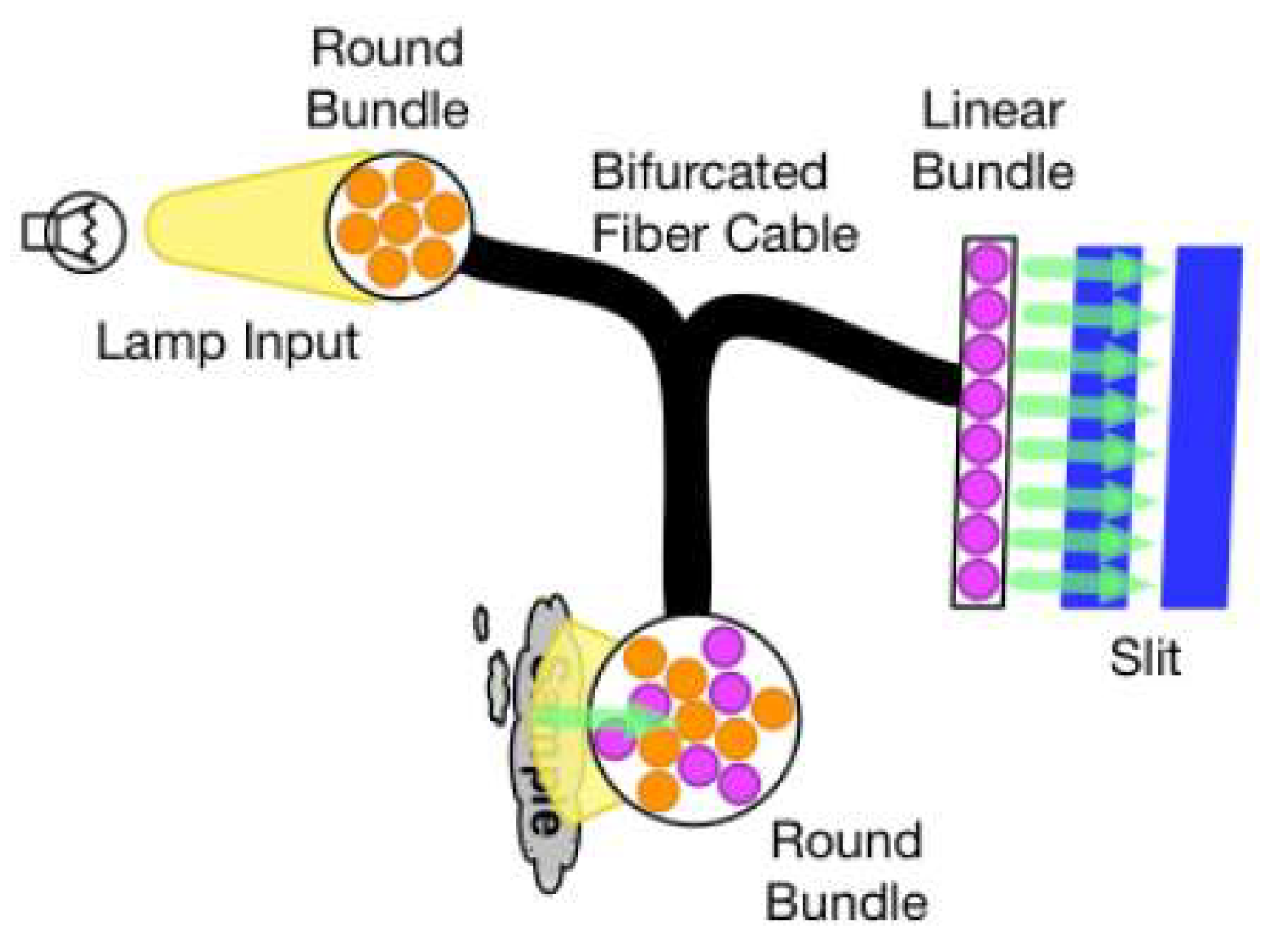
2. Near Infrared Spectroscopy
| Manufacturer | Model | Technology | Spectral Range (nm) |
Spectral Resolution (nm) |
|---|---|---|---|---|
| Texas Instruments | NIRscan Nano EVM |
Grating–MEMS DMD |
900–1700 | 10 |
| Viavi Solutions | MicroNIR Pro 1700 | LVF–Linear | 908–1676 | 12–25 |
| Si-Ware Systems |
NeoSpectra | MEMS–FT | 1250–1700 | 8–16 |
| Ocean Optics | Flame NIR | Grating | 970–1700 | ~10.0 |
| ITPhotonics | Polispec NIR | x | 900-1700 | 3,2 |
| Thermo Fisher Scientific | MicroPhazir | MEMS | 1596-2396 | 11 |
| SouthNest Tecnology | NanoFTIR | MEMS-Michaelson Interferometer | 800-2600 | 2.5-13 |
| Spectral Engines | NIROne Sensor | MEMS Fabry-Pérot Interferometer | 1350-2500 | 16 |
3. Raman Spectroscopy for Health and Food Safety Control
4. Hyperspectral imaging for Health and Food Safety Control
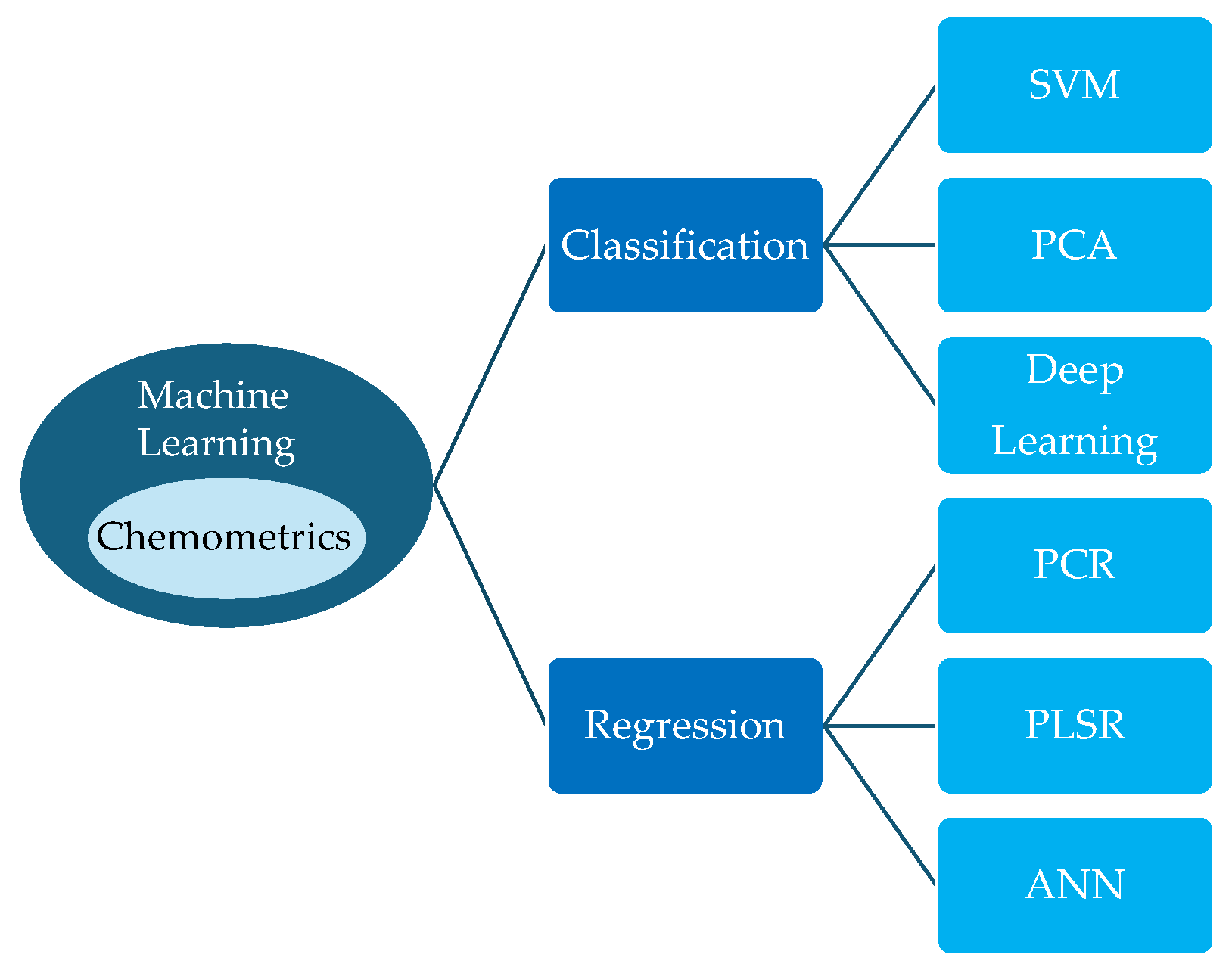
5. Combination of Vibrational Spectroscopy and chemometric analysis
6. Chemometrics Software Tools
7. Vibrational Spectroscopy Applications
8. Conclusion and Outlook
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Beć, K. B.; Grabska, J.; Huck, C. W. Principles and Applications of Miniaturized Near-Infrared (NIR) Spectrometers. Chemistry – A European Journal 2021, 27(5), 1514–1532. [Google Scholar] [CrossRef]
- Yang, Z.; Albrow-Owen, T.; Cai, W.; Hasan, T. Miniaturization of optical spectrometers. Science 2021, 371(6528). [Google Scholar] [CrossRef]
- Leone, M.; Consales, M.; Passeggio, G.; Buontempo, S.; Zaraket, H.; Youssef, A.; Persiano, G.V.; Cutolo, A.; Cusano, A. Fiber optic soil water content sensor for precision farming. Optics & Laser Technology 2022, 149–107816. [Google Scholar] [CrossRef]
- Beć, K. B.; Grabska, J.; Huck, C.K.; Chem. Principles and Applications of Miniaturized Near-Infrared (NIR) Spectrometers. Eur. J. 2021, 27, 1514. [Google Scholar] [CrossRef]
- Texas Instruments. DLP NIRscan Nano EVM User’s Guide; Texas Instruments: Dallas, TX, USA, 2017. [Google Scholar]
- Huang, W.; Luo, S.; Yang, D.; Zhang, S. Applications of smartphone-based near-infrared (NIR) imaging, measurement, and spectroscopy technologies to point-of-care (POC) diagnostics. J. Zhejiang Univ Sci B 2021, 22(3), 171–189. [Google Scholar] [CrossRef]
- Wang, H.-P.; Chen, P.; Dau, J.W.; Liu, D.; Li, J.Y.; Xu, Y.P.; Chu, X.L. Recent advances of chemometric calibration methods in modern spectroscopy: Algorithms, strategy, and related issues. TrAC Trends in Analytical Chemistry 2022, 153, 116648. [Google Scholar] [CrossRef]
- Xia, J.; Zhang, W.; Zhang, W.; Yang, Y.; Hu, G.; Ge, D.; Liu, H.; Cao, H. A cloud computing-based approach using the visible near-infrared spectrum to classify greenhouse tomato plants under water stress. Comput. Electron. Agr. 2021, 181, 105966. [Google Scholar] [CrossRef]
- Rego, C.; Ferrero, F; Valledor, M.; Campo, J.C.; Forcada, S.; Royo, L.; Soldado, A. A portable IoT NIR spectroscopic system to analyze the quality of dairy farm forage. Computers and Electronics in Agriculture 2020, 175, 105578. [Google Scholar] [CrossRef]
- Perezcampos Mayoral, C.; Gutiérrez Gutiérrez, J.; Cano Pérez, J.L.; Vargas Treviño, M.; Gallegos Velasco, I.B.; Hernández Cruz, P.A.; Torres Rosas, R.; Tepech Carrillo, L.; Arnaud Ríos, J.; Apreza, E.L.; et al. Fiber Optic Sensors for Vital Signs Monitoring. A Review of Its Practicality in the Health Field. Biosensors 2021, 11, 58. [Google Scholar] [CrossRef] [PubMed]
- Pasquini, C. Near infrared spectroscopy: A mature analytical technique with new perspectives – A review. Anal. Chim. Acta 2018, 1026, 8–36. [Google Scholar] [CrossRef]
- Sun, Y.; Tang, H.; Zou, X.; Meng, G.; Wu, N. Raman spectroscopy for food quality assurance and safety monitoring: a review. Current Opinion in Food Science 2022, 47, 100910. [Google Scholar] [CrossRef]
- Surappa, S.; Multani, P.; Ugur Parlatan, U.; Sinawang, P.D.; Kaifi, J.; Akinac de, D.; Demirci, U. Integrated “lab-on-a-chip” microfluidic systems for isolation, enrichment, and analysis of cancer biomarkers. Lab Chip 2023, 23, 2942–2958. [Google Scholar] [CrossRef]
- Nie, Ch; Shaw, I.; Chen, C. Application of microfluidic technology based on surface-enhanced Raman scattering in cancer biomarker detection: A review. Journal of Pharmaceutical Analysis 2023, Volume 13(Issue 12), Pages 1429–1451. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Wang, H.; Zhong, W.; et al. The development and application of SERS-based lat-eral flow immunochromatography in the field of food safety. Microchim Acta 2025, 192, 246. [Google Scholar] [CrossRef]
- Bai, J.; Dong, D. Observing and analyzing living plants using handheld and miniaturized Raman spectrometers: A review. Trends in Analytical Chemistry 2025, 193, 118437. [Google Scholar] [CrossRef]
- Pallua, J. D.; Brunner, A.; Zelger, B.; Huck, C.W.; Schirmer, M.; Laimer, J.; Putzer, D.; Thaler, M.; Zelger, B. New perspectives of hyperspectral imaging for clinical research. NIR news 2021, 32(3-4), 5–13. [Google Scholar] [CrossRef]
- Kumar, V.; Shankar, R.; Rambabu, M.; Dua, Y. Deep learning for hyperspectral image classification: A survey. Computer Science Review 2024, Volume 53, 100658. [Google Scholar] [CrossRef]
- Bruno, F.; Mishra, P.; Davies, B.; Sturrock, K.; Fiore, A. Portable and handheld hyperspectral imaging for non-destructive acrylamide prediction in ground potato crisps. Food Control 2025, Volume 178, 111512. [Google Scholar] [CrossRef]
- Neri, I.; Caponi, S.; Bonacci, F.; Clementi, G.; Cottone, F.; Gammaitoni, L.; Figorilli, S.; Ortenzi, L.; Aisa, S.; Pallottino, F.; Mattarelli, M. Real-Time AI-Assisted Push-Broom Hyperspectral System for Precision Agriculture. Sensors 2023, 24(2), 344. [Google Scholar] [CrossRef]
- Thomas, J.; Lapray, P.; Le Moan, S. Trends in Snapshot Spectral Imaging: Systems, Processing, and Quality. Sensors 2024, 25(3), 675. [Google Scholar] [CrossRef]
- Medina–García, M.; Amigo, J.M.; Martínez-Domingo, M.A.; Valero, E.M.; Jiménez–Carvelo, A.M. Strategies for analysing hyperspectral imaging data for food quality and safety issues – A critical review of the last 5 years. Microchemical Journal 214, 113994. [CrossRef]
- Jiang, Z.; Li, J.; Xu, S.; Liu, Z.; Ma, D.; Wang, Q.; Yuan, Y. Cross-domain hyperspectral image classification. Pattern Recognition 2025, Volume 168(2025), 111836. [Google Scholar] [CrossRef]
- Li, W.; Wu, Y.; Du, L.; Shang, X.; Shi, J. Hyperspectral Imaging for Foreign Matter Detection in Foods: Advances, Challenges, and Future Directions. Foods 2024, 14(17), 3026. [Google Scholar] [CrossRef]
- Guo, Z.; Zhang, J.; Wang, H.; Li, S.; Shao, X.; Xia, L.; Darwish, I.A.; Guo, Y.; Sun, X. Advancing detection of fungal and mycotoxins contamination in grains and oilseeds: Hyperspectral imaging for enhanced food safety. Food Chemistry 2025, 470, 142689. [Google Scholar] [CrossRef]
- Ali, H.M.; Xiao, Y.; Kersten-Oertel, M. Surgical hyperspectral imaging: a systematic review. Comput Assist Surg (Abingdon) 2025, 30(1), 2546819. [Google Scholar] [CrossRef]
- Mishra, P.; Roger, J.M.; Jouan-Rimbaud-Bouveresse, D.; Biancolillo, A.; Marini, F.; Nordon, A.; Rutledge, D.N. Recent trends in multi-block data analysis in chemometrics for multi-source data integration. TrAC Trends in Analytical Chemistry 2021, 137, 116206. [Google Scholar] [CrossRef]
- Smilde, A. K.; Mage, I.; Naes, T.; Hankemeier, T.; Lips, M. A.; Kiers, H.A-L-; Acar, E.; Bro, R. Common and distinct components in data fusion. Journal of Chemometrics 2017, 31(7), p. e2900. [Google Scholar] [CrossRef]
- Song, Y.; Westerhuis, J. A.; Smilde, A. K. Separating common (global and local) and distinct variation in multiple mixed types data sets. Journal of Chemometrics 2020, 34(1), e3197. [Google Scholar] [CrossRef]
- Zhang, X.; Yang, J.; Lin, T.; Ying, Y. Food and agro-product quality evaluation based on spectroscopy and deep learning: A review. Trends in Food Science & Technology 2021, 112, 431–441. [Google Scholar] [CrossRef]
- Mishra, P.; Klont, R.; Verkleij, T.; Wisse, S. Translating near-infrared spectroscopy from laboratory to commercial slaughterhouse: Existing challenges and solutions. Infrared Physics & Technology 2021, 119, 103918. [Google Scholar] [CrossRef]
- Yang, C.; Guo, Z.; Barbin, D. F.; Dai, Z.; Watson, N.; Povey, M.; Zou, X. Hyperspectral Imaging and Deep Learning for Quality and Safety Inspection of Fruits and Vegetables: A Review. Journal of Agricultural and Food Chemistry 2025, 73(17), 10019–10035. [Google Scholar] [CrossRef]
- Benelli, A.; Cevoli, C.; Fabbri, A.; Ragni, L. Ripeness evaluation of kiwifruit by hyperspectral imaging. Biosyst. Eng. 2022, 42–52. [Google Scholar] [CrossRef]
- Zhang, Q.; Lu, Z.; Liu, Z.; Li, J.; Chang, M.; Zuo, M. Application of Machine Learning in Food Safety Risk Assessment. Foods 2024, 14(23), 4005. [Google Scholar] [CrossRef]
- Entrenas, J.-A.; Pérez-Marín, D.; Torres, I.; Garrido-Varo, A.; Sánchez, M.-T. Simultaneous detection of quality and safety in spinach plants using a new generation of NIRS sensors. Postharvest Biology and Technology 2020, 160, 111026. [Google Scholar] [CrossRef]
- Parastar, H.; van Kollenburg, G.; Weesepoel, Y.; van den Doel, A.; Buydens, L.; Jansen, J. Integration of handheld NIR and machine learning to “Measure & Monitor” chicken meat authenticity. Food Control 2020, 112, 107149. [Google Scholar] [CrossRef]
- de la Roza-Delgado, B.; Garrido-Varo, A.; Soldado, A.; González Arrojo, A.; Cuevas Valdés, M.; Maroto, F.; Pérez Marín, D. Matching portable NIRS instruments for in situ monitoring indicators of milk composition. Food Control 2017, 76, 74–81. [Google Scholar] [CrossRef]
- Yakes, B. J.; Ellsworth, Z.; Karunathilaka, S. R.; Crump, E. Evaluation of Portable Sensor and Spectroscopic Devices for Seafood Decomposition Determination. Food Anal. Methods 2021, 14(11), 2346–2356. [Google Scholar] [CrossRef]
- Beć, K. B.; Grabska, J.; Huck, C. W. Miniaturized NIR Spectroscopy in Food Analysis and Quality Control: Promises, Challenges, and Perspectives. Foods 2022, 11(10). [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Gao, X.; Qi, W-L.; QWang, Y.; Wang, X.; Zhou, J.; Lu, D.; Chen, B. Advances in differentiation and identification of foodborne bacteria using near infrared spectroscopy. Anal. Methods 2021, 13(23), 2558–2566. [Google Scholar] [CrossRef]
- Soni, A.; Dixit, Y.; Reis, M. M.; Brightwell, G. Hyperspectral imaging and machine learning in food microbiology: Developments and challenges in detection of bacterial, fungal, and viral contaminants. Comprehensive Reviews in Food Science and Food Safety 2022, 21(4), 3717–3745. [Google Scholar] [CrossRef]
- Hardy, M.; Moser, B.; Haughey, S. A.; Elliott, C. T. Does the fish rot from the head? Hyperspectral imaging and machine learning for the evaluation of fish freshness. Chemometrics and Intelligent Laboratory Systems 2024, 245, 105059. [Google Scholar] [CrossRef]
- Aramendia, J.; García-Velasco, N.; Amigo, J.M.; Izagirre, U.; Seifert, A.; Soto, M.; Cstro, K. Evidence of internalized microplastics in mussel tissues detected by volumetric Raman imaging. Science of The Total Environment 2024, 914, 169960. [Google Scholar] [CrossRef]
- Wu, Z.; Pu, H.; Sun, D.-W. Fingerprinting and tagging detection of mycotoxins in agri-food products by surface-enhanced Raman spectroscopy: Principles and recent applications. Trends in Food Science & Technology 2021, 393–404. [Google Scholar] [CrossRef]
- Logan, B. G.; Hopkins, D. L.; Schmidtke, L. M.; Fowler, S. M. Authenticating common Australian beef production systems using Raman spectroscopy. Food Control 2021, 121, 107652. [Google Scholar] [CrossRef]
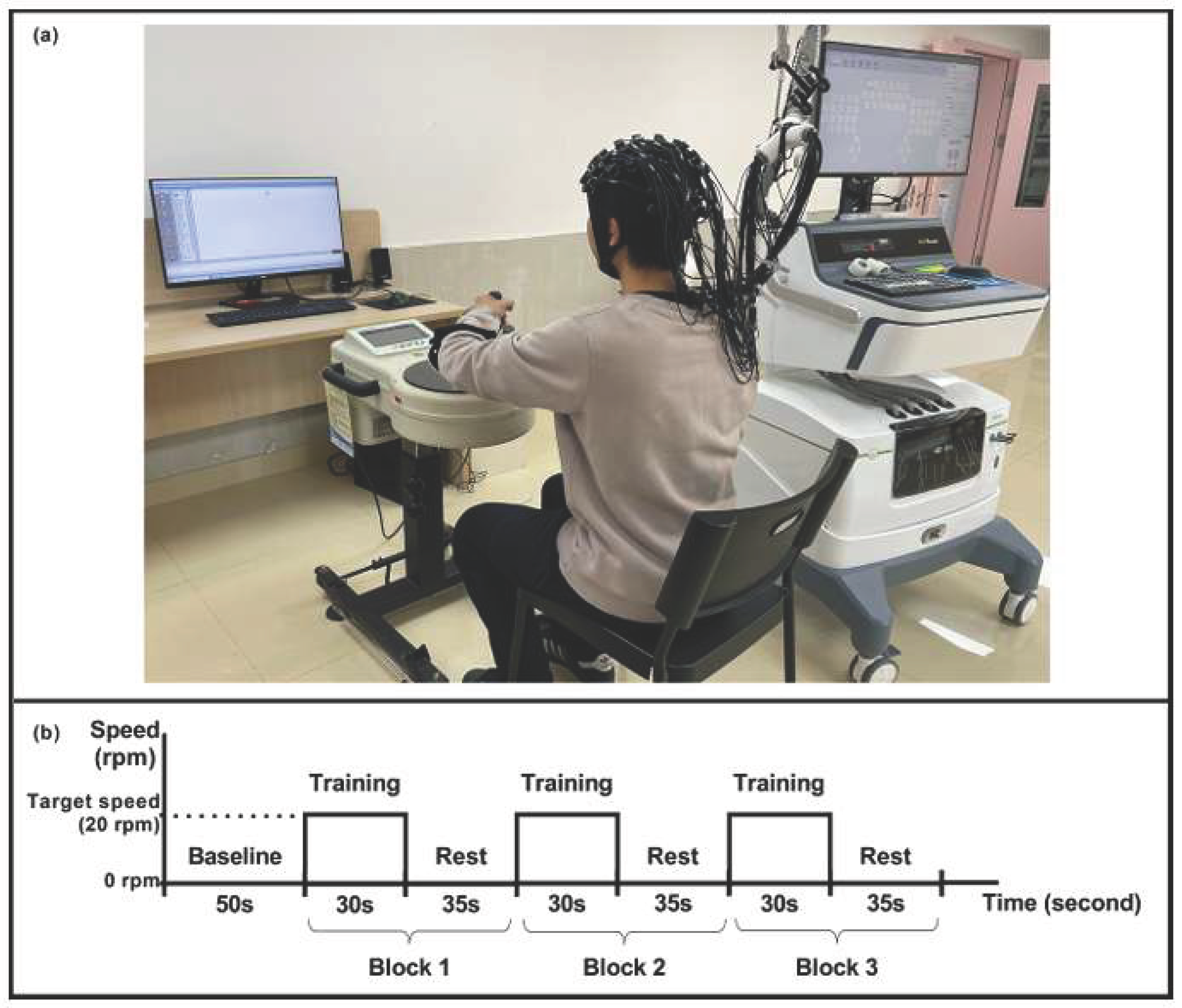
- Xia, W.; Dai, R.; Xu, X.; Huai, B.; Bai, Z.; Zhang, J.; Jin, M.; Niu, W. Cortical mapping of active and passive upper limb training in stroke patients and healthy people: A functional near-infrared spectroscopy study. Brain Research 2022, 1788, 147935. [Google Scholar] [CrossRef]
- Phillips, Z.; Canoy, R. J.; Paik, S.; Lee, S.H.; Kim, B.M. Functional Near-Infrared Spectroscopy as a Personalized Digital Healthcare Tool for Brain Monitoring. J. Clin Neurol. 2023, 19(2), 115–124. [Google Scholar] [CrossRef]
- Yim, A.; Alberto, M.; Sharma, V.; Gree, A.; Mclean, A.; Plessis, J.; Wong, L.M.; Wood, B.; Ischia, J.; Raman, J.; Bolton, D. Near-infrared spectroscopy as a novel method of ex vivo bladder cancer tissue characterisation. BJU International 2024. [Google Scholar] [CrossRef] [PubMed]
- Yu M.; Yi C.; Lin S.; Ye H., Xue J.; Yin J.; Zhong R.; Yao H., Liao H.; Zhang Y.; Yang J.; Guo Y.; Song X-J; Ye T-T. e-Bandage: Exploiting Smartphone as a Therapeutic Device for Cutaneous Wound Treatment. Advanced Intelligent Systems 2024, 6(3), 2300494. [CrossRef]
- Bratchenko, I. A.; Bratchenko, L.A.; Moryatov, A. A.; Khristoforova, Y.A.; Artemyev, D.N.; Myakinin, O.O.; Orlov, A.E.; Kozlov, S.V.; Zakharov, V.P. In vivo diagnosis of skin cancer with a portable Raman spectroscopic device. Experimental Dermatology 2021, 30(5), 652–663. [Google Scholar] [CrossRef]
- Yang, G.; Wang, Z. A Deep Transfer Contrastive Learning Network for Few-Shot Hyperspectral Image Classification. Remote Sensing 2024, 17(16), 2800. [Google Scholar] [CrossRef]


| Model | Laser Wavelength |
Laser Power | Spectral Range (cm−1) |
Spectral resolution (FWHM) |
|---|---|---|---|---|
| Pendar X10 | ~ 830 nm Difference Raman Spectroscopy |
Max. 90 mW | Measures from a distance (“standoff”) from 0.3 to 2 m, No direct contact |
X |
| BRAVO (Bruker) | Laser Duo: 785 nm and 852 nm |
Class 1 Laser | ~ 170 cm−1 – ~ 2.200 cm−1 |
X |
| B&W Tek i-Raman Plus | 532 nm or 785 nm | ~ 340 mW (785 nm) |
~ 65 cm−1 – ~ 4200 cm−1 | < ~ 4.5 cm−1 |
| miniRaman (Lightnovo) | Standard dual: 785 nm and 660/675 nm |
5-50 mW; 5-40/5-75 mW | 2336-460 cm-1 | 2 cm-1 |
| Metrohm NanoRam-1064 | 1064 nm | 420 ± 30 mW | 176 – 2500 cm−1 | ~ 10 – 11 cm−1 |
| Name/Type | Key features (short) | Strengths | Weaknesses | Typical users |
|---|---|---|---|---|
|
PLS_Toolbox/Solo (Eigenvector)/ Commercial (MATLAB add-on) |
PLS, PCR, multiway (PARAFAC/Tucker), preprocessing, validation, many visualization tools. | Very feature-rich for spectroscopy & calibration; tight MATLAB integration; mature. | Requires MATLAB (unless using Solo); commercial cost. | Analytical chemists, spectroscopists, model developers. |
|
The Unscrambler X (CAMO / AspenTech)/ Commercial |
PCA, PLS, calibration, process monitoring, DoE, SPC, strong visualization. | GUI-driven, industry-proven for spectroscopy and PAT; lots of built-in workflows. | Commercial cost; less code/scriptable than Python/R. | Industrial spectroscopy labs, process analytics teams. |
|
SIMCA (Sartorius/Umetrics)/ Commercial |
Multivariate process monitoring, PCA/PLS/SIMCA models, real-time monitoring. | Built for process control/PAT and real-time batch monitoring; strong automation and deployment. | Commercial; enterprise focus can be heavyweight for small projects. | Biopharma/process industries, PAT teams. |
|
Pirouette (Infometrix)/ Commercial |
PCA, PLS, classification, mixture analysis, automation tools. | Fast, focused chemometrics GUI; long history in spectroscopy communities. | Smaller vendor; fewer ecosystem integrations than MATLAB/Python. | QC labs, spectroscopy analysts who want GUI workflows. |
|
Mnova — Advanced Chemometrics (Mestrelab) Commercial (plugin/module) |
PCA, PLS, SIMCA, MCR-ALS, spectral preprocessing integrated with NMR/LC/MS workflows. | Excellent if you already use Mnova for spectra — combines spectral processing + chemometrics in one place. | Requires Mnova license; more specialized toward NMR/LC-MS users. | Spectroscopists, NMR / LC-MS users who want integrated workflow. |
|
OPUS (Bruker)/ Commercial |
Spectrometer control + spectral processing + multivariate analysis extensions. | Industry-leading spectroscopy software with instrument integration and reaction monitoring plugins. | Mainly tied to Bruker instruments; commercial. | FT-IR / Raman / NIR users using Bruker instruments. |
|
TQ Analyst / OMNIC (Thermo Fisher)/ Commercial |
Spectral chemometrics (PCR/PLS), spectral libraries, method dev & deployment. | Designed for spectroscopists and manufacturing deployment; vendor support for regulatory environments. | Commercial; tied to Thermo ecosystem for full integration. | QA/QC labs, spectroscopy method developers. |
| mdatools (R package) | PCA, PLS, preprocessing, validation, visualization, tutorials & docs. | Free, actively maintained, good for reproducible scripting and teaching. | Requires R programming; GUI options limited. | Academics, students, data scientists preferring R. |
|
chemometrics (Python package)/ Open source |
Spectral preprocessing, plotting, PLS, PCA — built on scikit-learn. | Python-friendly, integrates with scikit-learn ecosystem for ML + chemometrics. | Smaller feature set vs mature commercial suites; requires coding. | Data scientists & researchers who prefer Python. |
|
pyChemometrics / chemotools / scikit-spectra (Python eco.)/ Open source |
PCA/PLS wrappers, spectral transformers, scikit-learn integration, spectroscopy datatypes. | Flexible, ideal for building reproducible pipelines; many specialized transformers available. | Fragmented ecosystem (multiple small packages); need to assemble pipeline. | Python users building production or research pipelines. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




